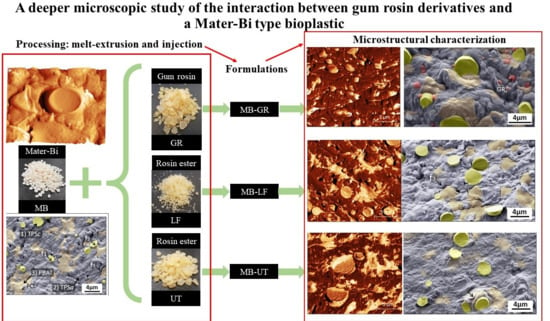
A Deeper Microscopic Study of the Interaction between Gum Rosin Derivatives and a Mater-Bi Type Bioplastic
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Samples Preparation
2.3. Sample Characterization
2.3.1. Tensile Test
2.3.2. Dynamic Thermo-Mechanical Analysis (DTMA)
2.3.3. Microscopic Characterization
2.3.4. Oxygen Transmission Rate (OTR)
2.3.5. Static Contact Angle Measurements
3. Results and Discussion
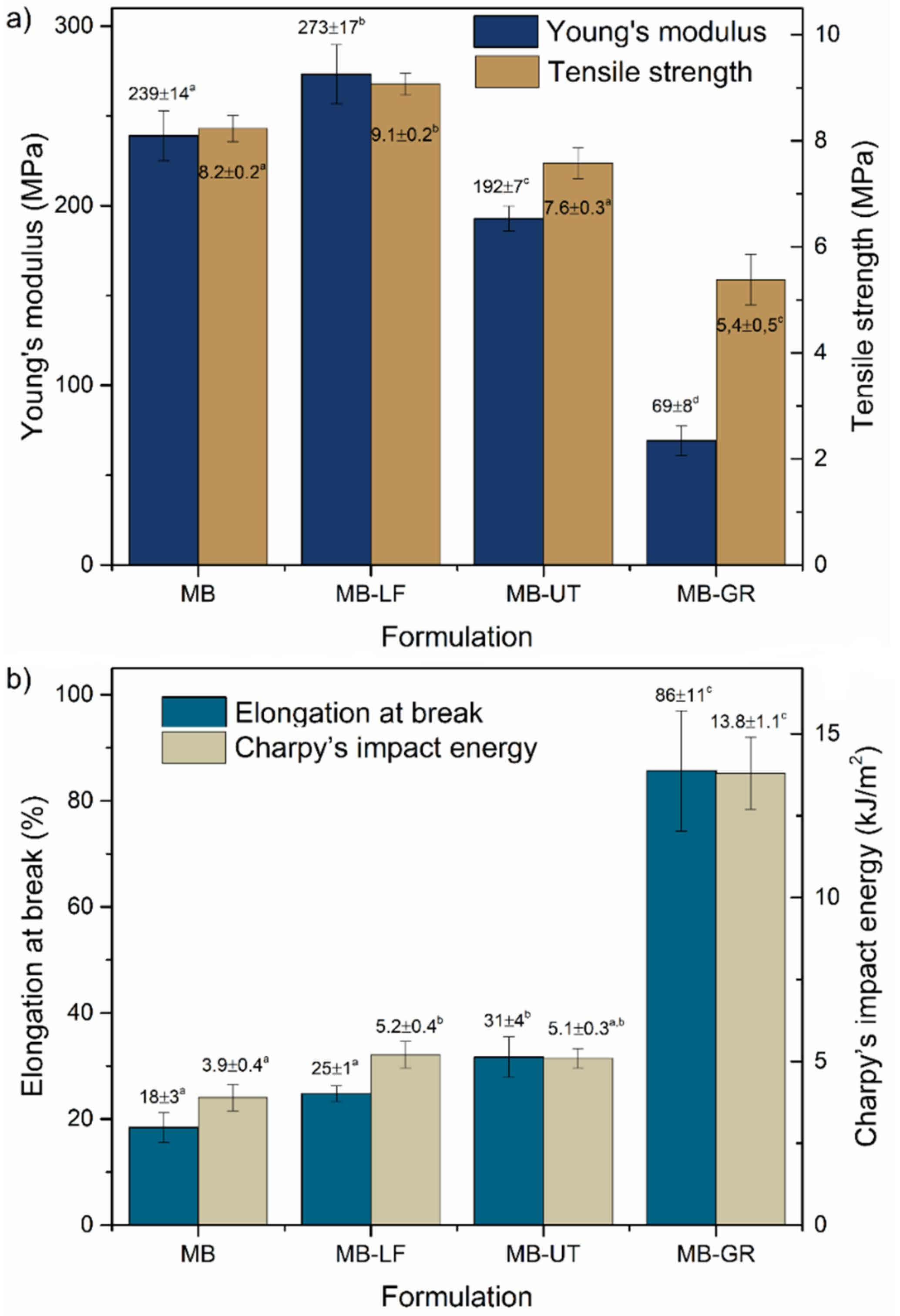
3.1. Tensile Test
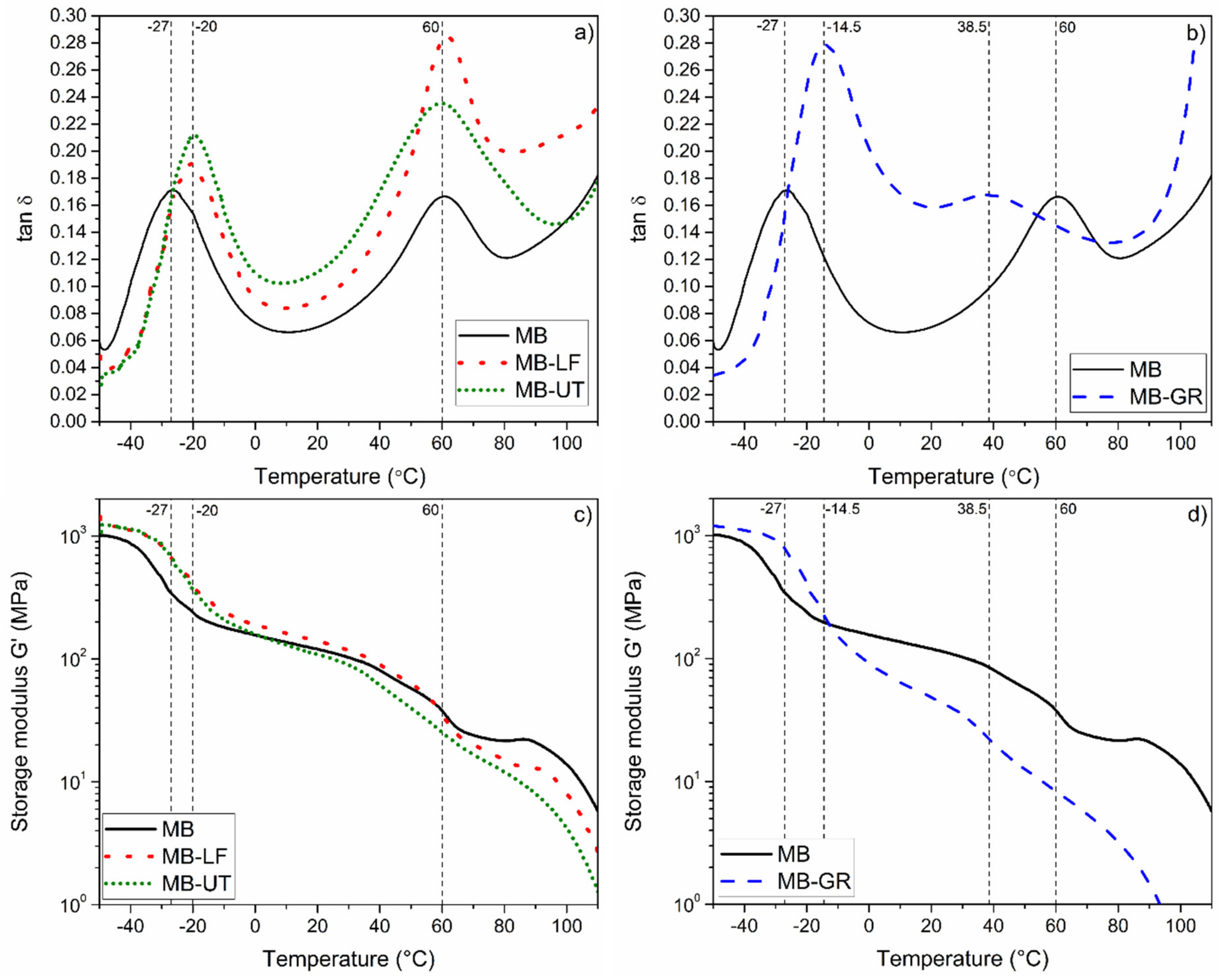
3.2. Dynamic Thermo-Mechanical Analysis (DTMA) Characterization
3.3. Field Emission Scanning Electron Microscopy (FE-SEM) Microstructural Characterization
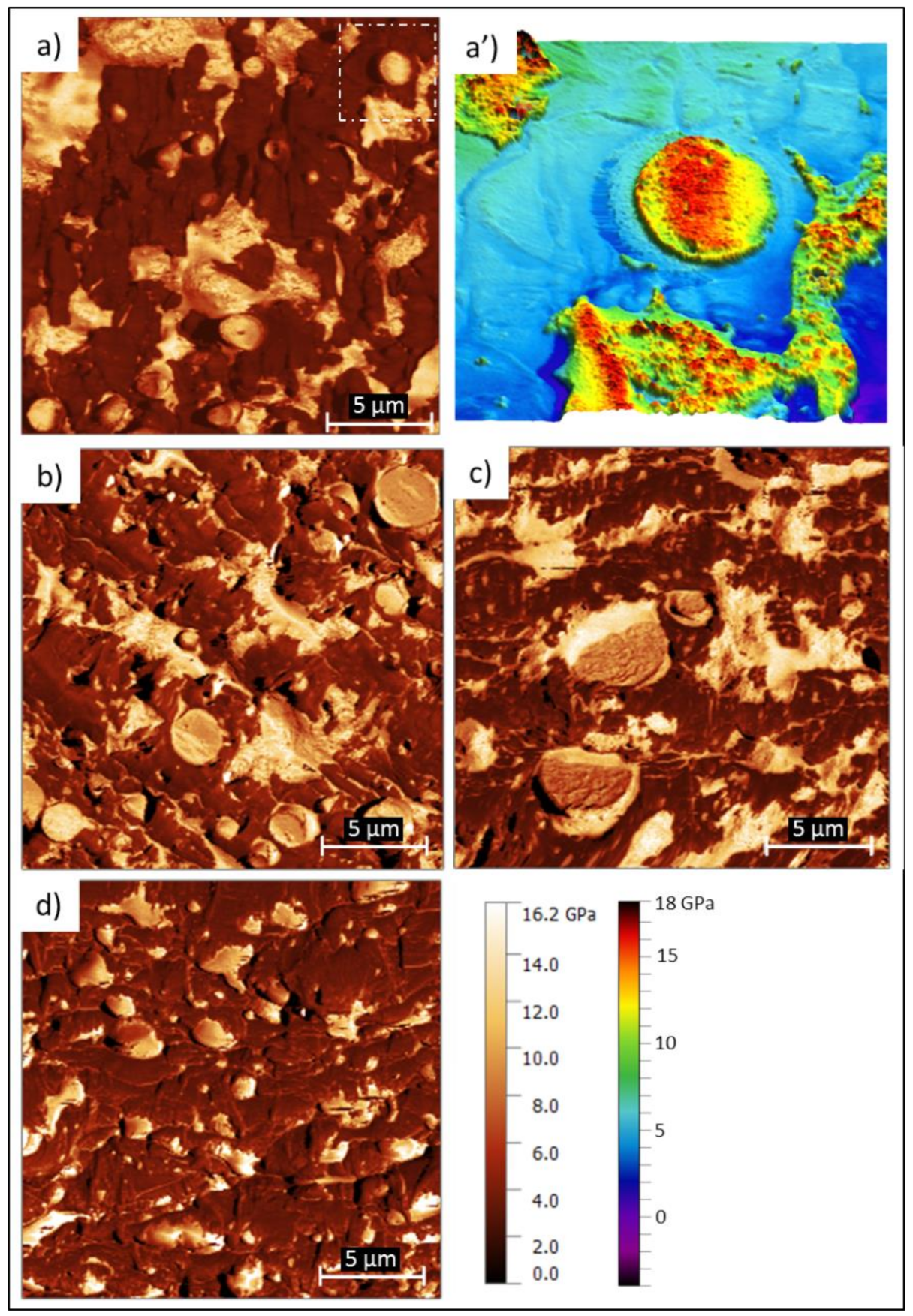
3.4. Atomic Force Microscopy with Nanomechanical Assessment (AFM-QNM) Microstructural Characterization
3.5. Oxygen Permeability and Wettability
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Keshavarz, T.; Roy, I. Polyhydroxyalkanoates: Bioplastics with a green agenda. Curr. Opin. Microbiol. 2010, 13, 321–326. [Google Scholar] [CrossRef]
- Aldas, M.; Paladines, A.; Valle, V.; Pazmiño, M.; Quiroz, F. Effect of the prodegradant-additive plastics incorporated on the polyethylene recycling. Int. J. Polym. Sci. 2018, 2018, 2474176. [Google Scholar] [CrossRef]
- Arrieta, M.P.; Peponi, L.; López, D.; Fernández-García, M. Recovery of yerba mate (Ilex paraguariensis) residue for the development of PLA-based bionanocomposite films. Ind. Crops Prod. 2018, 111, 317–328. [Google Scholar] [CrossRef]
- Akrami, M.; Ghasemi, I.; Azizi, H.; Karrabi, M.; Seyedabadi, M. A new approach in compatibilization of the poly (lactic acid)/thermoplastic starch (pla/tps) blends. Carbohydr. Polym. 2016, 144, 254–262. [Google Scholar] [CrossRef] [PubMed]
