New Antibacterial Silver(I) Coordination Polymers Based on a Flexible Ditopic Pyrazolyl-Type Ligand
Abstract
1. Introduction
2. Materials and Methods
2.1. General
- All reagents and solvents were purchased from Sigma-Aldrich (Darmstadt, Germany) and Alfa Aesar (Kandel, Germany), and were used as received.
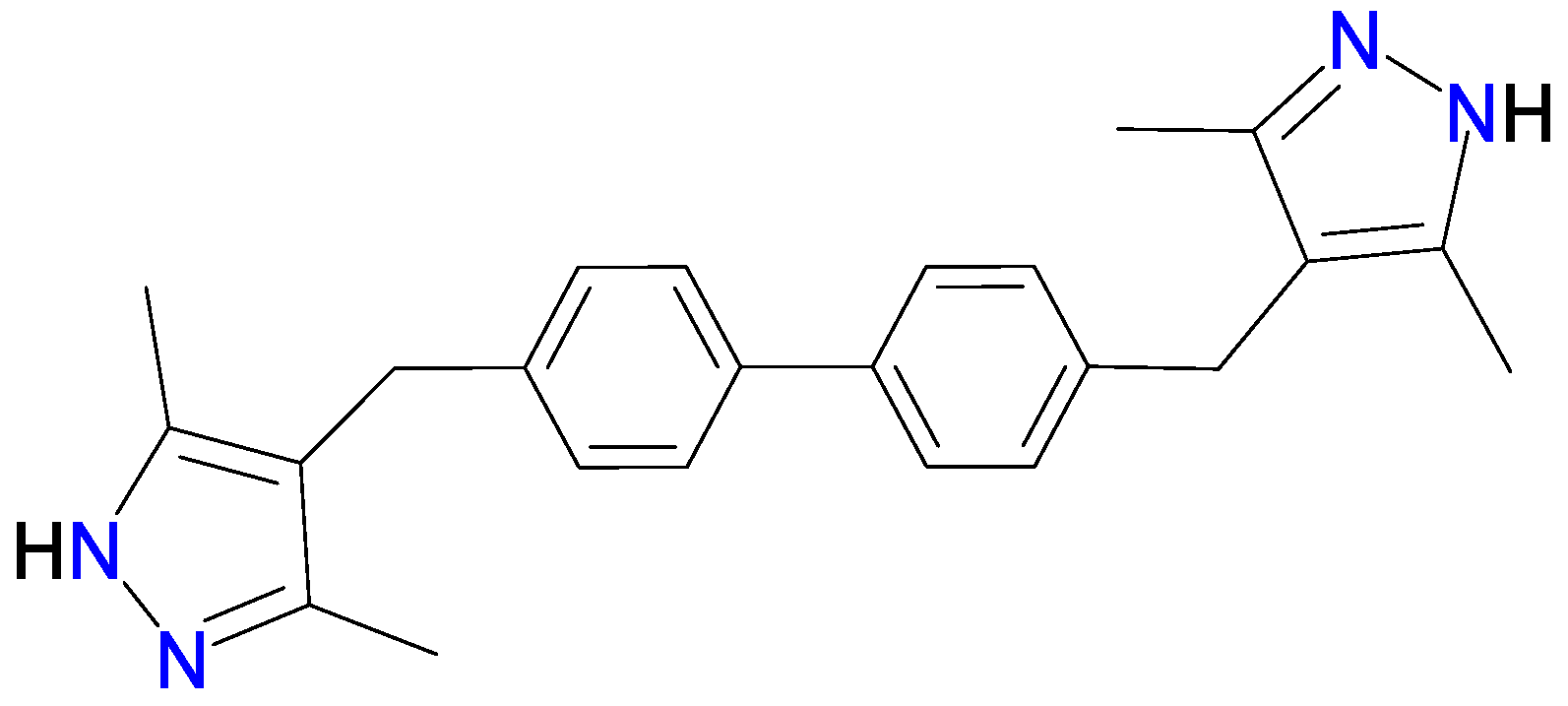
- The ligand 4,4′-bis((3,5-dimethyl-1H-pyrazol-4-yl)methyl)biphenyl (H2DMPMB) was synthesized according to an optimized method previously reported in the literature [60].
- FTIR spectra were recorded from 4000 to 650 cm‒1 with a Perkin–Elmer Spectrum 100 instrument (Perkin-Elmer, Shelton, CT, USA) by total reflectance on a ZnSe crystal. In the following, the IR bands are classified as very weak (vw), weak (w), medium (m), strong (s), and very strong (vs).
- Elemental analyses (C, H, N, and S) were performed in-house with Fisons Instruments 1108 CHNS-O Elemental Analyzer (Thermo Scientific, Waltham, MA, USA). Before performing the analytical characterization, all samples were dried in vacuo (50 °C, ~10‒4 bar) until a constant weight was reached.
