Creation of Superhydrophobic Poly(L-phenylalanine) Nonwovens by Electrospinning
Abstract
:1. Introduction
2. Materials and Methods
2.1. Synthesis and Characterization of PolyPhe and Investigation of Its Solubility in Various Solvents
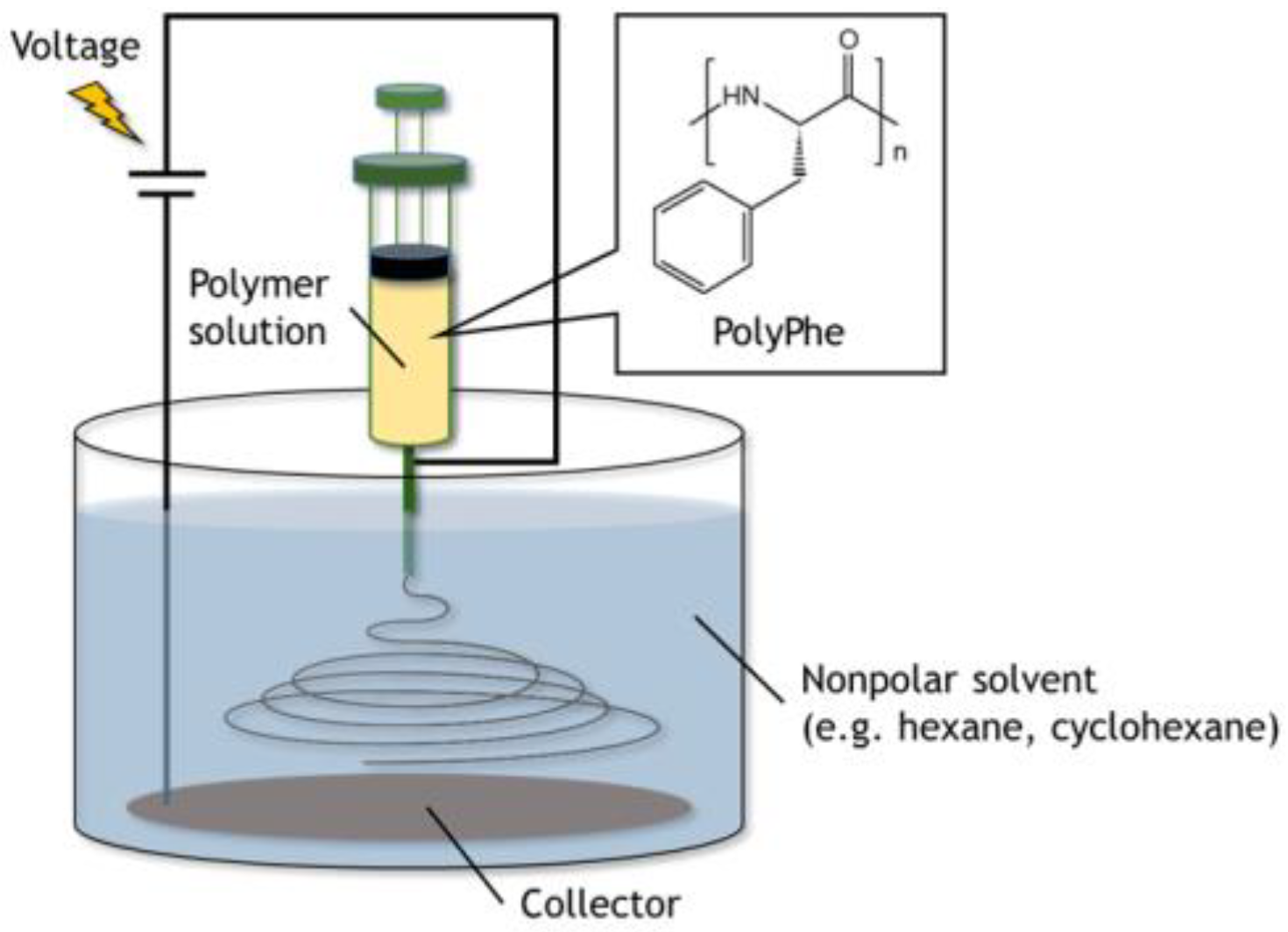
2.2. Electrospinning of PolyPhe in the Air and in the Nonpolar Solvents
2.3. Water Contact Angle (CA) Measurement on the Surface of PolyPhe Nonwovens
2.4. Hydrolysis Test of PolyPhe Nonwovens under Acidic, Basic, Enzymatic Conditions
3. Results and Discussion
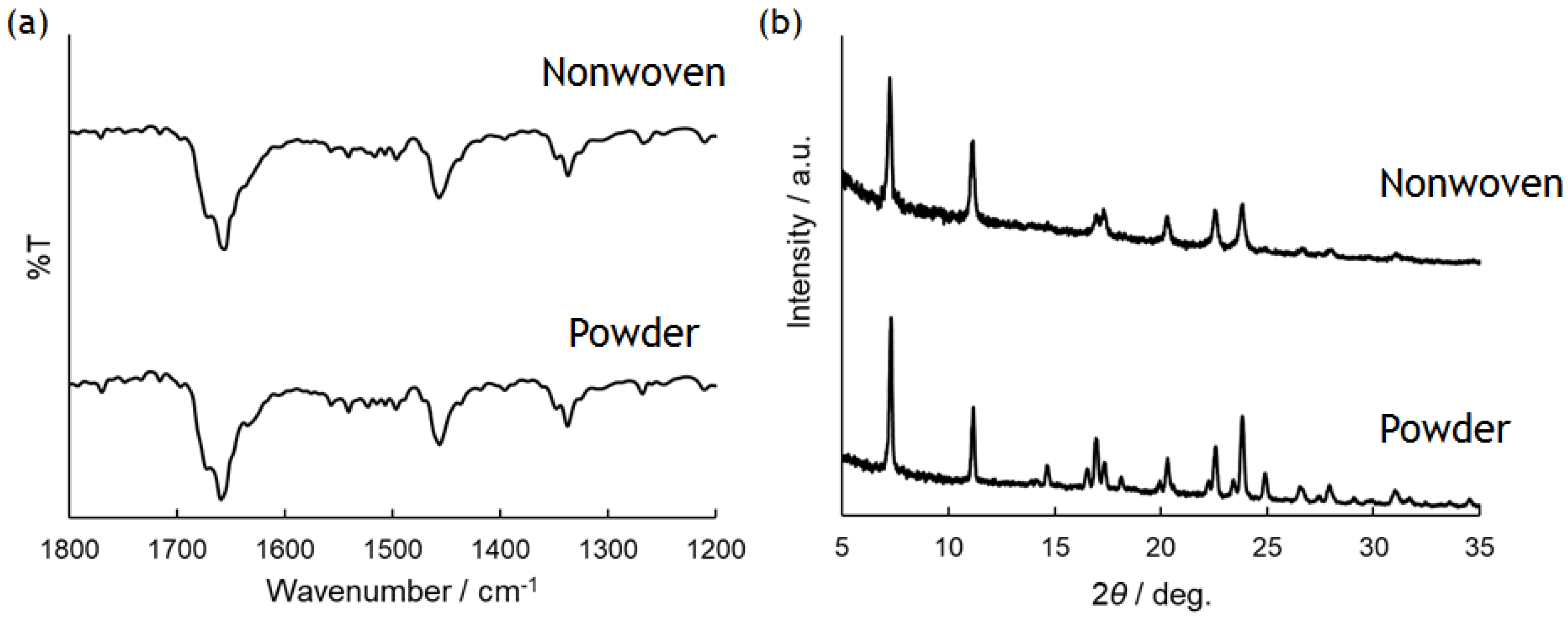
3.1. Solubility of PolyPhe in Various Solvents
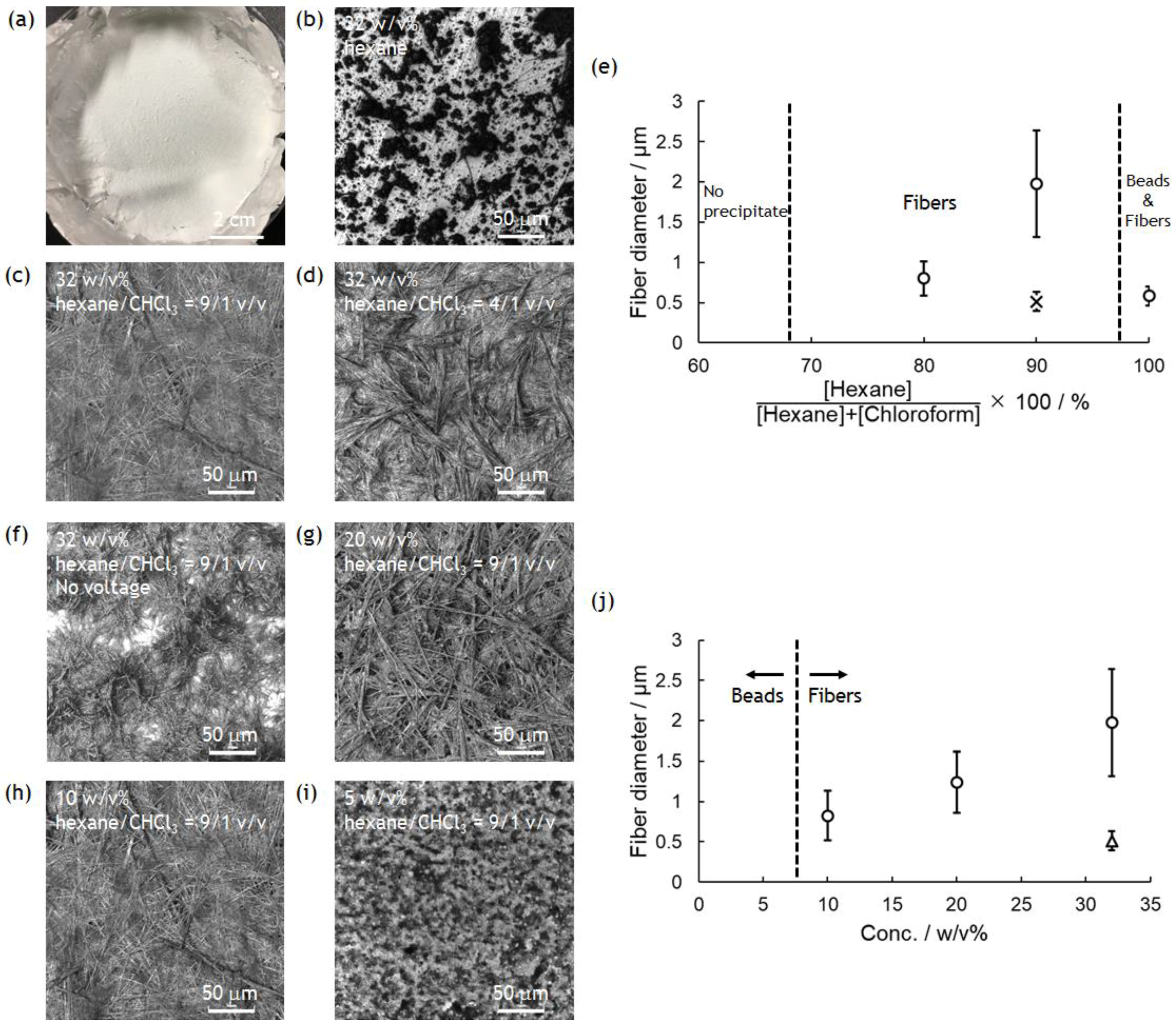
3.2. Electrospinning for the Preparation of PolyPhe Nonwovens
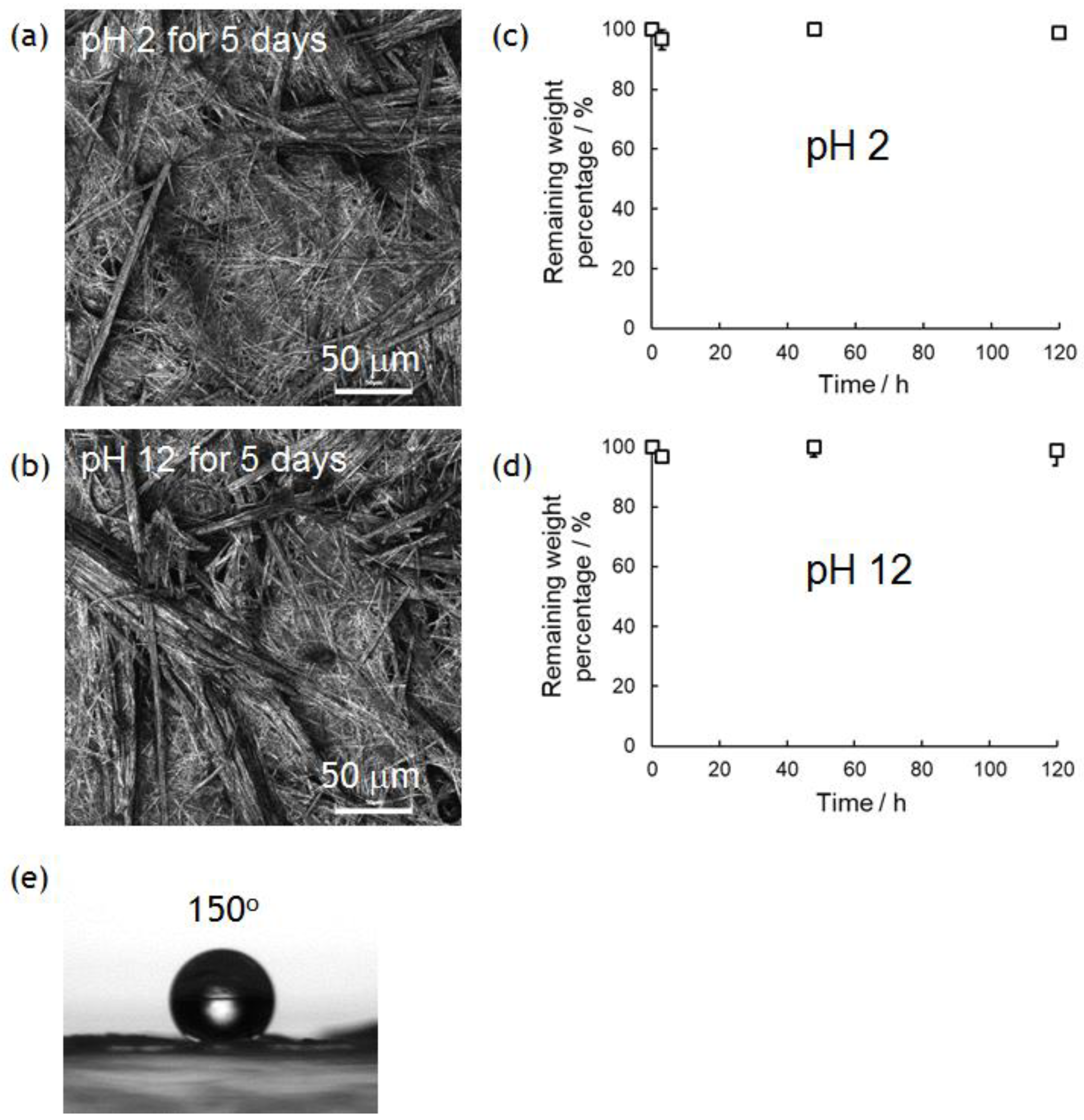
3.3. Wettability of PolyPhe Nonwovens
3.4. Chemical Stability of PolyPhe Nonwovens
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Feng, X.; Jiang, L. Design and creation of superwetting/antiwetting surfaces. Adv. Mater. 2006, 18, 3063–3078. [Google Scholar] [CrossRef]
- Gao, L.; McCarthy, T.J. Artificial lotus leaf prepared using a 1945 patent and a commercial textile. Langmuir 2006, 22, 5998–6000. [Google Scholar] [CrossRef] [PubMed]
- Roach, P.; Shirtcliffe, N.J.; Newton, M.I. Progress in superhydrophobic surface development. Soft Matter 2008, 4, 224–240. [Google Scholar] [CrossRef]
