Chromogenic Mechanism and Chromaticity Study of Brazilian Aquamarine
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods
3. Results
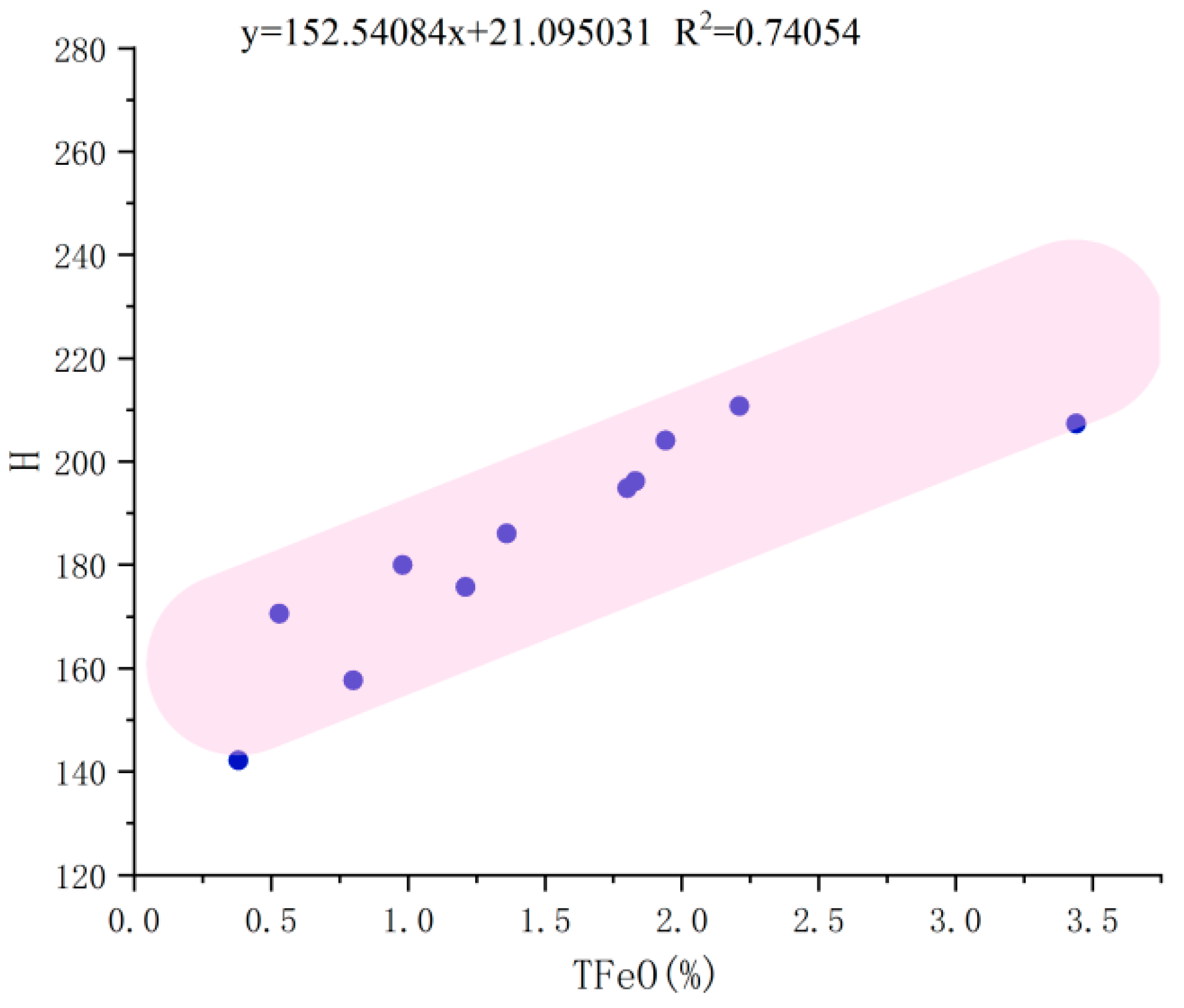
3.1. Electron Probe Microanalysis (EPMA)
3.2. UV-Vis Spectroscopy Analysis
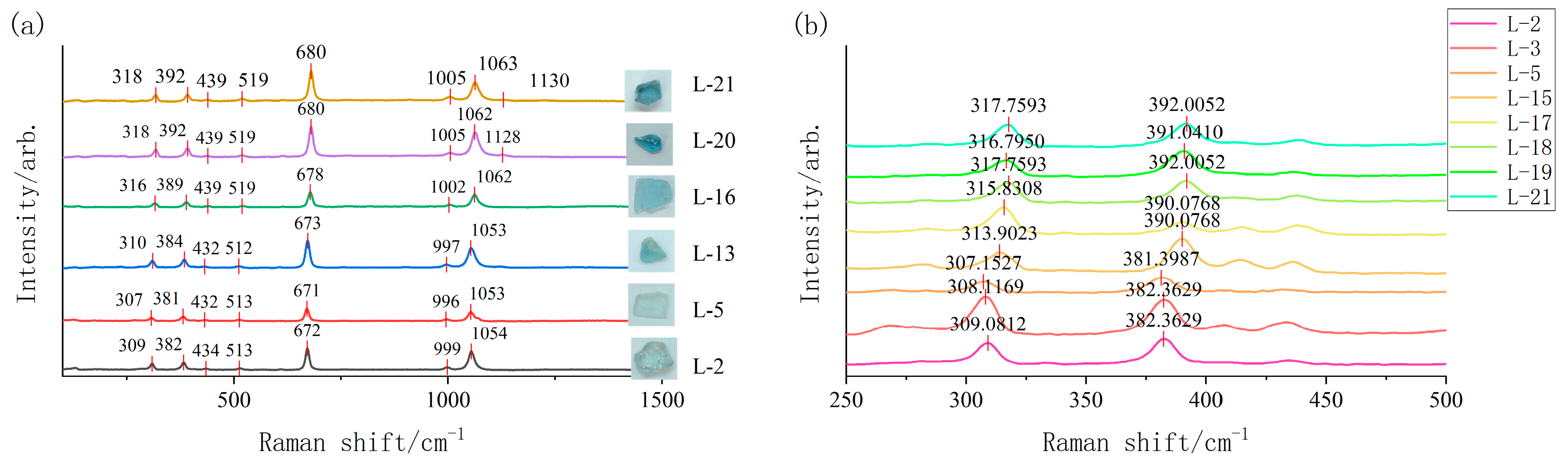
3.3. Raman Spectroscopy Analysis
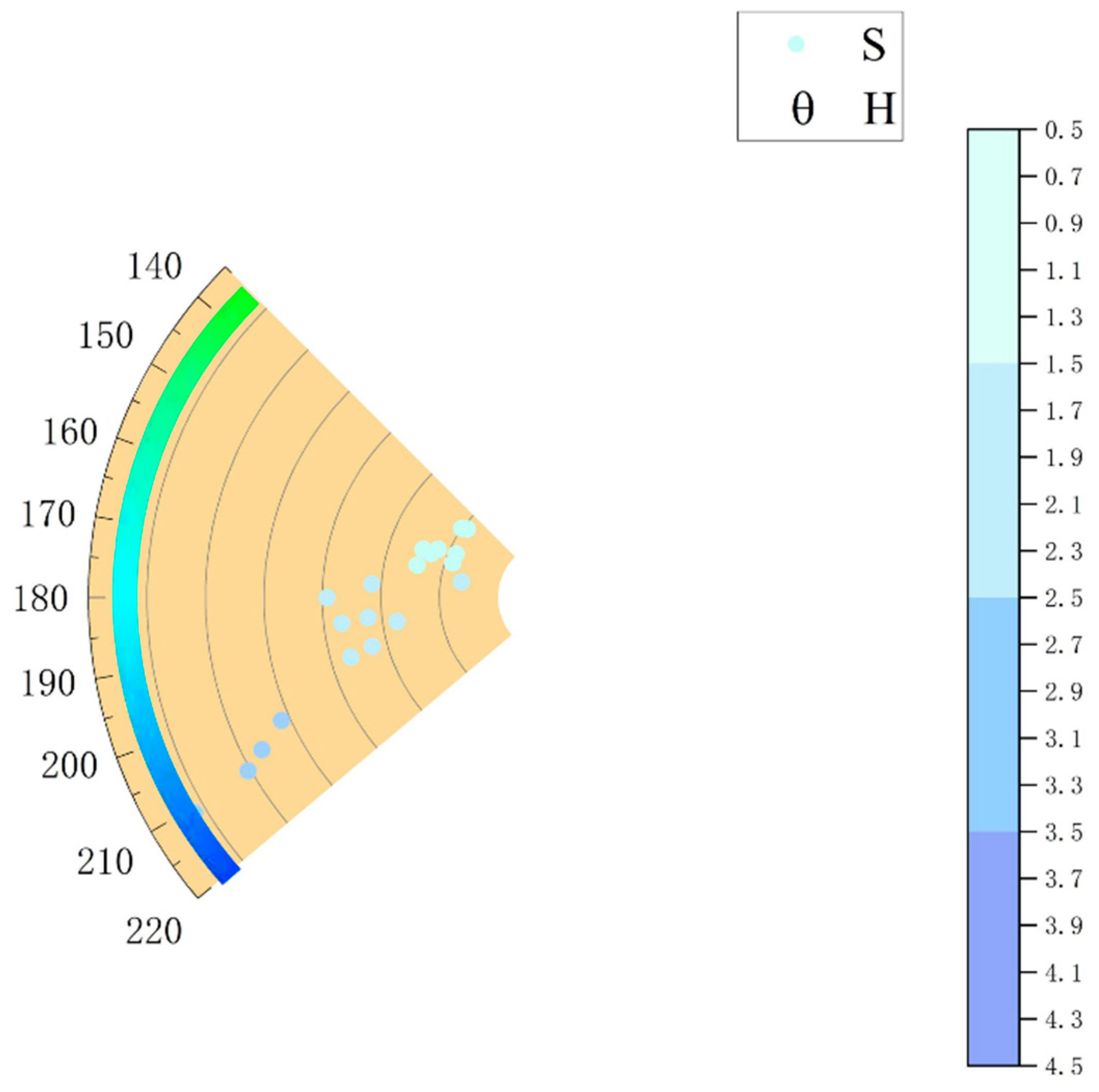
3.4. Chromaticity Experiment Analysis
4. Discussion
5. Conclusions
6. Patents
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zheng, Y.Y.; Yu, X.Y.; Xu, B.; Guo, H.S.; Yan, Y.; Zhang, Y.; Tang, J.; Zhao, S.Y. A review on origin identification of emeralds. Acta Mineral. Sin. 2024, 43, 525–561. [Google Scholar] [CrossRef]
- Gorshunov, B.P.; Zhukova, E.S.; Torgashev, V.I.; Lebedev, V.V.; Shakurov, G.S.; Kremer, R.K.; Pestrjakov, E.V.; Thomas, V.G.; Fursenko, D.A.; Dressel, M. Quantum Behavior of Water Molecules Confined to Nanocavities in Gemstones. J. Phys. Chem. Lett. 2013, 4, 2015–2020. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.; Gan, F.X.; Zhao, H.X. Non-destructive analysis of chemical composition, structure, and phases in natural beryl gemstones. J. Chin. Ceram. Soc. 2015, 43, 10. [Google Scholar] [CrossRef]
- Wu, J.M.; Hu, X.K.; Li, Y.X. Research status and geological implications of beryl. Adv. Geosci. 2022, 12, 1427–1433. [Google Scholar] [CrossRef]