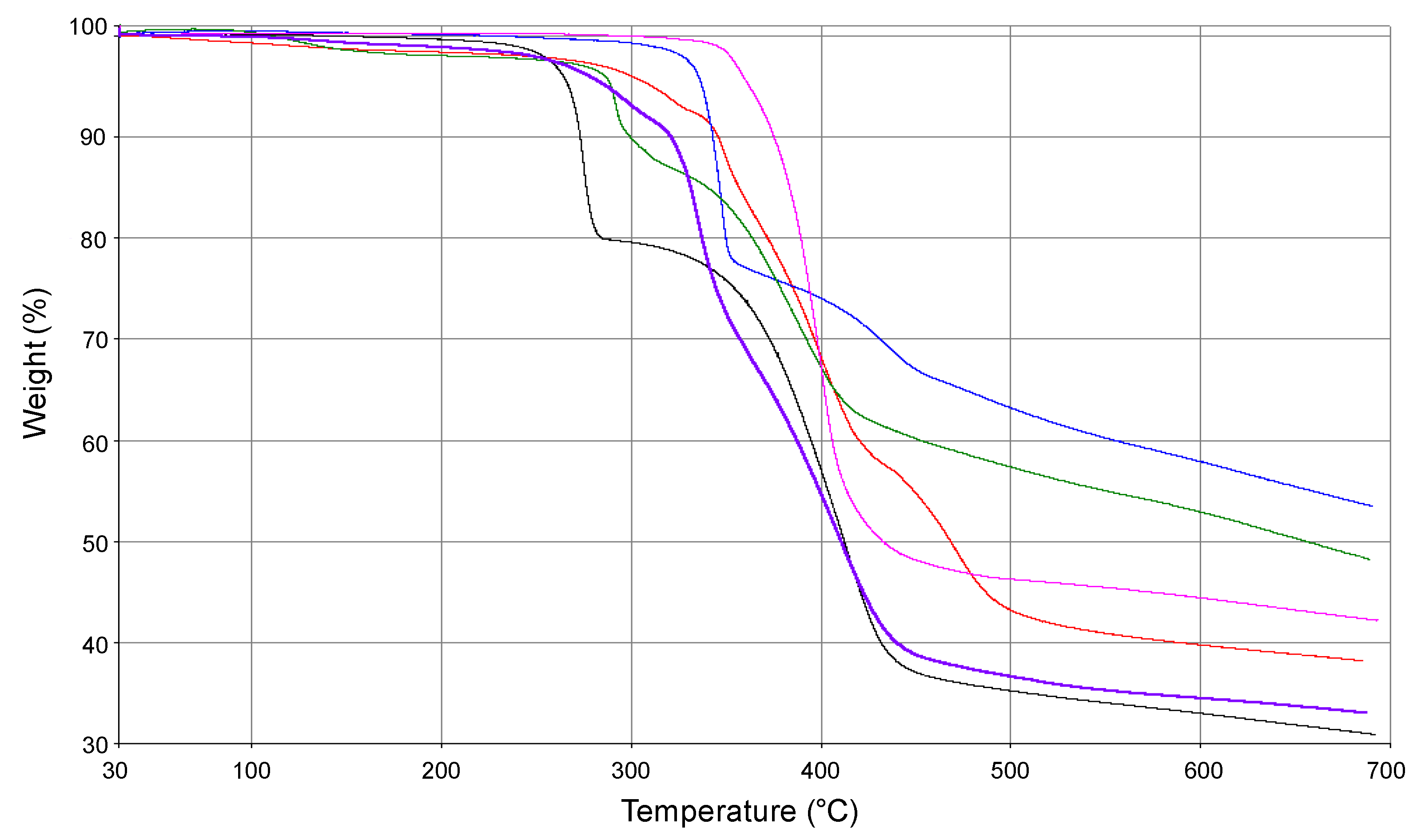
- Termogravimetric analyses (TGA) were carried out from 30 to 700 °C in a N2 stream with a Perkin–Elmer STA 6000 simultaneous thermal analyzer (Perkin-Elmer, Shelton, CT, USA) with the heating rate of 7 °C/min.
2.2. Syntheses
2.3. Bacteria Strain and Growth Conditions
2.4. Antibacterial Activity
3. Results and Discussion
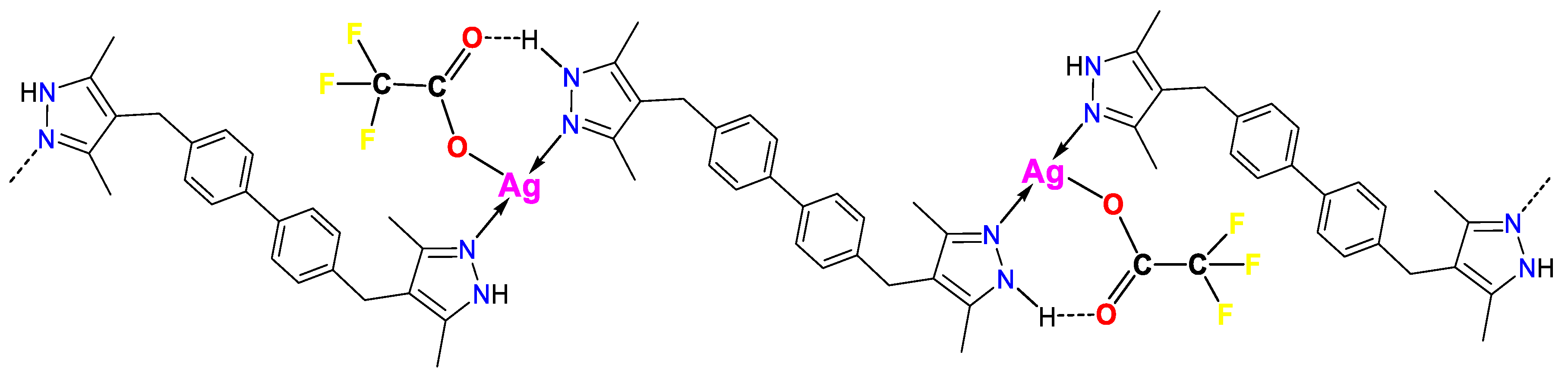
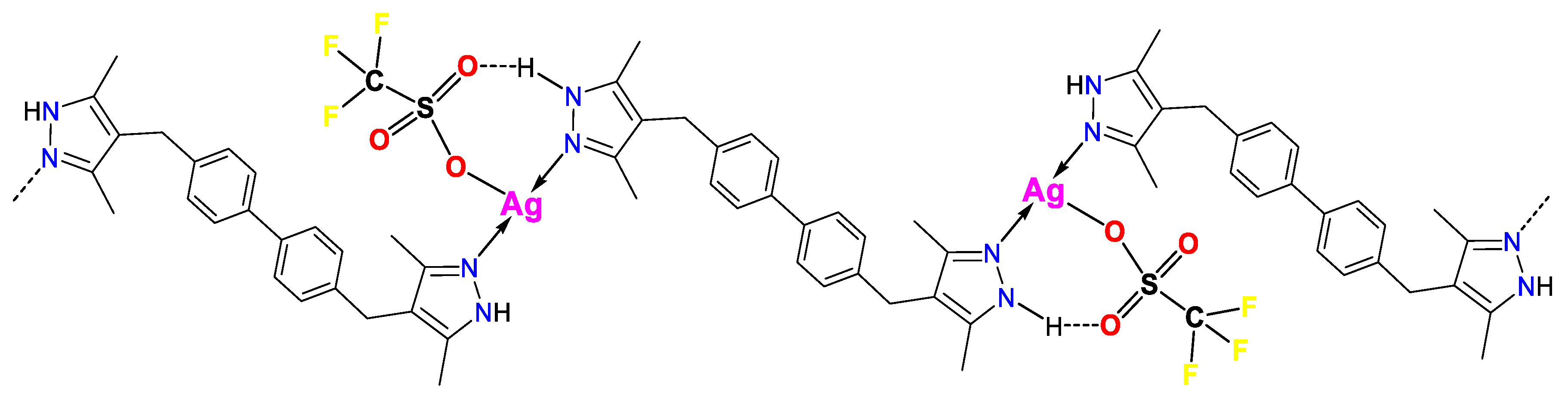
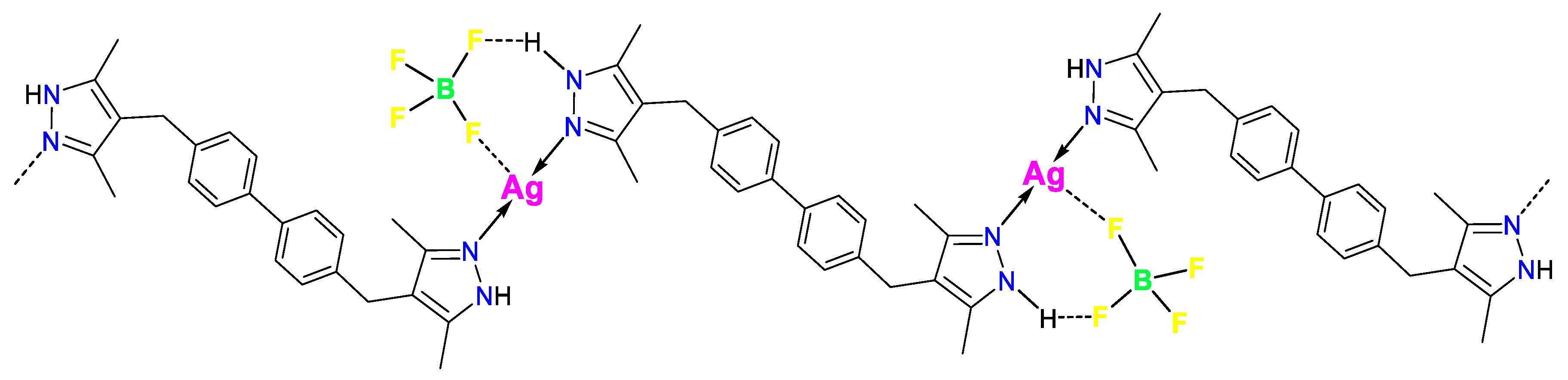
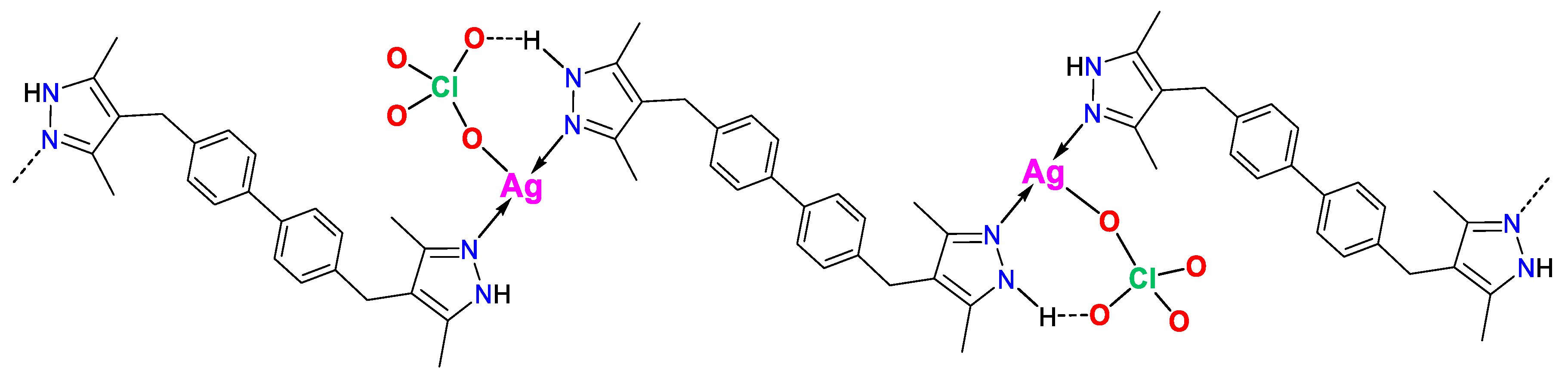
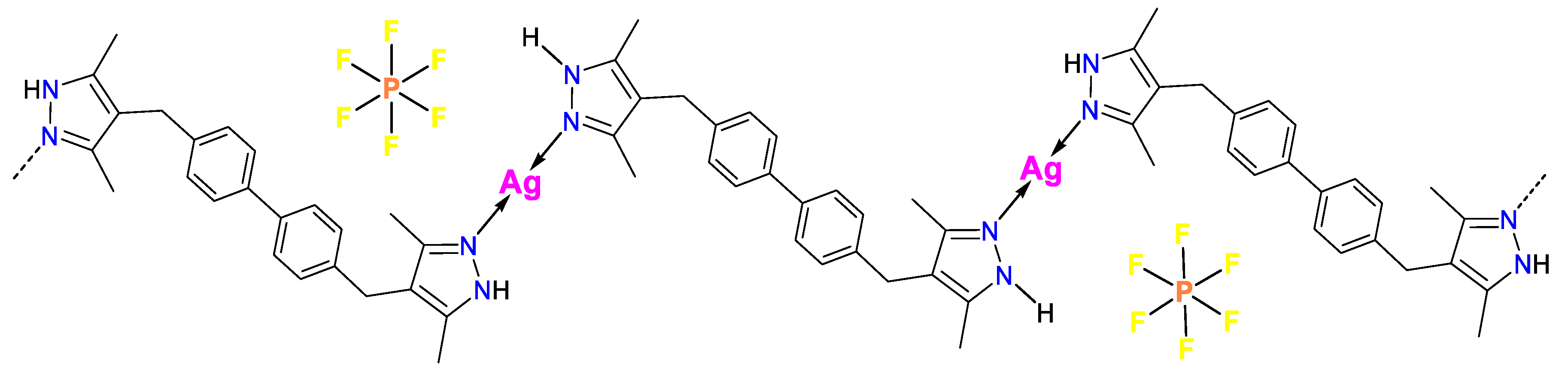
3.1. Synthesis and FTIR Spectroscopy
3.2. Thermogravimetric Analysis
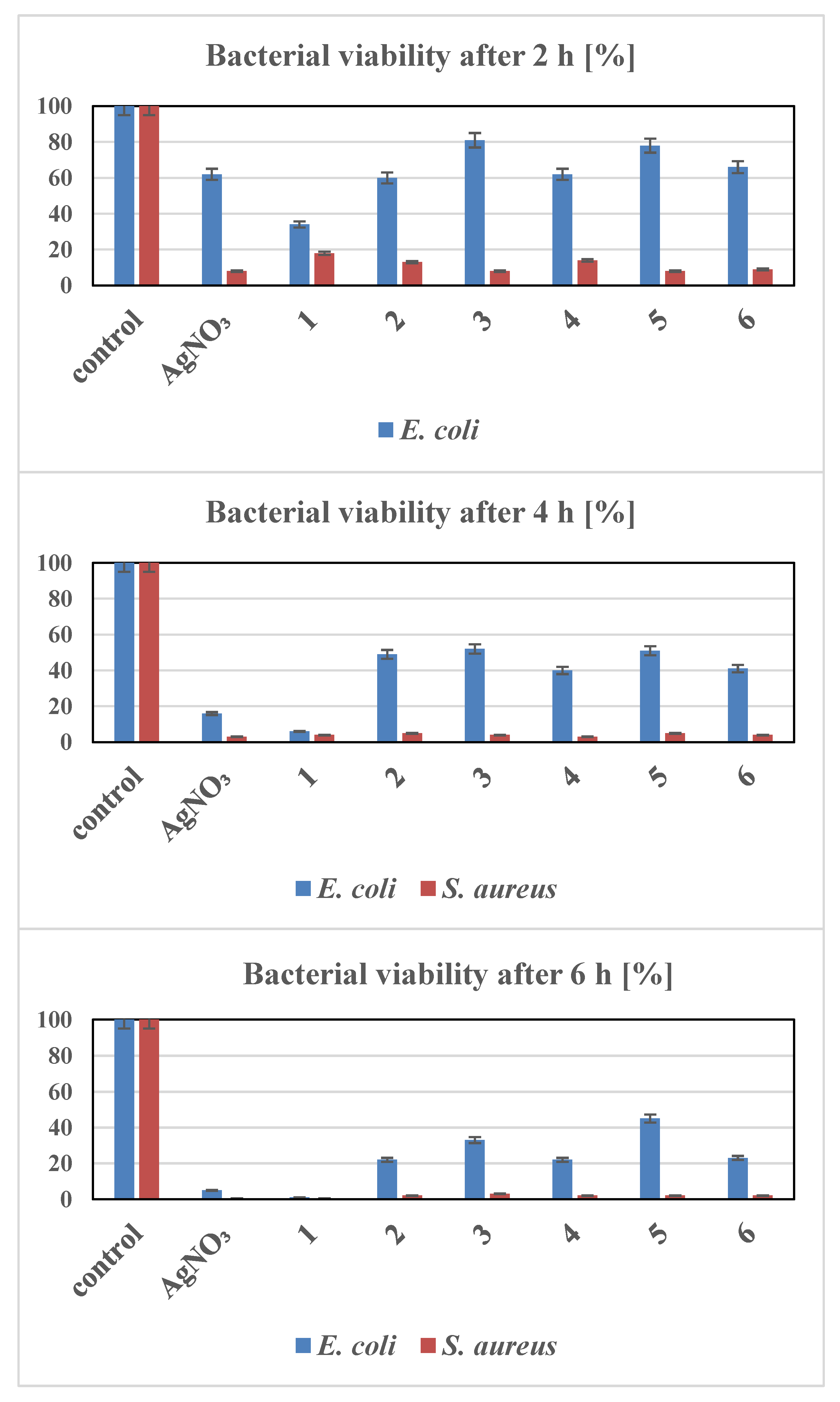
3.3. Antibacterial Activity
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Patel, V. Synthesis, Characterization and Application of Coordination Polymers; Lambert Academic Publishing: Saarbrücken, Germany, 2015. [Google Scholar]
- Batten, S.R.; Neville, S.M.; Turner, D.R. Coordination Polymers: Design, Analysis and Application; Springer: New York, NY, USA, 2010. [Google Scholar]
- Hong, M.-C.; Chen, L. Design and Construction of Coordination Polymers; John Wiley & Sons Inc.: Hoboken, NJ, USA, 2009. [Google Scholar]
- Morsali, A.; Hashemi, L. Main Group Metal Coordination Polymers: Structures and Nanostructures; John Wiley & Sons Inc.: Hoboken, NJ, USA, 2017. [Google Scholar]
- MacGillivray, L.R.; Lukehart, C.M. Metal-Organic Framework Materials; John Wiley & Sons Inc.: Weinheim, Germany, 2014. [Google Scholar]
- Kaskel, S. The Chemistry of Metal-Organic Frameworks: Synthesis, Characterization, and Applications; John Wiley & Sons Inc.: Weinheim, Germany, 2016. [Google Scholar]
- Seyyedi, B. Metal−Organic Frameworks: A New Class of Crystalline Porous Materials; Lambert Academic Publishing: Saarbrücken, Germany, 2014. [Google Scholar]
- Li, H.; Wang, K.; Sun, Y.; Lollar, C.T.; Li, J.; Zhou, H.-C. Recent advances in gas storage and separation using metal-organic frameworks. Mater. Today 2018, 21, 108–121. [Google Scholar] [CrossRef]
- Rogge, S.M.J.; Bavykina, A.; Hajek, J.; Garcia, H.; Olivos-Suarez, A.I.; Sepúlveda-Escribano, A.; Vimont, A.; Clet, G.; Bazin, P.; Kapteijn, F.; et al. Metal-organic frameworks and covalent organic frameworks as single-site catalysts. Chem. Soc. Rev. 2017, 46, 3134–3184. [Google Scholar] [CrossRef] [PubMed]
- Heine, J.; Müller-Buschbaum, K. Engineering metal-based luminescence in coordination polymers and metal-organic frameworks. Chem. Soc. Rev. 2013, 42, 9232–9242. [Google Scholar] [CrossRef]
- Zhang, X.; Wang, W.; Hu, Z.; Wang, G.; Uvdal, K. Coordination polymers for energy transfer: Preparations, properties, sensing applications, and perspectives. Coord. Chem. Rev. 2015, 284, 206–235. [Google Scholar] [CrossRef]
- Mínguez Espallargas, G.; Coronado, E. Magnetic functionalities in MOFs: From the framework to the pore. Chem. Soc. Rev. 2018, 47, 533–557. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.; Campbell, M.G.; Dinca, M. Electrically conductive porous metal-organic frameworks. Angew. Chem. Int. Ed. 2016, 55, 3566–3579. [Google Scholar] [CrossRef] [PubMed]