- Guo, Z.; Liu, W.; Su, B. Superhydrophobic surfaces: From natural to biomimetic to functional. J. Colloid Interface Sci. 2011, 353, 335–355. [Google Scholar] [CrossRef] [PubMed]
- Zhu, H.; Guo, Z.; Liu, W. Adhesion behaviors on superhydrophobic surfaces. Chem. Commun. 2014, 50, 3900–3913. [Google Scholar] [CrossRef] [PubMed]
- Lindström, A.B.; Strynar, M.J.; Libelo, E.L. Polyfluorinated compounds: Past, present, and future. Environ. Sci. Technol. 2011, 45, 7954–7961. [Google Scholar] [CrossRef] [PubMed]
- Osawa, S.; Yabe, M.; Miyamura, M.; Mizuno, K. Preparation of super-hydrophobic surface on biodegradable polymer by transcribing microscopic pattern of water-repellent leaf. Polymer 2006, 47, 3711–3714. [Google Scholar] [CrossRef]
- Shi, J.; Alves, N.M.; Mano, J.F. Towards bioinspired superhydrophobic poly (L-lactic acid) surfaces using phase inversion-based methods. Bioinspir. Biomim. 2008, 3, 034003. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Song, W.; Veiga, D.D.; Custodio, C.A.; Mano, J.F. Bioinspired degradable substrates with extreme wettability properties. Adv. Mater. 2009, 21, 1830–1834. [Google Scholar] [CrossRef]
- Yoshida, H.; Klee, D.; Möller, M.; Akashi, M. Creation of superhydrophobic electrospun nonwovens fabricated from naturally occurring poly(amino acid) derivatives. Adv. Funct. Mater. 2014, 24, 6359–6364. [Google Scholar] [CrossRef]
- Jacobs, R.E.; White, S.H. The nature of the hydrophobic binding of small peptides at the bilayer interface: Implications for the insertion of transbilayer helices. Biochemistry 1989, 28, 3421–3437. [Google Scholar] [CrossRef] [PubMed]
