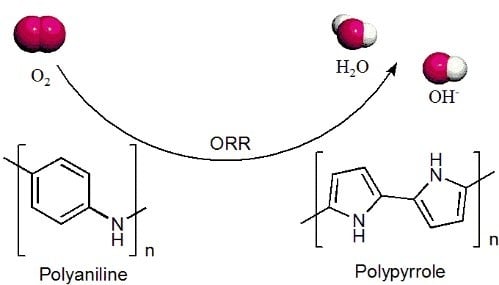
Recent Advance on Polyaniline or Polypyrrole-Derived Electrocatalysts for Oxygen Reduction Reaction
Abstract
1. Introduction
2. Polyaniline-Derived Catalysts for Oxygen Reduction Reaction
2.1. Metal-Free Polyaniline-Based Catalysts
2.2. Noble Metal-Free Polyaniline-Based Catalysts
2.3. Noble Metal Polyaniline-Based Catalysts
3. Polypyrrole-Derived Catalysts for Oxygen Reduction Reaction
3.1. Metal-Free Polypyrrole-Derived Catalysts
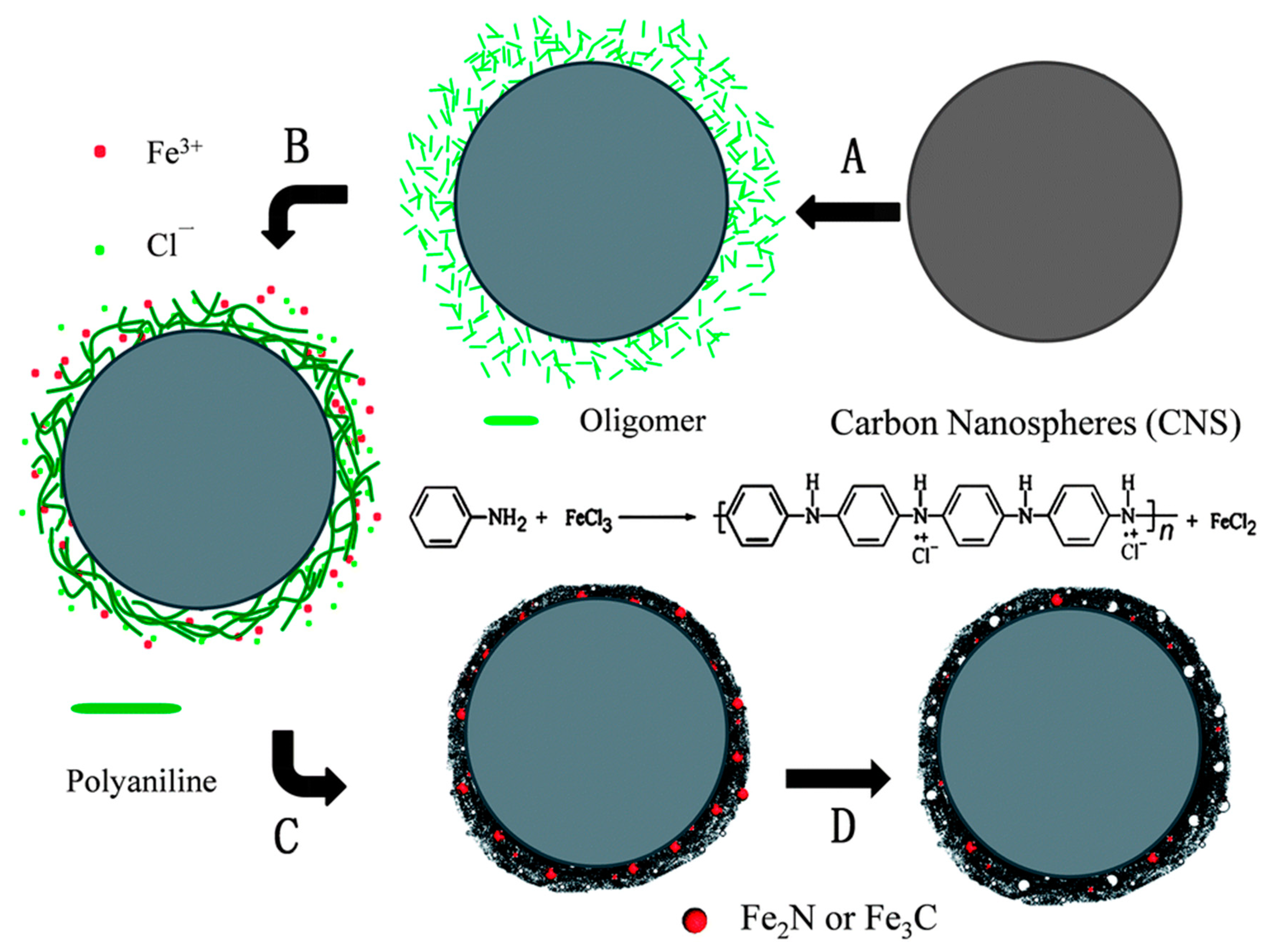
3.2. Non-Noble Metal Polypyrrole-Derived Catalysts
3.3. Noble Metal Polypyrrole-Based Catalysts
4. Summary and Perspective
Funding
Conflicts of Interest
References
- Chen, H.C.; Song, Z.; Zhao, X.; Zhang, T.; Pei, P.C.; Liang, C. A review of durability test protocols of the proton exchange membrane fuel cells for vehicle. Appl. Energy 2018, 224, 289–299. [Google Scholar] [CrossRef]
- Wang, G.J.; Yu, Y.; Liu, H.; Gong, C.L.; Wen, S.; Wang, X.H.; Tu, Z.K. Progress on design and development of polymer electrolyte membrane fuel cell systems for vehicle applications: A review. Fuel Process. Technol. 2018, 179, 203–228. [Google Scholar] [CrossRef]
- Priya, K.; Sathishkumar, K.; Rajasekar, N. A comprehensive review on parameter estimation techniques for Proton Exchange Membrane fuel cell modelling. Renew. Sustain. Energy Rev. 2018, 93, 121–144. [Google Scholar] [CrossRef]
- Zhou, X.; Qiao, J.; Yang, L.; Zhang, J. A Review of Graphene-Based Nanostructural Materials for Both Catalyst Supports and Metal-Free Catalysts in PEM Fuel Cell Oxygen Reduction Reactions. Adv. Energy Mater. 2014, 4. [Google Scholar] [CrossRef]
- Klingele, M.; Van Pham, C.; Fischer, A.; Thiele, S. A Review on Metal-Free Doped Carbon Materials Used as Oxygen Reduction Catalysts in Solid Electrolyte Proton Exchange Fuel Cells. Fuel Cells 2016, 16, 522–529. [Google Scholar] [CrossRef]
- Banham, D.; Ye, S.; Pei, K.; Ozaki, J.-I.; Kishimoto, T.; Imashiro, Y. A review of the stability and durability of non-precious metal catalysts for the oxygen reduction reaction in proton exchange membrane fuel cells. J. Power Sources 2015, 285, 334–348. [Google Scholar] [CrossRef]
- Zhang, C.L.; Shen, X.C.; Pan, Y.B.; Peng, Z.M. A review of Pt-based electrocatalysts for oxygen reduction reaction. Front. Energy 2017, 11, 268–285. [Google Scholar] [CrossRef]
- Lafforgue, C.; Zadick, A.; Dubau, L.; Maillard, F.; Chatenet, M. Selected Review of the Degradation of Pt and Pd-based Carbon-supported Electrocatalysts for Alkaline Fuel Cells: Towards Mechanisms of Degradation. Fuel Cells 2018, 18, 229–238. [Google Scholar] [CrossRef]
- Sui, S.; Wang, X.Y.; Zhou, X.T.; Su, Y.H.; Riffatc, S.; Liu, C.J. A comprehensive review of Pt electrocatalysts for the oxygen reduction reaction: Nanostructure, activity, mechanism and carbon support in PEM fuel cells. J. Mater. Chem. A 2017, 5, 1808–1825. [Google Scholar] [CrossRef]
