Passivation Mechanism of Nitrogen in ZnO under Different Oxygen Ambience
Abstract
:1. Introduction
2. Experimental
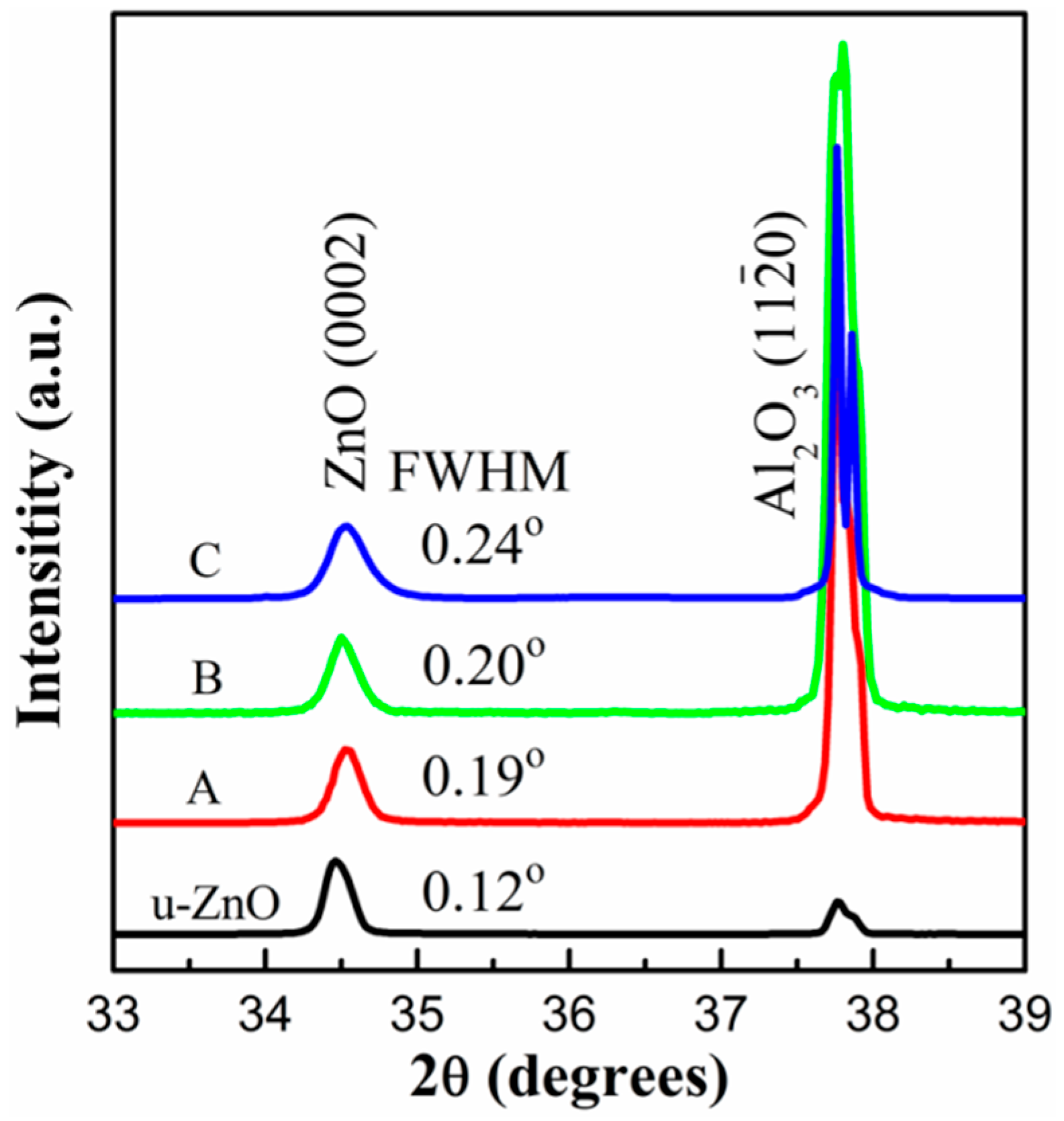
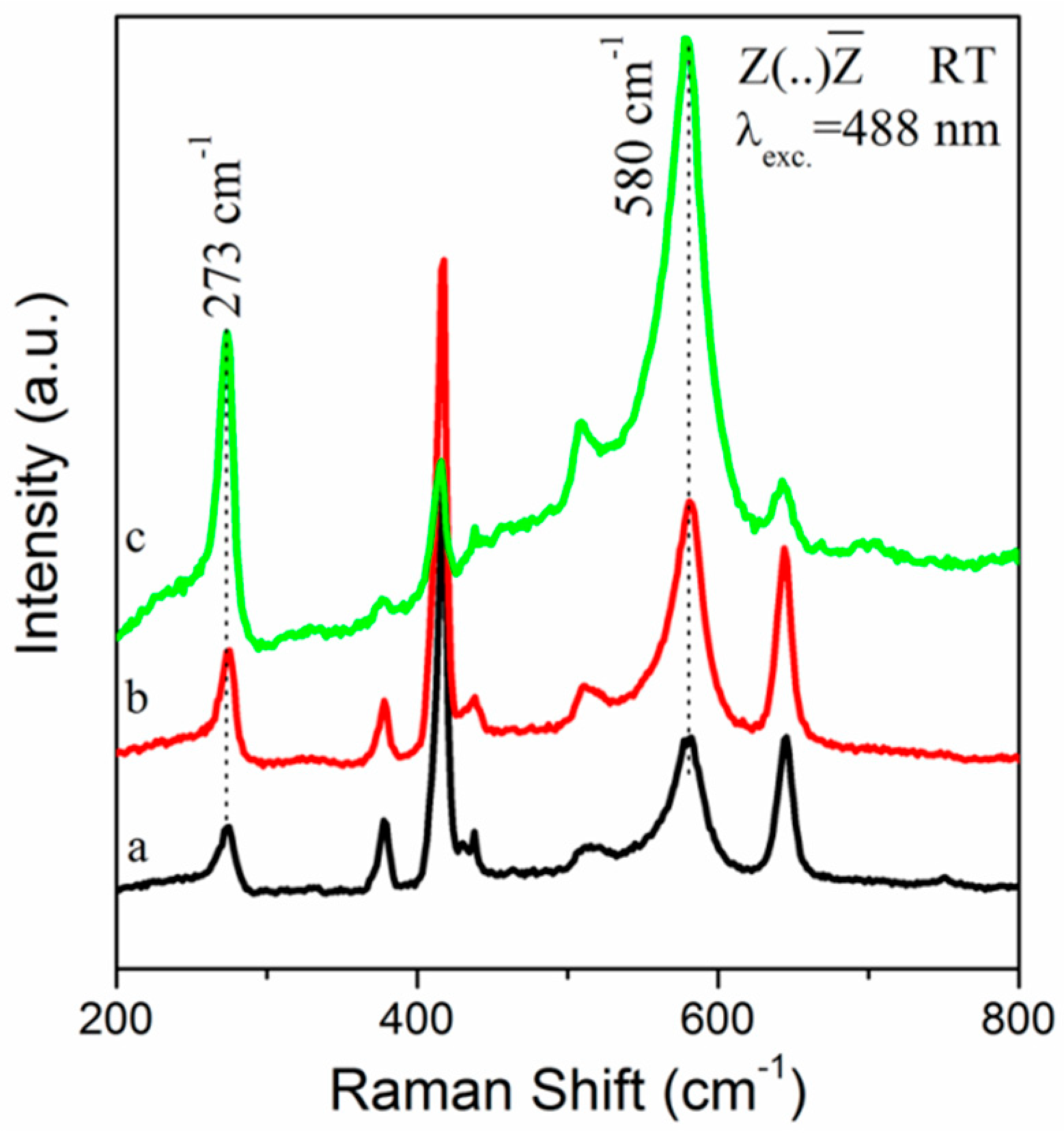
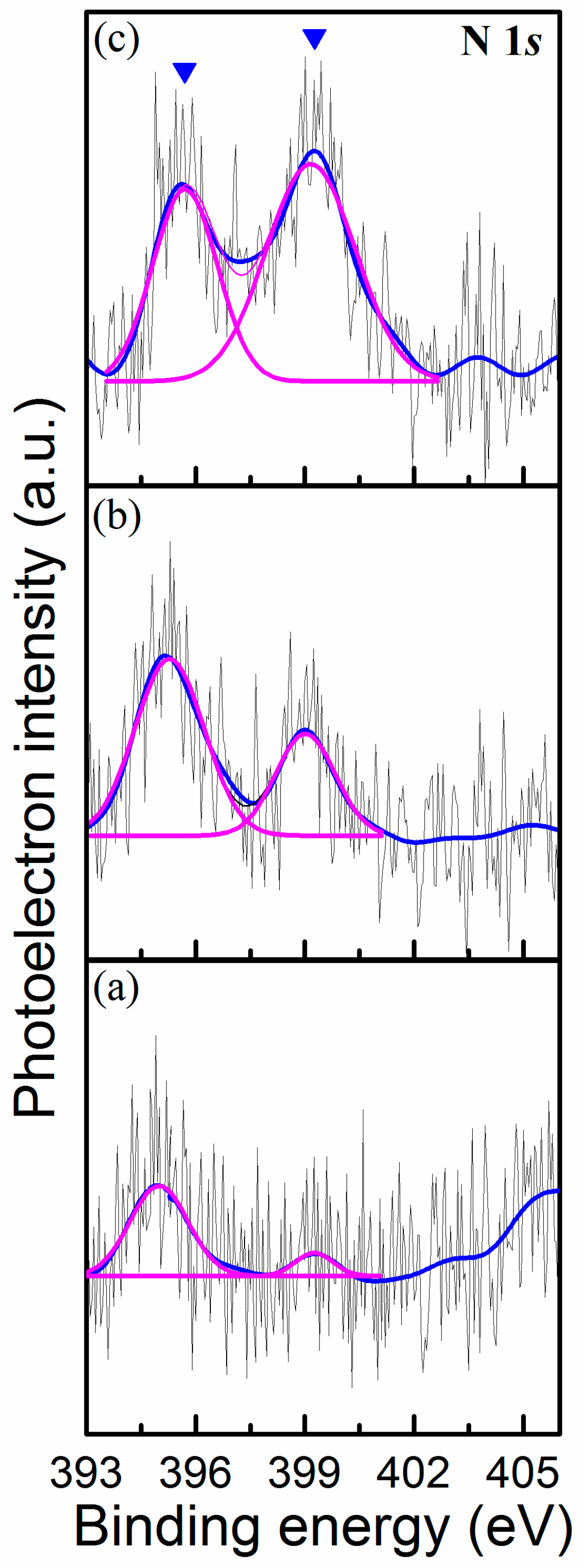
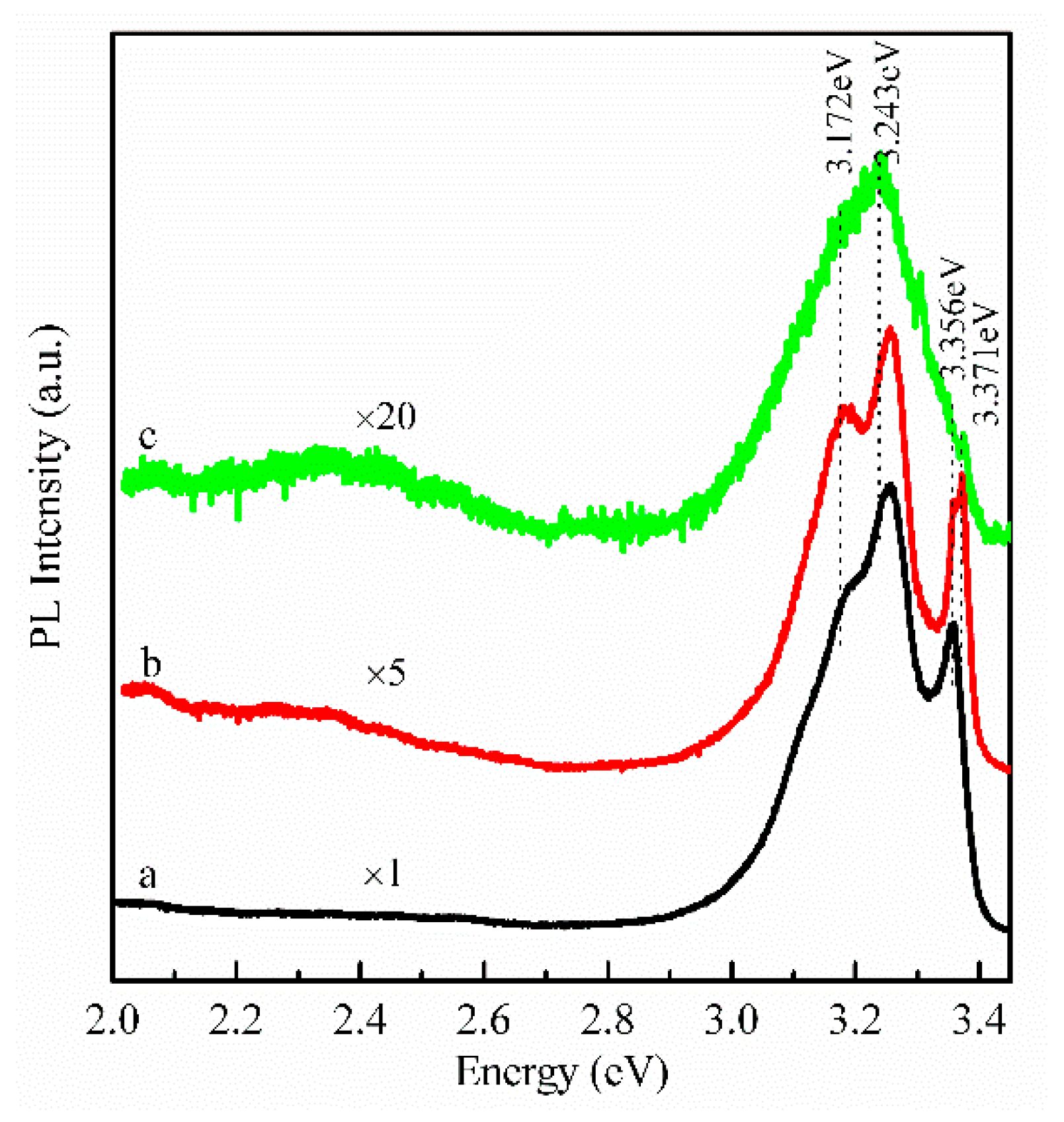
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Bagnall, D.M.; Chen, Y.F.; Zhu, Z.; Yao, T.; Koyama, S.; Shen, M.Y.; Goto, T. Optically pumped lasing of ZnO at room temperature. Appl. Phys. Lett. 1997, 70, 2230–2232. [Google Scholar] [CrossRef]
- Service, R.F. Materials science—Will UV lasers beat the blues? Science 1997, 276, 895. [Google Scholar] [CrossRef]
- Norton, D.P.; Heo, Y.W.; Ivill, M.P.; Ip, K.; Pearton, S.J.; Chisholm, M.F.; Steiner, T. ZnO: Growth, doping & processing. Mater. Today 2004, 7, 34–40. [Google Scholar]
- Bian, J.M.; Li, X.M.; Gao, X.D.; Yu, W.D.; Chen, L.D. Deposition and electrical properties of N-In codoped p-type ZnO films by ultrasonic spray pyrolysis. Appl. Phys. Lett. 2004, 84, 541–543. [Google Scholar] [CrossRef]
- Tsukazaki, A.; Ohtomo, A.; Onuma, T.; Ohtani, M.; Makino, T.; Sumiya, M.; Ohtani, K.; Chichibu, S.F.; Fuke, S.; Segawa, Y.; et al. Repeated temperature modulation epitaxy for p-type doping and light-emitting diode based on ZnO. Nat. Mater. 2005, 4, 42–46. [Google Scholar] [CrossRef]
