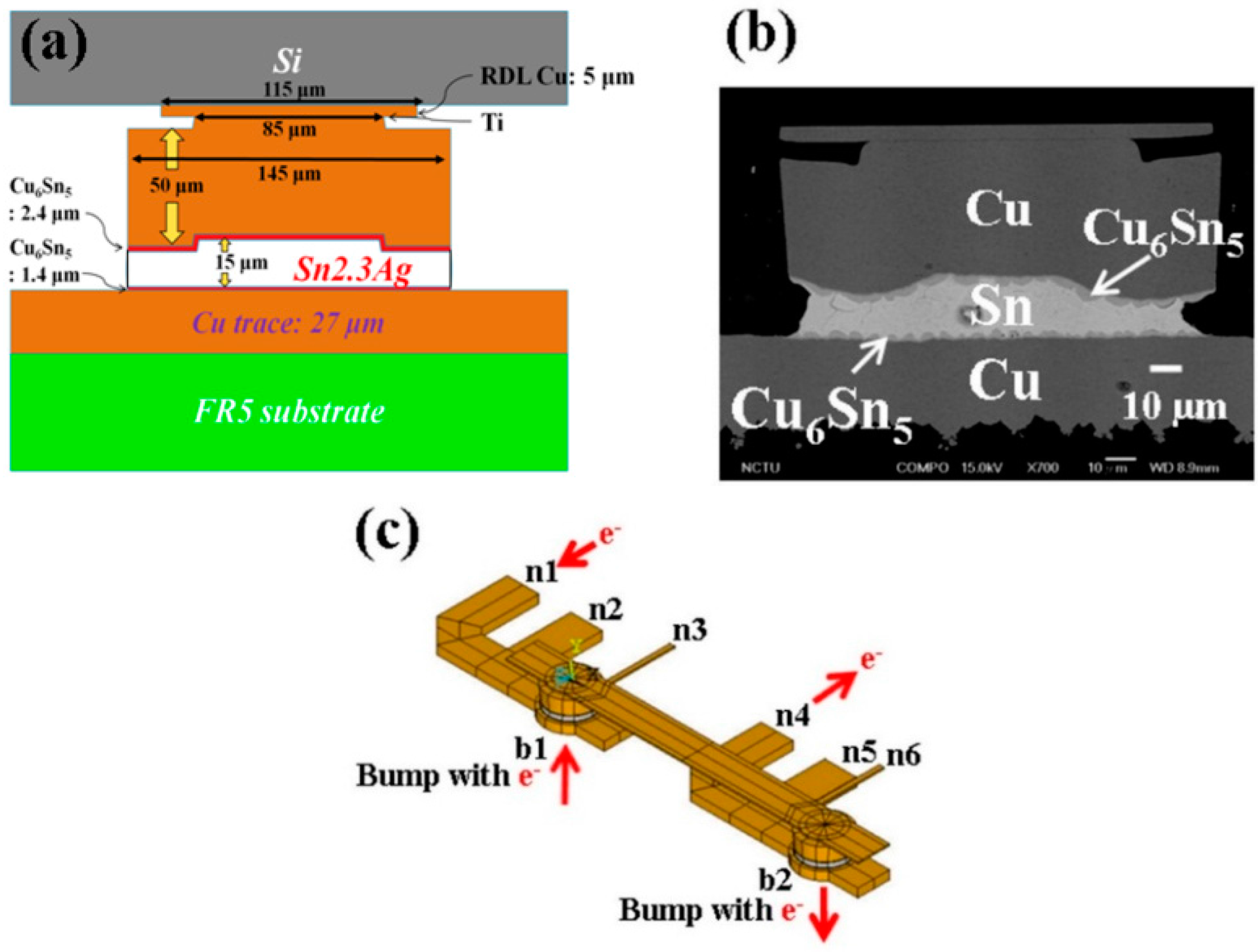
3.2. Formation of Porous Cu3Sn
It is interesting that porous Cu
3Sn IMCs may form at later stages of electromigration tests.
Figure 3 and
Figure 4 illustrate the cross-sectional SEM images at different stages in the EM tests with opposite direction of electron flow.
Figure 3 shows the EM test results with upward electron flow.
