Carbon-Based Polymer Nanocomposite for High-Performance Energy Storage Applications
Abstract
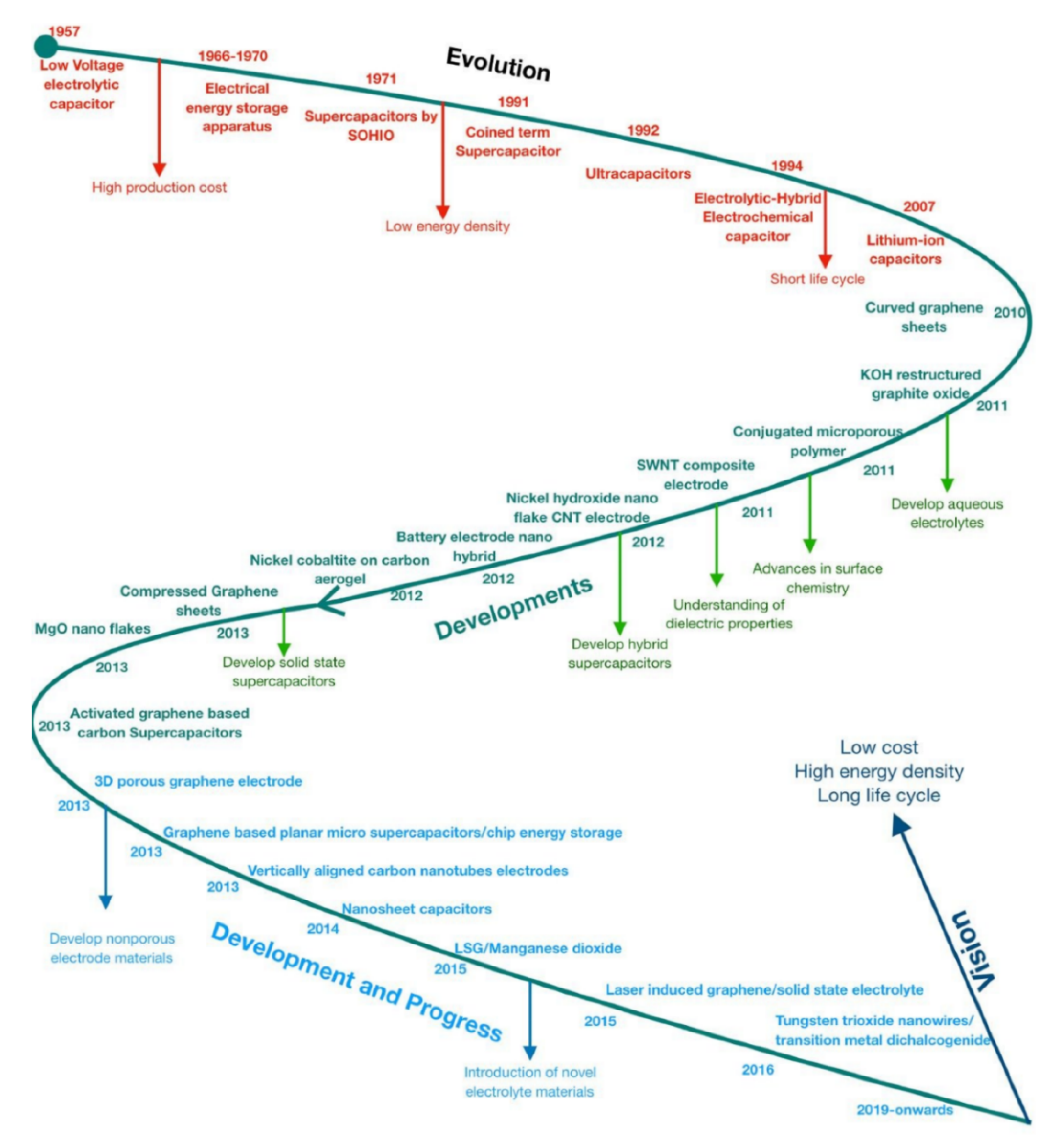
1. Introduction
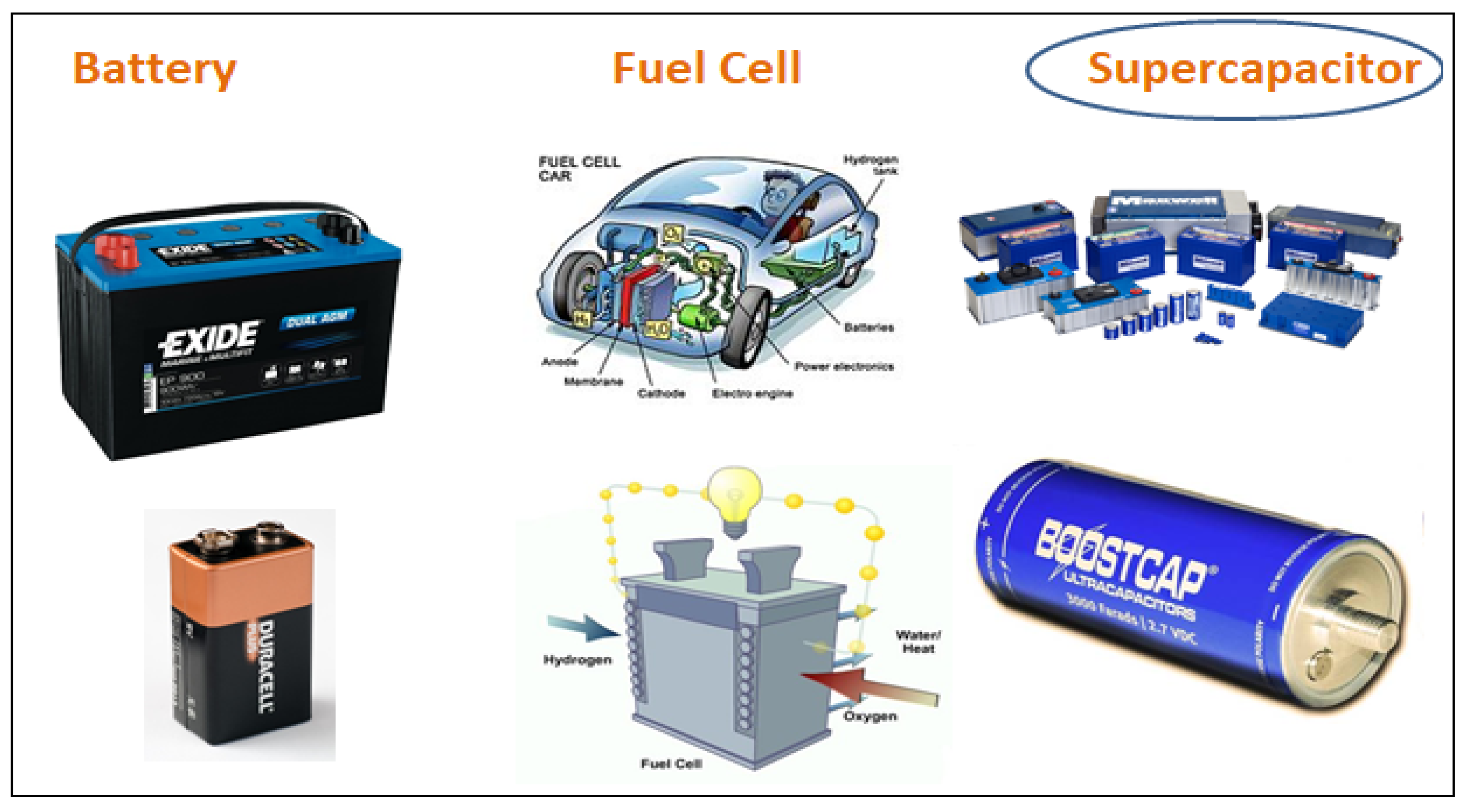
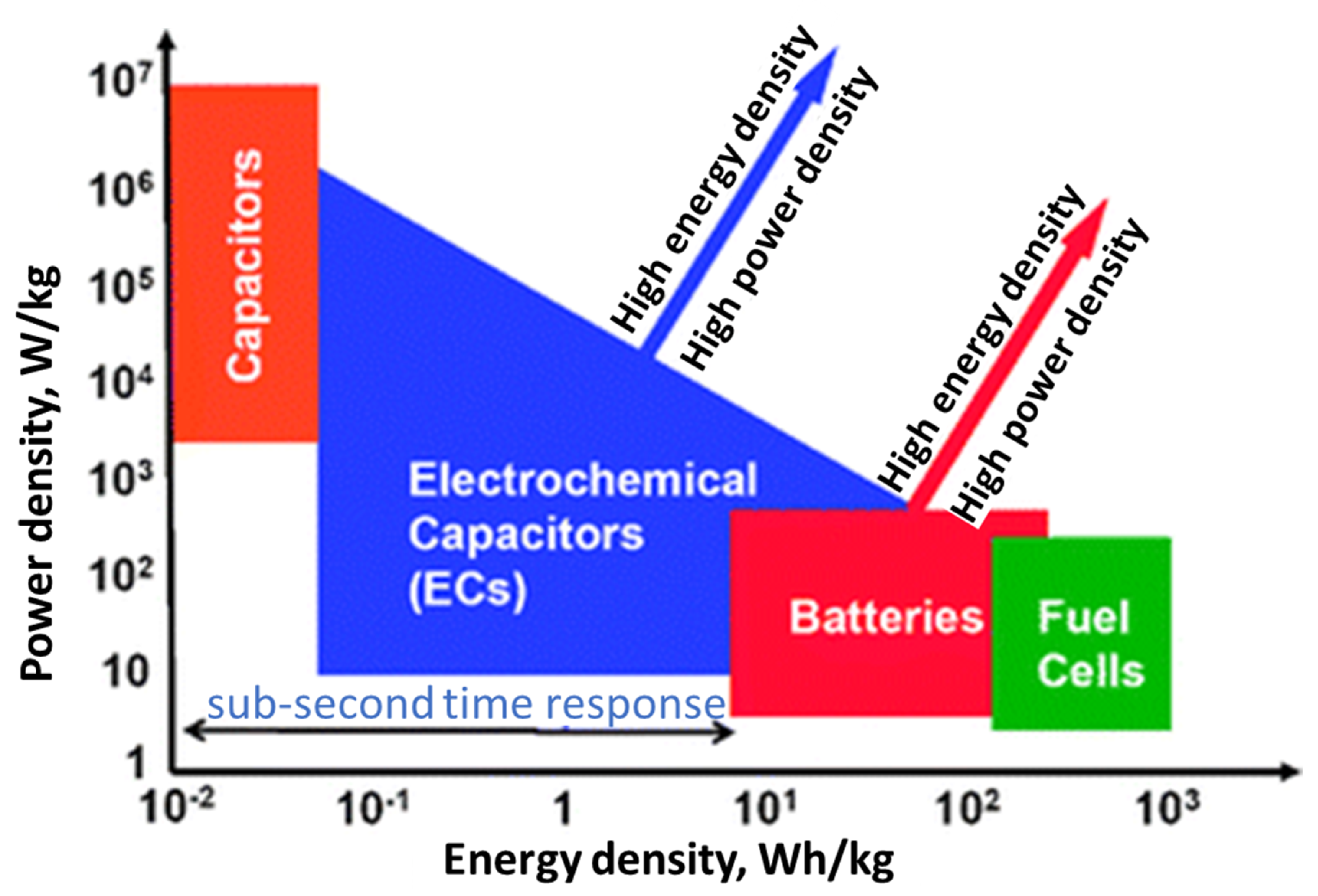
2. Electrochemical Energy Storage Applications
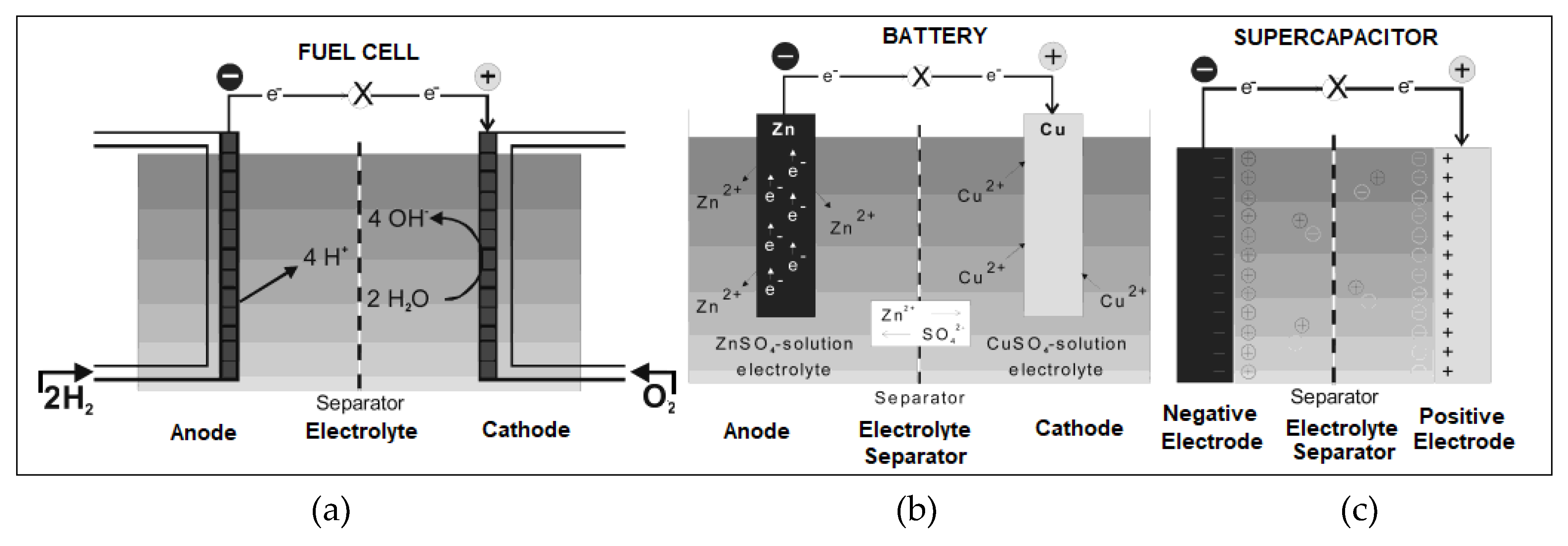
2.1. Fuel Cells
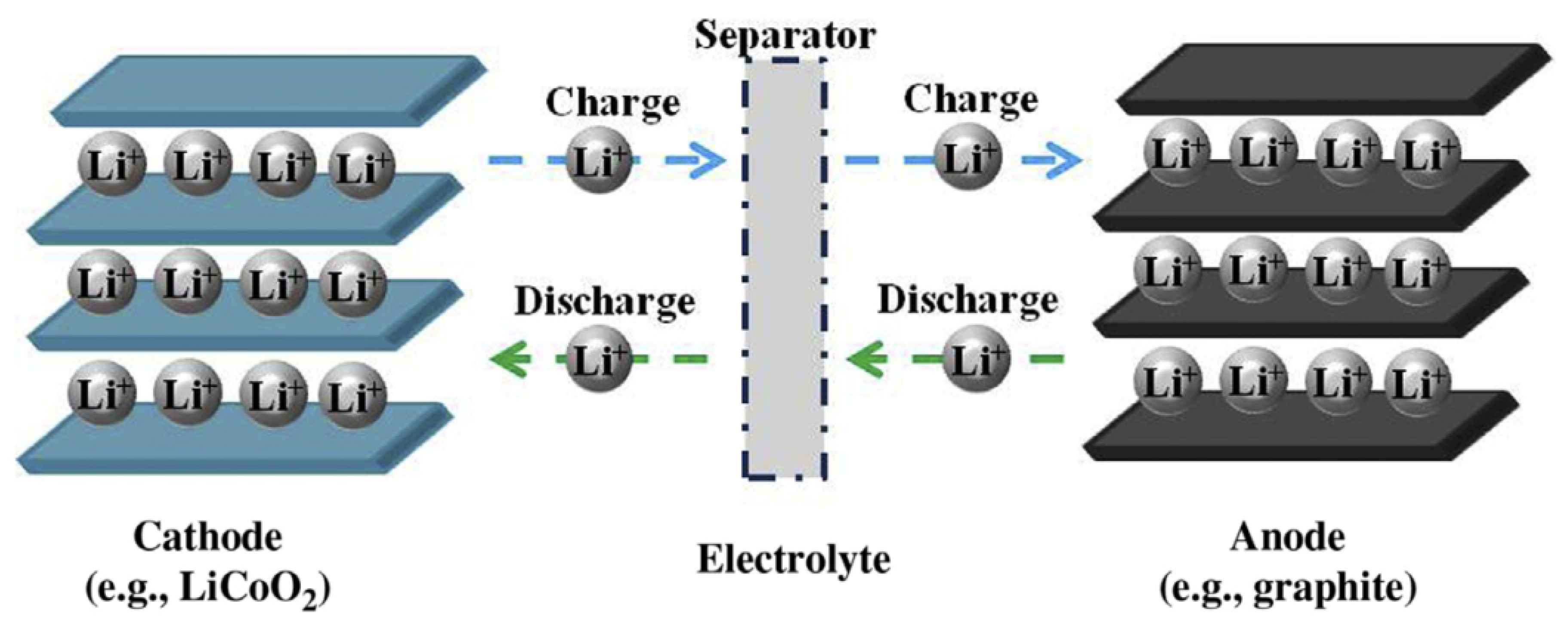
2.2. Battery
2.3. Supercapacitor
3. Carbon-Based Polymer Nanocomposites
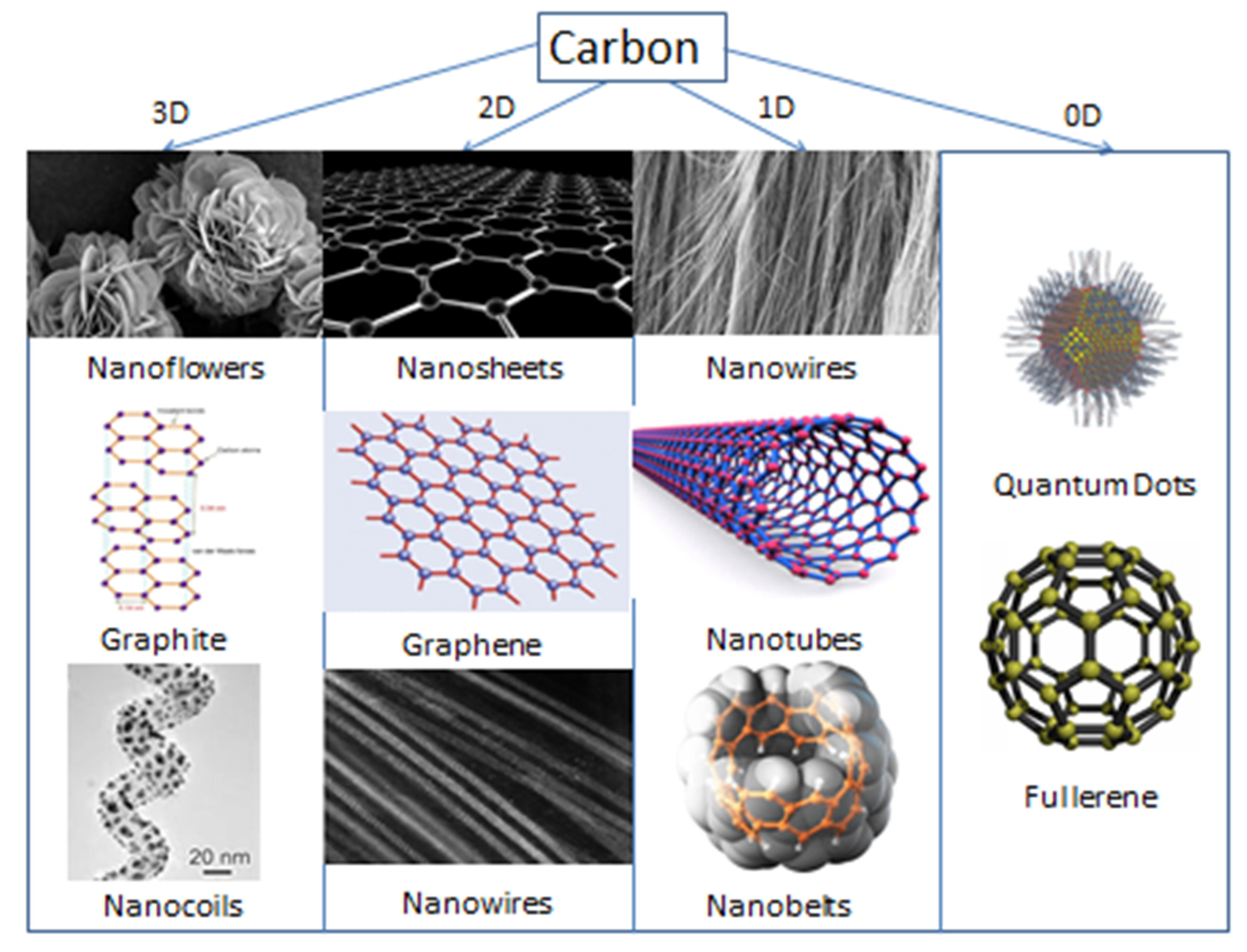
3.1. Carbon Materials
3.1.1. Carbon Nanotubes
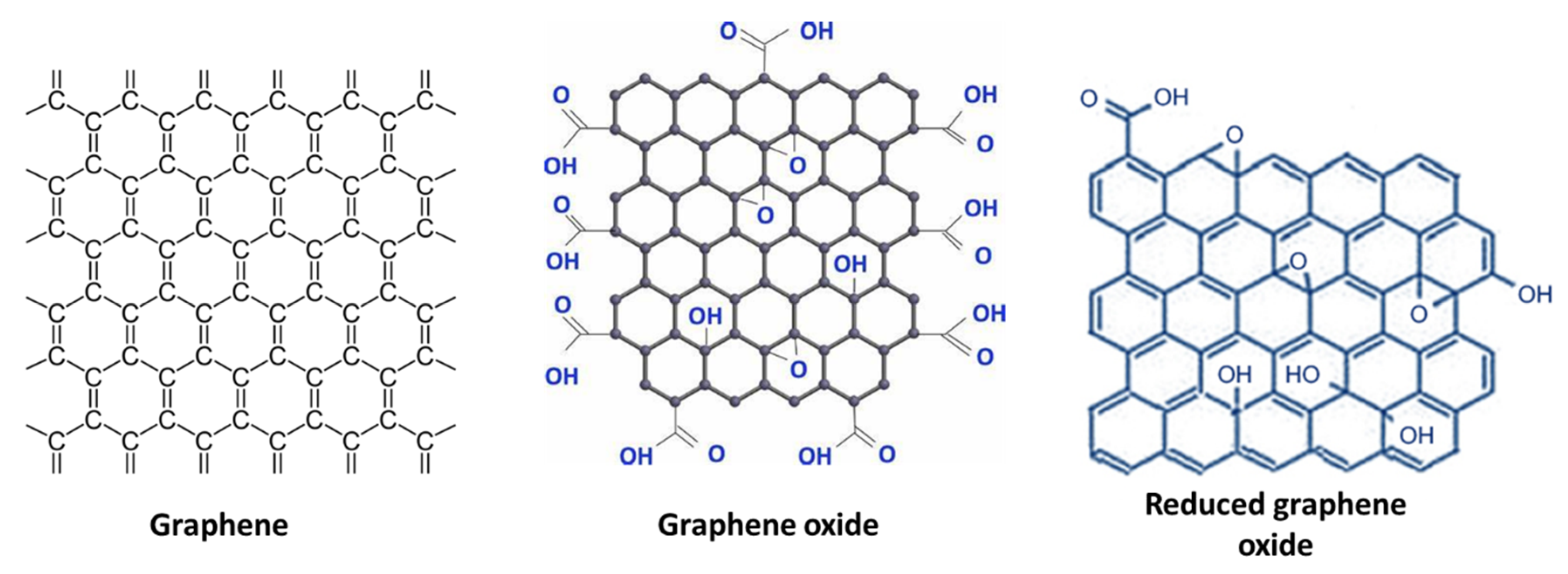
3.1.2. Graphene, Graphene Oxide, and Reduced Graphene Oxide
3.1.3. Other Carbon Materials
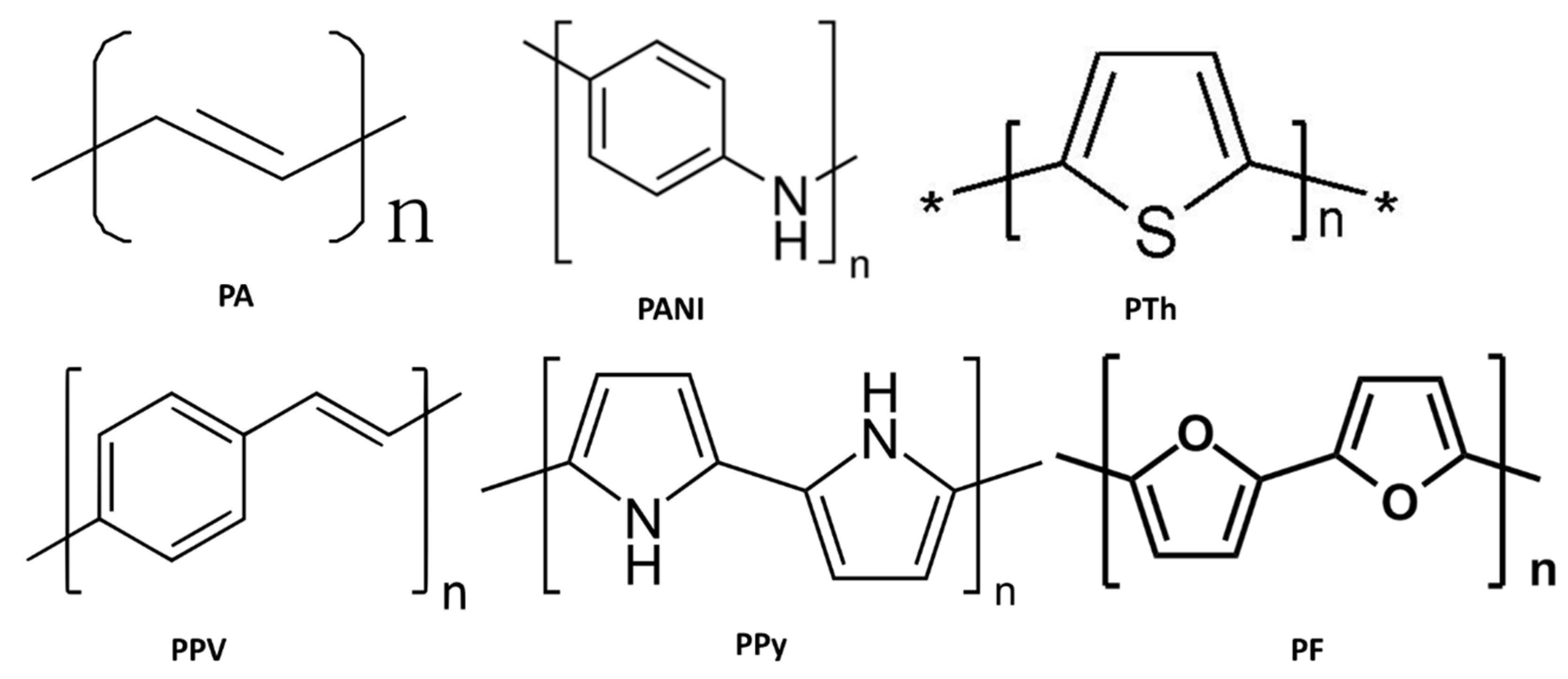
3.2. Polymer Materials
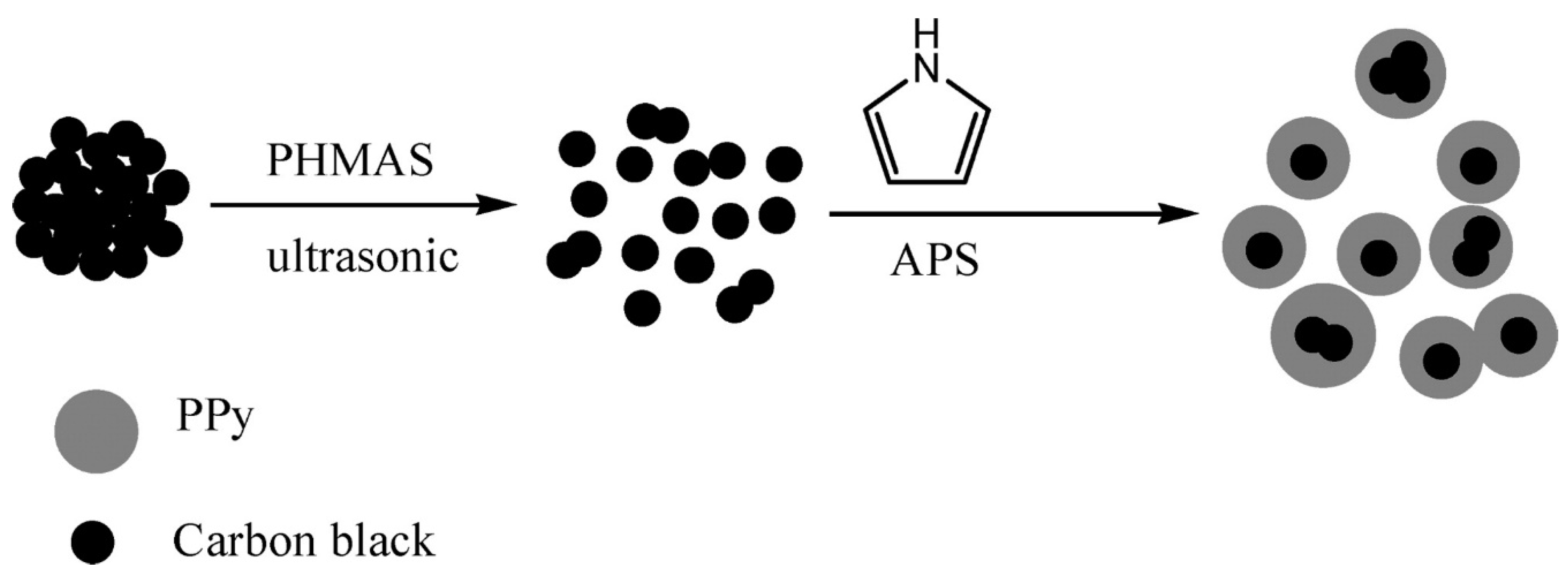
3.3. Carbon Polymer Nanocomposites
4. Applications of Carbon-based Polymer Nanocomposites
4.1. Applications of Carbon-Based Polymer Nanocomposites (CPNCs) in Fuel Cells
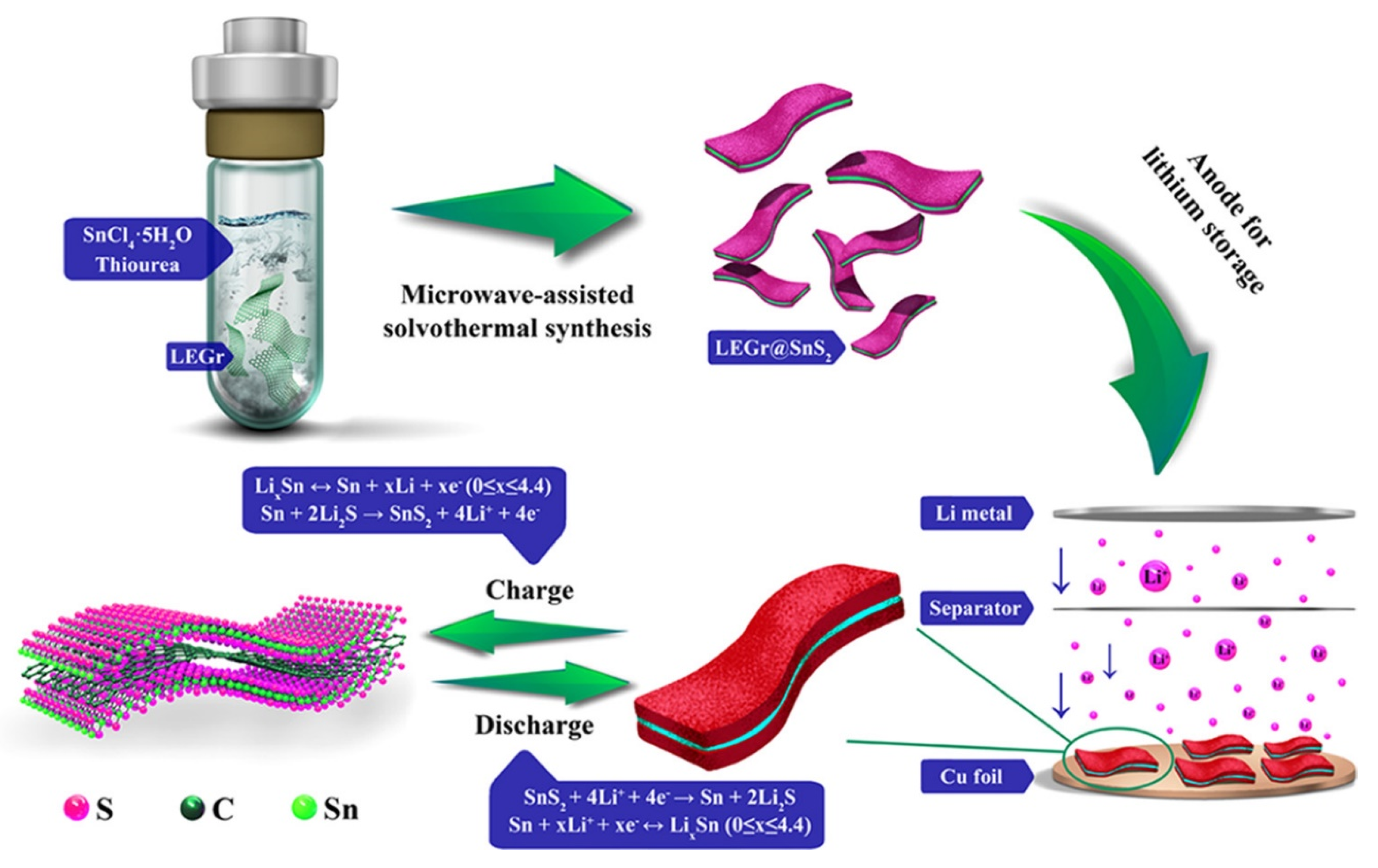
4.2. Applications of CPNCs for Li-Ion Battery
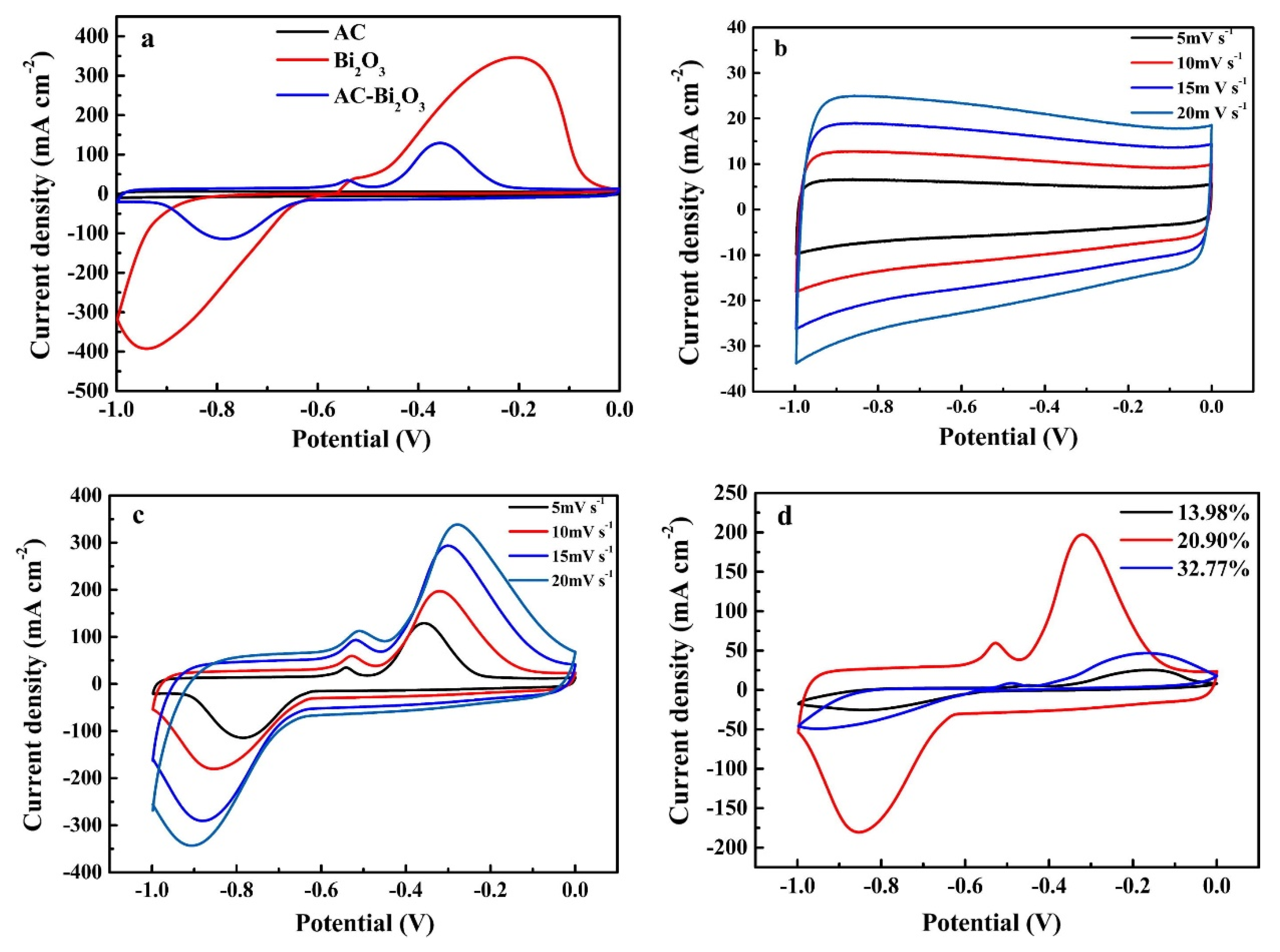
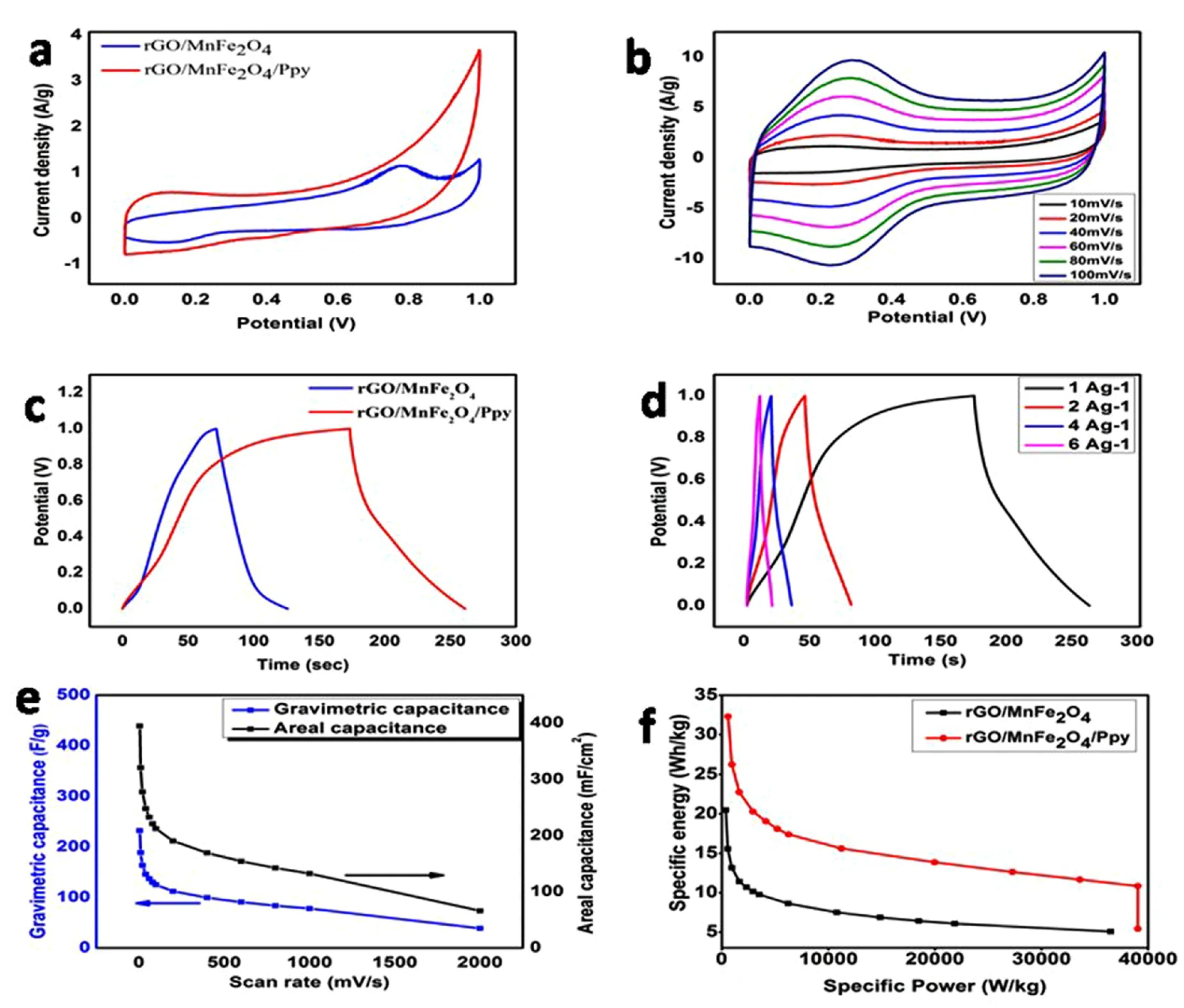
4.3. Applications of CPNCs for Supercapacitors
5. Conclusions and Future Prospects
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Davis, S.E.; Ide, M.S.; Davis, R.J.; Bowman, S. Selective oxidation of alcohols and aldehydes over supported metal nanoparticles. Green Chem. 2013, 15, 17–45. [Google Scholar] [CrossRef]
- Siwal, S.; Devi, N.; Perla, V.K.; Ghosh, S.K.; Mallick, K. Promotional role of gold in electrochemical methanol oxidation. Catal. Struct. React. 2019, 5, 1–9. [Google Scholar] [CrossRef]
- Siwal, S.; Devi, N.; Perla, V.; Barik, R.; Ghosh, S.; Mallick, K. The influencing role of oxophilicity and surface area of the catalyst for electrochemical methanol oxidation reaction: A case study. Mater. Res. Innov. 2018, 23, 440–447. [Google Scholar] [CrossRef]
- Samarjeet, S.; Sarit, G.; Debkumar, N.; Nishu, D.; Venkata, K.P.; Rasmita, B.; Malick, K. Synergistic effect of graphene oxide on the methanol oxidation for fuel cell application. Mater. Res. Exp. 2017, 4, 095306. [Google Scholar] [CrossRef]
- Siwal, S.; Matseke, S.; Mpelane, S.; Hooda, N.; Nandi, D.; Mallick, K. Palladium-polymer nanocomposite: An anode catalyst for the electrochemical oxidation of methanol. Int. J. Hydrogen Energy 2017, 42, 23599–23605. [Google Scholar] [CrossRef]
- Siwal, S.S.; Choudhary, M.; Mpelane, S.; Brink, R.; Mallick, K. Single step synthesis of a polymer supported palladium composite: A potential anode catalyst for the application of methanol oxidation. RSC Adv. 2016, 6, 47212–47219. [Google Scholar] [CrossRef]
- Lei, H.; Li, X.; Sun, C.; Zeng, J.; Siwal, S.S.; Zhang, Q. Galvanic Replacement–Mediated Synthesis of Ni-Supported Pd Nanoparticles with Strong Metal–Support Interaction for Methanol Electro-oxidation. Small 2019, 15, 1804722. [Google Scholar] [CrossRef]
- Siwal, S.S.; Thakur, S.; Zhang, Q.; Thakur, V.K. Electrocatalysts for electrooxidation of direct alcohol fuel cell: Chemistry and applications. Mater. Today Chem. 2019, 14, 100182. [Google Scholar] [CrossRef]
- Choudhary, M.; Siwal, S.; Nandi, D.; Mallick, K. Charge storage ability of the gold nanoparticles: Towards the performance of a supercapacitor. Appl. Surf. Sci. 2017, 424, 151–156. [Google Scholar] [CrossRef]
- Li, Y.; Ye, D. Carbon-Based Polymer Nanocomposite for Lithium-Ion Batteries. In Carbon-Based Polymer Nanocomposites for Environmental and Energy Applications; Elsevier: Amsterdam, The Netherlands, 2018; pp. 537–557. [Google Scholar]
- Abbasi, H.; Antunes, M.; Velasco, J.I. Recent advances in carbon-based polymer nanocomposites for electromagnetic interference shielding. Prog. Mater. Sci. 2019, 103, 319–373. [Google Scholar] [CrossRef]
- Mohan, V.B.; Lau, K.T.; Hui, D.; Bhattacharyya, D. Graphene-based materials and their composites: A review on production, applications and product limitations. Compos. Part B Eng. 2018, 142, 200–220. [Google Scholar] [CrossRef]
- Dang, Z.-M.; Yuan, J.; Yao, S.-H.; Liao, R.-J. Flexible Nanodielectric Materials with High Permittivity for Power Energy Storage. Adv. Mater. 2013, 25, 6334–6365. [Google Scholar] [CrossRef] [PubMed]
- Devi, N.; Ghosh, S.K.; Perla, V.K.; Pal, T.; Mallick, K. Laboratory based synthesis of the pure form of gananite (BiF3) nanoparticles: A potential material for electrochemical supercapacitor application. New J. Chem. 2019, 43, 18369–18376. [Google Scholar] [CrossRef]
- Al-Saleh, M. Electrically conductive carbon nanotube/polypropylene nanocomposite with improved mechanical properties. Mater. Des. 2015, 85, 76–81. [Google Scholar] [CrossRef]
- Tang, H.; Chen, G.-X.; Li, Q. Epoxy-based high-k composites with low dielectric loss caused by reactive core-shell-structured carbon nanotube hybrids. Mater. Lett. 2016, 184, 143–147. [Google Scholar] [CrossRef]
- Romasanta, J.L.; Santana, M.H.; López-Manchado, M.A.; Verdejo, R. Functionalised graphene sheets as effective high dielectric constant fillers. Nanoscale Res. Lett. 2011, 6, 508. [Google Scholar] [CrossRef]
- Wang, F.; Wang, H.; Mao, J. Aligned-graphene composites: A review. J. Mater. Sci. 2018, 54, 36–61. [Google Scholar] [CrossRef]
- Baek, J.E.; Kim, J.Y.; Jin, H.M.; Kim, B.H.; Lee, K.E.; Kim, S.O. Single-step self-assembly of multilayer graphene based dielectric nanostructures. FlatChem 2017, 4, 61–67. [Google Scholar] [CrossRef]
- Lin, B.; Li, Z.-T.; Yang, Y.; Li, Y.; Lin, J.-C.; Zheng, X.-M.; He, F.-A.; Lam, K.H. Enhanced dielectric permittivity in surface-modified graphene/PVDF composites prepared by an electrospinning-hot pressing method. Compos. Sci. Technol. 2019, 172, 58–65. [Google Scholar] [CrossRef]
