Applications of Biopolymers for Drugs and Probiotics Delivery
Abstract
1. Introduction
2. Biopolymers vs. Conventional Synthetic Materials
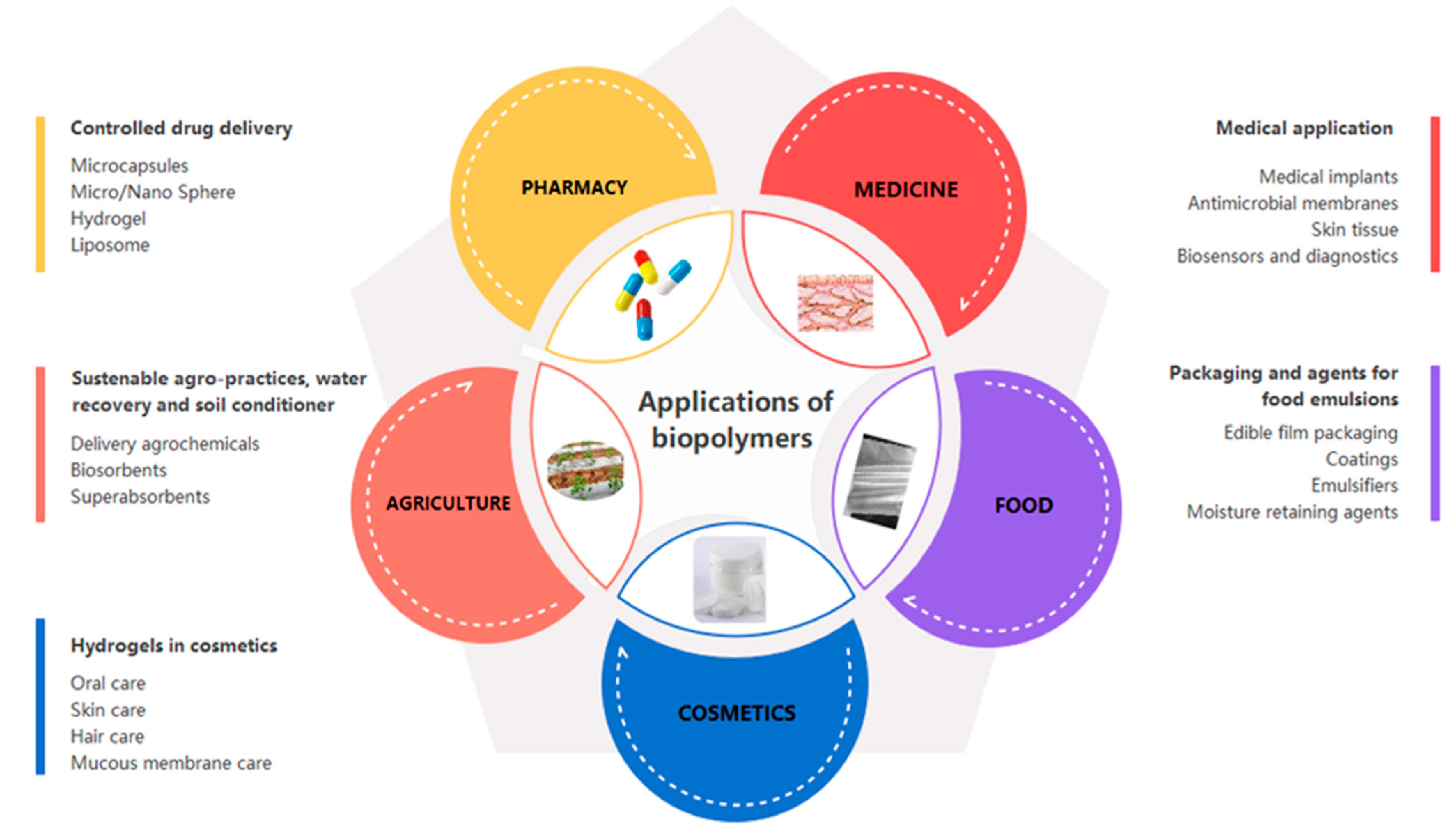
3. Applications of Biopolymers
3.1. Biopolymers in the Pharmaceutical Industry
3.1.1. Biopolymers for Controlled Drug Release
Alginate and Its Use for Drug Delivery
Chitosan and Its Use for Drug Delivery
Agar and Its Use for Drug Delivery
Starch and Its Use for Drug Delivery
Cellulose and Its Use for Drug Delivery
3.2. Biopolymers in Probiotic Encapsulation and Delivery
- (1)
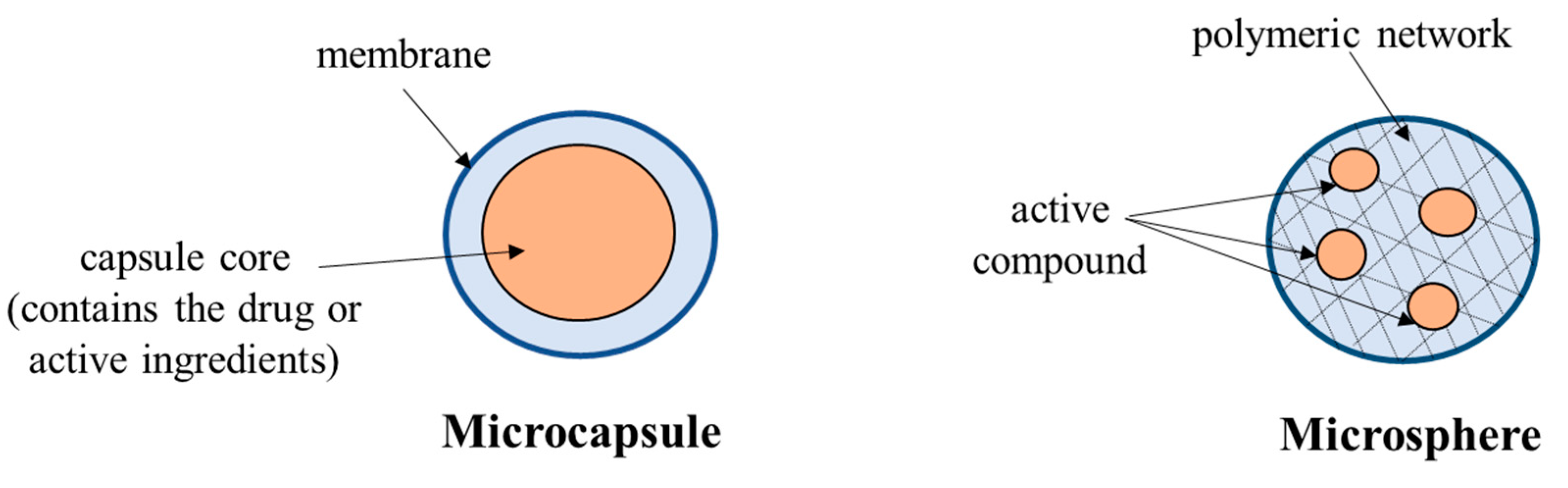
- Microencapsulation represents a physicochemical or mechanical process used to trap a substance (active agent) into a coating material (defined as wall material). In this way, spherical particles have a thin and strong but semipermeable membrane with a diameter from nanometers to a few millimeters [174]. The purpose of the procedure is to protect compounds or viable cells against environmental agents that can destroy the core [181].
- (2)
- Spray-drying technique is suitable for industrial applications on a large scale, involving atomization of a liquid mixture and the solvent is evaporated at contact with hot air or gas.
- (3)
- Lyophilization involves freezing the cells with the material used for encapsulation (usually at freezing temperatures), followed by vacuum elimination of water at a pressure between 0.05 to 0.1 mBar and temperature between −50 °C to −30 °C. To preserve and stabilize the activity of lyophilized probiotics, cryoprotectants are added, such as lactose, trehalose, sorbitol, sucrose, milk protein, or skim milk.
- (4)
- Extrusion is the most common technique to use biopolymers as encapsulation materials. The method involves obtaining a hydrocolloid solution, followed by the addition of microorganisms, formation of droplets using a syringe needle (pilot scale) or an extruder (industrial scale), and their release into a hardening solution (typically calcium chloride) [182].
- (5)
- Emulsion is when a small volume of a hydrocolloid suspension containing microorganisms (discontinuous phase) is added to a larger volume of vegetable oil (continuous phase). Using an emulsifier, the mixture is homogenized. After emulsion formation, it can be insolubilized to manufacture gel capsules. The big disadvantage of this method is that the particles obtained vary greatly in shape and size, although bead sizes can be reduced by mechanical homogenization [183].
- (6)
- Spray–freeze-drying is a combined procedure that involves steps used in lyophilization (freeze-drying) and spray-drying. The advantage is that it provides capsules with a controlled size and higher specific surface area, unlike those obtained by spray-drying. The disadvantages of the method refers to high costs (approximately 50 times higher than the classic spray-drying version), long processing times, and the high-energy requirement.
- (7)
- Layer-by-layer is technology based on alternating coating layers of cationic (e.g., chitosan) with anionic (e.g., alginate) biopolymers on cells via electrostatic interaction [184]. It has the advantage of enhanced bacterial viability throughout the gastrointestinal tract, along with the survival of probiotic cells against acidic and bile salt insults, mucoadhesion and growth on intestinal tissues, and in vivo survival [179].
3.2.1. Alginate’s Use for Probiotic Delivery
3.2.2. Chitosan Use for Probiotic Delivery
3.2.3. Agar Use for Probiotic Delivery
3.2.4. Starch Use for Probiotic Delivery
3.2.5. Cellulose Use for Probiotic Delivery
4. Challenges and Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Rebelo, R.; Fernandes, M.; Fangueiro, R. Biopolymers in medical implants: A brief review. Procedia Eng. 2017, 200, 236–243. [Google Scholar] [CrossRef]
- Bala, I.A.; Abdullahi, M.R.; Bashir, S.S. A review on formulation of enzymatic solution for biopolymer hydrolysis. J. Chem. 2017, 6, 9–13. [Google Scholar]
- Yadav, P.; Yadav, H.; Shah, V.G.; Shah, G.; Dhaka, G. Biomedical biopolymers, their origin and evolution in biomedical sciences: A systematic review. J. Clin. Diagnostic Res. 2015, 9, 21–25. [Google Scholar] [CrossRef]
- Aggarwal, J.; Sharma, S.; Kamyab, H.; Kumar, A. The Realm of Biopolymers and Their Usage: An Overview. Available online: http://www.jett.dormaj.com (accessed on 24 June 2021).
- Udayakumar, G.P.; Muthusamy, S.; Selvaganesh, B.; Sivarajasekar, N.; Rambabu, K.; Banat, F.; Sivamani, S.; Sivakumar, N.; Hosseini-Bandegharaei, A.; Show, P.L. Biopolymers and composites: Properties, characterization and their applications in food, medical and pharmaceutical industries. J. Environ. Chem. Eng. 2021, 9, 105322. [Google Scholar] [CrossRef]
- Pattanashetti, N.A.; Heggannavar, G.B.; Kariduraganavar, M.Y. Smart biopolymers and their biomedical applications. Procedia Manuf. 2017, 12, 263–279. [Google Scholar] [CrossRef]
- Rendón-Villalobos, R.; Ortíz-Sánchez, A.; Tovar-Sánchez, E.; Flores-Huicochea, E. The role of biopolymers in obtaining environmentally friendly materials. In Composites from Renewable and Sustainable Materials; InTech: Houston, TX, USA, 2016. [Google Scholar]
- Nath, K.; Bhattacharyya, S.K.; Das, N.C. Biodegradable polymeric materials for EMI shielding. In Materials for Potential EMI Shielding Applications; Elsevier: Amsterdam, The Netherlands, 2020; pp. 165–178. [Google Scholar]
- Kumar, S.; Thakur, K. Bioplastics—Classification, production and their potential food applications. J. Hill Agric. 2017, 8, 118. [Google Scholar] [CrossRef]
- Prajapati, S.K.; Jain, A.; Jain, A.; Jain, S. Biodegradable polymers and constructs: A novel approach in drug delivery. Eur. Polym. J. 2019, 120, 109191. [Google Scholar] [CrossRef]
- Arora, S. Biopolymers as Packaging Material in Food and Allied Industry Value Addition of Makhana and Its by-Products View Project. 2018. Available online: https://www.researchgate.net/publication/342765641 (accessed on 1 February 2021).
