Photocurable Bioinks for the 3D Pharming of Combination Therapies
Abstract
1. Introduction
2. Materials and Methods
2.1. Hydrophilic Photocurable Bioink Preparation
2.2. Hydrophobic Photocurable Bioink Preparation
2.3. Bioinks Gelation and Mechanical Properties
2.4. Scanning Electron Microscopy (SEM)
2.5. Preform Tablet Fabrication and Characterization
2.6. Drug Release Kinetics
2.7. Statistical Analysis
3. Results and Discussion
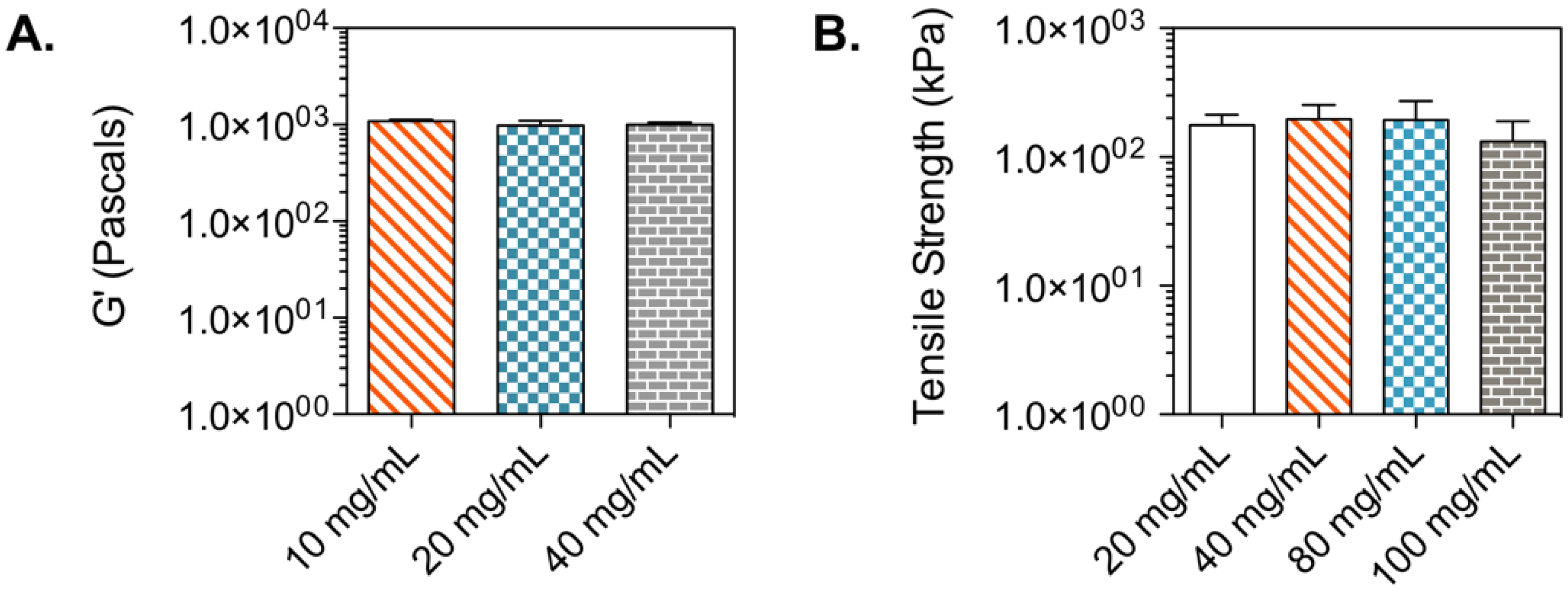
3.1. Bioinks Characterization
3.2. Droplet Formation
3.3. Preform Tablet Characterization
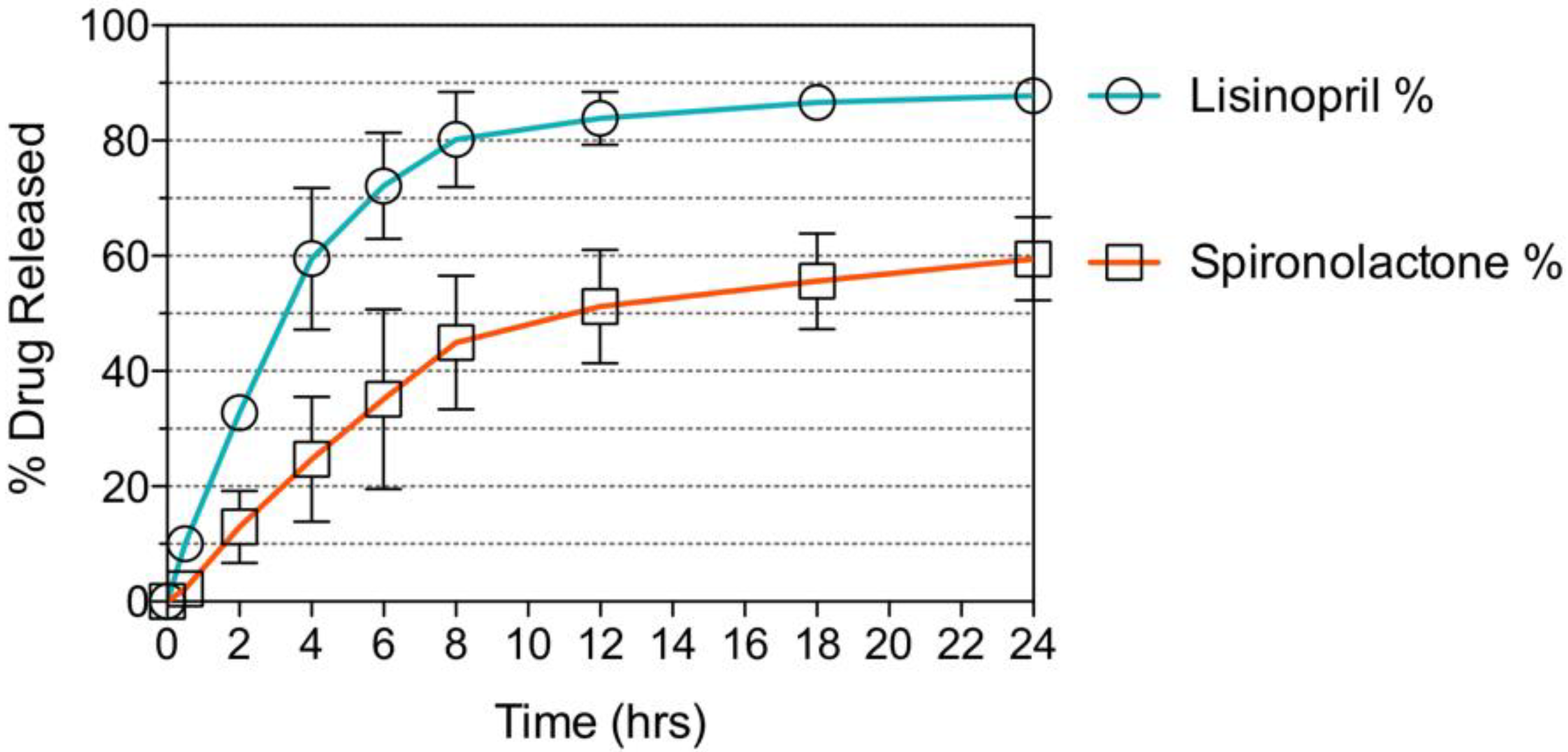
3.4. Polypill Dissolution Test
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Banks, J. Adding value in additive manufacturing: Researchers in the United Kingdom and Europe look to 3D printing for customization. IEEE Pulse 2013, 4, 22–26. [Google Scholar] [CrossRef] [PubMed]
- Kaye, R.; Goldstein, T.; Zeltsman, D.; Grande, D.A.; Smith, L.P. Three dimensional printing: A review on the utility within medicine and otolaryngology. Int. J. Pediatr. Otorhinolaryngol. 2016, 89, 145–148. [Google Scholar] [CrossRef] [PubMed]
- Zopf, D.A.; Hollister, S.J.; Nelson, M.E.; Ohye, R.G.; Green, G.E. Bioresorbable Airway Splint Created with a Three-Dimensional Printer. N. Engl. J. Med. 2013, 368, 2043–2045. [Google Scholar] [CrossRef] [PubMed]
- Acosta-Vélez, G.F.; Wu, B.M. 3D Pharming: Direct Printing of Personalized Pharmaceutical Tablets Abstract Powder Bed Inkjet 3D Printing. Polym. Sci. 2016, 1, 1–10. [Google Scholar]
- Norman, J.; Madurawe, R.D.; Moore, C.M.V.; Khan, M.A.; Khairuzzaman, A. A new chapter in pharmaceutical manufacturing: 3D-printed drug products. Adv. Drug Deliv. Rev. 2017, 108, 39–50. [Google Scholar] [CrossRef] [PubMed]
