Novel Dual-Component Microencapsulated Hydrophobic Amine and Microencapsulated Isocyanate Used for Self-Healing Anti-Corrosion Coating
Abstract
:1. Introduction
2. Experimental
2.1. Materials
2.2. Microcapsules Preparation
2.2.1. Fabrication of MF Prepolymer
2.2.2. Preparation of IPDI Microcapsules
2.2.3. Microencapsulation of Amine
2.3. Preparation of Dual-Component Self-Healing Coating
2.4. Characterization
3. Results and Discussion
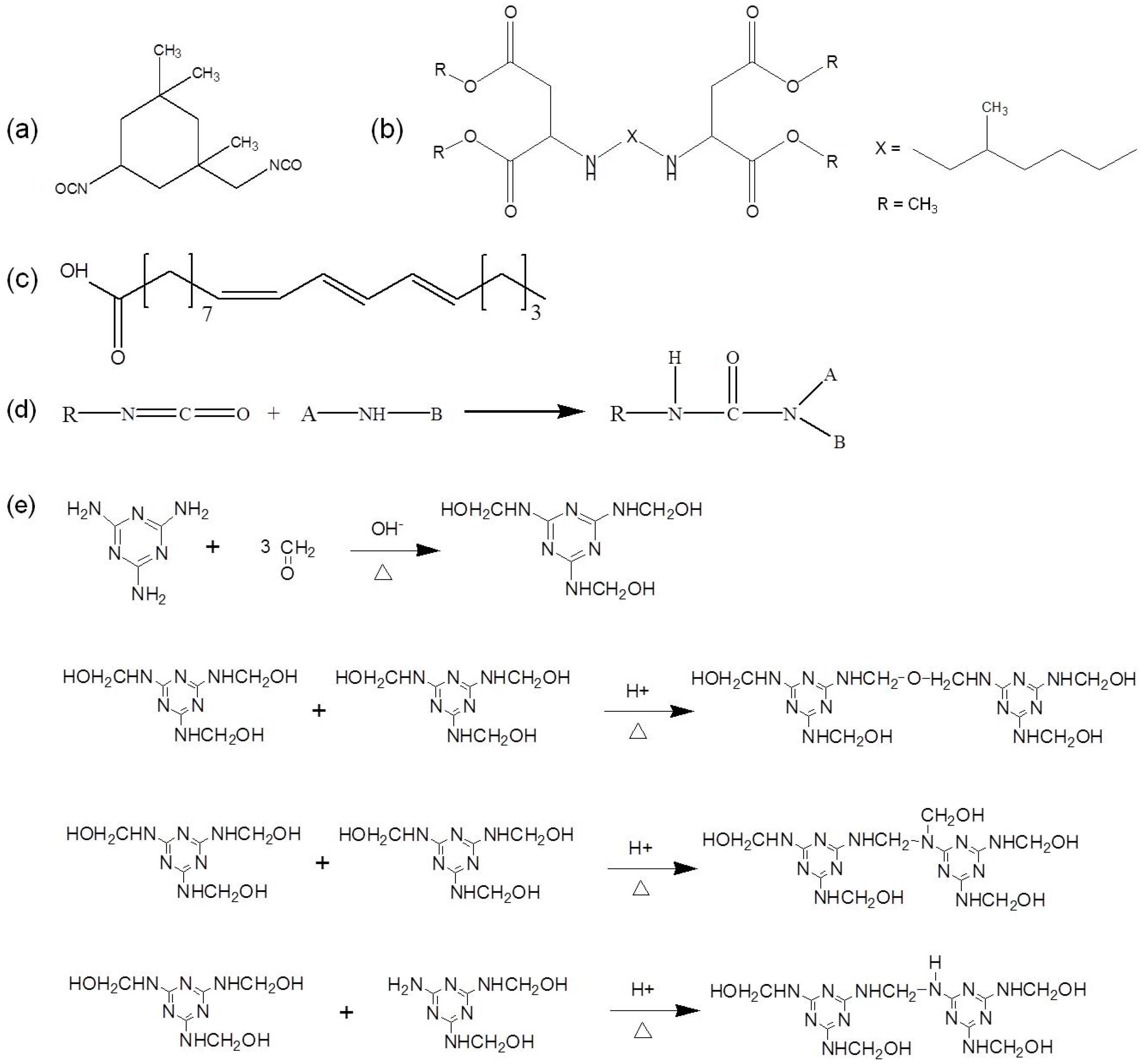
3.1. Self-Healing Agent and Microencapsulation Mechanism
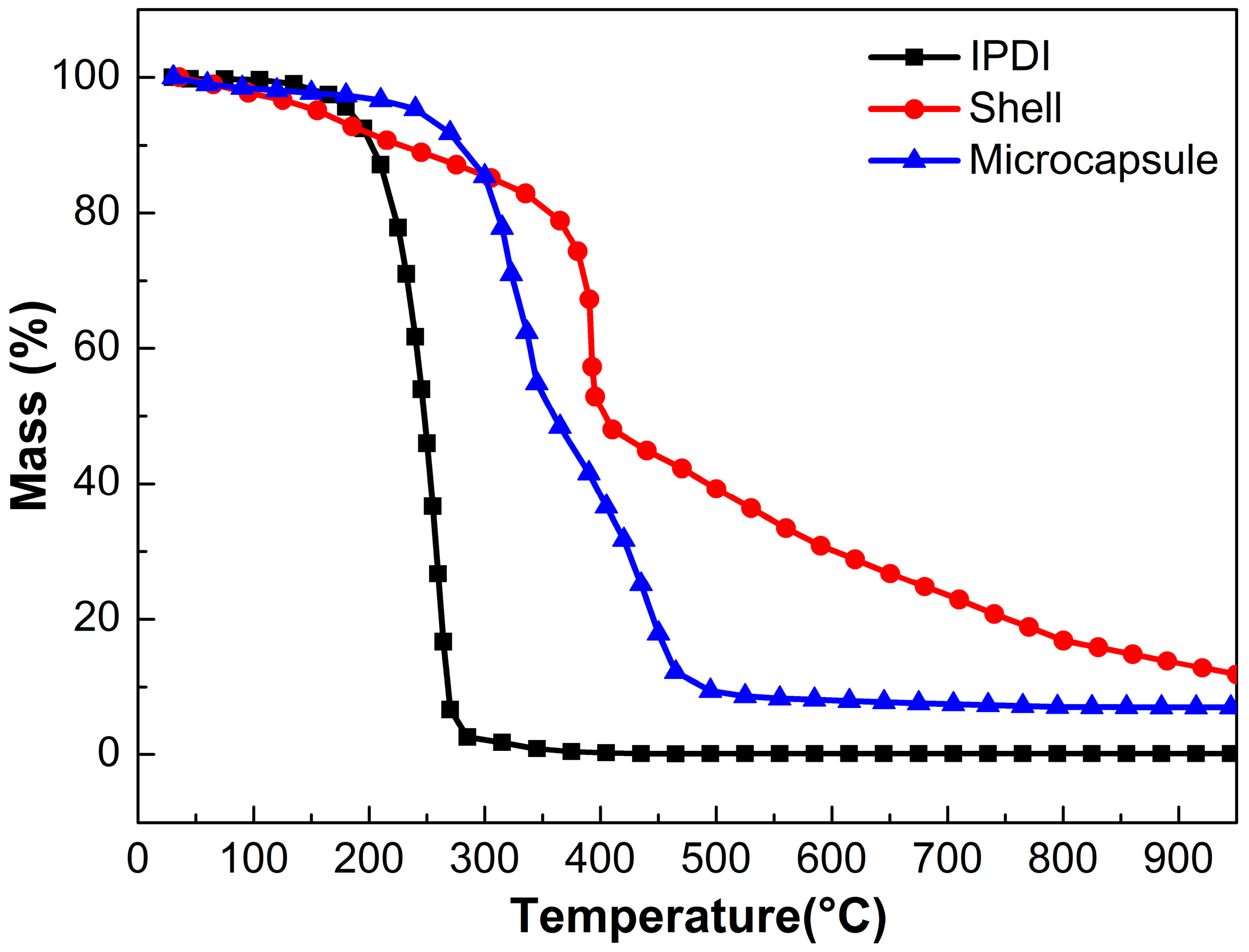
3.2. Microencapsulation of IPDI
3.3. Microencapsulation of Hydrophobic PAE-TO
3.4. Performance of Self-Healing Coating Containing Dual-Component Microcapsule
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Huang, M.; Zhang, H.; Yang, J. Synthesis of organic silane microcapsules for self-healing corrosion resistant polymer coatings. Corros. Sci. 2012, 65, 561–566. [Google Scholar] [CrossRef]
- Behzadnasab, M.; Mirabedini, S.M.; Esfandeh, M.; Farnood, R.R. Evaluation of corrosion performance of a self-healing epoxy-based coating containing linseed oil-filled microcapsules via electrochemical impedance spectroscopy. Prog. Org. Coat. 2017, 105, 212–224. [Google Scholar] [CrossRef]
- Liu, Z.F.; Liu, H.S.; Zhang, L.H.; Yan, M.F. Study on performance of scale and corrosion inhibition of polyaspartic acid composite material. Adv. Mater. Res. 2011, 233–235, 50–53. [Google Scholar] [CrossRef]
- Sun, D.; Zhang, H.; Tang, X.-Z.; Yang, J. Water resistant reactive microcapsules for self-healing coatings in harsh environments. Polymer 2016, 91, 33–40. [Google Scholar] [CrossRef]
- Zhu, D.Y.; Rong, M.Z.; Zhang, M.Q. Self-healing polymeric materials based on microencapsulated healing agents: From design to preparation. Prog. Polym. Sci. 2015, 49–50, 175–220. [Google Scholar] [CrossRef]
