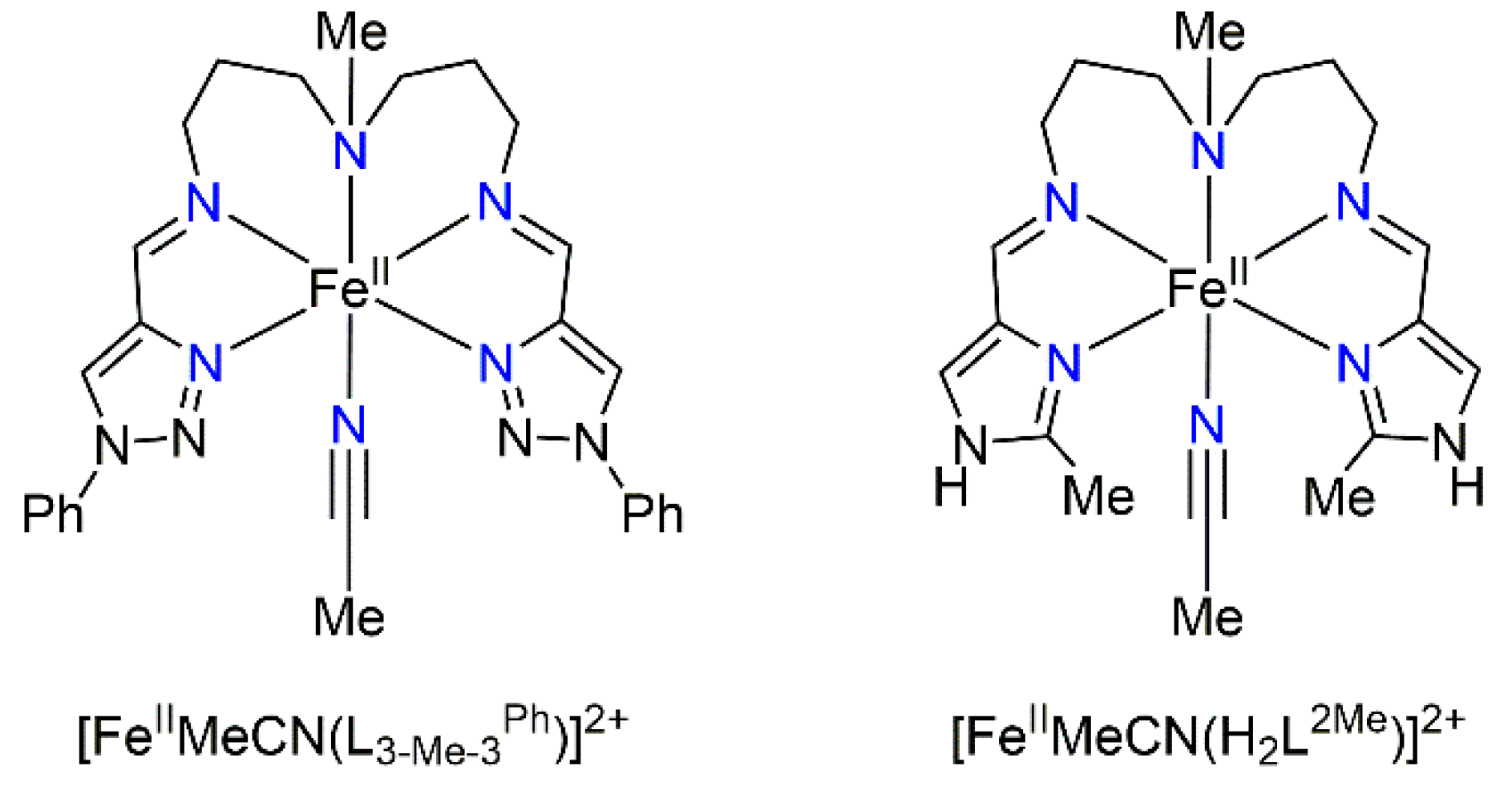
Iron(II) Spin Crossover Complex with the 1,2,3-Triazole-Containing Linear Pentadentate Schiff-Base Ligand and the MeCN Monodentate Ligand
Abstract
1. Introduction
2. Materials and Methods
2.1. Synthesis of FeII Complex
2.1.1. General
2.1.2. Synthesis of the Linear Pentadentate N5 Ligand L3-Me-3Ph = bis(N,N′-1-Phenyl-1H-1,2,3-triazol-4-yl-methylideneaminopropyl)methylamine
2.1.3. Preparation of [FeIIMeCN(L3-Me-3Ph)](BPh4)2·MeCN·H2O (1)
2.2. Physical Measurements
2.3. Crystallographic Data Collection and Structure Analyses
2.4. Computational Details
3. Results and Discussion
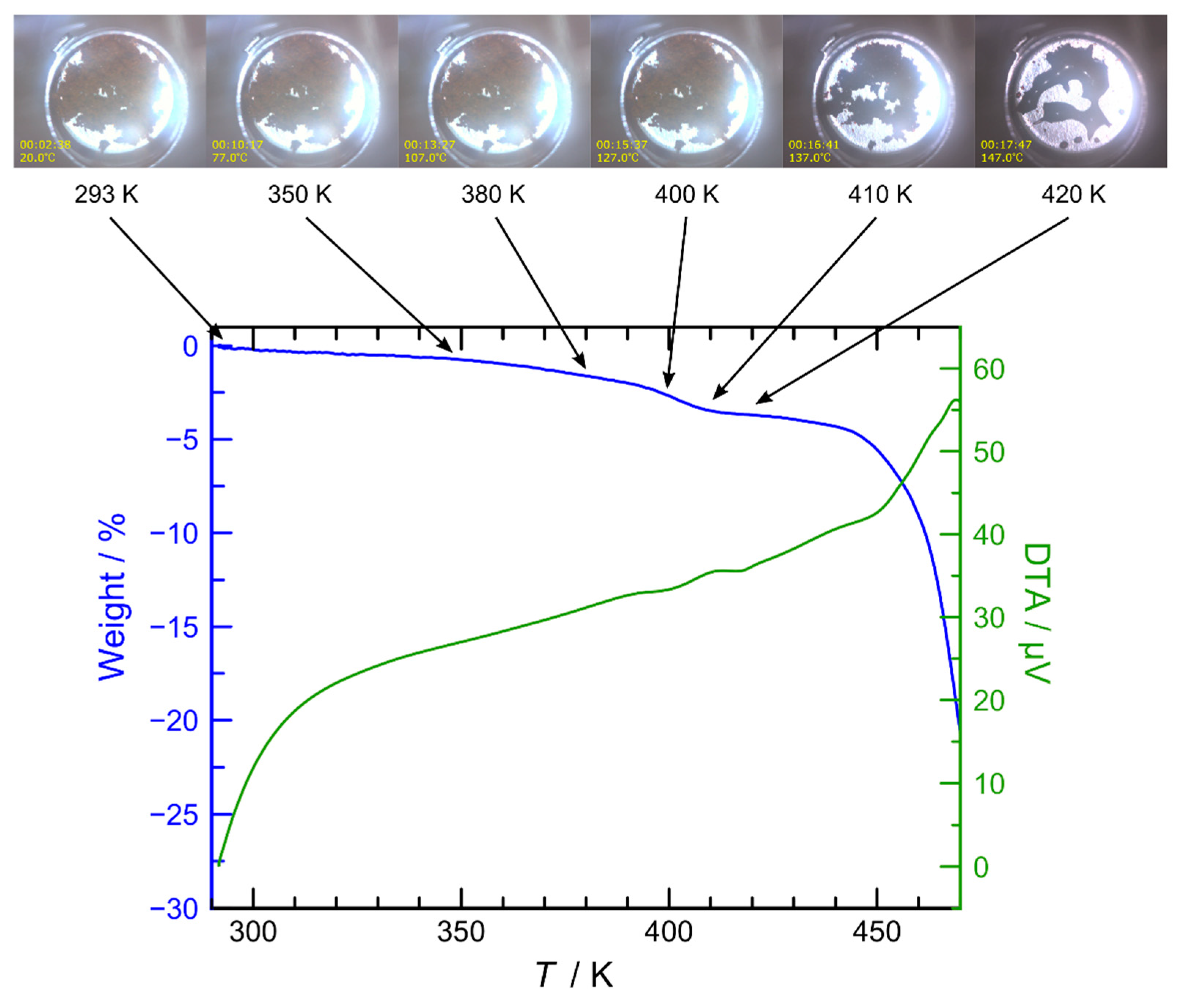
3.1. Synthesis and Characterization
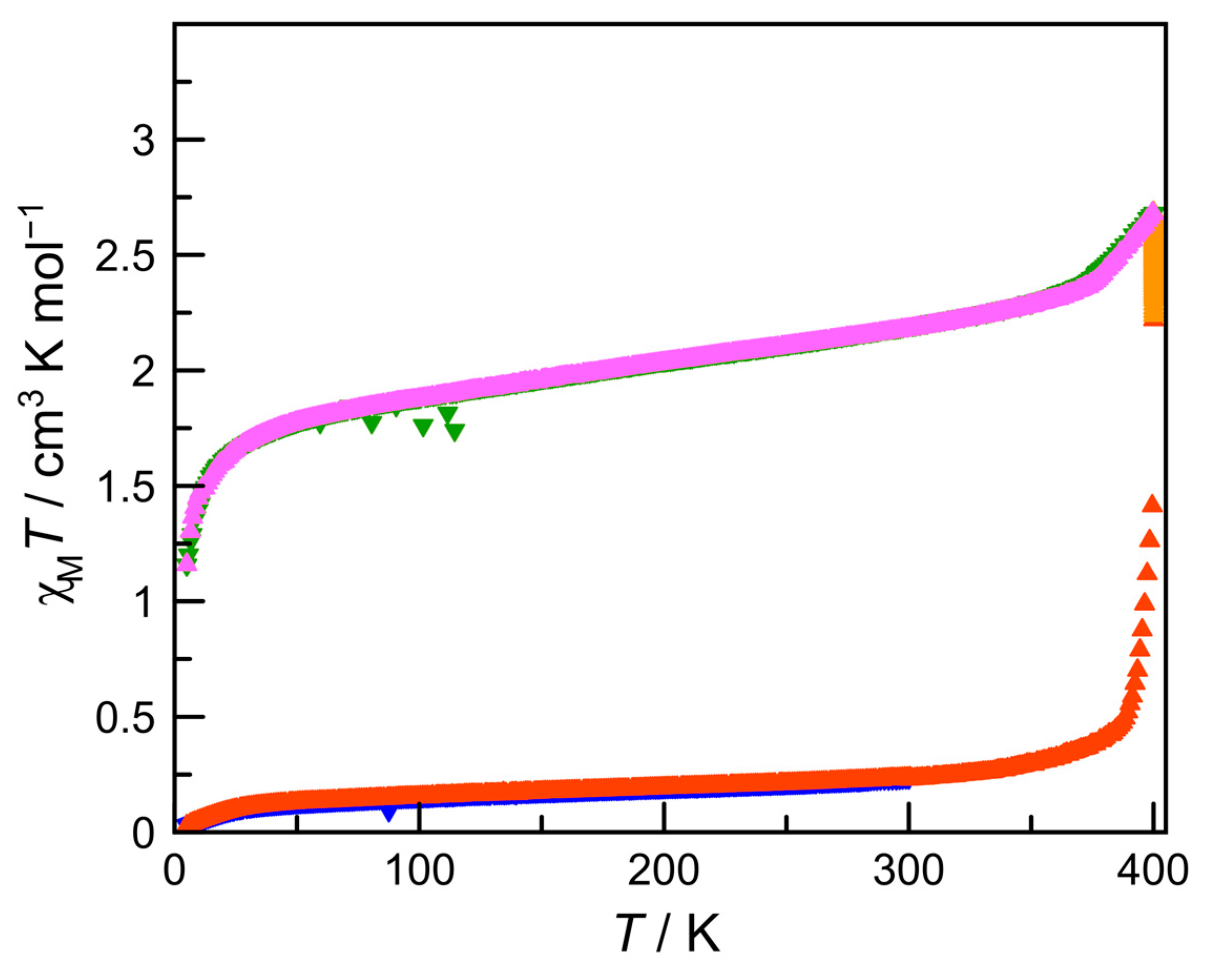
3.2. Magnetic Properties
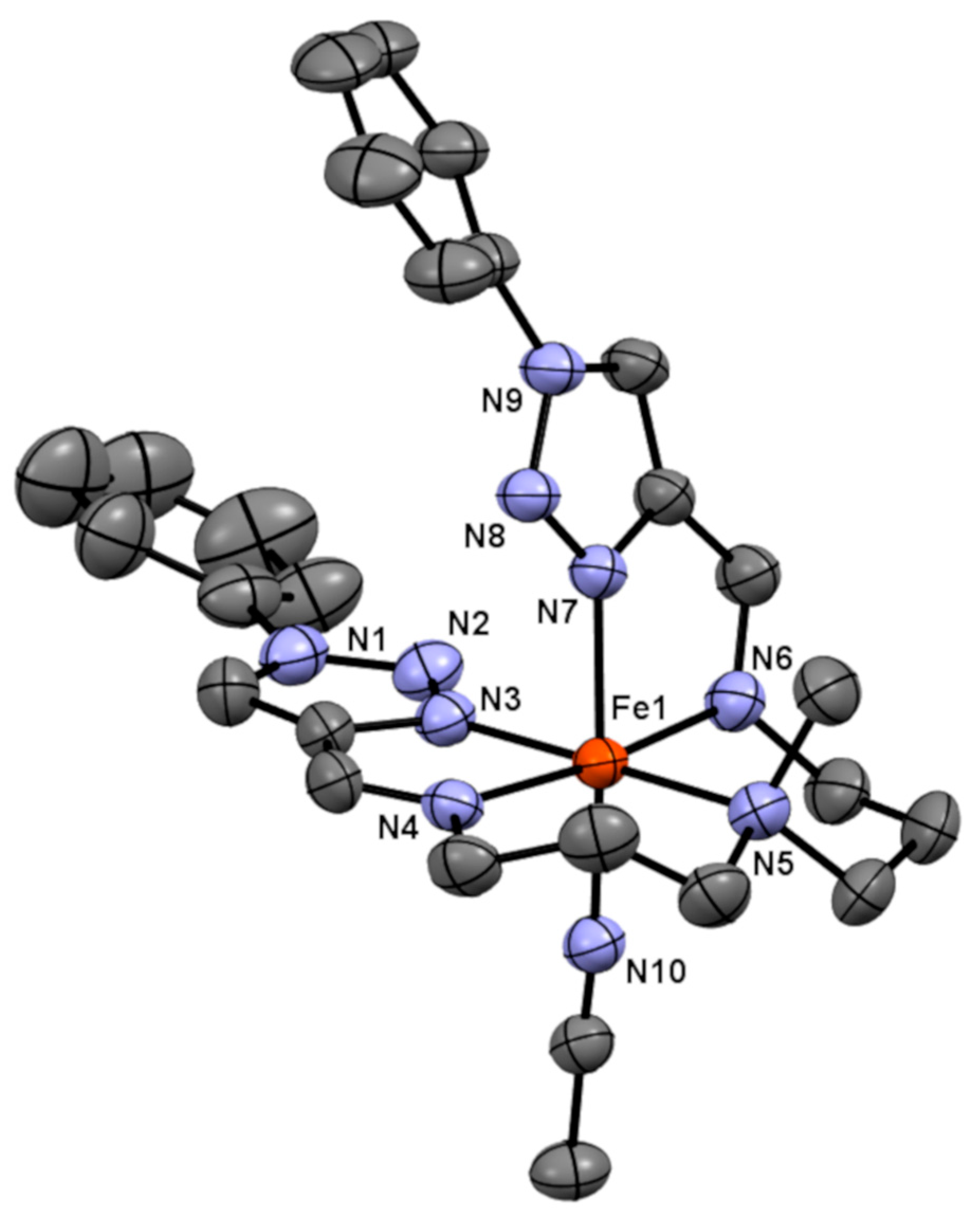
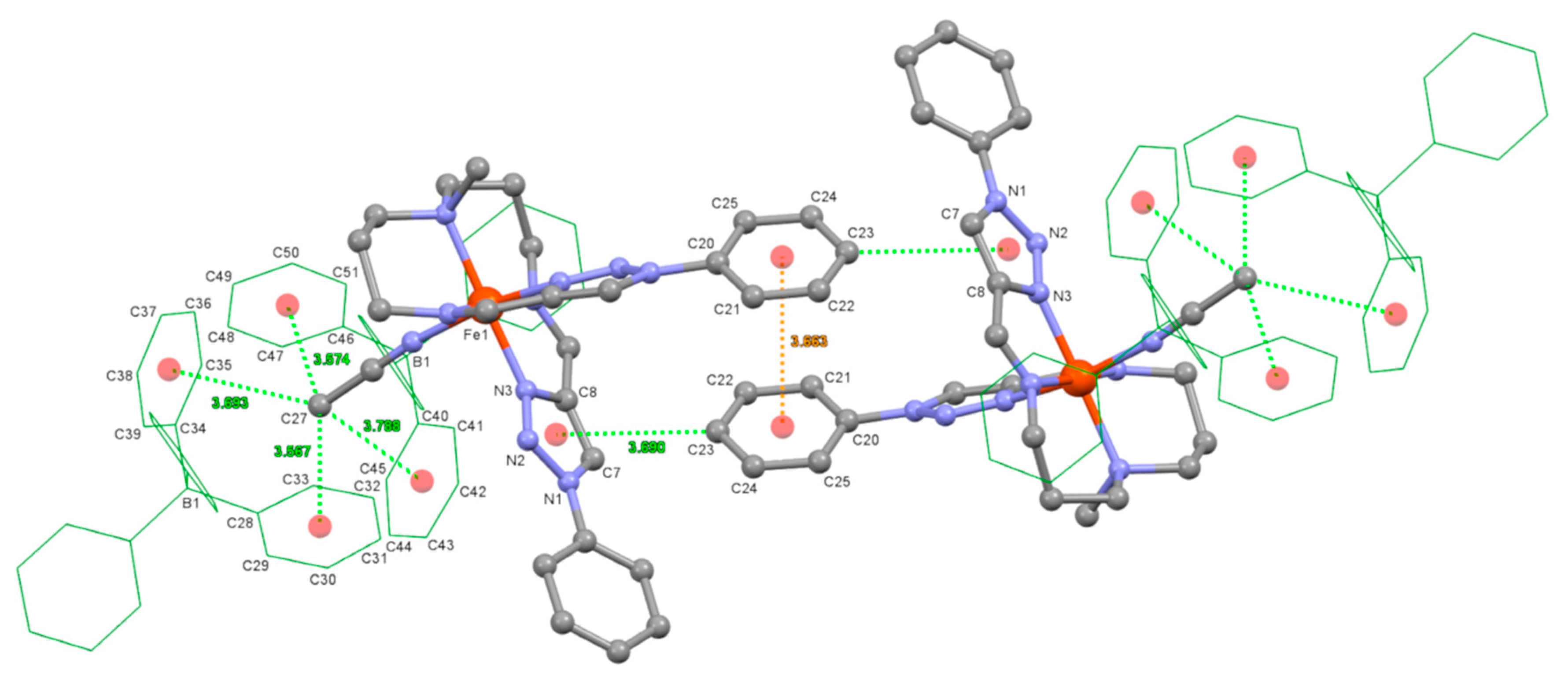
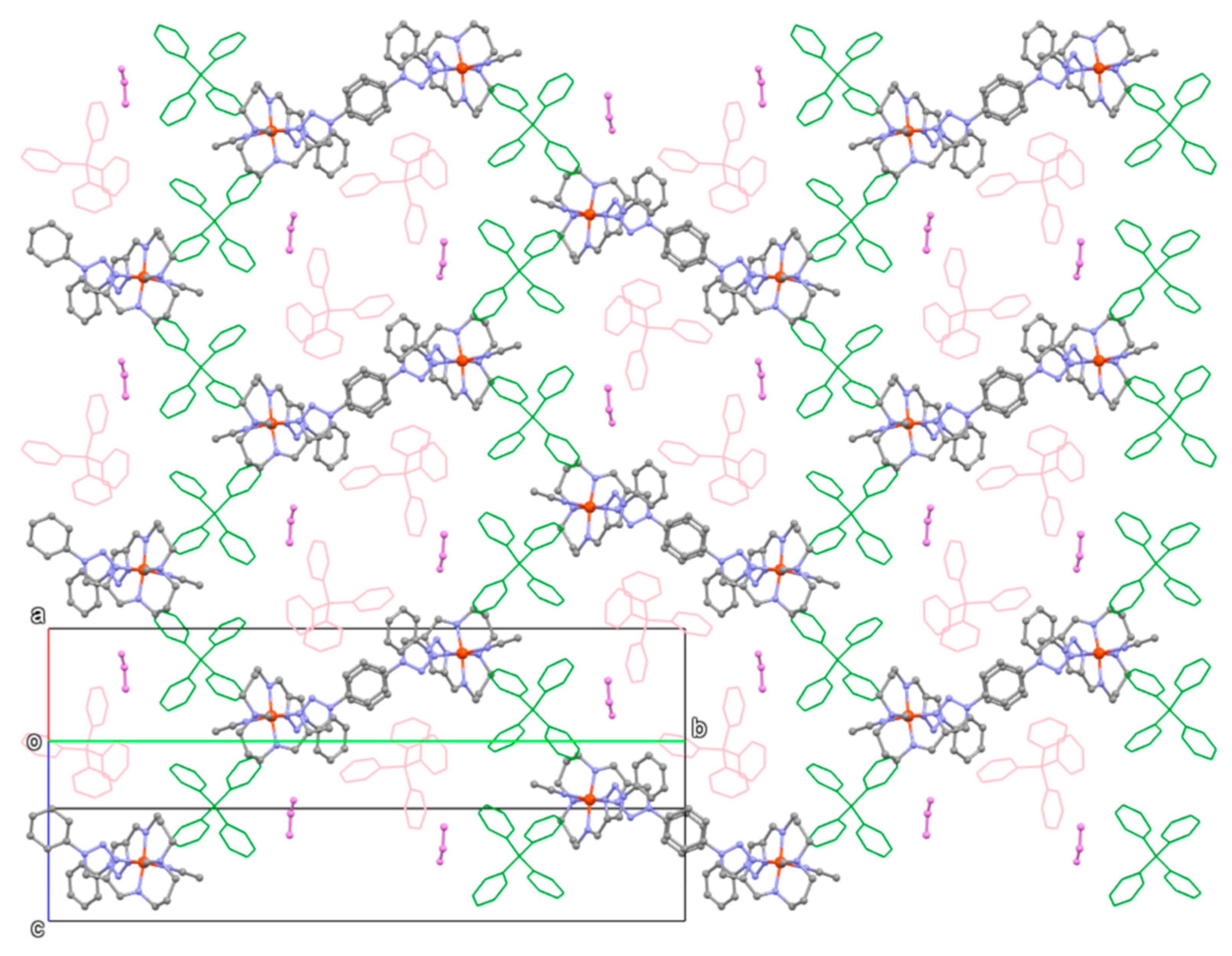
3.3. Crystal Structures
3.4. DFT Calculations
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Gütlich, P.; Goodwin, H.A. (Eds.) Spin Crossover in Transition Metal Compounds I-III; Topics in Current Chemistry; Springer: Berlin, Germany, 2004; Volumes 233–235. [Google Scholar]
- Halcrow, M.A. (Ed.) Spin-Crossover Materials–Properties and Applications; John Wiley & Sons: Chichester, UK, 2013. [Google Scholar]
- Gütlich, P.; Gaspar, A.B.; Garcia, Y. Spin state switching in iron coordination compounds. Beilstein J. Org. Chem. 2013, 9, 342–391. [Google Scholar] [CrossRef] [PubMed]
- Linares, J.; Codjovi, E.; Garcia, Y. Pressure and Temperature Spin Crossover Sensors with Optical Detection. Sensors 2012, 12, 4479–4492. [Google Scholar] [CrossRef] [PubMed]
- Gentili, D.; Demitri, N.; Schäfer, B.; Liscio, F.; Bergenti, I.; Ruani, G.; Ruben, M.; Cavallini, M. Multi-modal sensing in spin crossover compounds. J. Mater. Chem. C 2015, 3, 7836–7844. [Google Scholar] [CrossRef]
- Halcrow, M.A. Spin-crossover Compounds with Wide Thermal Hysteresis. Chem. Lett. 2014, 43, 1178–1188. [Google Scholar] [CrossRef]
- Brooker, S. Spin crossover with thermal hysteresis: Practicalities and lessons learnt. Chem. Soc. Rev. 2015, 44, 2880–2892. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, T.; Newton, G.N.; Shiga, T.; Hayami, S.; Matsui, Y.; Okamoto, H.; Kumai, R.; Murakami, Y.; Oshio, H. Programmable spin-state switching in a mixed-valence spin-crossover iron grid. Nat. Commun. 2014, 5, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.-Y.; Ohtsu, H.; Kojima, T.; Dai, J.-W.; Yoshida, T.; Breedlove, B.K.; Zhang, W.-X.; Iguchi, H.; Sato, O.; Kawano, M.; et al. Direct Observation of Ordered High-Spin-Low-Spin Intermediate States of an Iron(III) Three-Step Spin-Crossover Complex. Angew. Chem. Int. Ed. 2016, 55, 5184–5189. [Google Scholar] [CrossRef]
- Murphy, M.J.; Zenere, K.A.; Ragon, F.; Southon, P.D.; Kepert, C.J.; Neville, S.M. Guest programmable multlstep spin crossover in a porous 2-D Hofmann-type material. J. Am. Chem. Soc. 2017, 139, 1330–1335. [Google Scholar] [CrossRef]
- Zhang, D.; Trzop, E.; Valverde-Muñoz, F.J.; Piñeiro-López, L.; Muñoz, M.C.; Collet, E.; Real, J.A. Competing Phases Involving Spin-State and Ligand Structural Orderings in a Multistable Two-Dimensional Spin Crossover Coordination Polymer. Cryst. Growth Des. 2017, 17, 2736–2745. [Google Scholar] [CrossRef]