- Díez-Pascual, A.M. Synthesis and applications of biopolymer composites. Int. J. Mol. Sci. 2019, 20, 2321. [Google Scholar] [CrossRef] [PubMed]
- Qasim, U.; Osman, A.I.; Al-Muhtaseb, A.H.; Farrell, C.; Al-Abri, M.; Ali, M.; Vo, D.V.N.; Jamil, F.; Rooney, D.W. Renewable cellulosic nanocomposites for food packaging to avoid fossil fuel plastic pollution: A review. Environ. Chem. Lett. 2021, 19, 613–641. [Google Scholar] [CrossRef]
- Hamouda, T. Sustainable packaging from coir fibers. In Biopolymers and Biocomposites from Agro-Waste for Packaging Applications; Elsevier: Amsterdam, The Netherlands, 2021; pp. 113–126. [Google Scholar]
- Shivam, P. Recent developments on biodegradable polymers and their future trends. Int. Res. J. Sci. Eng. 2016, 4, 17–26. [Google Scholar]
- Chen, H.; Wang, J.; Cheng, Y.; Wang, C.; Liu, H.; Bian, H.; Pan, Y.; Sun, J.; Han, W. Application of protein-based films and coatings for food packaging: A review. Polymers 2019, 11, 2039. [Google Scholar] [CrossRef]
- Swain, S.K.; Pattanayak, A.J.; Sahoo, A.P. Functional Biopolymer Composites; Springer: Berlin, Germany, 2018; pp. 159–182. [Google Scholar]
- Varma, K.; Gopi, S. Biopolymers and their role in medicinal and pharmaceutical applications. In Biopolymers and Their Industrial Applications; Elsevier: Amsterdam, The Netherlands, 2021; pp. 175–191. [Google Scholar]
- Carvalho, A.J.F. Starch: Major sources, properties and applications as thermoplastic materials. In Monomers, Polymers and Composites from Renewable Resources; Elsevier: Amsterdam, The Netherlands, 2008; pp. 321–342. ISBN 9780080453163. [Google Scholar]
- Temesgen, S.; Rennert, M.; Tesfaye, T.; Nase, M. Review on spinning of biopolymer fibers from starch. Polymers 2021, 13, 1121. [Google Scholar] [CrossRef] [PubMed]
- Gustafsson, J.; Landberg, M.; Bátori, V.; Åkesson, D.; Taherzadeh, M.J.; Zamani, A. Development of bio-based films and 3D objects from apple pomace. Polymers 2019, 11, 289. [Google Scholar] [CrossRef]
- Liu, W.; Misra, M.; Askeland, P.; Drzal, L.T.; Mohanty, A.K. “Green” composites from soy based plastic and pineapple leaf fiber: Fabrication and properties evaluation. Polymer 2005, 46, 2710–2721. [Google Scholar] [CrossRef]
- Maraveas, C. Production of sustainable and biodegradable polymers from agricultural waste. Polymers 2020, 12, 1127. [Google Scholar] [CrossRef] [PubMed]
- Heredia-Guerrero, J.A.; Heredia, A.; Domínguez, E.; Cingolani, R.; Bayer, I.S.; Athanassiou, A.; Benítez, J.J. Cutin from agro-waste as a raw material for the production of bioplastics. J. Exp. Bot. 2017, 68, 5401–5410. [Google Scholar] [CrossRef] [PubMed]
- Khrunyk, Y.; Lach, S.; Petrenko, I.; Ehrlich, H. Progress in modern marine biomaterials research. Mar. Drugs 2020, 18, 589. [Google Scholar] [CrossRef]
- Kaur, S.; Dhillon, G.S. The versatile biopolymer chitosan: Potential sources, evaluation of extraction methods and applications. Crit. Rev. Microbiol. 2014, 40, 155–175. [Google Scholar] [CrossRef]
- Torres, F.G.; Troncoso, O.P.; Pisani, A.; Gatto, F.; Bardi, G. Natural polysaccharide nanomaterials: An overview of their immunological properties. Int. J. Mol. Sci. 2019, 20, 5092. [Google Scholar] [CrossRef]
- Velu, R.; Calais, T.; Jayakumar, A.; Raspall, F. A comprehensive review on bio-nanomaterials for medical implants and feasibility studies on fabrication of such implants by additive manufacturing technique. Materials 2020, 13, 92. [Google Scholar] [CrossRef]
- Chaitanya, S.; Singh, I. Processing of PLA/sisal fiber biocomposites using direct- and extrusion-injection molding. Mater. Manuf. Processes 2017, 32, 468–474. [Google Scholar] [CrossRef]
- Khanna, S.; Srivastava, A.K. A simple structured mathematical model for biopolymer (PHB) production. Biotechnol. Prog. 2005, 21, 830–838. [Google Scholar] [CrossRef] [PubMed]
- Ncube, L.K.; Ude, A.U.; Ogunmuyiwa, E.N.; Zulkifli, R.; Beas, I.N. Environmental impact of food packaging materials: A review of contemporary development from conventional plastics to polylactic acid based materials. Materials 2020, 13, 4994. [Google Scholar] [CrossRef]
- Wankhade, V. Animal-derived biopolymers in food and biomedical technology. In Biopolymer-Based Formulations: Biomedical and Food Applications; Elsevier: Amsterdam, The Netherlands, 2020; pp. 139–152. ISBN 9780128168981. [Google Scholar]
- Parker, G. Measuring the environmental performance of food packaging: Life cycle assessment. In Environmentally Compatible Food Packaging; Elsevier: Amsterdam, The Netherlands, 2008; pp. 211–237. ISBN 9781845691943. [Google Scholar]
- Shankar, S.; Rhim, J.-W. Bionanocomposite films for food packaging applications. In Reference Module in Food Science; Elsevier: Amsterdam, The Netherlands, 2018. [Google Scholar]
- Hassan, M.E.; Bai, J.; Dou, D.Q. Biopolymers; Definition, classification and applications. Egypt. J. Chem. 2019, 62, 1725–1737. [Google Scholar] [CrossRef]
- Soldo, A.; Miletić, M.; Auad, M.L. Biopolymers as a sustainable solution for the enhancement of soil mechanical properties. Sci. Rep. 2020, 10, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Song, J.; Winkeljann, B.; Lieleg, O. Biopolymer-based coatings: Promising strategies to improve the biocompatibility and functionality of materials used in biomedical engineering. Adv. Mater. Interfaces 2020, 7, 2000850. [Google Scholar] [CrossRef]
- Gabor, D.; Tita, O. Biopolymers used in food packaging: A review. Acta Univ. Cibiniensis Ser. E Food Technol. 2012, 16, 3–19. [Google Scholar]
- Sadasivuni, K.K.; Saha, P.; Adhikari, J.; Deshmukh, K.; Ahamed, M.B.; Cabibihan, J.J. Recent advances in mechanical properties of biopolymer composites: A review. Polym. Compos. 2020, 41, 32–59. [Google Scholar] [CrossRef]
- Jummaat, F.; Bashir Yahya, E.; Khalil HPS, A.; Adnan, A.S.; Mohammed Alqadhi, A.; Abdullah, C.K.; Sofea, A.A.; Olaiya, N.G.; Abdat, M.; Hps, K. The role of biopolymer-based materials in obstetrics and gynecology applications: A review. Polymers 2021, 13, 633. [Google Scholar] [CrossRef]
- Reddy, M.S.B.; Ponnamma, D.; Choudhary, R.; Sadasivuni, K.K. A comparative review of natural and synthetic biopolymer composite scaffolds. Polymers 2021, 13, 1105. [Google Scholar] [CrossRef]
- Olivia, M.; Jingga, H.; Toni, N.; Wibisono, G. Biopolymers to improve physical properties and leaching characteristics of mortar and concrete: A review. In IOP Conference Series: Materials Science and Engineering; Institute of Physics Publishing: Bristol, UK, 2018; Volume 345, p. 012028. [Google Scholar]
- Gurgel Adeodato Vieira, M.; Altenhofen da Silva, M.; Oliveira dos Santos, L.; Masumi Beppu, M. Natural-based plasticizers and biopolymer films: A review. Eur. Polym. J. 2011, 47, 254–263. [Google Scholar] [CrossRef]
- Polman, E.M.N.; Gruter, G.J.M.; Parsons, J.R.; Tietema, A. Comparison of the aerobic biodegradation of biopolymers and the corresponding bioplastics: A review. Sci. Total Environ. 2021, 753, 131953. [Google Scholar] [CrossRef]
- Mohan, S.; Oluwafemi, O.S.; Kalarikkal, N.; Thomas, S.; Songca, S.P. Biopolymers—Application in nanoscience and nanotechnology. In Recent Advances in Biopolymers; InTech: Houston, TX, USA, 2016. [Google Scholar]
- Imre, B.; Pukánszky, B. Compatibilization in bio-based and biodegradable polymer blends. Eur. Polym. J. 2013, 49, 1215–1233. [Google Scholar] [CrossRef]
- Jayanth, D.; Kumar, P.S.; Nayak, G.C.; Kumar, J.S.; Pal, S.K.; Rajasekar, R. A review on biodegradable polymeric materials striving towards the attainment of green environment. J. Polym. Environ. 2018, 26, 838–865. [Google Scholar] [CrossRef]
- Cziple, F.A.; Velez Marques, A.J. Biopolymers Versus Synthetic Polymers. Available online: https://creativepegworks.wordpress.com/2015/12/01/biopolymers-vs-synthetic-polymers/ (accessed on 26 June 2021).
- Simionescu, B.C.; Ivanov, D. Natural and synthetic polymers for designing composite materials. In Handbook of Bioceramics and Biocomposites; Springer: Berlin, Germany, 2016; pp. 233–286. ISBN 9783319124605. [Google Scholar]
- Gowthaman, N.S.K.; Lim, H.N.; Sreeraj, T.R.; Amalraj, A.; Gopi, S. Advantages of biopolymers over synthetic polymers. In Biopolymers and Their Industrial Applications; Elsevier: Amsterdam, The Netherlands, 2021; pp. 351–372. [Google Scholar]
- Luh, T.-Y.; Yang, H.-C.; Lin, N.-T.; Lin, S.-Y.; Lee, S.-L.; Chen, C.-H. OMCOS for functional polymers-double-stranded DNA-like polymers. Pure Appl. Chem. 2008, 80, 819–829. [Google Scholar] [CrossRef]
- Colmenares, J.C.; Kuna, E. Photoactive hybrid catalysts based on natural and synthetic polymers: A comparative overview. Molecules 2017, 22, 790. [Google Scholar] [CrossRef] [PubMed]
- Ryder, K.; Ali, M.A.; Carne, A.; Billakanti, J. The potential use of dairy by-products for the production of nonfood biomaterials. Crit. Rev. Environ. Sci. Technol. 2017, 47, 621–642. [Google Scholar] [CrossRef]
- Maraveas, C. Environmental sustainability of greenhouse covering materials. Sustainability 2019, 11, 6129. [Google Scholar] [CrossRef]
- Mtibe, A.; Motloung, M.P.; Bandyopadhyay, J.; Ray, S.S. Synthetic biopolymers and their composites: Advantages and limitations—An overview. Macromol. Rapid Commun. 2021, 42, 2100130. [Google Scholar] [CrossRef] [PubMed]
- Ortelli, S.; Costa, A.L.; Torri, C.; Samorì, C.; Galletti, P.; Vineis, C.; Varesano, A.; Bonura, L.; Bianchi, G. Innovative and sustainable production of biopolymers. In Factories of the Future: The Italian Flagship Initiative; Springer: Berlin, Germany, 2019; pp. 131–148. [Google Scholar] [CrossRef]
- Adeyeye, O.A.; Sadiku, E.R.; Reddy, A.B.; Ndamase, A.S.; Makgatho, G.; Sellamuthu, P.S.; Perumal, A.B.; Nambiar, R.B.; Fasiku, V.O.; Inrahim, I.D.; et al. The use of biopolymers in food packaging. In Green Polymers and Their Nanocomposites; Springer: Singapore, 2019; pp. 137–158. [Google Scholar] [CrossRef]
- Wróblewska-Krepsztul, J.; Rydzkowski, T.; Michalska-Pożoga, I.; Thakur, V.K. Biopolymers for biomedical and pharmaceutical applications: Recent advances and overview of alginate electrospinning. Nanomaterials 2019, 9, 404. [Google Scholar] [CrossRef]
- Ferraris, S.; Spriano, S.; Scalia, A.C.; Cochis, A.; Rimondini, L.; Cruz-Maya, I.; Guarino, V.; Varesano, A.; Vineis, C. Topographical and biomechanical guidance of electrospun fibers for biomedical applications. Polymers 2020, 12, 2896. [Google Scholar] [CrossRef]
- Dhandayuthapani, B.; Yoshida, Y.; Maekawa, T.; Kumar, D.S. Polymeric scaffolds in tissue engineering application: A review. Int. J. Polym. Sci. 2011, 2011, 290602. [Google Scholar] [CrossRef]
- Ogueri, K.S.; Jafari, T.; Escobar, I.J.L.; Laurencin, C.T. Polymeric biomaterials for scaffold-based bone regenerative engineering. Regen. Eng. Transl. Med. 2019, 5, 128–154. [Google Scholar] [CrossRef]
- Bhatia, S. Natural polymers vs. synthetic polymer. In Natural Polymer Drug Delivery Systems; Springer: Berlin, Germany, 2016; pp. 95–118. [Google Scholar]
- Innovative Packaging of Medicines-IJAHR. Available online: https://www.ipinnovative.com/journals/IJAHR/article-full-text/10648 (accessed on 27 June 2021).