- Borchani, K.E.; Carrot, C.; Jaziri, M. Biocomposites of alfa fibers dispersed in the mater-bi® type bioplastic: Morphology, mechanical and thermal properties. Compos. Part A Appl. Sci. Manuf. 2015, 78, 371–379. [Google Scholar] [CrossRef]
- Sessini, V.; Arrieta, M.P.; Fernández-Torres, A.; Peponi, L. Humidity-activated shape memory effect on plasticized starch-based biomaterials. Carbohydr. Polym. 2018, 179, 93–99. [Google Scholar] [CrossRef] [PubMed]
- Arrieta, M.P.; Samper, M.D.; Aldas, M.; López, J. On the use of PLA-PHB blends for sustainable food packaging applications. Materials 2017, 10, 1008. [Google Scholar] [CrossRef] [PubMed]
- Aldas, M.; Ferri, J.; Lopez-Martinez, J.; Samper, M.; Arrieta, M. Effect of pine resin derivatives on the structural, thermal, and mechanical properties of mater-bi type bioplastic. J. Appl. Polym. Sci. 2019. [Google Scholar] [CrossRef]
- Sessini, V.; Navarro-Baena, I.; Arrieta, M.P.; Dominici, F.; López, D.; Torre, L.; Kenny, J.M.; Dubois, P.; Raquez, J.-M.; Peponi, L. Effect of the addition of polyester-grafted-cellulose nanocrystals on the shape memory properties of biodegradable PLA/PCL nanocomposites. Polym. Degrad. Stab. 2018, 152, 126–138. [Google Scholar] [CrossRef]
- Sessini, V.; Arrieta, M.P.; Raquez, J.-M.; Dubois, P.; Kenny, J.M.; Peponi, L. Thermal and composting degradation of eva/thermoplastic starch blends and their nanocomposites. Polym. Degrad. Stab. 2019, 159, 184–198. [Google Scholar] [CrossRef]
- Kaseem, M.; Hamad, K.; Deri, F. Thermoplastic starch blends: A review of recent works. Polym. Sci. Ser. A 2012, 54, 165–176. [Google Scholar] [CrossRef]
- Olivato, J.B.; Nobrega, M.M.; Müller, C.M.O.; Shirai, M.A.; Yamashita, F.; Grossmann, M.V.E. Mixture design applied for the study of the tartaric acid effect on starch/polyester films. Carbohydr. Polym. 2013, 92, 1705–1710. [Google Scholar] [CrossRef] [PubMed]