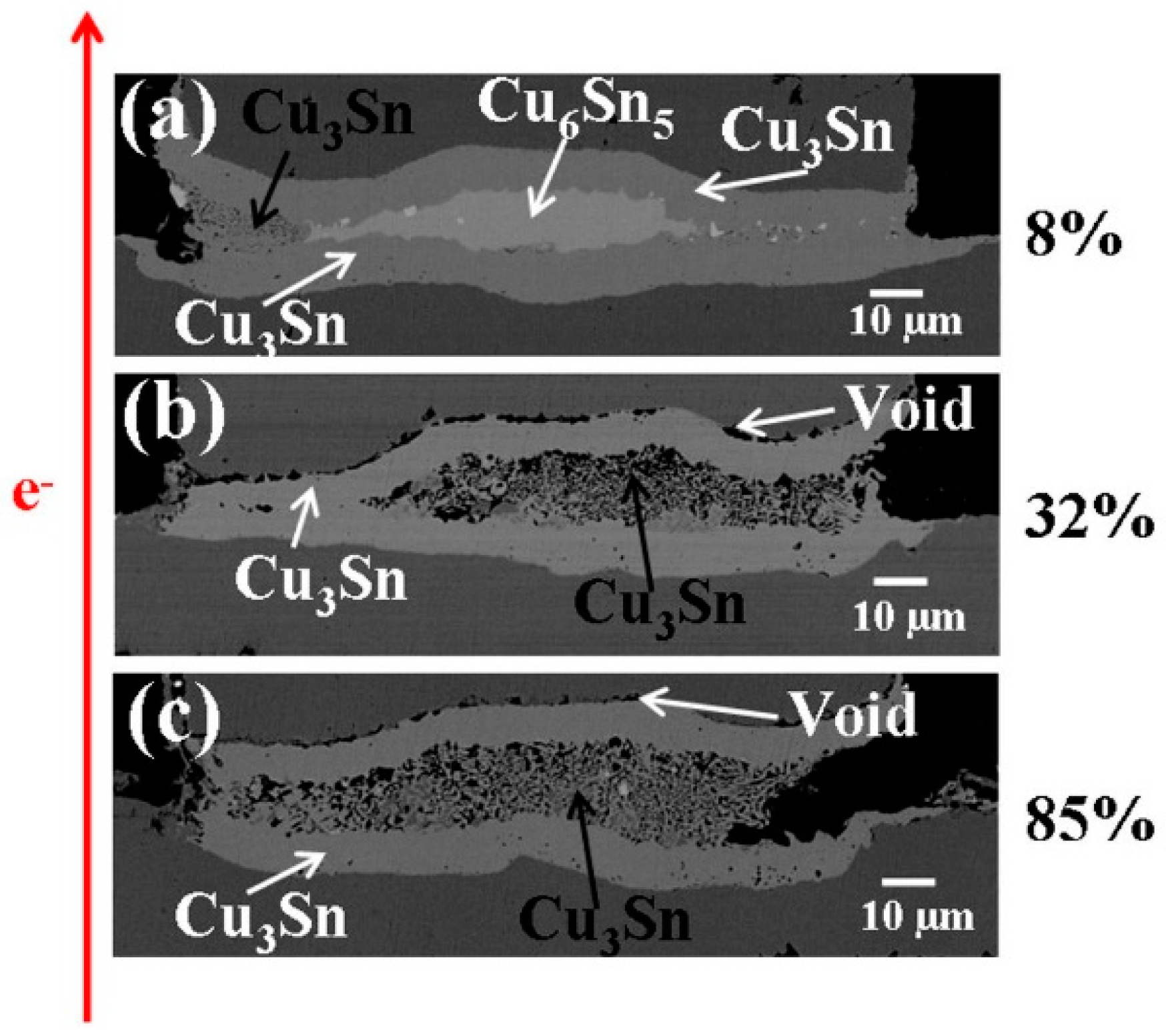
Figure 3a–c are the cross-sectional SEM images with bump resistance increase, 8% (after 517 h), 32% (after 217 h), and 85% (after 429 h) of its initial value, respectively. When the bump resistance increased to 8% of its initial value, the layer-type Cu
6Sn
5 in the middle started to transform into porous-type Cu
3Sn, as shown in
Figure 3a. We note that the porous-type Cu
3Sn first formed on the periphery of the bump. As the change in bump resistance increased to 32%, shown in
Figure 3b, the layer-type Cu
6Sn
5 IMC had fully transformed into porous-type Cu
3Sn IMC.
Figure 3c shows similar results, from a different bump, as
Figure 3b which means the Cu
6Sn
5 IMC had also totally transformed into porous-type Cu
3Sn IMCs in the later stage of the EM tests.
Figure 3.
Cross-sectional SEM images of solder bumps with Cu column UBM stressed at 1.45 × 104 A/cm2 with upward electron flow at 185 °C with bump resistance increases (a) 8%, (b) 32%, and (c) 85% of its initial value.
Figure 3.
Cross-sectional SEM images of solder bumps with Cu column UBM stressed at 1.45 × 104 A/cm2 with upward electron flow at 185 °C with bump resistance increases (a) 8%, (b) 32%, and (c) 85% of its initial value.
