Fiber Forming Capability of Binary and Ternary Compositions in the Polymer System: Bacterial Cellulose–Polycaprolactone–Polylactic Acid
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of Blend Solutions
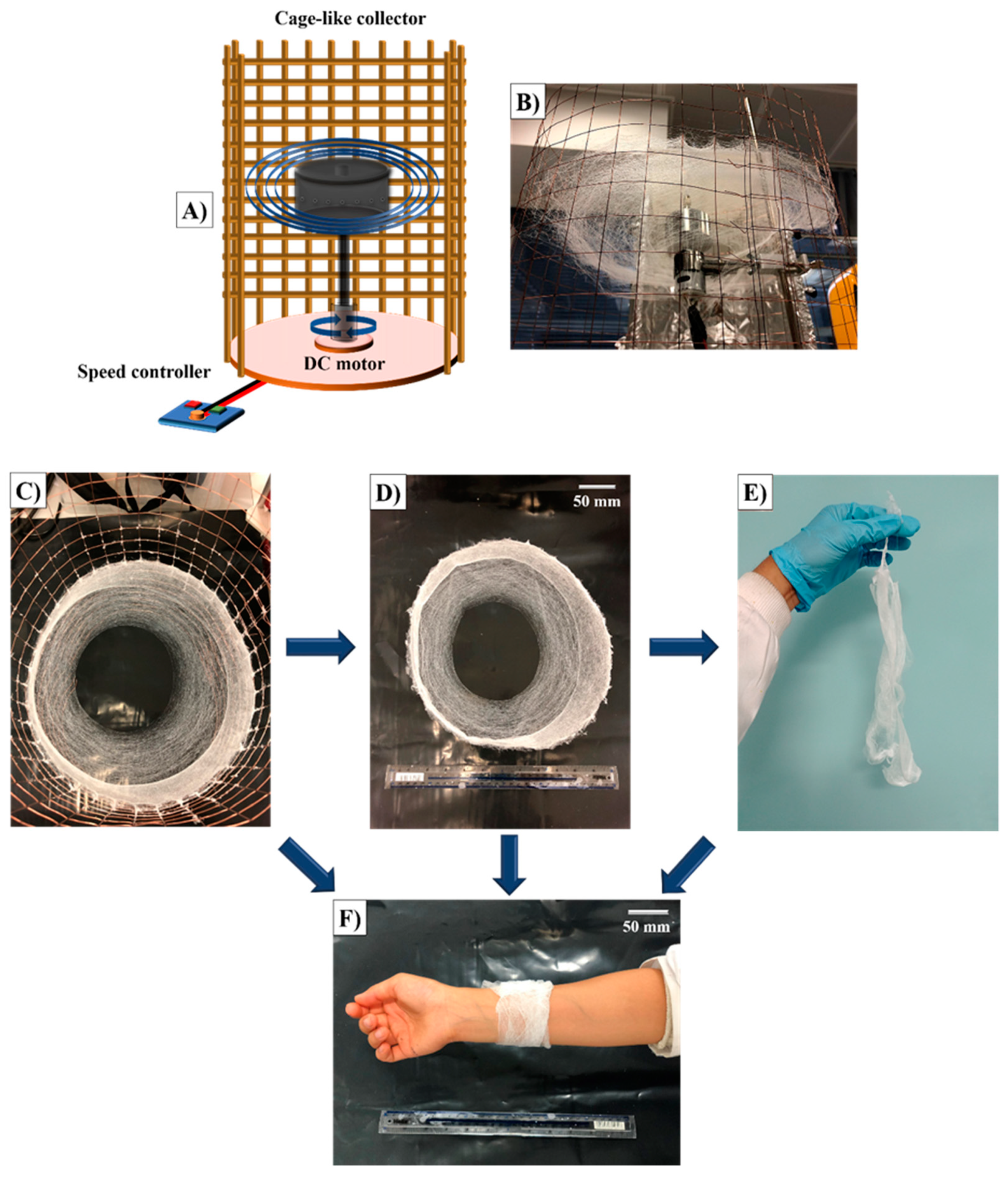
2.3. Bandage-Like Scaffold Production
2.4. Characterization
2.4.1. Physical
2.4.2. Morphological
2.4.3. Chemical
2.4.4. Mechanical
3. Results and Discussion
3.1. Yield Calculations
3.2. Morphological Features of the Scaffolds
3.3. Chemical Characterization of the Scaffolds
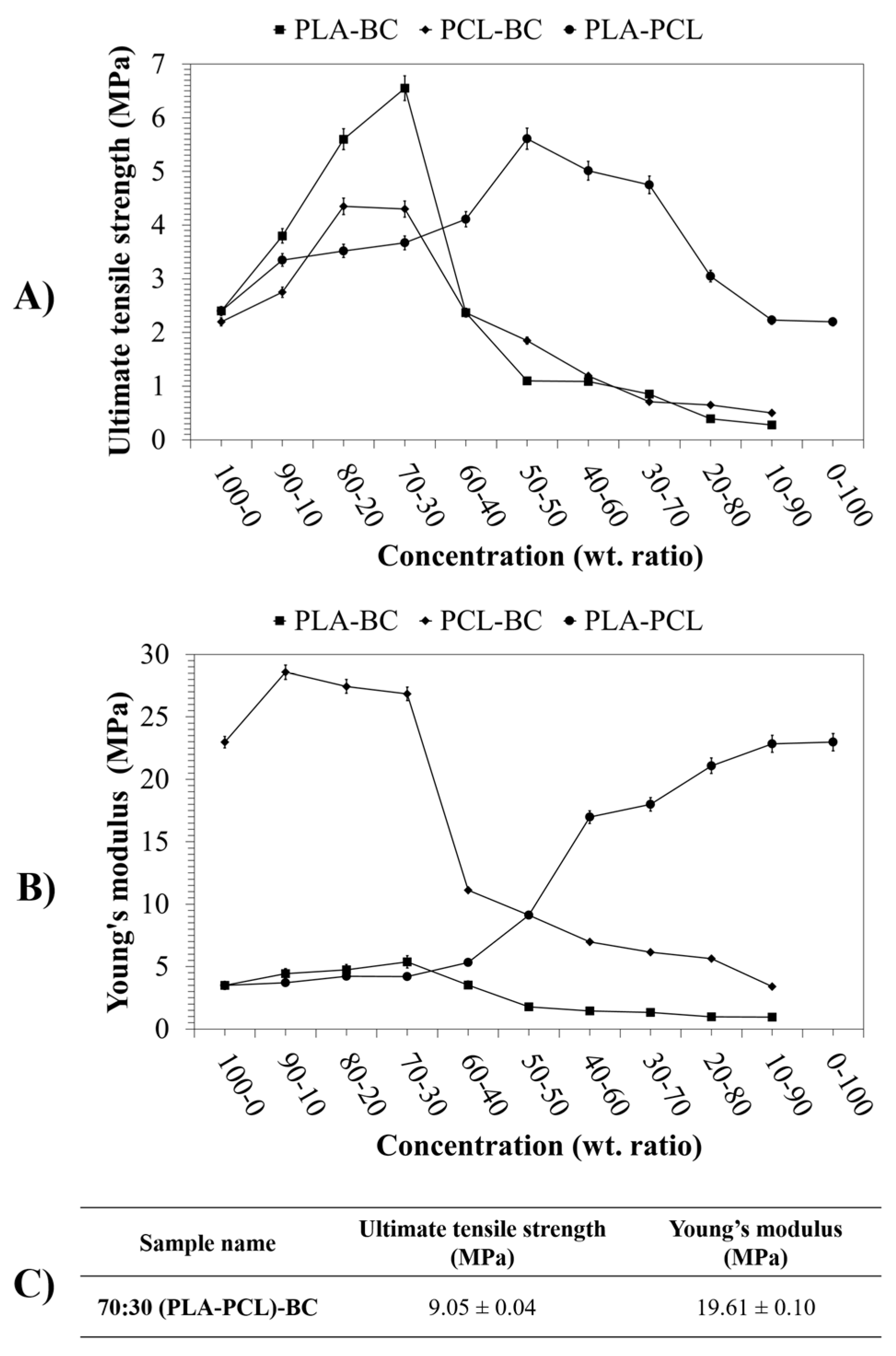
3.4. Mechanical Properties of the Scaffolds
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Huang, X.; Zhang, Y.; Zhang, X.; Xu, L.; Chen, X.; Wei, S. Influence of radiation crosslinked carboxymethyl-chitosan/gelatin hydrogel on cutaneous wound healing. Mater. Sci. Eng. C 2013, 33, 4816–4824. [Google Scholar] [CrossRef] [PubMed]
- Rittie, L. Cellular mechanisms of skin repair in humans and other mammals. J. Cell Commun. Signal. 2016, 10, 103–120. [Google Scholar] [CrossRef] [PubMed]
- Yildirimer, L.; Thanh, N.T.; Seifalian, A.M. Skin regeneration scaffolds: A multimodal bottom-up approach. Trends Biotechnol. 2012, 30, 638–648. [Google Scholar] [CrossRef] [PubMed]
- Altun, E.; Aydogdu, M.O.; Crabbe-Mann, M.; Ahmed, J.; Brako, F.; Karademir, B.; Aksu, B.; Sennaroglu, M.; Eroglu, M.S.; Ren, G.; et al. Co-culture of keratinocyte-staphylococcus aureus on cu-ag-zn/cuo and cu-ag-w nanoparticle loaded bacterial cellulose: Pmma bandages. Macromol. Mater. Eng. 2018, 1800537. [Google Scholar] [CrossRef]
- Kuang, T.; Chen, F.; Chang, L.; Zhao, Y.; Fu, D.; Gong, X.; Peng, X. Facile preparation of open-cellular porous poly (l-lactic acid) scaffold by supercritical carbon dioxide foaming for potential tissue engineering applications. Chem. Eng. J. 2017, 307, 1017–1025. [Google Scholar] [CrossRef]
- Cullum, N.; Buckley, H.L.; Dumville, J.; Hall, J.; Lamb, K.; Madden, M.T.; Morley, R.; O’Meara, S.M.; Saramago Goncalves, P.R.; Soares, M.O.; et al. Wounds research for patient benefit. Health Technol. Assess. 2016, 4, 1–334. [Google Scholar]
- Aydogdu, M.O.; Altun, E.; Crabbe-Mann, M.; Brako, F.; Koc, F.; Ozen, G.; Kuruca, S.E.; Edirisinghe, U.; Luo, C.J.; Gunduz, O.; et al. Cellular interactions with bacterial cellulose: Polycaprolactone nanofibrous scaffolds produced by a portable electrohydrodynamic gun for point-of-need wound dressing. Int. Wound J. 2018, 15, 789–797. [Google Scholar] [CrossRef]