- Wojtowicz, A.; Janssen, L.; Moscicki, L. Blends of natural and synthetic polymers. In Thermoplastic Starch. Janssen LPBM; Moscicki, L., Ed.; WILEY-VCH Verlag GmbH& Co. KGaA: Weinheim, Germany, 2009. [Google Scholar]
- Yoshida, Y.; Uemura, T. Properties and applications of “mater-bi”. In Studies in Polymer Science; Doi, Y., Fukuda, K., Eds.; Elsevier: Amsterdam, The Netherlands, 1994; Volume 12, pp. 443–450. [Google Scholar]
- Bastioli, C.; Milizia, T.; Floridi, G.; Cella, G.; Tosin, M.; Scaffid, A. Poliésteres Alifáticos-Aromáticos. Biodegradable. Patent ES2342192T3, 2 July 2010. [Google Scholar]
- Nainggolan, H.; Gea, S.; Bilotti, E.; Peijs, T.; Hutagalung, S.D. Mechanical and thermal properties of bacterial-cellulose-fibre-reinforced mater-bi® bionanocomposite. Beilstein J. Nanotechnol. 2013, 4, 325–329. [Google Scholar] [CrossRef] [PubMed]
- Ferri, J.; Garcia-Garcia, D.; Sánchez-Nacher, L.; Fenollar, O.; Balart, R. The effect of maleinized linseed oil (mlo) on mechanical performance of poly (lactic acid)-thermoplastic starch (pla-tps) blends. Carbohydr. Polym. 2016, 147, 60–68. [Google Scholar] [CrossRef] [PubMed]
- Morreale, M.; Scaffaro, R.; Maio, A.; La Mantia, F. Effect of adding wood flour to the physical properties of a biodegradable polymer. Compos. Part A Appl. Sci. Manuf. 2008, 39, 503–513. [Google Scholar] [CrossRef]
- Nayak, S. Biodegradable pbat/starch nanocomposites. Polym. Plast. Technol. Eng. 2010, 49, 1406–1418. [Google Scholar] [CrossRef]
- Gonzalez Seligra, P.; Eloy Moura, L.; Famá, L.; Druzian, J.I.; Goyanes, S. Influence of incorporation of starch nanoparticles in pbat/tps composite films. Polym. Int. 2016, 65, 938–945. [Google Scholar] [CrossRef]
