3.1. Purity of Materials
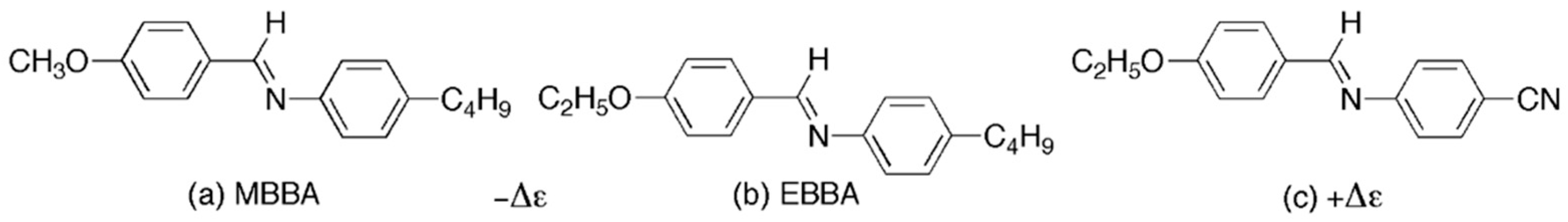
Common materials used in the USA and Europe in various liquid crystal applications, including displays, were based on the Schiff’s bases MBBA and EBBA [
9,
15], see
Figure 2, and on alkyl carbonato-alkoxyphenyl benzoates. Heilmeier at RCA reported privately a three-component formulation at that time, which gave the best transitions for a nematic phase of 24 to 76 °C. In the UK, it was well known that such materials were electrolytically unstable, easily oxidised and could degrade on exposure to UV radiation. This can happen in their preparation, storage and usage. Their use in dynamic scattering mode devices was found to result in displays having differing lifetimes depending on material purity. Hull worked on a variety of synthesized Schiff’s bases, stilbenes, carbonates, carboxylic esters, ultra-pure Schiff’s bases, etc., but to little effect. There were difficulties in purifying them. In particular, there were problems with low melting variants, which, when it came to recrystallisation at a low temperature, one would need to sit inside a large-size refrigerator to perform vacuum filtrations at −10 °C so that emulsification or the formation of lyotropic phases did not occur.
Although coloured, Schiff’s bases, in addition to having negative dielectric anisotropies, are relatively easy to prepare, thereby providing a variety of materials for use in multicomponent mixtures. However, even in the design and preparation of these materials they still had further problems due to exchange reactions occurring whilst being located in devices. As a consequence, the components of mixtures were required to possess structures where one of the exchangeable moieties is the same. For MBBA and EBBA, the exchangeable unit is the right-hand side of the molecules as shown in
Figure 2. The aniline part of the molecule can flip from MBBA to EBBA without changing the mixture composition. This of course limits the selection of material components for mixtures. The fact that electro- and photochemistry is still ongoing in devices is often not realised, particularly where small changes in purity can affect properties. Not only is this apparent in DSM devices, it also can occur in smectic A devices, dye devices, OLEDs, etc.
Nevertheless, MBBA was still particularly popular with the industrial laboratories in the 1970s because it was found to be a room temperature nematogen, however, the feedback on its transition temperatures was somewhat variable. It was not appreciated at the time that the purity of a material for use in an electronic device should be aimed at reaching a purity that was near that of the electronic components of the device. Therefore, it became an objective to provide a supply of ultrapure MBBA of purity greater than expected from organic laboratories.
There were also big problems of materials analysis in those days to evaluate purities as there were no Fourier transform instruments, or high-performance chromatography equipment, thus making pure materials dependent on the skills of the chemist and the use of thin-layer chromatography (TLC). To this day, there are many materials that are commercially available or from academic laboratories that are not pure enough. In the following, the preparation of MBBA is described using the methods of that era in the UK.
MBBA could be prepared via the condensation of equimolar amounts of 4-butylaniline and 4-methoxybenzaldehyde by heating them in ethanolic solution with a trace of glacial acetic acid as a catalyst. However, the products from such a method produced other materials that were not so easy to separate. So, the first point was to use a method that produced less in terms of by-products. Thus, it appeared important to allow the reaction mixture to stand in the dark for 12 h after the reaction had been completed, but longer times were found to yield more highly coloured products. Deep refrigeration of the reaction mixture gave crystals that could be filtered off, which were often difficult to separate from solution because any rise in temperature during filtration would result in the formation of a gel or lyotropic phase. The transition temperatures for a purified product were Cryst to N at 21 °C and N to liquid at 41–43 °C, which was similar to commercial materials. Two crystallisations with light petroleum (bp 40–60 °C) gave constant transition temperatures of Cryst to N at 21 °C and N to liquid at 45 °C, determined by thermal polarised light microscopy [
16,
17]. Distillation via vacuum sublimation under reduced pressure did not improve the situation. MBBA was also found to be sensitive to moisture, and prolonged evacuation over CaCl
2 was necessary. Various storage conditions were then applied, and it was found that being under vacuum in the presence of P
2O
5 at room temperature for several days improved the transition temperatures from Cryst to N at 21 °C and N to liquid at 47 °C. These constants have not been improved since, and the temperatures are considered to be those for pure MBBA. Such a product was found to give a single spot-on neutral TLC plate (N.B. slightly acidic or basic plates produce a cleavage of the Schiff’s base). The analysis of the purified material was found to be consistent with IR spectrum and mass spectrometric data for the structure of MBBA. Within the UK, pure samples of MBBA were supplied to various team members, either to be stored in sealed ampoules or when in use in a vacuum over a desiccant such a P
2O
5. Under these conditions, in comparison to commercial samples, the ultrapure material was much paler, and preliminary information indicated that an electro-optic effect was found under normal electrical addressing. For such a pure product, it was estimated that MBBA could be prepared at a cost of around 0.1 pence per gram for an overall yield of ~47%. In contrast, commercial samples of MBBA were found to be considerably impure, wet or both, and as such the physical studies of such samples were doubtful, and applications could be rendered worthless.