- Bao, X.; Guo, P.-H.; Liu, W.; Tucek, J.; Zhang, W.-X.; Leng, J.-D.; Chen, X.-M.; Gural’skiy, I.; Salmon, L.; Bousseksou, A.; et al. Remarkably high-temperature spin transition exhibited by new 2D metal–organic frameworks. Chem. Sci. 2012, 3, 1629. [Google Scholar] [CrossRef]
- Liu, W.; Bao, X.; Li, J.-Y.; Qin, Y.-L.; Chen, Y.-C.; Ni, Z.-P.; Tong, M.-L. High-Temperature Spin Crossover in Two Solvent-Free Coordination Polymers with Unusual High Thermal Stability. Inorg. Chem. 2015, 54, 3006–3011. [Google Scholar] [CrossRef]
- Zheng, S.; Reintjens, N.R.M.; Siegler, M.A.; Roubeau, O.; Bouwman, E.; Rudavskyi, A.; Havenith, R.W.A.; Bonnet, S. Stabilization of the Low-Spin State in a Mononuclear Iron(II) Complex and High-Temperature Cooperative Spin Crossover Mediated by Hydrogen Bonding. Chem. A Eur. J. 2016, 22, 331–339. [Google Scholar] [CrossRef]
- Hora, S.; Hagiwara, H. High-Temperature Wide Thermal Hysteresis of an Iron(II) Dinuclear Double Helicate. Inorganics 2017, 5, 49. [Google Scholar] [CrossRef]
- Craze, A.R.; Howard-Smith, K.J.; Bhadbhade, M.M.; Mustonen, O.; Kepert, C.J.; Marjo, C.E.; Li, F. Investigation of the High-Temperature Spin-Transition of a Mononuclear Iron(II) Complex Using X-ray Photoelectron Spectroscopy. Inorg. Chem. 2018, 57, 6503–6510. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, K.; Yamamoto, K.; Yamamoto, T.; Einaga, Y.; Shiota, Y.; Yoshizawa, K.; Mori, H. High-Temperature Cooperative Spin Crossover Transitions and Single-Crystal Reflection Spectra of [FeIII(qsal)2](CH3OSO3) and Related Compounds. Crystals 2019, 9, 81. [Google Scholar] [CrossRef]
- Hagiwara, H. High-temperature Spin Crossover of a Solvent-Free Iron(II) Complex with the Linear Hexadentate Ligand [Fe(L2-3-2Ph)](AsF6)2 (L2-3-2Ph = bis[N-(1-Phenyl-1H-1,2,3-triazol-4-yl)methylidene-2-aminoethyl]-1,3- propanediamine). Magnetochemistry 2019, 5, 10. [Google Scholar] [CrossRef]
- Weber, B.; Bauer, W.; Obel, J. An iron(II) spin-crossover complex with a 70 K wide thermal hysteresis loop. Angew. Chem. Int. Ed. 2008, 47, 10098–10101. [Google Scholar] [CrossRef]
- Hayami, S.; Gu, Z.Z.; Yoshiki, H.; Fujishima, A.; Sato, O. Iron(III) spin-crossover compounds with a wide apparent thermal hysteresis around room temperature. J. Am. Chem. Soc. 2001, 123, 11644–11650. [Google Scholar] [CrossRef] [PubMed]
- Kahn, O.; Martinez, C.J. Spin-transition polymers: From molecular materials toward memory devices. Science 1998, 279, 44–48. [Google Scholar] [CrossRef]
- Southon, P.D.; Liu, L.; Fellows, E.A.; Price, D.J.; Halder, G.J.; Chapman, K.W.; Moubaraki, B.; Murray, K.S.; Létard, J.F.; Kepert, C.J. Dynamic interplay between spin-crossover and host-guest function in a nanoporous metal-organic framework material. J. Am. Chem. Soc. 2009, 131, 10998–11009. [Google Scholar] [CrossRef]
- Arcis-Castíllo, Z.; Zheng, S.; Siegler, M.A.; Roubeau, O.; Bedoui, S.; Bonnet, S. Tuning the transition temperature and cooperativity of bapbpy-based mononuclear spin-crossover compounds: Interplay between molecular and crystal engineering. Chem. A Eur. J. 2011, 17, 14826–14836. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.-H.; Zhang, S.-L.; Shao, D.; Wang, X.-Y. Spin Crossover in [Fe(2-Picolylamine)3]2+ Adjusted by Organosulfonate Anions. Inorg. Chem. 2015, 54, 7857–7867. [Google Scholar] [CrossRef] [PubMed]