- Manzano, V.; García, N.L.; Ramírez, C.R.; D’Accorso, N.; Goyanes, S. Mulch plastic systems: Recent advances and applications. In Polymers for Agri-Food Applications; Springer: New York, NY, USA, 2019; pp. 265–290. [Google Scholar]
- Arrieta, M.P.; Samper, M.; Jiménez-López, M.; Aldas, M.; López, J. Combined effect of linseed oil and gum rosin as natural additives for PVC. Ind. Crops Prod. 2017, 99, 196–204. [Google Scholar] [CrossRef]
- Gutierrez, J.; Tercjak, A. Natural gum rosin thin films nanopatterned by poly(styrene)-block-poly (4-vinylpiridine) block copolymer. RSC Adv. 2014, 4, 32024–32030. [Google Scholar] [CrossRef]
- Wilbon, P.A.; Chu, F.; Tang, C. Progress in renewable polymers from natural terpenes, terpenoids, and rosin. Macromol. Rapid Commun. 2013, 34, 8–37. [Google Scholar] [CrossRef]
- Rodríguez-García, A.; Martín, J.A.; López, R.; Mutke, S.; Pinillos, F.; Gil, L. Influence of climate variables on resin yield and secretory structures in tapped pinus pinaster ait. In central spain. Agric. For. Meteorol. 2015, 202, 83–93. [Google Scholar] [CrossRef]
- Davis, G.; Song, J.H. Biodegradable packaging based on raw materials from crops and their impact on waste management. Ind. Crops Prod. 2006, 23, 147–161. [Google Scholar] [CrossRef]
- Yadav, B.K.; Gidwani, B.; Vyas, A. Rosin: Recent advances and potential applications in novel drug delivery system. J. Bioact. Compat. Polym. 2016, 31, 111–126. [Google Scholar] [CrossRef]
- Silvestre, A.J.; Gandini, A. Rosin: Major sources, properties and applications. In Monomers, Polymers and Composites from Renewable Resources; Elsevier: Amsterdam, The Netherlands, 2008; pp. 67–88. [Google Scholar]
- Abdel-Raouf, M.E.-S.; Abdul-Raheim, A.-R.M. Rosin: Chemistry, derivatives, and applications: A review. BAOJ Chem. 2018, 4, 39. [Google Scholar]