- Zadbuke, N.; Shahi, S.; Gulecha, B.; Padalkar, A.; Thube, M. Recent trends and future of pharmaceutical packaging technology. J. Pharm. Bioallied Sci. 2013, 5, 98–110. [Google Scholar] [CrossRef]
- Gheorghita Puscaselu, R.; Amariei, S.; Norocel, L.; Gutt, G. New edible packaging material with function in shelf life extension: Applications for the meat and cheese industries. Foods 2020, 9, 562. [Google Scholar] [CrossRef]
- Janes, M.E.; Dai, Y. Edible films for meat, poultry and seafood. In Advances in Meat, Poultry and Seafood Packaging; Elsevier: Amsterdam, The Netherlands, 2012; pp. 504–521. ISBN 9781845697518. [Google Scholar]
- Avila-Sosa, R.; Palou, E.; López-Malo, A. Essential oils added to edible films. In Essential Oils in Food Preservation, Flavor and Safety; Elsevier: Amsterdam, The Netherlands, 2016; pp. 149–154. ISBN 9780124166448. [Google Scholar]
- Zhao, Y.; Mcdaniel, M. Sensory quality of foods associated with edible film and coating systems and shelf-life extension. In Innovations in Food Packaging; Elsevier: Amsterdam, The Netherlands, 2005; pp. 434–453. ISBN 9780123116321. [Google Scholar]
- Min, S.; Krochta, J.M. Antimicrobial films and coatings for fresh fruit and Vegetables. In Improving the Safety of Fresh Fruit and Vegetables; Elsevier: Amsterdam, The Netherlands, 2005; pp. 454–492. ISBN 9781855739567. [Google Scholar]
- Mcclements, D.J. Biopolymers in food emulsions. In Modern Biopolymer Science; Elsevier: Amsterdam, The Netherlands, 2009; pp. 129–166. ISBN 9780123741950. [Google Scholar]
- Ahmed, Z.; Ahmad, A. Biopolymer produced by the lactic acid bacteria: Production and practical application. In Microbial Production of Food Ingredients and Additives; Elsevier: Amsterdam, The Netherlands, 2017; pp. 217–257. [Google Scholar]
- Martau, G.A.; Mihai, M.; Vodnar, D.C. The use of chitosan, alginate, and pectin in the biomedical and food sector-biocompatibility, bioadhesiveness, and biodegradability. Polymers 2019, 11, 1837. [Google Scholar] [CrossRef]
- De Vos, P.; Lazarjani, H.A.; Poncelet, D.; Faas, M.M. Polymers in cell encapsulation from an enveloped cell perspective. Adv. Drug Deliv. Rev. 2014, 67–68, 15–34. [Google Scholar] [CrossRef]
- Madene, A.; Jacquot, M.; Scher, J.; Desobry, S. Flavour encapsulation and controlled release—A review. Int. J. Food Sci. Technol. 2006, 41, 1–21. [Google Scholar] [CrossRef]
- Therapeutic Applications of Cell Microencapsulation—Google Cărți. Available online: https://books.google.ro/books?id=7u9d2fkYHvMC&pg=PA1&hl=ro&source=gbs_toc_r&cad=3#v=onepage&q&f=false (accessed on 9 August 2021).
- Biopolymer Nanoparticles: A Review of Prospects for Application as Carrier for Therapeutics and Diagnostics—Pharmaceutical Research and Allied Sciences. Available online: https://ijpras.com/article/biopolymer-nanoparticles-a-review-of-prospects-for-application-as-carrier-for-therapeutics-and-diagnostics (accessed on 9 August 2021).
- Lee, M.K.; Bae, Y.H. Cell transplantation for endocrine disorders. Adv. Drug Deliv. Rev. 2000, 42, 103–120. [Google Scholar] [CrossRef]
- Sharma, K.; Singh, V.; Arora, A. Natural biodegradable polymers as matrices intransdermal drug delivery. Int. J. Drug Dev. Res. 2011, 3, 85–103. [Google Scholar]
- DJ, M. Designing biopolymer microgels to encapsulate, protect and deliver bioactive components: Physicochemical aspects. Adv. Colloid Interface Sci. 2017, 240, 31–59. [Google Scholar] [CrossRef]
- Altomare, L.; Bonetti, L.; Campiglio, C.E.; de Nardo, L.; Draghi, L.; Tana, F.; Farè, S. Biopolymer-based strategies in the design of smart medical devices and artificial organs. Int. J. Artif. Organ 2018, 41, 337–359. [Google Scholar] [CrossRef]
- Schmaljohann, D. Thermo- and pH-responsive polymers in drug delivery. Adv. Drug Deliv. Rev. 2006, 58, 1655–1670. [Google Scholar] [CrossRef]
- George, A.; Shah, P.A.; Shrivastav, P.S. Natural biodegradable polymers based nano-formulations for drug delivery: A review. Int. J. Pharm. 2019, 561, 244–264. [Google Scholar] [CrossRef] [PubMed]
- Polymers in Pharmaceutical Drug Delivery System: A Review. Available online: https://www.researchgate.net/publication/285986312_Polymers_in_pharmaceutical_drug_delivery_system_A_review (accessed on 26 July 2021).
- Paques, J.P.; Van Der Linden, E.; Van Rijn, C.J.M.; Sagis, L.M.C. Preparation methods of alginate nanoparticles. Adv. Colloid Interface Sci. 2014, 209, 163–171. [Google Scholar] [CrossRef]
- Severino, P.; da Silva, C.F.; Andrade, L.N.; de Lima Oliveira, D.; Campos, J.; Souto, E.B. Alginate nanoparticles for drug delivery and targeting. Curr. Pharm. Des. 2019, 25, 1312–1334. [Google Scholar] [CrossRef]
- De, S.; Robinson, D. Polymer relationships during preparation of chitosan-alginate and poly-l-lysine-alginate nanospheres. J. Control. Release 2003, 89, 101–112. [Google Scholar] [CrossRef]
- Smith, A.M.; Miri, T. Alginates in Foods. In Practical Food Rheology: An Interpretive Approach; John Wiley&Sons: Hoboken, NJ, USA, 2010; pp. 113–132. ISBN 9781405199780. [Google Scholar]
- Espevik, T.; Otterlei, M.; Skjåk-Braek, G.; Ryan, L.; Wright, S.D.; Sundan, A. The involvement of CD14 in stimulation of cytokine production by uronic acid polymers. Eur. J. Immunol. 1993, 23, 255–261. [Google Scholar] [CrossRef]
- De Vos, P.; De Haan, B.; Pater, J.; Van Schilfgaarde, R. Association between capsule diameter, adequacy of encapsulation, and survival of microencapsulated rat islet allografts. Transplantation 1996, 62, 893–899. [Google Scholar] [CrossRef] [PubMed]
- Lim, F.; Sun, A.M. Microencapsulated islets as bioartificial endocrine pancreas. Science 1980, 210, 908–910. [Google Scholar] [CrossRef]
- Gattás-Asfura, K.M.; Stabler, C.L. Chemoselective cross-linking and functionalization of alginate via Staudinger ligation. Biomacromolecules 2009, 10, 3122–3129. [Google Scholar] [CrossRef] [PubMed]
- Thomas, D.; KurienThomas, K.; Latha, M.S. Preparation and evaluation of alginate nanoparticles prepared by green method for drug delivery applications. Int. J. Biol. Macromol. 2020, 154, 888–895. [Google Scholar] [CrossRef] [PubMed]
- Design and Characterization of Ibuprofen Loaded Alginate Microspheres Prepared by Ionic Gelation Method. Available online: https://www.researchgate.net/publication/327743699_Design_and_Characterization_of_Ibuprofen_Loaded_Alginate_Microspheres_Prepared_by_Ionic_Gelation_Method (accessed on 24 July 2021).