- Zhang, B.W.; Yang, H.L.; Wang, Y.X.; Dou, S.X.; Liu, H.K. A Comprehensive Review on Controlling Surface Composition of Pt-Based Bimetallic Electrocatalysts. Adv. Energy Mater. 2018, 8. [Google Scholar] [CrossRef]
- Asset, T.; Chattot, R.; Fontana, M.; Mercier-Guyon, B.; Job, N.; Dubau, L.; Maillard, F. A Review on Recent Developments and Prospects for the Oxygen Reduction Reaction on Hollow Pt-alloy Nanoparticles. Chemphyschem 2018, 19, 1552–1567. [Google Scholar] [CrossRef] [PubMed]
- Gomez-Marin, A.M.; Ticianelli, E.A. A reviewed vision of the oxygen reduction reaction mechanism on Pt-based catalysts. Curr. Opin. Electrochem. 2018, 9, 129–136. [Google Scholar] [CrossRef]
- Antolini, E. The oxygen reduction on Pt-Ni and Pt-Ni-M catalysts for low-temperature acidic fuel cells: A review. Int. J. Energy Res. 2018, 42, 3747–3769. [Google Scholar] [CrossRef]
- Gong, K.; Du, F.; Xia, Z.; Durstock, M.; Dai, L. Nitrogen-Doped Carbon Nanotube Arrays with High Electrocatalytic Activity for Oxygen Reduction. Science 2009, 323, 760–764. [Google Scholar] [CrossRef]
- Guo, D.; Shibuya, R.; Akiba, C.; Saji, S.; Kondo, T.; Nakamura, J. Active sites of nitrogen-doped carbon materials for oxygen reduction reaction clarified using model catalysts. Science 2016, 351, 361–365. [Google Scholar] [CrossRef] [PubMed]
- Chung, H.T.; Cullen, D.A.; Higgins, D.; Sneed, B.T.; Holby, E.F.; More, K.L.; Zelenay, P. Direct atomic-level insight into the active sites of a high-performance PGM-free ORR catalyst. Science 2017, 357, 479–483. [Google Scholar] [CrossRef] [PubMed]
- Liu, T.; Zhao, P.; Hua, X.; Luo, W.; Chen, S.; Cheng, G. An Fe-N-C hybrid electrocatalyst derived from a bimetal-organic framework for efficient oxygen reduction. J. Mater. Chem. A 2016, 4, 11357–11364. [Google Scholar] [CrossRef]
- Zhang, R.; He, S.; Lu, Y.; Chen, W. Fe, Co, N-functionalized carbon nanotubes in situ grown on 3D porous N-doped carbon foams as a noble metal-free catalyst for oxygen reduction. J. Mater. Chem. A 2015, 3, 3559–3567. [Google Scholar] [CrossRef]
- Yang, F.; Zhao, P.; Hua, X.; Luo, W.; Cheng, G.; Xing, W.; Chen, S. A cobalt-based hybrid electrocatalyst derived from a carbon nanotube inserted metal-organic framework for efficient water-splitting. J. Mater. Chem. A 2016, 4, 16057–16063. [Google Scholar] [CrossRef]
- Bu, L.; Zhang, N.; Guo, S.; Zhang, X.; Li, J.; Yao, J.; Wu, T.; Lu, G.; Ma, J.-Y.; Su, D.; et al. Biaxially strained PtPb/Pt core/shell nanoplate boosts oxygen reduction catalysis. Science 2016, 354, 1410–1414. [Google Scholar] [CrossRef]
- Li, M.; Zhao, Z.; Cheng, T.; Fortunelli, A.; Chen, C.-Y.; Yu, R.; Zhang, Q.; Gu, L.; Merinov, B.V.; Lin, Z.; et al. Ultrafine jagged platinum nanowires enable ultrahigh mass activity for the oxygen reduction reaction. Science 2016, 354, 1414–1419. [Google Scholar] [CrossRef] [PubMed]