- White, S.R.; Sottos, N.R.; Geubelle, P.H.; Moore, J.S.; Kessler, M.R.; Sriram, S.R. Autonomic healing of polymer composites. Nature 2001, 409, 794–797. [Google Scholar] [CrossRef] [PubMed]
- Gao, L.; He, J.; Hu, J.; Wang, C. Photoresponsive self-healing polymer composite with photoabsorbing hybrid microcapsules. ACS Appl. Mater. Interfaces 2015, 7, 25546–25552. [Google Scholar] [CrossRef] [PubMed]
- Hillewaere, X.K.D.; Du Prez, F.E. Fifteen chemistries for autonomous external self-healing polymers and composites. Prog. Polym. Sci. 2015, 49–50, 121–153. [Google Scholar] [CrossRef]
- Yang, Y.; Ding, X.; Urban, M.W. Chemical and physical aspects of self-healing materials. Prog. Polym. Sci. 2015, 49–50, 34–59. [Google Scholar] [CrossRef]
- He, Z.; Jiang, S.; Li, Q.; Wang, J.; Zhao, Y.; Kang, M. Facile and cost-effective synthesis of isocyanate microcapsules via polyvinyl alcohol-mediated interfacial polymerization and their application in self-healing materials. Compos. Sci. Technol. 2017, 138, 15–23. [Google Scholar] [CrossRef]
- Wang, W.; Xu, L.; Liu, F.; Li, X.; Xing, L. Synthesis of isocyanate microcapsules and micromechanical behavior improvement of microcapsule shells by oxygen plasma treated carbon nanotubes. J. Mater. Chem. A 2013, 1, 776–782. [Google Scholar] [CrossRef]
- Yi, H.; Yang, Y.; Gu, X.; Huang, J.; Wang, C. Multilayer composite microcapsules synthesized by pickering emulsion templates and their application in self-healing coating. J. Mater. Chem. A 2015, 3, 13749–13757. [Google Scholar] [CrossRef]
- Huang, M.; Yang, J. Facile microencapsulation of HDI for self-healing anticorrosion coatings. J. Mater. Chem. 2011, 21, 11123–11130. [Google Scholar] [CrossRef]