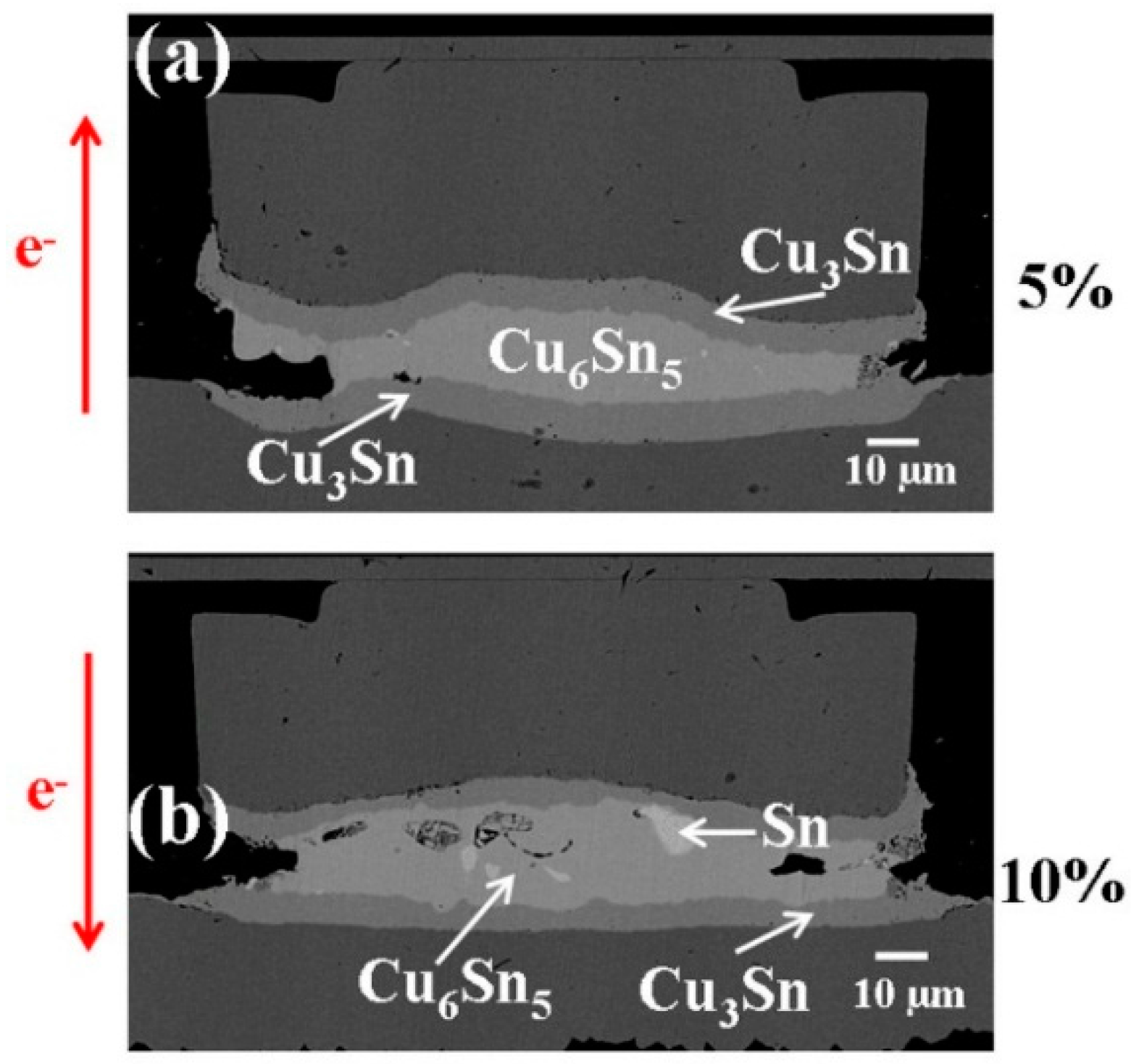
Similarly,
Figure 4 shows the EM tests results with downward electron flow, where
Figure 4a–c illustrate the cross-sectional SEM images with the bump resistance increase 15% (after 517 h), 20% (after 217 h), and 100% (after 429 h) of its initial value, respectively. The results are similar to those in
Figure 3. As the bump resistance increased, the layer-type Cu
6Sn
5 in the middle started to transform into porous-type Cu
3Sn.
In order to rule out the possibility that the porous structure was generated in the polishing process or by other external forces, FIB were utilized to provide a deeper cross-section in a selected solder joint, stressed at 1.45 × 10
4 A/cm
2 for 429 h as illustrated in
Figure 5. The downward direction of the electron flow was labeled in the figure. This second cross-section view indicated that the porous structure not only formed on the polished surface but in the entire bump.