- Bakakin, V.V.; Rylov, G.M.; Belov, N.V. Crystal structure of lithium-bearing beryl. Dokl. Akad. Nauk SSSR 1969, 188, 659–662. [Google Scholar]
- Andersson, L.O. The yellow color center and trapped electrons in beryl. Can. Mineral. 2013, 51, 15–25. [Google Scholar] [CrossRef]
- Spinolo, G.; Fontana, I.; Galli, A. Optical absorption spectra of Fe2+ and Fe3+ in beryl crystals. Phys. Status Solidi B 2010, 244, 4360–4369. [Google Scholar] [CrossRef]
- Qi, L.J.; Ye, S.; Xiang, C.J.; Pei, J.C.; Luo, Y.A. Color centers and chromogenic mechanisms in irradiated beryl. Geol. Sci. Technol. Inf. 2001, 20, 25–28. [Google Scholar] [CrossRef]
- Wu, X.; Han, X.Z.; Feng, X.Q.; Guo, S.G. Lattice occupancy and chromogenic mechanism of iron ions in beryl. Acta Mineral. Sin. 2023, 42, 577–590. [Google Scholar] [CrossRef]
- Dong, J.Y.; Huang, F.; Wang, D.H. Geochemical characteristics and geological implications of beryl from different types of beryllium deposits. Acta Petrol. Sin. 2023, 39, 2153–2166. [Google Scholar] [CrossRef]
- Cui, S.Y.; Shen, J.Q.; Xu, B. Gem-mineralogical characteristics of aquamarine from three origins. China Gems Jades 2022, 32, 8. [Google Scholar]
- Ding, Z.L.; Jiang, Y.; Yu, L.; Yang, Y.M.; Zuo, Y.; Tang, Y.T. Study on the spectral characteristics of aquamarine from Renli, Hunan Province. World Nonferrous Met. 2022, 37, 152–155. [Google Scholar] [CrossRef]
