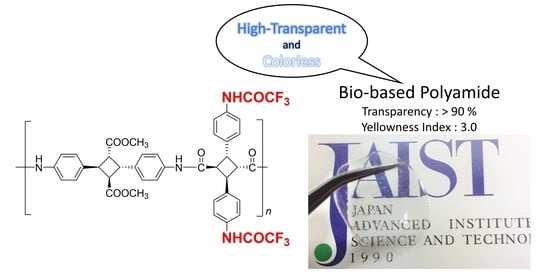
Fluorinated and Bio-Based Polyamides with High Transparencies and Low Yellowness Index
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Instruments
2.3. Synthesis of 4-(Trifluoroacetamido)cinnamic Acid
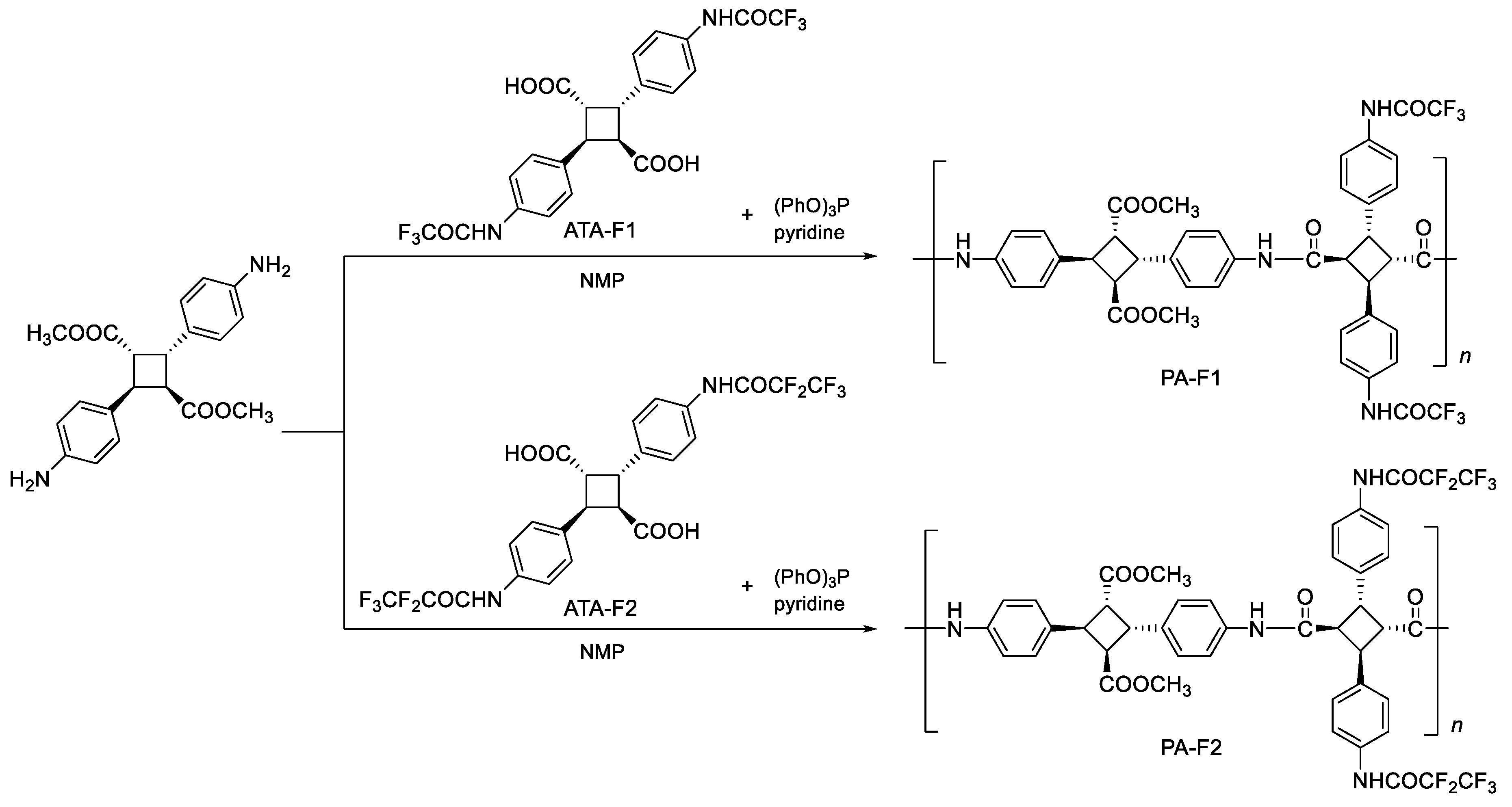
2.4. Synthesis of 4,4′-Bis(trifluoroacetamido)-α-Truxillic Acid (ATA-F1)
2.5. Synthesis of 4,4′-Bis(pentafluoropropionamido)-α-Truxillic Acid (ATA-F2)
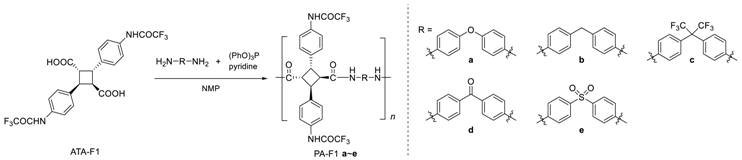
2.6. Synthesis of Bio-Based Polyamide Using ATA-F1, ATA-F2
3. Results and Discussion
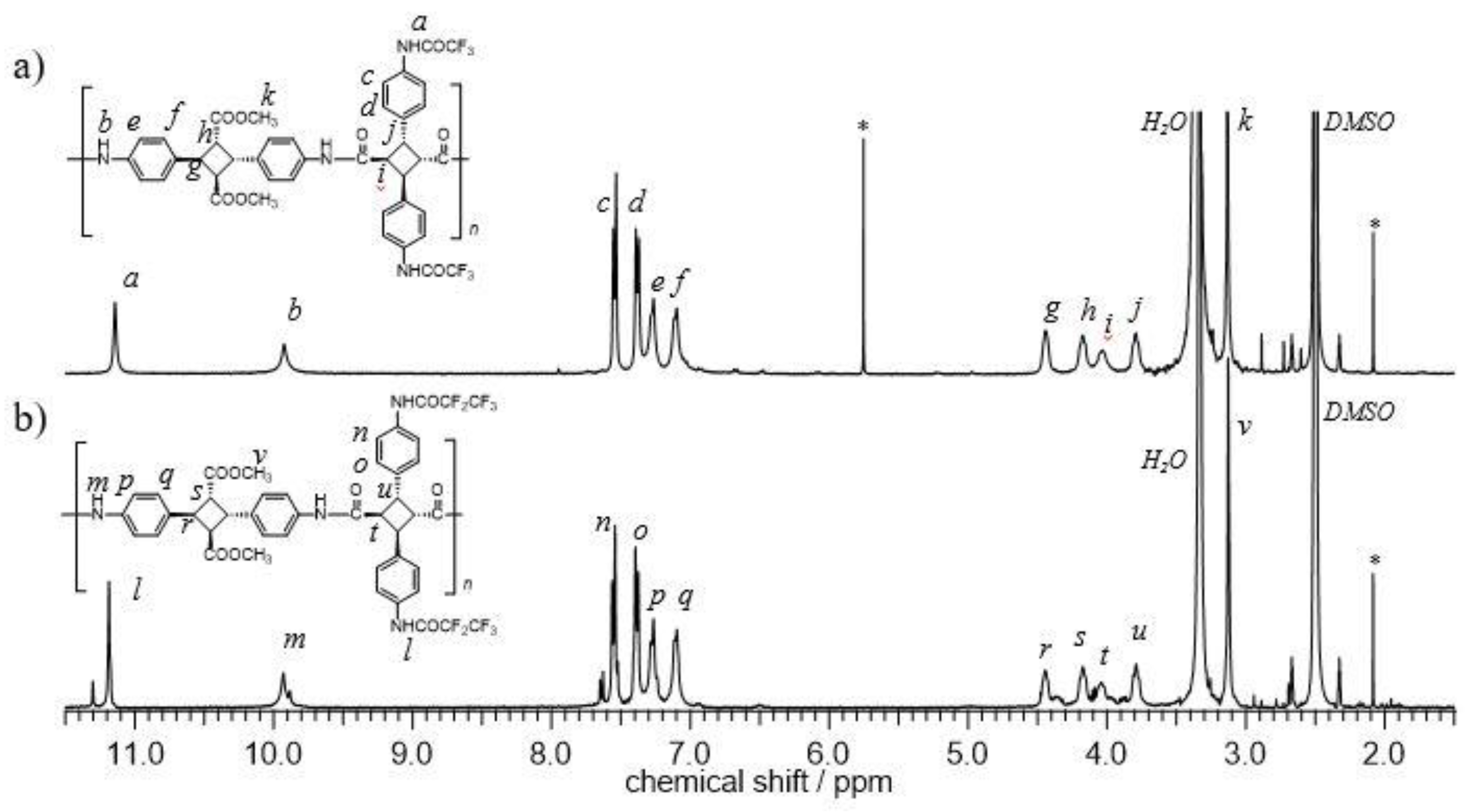
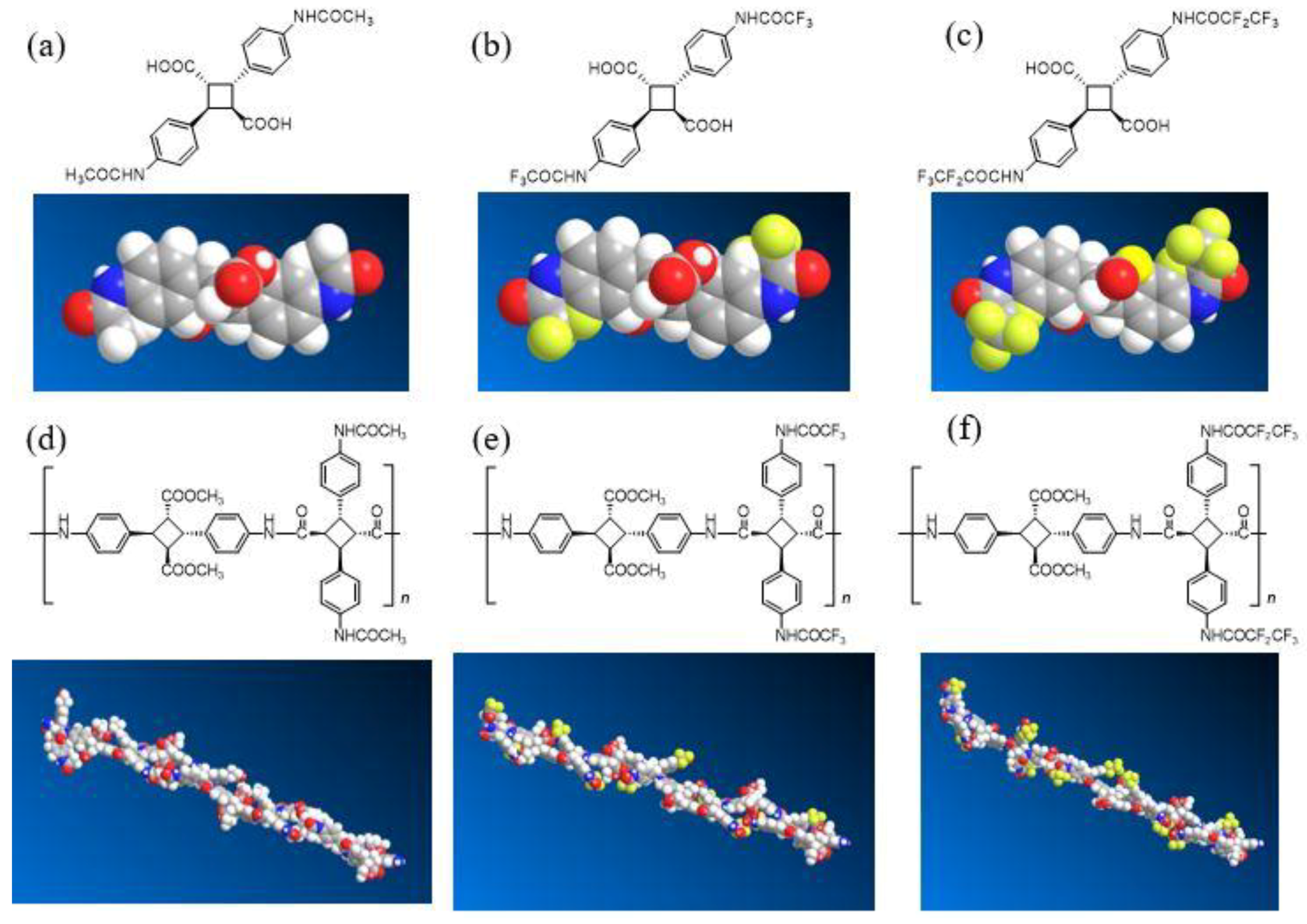
3.1. Synthesis and Structural Analysis of Fluorinated Bio-Based Polyamides (PA-F1 and PA-F2)
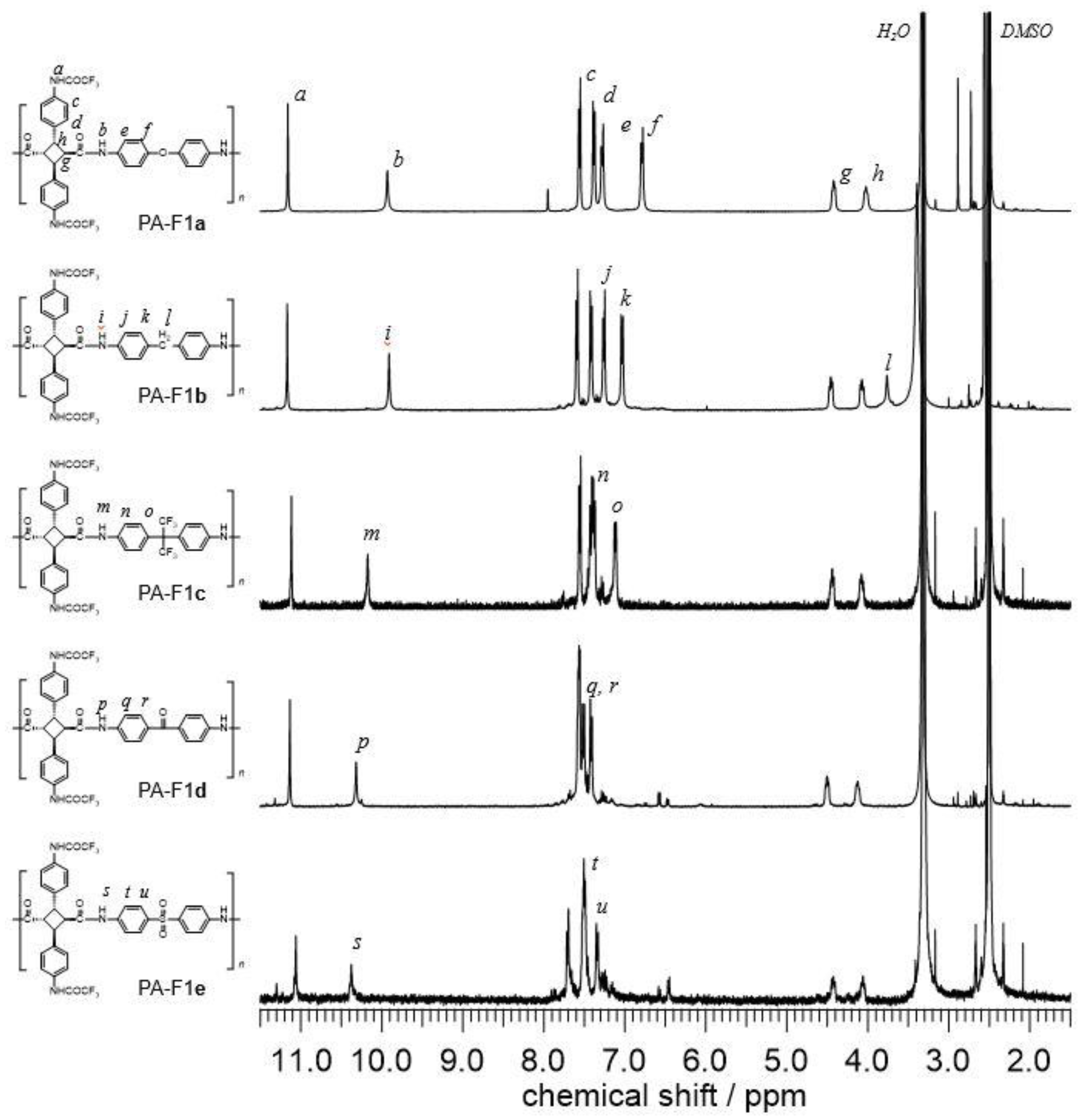