- Michael, S.; Berger, A. The terminal groups of poly-α-amino acids. J. Am. Chem. Soc. 1955, 77, 1893–1898. [Google Scholar]
- Daly, W.H.; Poché, D. The preparation of N-carboxyanhydrides of α-amino acids using bis (trichloromethyl) carbonate. Tetrahedron Lett. 1998, 29, 5859–5862. [Google Scholar] [CrossRef]
- Kricheldorf, H.R.; Mueller, D. Secondary structure of peptides. 3. Carbon-13 NMR cross polarization/magic angle spinning spectroscopic characterization of solid polypeptides. Macromolecules 1983, 16, 615–623. [Google Scholar] [CrossRef]
- Kamei, Y.; Sudo, A.; Nishida, H.; Kikukawa, K.; Endo, T. Synthesis of polypeptides from activated urethane derivatives of α-amino acids. J. Polym. Sci. A Polym. Chem. 2008, 46, 2525–2535. [Google Scholar] [CrossRef]
- Matsusaki, M.; Matsumoto, M.; Waku, T.; Akashi, M. Self-assembled structure of peptide nanospheres induces high stability against hydrolysis and sterilization. J. Biomater. Sci. Polym. Ed. 2011, 22, 1035–1048. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Xia, Y. Electrospinning of nanofibers: Reinventing the wheel? Adv. Mater. 2004, 16, 1151–1170. [Google Scholar] [CrossRef]
- Xu, C.Y.; Inai, R.; Kotaki, M.; Ramakrishna, S. Aligned biodegradable nanofibrous structure: A potential scaffold for blood vessel engineering. Biomaterials 2004, 25, 877–886. [Google Scholar] [CrossRef]
