Microscopic Structure of Swollen Hydrogels by Scanning Electron and Light Microscopies: Artifacts and Reality
Abstract
1. Introduction
2. Experimental
2.1. Chemicals
2.2. Synthesis of Hydrogels
2.3. Coding of Samples
2.4. Electron Microscopy
2.5. Wide-Field Light Microscopy
2.6. Laser Scanning Confocal Microscopy
2.7. Equilibrium Water Content
2.8. Tensile Measurements
3. Results and Discussion
3.1. Microstructure of Highly Swollen Homogeneous PGMA Hydrogel
3.2. PGMA Hydrogels Prepared in the Presence of Water as a Diluent
3.3. PHEMA Hydrogels
3.4. IPN Hydrogels
3.5. Effect of Plunge-Freezing at Cryo-SEM on the Hydrogel Microstructure
4. Conclusions
Supplementary Materials
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Hoffman, A.S. Hydrogels for biomedical applications. Adv. Drug Deliv. Rev. 2002, 54, 3–12. [Google Scholar] [CrossRef]
- Macková, H.; Plichta, Z.; Hlídková, H.; Sedláček, O.; Konefal, R.; Sadakbayeva, Z.; Dušková-Smrčková, M.; Horák, D.; Kubinová, Š. Reductively Degradable Poly(2-hydroxyethyl methacrylate) Hydrogels with Oriented Porosity for Tissue Engineering Applications. ACS Appl. Mater. Interf. 2017, 9, 10544–10553. [Google Scholar] [CrossRef] [PubMed]
- Šprincl, L.; Kopeček, J.; Lím, D. Effect of porosity of heterogeneous poly(glycol monomethacrylate) gels on the healing-in of test implants. J. Biomed. Mater. Res. 1971, 4, 447–458. [Google Scholar] [CrossRef] [PubMed]
- Kopeček, J.; Yang, J. Hydrogels as smart biomaterials. Polym. Int. 2007, 56, 1078–1098. [Google Scholar] [CrossRef]
- Savina, I.N.; Cnudde, V.; D’Hollander, S.; van Hoorebeke, L.; Mattiasson, B.; Galaev, I.Y.; Du Prez, F. Cryogels from poly(2-hydroxyethyl methacrylate): Macroporous, interconnected materials with potential as cell scaffolds. Soft Matter. 2007, 3, 1176–1184. [Google Scholar] [CrossRef]
- Allen, P.E.M.; Bennett, D.J.; Williams, D.R.G. Water in methacrylates-I. Sorption and desorption properties of poly(2-hydroxyethyl methacrylate-co-glycol dimethacrylate) networks. Eur. Polym. J. 1992, 28, 347–352. [Google Scholar] [CrossRef]
- Orakdogen, N.; Okay, O. Influence of the initiator system on the spatial inhomogeneity in acrylamide-based hydrogels. J. Appl. Polym. Sci. 2007, 103, 3228–3237. [Google Scholar] [CrossRef]
- Shibayama, M. Universality and specificity of polymer gels viewed by scattering methods. Bull. Chem. Soc. Jpn. 2006, 79, 1799–1819. [Google Scholar] [CrossRef]