- Mohanapriya, M.K.; Deshmukh, K.; Chidambaram, K.; Ahamed, M.B.; Sadasivuni, K.K.; Ponnamma, D.; AlMaadeed, M.A.-A.; Deshmukh, R.R.; Pasha, S.K.K. Polyvinyl alcohol (PVA)/polystyrene sulfonic acid (PSSA)/carbon black nanocomposite for flexible energy storage device applications. J. Mater. Sci. Mater. Electron. 2017, 28, 6099–6111. [Google Scholar] [CrossRef]
- Fan, Z.; Wang, D.; Yuan, Y.; Wang, Y.; Cheng, Z.; Liu, Y.; Xie, Z. A lightweight and conductive MXene/graphene hybrid foam for superior electromagnetic interference shielding. Chem. Eng. J. 2020, 381, 122696. [Google Scholar] [CrossRef]
- Al-Saleh, M. Carbon-based polymer nanocomposites as dielectric energy storage materials. Nanotechnology 2018, 30, 062001. [Google Scholar] [CrossRef] [PubMed]
- Cai, C.; Liu, L.; Fu, Y. Processable conductive and mechanically reinforced polylactide/graphene bionanocomposites through interfacial compatibilizer. Polym. Compos. 2017, 40, 389–400. [Google Scholar] [CrossRef]
- Beaudin, M.; Zareipour, H.; Schellenberglabe, A.; Rosehart, W. Energy storage for mitigating the variability of renewable electricity sources: An updated review. Energy Sustain. Dev. 2010, 14, 302–314. [Google Scholar] [CrossRef]
- Arani, A.A.K.; Karami, H.; Soleymani, S.; Hejazi, M. Review of Flywheel Energy Storage Systems structures and applications in power systems and microgrids. Renew. Sustain. Energy Rev. 2017, 69, 9–18. [Google Scholar] [CrossRef]
- Elliman, R.; Gould, C.; Al-Tai, M. Review of current and future electrical energy storage devices. In Proceedings of the 2015 50th International Universities Power Engineering Conference (UPEC), Stoke on Trent, UK, 1–4 September 2015; pp. 1–5. [Google Scholar]
- Hannan, M.; Hoque, M.; Mohamed, A.; Ayob, A. Review of energy storage systems for electric vehicle applications: Issues and challenges. Renew. Sustain. Energy Rev. 2017, 69, 771–789. [Google Scholar] [CrossRef]
- Hossain, S.; Hoque, M. Polymer nanocomposite materials in energy storage: Properties and applications. In Polymer-based Nanocomposites for Energy and Environmental Applications; Elsevier: Amsterdam, The Netherlands, 2018; pp. 239–282. [Google Scholar]
- Winter, M.; Brodd, R.J. What Are Batteries, Fuel Cells, and Supercapacitors? Chem. Rev. 2004, 104, 4245–4270. [Google Scholar] [CrossRef]
- Rolison, D.R.; Long, J.W.; Lytle, J.C.; Fischer, A.E.; Rhodes, C.P.; McEvoy, T.M.; Bourg, M.E.; Lubers, A.M. Multifunctional 3D nanoarchitectures for energy storage and conversion. Chem. Soc. Rev. 2009, 38, 226–252. [Google Scholar] [CrossRef]
- Zuo, W.; Li, R.; Zhou, C.; Li, Y.; Xia, J.; Liu, X. Battery-Supercapacitor Hybrid Devices: Recent Progress and Future Prospects. Adv. Sci. 2017, 4, 1600539. [Google Scholar] [CrossRef]
- Xiu, Y.; Cheng, L.; Chunyan, L. Research on Hybrid Energy Storage System of Super-capacitor and Battery Optimal Allocation. J. Int. Counc. Electr. Eng. 2014, 4, 341–347. [Google Scholar] [CrossRef][Green Version]
- Manzetti, S.; Mariasiu, F. Electric vehicle battery technologies: From present state to future systems. Renew. Sustain. Energy Rev. 2015, 51, 1004–1012. [Google Scholar] [CrossRef]
- Hoffert, M.I.; Caldeira, K.; Benford, G.; Criswell, D.R.; Green, C.; Herzog, H.; Jain, A.K.; Kheshgi, H.S.; Lackner, K.S.; Lewis, J.S.; et al. Advanced Technology Paths to Global Climate Stability: Energy for a Greenhouse Planet. Science 2002, 298, 981–987. [Google Scholar] [CrossRef] [PubMed]
- Chauhan, A.; Saini, R. A review on Integrated Renewable Energy System based power generation for stand-alone applications: Configurations, storage options, sizing methodologies and control. Renew. Sustain. Energy Rev. 2014, 38, 99–120. [Google Scholar] [CrossRef]
- Hossain, A.; Bandyopadhyay, P.; Guin, P.S.; Roy, S. Recent developed different structural nanomaterials and their performance for supercapacitor application. Appl. Mater. Today 2017, 9, 300–313. [Google Scholar] [CrossRef]
- Jagadale, A.; Zhou, X.; Xiong, R.; Dubal, D.; Xu, J.; Yang, S. Lithium ion capacitors (LICs): Development of the materials. Energy Storage Mater. 2019, 19, 314–329. [Google Scholar] [CrossRef]
- Lin, Z.; Goikolea, E.; Balducci, A.; Naoi, K.; Taberna, P.; Salanne, M.; Yushin, G.; Simon, P. Materials for supercapacitors: When Li-ion battery power is not enough. Mater. Today 2018, 21, 419–436. [Google Scholar] [CrossRef]
- Wang, Y.; Fu, X.; Zheng, M.; Zhong, W.-H.; Cao, G. Strategies for Building Robust Traffic Networks in Advanced Energy Storage Devices: A Focus on Composite Electrodes. Adv. Mater. 2018, 31, 1700322. [Google Scholar] [CrossRef]
- Miller, E.E.; Hua, Y.; Tezel, F.H. Materials for energy storage: Review of electrode materials and methods of increasing capacitance for supercapacitors. J. Energy Storage 2018, 20, 30–40. [Google Scholar] [CrossRef]
- Muzaffar, A.; Ahamed, M.B.; Deshmukh, R.; Thirumalai, J. A review on recent advances in hybrid supercapacitors: Design, fabrication and applications. Renew. Sustain. Energy Rev. 2019, 101, 123–145. [Google Scholar] [CrossRef]
- Farzana, R.; Rajarao, R.; Bhat, B.R.; Sahajwalla, V. Performance of an activated carbon supercapacitor electrode synthesised from waste Compact Discs (CDs). J. Ind. Eng. Chem. 2018, 65, 387–396. [Google Scholar] [CrossRef]
- Tian, J.; Wu, S.; Yin, X.; Wu, W. Novel preparation of hydrophilic graphene/graphene oxide nanosheets for supercapacitor electrode. Appl. Surf. Sci. 2019, 496. [Google Scholar] [CrossRef]
- Zhou, Y.; Zhou, X.; Ge, C.; Zhou, W.; Zhu, Y.; Xu, B. Branched carbon nanotube/carbon nanofiber composite for supercapacitor electrodes. Mater. Lett. 2019, 246, 174–177. [Google Scholar] [CrossRef]