- Bose, P.S.; Reddy, P.S.; Nagaraju, R. Preparation and evaluation of indomethacin loaded alginate microspheres. Ceska Slov. Farm. 2016, 65, 104–110. [Google Scholar]
- Hariyadi, D.M.; Hendradi, E.; Erawati, T.; Jannah, E.N.; Febrina, W. Influence of drug-polymer ratio on physical characteristics and release of metformin hydrochloride from metformin alginate microspheres. Trop. J. Pharm. Res. 2018, 17, 1229–1233. [Google Scholar] [CrossRef]
- Ahmed, M.M.; El-Rasoul, S.A.; Auda, S.H.; Ibrahim, M.A. Emulsification/internal gelation as a method for preparation of diclofenac sodium–sodium alginate microparticles. Saudi Pharm. J. 2013, 21, 61–69. [Google Scholar] [CrossRef] [PubMed]
- Patel, N.; Lalwani, D.; Gollmer, S.; Injeti, E.; Sari, Y.; Nesamony, J. Development and evaluation of a calcium alginate based oral ceftriaxone sodium formulation. Prog. Biomater. 2016, 5, 117–133. [Google Scholar] [CrossRef]
- Setty, C.M.; Sahoo, S.S.; Sa, D.B. Alginate-coated alginate-polyethyleneimine beads for prolonged release of furosemide in simulated intestinal fluid. Drug Dev. Ind. Pharm. 2008, 31, 435–446. [Google Scholar] [CrossRef] [PubMed]
- Rastogi, R.; Sultana, Y.; Aqil, M.; Ali, A.; Kumar, S. Alginate microspheres of isoniazid for oral sustained drug delivery. Int. J. Pharm. 2007, 334, 71–77. [Google Scholar] [CrossRef]
- Veronovski, A.; Knez, Ž.; Novak, Z. Preparation of multi-membrane alginate aerogels used for drug delivery. J. Supercrit. Fluids 2013, 79, 209–215. [Google Scholar] [CrossRef]
- Lee, S.; Kim, Y.C.; Park, J.H. Zein-alginate based oral drug delivery systems: Protection and release of therapeutic proteins. Int. J. Pharm. 2016, 515, 300–306. [Google Scholar] [CrossRef] [PubMed]
- Varshosaz, J. The promise of chitosan microspheres in drug delivery systems. Expert Opin. Drug Deliv. 2007, 4, 263–273. [Google Scholar] [CrossRef]
- Bakshi, P.S.; Selvakumar, D.; Kadirvelu, K.; Kumar, N.S. Chitosan as an environment friendly biomaterial—A review on recent modifications and applications. Int. J. Biol. Macromol. 2020, 150, 1072–1083. [Google Scholar] [CrossRef]
- Negm, N.A.; Hefni, H.H.H.; Abd-Elaal, A.A.A.; Badr, E.A.; Abou Kana, M.T.H. Advancement on modification of chitosan biopolymer and its potential applications. Int. J. Biol. Macromol. 2020, 152, 681–702. [Google Scholar] [CrossRef]
- Jayakumar, R.; Reis, R.L.; Mano, J.F. Chemistry and applications of phosphorylated chitin and chitosan. E Polym. 2006, 6, 1–16. [Google Scholar] [CrossRef]
- Raza, Z.A.; Khalil, S.; Ayub, A.; Banat, I.M. Recent developments in chitosan encapsulation of various active ingredients for multifunctional applications. Carbohydr. Res. 2020, 492, 108004. [Google Scholar] [CrossRef] [PubMed]
- Hussain, S.A.; Abdelkader, H.; Abdullah, N.; Kmaruddin, S. Review on micro-encapsulation with Chitosan for pharmaceuticals applications. MOJ Curr. Res. Rev. 2018, 1, 77–84. [Google Scholar] [CrossRef]
- Koppolu, B.P.; Smith, S.G.; Ravindranathan, S.; Jayanthi, S.; Suresh Kumar, T.K.; Zaharoff, D.A. Controlling chitosan-based encapsulation for protein and vaccine delivery. Biomaterials 2014, 35, 4382–4389. [Google Scholar] [CrossRef]
- Prabaharan, M. Review paper: Chitosan derivatives as promising materials for controlled drug delivery. J. Biomater. Appl. 2008, 23, 5–36. [Google Scholar] [CrossRef] [PubMed]
- Yu, S.; Xu, X.; Feng, J.; Liu, M.; Hu, K. Chitosan and chitosan coating nanoparticles for the treatment of brain disease. Int. J. Pharm. 2019, 560, 282–293. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Chi, N.; Tang, X. Preparation of estradiol chitosan nanoparticles for improving nasal absorption and brain targeting. Eur. J. Pharm. Biopharm. 2008, 70, 735–740. [Google Scholar] [CrossRef]
- Panos, I.; Acosta, N.; Heras, A. New drug delivery systems based on chitosan. Curr. Drug Discov. Technol. 2008, 5, 333–341. [Google Scholar] [CrossRef]
- Rokhade, A.P.; Patil, S.A.; Aminabhavi, T.M. Synthesis and characterization of semi-interpenetrating polymer network microspheres of acrylamide grafted dextran and chitosan for controlled release of acyclovir. Carbohydr. Polym. 2007, 67, 605–613. [Google Scholar] [CrossRef]
- Giunchedi, P.; Juliano, C.; Gavini, E.; Cossu, M.; Sorrenti, M. Formulation and in vivo evaluation of chlorhexidine buccal tablets prepared using drug-loaded chitosan microspheres. Eur. J. Pharm. Biopharm. 2002, 53, 233–239. [Google Scholar] [CrossRef]
- Cerchiara, T.; Luppi, B.; Bigucci, F.; Petrachi, M.; Orienti, I.; Zecchi, V. Controlled release of vancomycin from freeze-dried chitosan salts coated with different fatty acids by spray-drying. J. Microencapsul. 2003, 20, 473–478. [Google Scholar] [CrossRef]
- Govender, S.; Pillay, V.; Chetty, D.J.; Essack, S.Y.; Dangor, C.M.; Govender, T. Optimisation and characterisation of bioadhesive controlled release tetracycline microspheres. Int. J. Pharm. 2005, 306, 24–40. [Google Scholar] [CrossRef]
- Almeida, A.; Araújo, M.; Novoa-Carballal, R.; Andrade, F.; Gonçalves, H.; Reis, R.L.; Lúcio, M.; Schwartz, S.; Sarmento, B. Novel amphiphilic chitosan micelles as carriers for hydrophobic anticancer drugs. Mater. Sci. Eng. C 2020, 112, 110920. [Google Scholar] [CrossRef]
- Nair, S.; Anoop, K. Design and in vitro evaluation of controlled release Satranidazole subgingival films for periodontitis therapy. Recent Res. Sci. Technol. 2014, 2, 6–11. [Google Scholar]
- Sohail, R.; Abbas, S.R. Evaluation of amygdalin-loaded alginate-chitosan nanoparticles as biocompatible drug delivery carriers for anticancerous efficacy. Int. J. Biol. Macromol. 2020, 153, 36–45. [Google Scholar] [CrossRef] [PubMed]
- Shariatinia, Z.; Mazloom-Jalali, A. Chitosan nanocomposite drug delivery systems designed for the ifosfamide anticancer drug using molecular dynamics simulations. J. Mol. Liq. 2019, 273, 346–367. [Google Scholar] [CrossRef]
- Hanna, D.H.; Saad, G.R. Encapsulation of ciprofloxacin within modified xanthan gum-chitosan based hydrogel for drug delivery. Bioorg. Chem. 2019, 84, 115–124. [Google Scholar] [CrossRef] [PubMed]
- Cánepa, C.; Imperiale, J.C.; Berini, C.A.; Lewicki, M.; Sosnik, A.; Biglione, M.M. Development of a drug delivery system based on chitosan nanoparticles for oral administration of interferon-α. Biomacromolecules 2017, 18, 3302–3309. [Google Scholar] [CrossRef]
- Sathiyaseelan, A.; Saravanakumar, K.; Anand Mariadoss, A.V.; Wang, M.H. Biocompatible fungal chitosan encapsulated phytogenic silver nanoparticles enhanced antidiabetic, antioxidant and antibacterial activity. Int. J. Biol. Macromol. 2020, 153, 63–71. [Google Scholar] [CrossRef] [PubMed]
- Ali, A.; Ahmed, S. A review on chitosan and its nanocomposites in drug delivery. Int. J. Biol. Macromol. 2018, 109, 273–286. [Google Scholar] [CrossRef]
- Mostafavi, F.S.; Zaeim, D. Agar-based edible films for food packaging applications—A review. Int. J. Biol. Macromol. 2020, 159, 1165–1176. [Google Scholar] [CrossRef]
- Bertasa, M.; Dodero, A.; Alloisio, M.; Vicini, S.; Riedo, C.; Sansonetti, A.; Scalarone, D.; Castellano, M. Agar gel strength: A correlation study between chemical composition and rheological properties. Eur. Polym. J. 2020, 123, 109442. [Google Scholar] [CrossRef]
- Zhang, C.; An, D.; Xiao, Q.; Weng, H.; Zhang, Y.; Yang, Q.; Xiao, A. Preparation, characterization, and modification mechanism of agar treated with hydrogen peroxide at different temperatures. Food Hydrocoll. 2020, 101, 105527. [Google Scholar] [CrossRef]
- Xiao, Q.; An, D.; Zhang, C.; Weng, H.; Zhang, Y.; Chen, F.; Xiao, A. Agar quality promotion prepared by desulfation with hydrogen peroxide. Int. J. Biol. Macromol. 2020, 145, 492–499. [Google Scholar] [CrossRef] [PubMed]
- El-Helw, A.E.R.; El-Said, Y. Preparation and characterization of agar beads containing phenobarbitone sodium. J. Microencapsul. 1988, 5, 159–163. [Google Scholar] [CrossRef]
- Evandro, M.; Denis, R.; Zenia, A.; Emre, K.; Denis, P. Oil encapsulation in core-shell alginate capsules by inverse gelation. I: Dripping methodology. J. Microencapsul. 2017, 34, 82–90. [Google Scholar] [CrossRef]
- Rivadeneira, J.; Audisio, M.; Gorustovich, A. Films based on soy protein-agar blends for wound dressing: Effect of different biopolymer proportions on the drug release rate and the physical and antibacterial properties of the films. J. Biomater. Appl. 2018, 32, 1231–1238. [Google Scholar] [CrossRef]
- Zhu, F. Encapsulation and delivery of food ingredients using starch based systems. Food Chem. 2017, 229, 542–552. [Google Scholar] [CrossRef] [PubMed]
- Queiroz, V.M.; Kling, I.C.S.; Eltom, A.E.; Archanjo, B.S.; Prado, M.; Simão, R.A. Corn starch films as a long-term drug delivery system for chlorhexidine gluconate. Mater. Sci. Eng. C 2020, 112, 110852. [Google Scholar] [CrossRef]
- Qi, X.; Tester, R.F. Starch granules as active guest molecules or microorganism delivery systems. Food Chem. 2019, 271, 182–186. [Google Scholar] [CrossRef] [PubMed]
- Hoyos-Leyva, J.D.; Bello-Pérez, L.A.; Alvarez-Ramirez, J.; Garcia, H.S. Microencapsulation using starch as wall material: A review. Food Rev. Int. 2018, 34, 148–161. [Google Scholar] [CrossRef]
- Santana, Á.L.; Angela, M.; Meireles, A. New starches are the trend for industry applications: A review. Food Public Health 2014, 2014, 229–241. [Google Scholar] [CrossRef]
- Chen, J.; Wang, Y.; Liu, J.; Xu, X. Preparation, characterization, physicochemical property and potential application of porous starch: A review. Int. J. Biol. Macromol. 2020, 148, 1169–1181. [Google Scholar] [CrossRef]
- Šárka, E.; Dvořáček, V. New processing and applications of waxy starch (a review). J. Food Eng. 2017, 206, 77–87. [Google Scholar] [CrossRef]
- Hemamalini, T.; Giri Dev, V.R. Comprehensive review on electrospinning of starch polymer for biomedical applications. Int. J. Biol. Macromol. 2018, 106, 712–718. [Google Scholar] [CrossRef]
- Wang, B.; Sui, J.; Yu, B.; Yuan, C.; Guo, L.; Abd El-Aty, A.M.; Cui, B. Physicochemical properties and antibacterial activity of corn starch-based films incorporated with Zanthoxylum bungeanum essential oil. Carbohydr. Polym. 2021, 254, 117314. [Google Scholar] [CrossRef]
- Singh, A.; Nath, L.; Singh, A. Pharmaceutical, food and non-food applications of modified starches: A critical review. Electron. J. Environ. Agric. Food Chem. 2010, 9, 1214–1221. [Google Scholar]
- Rodrigues, A.; Emeje, M. Recent applications of starch derivatives in nanodrug delivery. Carbohydr. Polym. 2012, 87, 987–994. [Google Scholar] [CrossRef]
- Saboktakin, M. In-vitro and in-vivo characterizations of novel modified starch transdermal drug delivery system. MOJ Proteom. Bioinform. 2017, 5, 132–135. [Google Scholar] [CrossRef][Green Version]
- Pal, K. Starch based hydrogel with potential biomedical application as artificial skin. Afr. J. Biomed. Res. 2006, 9, 23–29. [Google Scholar] [CrossRef]
- Malafaya, P.B.; Elvira, C.; Gallardo, A.; San Román, J.; Reis, R.L. Porous starch-based drug delivery systems processed by a microwave route. J. Biomater. Sci. Polym. Ed. 2001, 12, 1227–1241. [Google Scholar] [CrossRef]
- Janaswamy, S. Encapsulation altered starch digestion: Toward developing starch-based delivery systems. Carbohydr. Polym. 2014, 101, 600–605. [Google Scholar] [CrossRef] [PubMed]
- Sachan, N.; Bhattacharya, A. Modeling and characterization of drug release from glutinous rice starch based hydrogel beads for controlled drug delivery. Int. J. Health Res. 2010, 2, 55398. [Google Scholar] [CrossRef]
- Tesfay, D.; Abrha, S.; Yilma, Z.; Woldu, G.; Molla, F. Preparation, optimization, and evaluation of epichlorohydrin cross-linked enset (ensete ventricosum (welw.) cheeseman) starch as drug release sustaining excipient in microsphere formulation. BioMed Res. Int. 2020, 2020, 2147971. [Google Scholar] [CrossRef]
- Alabi, C.O.; Singh, I.; Odeku, O.A. Evaluation of starch-clay composites as a pharmaceutical excipient in tramadol tablet formulations. Polim. Med. 2020, 50, 33–40. [Google Scholar] [CrossRef]
- Starch-Chitosan Modified Blend as Long-Term Controlled Drug Release For Cancer Therapy. Available online: http://staff.uokufa.edu.iq/publication_details.php?khalidahk.abbas&recordID=6799 (accessed on 26 June 2021).