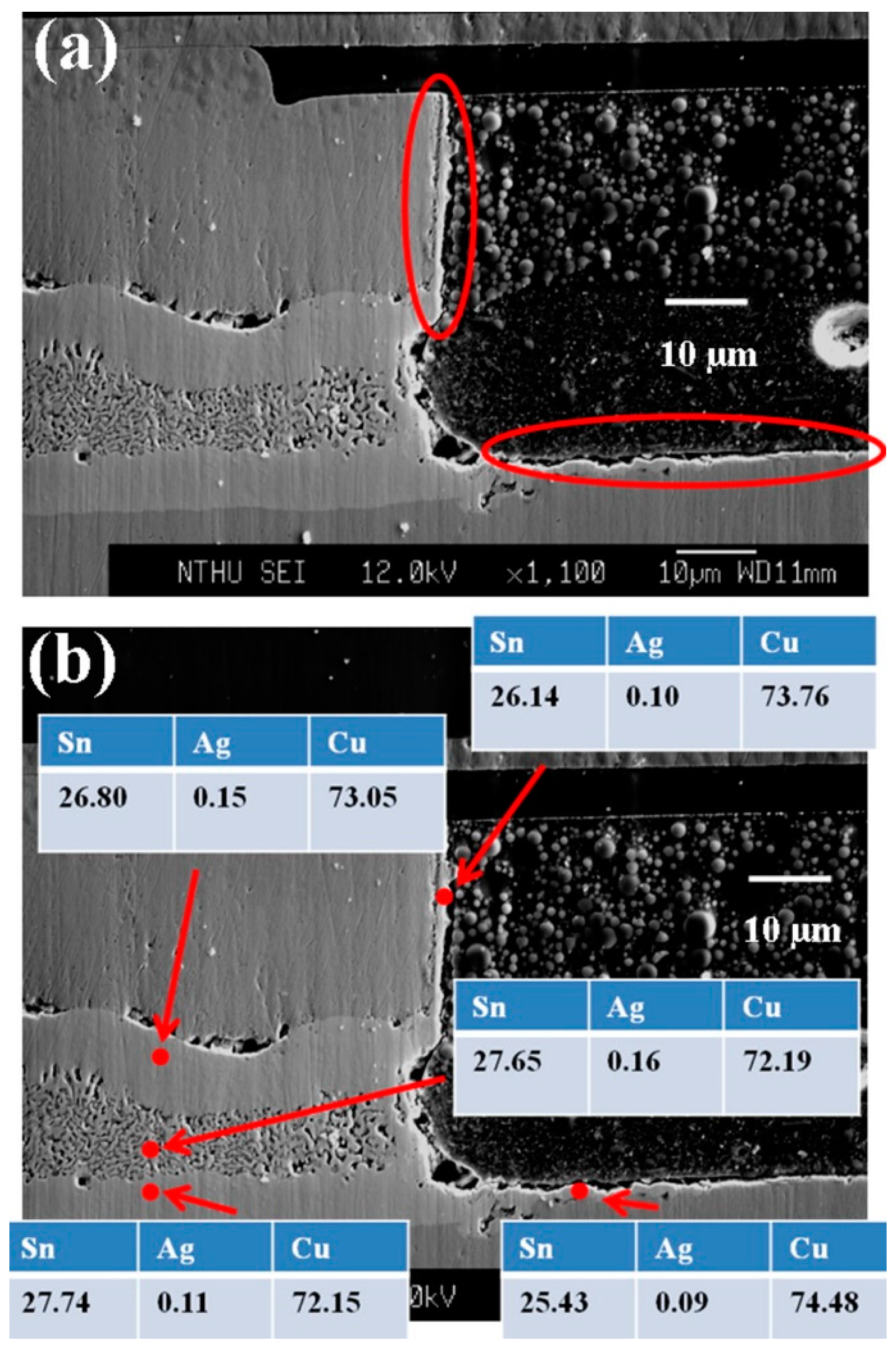
In the bumps with porous Cu
3Sn, we observed that the effect of side wall reaction is very serious. Park
et al. reported that a high stressing current may lead to a serious side wall reaction effect [
16].
Figure 6a shows the side wall of a bump. The bump has had porous-type Cu
3Sn IMCs formation. We can clearly observe that there are IMCs formation on the side wall, indicated by the ellipses. Since the thickness of the IMCs formed on the side wall is only about 3 μm, it is hard to verify the composition with EDS. Hence, EPMA was used instead.
Figure 6b illustrates the results that all the IMCs formed on the side wall are Cu
3Sn. The compositions of the layer-type and porous-type structures were also confirmed with EPMA to be Cu
3Sn, identical with the results measured with EDS. Therefore, the whole joint has transformed completely into Cu
3Sn with two different morphologies.
Figure 7 shows the cross-sectional TEM image and the electron diffraction patterns of point “a” in a porous Cu
3Sn. In the diffraction pattern, the spots correspond to the principle reflections of a basic hexagonal lattice. The zone axes are indexed with [01
2]. The results show that the porous-type structure is hexagonal Cu
3Sn.
Figure 4.
Cross-sectional SEM images of solder bumps with Cu column UBM stressed at 1.45 × 104 A/cm2 with downward electron flow at 185 °C with bump resistance increases (a) 15%, (b) 20%, and (c) 100% of its initial value. Cross-sectional SEM images of solder bumps with Cu column UBM stressed at 1.45 × 104 A/cm2 with downward electron flow at 185 °C with bump resistance increases (a) 15%, (b) 20%, and (c) 100% of its initial value.