- ISO 527-1:2012. Plastics—Determination of Tensile Properties—Part 1: General Principles; International Standards Organization: Geneva, Switzerland, 2012. [Google Scholar]
- ISO 179-1:2010. Plastics—Determination of Charpy Impact Properties/Part 1: Non-Instrumented Impact Test; International Standards Organization: Geneva, Switzerland, 2010. [Google Scholar]
- Butt, H.-J.; Cappella, B.; Kappl, M. Force measurements with the atomic force microscope: Technique, interpretation and applications. Surf. Sci. Rep. 2005, 59, 1–152. [Google Scholar] [CrossRef]
- Roa, J.; Rayon, E.; Morales, M.; Segarra, M. Contact mechanics at nanometric scale using nanoindentation technique for brittle and ductile materials. Recent Pat. Eng. 2012, 6, 116–126. [Google Scholar] [CrossRef]
- Hernández-Fernández, J.; Rayón, E.; López, J.; Arrieta, M.P. Enhancing the thermal stability of polypropylene by blending with low amounts of natural antioxidants. Macromol. Mater. Eng. 2019. [Google Scholar] [CrossRef]
- Taguet, A.; Huneault, M.A.; Favis, B.D. Interface/morphology relationships in polymer blends with thermoplastic starch. Polymer 2009, 50, 5733–5743. [Google Scholar] [CrossRef]
- Zhang, S.; He, Y.; Lin, Z.; Li, J.; Jiang, G. Effects of tartaric acid contents on phase homogeneity, morphology and properties of poly (butyleneadipate-co-terephthalate)/thermoplastic starch bio-composities. Polym. Test. 2019, 76, 385–395. [Google Scholar] [CrossRef]
- Mohammadi Nafchi, A.; Moradpour, M.; Saeidi, M.; Alias, A.K. Thermoplastic starches: Properties, challenges, and prospects. Starch-Stärke 2013, 65, 61–72. [Google Scholar] [CrossRef]
- Van Soest, J.; De Wit, D.; Vliegenthart, J. Mechanical properties of thermoplastic waxy maize starch. J. Appl. Polym. Sci. 1996, 61, 1927–1937. [Google Scholar] [CrossRef]