- Wu, M.-X.; Yang, Y.-W. Metal-organic framework (MOF)-based drug/cargo delivery and cancer therapy. Adv. Mater. 2017, 29, 1606134. [Google Scholar] [CrossRef]
- Janiak, C.; Vieth, J.K. MOFs, MILs and more: Concepts, properties and applications for porous coordination networks (PCNs). New J. Chem. 2010, 34, 2366–2388. [Google Scholar] [CrossRef]
- Robin, A.Y.; Fromm, K.M. Coordination polymer networks with O- and N-donors: What they are, why and how they are made. Coord. Chem. Rev. 2006, 250, 2127–2157. [Google Scholar] [CrossRef]
- Tabacaru, A.; Pettinari, C.; Galli, S. Coordination polymers and metal-organic frameworks built up with poly(tetrazolate) ligands. Coord. Chem. Rev. 2018, 372, 1–30. [Google Scholar] [CrossRef]
- Pettinari, C.; Tabacaru, A.; Galli, S. Coordination polymers and metal-organic frameworks based on poly(pyrazole)-containing ligands. Coord. Chem. Rev. 2016, 307, 1–31. [Google Scholar] [CrossRef]
- Liao, P.-Q.; He, C.-T.; Zhou, D.-D.; Zhang, J.-P.; Chen, X.-M. Porous metal azolate frameworks. In The Chemistry of Metal-Organic Frameworks: Synthesis, Characterization, and Applications; Kaskel, S., Ed.; Wiley: Weinheim, Germany, 2016; Volume 1, pp. 309–343. [Google Scholar]
- Biradha, K.; Sarkar, M.; Rajput, L. Crystal engineering of coordination polymers using 4,4′-bipyridine as a bond between transition metal atoms. Chem. Commun. 2006, 4169–4179. [Google Scholar] [CrossRef] [PubMed]
- Gagnon, K.J.; Perry, H.P.; Clearfield, A. Conventional and unconventional metal-organic frameworks based on phosphonate ligands: MOFs and UMOFs. Chem. Rev. 2012, 112, 1034–1054. [Google Scholar] [CrossRef] [PubMed]
- Su, C.-Y.; Chen, C.-L.; Zhang, J.-Y.; Kang, B.-S. Silver(I) coordination polymers. In Design and Construction of Coordination Polymers; Hong, M.-C., Chen, L., Eds.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2009; pp. 111–144. [Google Scholar]
- Steel, P.J.; Fitchett, C.M. Metallosupramolecular silver(I) assemblies based on pyrazine and related ligands. Coord. Chem. Rev. 2008, 252, 990–1006. [Google Scholar] [CrossRef]
- Mak, T.C.; Zhao, X.-L. Silver: Inorganic and coordination chemistry. In Encyclopedia of Inorganic and Bioinorganic Chemistry; King, R.B., Ed.; John Wiley & Sons, Inc.: Chichester, UK, 2005; pp. 5187–5197. [Google Scholar]
- Khlobystov, A.N.; Blake, A.J.; Champness, N.R.; Lemenovskii, D.A.; Majouga, A.G.; Zyk, N.V.; Schröder, M. Supramolecular design of one-dimensional coordination polymers based on silver(I) complexes of aromatic nitrogen-donor ligands. Coord. Chem. Rev. 2001, 222, 155–192. [Google Scholar] [CrossRef]
- Yan, J.; Wilbraham, L.; Basa, P.N.; Schüttel, M.; Macdonald, J.C.; Ciofini, I.; Coudert, F.-X.; Burdette, S.C. Emissive azobenzenes delivered on a silver coordination polymer. Inorg. Chem. 2018, 57, 15009–15022. [Google Scholar] [CrossRef]
- Huang, R.-W.; Wei, Y.-S.; Dong, X.-Y.; Wu, X.-H.; Du, C.-X.; Zang, S.-Q.; Mak, T.C. Hypersensitive dual-function luminescence switching of a silver-chalcogenolate cluster based metal-organic framework. Nat. Chem. 2017, 9, 689. [Google Scholar] [CrossRef]