Figure 4.
Cross-sectional SEM images of solder bumps with Cu column UBM stressed at 1.45 × 104 A/cm2 with downward electron flow at 185 °C with bump resistance increases (a) 15%, (b) 20%, and (c) 100% of its initial value. Cross-sectional SEM images of solder bumps with Cu column UBM stressed at 1.45 × 104 A/cm2 with downward electron flow at 185 °C with bump resistance increases (a) 15%, (b) 20%, and (c) 100% of its initial value.
Figure 5.
Cross-sectional FIB image of Cu3Sn IMC structures.
Figure 5.
Cross-sectional FIB image of Cu3Sn IMC structures.
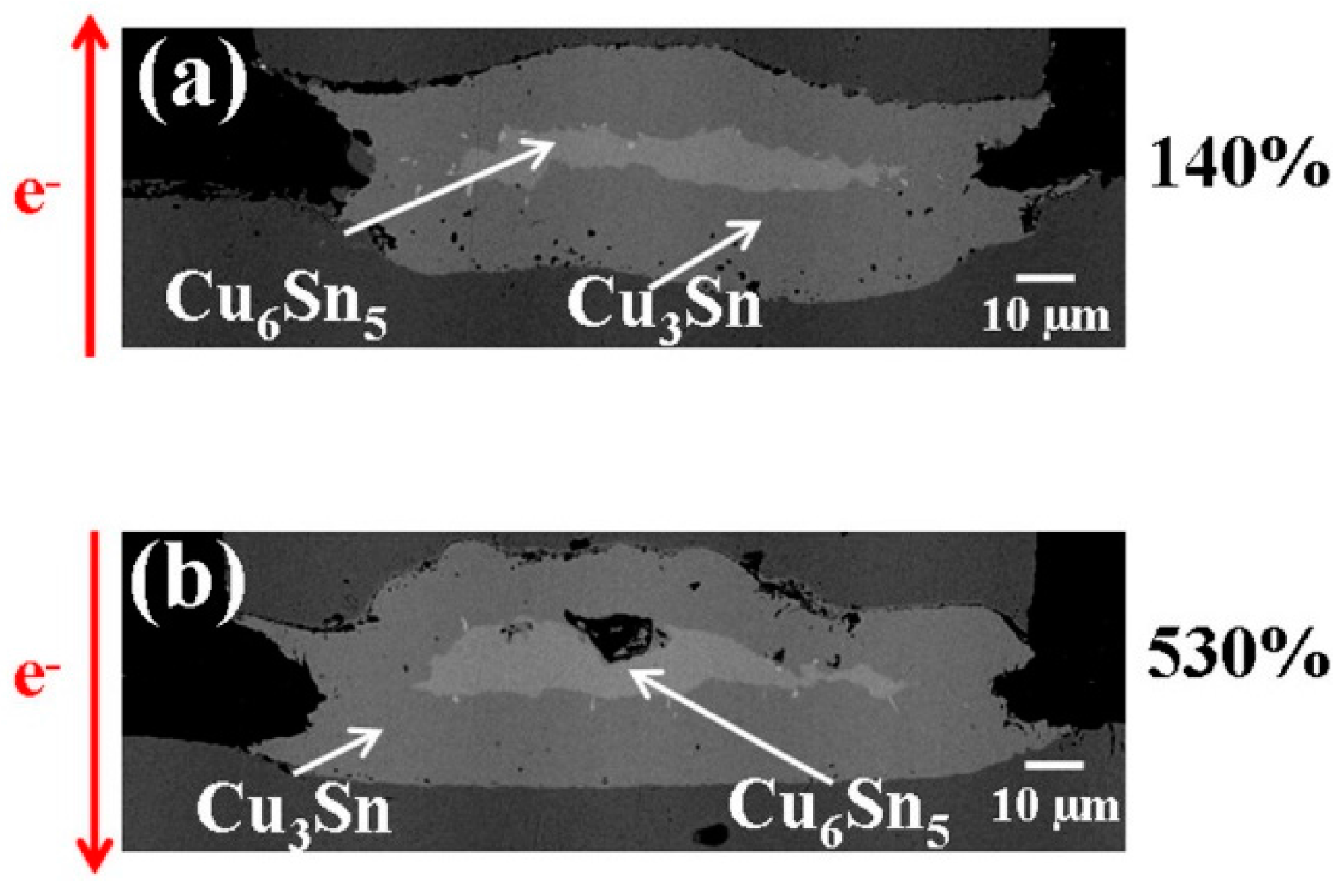
To determine the conditions under which porous Cu
3Sn could form, we stressed the sample at a lower current density and, hence, a lower temperature.