- Altun, E.; Aydogdu, M.O.; Togay, S.O.; Sengil, A.Z.; Ekren, N.; Haskoylu, M.E.; Oner, E.T.; Altuncu, N.A.; Ozturk, G.; Crabbe-Mann, M.; et al. Bioinspired scaffold induced regeneration of neural tissue. Eur. Polym. J. 2019, 114, 98–108. [Google Scholar] [CrossRef]
- Chuah, C.; Wang, J.; Tavakoli, J.; Tang, Y. Novel bacterial cellulose-poly (acrylic acid) hybrid hydrogels with controllable antimicrobial ability as dressings for chronic wounds. Polymers 2018, 10, 1323. [Google Scholar] [CrossRef]
- Weitz, R.T.; Harnau, L.; Rauschenbach, S.; Burghard, M.; Kern, K. Polymer nanofibers via nozzle-free centrifugal spinning. Nano Lett. 2008, 8, 1187–1191. [Google Scholar] [CrossRef]
- Zhang, X.; Lu, Y. Centrifugal spinning: An alternative approach to fabricate nanofibers at high speed and low cost. Polym. Rev. 2014, 54, 677–701. [Google Scholar] [CrossRef]
- Luo, C.J.; Stoyanov, S.D.; Stride, E.; Pelan, E.; Edirisinghe, M. Electrospinning versus fibre production methods: From specifics to technological convergence. Chem. Soc. Rev. 2012, 41, 4708–4735. [Google Scholar] [CrossRef] [PubMed]
- Tian, L.; Zhao, C.; Li, J.; Pan, Z. Multi-needle, electrospun, nanofiber filaments: Effects of the needle arrangement on the nanofiber alignment degree and electrostatic field distribution. Text. Res. J. 2014, 85, 621–631. [Google Scholar] [CrossRef]
- Cai, N.; Dai, Q.; Wang, Z.; Luo, X.; Xue, Y.; Yu, F. Preparation and properties of nanodiamond/poly(lactic acid) composite nanofiber scaffolds. Fibers Polym. 2014, 15, 2544–2552. [Google Scholar] [CrossRef]
- Nair, L.S.; Laurencin, C.T. Biodegradable polymers as biomaterials. Prog. Polym. Sci. 2007, 32, 762–798. [Google Scholar] [CrossRef]
- Altun, E.; Aydogdu Mehmet, O.; Koc, F.; Crabbe-Mann, M.; Brako, F.; Kaur-Matharu, R.; Ozen, G.; Kuruca Serap, E.; Edirisinghe, U.; Gunduz, O.; et al. Novel making of bacterial cellulose blended polymeric fiber bandages. Macromol. Mater. Eng. 2018, 303, 1700607. [Google Scholar] [CrossRef]
- Han, G.D.; Ahn, B.C.; Lee, J.-H.; Hwang, Y.; Kim, H.J.; Park, Y.K.; Lee, W.J.; Son, H.K. Optimization of electrospun poly(caprolactone) fiber diameter for vascular scaffolds to maximize smooth muscle cell infiltration and phenotype modulation. Polymers 2019, 11, 643. [Google Scholar] [CrossRef] [PubMed]