- Mehrizi, A.A. Synthesis and Characterization of Erythromycin Loaded PVA-Corn Starch Hydrogel Wound Dressing. Available online: https://www.academia.edu/15627564/Synthesis_and_characterization_of_Erythromycin_loaded_PVA_corn_starch_hydrogel_wound_dressing (accessed on 26 June 2021).
- Shibata, N.; Nishumura, A.; Naruhashi, K.; Nakao, Y.; Miura, R. Preparation and pharmaceutical evaluation of new sustained-release capsule including starch-sponge matrix (SSM). Biomed. Pharmacother. 2010, 64, 352–358. [Google Scholar] [CrossRef]
- Balmayor, E.R.; Tuzlakoglu, K.; Azevedo, H.S.; Reis, R.L. Preparation and characterization of starch-poly-ε-caprolactone microparticles incorporating bioactive agents for drug delivery and tissue engineering applications. Acta Biomater. 2009, 5, 1035–1045. [Google Scholar] [CrossRef]
- Xu, J.; Tan, X.; Chen, L.; Li, X.; Xie, F. Starch/microcrystalline cellulose hybrid gels as gastric-floating drug delivery systems. Carbohydr. Polym. 2019, 215, 151–159. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Liu, L.; Tian, H.; Luo, X.; Liu, S. Encapsulation of Lactobacillus plantarum in cellulose based microgel with controlled release behavior and increased long-term storage stability. Carbohydr. Polym. 2019, 223, 115065. [Google Scholar] [CrossRef]
- Ab’lah, N.; Wong, T.W. Starch as oral colon-specific nano- and microparticulate drug carriers. In Polymer Science and Innovative Applications; Elsevier: Amsterdam, The Netherlands, 2020; pp. 287–330. [Google Scholar]
- Yang, X.; Darko, O.K.; Huang, Y.; He, C.; Yang, H.; He, S.; Li, J.; Li, J.; Hocker, B.; Yin, Y. Resistant starch regulates gut microbiota: Structure, biochemistry and cell signalling. Cell Physiol. Biochem. 2017, 42, 306–318. [Google Scholar] [CrossRef] [PubMed]
- Fuentes-Zaragoza, E.; Sánchez-Zapata, E.; Sendra, E.; Sayas, E.; Navarro, C.; Fernández-Lõpez, J.; Pérez-Alvarez, J.A. Resistant starch as prebiotic: A review. Starch Staerke 2011, 63, 406–415. [Google Scholar] [CrossRef]
- Pooresmaeil, M.; Behzadi Nia, S.; Namazi, H. Green encapsulation of LDH(Zn/Al)-5-Fu with carboxymethyl cellulose biopolymer; new nanovehicle for oral colorectal cancer treatment. Int. J. Biol. Macromol. 2019, 139, 994–1001. [Google Scholar] [CrossRef]
- Li, Y.; Yu, S.; Chen, P.; Rojas, R.; Hajian, A.; Berglund, L. Cellulose nanofibers enable paraffin encapsulation and the formation of stable thermal regulation nanocomposites. Nano Energy 2017, 34, 541–548. [Google Scholar] [CrossRef]
- Baek, J.; Ramasamy, M.; Willis, N.C.; Kim, D.S.; Anderson, W.A.; Tam, K.C. Encapsulation and controlled release of vitamin C in modified cellulose nanocrystal/chitosan nanocapsules. Curr. Res. Food Sci. 2021, 4, 215–223. [Google Scholar] [CrossRef]
- Mitbumrung, W.; Suphantharika, M.; Mcclements, D.J.; Winuprasith, T. Encapsulation of vitamin D3 in pickering emulsion stabilized by nanofibrillated mangosteen cellulose: Effect of environmental stresses. J. Food Sci. 2019, 84, 3213–3221. [Google Scholar] [CrossRef]
- Pan-In, P.; Banlunara, W.; Chaichanawongsaroj, N.; Wanichwecharungruang, S. Ethyl cellulose nanoparticles: Clarithomycin encapsulation and eradication of H. pylori. Carbohydr. Polym. 2014, 109, 22–27. [Google Scholar] [CrossRef]
- Serna-Cock, L.; Vallejo-Castillo, V. Probiotic encapsulation. Afr. J. Microbiol. Res. 2013, 7, 4743–4753. [Google Scholar] [CrossRef]
- Salminen, S.; Collado, M.C.; Endo, A.; Hill, C.; Lebeer, S.; Quigley, E.M.M.; Sanders, M.E.; Shamir, R.; Swann, J.R.; Szajewska, H.; et al. The international scientific association of probiotics and prebiotics (ISAPP) consensus statement on the definition and scope of postbiotics. Nat. Rev. Gastroenterol. Hepatol. 2021, 2021, 1–19. [Google Scholar] [CrossRef]
- Silva, D.R.; Sardi, J.C.O.; Pitangui, N.S.; Roque, S.M.; da Silva, A.C.B.; Rosalen, P.L. Probiotics as an alternative antimicrobial therapy: Current reality and future directions. J. Funct. Foods 2020, 73, 104080. [Google Scholar] [CrossRef]
- Misra, S.; Pandey, P.; Mishra, H.N. Novel approaches for co-encapsulation of probiotic bacteria with bioactive compounds, their health benefits and functional food product development: A review. Trends Food Sci. Technol. 2021, 109, 340–351. [Google Scholar] [CrossRef]
- Kailasapathy, K. Microencapsulation of Probiotic Bacteria 39 Microencapsulation of Probiotic Bacteria: Technology and Potential Applications. Available online: https://www.caister.com/bacteria-plant (accessed on 27 July 2021).
- Burgain, J.; Gaiani, C.; Linder, M.; Scher, J. Encapsulation of probiotic living cells: From laboratory scale to industrial applications. J. Food Eng. 2011, 104, 467–483. [Google Scholar] [CrossRef]
- Kailasapathy, K. Encapsulation technologies for functional foods and nutraceutical product development. CAB Rev. 2009, 4, 33. [Google Scholar] [CrossRef]
- Viability and Survival of Free and Encapsulated Probiotic Bacteria in Cheddar Cheese|Western Sydney University ResearchDirect. Available online: https://researchdirect.westernsydney.edu.au/islandora/object/uws%3A5276 (accessed on 30 July 2021).
- Homayouni, A.; Azizi, A.; Ehsani, M.R.; Yarmand, M.S.; Razavi, S.H. Effect of microencapsulation and resistant starch on the probiotic survival and sensory properties of synbiotic ice cream. Food Chem. 2008, 111, 50–55. [Google Scholar] [CrossRef]
- Mcmaster, L.D.; Kokott, S.A. Micro-encapsulation of Bifidobacterium lactis for incorporation into soft foods. World J. Microbiol. Biotechnol. 2005, 21, 723–728. [Google Scholar] [CrossRef]
- Muthukumarasamy, P.; Holley, R.A. Survival of Escherichia coli O157:H7 in dry fermented sausages containing micro-encapsulated probiotic lactic acid bacteria. Food Microbiol. 2007, 24, 82–88. [Google Scholar] [CrossRef] [PubMed]
- Frakolaki, G.; Giannou, V.; Kekos, D.; Tzia, C. A review of the microencapsulation techniques for the incorporation of probiotic bacteria in functional foods. Crit. Rev. Food Sci. Nutr. 2021, 61, 1515–1536. [Google Scholar] [CrossRef]
- Zhao, C.; Zhu, Y.; Kong, B.; Huang, Y. Dual-core prebiotic microcapsule encapsulating probiotics for metabolic syndrome. ACS Appl. Mater. Interfaces 2020, 12, 42586–42594. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Lin, J.; Zhong, Q. The increased viability of probiotic Lactobacillus salivarius NRRL B-30514 encapsulated in emulsions with multiple lipid-protein-pectin layers. Food Res. Int. 2015, 71, 9–15. [Google Scholar] [CrossRef]
- Whorwell, P.J.; Morel, J.; Duane, C. Efficacy of an encapsulated probiotic Bifidobacterium infantis 35624 in women with irritable bowel syndrome. Am. J. Gastroenterol. 2006, 101, 1581–1590. [Google Scholar] [CrossRef]
- Anselmo, A.C.; Mchugh, K.J.; Webster, J.; Langer, R.; Jaklenec, A. Layer-by-layer encapsulation of probiotics for delivery to the microbiome. Adv. Mater. 2016, 28, 9486–9490. [Google Scholar] [CrossRef] [PubMed]
- Zheng, D.-W.; Li, R.-Q.; An, J.-X.; Xie, T.-Q.; Han, Z.-Y.; Xu, R.; Fang, Y.; Zhang, X.-Z. Prebiotics-encapsulated probiotic spores regulate gut microbiota and suppress colon cancer. Adv. Mater. 2020, 32, 2004529. [Google Scholar] [CrossRef] [PubMed]
- Solanki, H.K.; Pawar, D.D.; Shah, D.A.; Prajapati, V.D.; Jani, G.K.; Mulla, A.M.; Thakar, P.M. Development of microencapsulation delivery system for long-term preservation of probiotics as biotherapeutics agent. BioMed Res. Int. 2013, 2013, 620719. [Google Scholar] [CrossRef]
- Martín, M.J.; Lara-Villoslada, F.; Ruiz, M.A.; Morales, M.E. Microencapsulation of bacteria: A review of different technologies and their impact on the probiotic effects. Innov. Food Sci. Emerg. Technol. 2015, 27, 15–25. [Google Scholar] [CrossRef]
- Capela, P.; Hay, T.K.C.; Shah, N.P. Effect of homogenisation on bead size and survival of encapsulated probiotic bacteria. Food Res. Int. 2007, 40, 1261–1269. [Google Scholar] [CrossRef]
- Razavi, S.; Janfaza, S.; Tasnim, N.; Gibson, D.L.; Hoorfar, M. Nanomaterial-based encapsulation for controlled gastrointestinal delivery of viable probiotic bacteria. Nanoscale Adv. 2021, 3, 2699–2709. [Google Scholar] [CrossRef]
- Darjani, P.; Hosseini Nezhad, M.; Kadkhodaee, R.; Milani, E. Influence of prebiotic and coating materials on morphology and survival of a probiotic strain of Lactobacillus casei exposed to simulated gastrointestinal conditions. LWT Food Sci. Technol. 2016, 73, 162–167. [Google Scholar] [CrossRef]
- Anal, A.K.; Singh, H. Recent advances in microencapsulation of probiotics for industrial applications and targeted delivery. Trends Food Sci. Technol. 2007, 18, 240–251. [Google Scholar] [CrossRef]
- Tripathi, M.K.; Giri, S.K. Probiotic functional foods: Survival of probiotics during processing and storage. J. Funct. Foods 2014, 9, 225–241. [Google Scholar] [CrossRef]
- ISO 10993-5:2009(en), Biological Evaluation of Medical Devices—Part 5: Tests for In Vitro Cytotoxicity. Available online: https://www.iso.org/obp/ui/#iso:std:iso:10993:-5:ed-3:v1:en (accessed on 27 June 2021).