- Jiao, S.J.; Zhang, Z.Z.; Lu, Y.M.; Shen, D.Z.; Yao, B.; Zhang, J.Y.; Li, B.H.; Zhao, D.X.; Fan, X.W.; Tang, Z.K. ZnO p-n junction light-emitting diodes fabricated on sapphire substrates. Appl. Phys. Lett. 2006, 88, 031911. [Google Scholar] [CrossRef]
- Kato, H.; Yamamuro, T.; Ogawa, A.; Kyotani, C.; Sano, M. Impact of Mixture Gas Plasma of N-2 and O-2 as the N Source on ZnO-Based Ultraviolet Light-Emitting Diodes Fabricated by Molecular Beam Epitaxy. Appl. Phys. Express 2011, 4, 091105. [Google Scholar] [CrossRef]
- Lyons, J.L.; Janotti, A.; Van de Walle, C.G. Why nitrogen cannot lead to p-type conductivity in ZnO. Appl. Phys. Lett. 2009, 95, 252105. [Google Scholar] [CrossRef]
- Liu, L.; Xu, J.L.; Wang, D.D.; Jiang, M.M.; Wang, S.P.; Li, B.H.; Zhang, Z.Z.; Zhao, D.X.; Shan, C.X.; Yao, B.; et al. p-Type Conductivity in N-Doped ZnO: The Role of the N-Zn-V-O Complex. Phys. Rev. Lett. 2012, 108, 215501. [Google Scholar] [CrossRef]
- Nakahara, K.; Takasu, H.; Fons, P.; Yamada, A.; Iwata, K.; Matsubara, K.; Hunger, R.; Niki, S. Growth of N-doped and Ga plus N-codoped ZnO films by radical source molecular beam epitaxy. J. Cryst. Growth 2002, 237–239, 503–508. [Google Scholar] [CrossRef]
- Yan, Y.; Zhang, S.B.; Pantelides, S.T. Control of doping by impurity chemical potentials: Predictions for p-type ZnO. Phys. Rev. Lett. 2001, 86, 5723–5726. [Google Scholar] [CrossRef]