- Scaffaro, R.; Maio, A.; Sutera, F.; Gulino, F.E.; Morreale, M. Degradation and recycling of films based on biodegradable polymers: A short review. Polymers 2019, 11, 651. [Google Scholar] [CrossRef]
- Herrero-Herrero, M.; Gómez-Tejedor, J.A.; Vallés-Lluch, A. Pla/pcl electrospun membranes of tailored fibres diameter as drug delivery systems. Eur. Polym. J. 2018, 99, 445–455. [Google Scholar] [CrossRef]
- Vieira, A.C.; Vieira, J.C.; Ferra, J.M.; Magalhães, F.D.; Guedes, R.M.; Marques, A.T. Mechanical study of pla–pcl fibers during in vitro degradation. J. Mech. Behav. Biomed. Mater. 2011, 4, 451–460. [Google Scholar] [CrossRef]
- Klemm, D.; Schumann, D.; Udhardt, U.; Marsch, S. Bacterial synthesized cellulose—Artificial blood vessels for microsurgery. Prog. Polym. Sci. 2001, 26, 1561–1603. [Google Scholar] [CrossRef]
- Kwak, M.H.; Kim, J.E.; Go, J.; Koh, E.K.; Song, S.H.; Son, H.J.; Kim, H.S.; Yun, Y.H.; Jung, Y.J.; Hwang, D.Y. Bacterial cellulose membrane produced by acetobacter sp. A10 for burn wound dressing applications. Carbohydr. Polym. 2015, 122, 387–398. [Google Scholar] [CrossRef] [PubMed]
- Reiniati, I.; Hrymak, A.N.; Margaritis, A. Recent developments in the production and applications of bacterial cellulose fibers and nanocrystals. Crit. Rev. Biotechnol. 2017, 37, 510–524. [Google Scholar] [CrossRef] [PubMed]
- Ding, Q.; Xu, X.; Yue, Y.; Mei, C.; Huang, C.; Jiang, S.; Wu, Q.; Han, J. Nanocellulose-mediated electroconductive self-healing hydrogels with high strength, plasticity, viscoelasticity, stretchability, and biocompatibility toward multifunctional applications. ACS Appl. Mater. Interfaces 2018, 10, 27987–28002. [Google Scholar] [CrossRef] [PubMed]
- Han, J.; Yue, Y.; Wu, Q.; Huang, C.; Pan, H.; Zhan, X.; Mei, C.; Xu, X. Effects of nanocellulose on the structure and properties of poly(vinyl alcohol)-borax hybrid foams. Cellulose 2017, 24, 4433–4448. [Google Scholar] [CrossRef]
- Han, J.; Lu, K.; Yue, Y.; Mei, C.; Huang, C.; Wu, Q.; Xu, X. Nanocellulose-templated assembly of polyaniline in natural rubber-based hybrid elastomers toward flexible electronic conductors. Ind. Crops Prod. 2019, 128, 94–107. [Google Scholar] [CrossRef]