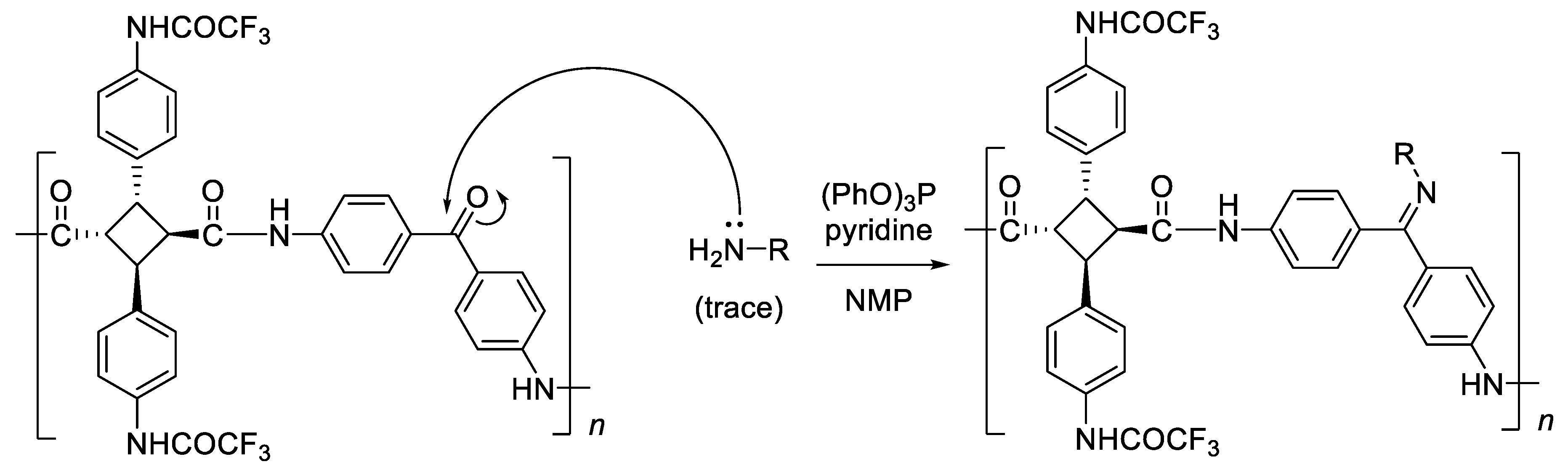
3.2. Synthesis of Various Types of Fluorinated Bio-Based Polyamides (PA-F1a–e)
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Rasal, R.M.; Janorkar, A.V.; Hirt, D.E. Poly(lactic acid) modifications. Prog. Polym. Sci. 2010, 35, 338–356. [Google Scholar] [CrossRef]
- Auras, R.; Harte, B.; Selke, S. An Overview of Polylactides as Packaging Materials. Macromol. Biosci. 2004, 4, 835–864. [Google Scholar] [CrossRef]
- Shin, H.; Wang, S.; Tateyama, S.; Kaneko, D.; Kaneko, T. Preparation of Ductile Biopolyimide Film by Copolymerization. Ind. Eng. Chem. Res. 2016, 55, 8761–8766. [Google Scholar] [CrossRef]