- Park, C.H.; Zhang, S.B.; Wei, S.H. Origin of p-type doping difficulty in ZnO: The impurity perspective. Phys. Rev. B 2002, 66, 073202. [Google Scholar] [CrossRef]
- Lee, E.C.; Kim, Y.S.; Jin, Y.G.; Chang, K.J. Compensation mechanism for N acceptors in ZnO. Phys. Rev. B 2001, 64, 085120. [Google Scholar] [CrossRef]
- Park, S.H.; Minegishi, T.; Lee, H.J.; Park, J.S.; Im, I.H.; Yao, T.; Oh, D.C.; Taishi, T.; Yonenaga, I.; Chang, J.H. Investigation of the crystallinity of N and Te codoped Zn-polar ZnO films grown by plasma-assisted molecular-beam epitaxy. J. Appl. Phys. 2010, 108, 093518. [Google Scholar] [CrossRef]
- Futsuhara, M.; Yoshioka, K.; Takai, O. Optical properties of zinc oxynitride thin films. Thin Solid Films 1998, 317, 322–325. [Google Scholar] [CrossRef]
- Singh, A.V.; Mehra, R.M.; Wakahara, A.; Yoshida, A. p-type conduction in codoped ZnO thin films. J. Appl. Phys. 2003, 93, 396–399. [Google Scholar] [CrossRef]
- Kim, Y.I.; Cadars, S.; Shayib, R.; Proffen, T.; Feigerle, C.S.; Chmelka, B.F.; Seshadri, R. Local structures of polar wurtzites Zn1−xMgxO studied by Raman and Zn-67/Mg-25 NMR spectroscopies and by total neutron scattering. Phys. Rev. B 2008, 78, 195205. [Google Scholar] [CrossRef]
- Manjón, F.J.; Marí, B.; Serrano, J.; Romero, A.H. Silent Raman modes in zinc oxide and related nitrides. J. Appl. Phys. 2005, 97, 053516. [Google Scholar] [CrossRef]