- Zhang, Y.; Rempel, C.; Liu, Q. Thermoplastic starch processing and characteristics—A review. Crit. Rev. Food Sci. Nutr. 2014, 54, 1353–1370. [Google Scholar] [CrossRef] [PubMed]
- Janssen, L.; Moscicki, L. Thermoplastic Starch: A Green Material for Various Industries; John Wiley & Sons: Hoboken, NJ, USA, 2009. [Google Scholar]
- Yu, J.; Gao, J.; Lin, T. Biodegradable thermoplastic starch. J. Appl. Polym. Sci. 1996, 62, 1491–1494. [Google Scholar] [CrossRef]
- Zullo, R.; Iannace, S. The effects of different starch sources and plasticizers on film blowing of thermoplastic starch: Correlation among process, elongational properties and macromolecular structure. Carbohydr. Polym. 2009, 77, 376–383. [Google Scholar] [CrossRef]
- Sousa, F.M.; Costa, A.R.M.; Reul, L.T.; Cavalcanti, F.B.; Carvalho, L.H.; Almeida, T.G.; Canedo, E.L. Rheological and thermal characterization of pcl/pbat blends. Polym. Bull. 2019, 76, 1573–1593. [Google Scholar] [CrossRef]
- Mittal, V.; Akhtar, T.; Luckachan, G.; Matsko, N. Pla, tps and pcl binary and ternary blends: Structural characterization and time-dependent morphological changes. Colloid Polym. Sci. 2015, 293, 573–585. [Google Scholar] [CrossRef]
- Arrieta, M.P.; López, J.; Hernández, A.; Rayón, E. Ternary PLA-PHB-limonene blends intended for biodegradable food packaging applications. Eur. Polym. J. 2014, 50, 255–270. [Google Scholar] [CrossRef]
- Burgos, N.; Martino, V.P.; Jiménez, A. Characterization and ageing study of poly (lactic acid) films plasticized with oligomeric lactic acid. Polym. Degrad. Stab. 2013, 98, 651–658. [Google Scholar] [CrossRef]
- Armentano, I.; Fortunati, E.; Burgos, N.; Dominici, F.; Luzi, F.; Fiori, S.; Jiménez, A.; Yoon, K.; Ahn, J.; Kang, S. Processing and Characterization of Plasticized PLA/PHB Blends for Biodegradable Multiphase Systems Express. Polym. Lett. 2015, 9, 583–596. [Google Scholar] [CrossRef]