- Kershaw Cook, L.J.; Kulmaczewski, R.; Mohammed, R.; Dudley, S.; Barrett, S.A.; Little, M.A.; Deeth, R.J.; Halcrow, M.A. A Unified Treatment of the Relationship between Ligand Substituents and Spin State in a Family of Iron(II) Complexes. Angew. Chem. Int. Ed. 2016, 55, 4327–4331. [Google Scholar] [CrossRef]
- Hagiwara, H.; Masuda, T.; Ohno, T.; Suzuki, M.; Udagawa, T.; Murai, K.I. Neutral Molecular Iron(II) Complexes Showing Tunable Bistability at Above, Below, and Just Room Temperature by a Crystal Engineering Approach: Ligand Mobility into a Three-Dimensional Flexible Supramolecular Network. Cryst. Growth Des. 2017, 17, 6006–6019. [Google Scholar] [CrossRef]
- Nakanishi, T.; Okazawa, A.; Sato, O. Halogen Substituent Effect on the Spin-Transition Temperature in Spin-Crossover Fe(III) Compounds Bearing Salicylaldehyde 2-Pyridyl Hydrazone-Type Ligands and Dicarboxylic Acids. Inorganics 2017, 5, 53. [Google Scholar] [CrossRef]
- Phonsri, W.; Macedo, D.S.; Vignesh, K.R.; Rajaraman, G.; Davies, C.G.; Jameson, G.N.L.; Moubaraki, B.; Ward, J.S.; Kruger, P.E.; Chastanet, G.; et al. Halogen Substitution Effects on N2O Schiff Base Ligands in Unprecedented Abrupt FeII Spin Crossover Complexes. Chem. A Eur. J. 2017, 23, 7052–7065. [Google Scholar] [CrossRef] [PubMed]
- Kimura, A.; Ishida, T. Spin-Crossover Temperature Predictable from DFT Calculation for Iron(II) Complexes with 4-Substituted Pybox and Related Heteroaromatic Ligands. ACS Omega 2018, 3, 6737–6747. [Google Scholar] [CrossRef]
- Nemec, I.; Herchel, R.; Boča, R.; Trávníček, Z.; Svoboda, I.; Fuess, H.; Linert, W. Tuning of spin crossover behaviour in iron(III) complexes involving pentadentate Schiff bases and pseudohalides. Dalton Trans. 2011, 40, 10090–10099. [Google Scholar] [CrossRef]
- Tsuchida, R. Absorption Spectra of Co-ordination Compounds. Bull. Chem. Soc. Jpn. 1938, 13, 388–400. [Google Scholar] [CrossRef]
- Shimura, Y. A Quantitative Scale of the Spectrochemical Series for the Mixed Ligand Complexes of d6 Metals. Bull. Chem. Soc. Jpn. 1988, 61, 693–698. [Google Scholar] [CrossRef]
- Ohta, S.; Yoshimura, C.; Matsumoto, N.; Okawa, H.; Ohyoshi, A. The Synthesis, Magnetic, and Spectroscopic Properties of Binuclear Iron(III) Complexes Bridged by Pyrazine, 1,1′-Tetramethylenebis(imidazol), or Bis(pyridine) Compounds Exhibiting a Spin-Equilibrium Behavior. Bull. Chem. Soc. Jpn. 1986, 59, 155–159. [Google Scholar] [CrossRef]
- Hayami, S.; Inoue, K.; Osaki, S.; Maeda, Y. Synthesis and Magnetic Properties of Binuclear Iron(III) Complexes Containing Photoisomerization Ligand. Chem. Lett. 1998, 27, 987–988. [Google Scholar] [CrossRef]
- Spiccia, L.; Fallon, G.D.; Grannas, M.J.; Nichols, P.J.; Tiekink, E.R.T. Synthesis and characterisation of mononuclear and binuclear iron(II) complexes of pentadentate and bis(pentadentate) ligands derived from 1,4,7-triazacyclononane. Inorg. Chim. Acta 1998, 279, 192–199. [Google Scholar] [CrossRef]