- Lu, X.; Wang, C.; Wei, Y. One-dimensional composite nanomaterials: Synthesis by electrospinning and their applications. Small 2009, 5, 2349–2370. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Wang, X.; Liu, X.; Lv, C.; Wang, Y.; Zheng, G.; Liu, H.; Liu, C.; Guo, Z.; Shen, C. Porous polyethylene bundles with enhanced hydrophobicity and pumping oil-recovery ability via skin-peeling. ACS Sustain. Chem. Eng. 2018, 6, 12580–12585. [Google Scholar] [CrossRef]
- Zhang, F.; Liu, X.; Zheng, G.; Guo, Z.; Liu, C.; Shen, C. Facile route to improve the crystalline memory effect: Electrospun composite fiber and annealing. Macromol. Chem. Phys. 2018, 219, 1800236. [Google Scholar] [CrossRef]
- Singh, G.; Bittner, A.M.; Loscher, S.; Malinowski, N.; Kern, K. Electrospinning of diphenylalanine nanotubes. Adv. Mater. 2008, 20, 2332–2336. [Google Scholar] [CrossRef]
- Akagi, T.; Watanabe, K.; Kim, H.; Akashi, M. Stabilization of polyion complex nanoparticles composed of poly (amino acid) using hydrophobic interactions. Langmuir 2009, 26, 2406–2413. [Google Scholar] [CrossRef] [PubMed]
- Zhu, P.; Yan, X.; Su, Y.; Yang, Y.; Li, J. Solvent-induced structural transition of self-assembled dipeptide: From organogels to microcrystals. Chem. Eur. J. 2010, 16, 3176–3183. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, H.; Klinkhammer, K.; Matsusaki, M.; Möller, M.; Klee, D.; Akashi, M. Disulfide-crosslinked electrospun poly (γ-glutamic acid) nonwovens as reduction-responsive scaffolds. Macromol. Biosci. 2009, 9, 568–574. [Google Scholar] [CrossRef] [PubMed]