- Sun, D.; An, J.; Wu, G.; Yang, J. Double-layered reactive microcapsules with excellent thermal and non-polar solvent resistance for self-healing coatings. J. Mater. Chem. A 2015, 3, 4435–4444. [Google Scholar] [CrossRef]
- Wu, G.; An, J.; Tang, X.-Z.; Xiang, Y.; Yang, J. A versatile approach towards multifunctional robust microcapsules with tunable, restorable, and solvent-proof superhydrophobicity for self-healing and self-cleaning coatings. Adv. Funct. Mater. 2014, 24, 6751–6761. [Google Scholar] [CrossRef]
- Nguyen, L.-T.T.; Hillewaere, X.K.D.; Teixeira, R.F.A.; van den Berg, O.; Du Prez, F.E. Efficient microencapsulation of a liquid isocyanate with in situ shell functionalization. Polym. Chem. 2015, 6, 1159–1170. [Google Scholar] [CrossRef]
- Ma, Y.; Jiang, Y.; Tan, H.; Zhang, Y.; Gu, J. A rapid and efficient route to preparation of isocyanate microcapsules. Polymers 2017, 9, 274. [Google Scholar] [CrossRef]
- Yang, J.L.; Keller, M.W.; Moore, J.S.; White, S.R.; Sottos, N.R. Microencapsulation of isocyanates for self-healing polymers. Macromolecules 2008, 41, 9650–9655. [Google Scholar] [CrossRef]
- Zainuddin, S.; Arefin, T.; Fahim, A.; Hosur, M.V.; Tyson, J.D.; Kumar, A.; Trovillion, J.; Jeelani, S. Recovery and improvement in low-velocity impact properties of e-glass/epoxy composites through novel self-healing technique. Compos. Struct. 2014, 108, 277–286. [Google Scholar] [CrossRef]
- Jin, H.; Mangun, C.L.; Griffin, A.S.; Moore, J.S.; Sottos, N.R.; White, S.R. Thermally stable autonomic healing in epoxy using a dual-microcapsule system. Adv. Mater. 2014, 26, 282–287. [Google Scholar] [CrossRef] [PubMed]
- Patrick, J.F.; Sottos, N.R.; White, S.R. Microvascular based self-healing polymeric foam. Polymer 2012, 53, 4231–4240. [Google Scholar] [CrossRef]