- Choi, C.H.; Chung, M.W.; Kwon, H.C.; Park, S.H.; Woo, S.I. B, N- and P, N-doped graphene as highly active catalysts for oxygen reduction reactions in acidic media. J. Mater. Chem. A 2013, 1, 3694–3699. [Google Scholar] [CrossRef]
- Yang, S.; Zhi, L.; Tang, K.; Feng, X.; Maier, J.; Muellen, K. Efficient Synthesis of Heteroatom (N or S)-Doped Graphene Based on Ultrathin Graphene Oxide-Porous Silica Sheets for Oxygen Reduction Reactions. Adv. Funct. Mater. 2012, 22, 3634–3640. [Google Scholar] [CrossRef]
- Meng, Y.; Voiry, D.; Goswami, A.; Zou, X.; Huang, X.; Chhowalla, M.; Liu, Z.; Asefa, T. N-, O-, and S-Tridoped Nanoporous Carbons as Selective Catalysts for Oxygen Reduction and Alcohol Oxidation Reactions. J. Am. Chem. Soc. 2014, 136, 13554–13557. [Google Scholar] [CrossRef] [PubMed]
- Qu, K.; Zheng, Y.; Dai, S.; Qiao, S.Z. Graphene oxide-polydopamine derived N, S-codoped carbon nanosheets as superior bifunctional electrocatalysts for oxygen reduction and evolution. Nano Energy 2016, 19, 373–381. [Google Scholar] [CrossRef]
- Zhao, Z.; Wang, S.; Liang, R.; Li, Z.; Shi, Z.; Chen, G. Graphene-wrapped chromium-MOF(MIL-101)/sulfur composite for performance improvement of high-rate rechargeable Li-S batteries. J. Mater. Chem. A 2014, 2, 13509–13512. [Google Scholar] [CrossRef]
- Wang, M.-Q.; Yang, W.-H.; Wang, H.-H.; Chen, C.; Zhou, Z.-Y.; Sun, S.-G. Pyrolyzed Fe-N-C Composite as an Efficient Non-precious Metal Catalyst for Oxygen Reduction Reaction in Acidic Medium. Acs Catal. 2014, 4, 3928–3936. [Google Scholar] [CrossRef]
- Jiang, W.-J.; Gu, L.; Li, L.; Zhang, Y.; Zhang, X.; Zhang, L.-J.; Wang, J.-Q.; Hu, J.-S.; Wei, Z.; Wan, L.-J. Understanding the High Activity of Fe-N-C Electrocatalysts in Oxygen Reduction: Fe/Fe3C Nanoparticles Boost the Activity of Fe-N-x. J. Am. Chem. Soc. 2016, 138, 3570–3578. [Google Scholar] [CrossRef]
- Lin, L.; Zhu, Q.; Xu, A.-W. Noble-Metal-Free Fe-N/C Catalyst for Highly Efficient Oxygen Reduction Reaction under Both Alkaline and Acidic Conditions. J. Am. Chem. Soc. 2014, 136, 11027–11033. [Google Scholar] [CrossRef]
- Maity, S.; Chatterjee, A. Conductive polymer-based electro-conductive textile composites for electromagnetic interference shielding: A review. J. Ind. Text. 2018, 47, 2228–2252. [Google Scholar] [CrossRef]
- Xie, L.; Zhu, Y.T. Tune the phase morphology to design conductive polymer composites: A review. Polym. Compos. 2018, 39, 2985–2996. [Google Scholar] [CrossRef]
- Amoabeng, D.; Velankar, S.S. A Review of Conductive Polymer Composites Filled With Low Melting Point Metal Alloys. Polym. Eng. Sci. 2018, 58, 1010–1019. [Google Scholar] [CrossRef]
- Deng, H.; Li, Q.; Liu, J.; Wang, F. Active sites for oxygen reduction reaction on nitrogen-doped carbon nanotubes derived from polyaniline. Carbon 2017, 112, 219–229. [Google Scholar] [CrossRef]
- Xu, J.; Lu, S.; Zhou, H.; Chen, X.; Wang, Y.; Xiao, C.; Ding, S. A Highly Efficient Electrocatalyst Derived from Polyaniline@CNTs-SPS** for the Oxygen Reduction Reaction. Chemelectrochem 2018, 5, 195–200. [Google Scholar] [CrossRef]