- Rana, A.; Jana, S.K.; Pal, T.; Puschmann, H.; Zangrando, E.; Dalai, S. Electrical conductivity and luminescence properties of two silver(I) coordination polymers with heterocyclic nitrogen ligands. J. Solid State Chem. 2014, 216, 49–55. [Google Scholar] [CrossRef]
- Zheng, X.-F.; Zhu, L.-G. Synthesis, structures and conductivity properties of silver 3-sulfobenzoate coordination polymers. Inorg. Chim. Acta 2011, 365, 419–429. [Google Scholar] [CrossRef]
- Yamada, S.; Ishida, T.; Nogami, T. Supramolecular triangular and linear arrays of metal-radical solids using pirazolato-silver(I) motifs. Dalton Trans. 2004, 898–903. [Google Scholar] [CrossRef]
- Rigamonti, L.; Vaccari, M.; Roncaglia, F.; Baschieri, C.; Forni, A. New silver(I) coordination polymer with Fe4 single-molecule magnets as long spacer. Magnetochemistry 2018, 4, 43. [Google Scholar] [CrossRef]
- May, L.J.; Shimizu, G.K.H. Highly selective intercalation of primary amines in a continuous layer Ag coordination network. Chem. Mater. 2005, 17, 217–220. [Google Scholar] [CrossRef]
- Yang, G.; Raptis, R.G. A robust, porous, cationic silver(I) 3,5-diphenyl-1,2,4-triazolate framework with a uninodal 49.66 net. Chem. Commun. 2004, 2058–2059. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Z.; He, C.; Yang, L.; Wang, Y.; Liu, T.; Duan, C. Alkyne activation by a porous silver coordination polymer for heterogeneous catalysis of carbon dioxide cycloaddition. ACS Catal. 2017, 7, 2248–2256. [Google Scholar] [CrossRef]
- Wang, C.C.; Jing, H.P.; Wang, P. Three silver-based complexes constructed from organic carboxylic acid and 4,4′-bipyridine-like ligands: Syntheses, structures and photocatalytic properties. J. Mol. Struct. 2014, 1074, 92–99. [Google Scholar] [CrossRef]
- Blake, A.J.; Champness, N.R.; Cooke, P.A.; Nicolson, J.E.B. Synthesis of a chiral adamantoid network – the role of solvent in the construction of new coordination netwroks with silver(I). Chem. Commun. 2000, 665–666. [Google Scholar] [CrossRef]
- Feazell, R.P.; Carson, C.R.; Klausmeyer, K.K. Silver(I) 3-aminomethylpyridine complexes, Part 1: Effect of ligand ration, π-stacking, and temperature with a noninteracting anion. Inorg. Chem. 2006, 45, 2627–2634. [Google Scholar] [CrossRef]
- Marchetti, F.; Pettinari, R.; Di Nicola, C.; Pettinari, C.; Paul, A.; Crispini, A.; Giorno, E.; Lelj, F.; Stoia, S.; Amati, M. Effect of methyl groups in a pyrimidine-based flexible ligand on the formation of silver(I) coordination networks. New J. Chem. 2018, 42, 13998–14008. [Google Scholar] [CrossRef]
- Patra, G.K.; Goldberg, I.; De, S.; Datta, D. Effect of the size of discrete anions on the nuclearity of a complex cation. CrystEngComm 2007, 9, 828–832. [Google Scholar] [CrossRef]
- Medici, S.; Peana, M.; Crisponi, G.; Nurchi, V.M.; Lachowicz, J.I.; Remelli, M.; Zoroddu, M.A. Silver coordination compounds: A new horizon in medicine. Coord. Chem. Rev. 2016, 327, 349–359. [Google Scholar] [CrossRef]