- Arrieta, M.P.; Peltzer, M.A.; López, J.; Garrigós, M.D.C.; Valente, A.J.M.; Jiménez, A. Functional properties of sodium and calcium caseinate antimicrobial active films containing carvacrol. J. Food Eng. 2014, 121, 94–101. [Google Scholar] [CrossRef]
- Hambleton, A.; Fabra, M.-J.; Debeaufort, F.; Dury-Brun, C.; Voilley, A. Interface and aroma barrier properties of iota-carrageenan emulsion–based films used for encapsulation of active food compounds. J. Food Eng. 2009, 93, 80–88. [Google Scholar] [CrossRef]



| Sample | OTR.e (cm3 mm m−2 day−1) | WCA |
|---|---|---|
| Mater-Bi® NF 866 | 57.0 ± 3.4 | 94.7 ± 1.3 |
| MB-LF | 118.9 ± 8.4 | 93.6 ± 1.5 |
| MB-UT | 55.7 ± 2.4 | 78.7 ± 1.2 |
| MB-GR | 78.6 ± 1.2 | 83.3 ± 1.6 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aldas, M.; Rayón, E.; López-Martínez, J.; Arrieta, M.P. A Deeper Microscopic Study of the Interaction between Gum Rosin Derivatives and a Mater-Bi Type Bioplastic. Polymers 2020, 12, 226. https://doi.org/10.3390/polym12010226
Aldas M, Rayón E, López-Martínez J, Arrieta MP. A Deeper Microscopic Study of the Interaction between Gum Rosin Derivatives and a Mater-Bi Type Bioplastic. Polymers. 2020; 12(1):226. https://doi.org/10.3390/polym12010226
Chicago/Turabian StyleAldas, Miguel, Emilio Rayón, Juan López-Martínez, and Marina P. Arrieta. 2020. "A Deeper Microscopic Study of the Interaction between Gum Rosin Derivatives and a Mater-Bi Type Bioplastic" Polymers 12, no. 1: 226. https://doi.org/10.3390/polym12010226
APA StyleAldas, M., Rayón, E., López-Martínez, J., & Arrieta, M. P. (2020). A Deeper Microscopic Study of the Interaction between Gum Rosin Derivatives and a Mater-Bi Type Bioplastic. Polymers, 12(1), 226. https://doi.org/10.3390/polym12010226