- Quilez-Bermejo, J.; Morallon, E.; Cazorla-Amoros, D. Oxygen-reduction catalysis of N-doped carbons prepared via heat treatment of polyaniline at over 1100 degrees C. Chem. Commun. 2018, 54, 4441–4444. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Yin, X.; Yu, X.; Tian, J.; Wu, W. Preparation of nitrogen-doped carbon materials based on polyaniline fiber and their oxygen reduction properties. Colloid Surface A 2018, 539, 163–170. [Google Scholar] [CrossRef]
- Zhou, F.; Wang, G.; Huang, F.; Zhang, Y.; Pan, M. Polyaniline derived N- and O-enriched high surface area hierarchical porous carbons as an efficient metal-free electrocatalyst for oxygen reduction. Electrochim. Acta 2017, 257, 73–81. [Google Scholar] [CrossRef]
- Xing, S.; Yu, X.; Wang, G.; Yu, Y.; Wang, Y.; Xing, Y. Confined polyaniline derived mesoporous carbon for oxygen reduction reaction. Eur. Polym. J. 2017, 88, 1–8. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhuang, X.; Su, Y.; Zhang, F.; Feng, X. Polyaniline nanosheet derived B/N co-doped carbon nanosheets as efficient metal-free catalysts for oxygen reduction reaction. J. Mater. Chem. A 2014, 2, 7742–7746. [Google Scholar] [CrossRef]
- Jiang, H.L.; Zhu, Y.H.; Feng, Q.; Su, Y.H.; Yang, X.L.; Li, C.Z. Nitrogen and Phosphorus Dual-Doped Hierarchical Porous Carbon Foams as Efficient Metal-Free Electrocatalysts for Oxygen Reduction Reactions. Chem.-Eur. J. 2014, 20, 3106–3112. [Google Scholar] [CrossRef]
- Quilez-Bermejo, J.; Gonzalez-Gaitan, C.; Morallon, E.; Cazorla-Amoros, D. Effect of carbonization conditions of polyaniline on its catalytic activity towards ORR. Some insights about the nature of the active sites. Carbon 2017, 119, 62–71. [Google Scholar] [CrossRef]
- Wei, Q.L.; Zhang, G.X.; Yang, X.H.; Fu, Y.Q.; Yang, G.H.; Chen, N.; Chen, W.F.; Sun, S.H. Litchi-like porous Fe/N/C spheres with atomically dispersed FeNx promoted by sulfur as highly efficient oxygen electrocatalysts for Zn-air batteries. J. Mater. Chem. A 2018, 6, 4605–4610. [Google Scholar] [CrossRef]
- Chenitz, R.; Kramm, U.I.; Lefevre, M.; Glibin, V.; Zhang, G.X.; Sun, S.H.; Dodelet, J.P. A specific demetalation of Fe-N-4 catalytic sites in the micropores of NC_Ar + NH3 is at the origin of the initial activity loss of the highly active Fe/N/C catalyst used for the reduction of oxygen in PEM fuel cells. Energy Environ. Sci. 2018, 11, 365–382. [Google Scholar] [CrossRef]
- Zhang, G.X.; Wei, Q.L.; Yang, X.H.; Tavares, A.C.; Sun, S.H. RRDE experiments on noble-metal and noble-metal-free catalysts: Impact of loading on the activity and selectivity of oxygen reduction reaction in alkaline solution. Appl. Catal. B-Environ. 2017, 206, 115–126. [Google Scholar] [CrossRef]