Generally, the measurements of dielectric coefficients ε
ıı and ε
⊥ became possible, along with the determination of the resistivities of nematogens. High purity MBBA had a resistivity of 2 × 10
11 Ωcm. It is interesting that at such high resistivities, dynamic scattering devices had relatively short lifetimes, lasting approximately 10 h, indicating that these devices required the incorporation of ionic dopants in order to generate more stable displays (the need to incorporate ionic dopants in “DSM-type” devices tended to make them unsuitable for commercial applications). Under these conditions, MBBA has a resistivity of ~10
10 Ωcm. In comparison, materials used in the twisted nematic construction [
18] were required to have resistivities of ~10
9 Ωcm. Consequently, high purity Schiff’s bases possessing terminal-substituted cyano-units and resistivities in the region of 10
10 Ωcm were a factor in obtaining good quality devices, and to chemists, measurements of the purities of their materials became determined by resistivity.
Thus, MBBA was found to be useful only as an experimental material that would never be applied in practice. For practical applications, a material should be as pure as possible, and in a simple experiment, TLC can be used to indicate if the material is a single species (such tests are used today in lateral flow tests for COVID infections). In the cases of electronic devices, purities should be in the region of 99% or have resistivities of ~10
10 Ωcm or better. A material should also give reproducible physical results. Its transition temperatures should remain constant with time and temperature, and without decomposition [
19]. As George Gray would retort: “
To be a reputable synthetic chemist is to supply collaborators with materials checked to be of the highest purities, and for recipients in doubt, TLC can be used as a check for worries over purities”.
3.2. Materials Designed to Fit
Once purity was recognised as important, two other items came into view in the development of device materials. With the inventions of new property-testing methods, property-structure correlations were amassing and allowing for the specific design of the structures of new molecular entities via feedback mechanisms. The utilisation of materials in various mixture formulations for a variety of devices requires upscaling for the production of materials, which meant at that time, the development of new synthetic methods in order to move from grams to tens of grams to kilograms of high purity compounds. With discovery aspects ongoing at Hull, BDH was contracted to provide a supply of selected materials, thereby increasing the pace of the activities of the UK Consortia. As noted above, MBBA still remained popular because its negative dielectric anisotropy suited applications in DSM devices. However, owing to its stability issues, with lifetimes rarely exceeding 3000 h, it started to become replaced by azo-benzenes and carbonate esters as reported by Heilmeier [
12], which also had negative dielectric anisotropies, but of a stronger level in comparison, due to the larger lateral molecular dipole. At Hull, azo-benzenes were not perused due to their poor UV stability, whereas the carbonate esters were more stable, but unfortunately, they had higher melting points. By placing a polar group at the terminus in analogues of MBBA, a dipole pointing along the molecular long axis was achieved, the dielectric anisotropy was positive, and the material was suitable for use in the twisted nematic display (TNLCD) of Schadt and Helfrich [
18,
20,
21]. Other devices also came into play [
22], including the phase change device and the electrically controlled birefringence (ECB) display, which now required further tuning to the molecular design.
Materials such as Schiff’s bases sufficed for the early work, but with low values of −Δε, they were soon replaced with carbonate esters, which had larger lateral dipoles. Carbonates are not particularly stable and so derivatives of trans-stilbene were thought to be suitable alternatives. For such materials, a lateral polar unit (Cl or CN) could be placed in the linking chain between the phenyl moieties. However, the stabilities of stilbenes with respect to cis–trans isomerisation were in question, as were the difficulties in their syntheses. At a similar time, heterocyclic systems were also being investigated where a phenyl unit was replaced with a pyridine ring, where the nitrogen atom could be located in different positions. Although of interest, they still had cis–trans stability issues and high transition temperatures. Therefore, the family of carbonate esters was also extended via the preparation of 4′-alkoxyphenyl 4-alkoxybenzoates.
Many materials at this particular time were unsuitable to be used in devices; they did not have suitable transition temperatures, were too high melting, unstable in devices, impure, difficult to synthesise, etc. Gray’s futuristic targets before the discovery of cyanobiphenyls in 1972, included materials based on the incorporation of bicyclo-octanes and bicyclo-octenes and ring systems such as cyclohexyl moieties [
23,
24], see
Figure 3, which no doubt he may have discussed previously with Dewer, who was his PhD examiner. Indeed, the abstract from Dewar and Goldberg’s 1972 paper [
25] states that
“Many compounds forming nematic mesophases contain p-phenylene units. It is shown that these perform a dual function, providing rigid linear groupings and contributing to the polarizability of the molecule. These conclusions are based on a comparison with compounds where benzene rings are replaced by cyclohexane or bicyclo[2.2.2.]octane”. The inclusion of such moieties in molecules possessing rod-like structures (see the top of
Figure 3) was probably not envisioned to be for materials with positive dielectric anisotropies, but the lower structures in the figure were probably being considered. It was not obvious at the time, nor was there an immediate need to produce them because of the synthetic complexities involved.
It should be noted that subsequent work by Gray and Kelly resulted in the production of bicylo-octane mimics of cyanobiphenyls [
26,
27,
28,
29,
30,
31]. The use of cyclohexyl moieties in molecular architectures was performed by Deutscher et al. [
32,
33,
34], Osman and Revesz [
35] and the well-known device materials by researchers at E. Merck [
36,
37,
38,
39,
40,
41]. Frustration, however, was growing as most materials were missing the mark. George Gray sought for a common feature in molecular design of all of the materials that he thought might be causing a problem, and he settled on the linking unit between phenyl rings. Its removal, he thought, could provide the change in structure that might make the materials useful liquid crystals.