- Genç, R.; Alaş, M.Ö.; Harputlu, E.; Repp, S.; Kremer, N.; Castellano, M.; Colak, S.G.; Ocakoglu, K.; Erdem, E. High-Capacitance Hybrid Supercapacitor Based on Multi-Colored Fluorescent Carbon-Dots. Sci. Rep. 2017, 7, 11222. [Google Scholar] [CrossRef]
- Saliger, R.; Fischer, U.; Herta, C.; Fricke, J. High surface area carbon aerogels for supercapacitors. J. Non-Cryst. Solids 1998, 225, 81–85. [Google Scholar] [CrossRef]
- Al Sheheri, S.Z.; Al-Amshany, Z.M.; Al Sulami, Q.A.; Tashkandi, N.Y.; Hussein, M.; El-Shishtawy, R.M. The preparation of carbon nanofillers and their role on the performance of variable polymer nanocomposites. Des. Monomers Polym. 2019, 22, 8–53. [Google Scholar] [CrossRef]
- Chen, X.; Paul, R.; Dai, L. Carbon-based supercapacitors for efficient energy storage. Natl. Sci. Rev. 2017, 4, 453–489. [Google Scholar] [CrossRef]
- Chen, T.; Dai, L. Carbon nanomaterials for high-performance supercapacitors. Mater. Today 2013, 16, 272–280. [Google Scholar] [CrossRef]
- Yang, C.; Liu, P.; Wang, T. Well-Defined Core−Shell Carbon Black/Polypyrrole Nanocomposites for Electrochemical Energy Storage. ACS Appl. Mater. Interfaces 2011, 3, 1109–1114. [Google Scholar] [CrossRef]
- Hahm, M.G.; Reddy, A.L.M.; Cole, D.P.; Rivera, M.; Vento, J.A.; Nam, J.; Jung, H.Y.; Kim, Y.L.; Narayanan, N.T.; Hashim, D.P.; et al. Carbon Nanotube–Nanocup Hybrid Structures for High Power Supercapacitor Applications. Nano Lett. 2012, 12, 5616–5621. [Google Scholar] [CrossRef]
- El-Kady, M.F.; Shao, Y.; Kaner, R.B. Graphene for batteries, supercapacitors and beyond. Nat. Rev. Mater. 2016, 1, 16033. [Google Scholar] [CrossRef]
- Ping, Y.; Gong, Y.; Fu, Q.; Pan, C. Preparation of three-dimensional graphene foam for high performance supercapacitors. Prog. Nat. Sci. 2017, 27, 177–181. [Google Scholar] [CrossRef]
- Yanilmaz, M.; Dirican, M.; Asiri, A.M.; Zhang, X. Flexible polyaniline-carbon nanofiber supercapacitor electrodes. J. Energy Storage 2019, 24, 100766. [Google Scholar] [CrossRef]
- Qu, D.; Wang, L.; Zheng, D.; Xiao, L.; Deng, B.; Qu, D. An asymmetric supercapacitor with highly dispersed nano-Bi2O3 and active carbon electrodes. J. Power Sources 2014, 269, 129–135. [Google Scholar] [CrossRef]
- Godse, L.; Karandikar, P.; Khaladkar, M. Study of Carbon Materials and Effect of its Ball Milling, on Capacitance of Supercapacitor. Energy Procedia 2014, 54, 302–309. [Google Scholar] [CrossRef]
- Crane, M.J.; Lim, M.B.; Zhou, X.; Pauzauskie, P.J. Rapid synthesis of transition metal dichalcogenide-carbon aerogel composites for supercapacitor electrodes. Microsyst. Nanoeng. 2017, 3. [Google Scholar] [CrossRef] [PubMed]
- He, S.; Hu, Y.; Wan, J.; Gao, Q.; Wang, Y.; Xie, S.; Qiu, L.; Wang, C.; Zheng, G.; Wang, B.; et al. Biocompatible carbon nanotube fibers for implantable supercapacitors. Carbon 2017, 122, 162–167. [Google Scholar] [CrossRef]
- Ke, Q.; Wang, J. Graphene-based materials for supercapacitor electrodes—A review. J. Mater. 2016, 2, 37–54. [Google Scholar] [CrossRef]
- Bose, S.; Kuila, T.; Mishra, A.K.; Rajasekar, R.; Kim, N.H.; Sharma, K. Carbon-based nanostructured materials and their composites as supercapacitor electrodes. J. Mater. Chem. 2012, 22, 767–784. [Google Scholar] [CrossRef]
- Wang, S.; Jin, C.; Qian, W. Bi2O3 with activated carbon composite as a supercapacitor electrode. J. Alloy. Compd. 2014, 615, 12–17. [Google Scholar] [CrossRef]
- Wang, G.; Zhang, L.; Zhang, J. A review of electrode materials for electrochemical supercapacitors. Chem. Soc. Rev. 2012, 41, 797–828. [Google Scholar] [CrossRef]
- Shao, Y.; El-Kady, M.F.; Wang, L.J.; Zhang, Q.; Li, Y.; Wang, H.; Mousavi, M.F.; Kaner, R.B. Graphene-based materials for flexible supercapacitors. Chem. Soc. Rev. 2015, 44, 3639–3665. [Google Scholar] [CrossRef] [PubMed]
- Nithya, V.; Hanitha, B.; Surendran, S.; Kalpana, D.; Selvan, R.K. Effect of pH on the sonochemical synthesis of BiPO4 nanostructures and its electrochemical properties for pseudocapacitors. Ultrason. Sonochem. 2015, 22, 300–310. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Kumar, S. Polymer/Carbon Nanotube Nano Composite Fibers—A Review. ACS Appl. Mater. Interfaces 2014, 6, 6069–6087. [Google Scholar] [CrossRef] [PubMed]
- Han, J.T.; Cho, J.Y.; Kim, J.H.; Jang, J.I.; Kim, J.S.; Lee, H.J.; Park, J.H.; Chae, J.S.; Roh, H.-K.; Lee, W.; et al. Structural Recovery of Highly Oxidized Single-Walled Carbon Nanotubes Fabricated by Kneading and Electrochemical Applications. Chem. Mater. 2019, 31, 3468–3475. [Google Scholar] [CrossRef]
- Park, J.W.; Park, S.J.; Kwon, O.S.; Lee, C.; Jang, J.; Jang, J.; Park, J.W.; Park, S.J.; Lee, C.; Kwon, O.S. In Situ Synthesis of Graphene/Polyselenophene Nanohybrid Materials as Highly Flexible Energy Storage Electrodes. Chem. Mater. 2014, 26, 2354–2360. [Google Scholar] [CrossRef]
- Krishnamoorthy, M.; Jha, N. Oxygen-Rich Hierarchical Porous Graphene as an Excellent Electrode for Supercapacitors, Aqueous Al-Ion Battery, and Capacitive Deionization. ACS Sustain. Chem. Eng. 2019, 7, 8475–8489. [Google Scholar] [CrossRef]
- Guan, L.; Pan, L.; Peng, T.; Gao, C.; Zhao, W.; Yang, Z.; Hu, H.; Wu, M. Synthesis of Biomass-Derived Nitrogen-Doped Porous Carbon Nanosheests for High-Performance Supercapacitors. ACS Sustain. Chem. Eng. 2019, 7, 8405–8412. [Google Scholar] [CrossRef]
- Manuel, J.; Zhao, X.; Cho, K.-K.; Kim, J.-K.; Ahn, J.-H. Ultralong Life Organic Sodium Ion Batteries Using a Polyimide/Multiwalled Carbon Nanotubes Nanocomposite and Gel Polymer Electrolyte. ACS Sustain. Chem. Eng. 2018, 6, 8159–8166. [Google Scholar] [CrossRef]
- Azman, N.H.N.; Nazir, S.M.M.; Ngee, L.H.; Sulaiman, Y. Graphene-based ternary composites for supercapacitors. Int. J. Energy Res. 2018, 42, 2104–2116. [Google Scholar] [CrossRef]