- Wei, Q.L.; Zhang, G.X.; Yang, X.H.; Chenitz, R.; Barham, D.; Yang, L.J.; Ye, S.Y.; Knights, S.; Sun, S.H. 3D Porous Fe/N/C Spherical Nanostructures As High-Performance Electrocatalysts for Oxygen Reduction in Both Alkaline and Acidic Media. Acs Appl. Mater. Interfaces 2017, 9, 36944–36954. [Google Scholar] [CrossRef] [PubMed]
- Wu, G.; More, K.L.; Johnston, C.M.; Zelenay, P. High-Performance Electrocatalysts for Oxygen Reduction Derived from Polyaniline, Iron, and Cobalt. Science 2011, 332, 443–447. [Google Scholar] [CrossRef] [PubMed]
- Jaouen, F.; Herranz, J.; Lefevre, M.; Dodelet, J.P.; Kramm, U.I.; Herrmann, I.; Bogdanoff, P.; Maruyama, J.; Nagaoka, T.; Garsuch, A.; et al. Cross-Laboratory Experimental Study of Non-Noble-Metal Electrocatalysts for the Oxygen Reduction Reaction. ACS Appl. Mater. Interfaces 2009, 1, 1623–1639. [Google Scholar] [CrossRef]
- Hu, Y.; Zhao, X.; Huang, Y.; Li, Q.; Bjerrum, N.J.; Liu, C.; Xing, W. Synthesis of self-supported non-precious metal catalysts for oxygen reduction reaction with preserved nanostructures from the polyaniline nanofiber precursor. J. Power Sources 2013, 225, 129–136. [Google Scholar] [CrossRef]
- Wang, X.; Zou, L.; Fu, H.; Xiong, Y.; Tao, Z.; Zheng, J.; Li, X. Noble Metal-Free Oxygen Reduction Reaction Catalysts Derived from Prussian Blue Nanocrystals Dispersed in Polyaniline. ACS Appl. Mater. Interfaces 2016, 8, 8436–8444. [Google Scholar] [CrossRef]
- Shi, K.-M.; Cheng, X.; Jia, Z.-Y.; Guo, J.-W.; Wang, C.; Wang, J. Oxygen reduction reaction of Fe-Polyaniline/Carbon Nanotube and Pt/C catalysts in alkali media. Int. J. Hydrog. Energy 2016, 41, 16903–16912. [Google Scholar] [CrossRef]
- Zhang, J.; He, D.; Su, H.; Chen, X.; Pan, M.; Mu, S. Porous polyaniline-derived FeNxC/C catalysts with high activity and stability towards oxygen reduction reaction using ferric chloride both as an oxidant and iron source. J. Mater. Chem. A 2014, 2, 1242–1246. [Google Scholar] [CrossRef]
- Wang, G.; Jiang, K.; Xu, M.; Min, C.; Ma, B.; Yang, X. A high activity nitrogen-doped carbon catalyst for oxygen reduction reaction derived from polyaniline-iron coordination polymer. J. Power Sources 2014, 266, 222–225. [Google Scholar] [CrossRef]
- Cao, S.; Han, N.; Han, J.; Hu, Y.; Fan, L.; Zhou, C.; Guo, R. Mesoporous Hybrid Shells of Carbonized Polyaniline/Mn2O3 as Non-Precious Efficient Oxygen Reduction Reaction Catalyst. ACS Appl. Mater. Interfaces 2016, 8, 6040–6050. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; Xu, Y.; Mei, X.; Du, N.; Jv, R.; Hu, Z.; Chen, S. Polyaniline/beta-MnO2 nanocomposites as cathode electrocatalyst for oxygen reduction reaction in microbial fuel cells. Chemosphere 2018, 198, 482–491. [Google Scholar] [CrossRef]
- Peng, S.; Jiang, H.; Zhang, Y.; Yang, L.; Wang, S.; Deng, W.; Tan, Y. Facile synthesis of cobalt and nitrogen co-doped graphene networks from polyaniline for oxygen reduction reaction in acidic solutions. J. Mater. Chem. A 2016, 4, 3678–3682. [Google Scholar] [CrossRef]