- Qi, L.J.; Ye, S.; Xiang, C.J.; Pei, J.C.; Shi, G.H. Vibrational spectroscopy and irradiation cleavage of complexes in beryl channels. Geol. Sci. Technol. Inf. 2001, 20, 18–22. [Google Scholar] [CrossRef]
- Karampelas, S.; Al-Shaybani, B.; Mohamed, F.; Sangsawong, S.; Al-Alawi, A. Emeralds from the Most Important Occurrences: Chemical and spectroscopic data. Minerals 2019, 9, 561. [Google Scholar] [CrossRef]
- Gao, R.; Chen, Q.L.; Ren, Y.N.; Bao, P.J.; Huang, H.Z. Gemological and spectroscopic characteristics of Kagem emeralds from Zambia. Spectrosc. Spect. Anal. 2023, 43, 3186–3192. [Google Scholar]
- Andersson, L.O. Comments on beryl colors and on other observations regarding iron-containing beryls. Can. Mineral. 2019, 57, 551–566. [Google Scholar] [CrossRef]
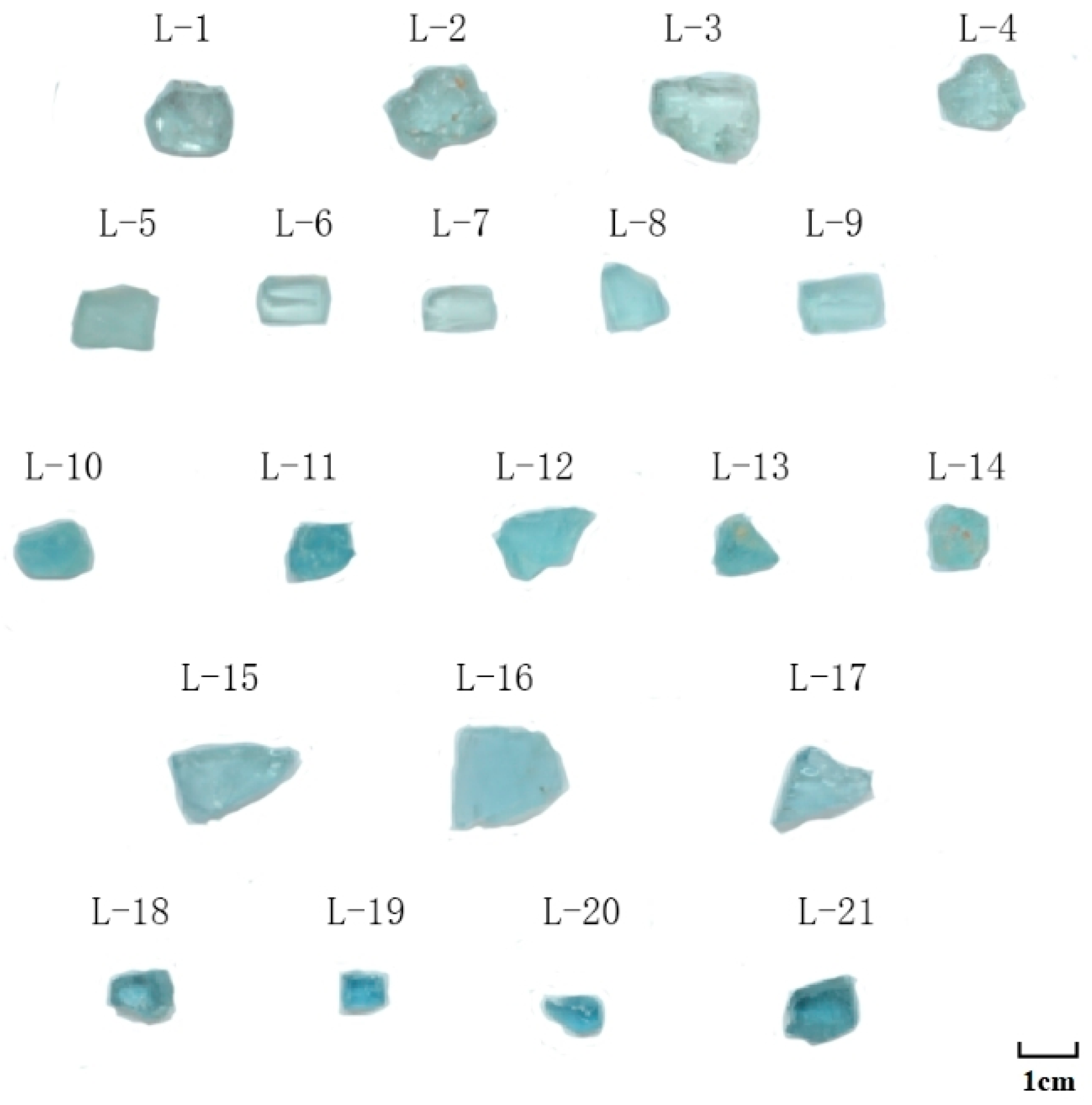



| L−3 | L−5 | L−8 | L−10 | L−12 | L−14 | L−16 | L−17 | L−18 | L−19 | L−20 | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Na2O | 0.56 | 0.66 | 0.68 | 0.64 | 1.30 | 0.91 | 0.38 | 0.80 | 1.47 | 1.98 | 1.73 |
| MgO | - | 0.19 | 0.04 | 0.13 | 0.42 | 0.83 | 0.21 | 0.42 | 1.38 | 1.03 | 1.02 |
| Al2O3 | 18.66 | 19.13 | 18.76 | 21.91 | 17.26 | 18.43 | 19.51 | 18.77 | 16.92 | 16.52 | 19.39 |
| SiO2 | 66.09 | 65.07 | 65.31 | 61.95 | 63.52 | 64.94 | 64.20 | 62.57 | 64.60 | 59.89 | 61.95 |
| P2O5 | 0.27 | - | 0.38 | 0.26 | 0.57 | 0.04 | - | 0.91 | - | 1.68 | - |
| K2O | 0.10 | 0.19 | 0.57 | 0.24 | 1.47 | 0.27 | 0.22 | 0.63 | 0.39 | 1.99 | 0.39 |