- Boudriau, S.; Hanzel, C.; Massicotte, J.; Sayegh, L.; Wang, J.; Lefebvre, M. Randomized Comparative Bioavailability of a Novel Three-Dimensional Printed Fast-Melt Formulation of Levetiracetam Following the Administration of a Single 1000-mg Dose to Healthy Human Volunteers Under Fasting and Fed Conditions. Drugs R D 2016, 16, 229–238. [Google Scholar] [CrossRef] [PubMed]
- Preis, M.; Öblom, H. 3D-Printed Drugs for Children—Are We Ready Yet? AAPS PharmSciTech 2017, 18, 303–308. [Google Scholar] [CrossRef] [PubMed]
- Chow, C.K.; Thakkar, J.; Bennett, A.; Hillis, G.; Burke, M.; Usherwood, T.; Vo, K.; Rogers, K.; Atkins, E.; Webster, R.; et al. Quarter-dose quadruple combination therapy for initial treatment of hypertension: Placebo-controlled, crossover, randomised trial and systematic review. Lancet 2017, 389, 1035–1042. [Google Scholar] [CrossRef]
- Weichselbaum, R.R.; Liang, H.; Deng, L.; Fu, Y.-X. Radiotherapy and immunotherapy: A beneficial liaison? Nat. Rev. Clin. Oncol. 2017, 14, 365–379. [Google Scholar] [CrossRef]
- Gradman, A.H.; Basile, J.N.; Carter, B.L.; Bakris, G.L. Combination therapy in hypertension. J. Am. Soc. Hypertens. 2016, 4, 42–50. [Google Scholar] [CrossRef]
- Cahn, A.; Cefalu, W.T. Clinical considerations for use of initial combination therapy in type 2 diabetes. Diabetes Care 2016, 39, S137–S145. [Google Scholar] [CrossRef] [PubMed]
- Melero, I.; Berman, D.M.; Aznar, M.A.; Korman, A.J.; Gracia, J.L.P.; Haanen, J. Evolving synergistic combinations of targeted immunotherapies to combat cancer. Nat. Rev. Cancer 2015, 15, 457–472. [Google Scholar] [CrossRef] [PubMed]
- Cicero, A.F. Combination Therapy with Olmesartan/Hydrochlorothiazide to Improve Blood Pressure Control. J. Clin. Exp. Cardiol. 2015, 6, 384. [Google Scholar] [CrossRef]