- Deng, Z.; Yi, Q.; Zhang, Y.; Nie, H.; Li, G.; Yu, L.; Zhou, X. Carbon Paper-Supported NiCo/C-N Catalysts Synthesized by Directly Pyrolyzing NiCo-Doped Polyaniline for Oxygen Reduction Reaction. Nano 2018, 13. [Google Scholar] [CrossRef]
- Zhang, L.; Liu, X.; Wang, Y.; Chen, G.; Xing, S. Dual Role of Polyaniline for Achieving Ag Dendrites and Enhancing Its Oxygen Reduction Reaction Catalytic Activity. Chemistryselect 2017, 2, 10300–10303. [Google Scholar] [CrossRef]
- Ye, B.; Cheng, K.; Li, W.; Liu, J.; Zhang, J.; Mu, S. Polyaniline and Perfluorosulfonic Acid Co-Stabilized Metal Catalysts for Oxygen Reduction Reaction. Langmuir 2017, 33, 5353–5361. [Google Scholar] [CrossRef]
- Luo, Y.; Estudillo-Wong, L.A.; Cavillo, L.; Granozzi, G.; Alonso-Vante, N. An easy and cheap chemical route using a MOF precursor to prepare Pd-Cu electrocatalyst for efficient energy conversion cathodes. J. Catal. 2016, 338, 135–142. [Google Scholar] [CrossRef]
- Liu, Y.; Chen, N.; Wang, F.; Cai, Y.; Zhu, H. Pt-Co deposited on polyaniline-modified carbon for the electro-reduction of oxygen: The interaction between Pt-Co nanoparticles and polyaniline. New J. Chem. 2017, 41, 6585–6592. [Google Scholar] [CrossRef]
- Kaewsai, D.; Hunsom, M. Comparative Study of the ORR Activity and Stability of Pt and PtM (M = Ni, Co, Cr, Pd) Supported on Polyaniline/Carbon Nanotubes in a PEM Fuel Cell. Nanomaterials 2018, 8, 299. [Google Scholar] [CrossRef] [PubMed]
- An, H.; Zhang, R.; Li, Z.; Zhou, L.; Shao, M.; Wei, M. Highly efficient metal-free electrocatalysts toward oxygen reduction derived from carbon nanotubes@polypyrrole core–shell hybrids. J. Mater. Chem. A 2016, 4, 18008–18014. [Google Scholar] [CrossRef]
- Zhang, Z.; Sun, J.; Dou, M.; Jo, J.; Wang, F. Nitrogen and Phosphorus Codoped Mesoporous Carbon Derived from Polypyrrole as Superior Metal-Free Electrocatalyst toward the Oxygen Reduction Reaction. ACS Appl. Mater. Interfaces 2017, 9, 16236–16242. [Google Scholar] [CrossRef] [PubMed]
- Xia, W.; Qu, C.; Liang, Z.; Zhao, B.; Dai, S.; Qiu, B.; Jiao, Y.; Zhang, Q.; Huang, X.; Guo, W.; et al. High-Performance Energy Storage and Conversion Materials Derived from a Single Metal Organic Framework/Graphene Aerogel Composite. Nano Lett. 2017, 17, 2788–2795. [Google Scholar] [CrossRef] [PubMed]
- Yang, M.; Liu, Y.; Chen, H.; Yang, D.; Li, H. Porous N-Doped Carbon Prepared from Triazine-Based Polypyrrole Network: A Highly Efficient Metal-Free Catalyst for Oxygen Reduction Reaction in Alkaline Electrolytes. ACS Appl. Mater. Interfaces 2016, 8, 28615–28623. [Google Scholar] [CrossRef] [PubMed]
- Morozan, A.; Jegou, P.; Campidelli, S.; Palacin, S.; Jousselme, B. Relationship between polypyrrole morphology and electrochemical activity towards oxygen reduction reaction. Chem. Commun. 2012, 48, 4627–4629. [Google Scholar] [CrossRef] [PubMed]