- Singh, P.; Medronho, B.; dos Santos, T.; Nunes-Correia, I.; Granja, P.; Miguel, M.G.; Lindman, B. On the viability, cytotoxicity and stability of probiotic bacteria entrapped in cellulose-based particles. Food Hydrocoll. 2018, 82, 457–465. [Google Scholar] [CrossRef]
- Puscaselu, R.G.; Lobiuc, A.; Dimian, M.; Covasa, M. Alginate: From food industry to biomedical applications and management of metabolic disorders. Polymers 2020, 12, 2417. [Google Scholar] [CrossRef] [PubMed]
- Lopes, S.; Bueno, L.; De Aguiar Júnior, F.; Finkler, C.L.L. Preparation and characterization of alginate and gelatin microcapsules containing Lactobacillus rhamnosus. An. Acad. Bras. Cienc. 2017, 89, 1601–1613. [Google Scholar] [CrossRef] [PubMed]
- Ramos, P.E.; Silva, P.; Alario, M.M.; Pastrana, L.M.; Teixeira, J.A.; Cerqueira, M.A.; Vicente, A.A. Effect of alginate molecular weight and M/G ratio in beads properties foreseeing the protection of probiotics. Food Hydrocoll. 2018, 77, 8–16. [Google Scholar] [CrossRef]
- Zhang, Z.; Gu, M.; You, X.; Sela, D.A.; Xiao, H.; Mcclements, D.J. Encapsulation of bifidobacterium in alginate microgels improves viability and targeted gut release. Food Hydrocoll. 2021, 116, 106634. [Google Scholar] [CrossRef]
- Sathyabama, S.; Kumar, M.R.; Devi, P.B.; Vijayabharathi, R.; Priyadharisini, V.B. Co-encapsulation of probiotics with prebiotics on alginate matrix and its effect on viability in simulated gastric environment. LWT Food Sci. Technol. 2014, 57, 419–425. [Google Scholar] [CrossRef]
- Shi, L.E.; Li, Z.H.; Li, D.T.; Xu, M.; Chen, H.Y.; Zhang, Z.L.; Tang, Z.X. Encapsulation of probiotic lactobacillus bulgaricus in alginate’milk microspheres and evaluation of the survival in simulated gastrointestinal conditions. J. Food Eng. 2013, 117, 99–104. [Google Scholar] [CrossRef]
- Martin, M.J.; Lara-Villoslada, F.; Ruiz, M.A.; Morales, M.E. Effect of unmodified starch on viability of alginate-encapsulated Lactobacillus fermentum CECT5716. LWT Food Sci. Technol. 2013, 53, 480–486. [Google Scholar] [CrossRef]
- Cheow, W.S.; Kiew, T.Y.; Hadinoto, K. Controlled release of Lactobacillus rhamnosus biofilm probiotics from alginate-locust bean gum microcapsules. Carbohydr. Polym. 2014, 103, 587–595. [Google Scholar] [CrossRef]
- Song, H.; Yu, W.; Liu, X.; Ma, X. Improved probiotic viability in stress environments with post-culture of alginate-chitosan microencapsulated low density cells. Carbohydr. Polym. 2014, 108, 10–16. [Google Scholar] [CrossRef]
- Kamalian, N.; Mirhosseini, H.; Mustafa, S.; Manap, M.Y.A. Effect of alginate and chitosan on viability and release behavior of Bifidobacterium pseudocatenulatum G4 in simulated gastrointestinal fluid. Carbohydr. Polym. 2014, 111, 700–706. [Google Scholar] [CrossRef] [PubMed]
- Cook, M.T.; Tzortzis, G.; Khutoryanskiy, V.V.; Charalampopoulos, D. Layer-by-layer coating of alginate matrices with chitosan-alginate for the improved survival and targeted delivery of probiotic bacteria after oral administration. J. Mater. Chem. B 2013, 1, 52–60. [Google Scholar] [CrossRef] [PubMed]
- de Araújo Etchepare, M.; Raddatz, G.C.; de Moraes Flores, É.M.; Zepka, L.Q.; Jacob-Lopes, E.; Barin, J.S.; Ferreira Grosso, C.R.; de Menezes, C.R. Effect of resistant starch and chitosan on survival of Lactobacillus acidophilus microencapsulated with sodium alginate. LWT Food Sci. Technol. 2016, 65, 511–517. [Google Scholar] [CrossRef]
- Fareez, I.M.; Lim, S.M.; Mishra, R.K.; Ramasamy, K. Chitosan coated alginate-xanthan gum bead enhanced pH and thermotolerance of Lactobacillus plantarum LAB12. Int. J. Biol. Macromol. 2015, 72, 1419–1428. [Google Scholar] [CrossRef]
- Prata, A.S.; Grosso, C.R.F. Production of microparticles with gelatin and chitosan. Carbohydr. Polym. 2015, 116, 292–299. [Google Scholar] [CrossRef]
- Morrison, I.D.; Ross, S. Colloidal Dispersions: Suspensions, Emulsions, and Foams; John Wiley&Sons: Hoboken, NJ, USA, 2002; p. 616. [Google Scholar]
- Kwiecień, I.; Kwiecień, M. Application of polysaccharide-based hydrogels as probiotic delivery systems. Gels 2018, 4, 47. [Google Scholar] [CrossRef] [PubMed]
- Albadran, H.A.; Monteagudo-Mera, A.; Khutoryanskiy, V.V.; Charalampopoulos, D. Development of chitosan-coated agar-gelatin particles for probiotic delivery and targeted release in the gastrointestinal tract. Appl. Microbiol. Biotechnol. 2020, 104, 5749–5757. [Google Scholar] [CrossRef]
- Jia, S.; Zhou, K.; Pan, R.; Wei, J.; Liu, Z.; Xu, Y. Oral immunization of carps with chitosan–alginate microcapsule containing probiotic expressing spring viremia of carp virus (SVCV) G protein provides effective protection against SVCV infection. Fish Shellfish Immunol. 2020, 105, 327–329. [Google Scholar] [CrossRef]
- Argin, S.; Kofinas, P.; Lo, Y.M. The cell release kinetics and the swelling behavior of physically crosslinked xanthan–chitosan hydrogels in simulated gastrointestinal conditions. Food Hydrocoll. 2014, 40, 138–144. [Google Scholar] [CrossRef]
- Cook, M.T.; Tzortis, G.; Charalampopoulos, D.; Khutoryanskiy, V.V. Production and evaluation of dry alginate-chitosan microcapsules as an enteric delivery vehicle for probiotic bacteria. Biomacromolecules 2011, 12, 2834–2840. [Google Scholar] [CrossRef] [PubMed]
- De Prisco, A.; Maresca, D.; Ongeng, D.; Mauriello, G. Microencapsulation by vibrating technology of the probiotic strain Lactobacillus reuteri DSM 17938 to enhance its survival in foods and in gastrointestinal environment. LWT Food Sci. Technol. 2015, 61, 452–462. [Google Scholar] [CrossRef]
- Graff, S.; Hussain, S.; Chaumeil, J.-C.; Charrueau, C. Increased intestinal delivery of viable saccharomyces boulardii by encapsulation in microspheres. Pharm. Res. 2008, 25, 1290–1296. [Google Scholar] [CrossRef] [PubMed]
- Krasaekoopt, W.; Watcharapoka, S. Effect of addition of inulin and galactooligosaccharide on the survival of microencapsulated probiotics in alginate beads coated with chitosan in simulated digestive system, yogurt and fruit juice. LWT Food Sci. Technol. 2014, 57, 761–766. [Google Scholar] [CrossRef]
- Mirtič, J.; Rijavec, T.; Zupančič, Š.; Zvonar Pobirk, A.; Lapanje, A.; Kristl, J. Development of probiotic-loaded microcapsules for local delivery: Physical properties, cell release and growth. Eur. J. Pharm. Sci. 2018, 121, 178–187. [Google Scholar] [CrossRef]
- Rodklongtan, A.; La-ongkham, O.; Nitisinprasert, S.; Chitprasert, P. Enhancement of Lactobacillus reuteri KUB-AC5 survival in broiler gastrointestinal tract by microencapsulation with alginate–chitosan semi-interpenetrating polymer networks. J. Appl. Microbiol. 2014, 117, 227–238. [Google Scholar] [CrossRef]
- Wu, Q.X.; Xu, X.; Xie, Q.; Tong, W.Y.; Chen, Y. Evaluation of chitosan hydrochloride-alginate as enteric micro-probiotic-carrier with dual protective barriers. Int. J. Biol. Macromol. 2016, 93, 665–671. [Google Scholar] [CrossRef]
- Çakmak, H.; Özselek, Y.; Turan, O.Y.; Fıratlıgil, E.; Karbancioğlu-Güler, F. Whey protein isolate edible films incorporated with essential oils: Antimicrobial activity and barrier properties. Polym. Degrad. Stab. 2020, 179, 109285. [Google Scholar] [CrossRef]
- Zhang, B.; Liu, Y.; Wang, H.; Liu, W.; Cheong, K.; Teng, B. Characterization of seaweed polysaccharide-based bilayer films containing essential oils with antibacterial activity. LWT Food Sci. Technol. 2021, 150, 111961. [Google Scholar] [CrossRef]
- Alfaro-Galarza, O.; López-Villegas, E.O.; Rivero-Perez, N.; Tapia- Maruri, D.; Jiménez-Aparicio, A.R.; Palma-Rodríguez, H.M.; Vargas-Torres, A. Protective effects of the use of taro and rice starch as wall material on the viability of encapsulated Lactobacillus paracasei subsp. Paracasei. LWT Food Sci. Technol. 2020, 117, 108686. [Google Scholar] [CrossRef]
- Ashwar, B.A.; Gani, A.; Gani, A.; Shah, A.; Masoodi, F.A. Production of RS4 from rice starch and its utilization as an encapsulating agent for targeted delivery of probiotics. Food Chem. 2018, 239, 287–294. [Google Scholar] [CrossRef]
- Dafe, A.; Etemadi, H.; Dilmaghani, A.; Mahdavinia, G.R. Investigation of pectin/starch hydrogel as a carrier for oral delivery of probiotic bacteria. Int. J. Biol. Macromol. 2017, 97, 536–543. [Google Scholar] [CrossRef]
- Benavent-Gil, Y.; Rodrigo, D.; Rosell, C.M. Thermal stabilization of probiotics by adsorption onto porous starches. Carbohydr. Polym. 2018, 197, 558–564. [Google Scholar] [CrossRef]
- Zanjani, M.A.K.; Ehsani, M.R.; Ghiassi Tarzi, B.; Sharifan, A. Promoting Lactobacillus casei and Bifidobacterium adolescentis survival by microencapsulation with different starches and chitosan and poly L-lysine coatings in ice cream. J. Food Process. Preserv. 2018, 42, e13318. [Google Scholar] [CrossRef]
- Lancuški, A.; Aiman, A.A.; Avrahami, R.; Vilensky, R.; Vasilyev, G.; Zussman, E. Design of starch-formate compound fibers as encapsulation platform for biotherapeutics. Carbohydr. Polym. 2017, 158, 68–76. [Google Scholar] [CrossRef]
- Reyes, V.; Chotiko, A.; Chouljenko, A.; Sathivel, S. Viability of Lactobacillus acidophilus NRRL B-4495 encapsulated with high maize starch, maltodextrin, and gum arabic. LWT Food Sci. Technol. 2018, 96, 642–647. [Google Scholar] [CrossRef]
- Thangrongthong, S.; Puttarat, N.; Ladda, B.; Itthisoponkul, T.; Pinket, W.; Kasemwong, K.; Taweechotipatr, M. Microencapsulation of probiotic Lactobacillus brevis ST-69 producing GABA using alginate supplemented with nanocrystalline starch. Food Sci. Biotechnol. 2020, 29, 1475–1482. [Google Scholar] [CrossRef] [PubMed]
- Sultana, K.; Godward, G.; Reynolds, N.; Arumugaswamy, R.; Peiris, P.; Kailasapathy, K. Encapsulation of probiotic bacteria with alginate–starch and evaluation of survival in simulated gastrointestinal conditions and in yoghurt. Int. J. Food Microbiol. 2000, 62, 47–55. [Google Scholar] [CrossRef]
- Zabihollahi, N.; Alizadeh, A.; Almasi, H.; Hanifian, S.; Hamishekar, H. Development and characterization of carboxymethyl cellulose based probiotic nanocomposite film containing cellulose nanofiber and inulin for chicken fillet shelf life extension. Int. J. Biol. Macromol. 2020, 160, 409–417. [Google Scholar] [CrossRef] [PubMed]
- Singh, P.; Medronho, B.; Miguel, M.G.; Esquena, J. On the encapsulation and viability of probiotic bacteria in edible carboxymethyl cellulose-gelatin water-in-water emulsions. Food Hydrocoll. 2018, 75, 41–50. [Google Scholar] [CrossRef]
- Dafe, A.; Etemadi, H.; Zarredar, H.; Mahdavinia, G.R. Development of novel carboxymethyl cellulose/k-carrageenan blends as an enteric delivery vehicle for probiotic bacteria. Int. J. Biol. Macromol. 2017, 97, 299–307. [Google Scholar] [CrossRef] [PubMed]
- Chen, B.; Lin, X.; Lin, X.; Li, W.; Zheng, B.; He, Z. Pectin-microfibrillated cellulose microgel: Effects on survival of lactic acid bacteria in a simulated gastrointestinal tract. Int. J. Biol. Macromol. 2020, 158, 826–836. [Google Scholar] [CrossRef]
- Chitprasert, P.; Sudsai, P.; Rodklongtan, A. Aluminum carboxymethyl cellulose–rice bran microcapsules: Enhancing survival of Lactobacillus reuteri KUB-AC5. Carbohydr. Polym. 2012, 90, 78–86. [Google Scholar] [CrossRef] [PubMed]
- Singh, P.; Medronho, B.; Alves, L.; da Silva, G.J.; Miguel, M.G.; Lindman, B. Development of carboxymethyl cellulose-chitosan hybrid micro- and macroparticles for encapsulation of probiotic bacteria. Carbohydr. Polym. 2017, 175, 87–95. [Google Scholar] [CrossRef]
- Huq, T.; Fraschini, C.; Khan, A.; Riedl, B.; Bouchard, J.; Lacroix, M. Alginate based nanocomposite for microencapsulation of probiotic: Effect of cellulose nanocrystal (CNC) and lecithin. Carbohydr. Polym. 2017, 168, 61–69. [Google Scholar] [CrossRef] [PubMed]