3.3. A Scientific Revolution
Across research laboratories in the USA, UK and Europe, by the middle of 1972, it appeared that there were still problems with the purities of Schiff’s bases, the quantities of materials were still tight, the temperature ranges not suitable, and the dynamic scattering mode was not fully reproducible even though it appeared better than the twisted nematic device; something had to give. Gray’s futuristic concepts were set aside, and the simple idea of eliminating the central linkage (-CH=N-, -CH=CH-, -COO-) from the materials previously prepared to give 4,4′-substituted biphenyls took precedence. Gray had already used biphenyls along with lateral fluorination (see later) 15 years earlier [
42] in the study of smectic liquid crystals. A second accompanying idea was the use of the nitrile (CN) group in the terminal position to give colourless materials with strong positive dielectric anisotropies. The cyano-equivalent in Schiff’s bases were first reported in Castellano in 1968 [
43], and later by Boller and Scherrer [
44]. The remarkable combination of biphenyls and nitrile resulted in nematic materials that would operate at room temperature in devices that used materials with positive dielectric anisotropy [
45,
46,
47]. Thus, in 1972, with many still working in the area of light scattering devices, 4-pentyl-4′-cyanobiphenyl (5CB, K15) took to the stage, and when it was incorporated into the Schadt-Helfrich (+Δε) device, the revolutionary twisted nematic display (TNLCD) [
18] was born.
As a consequence, Gray would often use the discovery of cyanobiphenyls as an example of the importance of basic research; to quote him from his award of the 1995 Kyoto Prize of the Inamori Foundation of Japan [
48], he said, “
I knew what I was doing by using the cyano-group to compensate for loss of molecular length, while at the same time providing the strongly polar molecular structure needed for the electric field to switch on the display. This I stress was not luck…the fundamental science was secure…we knew what we were doing”.
To us, and many others, George Gray emphasised “the idea behind the programme was simply the elimination of the central linkage in all of the previous systems to give stable 4′,4-disubstituted biphenyls, and that Ken Harrison undertook their syntheses”. However, we knew about the extensive fundamental studies on liquid crystals Gray had performed over the previous 20 years.
In reporting the invention of the cyanobiphenyls to the Consortium, members were reminded of the confidential nature of the work and that the new materials were the invention and property of the group in Hull, and it was the duty of the rest of the group to protect their position. The realisation of the Hull outcome struck home with the need for a more secure source of materials. So, it was decided that BDH would also supply the new materials, leaving the Hull group free to carry on with research, and BDH would become subsequently a full member of the network.
Gray also raised a number of points that he thought were important for the development of cyanobiphenyls [
48], of which we identify a few that are relevant here.
“Production of a new class of commercial materials cannot be driven to maximum advantage by an individual or a university group. The success therefore owed much to the partnership we had with the Defence Research Agency at Malvern and the commercial producer of our materials—BDH Chemicals Ltd. in the UK-now Merck (UK) Ltd. (London, UK). Without these alliances, the impact of the materials would not have occurred. The importance of collaboration also lies in the ease with which development problems and new needs can be tackled swiftly. The rapid commercialisation of the new materials owed a great deal to the easy relationship which developed between the University chemists and the large-scale industrial chemists and to the marketing skills of the staff at BDH Ltd. (Poole, UK)”
After the discovery and development of the cyanobiphenyls, the group at Hull became involved with the syntheses of various analogues of the cyanobiphenyls. The family of biphenyls included the alkoxy-cyanobiphenyls, alkyl- and alkoxy-terphenyls, and chiral cyanobiphenyls, di-esters and fluorenes, as shown in
Figure 4. It is interesting that many of these motifs for materials construction were discussed in Gray’s textbook “
Molecular Structure and the Properties of Liquid Crystals”, published by the Academic Press Incorporated, London and New York, in 1962 [
42].
Figure 4 shows how close Gray was to preparing cyanobiphenyls in 1962 and even to materials of interest today [
49]. For example, the nitrobiphenyls and nitroterphenyls shown in
Figure 4, are mimics of the cyano analogues as they have strong longitudinal polarities and large positive dielectric anisotropies, the biphenyl carboxylic acids are synthetically only one step away from the cyano-materials, whereas the lateral halogeno-materials are the forerunners of lateral-fluorinated compounds that dominated material design in the 1990s through to today. Surprisingly, Gray also prepared a lateral nitro-substituted biphenyl-carboxylic acid that was later found to exhibit a new phase called the D phase [
50], and subsequently characterised as being cubic or a bicontinuous phase [
51,
52].
In terms of lateral substituents in Schiff’s bases, Gray also examined many property–structure correlations for smectics and nematics and found many trends. In combination with Dewer [
25], the group efficiency order obtained by Dewer was
After the initial discovery of 4-pentyl-4′-cyanobiphenyl (5CB), various alkyl and alkoxy cyano-substituted materials, as shown in
Figure 5, were prepared in order to provide a range of materials for mixture formulations. The terphenyls were to be used to raise the clearing points, and the chiral analogue, derived from (
S)-2-methylbutanol, was used for chiral nematic mixtures and potential applications in phase change devices. The fluorene materials were designed to give the phenyl units in a potentially flat molecular architecture in the hope of improving relative physical properties. However, their melting points were much higher than the biphenyl analogues, and either the materials were non-mesogenic or had very low clearing points, and so they were not pursued further.
In 1962, Gray [
42] also reported on 4-methoxy-4’-nitrobiphenyl, as shown in
Figure 4; but as it was nonmesogenic, a question was left open—could the nitro terminal group offer a better option than the nitrile as a terminal substituent? Therefore, a separate property–structure investigation was launched to prepare the nitro analogues of the alkoxy cyanobiphenyls as shown in
Table 1. The table shows the comparative transition temperatures for the alkoxy cyano- and nitro-biphenyls with the same alkyl chain lengths (C
5 to C
8). It can be seen that the nitro-analogues had comparatively lower clearing (isotropisation) points than the nitrile compounds, and also shorter mesophase temperature ranges. It was concluded for practical purposes that the nitro-compounds were not competitive with the nitrile-analogues. Thus, this was the start of the development of numerous
structure–property correlations for applications of materials in devices.