- Khadka, D.B.; Haynie, D.T. Insoluble synthetic polypeptide mats from aqueous solution by electrospinning. ACS Appl. Mater. Interfaces 2010, 2, 2728–2732. [Google Scholar] [CrossRef] [PubMed]
- Farrar, D.; Ren, K.; Cheng, D.; Kim, S.; Moon, W.; Wilson, W.L.; Yu, S.M. Permanent polarity and piezoelectricity of electrospun α-helical poly (α-amino acid) fibers. Adv. Mater. 2011, 23, 3954–3958. [Google Scholar] [CrossRef] [PubMed]
- Khadka, D.B.; Cross, M.C.; Haynie, D.T. A synthetic polypeptide electrospun biomaterial. ACS Appl. Mater. Interfaces 2011, 3, 2994–3001. [Google Scholar] [CrossRef] [PubMed]
- Wakisaka, A.; Tsuchiya, M. Submerged Electrospray Method and Submerged Electrospray Device. JP Patent 5845525B2, 20 January 2015. [Google Scholar]
- Tsuchiya, M.; Wakisaka, A.; Kobara, H. Influence of liquid medium on the electrospinning of cellulose acetate in the liquid. Polym. Prep. Jpn. 2015, 64, 2Pb014. [Google Scholar]
- Young, C.J.; Poole-Warren, L.A.; Martens, P.J. Combining submerged electrospray and UV photopolymerization for production of synthetic hydrogel microspheres for cell encapsulation. Biotechnol. Bioeng. 2012, 109, 1561–1570. [Google Scholar] [CrossRef] [PubMed]