| CaO | 0.02 | 0.01 | 0.01 | 0.01 | 0.26 | - | 0.04 | 0.22 | 0.03 | 0.12 | 0.03 |
| TiO2 | 0.09 | 0.15 | 0.14 | - | 0.77 | 0.12 | 0.28 | 0.40 | - | 0.25 | - |
| V2O5 | 0.01 | 0.12 | - | - | 0.15 | - | 0.04 | 0.04 | - | - | - |
| Cr2O3 | 0.11 | 0.14 | - | - | - | 0.12 | - | 0.20 | - | 0.03 | 0.16 |
| TFeO | 0.38 | 0.80 | 0.53 | 1.36 | 0.98 | 1.21 | 1.80 | 1.83 | 1.94 | 3.44 | 2.21 |
| NiO | 0.02 | - | - | 0.09 | 0.05 | - | - | - | - | - | - |
| ZnO | 0.45 | 0.36 | 0.41 | 0.31 | 0.22 | - | 0.22 | 0.15 | 0.30 | 0.35 | 0.16 |
| BeO | 12.90 | 13.11 | 13.12 | 13.07 | 12.97 | 13.08 | 13.05 | 13.02 | 12.94 | 12.66 | 12.92 |
| H2O | 1.31 | 1.40 | 1.41 | 1.38 | 1.94 | 1.61 | 1.16 | 1.52 | 2.08 | 2.52 | 2.31 |
| Total | 100.97 | 101.33 | 101.36 | 100.35 | 101.88 | 101.56 | 10 1.11 | 101.48 | 102.05 | 102.46 | 102.27 |
| Na | 0.099 | 0.118 | 0.121 | 0.114 | 0.232 | 0.161 | 0.068 | 0.143 | 0.263 | 0.362 | 0.311 |
| Mg | - | 0.027 | 0.006 | 0.018 | 0.058 | 0.114 | 0.029 | 0.058 | 0.190 | 0.145 | 0.141 |
| Al | 2.317 | 2.380 | 2.337 | 2.359 | 1.873 | 2.309 | 2.429 | 2.033 | 2.166 | 1.832 | 2.111 |
| Si | 5.724 | 5.662 | 5.678 | 5.659 | 5.848 | 5.661 | 5.602 | 5.748 | 5.682 | 5.634 | 5.721 |
| P | 0.021 | - | 0.029 | 0.021 | 0.045 | 0.003 | - | 0.071 | - | 0.134 | - |
| K | 0.012 | 0.023 | 0.067 | 0.028 | 0.174 | 0.032 | 0.026 | 0.074 | 0.046 | 0.239 | 0.047 |