- Nomyia, K.; Tsuda, K.; Sudoh, T.; Oda, M. Ag(I)‒N bond-containing compound showing wide spectra in effective antimicrobial activities: Polymeric silver(I) imidazolate. J. Inorg. Biochem. 1997, 68, 39–44. [Google Scholar] [CrossRef]
- Nomiya, K.; Tsuda, K.; Kasuga, N.C. Synthesis and X-ray characterization of helical polymer complexes [Ag (1,2,3-L)(PPh3)2]n and [Ag(1,2,4-L)(PPh3)2]n (HL = triazole) and their antimicrobial activities. J. Chem. Soc. Dalton Trans. 1998, 1653–1659. [Google Scholar] [CrossRef]
- Nomiya, K.; Noguchi, R.; Oda, M. Complexes with tetrazole (Htetz) and triphenylphosphine ligands, and their antimicrobial activities. A helical polymer of silver (I) complex [Ag (tetz)(PPh3)2]n and a monomeric gold (I) complex [Au(tetz)(PPh3)]. Inorg. Chim. Acta 2000, 298, 24–32. [Google Scholar] [CrossRef]
- Rogovoy, M.I.; Berezin, A.S.; Kozlova, Y.N.; Samsonenko, D.G.; Artem’ev, A.V. A layered Ag(I)-based coordination polymer showing sky-blue luminescence and antibacterial activity. Inorg. Chem. Commun. 2019, 108, 107513. [Google Scholar] [CrossRef]
- Liu, A.; Wang, C.C.; Wang, C.Z.; Fu, H.-F.; Peng, W.; Cao, Y.-L.; Chu, H.-Y.; Du, A.-F. Selective adsorption activities toward organic dyes and antibacterial performance of silver based coordination polymers. J. Colloid Interface Sci. 2018, 512, 730–739. [Google Scholar] [CrossRef]
- Marchetti, F.; Palmucci, J.; Pettinari, C.; Pettinari, R.; Scuri, S.; Grappasonni, I.; Cocchioni, M.; Amati, M.; Lelj, F.; Crispini, A. Linkage isomerism in silver acylpyrazolonato complexes and correlation with their antibacterial activity. Inorg. Chem. 2016, 55, 5453–5466. [Google Scholar] [CrossRef]
- Alisir, S.H.; Demir, S.; Sariboga, B.; Buyukgungor, O. A disparate 3-D silver(I) coordination polymer of pyridine-3,5-dicarboxylate and pyrimidine with strong intermetallic interactions: X-ray crystallography, photoluminescence and antimicrobial activity. J. Coord. Chem. 2015, 68, 155–168. [Google Scholar] [CrossRef]
- Lu, X.; Ye, J.; Sun, Y.; Bogale, R.F.; Zhao, L.; Tian, P.; Ning, G. Ligand effects on the structural dimensionality and antibacterial activities of silver-based coordination polymers. Dalton Trans. 2014, 43, 10104–10113. [Google Scholar] [CrossRef]
- Jaros, S.W.; Smoleński, P.; Guedes da Silva, M.F.C.; Florek, M.; Król, J.; Staroniewicz, Z.; Pombeiro, A.J.L.; Kirillov, A.M. New silver BioMOFs driven by 1,3,5-triaza-7- phosphaadamantane-7-sulfide (PTALS): Synthesis, topological analysis and antimicrobial activity. CrystEngComm 2013, 15, 8060–8064. [Google Scholar] [CrossRef]
- Lotlikar, S.R.; Gallaway, E.; Grant, T.; Popis, S.; Whited, M.; Guragain, M.; Rogers, R.; Hamilton, S.; Gerasimchuk, N.G.; Patrauchan, M.A. Polymeric composites with silver(I) cyanoximates inhibit biofilm formation of Gram-positive and Gram-negative bacteria. Polymers 2019, 11, 1018. [Google Scholar] [CrossRef]