- Park, J.; Cho, Y.S.; Sung, S.J.; Byeon, M.; Yang, S.J.; Park, C.R. Characteristics tuning of graphene-oxide-based-graphene to various end-uses. Energy Storage Mater. 2018, 14, 8–21. [Google Scholar] [CrossRef]
- Hou, D.; Liu, Q.; Wang, X.; Quan, Y.; Qiao, Z.; Yu, L.; Ding, S. Facile synthesis of graphene via reduction of graphene oxide by artemisinin in ethanol. J. Mater. 2018, 4, 256–265. [Google Scholar] [CrossRef]
- Zaaba, N.; Foo, K.L.; Hashim, U.; Tan, S.; Liu, W.W.; Voon, C. Synthesis of Graphene Oxide using Modified Hummers Method: Solvent Influence. Procedia Eng. 2017, 184, 469–477. [Google Scholar] [CrossRef]
- Qorbani, M.; Esfandiar, A.; Mehdipour, H.; Chaigneau, M.; Zad, A.I.; Moshfegh, A.Z. Shedding Light on Pseudocapacitive Active Edges of Single-Layer Graphene Nanoribbons as High-Capacitance Supercapacitors. ACS Appl. Energy Mater. 2019, 2, 3665–3675. [Google Scholar] [CrossRef]
- Pykal, M.; Langer, M.; Prudilova, B.B.; Banas, P.; Otyepka, M. Ion Interactions across Graphene in Electrolyte Aqueous Solutions. J. Phys. Chem. C 2019, 123, 9799–9806. [Google Scholar] [CrossRef]
- Liu, X.; Lai, C.; Xiao, Z.; Zou, S.; Liu, K.; Yin, Y.; Liang, T.; Wu, Z. Superb Electrolyte Penetration/Absorption of Three-Dimensional Porous Carbon Nanosheets for Multifunctional Supercapacitor. ACS Appl. Energy Mater. 2019, 2, 3185–3193. [Google Scholar] [CrossRef]
- Devadas, B.; Imae, T. Effect of Carbon Dots on Conducting Polymers for Energy Storage Applications. ACS Sustain. Chem. Eng. 2017, 6, 127–134. [Google Scholar] [CrossRef]
- Qian, H.; Kucernak, A.R.; Greenhalgh, E.S.; Bismarck, A.; Shaffer, M.S.P. Multifunctional Structural Supercapacitor Composites Based on Carbon Aerogel Modified High Performance Carbon Fiber Fabric. ACS Appl. Mater. Interfaces 2013, 5, 6113–6122. [Google Scholar] [CrossRef] [PubMed]
- Luo, H.; Zhou, X.; Ellingford, C.; Zhang, Y.; Chen, S.; Zhou, K.; Zhang, D.; Bowen, C.R.; Wan, C. Interface design for high energy density polymer nanocomposites. Chem. Soc. Rev. 2019, 48, 4424–4465. [Google Scholar] [CrossRef]
- Yang, L.; Huang, X.; Gogoll, A.; Strømme, M.; Sjödin, M. Conducting Redox Polymer Based Anode Materials for High Power Electrical Energy Storage. Electrochim. Acta 2016, 204, 270–275. [Google Scholar] [CrossRef]
- Eftekhari, A.; Li, L.; Yang, Y. Polyaniline supercapacitors. J. Power Sources 2017, 347, 86–107. [Google Scholar] [CrossRef]
- Xie, Y.; Wang, D.; Ji, J. Preparation and Supercapacitor Performance of Freestanding Polypyrrole/Polyaniline Coaxial Nanoarrays. Energy Technol. 2016, 4, 714–721. [Google Scholar] [CrossRef]
- Patil, B.H.; Patil, S.J.; Lokhande, C.D. Electrochemical Characterization of Chemically Synthesized Polythiophene Thin Films: Performance of Asymmetric Supercapacitor Device. Electroanalysis 2014, 26, 2023–2032. [Google Scholar] [CrossRef]
- Singh, K.; Kumar, S.; Agarwal, K.; Soni, K.; Gedela, V.R.; Ghosh, K. Three-dimensional Graphene with MoS2 Nanohybrid as Potential Energy Storage/Transfer Device. Sci. Rep. 2017, 7, 9458. [Google Scholar] [CrossRef] [PubMed]
- Yun, S.; Freitas, J.N.; Nogueira, A.F.; Wang, Y.; Ahmad, S.; Wang, Z.-S. Dye-sensitized solar cells employing polymers. Prog. Polym. Sci. 2016, 59, 1–40. [Google Scholar] [CrossRef]
- Naveen, M.H.; Gurudatt, N.G.; Shim, Y.-B. Applications of conducting polymer composites to electrochemical sensors: A review. Appl. Mater. Today 2017, 9, 419–433. [Google Scholar] [CrossRef]
- Gomez, I.; Leonet, O.; Blázquez, J.A.; Grande, H.-J.; Mecerreyes, D. Poly(anthraquinonyl sulfides): High Capacity Redox Polymers for Energy Storage. ACS Macro Lett. 2018, 7, 419–424. [Google Scholar] [CrossRef]
- Li, S.; Chen, Y.; He, X.; Mao, X.; Zhou, Y.; Xu, J.; Yang, Y. Modifying Reduced Graphene Oxide by Conducting Polymer Through a Hydrothermal Polymerization Method and its Application as Energy Storage Electrodes. Nanoscale Res. Lett. 2019, 14, 226. [Google Scholar] [CrossRef]
- Yang, C.; Guan, L.; Wei, H.; Guo, Z.; Wang, Y.; Yan, X.; Zhang, X. Polymer nanocomposites for energy storage, energy saving, and anticorrosion. J. Mater. Chem. A 2015, 3, 14929–14941. [Google Scholar] [CrossRef]
- Naresh, V.; Elias, L.; Martha, S.K. Poly(3,4-ethylenedioxythiophene) coated lead negative plates for hybrid energy storage systems. Electrochim. Acta 2019, 301, 183–191. [Google Scholar] [CrossRef]
- Wang, H.; Lin, J.; Shen, Z.X. Polyaniline (PANi) based electrode materials for energy storage and conversion. J. Sci. Adv. Mater. Devices 2016, 1, 225–255. [Google Scholar] [CrossRef]
- Dubal, D.; Caban-Huertas, Z.; Holze, R.; Gómez-Romero, P. Growth of polypyrrole nanostructures through reactive templates for energy storage applications. Electrochim. Acta 2016, 191, 346–354. [Google Scholar] [CrossRef]
- Zhao, S.; Chen, H.; Li, J.; Zhang, J. Synthesis of polythiophene/graphite composites and their enhanced electrochemical performance for aluminum ion batteries. New J. Chem. 2019, 43, 15014–15022. [Google Scholar] [CrossRef]
- Kim, K.-G.; Kim, S.Y. Increase in Interfacial Adhesion and Electrochemical Charge Storage Capacity of Polypyrrole on Au Electrodes Using Polyethyleneimine. Sci. Rep. 2019, 9, 2169. [Google Scholar] [CrossRef] [PubMed]
- Dhibar, S.; Bhattacharya, D.-P.; Ghosh, D.; Hatui, G.; Das, C. Graphene–Single-Walled Carbon Nanotubes–Poly(3-methylthiophene) Ternary Nanocomposite for Supercapacitor Electrode Materials. Ind. Eng. Chem. Res. 2014, 53, 13030–13045. [Google Scholar] [CrossRef]
- Thakur, V.K.; Gupta, R. Recent Progress on Ferroelectric Polymer-Based Nanocomposites for High Energy Density Capacitors: Synthesis, Dielectric Properties, and Future Aspects. Chem. Rev. 2016, 116, 4260–4317. [Google Scholar] [CrossRef]