- Suvannasara, P.; Tateyama, S.; Miyasato, A.; Matsumura, K.; Shimoda, T.; Ito, T.; Yamagata, Y.; Fujita, T.; Takaya, N.; Kaneko, T. Bio-based polyimides from 4-aminocinnamic acid photodimer. Macromolecules 2014, 47, 1586–1593. [Google Scholar] [CrossRef]
- Tateyama, S.; Masuo, S.; Suvannasara, P.; Oka, Y.; Miyasato, A.; Yasaki, K.; Teerawatananond, T.; Muangsin, N.; Zhou, S.; Kawasaki, Y.; et al. Ultra-strong, transparent polytruxillamides derived from microbial photodimers. Macromolecules 2016, 49, 3336–3342. [Google Scholar] [CrossRef]
- Jin, X.; Tateyama, S.; Kaneko, T. Salt-induced reinforcement of anionic bio-polyureas with high transparency. Polym. J. 2015, 47, 727–732. [Google Scholar] [CrossRef]
- Mulliken, R.S. Molecular Compounds and their Spectra. II. J. Am. Chem. Soc. 1952, 74, 811–824. [Google Scholar] [CrossRef]
- Matsumura, T.; Hasuda, Y.; Nishi, S.; Yamada, N. Polyimide derived from 2,2′-bis(trifluoromethyl)-4,4′-diaminobiphenyl. 1. Synthesis and characterization of polyimides prepared with 2,2′-bis(3,4-dicarboxyphenyl)hexafluoropropane dianhydride or pyromellitic dianhydride. Macromolecules 1991, 24, 5001–5005. [Google Scholar] [CrossRef]
- Ando, S.; Matsuura, T.; Sasaki, S. Perfluorinated polyimide synthesis. Macromolecules 1992, 25, 5858–5860. [Google Scholar] [CrossRef]
- Dwivedi, S.; Sakamoto, S.; Kato, S.; Mitsumata, T.; Kaneko, T. Effect of biopolyimide molecular design on their silica hybrids thermo-mechanical, optical and electrical properties. RSC Adv. 2018, 8, 14009. [Google Scholar] [CrossRef]
| Polymer | Mn,GPC/g mol−1b | Mw/Mnb | Td5/°C c | Td10/°C c | Tg/°C d | Transparency (450 nm)/% e | YI e | Contact Angle/° |