- Nishi, Y.; Uryu, M.; Yamanaka, S.; Watanabe, K.; Kitamura, N.; Iguchi, M.; Mitsuhashi, S. The structure and mechanical properties of sheets prepared from bacterial cellulose. J. Mater. Sci. 1990, 25, 2997–3001. [Google Scholar] [CrossRef]
- Heseltine, P.L.; Ahmed, J.; Edirisinghe, M. Developments in pressurized gyration for the mass production of polymeric fibers. Macromol. Mater. Eng. 2018, 303, 1800218. [Google Scholar] [CrossRef]
- Li, Z.; Yin, X.; Qin, J.; Zhu, L. Preparation and hemocompatibility of electrospun bacteria cellulose sulfate/polyvinyl alcohol nanofibrous composite membrane. IOP Conf. Ser. Mater. Sci. Eng. 2018, 382, 022005. [Google Scholar] [CrossRef]
- Lu, L.; Wu, D.; Zhang, M.; Zhou, W. Fabrication of polylactide/poly(ε-caprolactone) blend fibers by electrospinning: Morphology and orientation. Ind. Eng. Chem. Res. 2012, 51, 3682–3691. [Google Scholar] [CrossRef]
- Ahmed, J.; Matharu, R.K.; Shams, T.; Illangakoon, U.E.; Edirisinghe, M. A comparison of electric-field-driven and pressure-driven fiber generation methods for drug delivery. Macromol. Mater. Eng. 2018, 1700577. [Google Scholar] [CrossRef]
- Huang, Z.-M.; Zhang, Y.Z.; Kotaki, M.; Ramakrishna, S. A review of polymer nanofibers by electrospinning and their applications in nanocomposites. Compos. Sci. Technol. 2003, 63, 2223–2253. [Google Scholar] [CrossRef]
- Christopherson, G.T.; Song, H.; Mao, H.Q. The influence of fiber diameter of electrospun substrates on neural stem cell differentiation and proliferation. Biomaterials 2009, 30, 556–564. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, J.; Altun, E.; Aydogdu, M.O.; Gunduz, O.; Kerai, L.; Ren, G.; Edirisinghe, M. Anti-fungal bandages containing cinnamon extract. Int. Wound J. 2019. [Google Scholar] [CrossRef] [PubMed]
- Mahalingam, S.; Edirisinghe, M. Forming of polymer nanofibers by a pressurised gyration process. Macromol. Rapid Commun. 2013, 34, 1134–1139. [Google Scholar] [CrossRef] [PubMed]
- Bechtel, S.E.; Forest, M.G.; Wang, Q. Non-isothermal modeling of fiber spinning. Am. Soc. Mech. Eng. 1992, 153, 37–48. [Google Scholar]
- Xu, Z.; Mahalingam, S.; Basnett, P.; Raimi-Abraham, B.; Roy, I.; Craig, D.; Edirisinghe, M. Making nonwoven fibrous poly(ε-caprolactone) constructs for antimicrobial and tissue engineering applications by pressurized melt gyration. Macromol. Mater. Eng. 2016, 301, 922–934. [Google Scholar] [CrossRef]