3.4. Formulation of Mixtures
Property–structure correlations certainly gave the synthesis of materials at least some form of prediction on target design, but for practical applications there is always a need to formulate mixtures with wide temperature ranges, including room temperature. This means there is a requirement to know details about the eutectic points, for example, the relative proportions of the components in the mixture at the eutectic point. Why the eutectic point?
For two-component systems, provided that the equilibrium relies only on temperature, pressure and concentration, the phase rule states that the number of degrees of freedom (F) of the system is related to the number of components (C) and of phases (P) present at equilibrium by the equation: F = C − P + 2. At the eutectic point, the solids of the two components are in equilibrium with the liquid phase. There are consequently three phases present, and since the system involves two components, there can be only one degree of freedom according to the phase rule. Since the pressure is arbitrarily fixed at 1 atm, this represents one degree of freedom, and therefore the system has effectively no degree of freedom. This means that the eutectic point is completely defined and there is only one temperature where this equilibrium is possible. The point at which this happens is the lowest temperature at which any liquid mixture can be in equilibrium with the solid phases of the two components and is also the lowest temperature at which any mixture of the two will melt. Thus, the eutectic point similarly appears to be the melting point of a pure compound. With the liquid-crystal-to-isotropic-liquid transition, the nematic and liquid phases are fluids, and the transition temperatures from the nematic phase to the liquid tend to vary linearly with concentration in binary mixtures. Therefore, the broadest temperature range nematic phase appears to be between the lowest melting point (eutectic) and the corresponding clearing point (N to I).
In principle, the eutectic mixtures from two different binary phase diagrams can be used to produce a third eutectic point, thereby reducing the relative melting point of the third eutectic composition of four components. This means that the solidification can be suppressed, whereas the clearing point can be weight-averaged. Ultimately multicomponent mixtures can be developed to further suppress the melting point while at the same time weight-averaging the clearing point. In practice, for binary mixtures the eutectic point can be determined experimentally, whereas for a multicomponent system determining the eutectic point is very time consuming, and a hit or miss process. Rapid evaluations are therefore required by theory and verified by experiment.
Early studies were performed on mixtures of MBBA and EBBA, but with cyanobiphenyls the aim by experiment was simply to obtain the lowest possible melting points consistent with not sacrificing nematic thermal stability too greatly. The objective being to obtain mixtures melting, as distinct from solidifying at <0 °C and giving nematic properties up to about 50 °C.
The results were obtained by optical microscopy using heated or cooled stages, whereby the confirmations of melting temperatures were obtained by differential thermal analysis (DTA) [
17]. DTA was used because of the problems of detecting the melting point by microscopy due to the paramorphotic defect textures of the solid state affecting or overpowering the textures of the liquid crystal. The results obtained were reasonable attempts to produce mixtures that could be compared to results found via various theories. However, to yield mixtures with the highest N-I value and the lowest mp at the time would have been serendipity. Better theories started to be deployed in 1973 for the estimations of eutectic points of mixtures, in the shape of the Schröder–van Laar Equation (1)
where ΔH
oi is the molar heat of fusion for component I, T
oi is the melting point of pure component i (K), R is the gas constant and x
i is the mol fraction of component i. The results obtained were not as accurate as desired and so the theory was extended for determining the eutectic point of multicomponent mixtures via a semiempirical form of the Schröder–van Laar equation [
57,
58,
59]. Melting points of eutectic mixtures were usually obtained to within 5 °C of the experimental results. These methodologies were used to create mixtures such as E7, E8, etc., as shown in
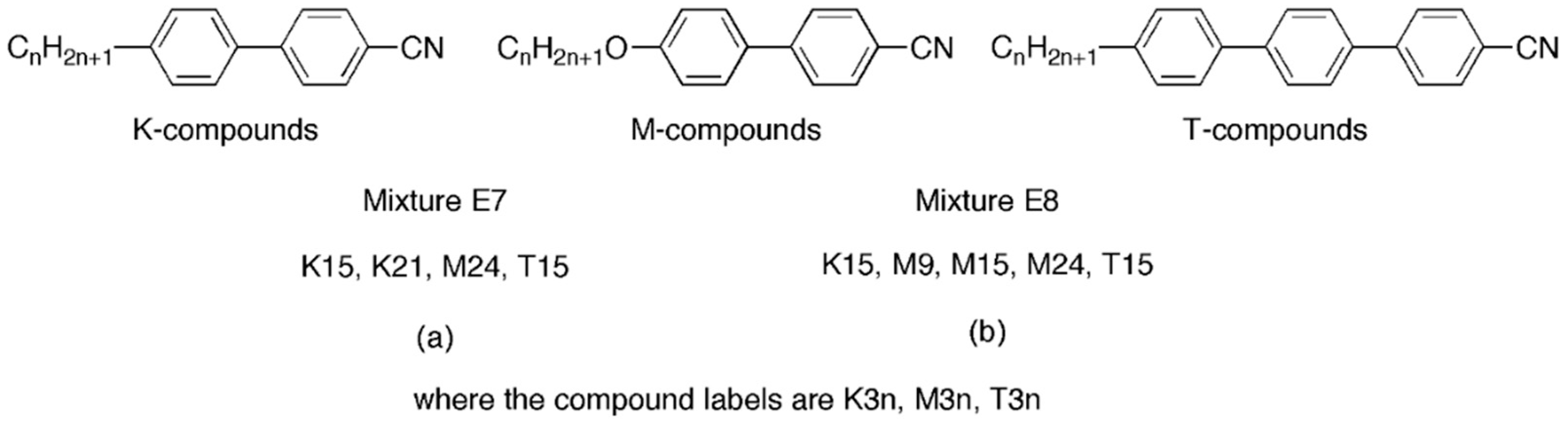
Figure 6, with E7 becoming one of the most popular formulations used in research.