- Fareez, I.M.; Lim, S.M.; Zulkefli, N.A.A.; Mishra, R.K.; Ramasamy, K. Cellulose Derivatives Enhanced Stability of Alginate-Based Beads Loaded with Lactobacillus plantarum LAB12 against Low pH, High Temperature and Prolonged Storage. Probiotics Antimicrob. Proteins 2017, 10, 543–557. [Google Scholar] [CrossRef]


| Characteristic of Materials | Biopolymers | Synthetic Polymers | References |
|---|---|---|---|
| Main source | Agro-resources | Petroleum and gas | [48] |
| Biodegradability/environmentally friendly | YES | NO/slow | [49,50] |
| Structure | Well defined | Stochastic | [48] |
| Chemical backbone structure | Carbon, oxygen, and nitrogen | Mostly carbon | [48] |
| Dispersity | Unity | ˃1 | [51] |
| Physicochemical resistance | Low | High | [52] |
| Toxicity | Low | High | [41] |
| Thermal stability | Low | High | [52] |
| Mechanical properties | Low | High | [53] |
| Sustainability | High | Low | [52] |
| Availability | High | Decreasing | [52] |
| Cost | High (depends on the type) | Low | [54] |
| Biopolymers | Entrapped Substances | Applications | Benefits | References |
|---|---|---|---|---|
| Alginate | Rifampicin | Drug delivery carriers | Nanoparticles are pH sensitive with the highest release of the active substance occurring at a pH of 7.4. Toxicity and safety tests were excellent with no systemic toxicity after oral administration of nanoparticles. | [92] |
| Alginate | Ibuprofen | Drug delivery system | Controlled drug release was maintained for 4 h (67.53% of the drug formulation). | [93] |
| Alginate and methylcellulose | Indomethacin | Drug delivery carrier | Controlled drug release. There was no interaction between the loaded drug and the polymers. | [94] |
| Alginate | Metformin hydrochloride | Drug delivery system | Good release time; microspheres may be used in the treatment of diabetes. | [95] |
| Alginate | Diclofenac sodium | Controlled-release microparticles | The drug: polymer (ratio 1:3) was obtained by emulsification and the drug release followed zero order kinetics, optimum for controlled drug release delivery. | [96] |
| Alginate and sodium carboxymethylcellulose | Ceftriaxone sodium | Multiarticulate beads | The use of the biopolymer matrix decreased drug release in gastric conditions but sustained it at intestinal pH. The beads swelled at pH 1.2 but particle diffusion and erosion occurred at pH 6.8. | [97] |
| Alginate | Furosemide | Controlled drug delivery beads | Drug release was controlled due to the thicker membrane and reduced beads swelling. Release of Furosemide depends on the conditions of the coating treatment. | [98] |
| Alginate | Isoniazid | Oral drug delivery | Microspheres were present in the intestinal lumen 4 h after administration and were detectable in the intestine after 24 h of oral administration. Approximately 26% of the drug was released in the gastrointestinal fluid (pH 1.2) in 6 h and 71.25% in the simulated intestinal fluid (pH 7.4) in 30 h. | [99] |
| Alginate | Nicotinic acid | Aerogels | The release of the drug was prolonged when the core was surrounded by several alginate-based membranes. Due to coating, 50% of the drug was released within 4 h. | [100] |
| Alginate, carboxymethylcellulose, and chitosan | Amoxicillin | Coated beads | In gastric pH conditions, the drug release was prolonged from 61 min to up to 8 h. |
| Biopolymers | Entrapped Substances | Applications | Benefits | References |
|---|---|---|---|---|
| Chitosan | Acyclovir | Drug delivery system | The grafting efficiency was 94% and the slow release of the drug was prolonged to 12 h. | [113] |
| Chitosan | Chlorhexidine diacetate | Buccal tablets | The tablets dissolved more quickly in vitro than chlorhexidine powder and both maintained and even improved the antimicrobial activity of the drug, particularly against Candida albicans, due to antimicrobial activity of the polymer itself. | [114] |
| Chitosan aspartate, glutamate, and hydrochloride | Vancomycin | Peptidic model drug | The sustained release from the microspheres minimized its solubilization in the upper gastrointestinal tract. | [115] |
| Chitosan | Tetracycline | Controlled drug system | The concentration of drug released was above the minimum limit required for the inhibition of Staphylococcus aureus. | [116] |
| Chitosan and oleic acid | Camptothecin | Controlled drug system | The encapsulation efficiency was about 78%. When its effectiveness in the simulated gastrointestinal fluids was tested, the drug was released slowly into the gastric environment. In intestinal fluids, the drug release was controlled. The drug embedded in chitosan was 75% protected from hydrolysis. | [117] |
| Chitosan | Satranidazole | Subgingival films for periodontitis | The drug was released for 96 h; the population of gram-positive bacteria was reduced. | [118] |
| Chitosan and alginate | Amygdalin | Drug delivery system | The controlled amygdalin release was performed for 10 h; the maximum amygdalin released was 70.46% at pH 3.1, 81.86% at pH 5.0, and 86.03% at pH 7.4. | [119] |
| Chitosan and graphene | Isosfamide | Sustained drug microspheres | The drug diffusion was the most controlled for when isosfamide was entrapped in microspheres. | [120] |
| Chitosan and xanthan gum | Ciprofloxacin | Controlled-release hydrogel | The entrapment efficiency of the prepared hydrogel increased with the drug increasing concentration and the maximum was reached at 93.8%. | [121] |
| Chitosan | Interferon-α | Nanoparticles for oral delivery | In mice, the nanoparticles were found in plasma at 1 h after administration, unlike the commercial interferon which could not be detected. | [122] |
| Biopolymers | Entrapped Substances | Applications | Benefits | References |
|---|---|---|---|---|
| Corn starch | Chlorhexidine gluconate | Long-term drug delivery system | In vitro drug release was observed for 21 days and it inhibited Staphylococcus aureus growth. | [134] |
| Corn starch | Clonidine | Transdermal patches | Transdermal patches with a single dose of 30 μg hydrogel had an effect 15 min after application in treated mice. | [143] |
| Corn starch | Salicylic acid | Hydrogel membrane | The diffusion of the drug through the membrane was 4.11 × 10−6 cm2/s. The hydrogel was tested as an artificial skin for transferring nutrients or medicines, or for healing substances to the target area. | [144] |
| Corn starch/ethylene vinyl alcohol blend | Non-steroid anti-inflammatory agent | Drug delivery carriers | In vitro tests showed an immediate burst effect, followed by a slower, controlled release of the drug that lasted up to 10 days. | [145] |
| Potato starch | Ibuprofen, benzocaine, and sulphapyridine | Starch-based stable carriers | Encapsulation altered starch digestion; resistant starch was available in the colon for fermentation. | [146] |
| Glutinous rice starch, sodium alginate, and calcium chloride | Metformin hydrochloride | Hydrogel beads for controlled drug delivery | The initial drug entrapment efficiency was very low for the metformin hydrochloride because of its high solubility. Encapsulation improved it when combined with pre-gelatinized starch gel. | [147] |
| Ensete ventricosum starch | Epichlorohydrin | Drug-release sustaining pharmaceutical excipient | The in vitro drug release profile showed a minimum burst release, followed by a sustained release for 12 h. | [148] |
| Starch-clay composites | Tramadol | Tablet formulations | The controlled drug release of tramadol from starch-clay biocomposites was achieved in approximately 350 min. | [149] |
| Starch-chitosan | Hydroxyurea | Cancer therapy | The drug release was sensitive to pH and increased in the acid environment. The drug/starch/chitosan had a toxicity effect and, at certain concentrations, killed cancer cells. | [150] |
| PVA-corn starch hydrogel | Erythromycin | Wound dressing | The release of erythromycin from the PVA/corn starch network was higher than the drug containing PVA hydrogel (after 1800 min, released 76.7 mg of the total drug). | [151] |
| Corn starch-sponge matrix | Uranine, indomethacin, and nifedipine | Sustained-release capsule | After intraduodenal administration, 2.5% of the capsule exhibited a sustained release of the drug in the plasma. | [152] |
| Starch-poly-ε caprolactone | Dexamethasone | Drug delivery and tissue engineering applications | The drug from the outermost layer of the microparticles was quickly released. In vitro tests showed a sustained-release pattern for 30 days. | [153] |
| High-amylose starch-microcrystalline cellulose | Ranitidine hydrochloride | Gastric-floating drug delivery systems | In vitro tests indicated that the system with 3:7 (wt./wt.) starch/cellulose ratio maintained the buoyancy for more than a day; the drug release was 45.87% in the first hour, followed by a sustained release for up to 10 h. | [154] |
| Maize starch | Probiotics, e.g., Lactobacillus plantarum | Microencapsulated probiotic | In low acid environments, L. plantarum encapsulated in the starch matrix was more stable. After simulated digestion and heating treatments, the cells maintained their high viability, unlike formulations with native starch | [155] |
| Biopolymers | Encapsulated Strain | Encapsulation Method | Benefits | References |
|---|---|---|---|---|
| Alginate and gelatin | Lactobacillus rhamnosus | Extrusion | The cells of L. rhamnosus survived in beads with 105 CFU/g after four months (initially 109 CFU/g). | [191] |
| Alginate | Lactococcus lactis spp. cremoris | Extrusion | No release of bacteria in the stomach simulated condition (first 120 min) or the survival in the intestinal fluid until 240 min. | [192] |
| Alginate | Bifidobacterium pseudocatenulatum | Extrusion | None of the uncoated probiotic cells survived after immersion in the simulated small intestine fluid. By contrast, 5.6 log10 CFU/g of viable probiotic cells remained in the tested microgels. | [193] |
| Alginate | Staphylococcus succinus and Enterococcus fecium | Extrusion | The encapsulated cells showed 98.75–88.75% of viability in simulated gastric fluids. Survival was constant throughout the storage time and decreased from 8.1 log CFU/mL to 7.9 log CFU/mL after 30 days of storage at 4 °C. | [194] |
| Alginate and milk | Lactobacillus bulgaricus | Extrusion | The viability of the encapsulated probiotic was the same after 120 min of incubation in an acid medium (simulated gastric fluid with pH 2.5). The viability of encapsulated L. bulgaricus was kept at 8 log CFU/g after 120 min of incubation at pH 2.0. Stability of the encapsulated probiotic can be preserved for one month after storage at 4 °C. | [195] |
| Alginate and starch | Lactobacillus fermentum | Lyophilization | The survival rate of the probiotic was significantly higher for microparticles blended with starch than those with no starch. | [196] |
| Alginate, chitosan, and locust beam | Lactobacillus rhamnosus | Freeze-drying | In contrast to the alginate-based capsules, the alginate locust beam capsules improved stress tolerance (6× for freeze-drying, 100× for thermotolerance, and 10× for acid). | [197] |
| Alginate and chitosan | Saccharomyces cerevisiae Y235 | Emulsification | The viable microencapsulated cells were kept at 7.00 log CFU/g after six months at −20 °C and remained 6.29 log CFU/g after incubation in SGF for 2 h and in SIF for 12 h, reaching the standard value (106–107 CFU/g). | [198] |