- Osmieri, L.; Videla, A.H.A.M.; Specchia, S. Optimization of a Fe-N-C electrocatalyst supported on mesoporous carbon functionalized with polypyrrole for oxygen reduction reaction under both alkaline and acidic conditions. Int. J. Hydrog. Energy 2016, 41, 19610–19628. [Google Scholar] [CrossRef]
- Osmieri, L.; Zafferoni, C.; Wang, L.; Videla, A.H.A.M.; Lavacchi, A.; Specchia, S. Polypyrrole-Derived Fe-Co-N-C Catalyst for the Oxygen Reduction Reaction: Performance in Alkaline Hydrogen and Ethanol Fuel Cells. Chemelectrochem 2018, 5, 1954–1965. [Google Scholar] [CrossRef]
- Sun, M.; Wu, X.; Liu, C.; Xie, Z.; Deng, X.; Zhang, W.; Huang, Q.; Huang, B. The in situ grown of activated Fe-N-C nanofibers derived from polypyrrole on carbon paper and its electro-catalytic activity for oxygen reduction reaction. J. Solid State Electrochem. 2018, 22, 1217–1226. [Google Scholar] [CrossRef]
- Sun, T.; Yang, M.; Chen, H.; Liu, Y.; Li, H. N-doped and N/Fe-codoped porous carbon spheres derived from tetrazine-based polypyrrole as efficient electrocatalysts for the oxygen reduction reaction. Appl. Catal. A Gen. 2018, 559, 102–111. [Google Scholar] [CrossRef]
- Sha, H.-D.; Yuan, X.; Li, L.; Ma, Z.; Ma, Z.-F.; Zhang, L.; Zhang, J. Experimental identification of the active sites in pyrolyzed carbon-supported cobalt-polypyrrole-4-toluenesulfinic acid as electrocatalysts for oxygen reduction reaction. J. Power Sources 2014, 255, 76–84. [Google Scholar] [CrossRef]
- Jia, X.; Gao, S.; Liu, T.; Li, D.; Tang, P.; Feng, Y. Fabrication and Bifunctional Electrocatalytic Performance of Ternary CoNiMn Layered Double Hydroxides/Polypyrrole/Reduced Graphene Oxide Composite for Oxygen Reduction and Evolution Reactions. Electrochim. Acta 2017, 245, 51–60. [Google Scholar] [CrossRef]
- Zhu, S.Y.; Tian, H.; Wang, N.; Chen, B.; Mai, Y.Y.; Feng, X.L. Patterning Graphene Surfaces with Iron-Oxide-Embedded Mesoporous Polypyrrole and Derived N-Doped Carbon of Tunable Pore Size. Small 2018, 14. [Google Scholar] [CrossRef]
- Ren, S.; Guo, Y.; Ma, S.; Mao, Q.; Wu, D.; Yang, Y.; Jing, H.; Song, X.; Hao, C. Co3O4 nanoparticles assembled on polypyrrole/graphene oxide for electrochemical reduction of oxygen in alkaline media. Chin. J. Catatl. 2017, 38, 1281–1290. [Google Scholar] [CrossRef]
- Ren, G.; Li, Y.; Guo, Z.; Xiao, G.; Zhu, Y.; Dai, L.; Jiang, L. A bio-inspired Co3O4-polypyrrole-graphene complex as an efficient oxygen reduction catalyst in one-step ball milling. Nano Res. 2015, 8, 3461–3471. [Google Scholar] [CrossRef]
- Xiao, D.; Ma, J.; Chen, C.; Luo, Q.; Ma, J.; Zheng, L.; Zuo, X. Oxygen-doped carbonaceous polypyrrole nanotubes-supported Ag nanoparticle as electrocatalyst for oxygen reduction reaction in alkaline solution. Mater. Res. Bull. 2018, 105, 184–191. [Google Scholar] [CrossRef]