From the beginning of the consortia in 1970, over a period of around 6 to 10 years, the development of the flat-panel industry was being revolutionised by international companies, universities and research establishments. Firstly, in the UK there were searches for information on displays and their developments from external sources, such as with companies as RCA, Bell Laboratories Ltd., TI Instruments and Ilixco, the formation of the Optel Corporation, input on materials from Merck, papers from conferences at the 1970 IEEE conference on Display Devices in NY, the International Conference on Liquid Crystals, articles from leaders (Castlelarno, Helfich and Schadt) in the field, etc. There were also an extremely large variety of inputs from the various members of the consortium, which included a number of government establishments, companies and surprisingly only one university (Hull). Externally, there were other inputs from materials suppliers and skill sets from universities and academics. People also joined the consortium, noticeably for Hull, as the university’s short contract was extended with an increase in research assistants to two, when Ken Harrison joined John Nash towards the end of 1971. Even in the first year of existence many new ideas were floating around the materials side of the network. For example, it was realised that for the development of the dynamic light-scattering mode (DSM) and electrically controlled birefringence (ECB) devices, materials with larger negative dielectric anisotropies (−Δε) were required, and in addition, methods for material formulations in mixtures were critical to expanding operational temperature ranges. For devices, new methods were being developed for the homeotropic and homogeneous alignment of the liquid crystal mixtures and for bonding devices.
3.5. Recognition
At this juncture, individual desired materials had been prepared in large quantities, they had been used in formulating various eutectic mixtures, which were finding ways into the marketplace for utilisation in flat, thin displays, and so the commercialisation process was under way. The research successes and transformative applications brought recognition in the form of the “
Queen’s Award for Technological Achievement” to various members of the consortia, including RRE, BDH and Hull University. For Hull, it was the first award of its type to a university in the UK.
Figure 7 shows a photograph of Kirton, Hilsum, Raynes (RRE), Gray (Hull), Sturgeon and Pellatt (BDH) at the University of Hull for the Queen’s Award, along with the physical appearance of 0.5 kg of 5CB in a one-litre flask at room temperature. Below is the quotation from the document announcing the Queen’s award for technological achievement.
THE DEPARTMENT OF CHEMISTRY, UNIVERSITY OF HULL
Greetings!
We being cognisant of the outstanding achievement of the said body as manifested in the application of Technology in Our United Kingdom of Great Britain and Northern Ireland, Our Channel Islands and Our Island of Man and being desirous of showing Our Royal Favour do hereby confer upon it
THE QUEEEN’S AWARD FOR TECHNOLOGICAL ACHIEVEMENT
For a period of five years from the twenty-first day of April 1979 until the twentieth day of April 1984 and do hereby give permission for the authorised flag of the said award to be flown during that time by the said body and for the device thereof to be displayed in the manner authorised by Our Warrant of the fifth day of April 1976.
And We do further hereby authorise the said body during the five years of the currency of this Our Award further to use and display in like manner the flags and devices of any current former Awards by it received as prescribed in the eighth Clause of Our said Warrant.
Given at Our Court at St. James’s under Our Royal Sign Manual this twenty-first day of April in the year of Our Lord 1979 in the twenty-eighth year of Our Reign.
By the Sovereign’s Command
The materials effort in the UK continued to focus on the development of new and improved nematogens for applications in displays such as the TNLCD device. Other displays, such as the dynamic scattering device, lost favour, whereas other new concepts vied for interest. Thus, there was a need for materials for the two-frequency switching mode, the supertwist nematic device (STN) and for multiplexed passive and active TFT addressed displays. These new applications required faster and sharper switching modes, bistable operation, better contrast and brightness, a wider viewing angle and lower operating voltages, and materials were sought with appropriate physical properties to meet these demands. In the meantime, the quantities of liquid crystals required grew into the tonnage scale. Applications other than displays also became of interest, for example in telecommunications, sensors, spatial light modulators, beam steering and switches, polymers for adhesives and alignment agents, etc. But for materials research? Gray commented on the discovery of the cyanobiphenyls,
“Many alternatives that did emerge during the next eight years were in fact cyanobiphenyl mimics or look-a-likes. Once chemists understood the strategy we used, cyclohexyl, pyrimidyl, and dioxanyl analogues appeared. The point is however that to be of greatest effect, an invention has to be timely—again bringing in something of the element of luck or chance.” [
48].