- Hillewaere, X.K.D.; Teixeira, R.F.A.; Nguyen, L.-T.T.; Ramos, J.A.; Rahier, H.; Du Prez, F.E. Autonomous self-healing of epoxy thermosets with thiol-isocyanate chemistry. Adv. Funct. Mater. 2014, 24, 5575–5583. [Google Scholar] [CrossRef]
- Zhang, H.; Wang, P.; Yang, J. Self-healing epoxy via epoxy–amine chemistry in dual hollow glass bubbles. Compos. Sci. Technol. 2014, 94, 23–29. [Google Scholar] [CrossRef]
- Yi, H.; Deng, Y.; Wang, C. Pickering emulsion-based fabrication of epoxy and amine microcapsules for dual core self-healing coating. Compos. Sci. Technol. 2016, 133, 51–59. [Google Scholar] [CrossRef]
- Liu, X.P.; Zheng, T.L. The Synthesis of a Polyaspartic Acid Ester and Development of the Coatings. Adv. Mater. Res 2013, 842, 297–301. [Google Scholar] [CrossRef]
- Liu, Z.-F.; Wang, Y.-J.; Gao, Y.-H.; Zhang, L.-H. Synergistic scale inhibition of polyaspartic acid composite with magnetic field. Front. Chem. Eng. China 2007, 1, 261–265. [Google Scholar] [CrossRef]
- Chen, Y.; Xia, C.; Shepard, Z.; Smith, N.; Rice, N.; Peterson, A.M.; Sakulich, A. Self-healing coatings for steel-reinforced concrete. ACS Sustain. Chem. Eng. 2017, 5, 3955–3962. [Google Scholar] [CrossRef]
- Zhang, H.; Li, W.; Huang, R.; Wang, N.; Wang, J.P.; Zhang, X.X. Microstructure regulation of microencapsulated bio-based n-dodecanol as phase change material by in-situ polymerization. New J. Chem. 2017, 41, 14696–14707. [Google Scholar] [CrossRef]
- Yang, Y.; Wei, Z.; Wang, C.; Tong, Z. Versatile fabrication of nanocomposite microcapsules with controlled shell thickness and low permeability. ACS Appl. Mater. Interfaces 2013, 5, 2495–2502. [Google Scholar] [CrossRef] [PubMed]












| Healing Agent | Temperature (°C) | Time | Healing Efficiency (%) |
|---|---|---|---|
| drying oil [19] | 25 | 2–4 day | 54 |
| Epoxy [8] | 45 | 2 day | 49 |
| Epoxy-amine [20] | 25 | 2 day | 91 |