- Chen, X.-L.; Zhang, L.; Feng, J.-J.; Wang, W.; Yuan, P.-X.; Han, D.-M.; Wang, A.-J. Facile solvothermal fabrication of polypyrrole sheets supported dendritic platinum-cobalt nanoclusters for highly efficient oxygen reduction and ethylene glycol oxidation. J. Colloid Interface Sci. 2018, 530, 394–402. [Google Scholar] [CrossRef]
- Escudero Cid, R.; Gomez de la Fuente, J.L.; Rojas, S.; Garcia Fierro, J.L.; Ocon, P. Polypyrrole-Modified-Carbon-Supported Ru-Pt Nanoparticles as Highly Methanol-Tolerant Electrocatalysts for the Oxygen-Reduction Reaction. Chemcatchem 2013, 5, 3680–3689. [Google Scholar] [CrossRef]
| Electrocatalyst | Medium | Catalysts Loading [mg cm−2] | Rotation Speed/rpm | Onset Potential/V vs. RHE | Halfwave Potential/V vs. RHE | Ref. |
|---|---|---|---|---|---|---|
| PANI-FeCo-C | 0.5 M H2SO4 | 900 | 0.93 | 0.79 | [46] | |
| 3.0 Fe-PANI-L | 0.5 M H2SO4 | 0.6 | 1000 | 0.905 | - | [48] |
| C-2PANI/PBA | 0.5 M H2SO4 | 0.36 | 1600 | 0.81 | 0.71 | [49] |
| FeNxC/C–F | 0.1 M HClO4 | 0.8 | 1600 | 0.88 | 0.76 | [51] |
| N/C/Fe-c | 0.5 M H2SO4 | 0.48 | 900 | 0.78 | 0.65 | [52] |
| Fe/N-Cs-900 | 0.5 M H2SO4 | - | 1600 | 0.845 | 0.717 | [70] |
| (Co-PPy-TsOH/C)P-A-P | 0.5 M H2SO4 | 900 | 0.78 | 0.71 | [71] | |
| CPANI/Mn2O3 | 0.1 M KOH | 0.28 | 1600 | 0.83 | 0.68 | [53] |
| Fe-Co-N-C | 0.1 M KOH | 0.635 | 800 | 0.85 | 0.78 | [68] |
| 0.1-Fe-N-C ANFs | 0.1 M KOH | - | 1600 | 0.83 | 0.74 | [69] |
| mNC-Fe3O4@rGO-2 | 0.1 M KOH | 0.24 | 1600 | 0.96 | 0.83 | [73] |
| Electrocatalyst | Medium | Catalysts Loading [mg cm−2] | Rotation Speed/rpm | Onset Potential/V vs. RHE | Halfwave Potential/V vs. RHE | Ref. |
|---|---|---|---|---|---|---|
| NCNT-700 | 1 M NaOH | 0.4 | 1600 | 0.88 at RRDE | 0.8 V at RRDE | [33] |
| NCNTs-900 | 0.1 M KOH | 0.64 | 1600 | 0.96 | 0.82 | [34] |
| MEP-NC850 | 0.1 M KOH | 0.25 | 1600 | 0.94 | 0.82 | [37] |
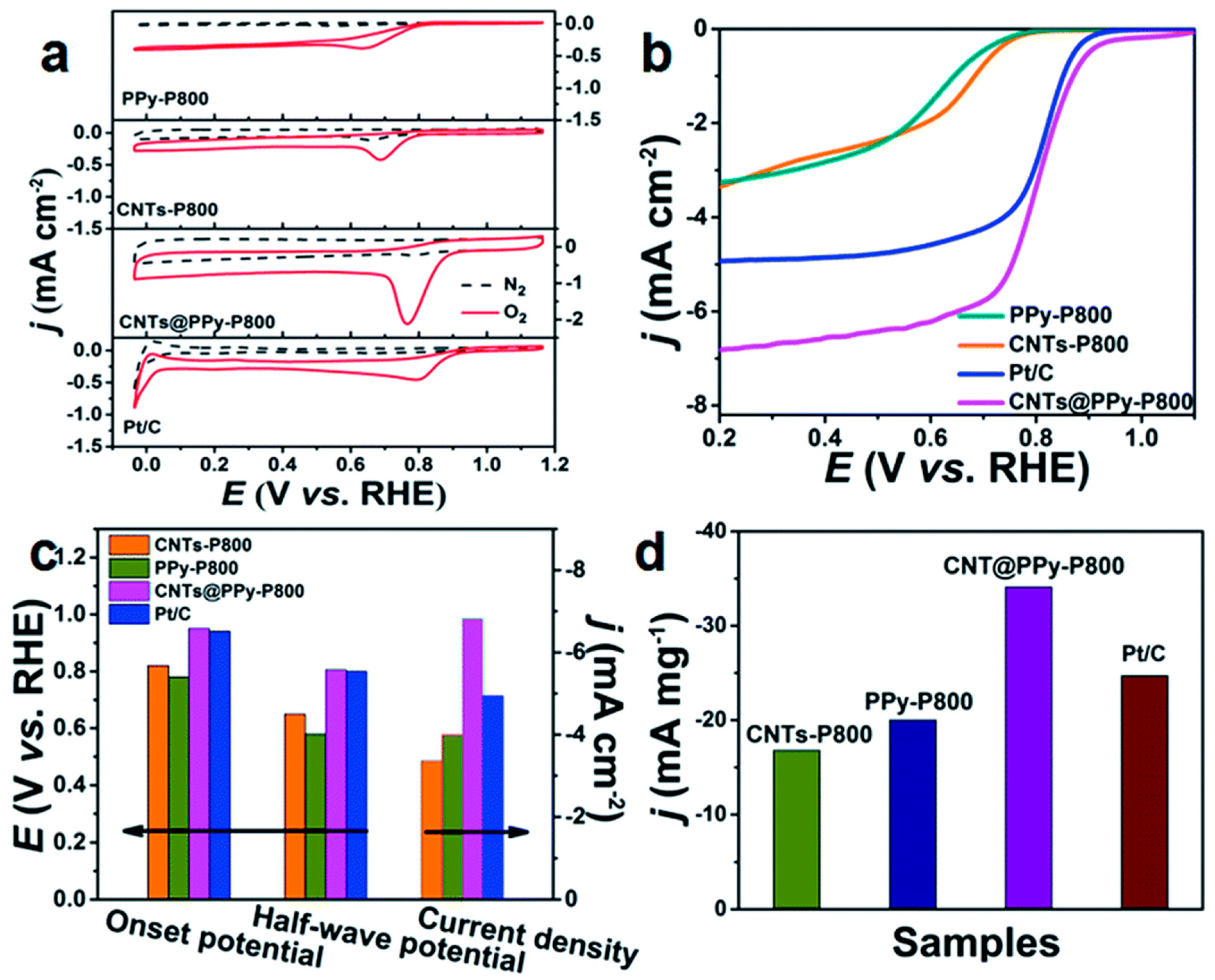
| CNTs@PPy-P800 | 0.1 M KOH | 0.2 | 1600 | 0.92 | 0.81 | [59] |
| N,P-MC | 0.1 M KOH | 0.2 | 1600 | 0.93 | 0.84 | [63] |
| NCNFs-900 | 0.1 M KOH | 1600 | 0.92 | 0.82 | [64] | |
| NC-900 | 0.1 M KOH | 1600 | 0.93 | 0.84 | [65] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jiang, Z.; Yu, J.; Huang, T.; Sun, M. Recent Advance on Polyaniline or Polypyrrole-Derived Electrocatalysts for Oxygen Reduction Reaction. Polymers 2018, 10, 1397. https://doi.org/10.3390/polym10121397
Jiang Z, Yu J, Huang T, Sun M. Recent Advance on Polyaniline or Polypyrrole-Derived Electrocatalysts for Oxygen Reduction Reaction. Polymers. 2018; 10(12):1397. https://doi.org/10.3390/polym10121397
Chicago/Turabian StyleJiang, Zhankun, Jiemei Yu, Taizhong Huang, and Min Sun. 2018. "Recent Advance on Polyaniline or Polypyrrole-Derived Electrocatalysts for Oxygen Reduction Reaction" Polymers 10, no. 12: 1397. https://doi.org/10.3390/polym10121397
APA StyleJiang, Z., Yu, J., Huang, T., & Sun, M. (2018). Recent Advance on Polyaniline or Polypyrrole-Derived Electrocatalysts for Oxygen Reduction Reaction. Polymers, 10(12), 1397. https://doi.org/10.3390/polym10121397