- Gao, L.L.; Yao, B.; Liu, B.; Liu, L.; Yang, T.; Liu, B.B.; Shen, D.Z. Effects of Mg concentration on solubility and chemical state of N in N-doped MgZnO alloy. J. Chem. Phys. 2010, 129, 024514. [Google Scholar] [CrossRef]
- Zhang, B.Y.; Yao, B.; Li, Y.F.; Zhang, Z.Z.; Li, B.H.; Shan, C.X.; Zhao, D.X.; Shen, D.Z. Investigation on the formation mechanism of p-type Li-N dual-doped ZnO. Appl. Phys. Lett. 2010, 97, 222101. [Google Scholar] [CrossRef]
- Yang, D.X.; Velamakanni, A.; Bozoklu, G.; Park, S.; Stoller, M.; Piner, R.D.; Stankovich, S.; Jung, I.; Field, D.A.; Ventrice, C.A., Jr.; et al. Chemical analysis of graphene oxide films after heat and chemical treatments by X-ray photoelectron and Micro-Raman spectroscopy. Carbon 2009, 47, 145–152. [Google Scholar] [CrossRef]
- Perkins, C.L.; Lee, S.H.; Li, X.N.; Asher, S.E.; Coutts, T.J. Identification of nitrogen chemical states in N-doped ZnO via X-ray photoelectron spectroscopy. J. Appl. Phys. 2005, 97, 034907. [Google Scholar] [CrossRef]
- Look, D.C.; Hemsky, J.W.; Sizelove, J.R. Residual native shallow donor in ZnO. Phys. Rev. Lett. 1999, 82, 2552–2555. [Google Scholar] [CrossRef]
- Limpijumnong, S.; Li, X.N.; Wei, S.H.; Zhang, S.B. Substitutional diatomic molecules NO, NC, CO, N-2, and O-2: Their vibrational frequencies and effects on p doping of ZnO. Appl. Phys. Lett. 2005, 86, 151910. [Google Scholar] [CrossRef]