- Moniruzzaman, M.; Winey, K.I. Polymer Nanocomposites Containing Carbon Nanotubes. Macromolecules 2006, 39, 5194–5205. [Google Scholar] [CrossRef]
- Zhang, Y.; Li, S.; Zhang, J.; Guo, P.; Zheng, J.; Zhao, X.S. Enhancement of Electrochemical Performance of Macroporous Carbon by Surface Coating of Polyaniline. Chem. Mater. 2010, 22, 1195–1202. [Google Scholar] [CrossRef]
- Muthulakshmi, B.; Kalpana, D.; Pitchumani, S.; Renganathan, N. Electrochemical deposition of polypyrrole for symmetric supercapacitors. J. Power Sources 2006, 158, 1533–1537. [Google Scholar] [CrossRef]
- Jurewicz, K.; Delpeux, S.; Bertagna, V.; Beguin, F.; Frackowiak, E. Supercapacitors from nanotubes/polypyrrole composites. Chem. Phys. Lett. 2001, 347, 36–40. [Google Scholar] [CrossRef]
- Zhang, Q.-W.; Zhou, X.; Yang, H.-S. Capacitance properties of composite electrodes prepared by electrochemical polymerization of pyrrole on carbon foam in aqueous solution. J. Power Sources 2004, 125, 141–147. [Google Scholar] [CrossRef]
- Kim, J.-H.; Lee, Y.-S.; Sharma, A.K.; Liu, C.G. Polypyrrole/carbon composite electrode for high-power electrochemical capacitors. Electrochim. Acta 2006, 52, 1727–1732. [Google Scholar] [CrossRef]
- Chauhan, N.P.; Solanki, M.S. Approaches and Challenges of Polyaniline–Graphene Nanocomposite for Energy Application. In Carbon-Based Polymer Nanocomposites for Environmental and Energy Applications; Elsevier: Amsterdam, The Netherlands, 2018; pp. 415–436. [Google Scholar]
- Ishaq, S.; Moussa, M.; Kanwal, F.; Ehsan, M.; Saleem, M.; Van, T.N.; Losic, D. Facile synthesis of ternary graphene nanocomposites with doped metal oxide and conductive polymers as electrode materials for high performance supercapacitors. Sci. Rep. 2019, 9, 5974. [Google Scholar] [CrossRef] [PubMed]
- Idumah, C.; Hassan, A. Emerging trends in graphene carbon based polymer nanocomposites and applications. Rev. Chem. Eng. 2016, 32, 223. [Google Scholar] [CrossRef]
- Wang, Y.; Wei, H.; Lu, Y.; Wei, S.; Wujcik, E.K.; Guo, Z. Multifunctional Carbon Nanostructures for Advanced Energy Storage Applications. Nanomaterials 2015, 5, 755–777. [Google Scholar] [CrossRef] [PubMed]
- Abouzari-Lotf, E.; Etesami, M.; Nasef, M.M. Carbon-Based Nanocomposite Proton Exchange Membranes for Fuel Cells. In Carbon-Based Polymer Nanocomposites for Environmental and Energy Applications; Elsevier: Amsterdam, The Netherlands, 2018; pp. 437–461. [Google Scholar]
- Peighambardoust, S.; Rowshanzamir, S.; Amjadi, M. Review of the proton exchange membranes for fuel cell applications. Int. J. Hydrog. Energy 2010, 35, 9349–9384. [Google Scholar] [CrossRef]
- Zhang, R.; Chen, Y.; Montazami, R. Ionic Liquid-Doped Gel Polymer Electrolyte for Flexible Lithium-Ion Polymer Batteries. Materials 2015, 8, 2735–2748. [Google Scholar] [CrossRef]
- Cai, C.; Wang, Y. Novel Nanocomposite Materials for Advanced Li-Ion Rechargeable Batteries. Materials 2009, 2, 1205–1238. [Google Scholar] [CrossRef]
- Shi, Y.; Peng, L.; Ding, Y.; Zhao, Y.; Yu, G. Nanostructured conductive polymers for advanced energy storage. Chem. Soc. Rev. 2015, 44, 6684–6696. [Google Scholar] [CrossRef]
- Nyholm, L.; Nyström, G.; Mihranyan, A.; Strømme, M. Toward Flexible Polymer and Paper-Based Energy Storage Devices. Adv. Mater. 2011, 23, 3751–3769. [Google Scholar] [CrossRef]
- Cheng, F.; Tang, W.; Li, C.; Chen, J.; Liu, H.K.; Shen, P.; Dou, S.X. Conducting Poly(aniline) Nanotubes and Nanofibers: Controlled Synthesis and Application in Lithium/Poly(aniline) Rechargeable Batteries. Chem. A Eur. J. 2006, 12, 3082–3088. [Google Scholar] [CrossRef]
- Deng, W.; Liang, X.; Wu, X.; Qian, J.; Cao, Y.; Ai, X.; Feng, J.; Yang, H. A low cost, all-organic Na-ion Battery Based on Polymeric Cathode and Anode. Sci. Rep. 2013, 3, 2671. [Google Scholar] [CrossRef] [PubMed]
- Magu, T.O.; Agobi, A.U.; Hitler, L.; Dass, P.M. A Review on Conducting Polymers-Based Composites for Energy Storage Application. J. Chem. Rev. 2019, 1, 19–34. [Google Scholar]
- Liu, Y.; Zhou, G.; Liu, K.; Cui, Y. Design of Complex Nanomaterials for Energy Storage: Past Success and Future Opportunity. Acc. Chem. Res. 2017, 50, 2895–2905. [Google Scholar] [CrossRef] [PubMed]
- Ji, L.; Lin, Z.; Alcoutlabi, M.; Zhang, X. Recent developments in nanostructured anode materials for rechargeable lithium-ion batteries. Energy Environ. Sci. 2011, 4, 2682–2699. [Google Scholar] [CrossRef]
- Sarang, K.T.; Miranda, A.; An, H.; Oh, E.-S.; Verduzco, R.; Lutkenhaus, J.L. Poly(fluorene-alt-naphthalene diimide) as n-Type Polymer Electrodes for Energy Storage. ACS Appl. Polym. Mater. 2019, 1, 1155–1164. [Google Scholar] [CrossRef]
- Li, J.; Han, S.; Zhang, C.; Wei, W.; Gu, M.; Meng, L. High-Performance and Reactivation Characteristics of High-Quality, Graphene-Supported SnS2 Heterojunctions for a Lithium-Ion Battery Anode. ACS Appl. Mater. Interfaces 2019, 11, 22314–22322. [Google Scholar] [CrossRef]
- Brousse, T.; Crosnier, O.; Bélanger, D.; Long, J.W. Capacitive and Pseudocapacitive Electrodes for Electrochemical Capacitors and Hybrid Devices. In Metal Oxides in Supercapacitors; Elsevier: Amsterdam, The Netherlands, 2017; pp. 1–24. [Google Scholar]
- Han, H.; Lee, S.W.; Moon, K.H.; Cho, S. Fabrication of Solid-State Asymmetric Supercapacitors Based on Aniline Oligomers and Graphene Electrodes with Enhanced Electrochemical Performances. ACS Omega 2019, 4, 1244–1253. [Google Scholar] [CrossRef]
- Oschatz, M.; Borchardt, L.; Hippauf, F.; Nickel, W.; Kaskel, S.; Brunner, E. Chapter Four—Interactions Between Electrolytes and Carbon-Based Materials—NMR Studies on Electrical Double-Layer Capacitors, Lithium-Ion Batteries, and Fuel Cells. In Annual Reports on NMR Spectroscopy; Webb, G.A., Ed.; Academic Press: Cambridge, MA, USA, 2016; pp. 237–318. [Google Scholar]