|---|---|---|---|---|---|---|---|---|
| PA-F1 | 110,000 | 2.09 | 341 | 355 | 274 | 88 | 3.0 | 80 |
| PA-F2 | 69,000 | 2.02 | 343 | 358 | 270 | 91 | 3.7 | 85 |
| PA f | 10,000 | 1.99 | 346 | 361 | 273 | 86 | 4.9 | 82 |
 | ||||||||
| Polymer | Mn,GPC/g mol−1b | Mw/Mnb | Td5/°C c | Td10/°C c | Tg/°C d | Transparency (450 nm)/% e | YI e | Contact Angle/° |
| PA-F1a | 190,000 | 1.59 | 346 | 359 | 283 | 97 | 3.0 | 81 |
| PA-F1b | 61,000 | 1.53 | 323 | 334 | 255 | 96 | 4.4 | 83 |
| PA-F1c | 47,400 | 1.37 | 314 | 329 | 254 | 98 | 4.3 | 82 |
| PA-F1d | 31,700 | 1.42 | 338 | 359 | 300 | 90 | 9.9 | 79 |
| PA-F1e | 34,200 | 1.37 | 339 | 355 | 263 | 97 | 3.4 | 78 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Takada, K.; Mae, Y.; Kaneko, T. Fluorinated and Bio-Based Polyamides with High Transparencies and Low Yellowness Index. Polymers 2018, 10, 1311. https://doi.org/10.3390/polym10121311
Takada K, Mae Y, Kaneko T. Fluorinated and Bio-Based Polyamides with High Transparencies and Low Yellowness Index. Polymers. 2018; 10(12):1311. https://doi.org/10.3390/polym10121311
Chicago/Turabian StyleTakada, Kenji, Yuko Mae, and Tatsuo Kaneko. 2018. "Fluorinated and Bio-Based Polyamides with High Transparencies and Low Yellowness Index" Polymers 10, no. 12: 1311. https://doi.org/10.3390/polym10121311
APA StyleTakada, K., Mae, Y., & Kaneko, T. (2018). Fluorinated and Bio-Based Polyamides with High Transparencies and Low Yellowness Index. Polymers, 10(12), 1311. https://doi.org/10.3390/polym10121311