- Boča, R.; Fukuda, Y.; Gembický, M.; Herchel, R.; Jaroščiak, R.; Linert, W.; Renz, F.; Yuzurihara, J. Spin crossover in mononuclear and binuclear iron(III) complexes with pentadentate Schiff-base ligands. Chem. Phys. Lett. 2000, 325, 411–419. [Google Scholar] [CrossRef]
- Hayami, S.; Hosokoshi, Y.; Inoue, K.; Einaga, Y.; Sato, O.; Maeda, Y. Pressure-Stabilized Low-Spin State for Binuclear Iron(III) Spin-Crossover Compounds. Bull. Chem. Soc. Jpn. 2001, 74, 2361–2368. [Google Scholar] [CrossRef]
- Kitashima, R.; Imatomi, S.; Yamada, M.; Matsumoto, N.; Maeda, Y. Gradual Two-step Spin Crossover Behavior of Binuclear Iron(III) Complex Bridged by trans -1,2-Bis(4-pyridyl)ethylene. Chem. Lett. 2005, 34, 1388–1389. [Google Scholar] [CrossRef]
- Šalitroš, I.; Boča, R.; Dlháň, L.; Gembický, M.; Kožíšek, J.; Linares, J.; Moncol, J.; Nemec, I.; Perašínová, L.; Renz, F.; et al. Unconventional spin crossover in dinuclear and trinuclear iron(III) complexes with cyanido and metallacyanido bridges. Eur. J. Inorg. Chem. 2009, 3141–3154. [Google Scholar] [CrossRef]
- Nemec, I.; Boča, R.; Herchel, R.; Trávníček, Z.; Gembický, M.; Linert, W. Dinuclear Fe(III) complexes with spin crossover. Monatsh. Chem. 2009, 140, 815–828. [Google Scholar] [CrossRef]
- Djukic, B.; Poddutoori, P.K.; Dube, P.A.; Seda, T.; Jenkins, H.A.; Lemaire, M.T. Bimetallic Iron(3+) Spin-Crossover Complexes Containing a 2,2′-Bithienyl Bridging bis-QsalH Ligand. Inorg. Chem. 2009, 48, 6109–6116. [Google Scholar] [CrossRef]
- Fujinami, T.; Nishi, K.; Kitashima, R.; Murakami, K.; Matsumoto, N.; Iijima, S.; Toriumi, K. One-step and two-step spin crossover binuclear iron(III) complexes bridged by 4,4′-bipyridine. Inorg. Chim. Acta 2011, 376, 136–143. [Google Scholar] [CrossRef]
- Boča, R.; Nemec, I.; Šalitroš, I.; Pavlik, J.; Herchel, R.; Renz, F. Interplay between spin crossover and exchange interaction in iron(III) complexes. Pure Appl. Chem. 2009, 81, 1357–1383. [Google Scholar] [CrossRef]
- Herchel, R.; Boča, R.; Gembický, M.; Kožíšek, J.; Renz, F. Spin Crossover in a Tetranuclear Cr(III)−Fe(III)3 Complex. Inorg. Chem. 2004, 43, 4103–4105. [Google Scholar] [CrossRef] [PubMed]
- Renz, F.; Jung, S.; Klein, M.; Menzel, M.; Thünemann, A.F. Molecular switching complexes with iron and tin as central atom. Polyhedron 2009, 28, 1818–1821. [Google Scholar] [CrossRef]
- Gembický, M.; Boča, R.; Renz, F. A heptanuclear Fe(II)–Fe(III)6 system with twelve unpaired electrons. Inorg. Chem. Commun. 2000, 3, 662–665. [Google Scholar] [CrossRef]
- Boča, R.; Šalitroš, I.; Kožíšek, J.; Linares, J.; Moncoľ, J.; Renz, F. Spin crossover in a heptanuclear mixed-valence iron complex. Dalton Trans. 2010, 39, 2198–2200. [Google Scholar] [CrossRef]
- Renz, F.; Kerep, P. Unprecedented multiple electronic spin transition in hepta- and nonanuclear complex compounds observed by Mössbauer spectroscopy. Polyhedron 2005, 24, 2849–2851. [Google Scholar] [CrossRef]
- Renz, F.; Hill, D.; Klein, M.; Hefner, J. Unprecedented multistability in dodecanuclear complex compound observed by Mössbauer spectroscopy. Polyhedron 2007, 26, 2325–2329. [Google Scholar] [CrossRef]