- Girard, J.; Joset, N.; Crochet, A.; Tan, M.; Holzheu, A.; Brunetto, P.S.; From, K.M. Synthesis of new polyether ether ketone derivatives with silver binding site and coordination compounds of their monomers with different silver salts. Polymers 2016, 8, 208. [Google Scholar] [CrossRef] [PubMed]
- Marchetti, F.; Palmucci, J.; Pettinari, C.; Pettinari, R.; Marangoni, M.; Ferraro, S.; Giovannetti, R.; Scuri, S.; Grappasonni, I.; Cocchioni, M.; et al. Preparation of polyethylene composites containing silver(I) acylpyrazolonato additives and SAR investigation of their antibacterial activity. ACS Appl. Mater. Interfaces 2016, 8, 29676–29687. [Google Scholar] [CrossRef] [PubMed]
- Marchetti, F.; Palmucci, J.; Pettinari, C.; Pettinari, R.; Condello, F.; Ferraro, S.; Marangoni, M.; Crispini, A.; Scuri, S.; Grappasonni, I.; et al. Novel composite plastics containing silver(I) acylpyrazolonato additives display potent antimicrobial activity by contact. Chem. Eur. J. 2015, 21, 836–850. [Google Scholar] [CrossRef] [PubMed]
- Tabacaru, A.; Pettinari, C.; Marchetti, F.; Di Nicola, C.; Domasevitch, K.V.; Galli, S.; Masciocchi, N.; Scuri, S.; Grappasonni, I.; Cocchioni, M. Antibacterial action of 4,4′-bipyrazolyl-based silver(I) coordination polymers embedded in PE disks. Inorg. Chem. 2012, 51, 9775–9788. [Google Scholar] [CrossRef] [PubMed]
- Brunetto, P.S.; Slenters, T.V.; Fromm, K.M. In vitro biocompatibility of new silver(I) coordination compound coated-surfaces for dental implant applications. Materials 2011, 4, 355–367. [Google Scholar] [CrossRef] [PubMed]
- Slenters, T.V.; Sagué, J.L.; Brunetto, P.S.; Zuber, S.; Fleury, A.; Mirolo, L.; Robin, A.Y.; Meuwly, M.; Gordon, O.; Landmann, R.; et al. Of chains and rings: Synthetic strategies and theoretical investigations for tuning the structure of silver coordination compounds and their applications. Materials 2010, 3, 3407–3429. [Google Scholar] [CrossRef]
- Gordon, O.; Slenters, T.V.; Brunetto, P.S.; Villaruz, A.E.; Sturdevant, D.E.; Otto, M.; Landmann, R.; Fromm, K.M. Silver coordination polymers for prevention of implant infection: Thiol interaction, impact on respiratory chain enzymes, and hydroxyl radical induction. Antimicrob. Agents Chemother. 2010, 54, 4208–4218. [Google Scholar] [CrossRef]
- Brunetto, P.S.; Fromm, K.M. Antimicrobial coatings for implant surfaces. Chimia 2008, 62, 249–252. [Google Scholar] [CrossRef]
- Slenters, T.V.; Hauser-Gerspach, I.; Daniels, A.U.; Fromm, K.M. Silver coordination compounds as light-stable, nano-structured and anti-bacterial coatings for dental implant and restorative materials. J. Mater. Chem. 2008, 18, 5359–5362. [Google Scholar] [CrossRef]
- Tabacaru, A.; Pettinari, C.; Marchetti, F.; Galli, S.; Masciocchi, N. Bis(pyrazolato)-based metal-organic frameworks fabricated with 4,4′-bis((3,5-dimethyl-1H-pyrazol-4-yl)methyl)biphenyl and late transition metals. Cryst. Growth Des. 2014, 14, 3142–3152. [Google Scholar] [CrossRef]
- Tabacaru, A.; Pettinari, C.; Masciocchi, N.; Galli, S.; Marchetti, F.; Angjellari, M. Pro-porous coordination polymers of the 1,4-bis((3,5-dimethyl-1Hpyrazol-4-yl)-methyl)benzene ligand with late transition metals. Inorg. Chem. 2011, 50, 11506–11513. [Google Scholar] [CrossRef] [PubMed]