- Annabi, N.; Mithieux, S.M.; Weiss, A.S.; Dehghani, F. The fabrication of elastin-based hydrogels using high pressure CO2. Biomaterials 2009, 30, 1–7. [Google Scholar] [CrossRef]
- Suchý, T.; Šupová, M.; Bartoš, M.; Sedláček, R.; Piola, M.; Soncini, M.; Fiore, G.B.; Sauerová, P.; Kalbáčová, M.H. Dry versus hydrated collagen scaffolds: Are dry states representative of hydrated states? J. Mater. Sci. Mater. Med. 2018, 29. [Google Scholar] [CrossRef]
- Dahl, R.; Staehelin, L.A. High-pressure freezing for the preservation of biological structure: Theory and practice. J. Electron. Micros. Tech. 1989, 12, 165–174. [Google Scholar] [CrossRef] [PubMed]
- Paterson, S.M.; Casadio, Y.S.; Brown, D.H.; Shaw, J.A.; Chirila, T.V.; Baker, M.V. Laser scanning confocal microscopy versus scanning electron microscopy for characterization of polymer morphology: Sample preparation drastically distorts morphologies of poly(2-hydroxyethyl methacrylate)-based hydrogels. J. Appl. Polym. Sci. 2013, 127, 4296–4304. [Google Scholar] [CrossRef]
- Trieu, H.H.; Qutubuddin, S. Polyvinyl alcohol hydrogels I. Microscopic structure by freeze-etching and critical point drying techniques. Colloid Polym. Sci. 1994, 3, 301–309. [Google Scholar] [CrossRef]
- Miller, D.R.; Peppas, N.A. Bulk characterization and scanning electron microscopy of hydrogels of P(VA-co-NVP). Biomaterials 1986, 7, 329–339. [Google Scholar] [CrossRef]
- Aston, R.; Sewell, K.; Klein, T.; Lawrie, G.; Grøndahl, L. Evaluation of the impact of freezing preparation techniques on the characterisation of alginate hydrogels by cryo-SEM. Eur. Polym. J. 2016, 82, 1–15. [Google Scholar] [CrossRef]
- Šlouf, M.; Vacková, T.; Lednický, F.; Wandrol, P. Polymer surface morphology: Characterization by electron microscopies. In Polymer Surface Characterization; Chapter 6; Sabbatini, L., Ed.; DE GRUYTER: Berlin, Germany; Boston, MA, USA, 2014; ISBN 9783110288117. [Google Scholar]
- Přádný, M.; Dušková-Smrčková, M.; Dušek, K.; Janoušková, O.; Sadakbayeva, Z.; Šlouf, M.; Michálek, M. Macroporous 2-hydroxyethyl methacrylate hydrogels of dual porosity for cell cultivation: Morphology, swelling, permeability, and mechanical behavior. J. Polym. Res. 2014, 21, 579. [Google Scholar] [CrossRef]
- Kim, J.; Yaszemski, M.J.; Lu, L. Three-Dimensional Porous Biodegradable Polymeric Scaffolds Fabricated with Biodegradable Hydrogel Porogens. Tissue Eng. Part. C Methods. 2009, 15, 583–594. [Google Scholar] [CrossRef]