- Ohyoshi, A.; Honbo, J.; Matsumoto, N.; Ohta, S.; Sakamoto, S. Spin-Equilibrium Behavior in Solution of an Iron(III) Complex [Bis[3-(3-methoxysalicylideneamine)propyl]amino-O,N,N′,N″,O′](pyridine)iron(III) Tetraphenylborate. Bull. Chem. Soc. Jpn. 1986, 59, 1611–1613. [Google Scholar] [CrossRef]
- Maeda, Y.; Noda, Y.; Oshio, H.; Takashima, Y. 57Fe Mössbauer Spectra, Crystal Structure, and Spin-Crossover Behavior of [Fe(mbpN)(lut)]BPh4. Bull. Chem. Soc. Jpn. 1992, 65, 1825–1831. [Google Scholar] [CrossRef]
- Hirose, S.; Hayami, S.; Maeda, Y. Magnetic Properties of Iron(III) Complexes with Photoisomerizable Ligands. Bull. Chem. Soc. Jpn. 2000, 73, 2059–2066. [Google Scholar] [CrossRef]
- Tanimura, K.; Kitashima, R.; Bréfuel, N.; Nakamura, M.; Matsumoto, N.; Shova, S.; Tuchagues, J.P. Infinite chain structure and steep spin crossover of a FeIII complex with a N3O2 pentadentate schiff-base ligand and 4-aminopyridine. Bull. Chem. Soc. Jpn. 2005, 78, 1279–1282. [Google Scholar] [CrossRef]
- Bannwarth, A.; Schmidt, S.O.; Peters, G.; Sönnichsen, F.D.; Thimm, W.; Herges, R.; Tuczek, F. FeIII spin-crossover complexes with photoisomerizable ligands: Experimental and theoretical studies on the ligand-driven light-induced spin change effect. Eur. J. Inorg. Chem. 2012, 2776–2783. [Google Scholar] [CrossRef]
- Herchel, R.; Trávníček, Z. 5-Aminotetrazole induces spin crossover in iron(III) pentadentate Schiff base complexes: Experimental and theoretical investigations. Dalton Trans. 2013, 42, 16279–16288. [Google Scholar] [CrossRef] [PubMed]
- Krüger, C.; Augustín, P.; Nemec, I.; Trávníček, Z.; Oshio, H.; Boča, R.; Renz, F. Spin crossover in iron(III) complexes with pentadentate schiff base ligands and pseudohalido coligands. Eur. J. Inorg. Chem. 2013, 902–915. [Google Scholar] [CrossRef]
- Krüger, C.; Augustín, P.; Dlháň, L.; Pavlik, J.; Moncol’, J.; Nemec, I.; Boča, R.; Renz, F. Iron(III) complexes with pentadentate Schiff-base ligands: Influence of crystal packing change and pseudohalido coligand variations on spin crossover. Polyhedron 2015, 87, 194–201. [Google Scholar] [CrossRef]
- Halcrow, M.A. The spin-states and spin-transitions of mononuclear iron(II) complexes of nitrogen-donor ligands. Polyhedron 2007, 26, 3523–3576. [Google Scholar] [CrossRef]
- Nishi, K.; Fujinami, T.; Kitabayashi, A.; Matsumoto, N. Tetrameric spin crossover iron(II) complex constructed by imidazole⋯chloride hydrogen bonds. Inorg. Chem. Commun. 2011, 14, 1073–1076. [Google Scholar] [CrossRef]
- Murakami, K.; Kitabayashi, A.; Yamauchi, S.; Nishi, K.; Fujinami, T.; Matsumoto, N.; Iijima, S.; Kojima, M. Iron(II) complexes with a linear pentadentate ligand H2L1 = bis(N,N′-2-methylimidazol-4-yl-methylideneaminopropyl) methylamine and a monodentate ligand X (X = N3−, NCS−, NCSe−). Inorg. Chim. Acta 2013, 400, 244–249. [Google Scholar] [CrossRef]
- Hagiwara, H.; Tanaka, T.; Hora, S. Synthesis, structure, and spin crossover above room temperature of a mononuclear and related dinuclear double helicate iron(II) complexes. Dalton Trans. 2016, 45, 17132–17140. [Google Scholar] [CrossRef]
- Hagiwara, H.; Okada, S. A polymorphism-dependent T1/2 shift of 100 K in a hysteretic spin-crossover complex related to differences in intermolecular weak CH⋯X hydrogen bonds (X = S vs. S and N). Chem. Commun. 2016, 52, 815–818. [Google Scholar] [CrossRef]