- Taylor, J.A.; Gerald, M.L.; Rabalais, J.W. Interactions of N2+ and NO+ ions with surfaces of graphite, diamond, teflon, and graphite monofluoride. J. Am. Chem. Soc. 1978, 100, 4441–4447. [Google Scholar] [CrossRef]
- Polo, M.C.; Aguiar, R.; Serra, P.; Clèries, L.; Varela, M.; Esteve, J. Carbon nitride thin films obtained by laser ablation of graphite in a nitrogen plasma. Appl. Surf. Sci. 1996, 96–98, 870–873. [Google Scholar] [CrossRef]
- Ding, M.; Zhao, D.X.; Yao, B.; Li, B.H.; Zhang, Z.Z.; Shen, D.Z. The p-type ZnO film realized by a hydrothermal treatment method. Appl. Phys. Lett. 2011, 98, 062102. [Google Scholar] [CrossRef]
- Sans, J.A.; Segura, A.; Mollar, M.; Marí, B. Optical properties of thin films of ZnO prepared by pulsed laser deposition. Thin Solid Films 2004, 453–454, 251–255. [Google Scholar] [CrossRef]
- Yao, B.; Shen, D.Z.; Zhang, Z.Z.; Wang, X.H.; Wei, Z.P.; Li, B.H.; Lv, Y.M.; Fan, X.W. Effects of nitrogen doping and illumination on lattice constants and conductivity behavior of zinc oxide grown by magnetron sputtering. J. Appl. Phys. 2006, 99, 123510. [Google Scholar] [CrossRef]
- Ahn, C.H.; Kim, Y.Y.; Kim, D.C.; Mohanta, S.K.; Choa, H.K. A comparative analysis of deep level emission in ZnO layers deposited by various methods. J. Appl. Phys. 2009, 105, 013502. [Google Scholar] [CrossRef]