Figure 8 shows cross-sectional SEM images of a pair of solder joints stressed with 1.2 × 10
4 A/cm
2 at 170 °C. The bump resistance increases 140% (after 5094 h) and 530% (after 5094 h) of its initial value as indicated in
Figure 8a,b, respectively. It can be seen that only layer-type Cu
3Sn and layer-type Cu
6Sn
5 have formed, with no evidence of porous Cu
3Sn even in the case of
Figure 8b. The results demonstrate that the solder joints do not transform into porous structures at an insufficient current density and temperature.
Figure 6.
Cross-sectional SEM images of (a) IMCs formed on the side wall and (b) EPMA analysis. The composition data were shown in atomic percent.
Figure 6.
Cross-sectional SEM images of (a) IMCs formed on the side wall and (b) EPMA analysis. The composition data were shown in atomic percent.
Figure 7.
(a) Cross-sectional TEM image of porous-type structure and (b) diffraction patterns of point A in (a).
Figure 7.
(a) Cross-sectional TEM image of porous-type structure and (b) diffraction patterns of point A in (a).
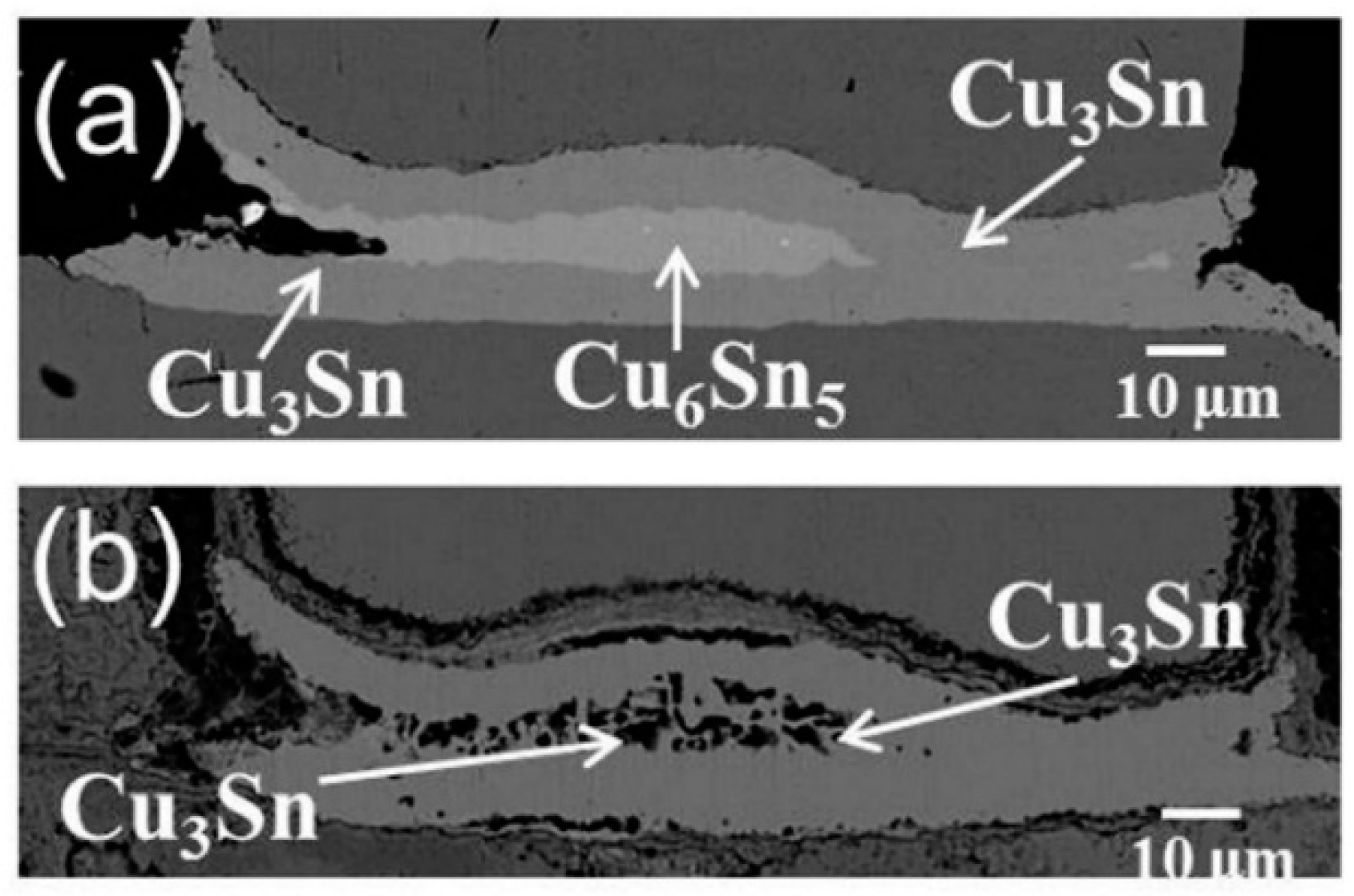
To further examine the temperature effect on the of porous Cu
3Sn formation, another set of bumps were aged at 185 °C in the oven without current stressing. In the case of 1000-h aging (
Figure 9a), the joints have fully transformed into IMC joints. Layer-type Cu
6Sn
5 has formed in the middle of the bump and layer-type Cu
3Sn has formed between Cu and Cu
6Sn
5. In the case of 2000 h aging (
Figure 9b), the layer-type Cu
6Sn
5 in the middle has completely transformed into porous Cu
3Sn. The results show that porous Cu
3Sn could form without current stressing at a sufficiently high temperature, which gives an independent confirmation of temperature’s critical role in the formation of porous Cu
3Sn.