- Hagiwara, H.; Minoura, R.; Okada, S.; Sunatsuki, Y. Synthesis, Structure, and Magnetic Property of a New Mononuclear Iron(II) Spin Crossover Complex with a Tripodal Ligand Containing Three 1,2,3-Triazole Groups. Chem. Lett. 2014, 43, 950–952. [Google Scholar] [CrossRef]
- Siddiki, A.A.; Takale, B.S.; Telvekar, V.N. One pot synthesis of aromatic azide using sodium nitrite and hydrazine hydrate. Tetrahedron Lett. 2013, 54, 1294–1297. [Google Scholar] [CrossRef]
- Pathigoolla, A.; Pola, R.P.; Sureshan, K.M. A versatile solvent-free azide-alkyne click reaction catalyzed by in situ generated copper nanoparticles. Appl. Catal. A Gen. 2013, 453, 151–158. [Google Scholar] [CrossRef]
- L’abbé, G.; Bruynseels, M.; Delbeke, P.; Toppet, S. Molecular rearrangements of 4-iminomethyl-1,2,3-triazoles. Replacement of 1-aryl substituents in 1 H -1,2,3-triazole-4-carbaldehydes. J. Heterocycl. Chem. 1990, 27, 2021–2027. [Google Scholar] [CrossRef]
- Kahn, O. Molecular Magnetism; VCH: Weinheim, Germany, 1993. [Google Scholar]
- Bain, G.A.; Berry, J.F. Diamagnetic Corrections and Pascal’s Constants. J. Chem. Educ. 2008, 85, 532. [Google Scholar] [CrossRef]
- SCALE3 ABSPACK, version 1.0.4; gui: 1.03; An oxford diffraction program; Oxford Diffraction Ltd.: Abingdon, UK, 2005.
- Rigaku Oxford Diffraction, CrysAlisPro Software system, version 1.171.39.46; Rigaku Corporation: Oxford, UK, 2018.
- Sheldrick, G.M. SHELXT—Integrated space-group and crystal-structure determination. Acta Crystallogr. Sect. A Found. Adv. 2015, 71, 3–8. [Google Scholar] [CrossRef] [PubMed]
- Sheldrick, G.M. Crystal structure refinement with SHELXL. Acta Crystallogr. Sect. C Struct. Chem. 2015, 71, 3–8. [Google Scholar] [CrossRef]
- Dolomanov, O.V.; Bourhis, L.J.; Gildea, R.J.; Howard, J.A.K.; Puschmann, H. OLEX2: A complete structure solution, refinement and analysis program. J. Appl. Crystallogr. 2009, 42, 339–341. [Google Scholar] [CrossRef]
- Llunell, M.; Casanova, D.; Cirera, J.; Alemany, P.; Alvarez, S. SHAPE2.1. Program for Calculating Continuous Shape Measures of Polyhedral Structures; Universitat de Barcelona: Barcelona, Spain, 2013. [Google Scholar]
- Zhao, Y.; Truhlar, D.G. The M06 suite of density functionals for main group thermochemistry, thermochemical kinetics, noncovalent interactions, excited states, and transition elements: Two new functionals and systematic testing of four M06-class functionals and 12 other functionals. Theor. Chem. Acc. 2008, 120, 215–241. [Google Scholar] [CrossRef]
- Frisch, M.J.; Trucks, G.W.; Schlegel, H.B.; Scuseria, G.E.; Robb, M.A.; Cheeseman, J.R.; Scalmani, G.; Barone, V.; Mennucci, B.; Petersson, G.A.; et al. Gaussian 09, Revision B.01, Gaussian, Inc.: Wallingford, CT, USA, 2010.
- Nakamoto, K. Infrared and Raman Spectra of Inorganic and Coordination Compounds, 6th ed.; John Wiley & Sons: Hoboken, NJ, USA, 2009. [Google Scholar]
- Boča, R.; Baran, P.; Boča, M.; Dlháň, L.; Fuess, H.; Haase, W.; Linert, W.; Papánková, B.; Werner, R. Spin crossover in bis(2,6-bis(benzimidazol-2-yl)pyridine) iron(II) tetraphenylborate. Inorg. Chim. Acta 1998, 278, 190–196. [Google Scholar] [CrossRef]