- Duan, P.-C.; Huang, Z.-D.; Zhang, F.-F.; Yang, G. Synthesis and crystal structure of copper(II) and silver(I) complexes of a biphenyl-bridged bipyrazolyl ligand. Transition Met. Chem. 2012, 37, 595–600. [Google Scholar] [CrossRef]
- Semitut, E.; Komarov, V.; Sukhikh, T.; Filatov, E.; Potapov, A. Synthesis, crystal structure and thermal stability of 1D linear silver(I) coordination polymers with 1,1,2,2-tetra(pyrazol-1-yl)ethane. Crystals 2016, 6, 138. [Google Scholar] [CrossRef]
- Moussa, S.H.; Tayel, A.A.; Al-Hassan, A.A.; Farouk, A. Tetrazolium/formazan test as an efficient method to determine fungal chitosan antimicrobial activity. J. Mycol. 2013, 2013, 753692. [Google Scholar] [CrossRef]
- Boldog, I.; Rusanov, E.B.; Chernega, A.N.; Sieler, J.; Domasevitch, K.V. One- and two-dimensional coordination polymers of 3,3′,5,5′-tetramethyl-4,4′-bipyrazolyl, a new perspective crystal engineering module. Polyhedron 2001, 20, 887–897. [Google Scholar] [CrossRef]
- Domasevitch, K.V.; Boldog, I.; Rusanov, E.B.; Hunger, J.; Blaurock, S.; Schöder, M.; Sieler, J. Helical bipyrazole networks conditioned by hydrothermal crystallization. Z. Anorg. Allg. Chem. 2005, 631, 1095–1100. [Google Scholar] [CrossRef]
- Banditelli, G.; Bandini, A.L.; Bonati, F.; Minghetti, G. Carbonylrhodium(I) derivatives of 3,5-dimethyl- and 3,5-bis(trifluoromethyl)pyrazole. J. Organomet. Chem. 1981, 218, 229–239. [Google Scholar] [CrossRef]
- Lever, A.B.P.; Mantovani, E.; Ramaswamy, B.S. Infrared combination frequencies in coordination complexes containing nitrate groups in various coordination environments. A probe for the metal–nitrate interaction. Can. J. Chem. 1971, 49, 1957–1964. [Google Scholar] [CrossRef]
- Nakamoto, K. Infrared and Raman Spectra of Inorganic and Coordination Compounds, Part A, sixth ed.; Wiley: Hoboken, NJ, USA, 2009. [Google Scholar]
- Effendy; Marchetti, F.; Pettinari, C.; Pettinari, R.; Ricciutelli, M.; Skelton, B.W.; White, A.H. (Bis(1,2,4-triazol-1-yl)methane)silver(I) phosphino complexes: Structures and spectroscopic properties of mixed-ligand coordination polymers. Inorg. Chem. 2004, 43, 2157–2165. [Google Scholar] [CrossRef]
- Nakamoto, K. Infrared and Raman Spectra of Inorganic and Coordination Compounds, Part B, sixth ed.; Wiley: Hoboken, NJ, USA, 2009. [Google Scholar]
- Burchell, T.J.; Puddephatt, R.J. Homochiral and heterochiral coordination polymers and networks of silver(I). Inorg. Chem. 2006, 45, 650–659. [Google Scholar] [CrossRef]
- Christe, K.O.; Naumann, D. Vibrational spectra of trifluoroacetates. Spectrochim. Acta A 1973, 29, 2017–2024. [Google Scholar] [CrossRef]
- Miles, M.G.; Doyle, G.; Cooney, R.P.; Tobias, R.S. Raman and infrared spectra and normal coordinates of the trifluoromethanesulfonate and trichloromethanesulfonate anions. Spectrochim. Acta A 1969, 25, 1515–1526. [Google Scholar] [CrossRef]