- Marin, A.G.; Loscertales, I.G.; Barrero, A. Surface tension effects on submerged electrosprays. Biomicrofluidics 2012, 6, 044104. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ido, Y.; Kobara, H.; Tominaga, K.I.; Wakisaka, A. A reactor system using electrospray in the liquid phase and its application in selective cyclosiloxane synthesis. Ind. Eng. Chem. Res. 2017, 56, 4878–4882. [Google Scholar] [CrossRef]
- Jackson, M.; Mantsch, H.H. The use and misuse of FTIR spectroscopy in the determination of protein structure. Crit. Rev. Biochem. Mol. 1995, 30, 95–120. [Google Scholar] [CrossRef] [PubMed]
- Rawal, A. Design parameters for a robust superhydrophobic electrospun nonwoven mat. Langmuir 2012, 28, 3285–3289. [Google Scholar] [CrossRef] [PubMed]
- Nishikawa, N.; Mayama, H.; Nonomura, Y.; Fujinaga, N.; Yokojima, S.; Nakamura, S.; Uchida, K. Theoretical explanation of the photoswitchable superhydrophobicity of diarylethene microcrystalline surfaces. Langmuir 2014, 30, 10643–10650. [Google Scholar] [CrossRef] [PubMed]
- Hu, L.; Zhang, S.; Zhang, Y.; Li, B. A flexible nanofiber-based membrane with superhydrophobic pinning properties. J. Colloid Interface Sci. 2016, 472, 167–172. [Google Scholar] [CrossRef] [PubMed]
- Wenzel, R.N. Resistance of solid surfaces to wetting by water. Ind. Eng. Chem. 1936, 28, 988–994. [Google Scholar] [CrossRef]
- Lembach, A.N.; Tan, H.-B.; Roisman, I.V.; Gambaryan-Roisman, T.; Zhang, Y.; Tropea, C.; Yarin, A.L. Drop impact, spreading, splashing, and penetration into electrospun nanofiber mats. Langmuir 2010, 26, 9516–9523. [Google Scholar] [CrossRef] [PubMed]