| Isocyanate [21] | 25 | 1 day | 100 |
| Thiol-isocyanate [22] | 25 | 8 h | 61 |
| Samples | Core Material Composition | Weight (g) | Emulsification Speed (rpm) |
|---|---|---|---|
| S1 | PAE | 20 g | 3000 |
| S2 | PAE | 20 g | 5000 |
| S3 | PAE/TO | 10 g/10 g | 5000 |
| S4 | TO | 20 g | 5000 |
| Self-Healing System | Content | Healing Efficiency |
|---|---|---|
| dual-component microcapsules | 5 wt % | 63% |
| dual-component microcapsules | 10 wt % | 84% |
| dual-component microcapsules | 15 wt % | 93% |
| IPDI microcapsules | 15 wt % | 78% |
| dual-component microcapsules | 20 wt % | 98% |
| dual-component microcapsules | 25 wt % | 100% |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guo, M.; Li, W.; Han, N.; Wang, J.; Su, J.; Li, J.; Zhang, X. Novel Dual-Component Microencapsulated Hydrophobic Amine and Microencapsulated Isocyanate Used for Self-Healing Anti-Corrosion Coating. Polymers 2018, 10, 319. https://doi.org/10.3390/polym10030319
Guo M, Li W, Han N, Wang J, Su J, Li J, Zhang X. Novel Dual-Component Microencapsulated Hydrophobic Amine and Microencapsulated Isocyanate Used for Self-Healing Anti-Corrosion Coating. Polymers. 2018; 10(3):319. https://doi.org/10.3390/polym10030319
Chicago/Turabian StyleGuo, Maolian, Wei Li, Na Han, Jianping Wang, Junfeng Su, Jianjie Li, and Xingxiang Zhang. 2018. "Novel Dual-Component Microencapsulated Hydrophobic Amine and Microencapsulated Isocyanate Used for Self-Healing Anti-Corrosion Coating" Polymers 10, no. 3: 319. https://doi.org/10.3390/polym10030319
APA StyleGuo, M., Li, W., Han, N., Wang, J., Su, J., Li, J., & Zhang, X. (2018). Novel Dual-Component Microencapsulated Hydrophobic Amine and Microencapsulated Isocyanate Used for Self-Healing Anti-Corrosion Coating. Polymers, 10(3), 319. https://doi.org/10.3390/polym10030319