- Dušek, K.; Sedláček, B. Phase separation in poly(2-hydroxyethyl methacrylate) gels in the presence of water. Eur. Polym. J. 1971, 7, 1275–1285. [Google Scholar] [CrossRef]
- Karpushkin, E.; Dušková-Smrčková, M.; Šlouf, M.; Dušek, K. Rheology and porosity control of poly(2-hydroxyethyl methacrylate) hydrogels. Polymer 2013, 54, 661–672. [Google Scholar] [CrossRef]
- Lozinsky, V.I. Cryogels on the basis of natural and synthetic polymers: Preparation, properties and application. Russ. Chem. Rev. 2002, 71, 489–511. [Google Scholar] [CrossRef]
- Hanson Shepherd, J.N.; Parker, S.T.; Shepherd, R.F.; Gillette, M.U.; Lewis, J.A.; Nuzzo, R.G. 3D microperiodic hydrogel scaffolds for robust neuronal cultures. Adv. Funct. Mater. 2011, 21, 47–54. [Google Scholar] [CrossRef] [PubMed]
- Woerly, S.; Marchand, R. Interactions of copolymeric poly( glyceryl methacrylate)-collagen hydrogels with neural tissue: Effects of structure and polar groups. Biomaterials 1991, 12, 197–203. [Google Scholar] [CrossRef]
- Dušková-Smrčková, M.; Sadakbayeva, Z.; Steinhart, M.; Dušek, K. The Manifold Varieties of Poly(2-Hydroxyethyl Methacrylate) Hydrogels−IPNs. Macromol. Symp. 2017, 372, 28–42. [Google Scholar] [CrossRef]
- Karpushkin, E.; Dušková-Smrčková, M.; Remmler, T.; Lapčíková, M.; Dušek, K. Rheological properties of homogeneous and heterogeneous poly(2-hydroxyethyl methacrylate) hydrogels. Polym. Int. 2012, 61, 328–336. [Google Scholar] [CrossRef]
- Refojo, M.F. Glyceryl methacrylate hydrogels. J. Appl. Polym. Sci. 1965, 9, 3161–3170. [Google Scholar] [CrossRef]
- Woerly, S.; Maghami, G.; Duncan, R. Poly(Glyceryl Methacrylate) Hydrogels—Effect of Composition and Crosslinking Density on Structure and Release of Dextran as a Model Macromolecule. J. Bioact. Compat. Polym. 1992, 7, 305–323. [Google Scholar] [CrossRef]
- Yu, B.; Wang, C.; Ju, Y.M.; West, L.; Harmon, J.; Moussy, Y.; Moussy, F. Use of hydrogel coating to improve the performance of implanted glucose sensors. Biosens. Bioelectron. 2008, 23, 1278–1284. [Google Scholar] [CrossRef]
- González-Méijome, J.M.; López-Alemany, A.; Almeida, J.B.; Parafita, M.A.; Refojo, M.F. Microscopic observations of superficial ultrastructure of unworn siloxane-hydrogel contact lenses by cryo-scanning electron microscopy. J. Biomed. Mater. Res. Part. B Appl. Biomater. 2006, 76, 419–423. [Google Scholar] [CrossRef]