- Eranka Illangakoon, U.; Mahalingam, S.; Wang, K.; Cheong, Y.K.; Canales, E.; Ren, G.G.; Cloutman-Green, E.; Edirisinghe, M.; Ciric, L. Gyrospun antimicrobial nanoparticle loaded fibrous polymeric filters. Mater. Sci. Eng. C 2017, 74, 315–324. [Google Scholar] [CrossRef]
- Beachley, V.; Wen, X. Effect of electrospinning parameters on the nanofiber diameter and length. Mater. Sci. Eng. C 2009, 29, 663–668. [Google Scholar] [CrossRef]
- Zhang, S.; Karaca, B.T.; VanOosten, S.K.; Yuca, E.; Mahalingam, S.; Edirisinghe, M.; Tamerler, C. Coupling infusion and gyration for the nanoscale assembly of functional polymer nanofibers integrated with genetically engineered proteins. Macromol. Rapid Commun. 2015, 36, 1322–1328. [Google Scholar] [CrossRef]
- Hong, X.; Harker, A.; Edirisinghe, M. Process modeling for the fiber diameter of polymer, spun by pressure-coupled infusion gyration. ACS Omega 2018, 3, 5470–5479. [Google Scholar] [CrossRef]
- Vo, P.P.; Doan, N.H.; Kinashi, K.; Sakai, W.; Tsutsumi, N.; Huynh, P.D. Centrifugally spun recycled pet: Processing and characterization. Polymers 2018, 10. [Google Scholar] [CrossRef] [PubMed]
- Yeniay, E.; Öcal, L.; Altun, E.; Giray, B.; Nuzhet Oktar, F.; Talat Inan, A.; Ekren, N.; Kilic, O.; Gunduz, O. Nanofibrous wound dressing material by electrospinning method. Int. J. Polym. Mater. Polym. Biomater. 2019, 68, 11–18. [Google Scholar] [CrossRef]
- Altun, E.; Ekren, N.; Kuruca, S.E.; Gunduz, O. Cell studies on electrohydrodynamic (ehd)-3d-bioprinted bacterial cellulose\polycaprolactone scaffolds for tissue engineering. Mater. Lett. 2019, 234, 163–167. [Google Scholar] [CrossRef]
- Aydogdu, M.O.; Chou, J.; Altun, E.; Ekren, N.; Cakmak, S.; Eroglu, M.; Osman, A.A.; Kutlu, O.; Oner, E.T.; Avsar, G.; et al. Production of the biomimetic small diameter blood vessels for cardiovascular tissue engineering. Int. J. Polym. Mater. Polym. Biomater. 2019, 68, 243–255. [Google Scholar] [CrossRef]
- Surma-Ślusarska, B.; Presler, S.; Danielewicz, D. Characteristics of bacterial cellulose obtained from acetobacter xylinum culture for application in papermaking. In Fibres and Textiles in Eastern Europe; INST Chemical Fibres: Lodz, Poland, 2008; Volume 16, pp. 108–111. [Google Scholar]