| Alginate and chitosan | Bifidobacterium pseudocatenulatum | The highest stability of B. pseudocatenulatum was at the highest concentrations of alginate (4.41 g/100 mL) and chitosan (0.56 g/100 mL). Resistance of alginate–chitosan capsule in SGF was better than in SIF. | [199] | |
| Alginate and chitosan | Bifidobacterium breve | Layer-by-layer | Three-layer coated matrix was the best method to increase viability from <3 log CFU/mL, seen in encapsulated cells, up to a maximum of 8.84 ± 0.17 log CFU/mL upon exposure to in vitro gastric conditions. Multilayer-coated alginate released their loads to the intestine with a gradual delivery over 240 min. | [200] |
| Alginate, starch, and chitosan | Lactobacillus acidophilus | Extrusion | Biopolymers ensured better stability of probiotics after exposure to SGF and SIF with 6.35 log CFU/g, while lower counts were noticed for freeze-dried microcapsules. During storage, cell viability of the probiotics stored in the freeze-dried form was up to six logs for 30 days and 135 days in the moist form when kept at room temperature. | [201] |
| Alginate, chitosan, and xanthan gum | Lactobacillus plantarum | Extrusion | Sequential incubation of biopolymers in SGF and SIF facilitated high survival of L. plantarum (95%) at pH < 2. Encapsulation improved storage stability of L. plantarum at 4 °C. | [202] |
| Biopolymers | Encapsulated Strain | Encapsulation Method | Benefits | References |
|---|---|---|---|---|
| Chitosan, agar, and gelatin | Lactobacillus plantarum | Emulsification | Particles with a diameter of approximately 6 mm did not solubilize in SGF 20 min after exposure. Cell viability in the biopolymer-free formula decreased completely after 2 h, unlike coated particles whose viability was 9.2 CFU/g after 2 h. | [206] |
| Chitosan and alginate | vaccine with Lactobacillus plantarum | Extrusion | The oral vaccine containing L. plantarum, used against spring viremia of carp virus, was effective even after 56 days due to the encapsulation. | [207] |
| Chitosan and xanthan gum | Pediococcus acidilactici | Extrusion | The encapsulated cells maintained their cell viability for 8 h in the gastrointestinal fluid with maximum release occurring after 24 h. The encapsulated cells maintained their viability for three days when tested in deionized water. | [208] |
| Chitosan and alginate | Bifidobacterium breve | Extrusion | In an acidic medium (pH 2), cell viability was maintained for 1 h. As pH increased (4 and 5), cell viability increased to 120 min. After 2 h, the swelling ratio decreased, a sign that the microcapsules began to disintegrate. Chitosan maintained cellular stability at pH 4 and 5, and alginate at pH 2. | [209] |
| Chitosan and alginate | Lactobacillus reuteri DSM 17938 | Vibration technology | Unencapsulated cells were more labile to gastrointestinal stress conditions (reduction by 2.09 log cycles after 3 h). The encapsulated ones resisted better with a reduction of 0.82 log cycles. | [210] |
| Chitosan and alginate | Saccharomyces boulardii | Extrusion | Encapsulation of strains with chitosan and alginate facilitated maintenance of cell viability up to 6 h after administration in mice. | [211] |
| Chitosan and alginate | L. acidophilus and L. casei | Extrusion | Galactooligosaccharides potentiated the effect of microencapsulation. Cell viability was reduced by 3.1 logs for L. acidophilus and 2.9 logs for L. casei when tested at a very low pH (1.55) of SIF. | [212] |
| Chitosan and alginate | Bacteria strain 4.1.Z (B. amyloliquefaciens, B. subtilis, and B. methylotrophicus) | Vibration and extrusion | After lyophilization, the microcapsules maintained their viability (106–107 CFU/g) for about two months under refrigeration. Chitosan maintained the integrity of capsules for 24 h. | [213] |
| Chitosan and alginate | Lactobacillus reuteri KUB-AC5 | Emulsification | The viability of non-encapsulated cells decreased in 40 min from 8 logs CFU/mL to <4 log CFU/mL, being completely eliminated after 1 h. The encapsulated cells were much more stable with a reduction of 1 log CFU/mL after 180 min at pH 1.8. | [214] |
| Chitosan and hydrochloride-alginate | Bacillus licheniformis | Orifice-polymerization method | The chitosan coating protected the microcapsules; cell release (6.19 CFU/mL) in 1 h in SGF (pH 2) and 4 h in the simulated intestinal fluid (pH 6). | [215] |
| Biopolymers | Encapsulated Strain | Encapsulation Method | Benefits | References |
|---|---|---|---|---|
| Rice starch | Lactobacillus casei, Lactobacillus brevis, and Lactobacillus plantarum | Extrusion | The viability of encapsulated cells (8.27/8.46/7.65 log CFU/g) was kept constant for two months at refrigeration. In contrast, non-encapsulated cells lost their viability by approximately 3 log CFU/g during storage. | [219] |
| Starch and pectin | Lactobacillus plantarum | Extrusion | Cell viability was reduced from 10 log CFU/g to 1 log CFU/g for free cells maintained for 2 h in gastric conditions (pH 1.5–3). Cells encapsulated in pectin had higher viability (4.6 log CFU/g) but the best protection was observed with the addition of starch to which the viability increased to 6.94 log CFU/g. | [220] |
| Starch from corn and rice | Lactobacillus plantarum | Freeze-drying | Encapsulated cells showed thermal stability and maintained their integrity for 35 min at 55 °C. Unencapsulated cells subjected to the same treatment lost their viability by 63% after only 10 min of exposure to 55 °C. | [221] |
| Starch, alginate, chitosan, and inulin | Lactobacillus casei and Bifidobacterium bifidum | Emulsification | Encapsulated L. casei and B. bifidum lost their viability when subjected to simulated gastric conditions for 120 min. Cell viability decreased from 25.10 × 1010 CFU/mL to 6.30 × 106 CFU/mL for L. casei. Encapsulated B. bifidum lost 4.65 log/mL of the bacterial culture, while the unencapsulated form had undetectable cell viability after 90 min. | [222] |
| Starch and alginate | Lactobacillus fermentum | Emulsification | Encapsulated in the matrix, cells maintained viability when stored at 4 °C for 45 days. In environmental conditions, however, cells showed a decrease of 1.7 log after 24 h, with complete loss after 2 weeks. | [196] |
| Starch | Lactobacillus paracasei | Electrospinning | Tested at different storage temperatures (4, 25, and 37 °C), L. paracasei cells maintained their initial viability of 13.6 × 10 CFU/mL when stored for three weeks at 4 °C and 25 °C but not at 37 °C. Unencapsulated cells lost about 90% of their viability regardless of the storage temperature. | [223] |
| Maize starch, maltodextrin, and gum arabic | Lactobacillus acidophilus | Spray-drying | After 30days of storage at room temperature, only strains encapsulated with maltodextrin, namely gum arabic, maintained their cell viability of 106 CFU/g. After 60 days, no encapsulating material prevented the loss of cell viability. Of the tested coatings, starch least protected the bacterial strains. | [224] |
| Taro and rice starch | Lactobacillus paracasei | Spray-drying | When stored, the taro–starch encapsulated strains were more stable; cells maintained their viability for a month, both at temperatures of 4 °C and 25 °C. | [218] |
| Cassava starch and alginate | Lactobacillus brevis | Emulsification | Encapsulation efficiency was higher than 89%. In gastrointestinal conditions, cell viability was better for microcapsules than free cells (96.07% compared to 76.51%). After 5 h of maintenance in the same conditions, viability of L. brevis encapsulated cells was 8.69 log CFU/mL, unlike the non-encapsulated ones with 6.87 log CFU/mL. | [225] |
| Starch and alginate | Lactobacillus casei | Extrusion | The addition of 2% starch to the alginate-based film-forming solution increased cell viability from 4 × 108 to 3.1 × 1011. Increasing starch did not change the results. Tested under simulated gastrointestinal conditions, cell viability was maintained for up to 6 h. | [226] |
| Biopolymers | Encapsulated Strain | Encapsulation Method | Benefits | References |
|---|---|---|---|---|
| CMC and gelatin | Lactobacillus rhamnosus | Emulsification | After 120 min of exposure to SGF and SIF, cell viability was maintained at approximately 77.5% (4 log CFU/mL) in capsules and 60% (5 log CFU/mL) in free cells. | [228] |
| CMC and κ-carrageenan | Lactobacillus plantarum | Extrusion | Cellular stability was greatly improved for encapsulated samples: in an acidic medium (pH 2), it decreased from 10 log CFU/g to 0 after 90 min (non-encapsulated cells) and to about 8 log CFU/g after 120 min (encapsulated cells). During storage for 30 days at 4 °C, cell stability changed from 10 log CFU/g to 2 log CFU/g (free cells) and from 10 log CFU/g to 7 log CFU/g (encapsulated cells). | [229] |
| Cellulose and pectin | lactic acid bacteria | High-pressure microfluidization | Viability of non-encapsulated cells decreased from 9.56 to 5.29 log CFU/mL in an acid medium, while encapsulation protected cells (decrease of 1.88 log CFU/mL after 2 h in the same conditions). | [230] |
| CMC and inulin | Lactobacillus plantarum | Casting | Cell viability decreased during storage whether or not probiotics were encapsulated. | [227] |
| CMC and rice bran | Lactobacillus reuteri | Emulsification | After heat exposure (85 °C, 25 s), cell viability decreased by more than 57%, although L. reuteri is a thermotolerant bacterium. However, the survival rate of encapsulated cells was approximately 6 log CFU/g. | [231] |
| CMC and chitosan | Lactobacillus rhamnosus | Extrusion | Microencapsulated strains were stable at pH 2–4; at the highest pH value tested (12.5), all microcapsules disintegrated. | [232] |
| Cellulose, alginate, starch, and lecithin | Lactobacillus rhamnosus | Extrusion | Under gastric conditions, viability of encapsulated cells was 37% higher than that of free ones. Encapsulation had a positive effect on storage, in which viability decreased by 1.23 log (25 degrees) and 1.08 log (4 degrees), unlike free cells in which stability decreased by 3.17 and 1.93. | [233] |
| Cellulose and alginate | Lactobacillus plantarum | Extrusion and lyophilization | Lyophilized encapsulated cells showed the best stability in the simulated gastrointestinal conditions: gradual release of 2.6 × 106 CFU/mL for 210 min. When refrigerated, encapsulated cells maintained viability for up to 160 days. | [155] |
| Cellulose and alginate | Lactobacillus plantarum | Extrusion-dripping | After 120 min in SGF, viability of non-encapsulated cells decreased by 66.6%, while encapsulated strains had a 58.4% better viability than that of free cells. The addition of cellulose protected the capsules from the action of pH. | [234] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gheorghita, R.; Anchidin-Norocel, L.; Filip, R.; Dimian, M.; Covasa, M. Applications of Biopolymers for Drugs and Probiotics Delivery. Polymers 2021, 13, 2729. https://doi.org/10.3390/polym13162729
Gheorghita R, Anchidin-Norocel L, Filip R, Dimian M, Covasa M. Applications of Biopolymers for Drugs and Probiotics Delivery. Polymers. 2021; 13(16):2729. https://doi.org/10.3390/polym13162729
Chicago/Turabian StyleGheorghita, Roxana, Liliana Anchidin-Norocel, Roxana Filip, Mihai Dimian, and Mihai Covasa. 2021. "Applications of Biopolymers for Drugs and Probiotics Delivery" Polymers 13, no. 16: 2729. https://doi.org/10.3390/polym13162729
APA StyleGheorghita, R., Anchidin-Norocel, L., Filip, R., Dimian, M., & Covasa, M. (2021). Applications of Biopolymers for Drugs and Probiotics Delivery. Polymers, 13(16), 2729. https://doi.org/10.3390/polym13162729