- Sun, W.; Sanderson, P.E.; Zheng, W. Drug combination therapy increases successful drug repositioning. Drug Discov. Today 2016, 21, 1189–1195. [Google Scholar] [CrossRef] [PubMed]
- Terrie, Y.C. Monitoring combination drug therapy. Pharm. Times 2010, 76, 1–6. [Google Scholar]
- Follath, F. Challenging the dogma of high target doses in the treatment of heart failure: Is more always better? Arch. Cardiovasc. Dis. 2009, 102, 785–789. [Google Scholar] [CrossRef] [PubMed]
- Farr, M.; Bacon, P.A. How and when should combination therapy be used? The role of an anchor drug. Br. J. Rheumatol. (UK) 1995, 34, 100–103. [Google Scholar] [CrossRef]
- Desai, D.; Wang, J.; Wen, H.; Li, X.; Timmins, P. Formulation design, challenges, and development considerations for fixed dose combination (FDC) of oral solid dosage forms. Pharm. Dev. Technol. 2013, 18, 1265–1276. [Google Scholar] [CrossRef]
- Pourkavoos, N. Unique Risks, Benefits, and Challenges of Developing Drug-Drug Combination Products in a Pharmaceutical Industrial Setting. Comb. Prod. Ther. 2012, 2, 1–31. [Google Scholar] [CrossRef]
- Goyanes, A.; Wang, J.; Buanz, A.; Martinez-Pacheco, R.; Telford, R.; Gaisford, S.; Basit, A.W. 3D printing of medicines: Engineering novel oral devices with unique design and drug release characteristics. Mol. Pharm. 2015, 12, 4077–4084. [Google Scholar] [CrossRef]
- Khaled, S.A.; Burley, J.C.; Alexander, M.R.; Yang, J.; Roberts, C.J. 3D printing of five-in-one dose combination polypill with defined immediate and sustained release profiles. J. Control. Release 2015, 217, 308–314. [Google Scholar] [CrossRef] [PubMed]
- Khaled, S.A.; Burley, J.C.; Alexander, M.R.; Yang, J.; Roberts, C.J. 3D printing of tablets containing multiple drugs with defined release profiles. Int. J. Pharm. 2015, 494, 643–650. [Google Scholar] [CrossRef] [PubMed]