| Ca | 0.003 | 0.002 | 0.001 | 0.001 | 0.027 | - | 0.005 | 0.022 | 0.003 | 0.013 | 0.003 |
| Ti | 0.006 | 0.011 | 0.010 | - | 0.053 | 0.009 | 0.019 | 0.028 | - | 0.018 | - |
| V | 0.001 | 0.008 | - | - | 0.010 | - | 0.003 | 0.003 | - | - | - |
| Cr | 0.008 | 0.011 | - | - | - | 0.009 | - | 0.015 | - | 0.003 | 0.012 |
| TFe | 0.029 | 0.062 | 0.041 | 0.104 | 0.076 | 0.093 | 0.138 | 0.141 | 0.150 | 0.271 | 0.171 |
| Ni | 0.002 | - | - | 0.007 | 0.004 | - | - | - | - | - | - |
| Zn | 0.031 | 0.024 | 0.028 | 0.021 | 0.015 | - | 0.015 | 0.010 | 0.021 | 0.025 | 0.011 |
| Be | 2.876 | 2.866 | 2.874 | 2.869 | 2.869 | 2.872 | 2.872 | 2.851 | 2.868 | 2.853 | 2.868 |
| Li | 0.124 | 0.134 | 0.126 | 0.131 | 0.131 | 0.128 | 0.128 | 0.149 | 0.132 | 0.147 | 0.132 |
| L-1 | L-2 | L-3 | L-4 | L-5 | L-6 | L-7 | |
| H | 156.37 | 143.73 | 142.21 | 161.48 | 157.7 | 159.91 | 166.76 |
| S | 9.29 | 9.98 | 9.58 | 9.3 | 10.88 | 8.18 | 10.19 |
| V | 99.67 | 100 | 100 | 100 | 99.2 | 100 | 100 |
| L-8 | L-9 | L-10 | L-11 | L-12 | L-13 | L-14 | |
| H | 170.59 | 160.55 | 186.07 | 186.85 | 180 | 188.57 | 175.74 |
| S | 15.09 | 11.3 | 16.17 | 18.44 | 19.55 | 13.73 | 15.78 |
| V | 99.34 | 100 | 100 | 100 | 100 | 100 | 100 |
| L-15 | L-16 | L-17 | L-18 | L-19 | L-20 | L-21 | |
| H | 195.3 | 194.87 | 196.21 | 204.09 | 207.35 | 210.77 | 209.34 |
| S | 18.34 | 16.25 | 18.26 | 25.7 | 28.29 | 34.94 | 30.2 |