- Kuhn, W.; Peterli, E.; Majer, H. Freezing point depression of gels produced by high polymer network. J. Polym. Sci. 1955, 16, 539–548. [Google Scholar] [CrossRef]
- Serp, D.; Mueller, M.; von Stockar, U.; Marison, I.W. Low-temperature electron microscopy for the study of polysaccharide ultrastructures in hydrogels. I. Theoretical and technical considerations, Biotechnol. Bioeng. 2002, 79, 243–252. [Google Scholar] [CrossRef]
- Shotton, D.M. A practical introduction to rapid freezing techniques. In Rapid Freezing, Freeze Fracture, and Deep Etching, 1st ed.; Severs, N.J., Shotton, D.M., Eds.; Wiley-Liss: New York, NY, USA, 1995; pp. 31–49. [Google Scholar]
- Harmon, M.E.; Schrof, W.; Frank, C.W. Fast-responsive semi-interpenetrating hydrogel networks imaged with confocal fluorescence microscopy. Polymer 2003, 44, 6927–6936. [Google Scholar] [CrossRef]
- Moor, H. Theory and Practice of High Pressure Freezing. In Cryotechniques in Biological Electron Microscopy; Steinbrecht, R.A., Zierold, K., Eds.; Springer: Berlin Heidelberg, Germany, 1987; pp. 175–191. ISBN 978-3-642-72815-0. [Google Scholar] [CrossRef]
- Apkarian, R.P.; Wright, E.R. Cryo and Cryo-Etch Methods for Quality Preservation of Hydrogels Imaged at High Magnification by Low Temperature SEM. Microsc. Microanal. 2005, 11, 1088–1089. [Google Scholar] [CrossRef][Green Version]
- Liu, Q.; Hedberg, E.L.; Liu, Z.; Bahulekar, R.; Meszlenyi, R.K.; Mikos, A.G. Preparation of macroporous poly(2-hydroxyethyl methacrylate) hydrogels by enhanced phase separation. Biomaterials 2000, 21, 2163–2169. [Google Scholar] [CrossRef]
- Baker, M.V.; Brown, D.H.; Casadio, Y.S.; Chirila, T.V. The preparation of poly(2-hydroxyethyl methacrylate) and poly{(2-hydroxyethyl methacrylate)-co-[poly(ethylene glycol) methyl ether methacrylate]} by photoinitiated polymerisation-induced phase separation in water. Polymer 2009, 50, 5918–5927. [Google Scholar] [CrossRef]
- Dušková-Smrčková, M.; Valentová, H.; Ďuračková, A.; Dušek, K. Diluent Induced Cyclization and Phase Separation in Polymer Networks. Macromol. Symp. 2011, 306, 67–76. [Google Scholar] [CrossRef]
- Sadakbayeva, Z.; Dušková-Smrčková, M.; Šturcová, A.; Pfleger, J.; Dušek, K. Microstructured poly(2-hydroxyethyl methacrylate)/poly(glycerol monomethacrylate) interpenetrating network hydrogels: UV-scattering induced accelerated formation and tensile behavior. Eur. Polym. J. 2018, 101, 304–313. [Google Scholar] [CrossRef]