- Acosta-Vélez, G.F.; Linsley, C.S.; Craig, M.C.; Wu, B.M. Photocurable Bioink for the Inkjet 3D Pharming of Hydrophilic Drugs. Bioengineering 2017, 4, 11. [Google Scholar] [CrossRef] [PubMed]
- Acosta-Vélez, G.F.; Zhu, T.Z.; Linsley, C.S.; Wu, B.M. Photocurable poly(ethylene glycol) as a bioink for the inkjet 3D pharming of hydrophobic drugs. Int. J. Pharm. 2018, 546, 145–153. [Google Scholar] [CrossRef] [PubMed]
- Trachtman, H.; Frymoyer, A.; Lewandowski, A.; Greenbaum, L.A.; Feig, D.I.; Gipson, D.S.; Warady, B.A.; Goebel, J.W.; Schwartz, G.J.; Lewis, K.; et al. Pharmacokinetics, pharmacodynamics, and safety of lisinopril in pediatric kidney transplant patients: Implications for starting dose selection. Clin. Pharmacol. Ther. 2015, 98, 25–33. [Google Scholar] [CrossRef] [PubMed]
- Snauwaert, E.; Vande Walle, J.; De Bruyne, P. Therapeutic efficacy and safety of ACE inhibitors in the hypertensive paediatric population: A review. Arch. Dis. Child. 2017, 102, 63–71. [Google Scholar] [CrossRef] [PubMed]
- Larik, F.A.; Saeed, A.; Shahzad, D.; Faisal, M.; El-Seedi, H.; Mehfooz, H.; Channar, P.A. Synthetic approaches towards the multi target drug spironolactone and its potent analogues/derivatives. Steroids 2017, 118, 76–92. [Google Scholar] [CrossRef] [PubMed]
- United States Pharmacopoeia Convention. <1217> Tablet Breaking Force; USP 37-NF 32; United States Pharmacopeia and the National Formulary; The United States Pharmacopeial Convention: Rockville, MD, USA, 2014. [Google Scholar]
- Jang, D.; Kim, D.; Moon, J.; Jang, D.; Kim, D.; Moon, J. Influence of Fluid Physical Properties on Ink-Jet Printability. Langmuir 2009, 25, 2629–2635. [Google Scholar] [CrossRef] [PubMed]
- Cole-Parmer Instrument Company. Available online: https://pim-resources.coleparmer.com/instruction manual/surface-tension-apparatus-instruction-manual.pdf (accessed on 12 January 2017).