- Sahu, R.P.; Sinha-Ray, S.; Yarin, A.L.; Pourdeyhimi, B. Drop impacts on electrospun nanofiber membranes. Soft Matter 2012, 8, 3957–3970. [Google Scholar] [CrossRef]
- Sahu, R.P.; Sett, S.; Yarina, A.L.; Pourdeyhimi, B. Impact of aqueous suspension drops onto non-wettable porousmembranes: Hydrodynamic focusing and penetration of nanoparticles. Colloids Surf. A Physicochem. Eng. Asp. 2015, 467, 31–45. [Google Scholar] [CrossRef]
- Baker, L.E. New synthetic substrates for pepsin. J. Biol. Chem. 1951, 193, 809–819. [Google Scholar] [PubMed]

© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yoshida, H.; Yanagisawa, K. Creation of Superhydrophobic Poly(L-phenylalanine) Nonwovens by Electrospinning. Polymers 2018, 10, 1212. https://doi.org/10.3390/polym10111212
Yoshida H, Yanagisawa K. Creation of Superhydrophobic Poly(L-phenylalanine) Nonwovens by Electrospinning. Polymers. 2018; 10(11):1212. https://doi.org/10.3390/polym10111212
Chicago/Turabian StyleYoshida, Hiroaki, and Kazuhiro Yanagisawa. 2018. "Creation of Superhydrophobic Poly(L-phenylalanine) Nonwovens by Electrospinning" Polymers 10, no. 11: 1212. https://doi.org/10.3390/polym10111212
APA StyleYoshida, H., & Yanagisawa, K. (2018). Creation of Superhydrophobic Poly(L-phenylalanine) Nonwovens by Electrospinning. Polymers, 10(11), 1212. https://doi.org/10.3390/polym10111212