| Sample | FNO (SCCM) | FN2 (SCCM) | [N] (cm−3) | µ (cm2 /V s) | nc (cm−3) | Resistivity (Ω cm) | Carriers Type |
|---|---|---|---|---|---|---|---|
| u-ZnO | … | … | … | 25 | ~1018 | 0.2 | n |
| A | 0.8 | 0.4 | 0.6 × 1020 | 1.84 | 6.35 × 1017 | 6 | n |
| B | 0.6 | 0.6 | 6.7 × 1020 | 0.31 | 3.27 × 1016 | 868 | p |
| C | 0.4 | 0.8 | 8.2 × 1020 | … | … | 11800 | indefinite |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, X.; Zhang, Z.; Zhang, Y.; Yao, B.; Li, B.; Gong, Q. Passivation Mechanism of Nitrogen in ZnO under Different Oxygen Ambience. Crystals 2019, 9, 204. https://doi.org/10.3390/cryst9040204
Chen X, Zhang Z, Zhang Y, Yao B, Li B, Gong Q. Passivation Mechanism of Nitrogen in ZnO under Different Oxygen Ambience. Crystals. 2019; 9(4):204. https://doi.org/10.3390/cryst9040204
Chicago/Turabian StyleChen, Xingyou, Zhenzhong Zhang, Yunyan Zhang, Bin Yao, Binghui Li, and Qian Gong. 2019. "Passivation Mechanism of Nitrogen in ZnO under Different Oxygen Ambience" Crystals 9, no. 4: 204. https://doi.org/10.3390/cryst9040204
APA StyleChen, X., Zhang, Z., Zhang, Y., Yao, B., Li, B., & Gong, Q. (2019). Passivation Mechanism of Nitrogen in ZnO under Different Oxygen Ambience. Crystals, 9(4), 204. https://doi.org/10.3390/cryst9040204