- Turner, J.S.; Cheng, Y.-L. Preparation of PDMS-PMAA Interpenetrating Polymer Network Membranes Using the Monomer Immersion Method. Macromolecules 2000, 33, 3714–3718. [Google Scholar] [CrossRef]
- Yin, L.; Fei, L.; Cui, F.; Tang, C.; Yin, C. Superporous hydrogels containing poly(acrylic acid-co-acrylamide)/O-carboxymethyl chitosan interpenetrating polymer networks. Biomaterials 2007, 28, 1258–1266. [Google Scholar] [CrossRef]
- Lee, F.; Kurisawa, M. Formation and stability of interpenetrating polymer network hydrogels consisting of fibrin and hyaluronic acid for tissue engineering. Acta Biomater. 2013, 9, 5143–5152. [Google Scholar] [CrossRef]








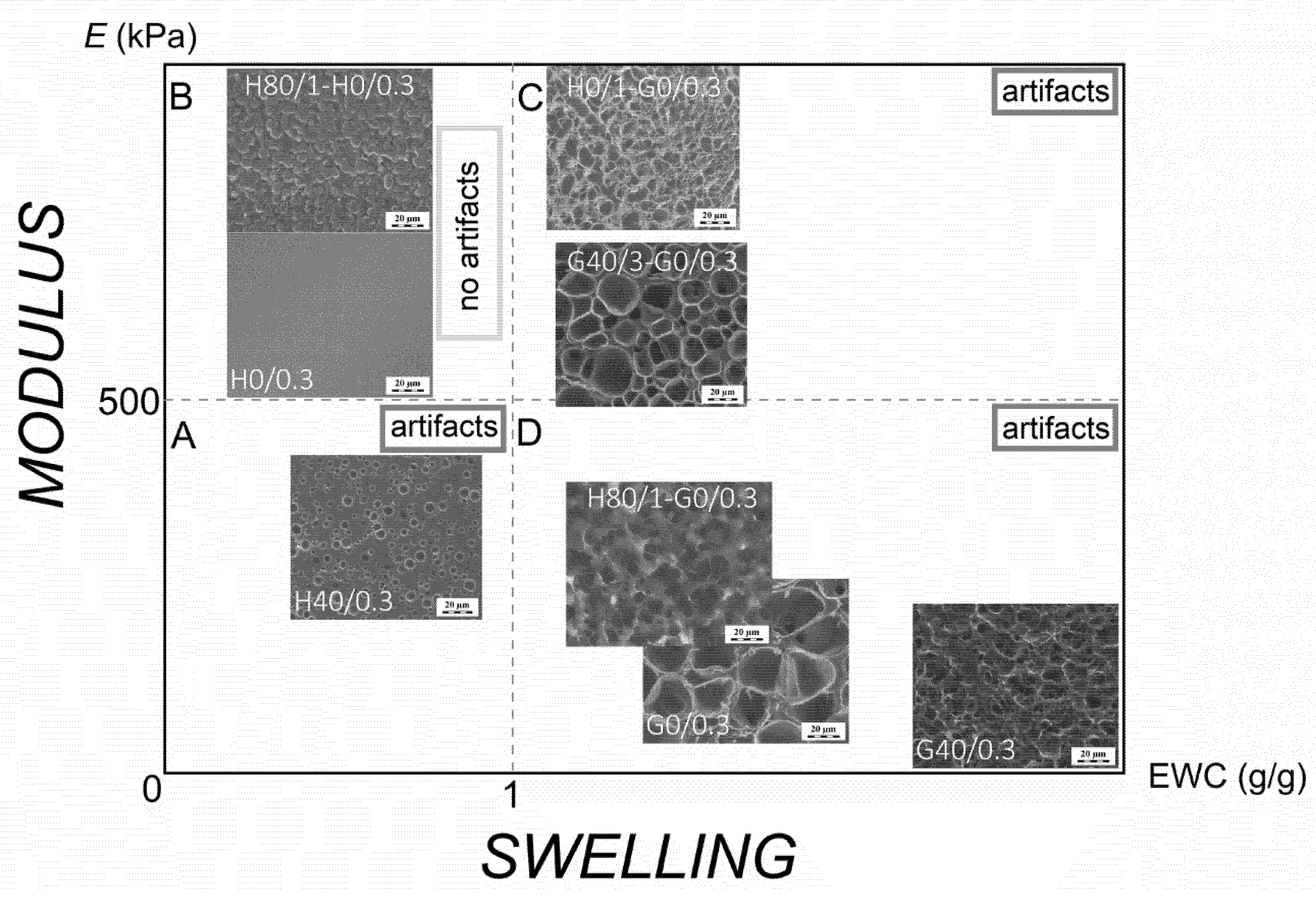
| Sample | Visual Appearance of Water-Swollen Gel | EWC (g/g) | Size of Pores in Cryo-SEM Image (μm) | E in Swollen State (kPa) |
|---|---|---|---|---|
| H0/0.3 | clear | 0.6 | no pores | 590 |
| H40/0.3 | clear | 0.7 | 0.6–10 | 430 |
| H80/1 | white | 4.4 | 2–5 | 4 |
| G0/0.3 | clear * | 2.8 | 2–40 | 250 |
| G40/0.3 | clear | 5.8 | 2–25 | 30 |
| H80/1-H0/0.3 | opaque | 0.6 | no pores | 980 |
| H80/1-G0/0.3 | opaque | 2.2 | 7–30 | 380 |
| G40/3-G0/0.3 | clear * | 1.9 | 2–25 | 540 |
| H0/1-G0/0.3 | clear * | 1.1 | 2–30 | 1010 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kaberova, Z.; Karpushkin, E.; Nevoralová, M.; Vetrík, M.; Šlouf, M.; Dušková-Smrčková, M. Microscopic Structure of Swollen Hydrogels by Scanning Electron and Light Microscopies: Artifacts and Reality. Polymers 2020, 12, 578. https://doi.org/10.3390/polym12030578
Kaberova Z, Karpushkin E, Nevoralová M, Vetrík M, Šlouf M, Dušková-Smrčková M. Microscopic Structure of Swollen Hydrogels by Scanning Electron and Light Microscopies: Artifacts and Reality. Polymers. 2020; 12(3):578. https://doi.org/10.3390/polym12030578
Chicago/Turabian StyleKaberova, Zhansaya, Evgeny Karpushkin, Martina Nevoralová, Miroslav Vetrík, Miroslav Šlouf, and Miroslava Dušková-Smrčková. 2020. "Microscopic Structure of Swollen Hydrogels by Scanning Electron and Light Microscopies: Artifacts and Reality" Polymers 12, no. 3: 578. https://doi.org/10.3390/polym12030578
APA StyleKaberova, Z., Karpushkin, E., Nevoralová, M., Vetrík, M., Šlouf, M., & Dušková-Smrčková, M. (2020). Microscopic Structure of Swollen Hydrogels by Scanning Electron and Light Microscopies: Artifacts and Reality. Polymers, 12(3), 578. https://doi.org/10.3390/polym12030578