| V | 99.45 | 98.71 | 97.65 | 100 | 96.6 | 98.88 | 94.47 |
| L-1 | L-2 | L-3 | L-4 | L-5 | L-6 | L-7 | L-9 | |
|---|---|---|---|---|---|---|---|---|
| Light blue |  |  |  |  |  |  |  |  |
| Simulate color blocks |  |  |  |  |  |  |  |  |
| L-8 | L-10 | L-11 | L-12 | L-13 | L-14 | L-15 | L-16 | L-17 | |
|---|---|---|---|---|---|---|---|---|---|
| Sky blue |  |  |  |  |  |  |  |  |  |
| Simulate color blocks |  |  |  |  |  |  |  |  |  |
| L-18 | L-19 | L-20 | L-21 | |
|---|---|---|---|---|
| Ocean blue |  |  |  |  |
| Simulate color blocks |  |  |  |  |
| H:230 S:40 | |
|---|---|
| Deep blue |  |
| Simulate color blocks |
| L-1 | L-2 | L-3 | L-5 | L-6 | L-7 | L-8 | L-9 | L-10 | L-11 | |
| Al-O (cm−1) | 308.12 | 309.08 | 308.12 | 307.15 | 309.08 | 309.08 | 307.15 | 307.15 | 309.08 | 308.12 |
| ∆H | 12.88 | 11.92 | 12.88 | 13.85 | 11.92 | 11.92 | 13.85 | 13.85 | 11.92 | 12.88 |
| L-12 | L-13 | L-14 | L-15 | L-16 | L-17 | L-18 | L-19 | L-20 | L-21 | |
| Al-O (cm−1) | 309.08 | 310.05 | 309.08 | 313.90 | 315.83 | 315.8 | 317.76 | 316.80 | 317.76 | 317.76 |
| ∆H | 11.92 | 10.95 | 11.92 | 7.10 | 5.17 | 5.17 | 3.24 | 4.2 1 | 3.24 | 3.24 |
| L-1 | L-2 | L-3 | L-5 | L-6 | L-7 | L-8 | |
| L | 97.83 | 97.85 | 97.92 | 97.13 | 98.36 | 98.05 | 96.68 |
| a | −9.38 | −11.07 | −10.73 | −10.82 | −8.07 | −9.49 | −13.42 |
| b | 1.83 | 4.54 | 4.65 | 1.87 | 1.01 | −0.13 | −1.24 |