- Czaja, W.; Krystynowicz, A.; Bielecki, S.; Brown, R.M., Jr. Microbial cellulose--the natural power to heal wounds. Biomaterials 2006, 27, 145–151. [Google Scholar] [CrossRef] [PubMed]
- Matta, A.K.; Rao, R.U.; Suman, K.N.S.; Rambabu, V. Preparation and characterization of biodegradable pla/pcl polymeric blends. Procedia Mater. Sci. 2014, 6, 1266–1270. [Google Scholar] [CrossRef]
- Abdellah Ali, S.F. Mechanical and thermal properties of promising polymer composites for food packaging applications. IOP Conf. Ser. Mater. Sci. Eng. 2016, 137, 012035. [Google Scholar] [CrossRef]
- Ostafinska, A.; Fortelny, I.; Nevoralova, M.; Hodan, J.; Kredatusova, J.; Slouf, M. Synergistic effects in mechanical properties of pla/pcl blends with optimized composition, processing, and morphology. RSC Adv. 2015, 5, 98971–98982. [Google Scholar] [CrossRef]
- Takayama, T.; Todo, M.; Tsuji, H. Effect of annealing on the mechanical properties of pla/pcl and pla/pcl/lti polymer blends. J. Mech. Behav. Biomed. Mater. 2011, 4, 255–260. [Google Scholar] [CrossRef]
- Krupa, A.; Tomasz Sobczyk, A.; Jaworek, A. Surface properties of plasma-modified poly(vinylidene fluoride) and poly(vinyl chloride) nanofibres. Fibres Text. East. Eur. 2014, 104, 35–39. [Google Scholar]
- Kriel, H.; Sanderson, R.D.; Smit, E. Single polymer composite yarns and films prepared from heat bondable poly(lactic acid) core-shell fibres with submicron fibre diameters. Fibres Text. East. Eur. 2013, 100, 44–47. [Google Scholar]
- Lin, W.-C.; Lien, C.-C.; Yeh, H.-J.; Yu, C.-M.; Hsu, S.-h. Bacterial cellulose and bacterial cellulose–chitosan membranes for wound dressing applications. Carbohydr. Polym. 2013, 94, 603–611. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.-B.; Yu, S.-H.; Mi, F.-L.; Wu, C.-W.; Shyu, S.-S.; Peng, C.-K.; Chao, A.-C. Preparation and characterization on mechanical and antibacterial properties of chitsoan/cellulose blends. Carbohydr. Polym. 2004, 57, 435–440. [Google Scholar] [CrossRef]








| PLA-BC Ratio (wt. ratio) | PCL-BC Ratio (wt. ratio) | PLA-PCL Ratio (wt. ratio) |
|---|---|---|
| 90:10 | 90:10 | 90:10 |
| 80:20 | 80:20 | 80:20 |
| 70:30 | 70:30 | 70:30 |
| 60:40 | 60:40 | 60:40 |
| 50:50 | 50:50 | 50:50 |
| 40:60 | 40:60 | 40:60 |
| 30:70 | 30:70 | 30:70 |
| 20:80 | 20:80 | 20:80 |
| 10:90 | 10:90 | 10:90 |
| Ratio | PLA-BC | PCL-BC | PLA-PCL | |||