- Yang, Q.; Li, Z.; Zhang, R.; Zhou, L.; Shao, M.; Wei, M. Carbon modified transition metal oxides/hydroxides nanoarrays toward high-performance flexible all-solid-state supercapacitors. Nano Energy 2017, 41, 408–416. [Google Scholar] [CrossRef]
- Pan, Y.; Xu, K.; Wu, C. Recent progress in supercapacitors based on the advanced carbon electrodes. Nanotechnol. Rev. 2019, 8, 299–314. [Google Scholar] [CrossRef]
- Dubal, D.; Chodankar, N.; Kim, D.-H.; Gómez-Romero, P. Towards flexible solid-state supercapacitors for smart and wearable electronics. Chem. Soc. Rev. 2018, 47, 2065–2129. [Google Scholar] [CrossRef]
- Soni, R.; Anothumakkool, B.; Kurungot, S. 1D Alignment of PEDOT in a Buckypaper for High-Performance Solid Supercapacitors. ChemElectroChem 2016, 3, 1329–1336. [Google Scholar] [CrossRef]
- Yang, C.; Zhang, L.; Hu, N.; Yang, Z.; Su, Y.; Xu, S.; Li, M.; Yao, L.; Hong, M.; Zhang, Y. Rational design of sandwiched polyaniline nanotube/layered graphene/polyaniline nanotube papers for high-volumetric supercapacitors. Chem. Eng. J. 2017, 309, 89–97. [Google Scholar] [CrossRef]
- Xiao, F.; Yang, S.; Zhang, Z.; Liu, H.; Xiao, J.; Wan, L.; Luo, J.; Wang, S.; Liu, Y. Scalable Synthesis of Freestanding Sandwich-structured Graphene/Polyaniline/Graphene Nanocomposite Paper for Flexible All-Solid-State Supercapacitor. Sci. Rep. 2015, 5, 9359. [Google Scholar] [CrossRef] [PubMed]
- Dubey, R.; Guruviah, V. Review of carbon-based electrode materials for supercapacitor energy storage. Ionics 2019, 25, 1419–1445. [Google Scholar] [CrossRef]
- Bashid, H.A.; Lim, H.N.; Hafiz, S.M.; Andou, Y.; Altarawneh, M.; Jiang, Z.T.; Huang, N.M. Modification of Carbon-Based Electroactive Materials for Supercapacitor Applications. In Carbon-Based Polymer Nanocomposites for Environmental and Energy Applications; Elsevier: Amsterdam, The Netherlands, 2018; pp. 393–413. [Google Scholar]
- Li, R.; He, C.; Han, X.; Yang, Y. Carbon-Based Polyaniline Nanocomposites for Supercapacitors. In Carbon-Based Polymer Nanocomposites for Environmental and Energy Applications; Elsevier: Amsterdam, The Netherlands, 2018; pp. 489–535. [Google Scholar]
- Zhang, S.; Shi, X.; Wróbel, R.; Chen, X.; Mijowska, E. Low-cost nitrogen-doped activated carbon prepared by polyethylenimine (PEI) with a convenient method for supercapacitor application. Electrochim. Acta 2019, 294, 183–191. [Google Scholar] [CrossRef]
- Xu, G.; Wang, N.; Wei, J.; Lv, L.; Zhang, J.; Chen, Z.; Xu, Q. Preparation of Graphene Oxide/Polyaniline Nanocomposite with Assistance of Supercritical Carbon Dioxide for Supercapacitor Electrodes. Ind. Eng. Chem. Res. 2012, 51, 14390–14398. [Google Scholar] [CrossRef]
- Biswas, S.; Drzal, L.T. Multilayered Nanoarchitecture of Graphene Nanosheets and Polypyrrole Nanowires for High Performance Supercapacitor Electrodes. Chem. Mater. 2010, 22, 5667–5671. [Google Scholar] [CrossRef]
- Park, J.H.; Ko, J.; Park, O.O.; Kim, D.-W. Capacitance properties of graphite/polypyrrole composite electrode prepared by chemical polymerization of pyrrole on graphite fiber. J. Power Sources 2002, 105, 20–25. [Google Scholar] [CrossRef]
- Chee, W.; Lim, H.N.; Harrison, I.; Chong, K.F.; Zainal, Z.; Ng, C.; Huang, N. Performance of Flexible and Binderless Polypyrrole/Graphene Oxide/Zinc Oxide Supercapacitor Electrode in a Symmetrical Two-Electrode Configuration. Electrochim. Acta 2015, 157, 88–94. [Google Scholar] [CrossRef]
- Lim, Y.; Tan, Y.; Lim, H.; Huang, N.M.; Tan, W.; Yarmo, M.; Yin, C.-Y. Potentiostatically deposited polypyrrole/graphene decorated nano-manganese oxide ternary film for supercapacitors. Ceram. Int. 2014, 40, 3855–3864. [Google Scholar] [CrossRef]
- Xiong, P.; Huang, H.; Wang, X. Design and synthesis of ternary cobalt ferrite/graphene/polyaniline hierarchical nanocomposites for high-performance supercapacitors. J. Power Sources 2014, 245, 937–946. [Google Scholar] [CrossRef]
- Han, C.; Shi, R.; Zhou, N.; Li, H.; Xu, L.; Zhang, T.; Li, J.; Kang, F.; Wang, G.; Li, B. High-Energy and High-Power Nonaqueous Lithium-Ion Capacitors Based on Polypyrrole/Carbon Nanotube Composites as Pseudocapacitive Cathodes. ACS Appl. Mater. Interfaces 2019, 11, 15646–15655. [Google Scholar] [CrossRef] [PubMed]
- Chen, T.; Dai, L. Flexible and wearable wire-shaped microsupercapacitors based on highly aligned titania and carbon nanotubes. Energy Storage Mater. 2016, 2, 21–26. [Google Scholar] [CrossRef]
- Ma, W.; Chen, S.; Yang, S.; Chen, W.; Cheng, Y.; Guo, Y.; Peng, S.; Ramakrishna, S.; Zhu, M.-F. Hierarchical MnO2 nanowire/graphene hybrid fibers with excellent electrochemical performance for flexible solid-state supercapacitors. J. Power Sources 2016, 306, 481–488. [Google Scholar] [CrossRef]
- Jiang, S.; Shi, T.; Zhan, X.; Long, H.; Xi, S.; Hu, H.; Tang, Z. High-performance all-solid-state flexible supercapacitors based on two-step activated carbon cloth. J. Power Sources 2014, 272, 16–23. [Google Scholar] [CrossRef]
- Zhao, D.; Chen, C.; Zhang, Q.; Chen, W.; Liu, S.; Wang, Q.; Liu, Y.; Li, J.; Yu, H. High Performance, Flexible, Solid-State Supercapacitors Based on a Renewable and Biodegradable Mesoporous Cellulose Membrane. Adv. Energy Mater. 2017, 7. [Google Scholar] [CrossRef]
- Wang, K.; Zhang, X.; Li, C.; Zhang, H.; Sun, X.; Xu, N.; Ma, Y. Flexible solid-state supercapacitors based on a conducting polymer hydrogel with enhanced electrochemical performance. J. Mater. Chem. A 2014, 2, 19726–19732. [Google Scholar] [CrossRef]
- Yu, J.; Wu, J.; Wang, H.; Zhou, A.; Huang, C.; Bai, H.; Li, L. Metallic Fabrics as the Current Collector for High-Performance Graphene-Based Flexible Solid-State Supercapacitor. ACS Appl. Mater. Interfaces 2016, 8, 4724–4729. [Google Scholar] [CrossRef]