- Hagiwara, H.; Hashimoto, S.; Matsumoto, N.; Iijima, S. Two-Dimensional Iron(II) Spin Crossover Complex Constructed of Bifurcated NH···O− Hydrogen Bonds and π−π Interactions: [FeII(HLH,Me)2](ClO4)2·1.5MeCN (HLH,Me = Imidazol-4-yl-methylidene-8-amino-2-methylquinoline). Inorg. Chem. 2007, 46, 3136–3143. [Google Scholar] [CrossRef] [PubMed]
- Costa, J.S.; Rodríguez-Jiménez, S.; Craig, G.A.; Barth, B.; Beavers, C.M.; Teat, S.J.; Aromí, G. Three-way crystal-to-crystal reversible transformation and controlled spin switching by a nonporous molecular material. J. Am. Chem. Soc. 2014, 136, 3869–3874. [Google Scholar] [CrossRef] [PubMed]
- Yang, F.-L.; Chen, X.; Wu, W.-H.; Zhang, J.-H.; Zhao, X.-M.; Shi, Y.-H.; Shen, F. Spin switching in tris(8-aminoquinoline)iron(II)(BPh4)2: Quantitative guest-losing dependent spin crossover properties and single-crystal-to-single-crystal transformation. Dalton Trans. 2018. [Google Scholar] [CrossRef] [PubMed]
- Bushuev, M.B.; Vinogradova, K.A.; Gatilov, Y.V.; Korolkov, I.V.; Nikolaenkova, E.B.; Krivopalov, V.P. Spin crossover in iron(II) hexafluorophosphate complexes with 2-(pyridin-2-yl)-4-(3,5-di-R-1H-pyrazol-1-yl)-6-methylpyrimidines. Inorg. Chim. Acta 2017, 467, 238–243. [Google Scholar] [CrossRef]
- Craze, A.R.; Bhadbhade, M.M.; Kepert, C.J.; Lindoy, L.F.; Marjo, C.E.; Li, F. Solvent Effects on the Spin-Transition in a Series of Fe(II) Dinuclear Triple Helicate Compounds. Crystals 2018, 8, 376. [Google Scholar] [CrossRef]
- Sertphon, D.; Harding, P.; Murray, K.S.; Moubaraki, B.; Neville, S.M.; Liu, L.; Telfer, S.G.; Harding, D.J. Solvent Effects on the Spin Crossover Properties of Iron(II) Imidazolylimine Complexes. Crystals 2019, 9, 116. [Google Scholar] [CrossRef]
- Jornet-Mollá, V.; Giménez-Saiz, C.; Romero, F.M. Synthesis, Structure, and Photomagnetic Properties of a Hydrogen-Bonded Lattice of [Fe(bpp)2]2+ Spin-Crossover Complexes and Nicotinate Anions. Crystals 2018, 8, 439. [Google Scholar] [CrossRef]
- Guionneau, P.; Marchivie, M.; Bravic, G.; Létard, J.-F.; Chasseau, D. Structural aspects of spin crossover. Examples of the [FeIILn(NCS)2] complexes. Top. Curr. Chem. 2004, 234, 97–128. [Google Scholar]
- Marchivie, M.; Guionneau, P.; Létard, J.-F.; Chasseau, D. Photo-induced spin-transition: The role of the iron(II) environment distortion. Acta Crystallogr. Sect. B 2005, 61, 25–28. [Google Scholar] [CrossRef]
- Spek, A.L. Structure validation in chemical crystallography. Acta Crystallogr. Sect. D Biol. Crystallogr. 2009, 65, 148–155. [Google Scholar] [CrossRef]
- Russell, V.; Scudder, M.; Dance, I. The crystal supramolecularity of metal phenanthroline complexes. J. Chem. Soc. Dalton Trans. 2001, 0, 789–799. [Google Scholar] [CrossRef]
| Temperature/K | 296 | 350 | 375 | 387 |
|---|---|---|---|---|
| Formula | C77H75B2FeN11 | |||
| Formula weight | 1231.95 | |||
| Crystal system | monoclinic | |||
| Space group | P21/n (No.14) | |||
| a/Å | 11.3072(2) | 11.3443(2) | 11.3962(3) | 11.4005(2) |
| b/Å | 41.0958(8) | 41.2866(9) | 41.3406(12) | 41.3938(10) |
| c/Å | 14.4788(4) | 14.4825(4) | 14.4308(5) | 14.4297(4) |
| β/deg | 92.977(2) | 92.796(2) | 92.513(3) | 92.473(2) |