|---|---|---|---|---|---|---|
| Viscosity (Pa s) | Surface Tension (mN m−1) | Viscosity (Pa s) | Surface Tension (mN m−1) | Viscosity (Pa s) | Surface Tension (mN m−1) | |
| 100:0 | 363 ± 1 | 52.8 ± 0.1 | 258 ± 1 | 38.4 ± 0.1 | 363 ± 1 | 52.8 ± 0.1 |
| 90:10 | 368 ± 2 | 53.9 ± 0.1 | 269 ± 2 | 39.2 ± 0.2 | 358 ± 1 | 51.3 ± 0.2 |
| 80:20 | 375 ± 2 | 55.2 ± 0.2 | 288 ± 3 | 41.4 ± 0.3 | 351 ± 2 | 50.8 ± 0.2 |
| 70:30 | 403 ± 1 | 57.2 ± 0.3 | 301 ± 1 | 43.3 ± 0.1 | 346 ± 1 | 49.5 ± 0.1 |
| 60:40 | 445 ± 01 | 59.5 ± 0.3 | 342 ± 1 | 46.4 ± 0.1 | 331 ± 2 | 48.2 ± 0.2 |
| 50:50 | 462 ± 2 | 60.1 ± 0.2 | 371 ± 2 | 48.8 ± 0.2 | 321 ± 2 | 47.1 ± 0.1 |
| 40:60 | 481 ± 1 | 61.5 ± 0.3 | 432 ± 2 | 51.3 ± 0.3 | 314 ± 1 | 44.2 ± 0.1 |
| 30:70 | 503 ± 1 | 63.3 ± 0.1 | 476 ± 3 | 54.2 ± 0.3 | 301 ± 3 | 43.8 ± 0.1 |
| 20:80 | 521 ± 2 | 65.5 ± 0.2 | 525 ± 3 | 57.6 ± 0.4 | 295 ± 2 | 41.4 ± 0.2 |
| 10:90 | 544 ± 3 | 67.2 ± 0.1 | 573 ± 2 | 59.4 ± 0.1 | 284 ± 2 | 39.2 ± 0.2 |
| 0:100 | N/A | N/A | N/A | N/A | 258 ± 1 | 38.4 ± 0.1 |
| Ternary Sample: 70:30 (PLA-PCL)-BC Viscosity: 359 ± 2 Pa s Surface Tension: 46.1 ± 0.3 mN m−1 | ||||||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aydogdu, M.O.; Altun, E.; Ahmed, J.; Gunduz, O.; Edirisinghe, M. Fiber Forming Capability of Binary and Ternary Compositions in the Polymer System: Bacterial Cellulose–Polycaprolactone–Polylactic Acid. Polymers 2019, 11, 1148. https://doi.org/10.3390/polym11071148
Aydogdu MO, Altun E, Ahmed J, Gunduz O, Edirisinghe M. Fiber Forming Capability of Binary and Ternary Compositions in the Polymer System: Bacterial Cellulose–Polycaprolactone–Polylactic Acid. Polymers. 2019; 11(7):1148. https://doi.org/10.3390/polym11071148
Chicago/Turabian StyleAydogdu, Mehmet Onur, Esra Altun, Jubair Ahmed, Oguzhan Gunduz, and Mohan Edirisinghe. 2019. "Fiber Forming Capability of Binary and Ternary Compositions in the Polymer System: Bacterial Cellulose–Polycaprolactone–Polylactic Acid" Polymers 11, no. 7: 1148. https://doi.org/10.3390/polym11071148
APA StyleAydogdu, M. O., Altun, E., Ahmed, J., Gunduz, O., & Edirisinghe, M. (2019). Fiber Forming Capability of Binary and Ternary Compositions in the Polymer System: Bacterial Cellulose–Polycaprolactone–Polylactic Acid. Polymers, 11(7), 1148. https://doi.org/10.3390/polym11071148