Figure 8.
Cross-sectional SEM images of solder bumps with Cu column UBM stressed at 1.20 × 104 A/cm2 at 170 °C with bump resistance increases (a) 140% with upward electron flow and (b) 530% with downward electron flow.
Figure 8.
Cross-sectional SEM images of solder bumps with Cu column UBM stressed at 1.20 × 104 A/cm2 at 170 °C with bump resistance increases (a) 140% with upward electron flow and (b) 530% with downward electron flow.
Figure 9.
Cross-sectional SEM images of solder bumps with Cu column UBM aging at 185 °C for (a) 1000 h and (b) 2000 h.
Figure 9.
Cross-sectional SEM images of solder bumps with Cu column UBM aging at 185 °C for (a) 1000 h and (b) 2000 h.
So far, we have observed that the porous-type Cu3Sn IMCs can form during high current stressing at high temperatures and the effect of side wall wetting is obvious. To explain its formation mechanism, a further discussion is presented in the next section.
3.3. Formation Mechanism of Porous Cu3Sn IMCs
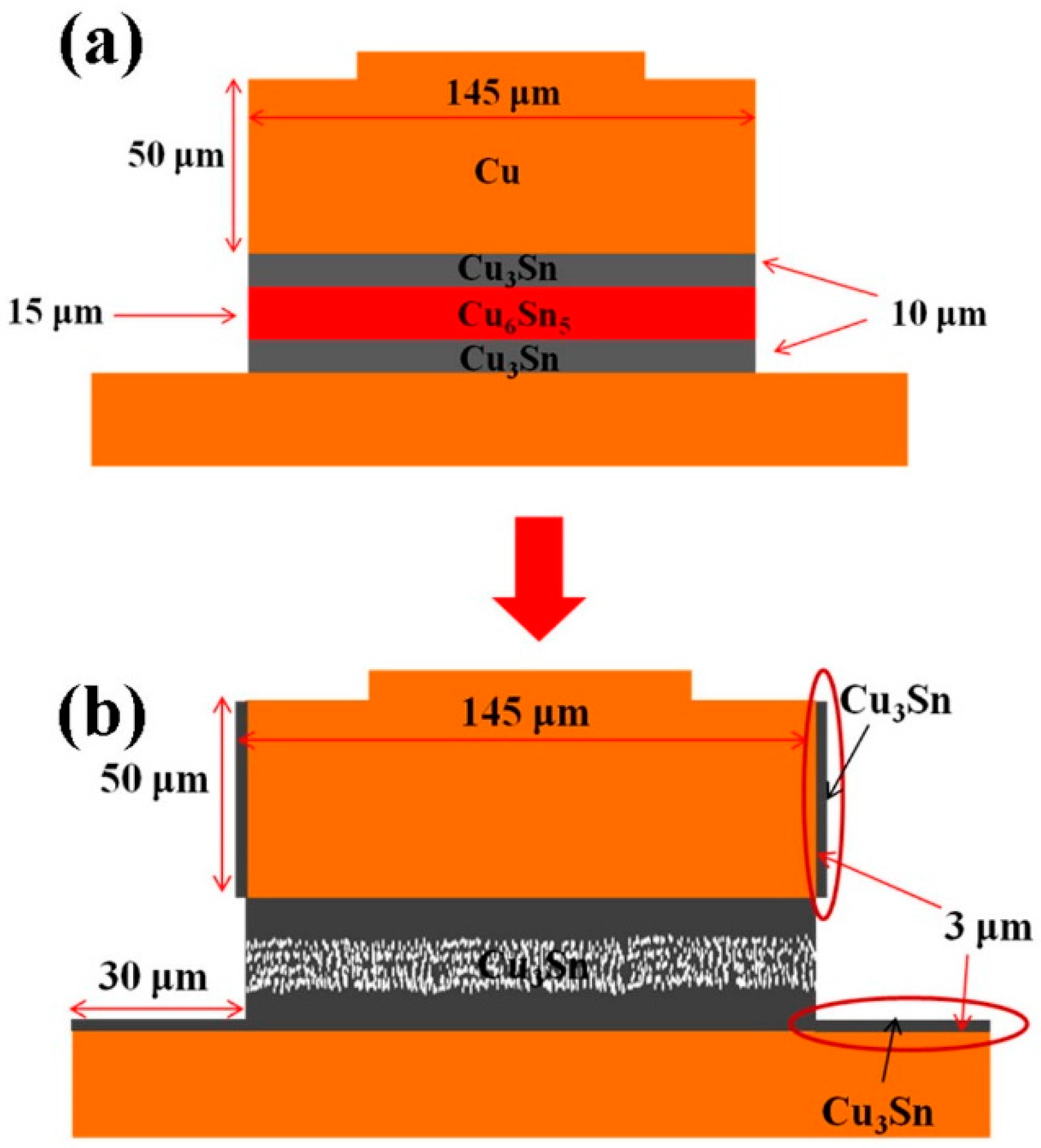
On the formation mechanism of porous Cu
3Sn, first, the temperature must be high enough. Second, the solder height must be low enough so that the Cu supply is enough for the whole joint to transform into IMC. This is because when there are unreacted solders in the joints, the major IMC is Cu
6Sn
5 after the metallurgical reactions, as illustrated in
Figure 1b. Only a thin layer of Cu
3Sn presented between the Cu metallization layer and the Cu
6Sn
5 IMCs. The transformation process is schematically illustrated in
Figure 10. In the early stage of the EM tests, layer-type Cu
6Sn
5 and Cu
3Sn formed first, as shown in
Figure 10a. As the bump resistance increased, the layer-type Cu
6Sn
5 started to transform into porous-type Cu
3Sn, as illustrated in
Figure 10b. The IMCs formed on the side walls due to the effect of side wall reaction are also shown in
Figure 10b, labeled by the ellipses.
According to the experimental results, we have developed the following mechanism to explain the formation of the porous-type Cu
3Sn. Previously, the Kirkendall void formation mechanism in Cu
3Sn was proposed [
17]. When the layer-type Cu
6Sn
5 decomposes into Cu
3Sn, it will release 3 Sn atoms:
The Sn atoms will then attract nine Cu atoms to form three more Cu3Sn.
Figure 10.
Schematic diagrams of transformation process at (a) early stage and (b) final stage in the electromigration tests.
Figure 10.
Schematic diagrams of transformation process at (a) early stage and (b) final stage in the electromigration tests.
The vacancies which enable the diffusion of the Cu atoms tend to form Kirkendall voids in Cu
3Sn. It implies that the growth of layer-type Cu