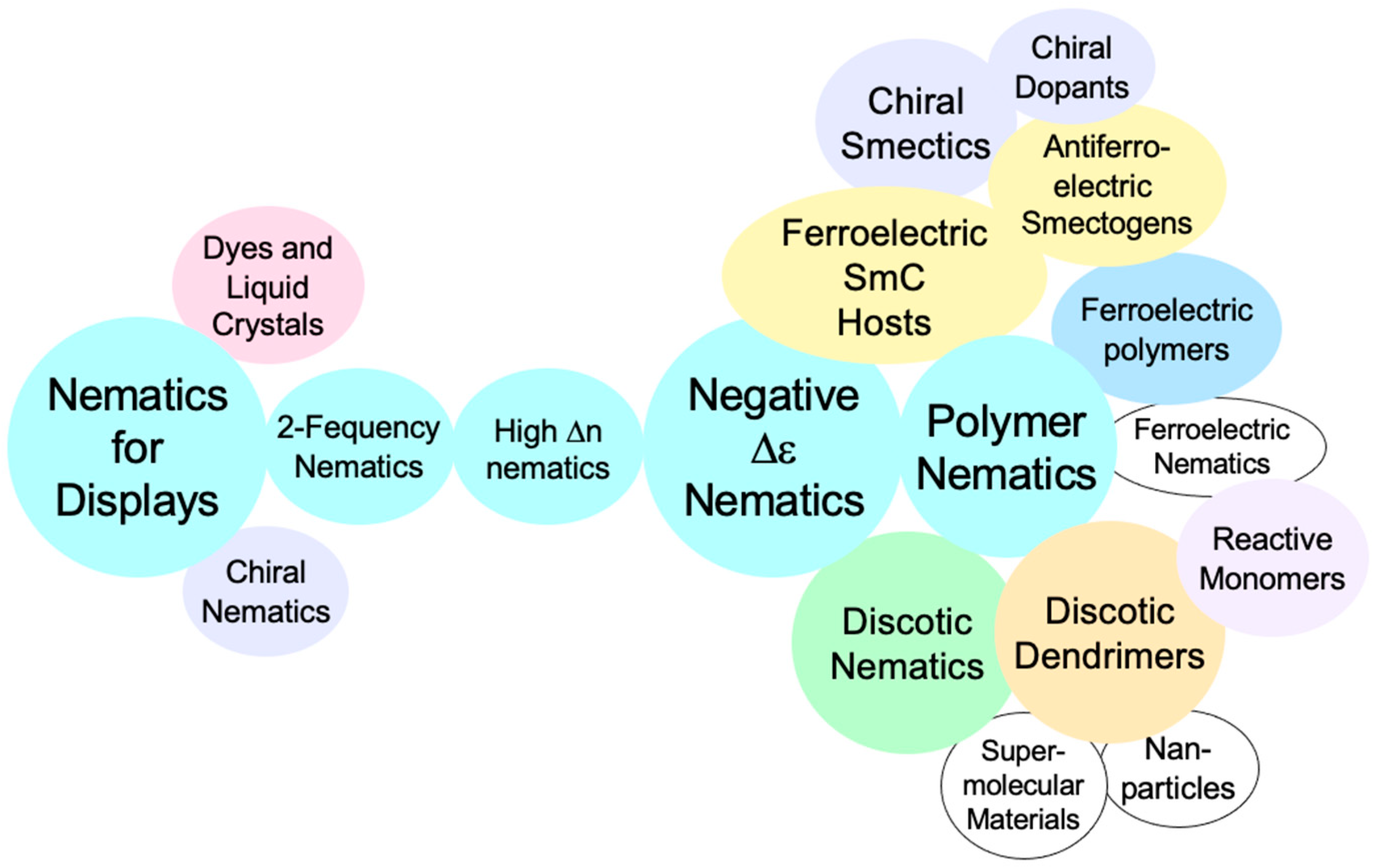
In its academia links to the consortia, Hull was free to research into other areas that that were still in their infancy. Liquid crystal research areas of interest included smectic and discotic phases, new synthetic methodologies which would allow access to nematogenic materials that were hitherto inaccessible, and high and low birefringent materials for display and nondisplay applications. Overarching these topics was also the possibility of introducing chirality either at a molecular level or in a structure of a mesophase. As Gray began this new frontier, he was joined by other academic staff in Hull, including Drs Toyne, Scrowston, Biggs and Lacey, and subsequently, near his retirement, he was joined by his selected successor Goodby, brought back from AT&T Bell Laboratories, USA, whom Gray had arranged to be an industrially funded reader in Hull by Thorn EMI and STL. A year later Goodby became professor and the consortium contract holder in Hull, head of the Liquid Crystals Group and Organic Chemistry, and subsequently head of the Department of Chemistry. The chart shown in
Figure 8 shows how the materials activities expanded rapidly in the new areas laid down by Gray and Goodby, starting with compounds based on cyano-biphenyl where interests lay now in their optical rather than electrical properties. Both high and low birefringencies were of interest, with high values being investigated for non-display applications and low values for thin display devices. Chirality and ferroelectricity were also topical for bistable fast switching devices. Therefore, the interconnected activities expanded rapidly as shown in the chart in
Figure 8, along with an expansion in research applications, reporting, publishing and patenting.
Conversely, the establishments and agencies of the Ministry of Defence (MoD) were reorganised and streamlined in 1991 by creating the Defence Research Agency (DRA), which included RAE and RSRE. In 1995, this metamorphosed to include other agencies in the formation of the Defence Evaluation and Research Agency (DERA). Subsequently in 2001, the MoD split the DERA into two: QinetiQ, which became the sixth largest defence contractor in the UK, and the Defence Science and Technology Laboratory (Dstl). Goodby steered the research activities of the consortium group at Hull through all of these changes until defence needs became redefined.
3.6. Nonlinear Optics
Nonlinear optics was a forthcoming area of interest to those working in liquid crystals in the 1980s, particularly on materials with positive dielectric anisotropies that would have donor–acceptor groups. Second and third order effects were being explored in the examination of the surface organisation of molecules, in beam steering devices and wave guides, light scattering modes, optical processing, optical filters and in various switching effects, e.g., for telecom devices [
60,
61].
Many organic materials that exhibited NLO effects had similar structures to cyanobiphenyls, and therefore it was easy to take a side-step switch to explore new material designs. Often infrared and microwave light were the target for novel device concepts, particularly in the areas of optical-switching, frequency-doubling and frequency-tripling applications. Often this meant having control over the birefringence of a mesophase; for example, high birefringence was of interest in microwave applications (Δn ~0.3 to 0.5), whereas low birefringence was of more interest in thin displays, such as those found in surface-stabilised ferroelectric devices (SSFLCDs, Δn ~ 0.05 to 0.2).
There were also differences between material systems possessing rod-like shaped molecules and those having disc-like molecules. With rod-like molecules, materials could be designed to have donor and acceptor groups, and hence could exhibit second and third order effects, whereas the symmetry of disc-like molecules meant that such materials favoured third-order properties. Thus, the Universities of Leeds and UEA joined the materials consortia collaborating with Hull on certain aspects of synthetic methods.