- Wu, P.; Cheng, S.; Yao, M.; Yang, L.; Zhu, Y.; Liu, P.; Xing, O.; Zhou, J.; Wang, M.; Luo, H.; et al. A Low-Cost, Self-Standing NiCo2O4@CNT/CNT Multilayer Electrode for Flexible Asymmetric Solid-State Supercapacitors. Adv. Funct. Mater. 2017, 27. [Google Scholar] [CrossRef]
- Ye, K.-H.; Liu, Z.-Q.; Xu, C.-W.; Li, N.; Chen, Y.-B.; Su, Y.-Z. MnO2/reduced graphene oxide composite as high-performance electrode for flexible supercapacitors. Inorg. Chem. Commun. 2013, 30, 1–4. [Google Scholar] [CrossRef]
- Liu, C.; Zhao, S.; Lu, Y.; Chang, Y.; Xu, D.; Wang, Q.; Dai, Z.; Bao, J.; Han, M. 3D Porous Nanoarchitectures Derived from SnS/S-Doped Graphene Hybrid Nanosheets for Flexible All-Solid-State Supercapacitors. Small 2017, 13. [Google Scholar] [CrossRef] [PubMed]
- Liao, Q.; Li, N.; Jin, S.; Yang, G.; Wang, C. All-Solid-State Symmetric Supercapacitor Based on Co3O4 Nanoparticles on Vertically Aligned Graphene. ACS Nano 2015, 9, 5310–5317. [Google Scholar] [CrossRef] [PubMed]
- Zhu, G.; He, Z.; Chen, J.; Zhao, J.; Feng, X.; Ma, Y.; Sun, P.; Wang, L.; Huang, W. Highly conductive three-dimensional MnO2–carbon nanotube–graphene–Ni hybrid foam as a binder-free supercapacitor electrode. Nanoscale 2014, 6, 1079–1085. [Google Scholar] [CrossRef] [PubMed]
- Qin, T.; Peng, S.; Hao, J.; Wen, Y.; Wang, Z.; Wang, X.; He, D.; Zhang, J.; Hou, J.; Cao, G. Flexible and Wearable All-Solid-State Supercapacitors with Ultrahigh Energy Density Based on a Carbon Fiber Fabric Electrode. Adv. Energy Mater. 2017, 7. [Google Scholar] [CrossRef]
- Ko, W.-Y.; Chen, Y.-F.; Lu, K.-M.; Lin, K.-J. Porous honeycomb structures formed from interconnected MnO2 sheets on CNT-coated substrates for flexible all-solid-state supercapacitors. Sci. Rep. 2016, 6. [Google Scholar] [CrossRef] [PubMed]
- Parvez, K.; Wu, Z.; Li, R.; Liu, X.; Graf, R.; Feng, X.; Müllen, K. Exfoliation of Graphite into Graphene in Aqueous Solutions of Inorganic Salts. J. Am. Chem. Soc. 2014, 136, 6083–6091. [Google Scholar] [CrossRef]
- Wang, X.; Sumboja, A.; Foo, W.L.; Yan, C.Y.; Tsukagoshi, K.; Lee, P.S. Rational design of a high performance all solid state flexible micro-supercapacitor on paper. RSC Adv. 2013, 3, 15827–15833. [Google Scholar] [CrossRef]
- Yao, B.; Yuan, L.; Xiao, X.; Zhang, J.; Qi, Y.; Zhou, J.; Zhou, J.; Hu, B.; Chen, W. Paper-based solid-state supercapacitors with pencil-drawing graphite/polyaniline networks hybrid electrodes. Nano Energy 2013, 2, 1071–1078. [Google Scholar] [CrossRef]
- Dai, S.; Xu, W.; Xi, Y.; Wang, M.; Gu, X.; Guo, N.; Hu, C. Charge storage in KCu7S4 as redox active material for a flexible all-solid-state supercapacitor. Nano Energy 2016, 19, 363–372. [Google Scholar] [CrossRef]
- Lee, H.; Kwon, J.; Suh, Y.D.; Moon, H.; Hong, S.; Yeo, J.; Ko, S.H. All-solid-state flexible supercapacitors by fast laser annealing of printed metal nanoparticle layers. J. Mater. Chem. A 2015, 3, 8339–8345. [Google Scholar] [CrossRef]
- Yun, J.; Kim, D.; Lee, G.; Ha, J.S. All-solid-state flexible micro-supercapacitor arrays with patterned graphene/MWNT electrodes. Carbon 2014, 79, 156–164. [Google Scholar] [CrossRef]
- Kang, Y.J.; Chung, H.; Kim, M.-S.; Kim, W. Enhancement of CNT/PET film adhesion by nano-scale modification for flexible all-solid-state supercapacitors. Appl. Surf. Sci. 2015, 355, 160–165. [Google Scholar] [CrossRef]
- Hu, H.; Zhang, K.; Li, S.; Ji, S.; Ye, C. Flexible, in-plane, and all-solid-state micro-supercapacitors based on printed interdigital Au/polyaniline network hybrid electrodes on a chip. J. Mater. Chem. A 2014, 2, 20916–20922. [Google Scholar] [CrossRef]
- Xu, X.; Shi, W.; Li, P.; Ye, S.; Ye, C.; Ye, H.; Lu, T.; Zheng, A.; Zhu, J.; Xu, L.; et al. Facile Fabrication of Three-Dimensional Graphene and Metal–Organic Framework Composites and Their Derivatives for Flexible All-Solid-State Supercapacitors. Chem. Mater. 2017, 29, 6058–6065. [Google Scholar] [CrossRef]
- Liu, X.; Qian, T.; Xu, N.; Zhou, J.; Guo, J.; Yan, C. Preparation of on chip, flexible supercapacitor with high performance based on electrophoretic deposition of reduced graphene oxide/polypyrrole composites. Carbon 2015, 92, 348–353. [Google Scholar] [CrossRef]
- Liu, S.; Xie, J.; Li, H.; Wang, Y.; Yang, H.Y.; Zhu, T.; Zhang, S.; Cao, G.; Zhao, X. Nitrogen-doped reduced graphene oxide for high-performance flexible all-solid-state micro-supercapacitors. J. Mater. Chem. A 2014, 2, 18125–18131. [Google Scholar] [CrossRef]
- Cao, X.; Zheng, B.; Shi, W.; Yang, J.; Fan, Z.; Luo, Z.; Rui, X.; Chen, B.; Yan, Q.; Zhang, H. Reduced Graphene Oxide-Wrapped MoO3Composites Prepared by Using Metal-Organic Frameworks as Precursor for All-Solid-State Flexible Supercapacitors. Adv. Mater. 2015, 27, 4695–4701. [Google Scholar] [CrossRef]
- Sun, G.; An, J.; Chua, C.K.; Pang, H.; Zhang, J.; Chen, P. Layer-by-layer printing of laminated graphene-based interdigitated microelectrodes for flexible planar micro-supercapacitors. Electrochem. Commun. 2015, 51, 33–36. [Google Scholar] [CrossRef]
- Xie, J.; Sun, X.; Zhang, N.; Xu, K.; Zhou, M.; Xie, Y. Layer-by-layer β-Ni(OH)2/graphene nanohybrids for ultraflexible all-solid-state thin-film supercapacitors with high electrochemical performance. Nano Energy 2013, 2, 65–74. [Google Scholar] [CrossRef]
- Fei, H.; Yang, C.; Bao, H.; Wang, G. Flexible all-solid-state supercapacitors based on graphene/carbon black nanoparticle film electrodes and cross-linked poly(vinyl alcohol)–H2SO4 porous gel electrolytes. J. Power Sources 2014, 266, 488–495. [Google Scholar] [CrossRef]