3Sn IMCs is at the expense of layer-type Cu
6Sn
5 IMCs. However, in our experimental results, it can be observed that as the layer-type Cu
3Sn IMCs grew to certain thickness, the layer-type Cu
6Sn
5 would not transform into the layer-type but porous-type Cu
3Sn. Hsiao
et al. found that Cu-Sn IMCs can become a diffusion barrier for the Cu/solder reaction [
18]. We assume that the layer-type Cu
3Sn served as a diffusion barrier and inhibited the Sn atoms released from the decomposition of Cu
6Sn
5 to react with Cu. Since the bumps were stressed with high current density at high temperature, the released Sn diffused to the side wall to form Cu
3Sn due to the effect of the side wall reaction. The place originally occupied by Sn became empty and led to the porous-type structure.
3.5. Theoretical Calculation of Pore Volume
This mechanism can be further verified by volume calculations. One mole of Cu6Sn5 decomposes into two moles of Cu3Sn plus three moles of Sn. Approximately, one mole of Cu6Sn5 has a volume of 117.7 cm3, two moles of Cu3Sn has a volume of 69.5 cm3, and three moles of Sn has a volume of 48.4 cm3. In our observation, the volume before and after the Cu6Sn5 to Cu3Sn conversion was nearly unchanged (117.7 to 117.9 cm3). Calculations also indicate that the volume change in phase transformation is negligible. On the other hand, the diffusion of Sn to the side walls would leave the pores behind. Assuming that the pore volume equals that of the released Sn, we have calculated the volume occupied by pores to be 41.2% (48.4/117.9) of the porous Cu3Sn volume, when all the released Sn atoms diffuse to the side walls of the UBMs. As a verification, we have used software to calculate the volume percentage of the pores in the actual bumps. Six bumps were input into the software to ensure accuracy. The average volume percentage of the pores was found to be 38.2%, a number close to the theoretical 41.2%. The volume of pores (or the volume of the released Sn) in the bumps is approximately 63,879 μm3. We assume that all of released Sn atoms have diffused to the side walls to form Cu3Sn. This implies that 63,879 μm3 of Sn would form 137,978 μm3 of Cu3Sn on the side walls. This is again close to the calculated value of 157,932 μm3 for the volume of Cu3Sn formed on the side walls in the actual bumps.
The above experimental results and calculation both indicate Sn diffusion to side walls of Cu UBM may be a possible mechanism for the formation of the porous Cu3Sn. The porous Cu3Sn will be a reliability issue for solder joints with Cu UBM. Yet, no solutions have been proposed so far to prevent it from happening. More studies need to be performed to solve this problem.