| L-9 | L-10 | L-11 | L-12 | L-13 | L-14 | L-15 | |
| L | 97.77 | 96.15 | 95.48 | 96.71 | 96.33 | 97.21 | 93.34 |
| a | −11.06 | −11.08 | −12.25 | −15.76 | −8.75 | −13.42 | −8.84 |
| b | 1.3 | −5.98 | −7.01 | −5.21 | −5.67 | −2.94 | −9.46 |
| L-16 | L-17 | L-18 | L-19 | L-20 | L-21 | ||
| L | 93.48 | 91.69 | 88.9 | 84.14 | 81.57 | 81 | |
| a | −8 | −8.32 | −7.13 | −5.49 | −3.62 | −4.34 | |
| b | −8.24 | −9.54 | −16.94 | −19.62 | −26.67 | −21.53 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, Z.; Zu, E.; He, X.; Wang, Z.; Wang, D.; Sun, Y.; Wang, Y.; Yang, S. Chromogenic Mechanism and Chromaticity Study of Brazilian Aquamarine. Crystals 2025, 15, 775. https://doi.org/10.3390/cryst15090775
Zhang Z, Zu E, He X, Wang Z, Wang D, Sun Y, Wang Y, Yang S. Chromogenic Mechanism and Chromaticity Study of Brazilian Aquamarine. Crystals. 2025; 15(9):775. https://doi.org/10.3390/cryst15090775
Chicago/Turabian StyleZhang, Zheng, Endong Zu, Xiaohu He, Zixuan Wang, Die Wang, Yicong Sun, Yigeng Wang, and Siqi Yang. 2025. "Chromogenic Mechanism and Chromaticity Study of Brazilian Aquamarine" Crystals 15, no. 9: 775. https://doi.org/10.3390/cryst15090775
APA StyleZhang, Z., Zu, E., He, X., Wang, Z., Wang, D., Sun, Y., Wang, Y., & Yang, S. (2025). Chromogenic Mechanism and Chromaticity Study of Brazilian Aquamarine. Crystals, 15(9), 775. https://doi.org/10.3390/cryst15090775