- Bala, R.; Sharma, R.P.; Bond, A.D. Second-sphere coordination in hexaamminecobalt(III) salt with organic sulphonate anions: Synthesis, characterization and X-ray crystal structure of [Co(NH3)6](CH3SO3)3. J. Mol. Struct. 2007, 830, 198–203. [Google Scholar] [CrossRef]
- Rosenthal, M.R. The mith of the non-coordinating anion. J. Chem. Educ. 1973, 50, 331–334. [Google Scholar] [CrossRef]
- Awaleh, M.P.; Badia, A.; Brisse, F. Coordination networks with flexible ligands based on silver(I) salts: Complexes of 1,3-bis(phenylthio)propane with silver(I) salts of PF6-, CF3COO-, CF3CF2COO-, CF3CF2CF2COO-, p-TsO-, and CF3SO3-. Inorg. Chem. 2005, 44, 7833–7845. [Google Scholar] [CrossRef]
- Zhang, J.-P.; Kitagawa, S. Supramolecular isomerism, framework flexibility, unsaturated metal center, and porous property of Ag(I)/Cu(I) 3,3′,5,5′-tetramethyl-4,4′-bipyrazolate. J. Am. Chem. Soc. 2008, 130, 907–917. [Google Scholar] [CrossRef]
- Masciocchi, N.; Moret, M.; Cairati, P.; Sironi, A.; Ardizzoia, G.A.; La Monica, G. The multiphase nature of the Cu(pz) and Ag(pz) (Hpz = pyrazole) systems: Selective synthesis and ab-initio X-ray powder diffraction structural characterization of copper(I) and silver(I) pyrazolates. J. Am. Chem. Soc. 1994, 116, 7668–7676. [Google Scholar] [CrossRef]
- Abate, G.; Aseffa, A.; Selassie, A.; Goshu, S.; Fekade, B.; WoldeMeskal, D.; Miörner, H. Direct colorimetric assay for rapid detection of rifampin-resistant Mycobacterium tuberculosis. J. Clin. Microbiol. 2004, 42, 871–873. [Google Scholar] [CrossRef]
- Thompson, N.C. Silver. In Comprehensive Inorganic Chemistry; Bailey, J.C., Ed.; Pergamon Press: Oxford, UK, 1973; Volume 3, pp. 79–128. [Google Scholar]
- Nawaz, M.; Han, M.Y.; Kim, T.; Manzoor, U.; Amin, M.T. Silver disinfection of Pseudomonas aeruginosa and E. coli in rooftop harvested rainwater for potable purposes. Sci. Total Environ. 2012, 431, 20–25. [Google Scholar] [CrossRef]
- Jung, W.K.; Koo, H.C.; Kim, K.W.; Shin, S.; Kim, S.H.; Park, Y.H. Antibacterial activity and mechanism of action of the silver ion in Staphylococcus aureus and Escherichia coli. Appl. Environ. Microbiol. 2008, 74, 2171–2178. [Google Scholar] [CrossRef]




© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tăbăcaru, A.; Pettinari, C.; Bușilă, M.; Dinică, R.M. New Antibacterial Silver(I) Coordination Polymers Based on a Flexible Ditopic Pyrazolyl-Type Ligand. Polymers 2019, 11, 1686. https://doi.org/10.3390/polym11101686
Tăbăcaru A, Pettinari C, Bușilă M, Dinică RM. New Antibacterial Silver(I) Coordination Polymers Based on a Flexible Ditopic Pyrazolyl-Type Ligand. Polymers. 2019; 11(10):1686. https://doi.org/10.3390/polym11101686
Chicago/Turabian StyleTăbăcaru, Aurel, Claudio Pettinari, Mariana Bușilă, and Rodica Mihaela Dinică. 2019. "New Antibacterial Silver(I) Coordination Polymers Based on a Flexible Ditopic Pyrazolyl-Type Ligand" Polymers 11, no. 10: 1686. https://doi.org/10.3390/polym11101686
APA StyleTăbăcaru, A., Pettinari, C., Bușilă, M., & Dinică, R. M. (2019). New Antibacterial Silver(I) Coordination Polymers Based on a Flexible Ditopic Pyrazolyl-Type Ligand. Polymers, 11(10), 1686. https://doi.org/10.3390/polym11101686