- Department of Health and Human Services, Food and Drug Administration, Center for Drug Evaluation and Research (CDER). Size, Shape, and Other Physical Attributes of Generic Tablets and Capsules; Guidance for Industry; Center for Drug Evaluation and Research (CDER): Silver Spring, MD, USA, 2015. [Google Scholar]
- Fraser, J.R.; Laurent, T.C.; Laurent, U.B. Hyaluronan: Its nature, distribution, functions and turnover. J. Intern. Med. 1997, 242, 27–33. [Google Scholar] [CrossRef] [PubMed]
- Allison, D.D.; Grande-Allen, K.J. Review. Hyaluronan: A Powerful Tissue Engineering Tool. Tissue Eng. Part A 2006, 12, 2131–2140. [Google Scholar] [CrossRef] [PubMed]
- Highley, C.B.; Prestwich, G.D.; Burdick, J.A. Recent advances in hyaluronic acid hydrogels for biomedical applications. Curr. Opin. Biotechnol. 2016, 40, 35–40. [Google Scholar] [CrossRef] [PubMed]
- Owen, S.C.; Fisher, S.A.; Tam, R.Y.; Nimmo, C.M.; Shoichet, M.S. Hyaluronic acid click hydrogels emulate the extracellular matrix. Langmuir 2013, 29, 7393–7400. [Google Scholar] [CrossRef] [PubMed]
- Kim, I.L.; Mauck, R.L.; Burdick, J.A. Hydrogel design for cartilage tissue engineering: A case study with hyaluronic acid. Biomaterials 2011, 32, 8771–8782. [Google Scholar] [CrossRef] [PubMed]
- Juban, A.; Nouguier-Lehon, C.; Briancon, S.; Hoc, T.; Puel, F. Predictive model for tensile strength of pharmaceutical tablets based on local hardness measurements. Int. J. Pharm. 2015, 490, 438–445. [Google Scholar] [CrossRef] [PubMed]
- Sheskey, P.J.; Cook, W.G.; Cable, C.G. Handbook Pharmaceutical Excipients, 6th ed.; Pharm Press: London, UK, 2009. [Google Scholar]
- Williams, R.O.; Reynolds, T.D.; Cabelka, T.D.; Sykora, M.A.; Mahaguna, V. Investigation of excipient type and level on drug release from controlled release tablets containing HPMC. Pharm. Dev. Technol. 2002, 7, 181–193. [Google Scholar] [CrossRef] [PubMed]
- Thomas, M.V.; Puleo, D.A. Calcium sulfate: Properties and clinical applications. J. Biomed. Mater. Res. Part B Appl. Biomater. 2009, 88, 597–610. [Google Scholar] [CrossRef] [PubMed]
- Singh, N.B.; Middendorf, B. Calcium sulphate hemihydrate hydration leading to gypsum crystallization. Prog. Cryst. Growth Charact. Mater. 2007, 53, 57–77. [Google Scholar] [CrossRef]
- Bešter-Rogač, M. Nonsteroidal Anti-Inflammatory Drugs Ion Mobility: A Conductometric Study of Salicylate, Naproxen, Diclofenac and Ibuprofen Dilute Aqueous Solutions. Acta Chim. Slov. 2009, 56, 70–77. [Google Scholar]







| Bioink | P (kg/m3) | (mPa·s) | Z | ||
|---|---|---|---|---|---|
| Hydrophilic | 0.08 | 1022.27 | 57.76 | 9.83 | 6.99 |
| Hydrophobic | 0.08 | 1048.00 | 31.41 | 4.88 | 10.52 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Acosta-Vélez, G.F.; Linsley, C.S.; Zhu, T.Z.; Wu, W.; Wu, B.M. Photocurable Bioinks for the 3D Pharming of Combination Therapies. Polymers 2018, 10, 1372. https://doi.org/10.3390/polym10121372
Acosta-Vélez GF, Linsley CS, Zhu TZ, Wu W, Wu BM. Photocurable Bioinks for the 3D Pharming of Combination Therapies. Polymers. 2018; 10(12):1372. https://doi.org/10.3390/polym10121372
Chicago/Turabian StyleAcosta-Vélez, Giovanny F., Chase S. Linsley, Timothy Z. Zhu, Willie Wu, and Benjamin M. Wu. 2018. "Photocurable Bioinks for the 3D Pharming of Combination Therapies" Polymers 10, no. 12: 1372. https://doi.org/10.3390/polym10121372
APA StyleAcosta-Vélez, G. F., Linsley, C. S., Zhu, T. Z., Wu, W., & Wu, B. M. (2018). Photocurable Bioinks for the 3D Pharming of Combination Therapies. Polymers, 10(12), 1372. https://doi.org/10.3390/polym10121372