Knowing how to control the magnitude of the birefringence became one of developing structure–property correlations. Raising the birefringence (Δn) became a task of basically increasing the number of delocalised π-electrons, and relative polarisability (Δα), within a molecular architecture without losing mesophase properties or other required materials properties, such as colour in dyed systems. Conversely, lowering the birefringence became a task of replacing π-bonds with σ-bonds. In
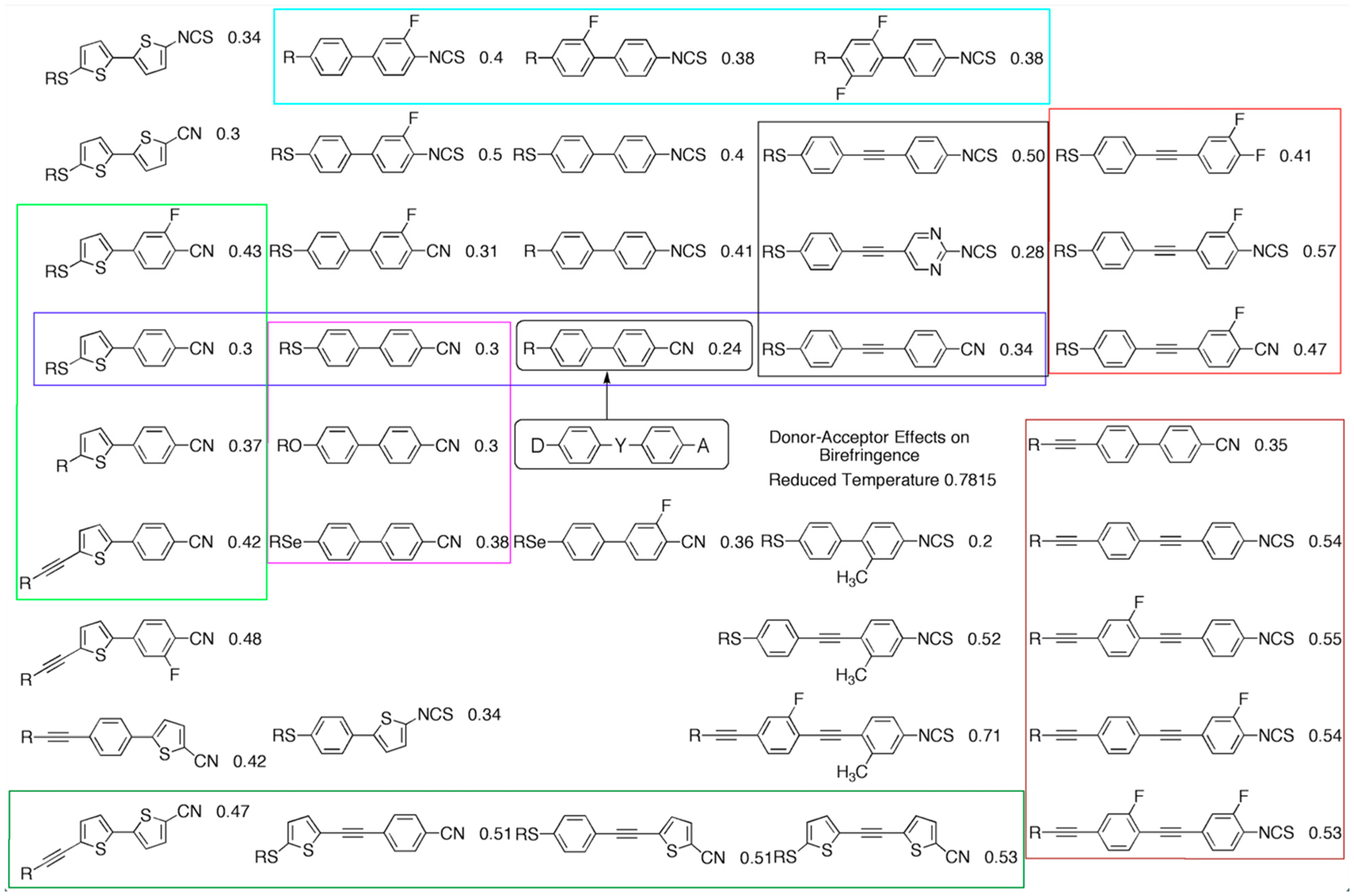
Figure 9, a chart is shown for pentyl-substituted molecules, with C
5H
11 denoted as R. Starting at the left-hand column, the structures of the molecules are essentially the same with the only major change to the acceptor groups on the right-hand side, i.e., CN to NCS and the addition of F; the terminal polar groups increase the longitudinal dipole and so the electrons are moving left to right and therefore the birefringence increases, which is mirrored by an increase in the polarisability, whereas the values for the order parameters remain roughly the same. For the centre column, the argument is slightly different as this time, the left-hand sides of the molecules are donating electrons into the central core, but the result is the same: the birefringence increases down the column but not to the extent that it does in the first column. In the right-hand column, the central core of the molecules is increased, extending the extent of the delocalised electrons. Coupled with this are changes to the terminal polar groups, but for the molecules, the longitudinal dipoles increase, thereby the birefringence also increases. For the three columns, the order parameter (S) does not appear to play a major role in determining birefringence, but the linked polarisability and the polarity have a degree of balance in determining the birefringence.
Such linear correlations shown in
Figure 9 can be expanded into a larger number with some crossovers between the correlations, thereby creating arrays of linkages between the physical properties of the materials. For instance,
Figure 10 shows how a number of property–structure activities can be linked together to create a two-dimensional landscape for materials that have donor–acceptor molecular structures. The arrays allow for projection beyond those materials that had been synthesised, thereby leading to the prospect of target selection at a distance away from the original development strands and improving the possibility of discovery and invention.