| V/Å3 | 6718.9(3) | 6775.1(3) | 6792.2(4) | 6803.2(3) |
| Z | 4 | 4 | 4 | 4 |
| dcalcd./g cm−3 | 1.218 | 1.208 | 1.205 | 1.203 |
| μ (Mo Kα)/mm−1 | 0.277 | 0.275 | 0.274 | 0.273 |
| R1a (I>2sigma(I)) | 0.0458 | 0.0500 | 0.0779 | 0.0601 |
| wR2b (I>2sigma(I)) | 0.1062 | 0.1176 | 0.1944 | 0.1511 |
| R1a (all data) | 0.0678 | 0.0826 | 0.1220 | 0.1030 |
| wR2b (all data) | 0.1161 | 0.1328 | 0.2177 | 0.1738 |
| S | 1.029 | 1.022 | 1.065 | 1.022 |
| CCDC number | 1911292 | 1911293 | 1911294 | 1911295 |
| Temperature/K | 296 | 350 | 375 | 387 |
|---|---|---|---|---|
| Fe1–N3 | 1.9770(13) | 1.9788(15) | 1.989(3) | 1.986(2) |
| Fe1–N4 | 1.9955(11) | 1.9976(13) | 2.004(2) | 1.9985(18) |
| Fe1–N5 | 2.0901(13) | 2.0918(15) | 2.088(3) | 2.093(2) |
| Fe1–N6 | 1.9477(12) | 1.9494(14) | 1.947(2) | 1.9533(19) |
| Fe1–N7 | 1.9577(12) | 1.9619(14) | 1.966(2) | 1.9683(17) |
| Fe1–N10 | 1.9580(13) | 1.9599(15) | 1.961(3) | 1.967(2) |
| Average Fe–N | 1.988 | 1.990 | 1.993 | 1.994 |
| N3–Fe1–N4 | 80.25(5) | 80.04(6) | 79.94(10) | 79.85(8) |
| N3–Fe1–N5 | 177.92(5) | 177.74(6) | 177.53(10) | 177.45(8) |
| N3–Fe1–N6 | 95.80(5) | 95.89(6) | 96.07(11) | 96.13(8) |
| N3–Fe1–N7 | 83.58(5) | 83.46(6) | 83.33(10) | 83.42(7) |
| N3–Fe1–N10 | 88.57(5) | 88.49(6) | 88.51(10) | 88.08(8) |
| N4–Fe1–N5 | 97.68(5) | 97.70(6) | 97.59(11) | 97.61(8) |
| N4–Fe1–N6 | 173.48(5) | 173.33(6) | 173.26(10) | 173.32(8) |
| N4–Fe1–N7 | 94.11(5) | 94.16(5) | 94.04(9) | 94.05(7) |
| N4–Fe1–N10 | 90.57(5) | 90.45(6) | 90.48(10) | 90.40(7) |
| N5–Fe1–N6 | 86.23(5) | 86.34(6) | 86.38(11) | 86.39(8) |
| N5–Fe1–N7 | 96.37(5) | 96.56(6) | 96.82(10) | 96.67(8) |
| N5–Fe1–N10 | 91.69(5) | 91.72(6) | 91.60(10) | 92.09(8) |
| N6–Fe1–N7 | 80.23(5) | 80.06(6) | 80.04(10) | 80.13(8) |
| N6–Fe1–N10 | 94.53(5) | 94.74(6) | 94.87(10) | 94.82(8) |
| N7–Fe1–N10 | 170.05(5) | 169.89(6) | 169.83(11) | 169.56(8) |
| Σ | 61.89 | 62.83 | 63.27 | 63.90 |
| Θ | 93.47 | 95.21 | 96.77 | 95.69 |
| S(Oh) | 0.721 | 0.742 | 0.747 | 0.753 |
| Octahedral volume (Å3) | 10.291 | 10.320 | 10.358 | 10.389 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Matsuyama, T.; Nakata, K.; Hagiwara, H.; Udagawa, T. Iron(II) Spin Crossover Complex with the 1,2,3-Triazole-Containing Linear Pentadentate Schiff-Base Ligand and the MeCN Monodentate Ligand. Crystals 2019, 9, 276. https://doi.org/10.3390/cryst9060276
Matsuyama T, Nakata K, Hagiwara H, Udagawa T. Iron(II) Spin Crossover Complex with the 1,2,3-Triazole-Containing Linear Pentadentate Schiff-Base Ligand and the MeCN Monodentate Ligand. Crystals. 2019; 9(6):276. https://doi.org/10.3390/cryst9060276
Chicago/Turabian StyleMatsuyama, Tomoe, Keishi Nakata, Hiroaki Hagiwara, and Taro Udagawa. 2019. "Iron(II) Spin Crossover Complex with the 1,2,3-Triazole-Containing Linear Pentadentate Schiff-Base Ligand and the MeCN Monodentate Ligand" Crystals 9, no. 6: 276. https://doi.org/10.3390/cryst9060276
APA StyleMatsuyama, T., Nakata, K., Hagiwara, H., & Udagawa, T. (2019). Iron(II) Spin Crossover Complex with the 1,2,3-Triazole-Containing Linear Pentadentate Schiff-Base Ligand and the MeCN Monodentate Ligand. Crystals, 9(6), 276. https://doi.org/10.3390/cryst9060276