- Li, X.; Zhao, T.; Chen, Q.; Li, P.; Wang, K.; Zhong, M.; Wei, J.; Wu, D.; Wei, B.; Zhu, H. Flexible all solid-state supercapacitors based on chemical vapor deposition derived graphene fibers. Phys. Chem. Chem. Phys. 2013, 15, 17752–17757. [Google Scholar] [CrossRef] [PubMed]
- Shao, Y.; Wang, H.; Zhang, Q.; Li, Y. Fabrication of large-area and high-crystallinity photoreduced graphene oxide films via reconstructed two-dimensional multilayer structures. NPG Asia Mater. 2014, 6, e119. [Google Scholar] [CrossRef]
- Chen, J.; Xu, J.; Zhou, S.; Zhao, N.; Wong, C.-P. Facile and scalable fabrication of three-dimensional Cu(OH)2nanoporous nanorods for solid-state supercapacitors. J. Mater. Chem. A 2015, 3, 17385–17391. [Google Scholar] [CrossRef]
- Niu, H.; Yang, X.; Jiang, H.; Zhou, D.; Li, X.; Zhang, T.; Liu, J.; Wang, Q.; Qu, F. Hierarchical core–shell heterostructure of porous carbon nanofiber@ZnCo2O4 nanoneedle arrays: Advanced binder-free electrodes for all-solid-state supercapacitors. J. Mater. Chem. A 2015, 3, 24082–24094. [Google Scholar] [CrossRef]
- Siwal, S.S.; Zhang, Q.; Sun, C.; Thakur, V.K. Graphitic Carbon Nitride Doped Copper-Manganese Alloy as High-Performance Electrode Material in Supercapacitor for Energy Storage. Nanomaterials 2019, 10, 2. [Google Scholar] [CrossRef] [PubMed]


| EDLC | Pseudocapacitor |
|---|---|
| Good cyclic stability | Greater specific capacitance |
| Good power performance | Greater energy density |
| Formation of the double layer at the interface | No such layer formation |
| No redox reaction i.e., non-faradaic | Redox reactions, i.e., Faradaic |
| No mechanism failure | Depends upon redox reactions |
| Fuel Cells | Anode Feed | Cathode Feed | Working Temp. (°C) | Power Density (mW/cm2) | Fuel Efficiency |
|---|---|---|---|---|---|
| Alkaline fuel cell | Pure H2 | O2 or air | 90–100 | 100–200 | 60 |
| PEMFCs | Pure H2 | O2 or air | 50–100 | 350 | 60 |
| Phosphoric acid fuel cell | Pure H2 | O2 or air | 150–200 | 200 | 40 |
| Molten carbonate fuel cell | H2 or natural gas | O2 or air | 600–700 | 100 | 45–50 |
| Solid oxide fuel cell | Gasoline or natural gas | O2 or air | 700–1000 | 240 | 60 |
| Substances | Specific Capacitance (Cs) | Cycling Durability | Ref. |
|---|---|---|---|
| CNT | 1.8 mF·cm−2 on 1 mA | 80%; 1000 runs | [142] |
| MnO2 nanowire/GN | 66.1 F·cm−3 on 60 mA·cm−3 | 96%; 10,000 runs on 0.12 A·cm−3 | [143] |
| AC cloth | 161.2 mF·cm−2 on 12.5 mA·cm−2 | 104%; 30,000 runs on 12.5 mA·cm−2 | [144] |
| AC | 153 mF·cm−2 on 10 mV·s−1 | 93.4%; 1000 runs on 200 mV·s−1 | [145] |
| PANI hydrogel | 430 F·g−1 on 5 mV·s−1 | 86%; 1000 runs on 7.5 A·g−1 | [146] |
| GN | 180.40 mF·cm−2 on 1 mA·cm−2 | 96.8%; 7500 runs on 8 mA·cm−2 | [147] |
| TiO2@PANI | 775.6 mF·cm−3 (28.3 F·g−1) on 10 mV·s−1 | 97.2%; 10,000 runs on 100 mV·s−1 | [147] |
| NiCo2O4@CNT/CNT | - | 95%; 5000 runs on 50 mV·s−1 | [148] |
| MnO2/rGO | 14 F·cm−2 (31.8 F·g−1) on 2 mV·s−1 | 100%; 5000 runs on 0.2 mA·cm−2 | [149] |
| SnS/S doped GN | 2.98 mF·cm−2 on 60 mA·cm−2 | 99%; 10,000 runs on 120 mA·m−2 | [150] |
| Co3O4/vertically aligned GN nanosheets | 580 F·g−1 on 1 A·g−1 | 86.3%; 20,000 runs on 20 A·g−1 | [151] |
| MnO2-CNT-GN | 107 F·g−1 | - | [152] |
| N/O-Enhanced carbon cloth | - | 116%; 5000 runs on 5 mA·cm−1 | [153] |
| MnO2/CNT | 324 F·g−1 on 0.5 A·g−1 | 100%; 5000 runs on 10 A·g−1 | [154] |
| GN sheets | 11.3 mF·cm−2 on 1 mV·s−1 | - | [155] |
| PANI-MnOx | 94.73 mF·cm−2 on 0.1 mA·cm−2 | - | [156] |
| Graphite/PANI | 77.8 mF·cm−2 on 0.1 mA·cm−2 | 83%; 10,000 runs on 1 mA·cm−2 | [157] |
| KCu7S4/GN | - | 92%; 5000 runs on 0.8 mA·cm−2 | [158] |
| Ag/AC | 45 mF·cm−2 on 0.3 mA·cm−2 | 86%; 1200 runs on 5 mA·cm−2 | [159] |
| GN/MWNT | 740.9 mF·cm−2 on 1 mA·cm−2 | 85%; 20,000 runs on 15 mA·cm−2 | [160] |
| SWCNTs | 17.5 F·g−1 on 2 A·g−1 | 87.5%; 10,000 runs on 5 A·g−1 | [161] |
| Au/PANI | 26.49 mF·cm−2 (67.06 F·cm−3) on 0.5 mA·cm−2 | 72.7%; 1000 runs on 200 mV·s−1 | [162] |
| GO/MOF | 250 mF·cm−3 on 6.4 mA·cm−3 | 96.3%; 5000 runs on 50.4 mA·cm−3 | [163] |
| rGO/PPy | 147.9 F· cm−3 on 5 A·cm−3 | 71.7%; 5000 runs on 10 A·cm−3 | [164] |
| N-Doped rGO | 3.4 mF·cm−2 on 20 mA·cm−2 | 98.4%; 2000 runs on 100 mA·cm−2 | [165] |
| rGO/MoO3 | 404 F·g−1 on 0.5 A·g−1 | 80%; 5000 runs on 2 A·g−1 | [166] |
| GN | 56.5 F·cm−3 on 0.06 A·cm−3 | - | [167] |
| b-Ni(OH)2/graphene | 2570 mF·cm−2 on 0.2 A·m−1 | 98.2%; 2000 runs on 0.1 A·m−2 | [168] |
| GN/carbon black nanoparticle | 144.5 F·g−1 on the current density of 0.5 A·g−1 | - | [169] |
| GN fibers/MnO2 fibers | 42.02 mF·cm−2 on 0.01 V·s−1 | 92%; 1000 runs on 1 mA·cm−2 | [170] |
| GO | 130 F·g−1 on 5 mV·s−1 | - | [171] |
| Cu(OH)2//AC | 26.4 F·g−1 on 4 A·g−1 | 90%; 5000 runs | [172] |
| ZnCo2O4//carbon nanofibers | 139.2 F·g−1 on 2 mV·s−1 | 90%; 3000 runs on 50 mV·s−1 | [173] |
| CuMnO2-gCN | 817.85 F·g−1 at 0.025 A·g−1 | 91% up to 1000 cycles | [174] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Siwal, S.S.; Zhang, Q.; Devi, N.; Thakur, V.K. Carbon-Based Polymer Nanocomposite for High-Performance Energy Storage Applications. Polymers 2020, 12, 505. https://doi.org/10.3390/polym12030505
Siwal SS, Zhang Q, Devi N, Thakur VK. Carbon-Based Polymer Nanocomposite for High-Performance Energy Storage Applications. Polymers. 2020; 12(3):505. https://doi.org/10.3390/polym12030505
Chicago/Turabian StyleSiwal, Samarjeet Singh, Qibo Zhang, Nishu Devi, and Vijay Kumar Thakur. 2020. "Carbon-Based Polymer Nanocomposite for High-Performance Energy Storage Applications" Polymers 12, no. 3: 505. https://doi.org/10.3390/polym12030505
APA StyleSiwal, S. S., Zhang, Q., Devi, N., & Thakur, V. K. (2020). Carbon-Based Polymer Nanocomposite for High-Performance Energy Storage Applications. Polymers, 12(3), 505. https://doi.org/10.3390/polym12030505