Acceptor groups for materials that might be predicted to exhibit mesomorphism include NO
2, CN and NCS, whereas donor groups include R, OR, SR, NH, etc. In addition, the length for a rod-like material affects the charge separation between the acceptor and donor (A and D) groups, and thereby the polarisability and dipole. The larger the longitudinal dipole, the greater the birefringence. However, the longer the molecule is on average, the greater the effect is on the melting point, with the bigger the molecule, the higher the melting point. Unfortunately, for certain applications a lower melting point was desired. For disc-like molecules, due to their symmetry, usually there are no donor and acceptor groups, and so the strength of any nonlinear effect is dependent on the number of π-electrons and the polarisability. Overall, numerous materials were prepared, and many examples of families of materials, their property–structure correlations, birefringencies (n
| |, n
⊥ and Δn), polarisabilities (Δα) and order parameters (S) were reported in the literature [
62,
63,
64,
65,
66,
67,
68,
69].
3.7. Nematogens and Smectogens with Negative Dielectric Anisotropies
This particularly important research programme was based on the Nobel Prize awarded to Heck, Suzuki and Miyaura in 2010, for innovations in synthetic chemistry utilising palladium-based coupling reactions [
70,
71,
72]. In the Suzuki variation, a double-bond-containing molecule is replaced by an organoboron substrate, with palladium acting as a catalyst in a cross-coupling procedure between two different substrates, one featuring the organoboron moiety and the other a good leaving group (Br, I, triflate), thereby it was possible to link two different phenyl units together to give asymmetrically substituted biphenyl and terphenyl products. Studying the publication by Suzuki and Miyaura in the late 1980s, Ken Toyne thought that the synthetic technique could be employed in the synthesis of laterally substituted fluorinated terphenyls, which might be used to make materials with negative dielectric anisotropies. Similar thoughts were ongoing at E. Merck (now owner of BDH), and so the two teams met through the consortium to discuss relative activities. At the beginning [
73,
74], both groups worked generally on the synthesis of a variety materials, but ultimately E. Merck took the lead in research on nematics for large area displays, whereas Hull tended to focus on smectics for small area microdisplays, and for larger area bistable multiplexed devices.
For nematics with negative dielectric anisotropy, it was already known that it was possible to create electrically controlled birefringence (ECB) displays. In the off state, the molecules are aligned orthogonally with respect to the substrates of a device. Upon applying an electric field, the molecules tilt away from the orthogonal state to give a bright on state. Such devices were being examined within the network in the early 1970s. This so-called vertically aligned configuration was much later called VA, after vertical alignment, and in the initial devices, switching times of around 25 ms [
75] were possible. It was also found that VA displays could provide a high brightness with good viewing angles. The reasonable response times and the possibility of creating controlled multidomains that could give symmetrical and wide-angle viewing meant that this mode might be adapted for TV applications.
For VAN/ECB-LCD modes, materials [
76] were needed that possessed large dipoles located across the molecular long axes, so that the materials would have a large negative dielectric anisotropy. In addition, they were required to have suitable birefringencies relative to the spacing thickness of the device, and with a relatively low viscosity for fast response times.
In comparison, materials with positive dielectric anisotropies possess longitudinal dipoles, and as such molecular rotation around the long axis, which is fast (1011 s−1) and has less effect than for materials with negative dielectric anisotropy, where the lateral dipole is more affected by the slower rotation of 106 s-1. In addition, the incorporation of multiple polar groups positioned along the long axis was not easy to achieve synthetically or to have them all point in the same direction across the molecule, unless they were fixed to the same phenyl ring. Furthermore, the lateral moiety that is polar would also have to be relatively small in order to retain mesomerism and at the same time not to increase viscosity. Consequently, fluorine was preferred over moieties such as nitrile, which has the adverse effect of raising viscosity due steric hinderance. So, as with nematogens of positive dielectric anisotropy, fluoro-substitution held the key to developing practical device materials for negative dielectric anisotropy. Achieving all of these goals was taking molecular engineering to a new level, which was only possible at the time via use of the Suzuki–Miyaura coupling methodologies in synthetic pathways.
Deploying fluorine instead of nitrile in the material design did have some drawbacks in terms dielectric anisotropy as shown in
Figure 11. Some examples of nematogens that possess two lateral polar groups (F and CN) fixed to the same side of a phenyl ring are shown along with the relative dielectric anisotropies. The materials have much larger negative dielectric anisotropies as one might expect. However, materials designed in this way, with two substituents on the same ring, have additive effects from the polarities of both polar groups. Comparatively, two fluorine atoms attached to adjacent positions on an aromatic ring, when combined, have a polarity a little bit less than a nitrile unit, but at the same time imparting less towards the viscosity, as shown in the figure. Moreover, additional fluoro-substituents can be added to give di-, tri, and tetra-analogues, with little change to the dielectricanisotropy. In addition, the conversion of one of the aromatic rings to cyclohexane can generate important nematogens that are suitable for displays.
Interestingly, the mesomorphic behaviour is determined by the lengths of the external aliphatic chains, and whether or not they are alkoxy or alkyl, as shown in
Table 2. Similarly, the location of the phenyl ring carrying lateral polar group(s) can be used to determine mesomorphic behaviour in terms of transition temperatures, phase sequences, dielectric anisotropies and viscosities, and to what devices the materials are best suited for. Apart from nematic devices, the materials as shown in
Table 2 also exhibit smectic C phases, as the aliphatic content is extensive. If the materials shown are substituted with a chiral aliphatic chain instead of a normal chain, the smectic C phase will also become chiral, or alternatively, if they are doped with a chiral material, then the mixture will also exhibit chirality. In both cases if this results in a chiral smectic C phase being formed, then it will exhibit ferroelectric properties. At this point a divergence had occurred in the research paths of Merck and the consortium, and Hull followed a path along research into the syntheses and various properties of ferroelectric materials.
In the design of smectogens that will either act as hosts or be chiral, it is important to understand the elements that will drive the molecules to tilt in the smectic state, and thereby to form synclinic (ferroelectric) smectic C or anticlinic (antiferroelectric) smectic C
A phases. The generation of the smectic C phase has been thought to be related to what were termed “outboard” terminal dipoles, or the location of a polar atom between a terminal aliphatic chain and a rigid core unit, as shown by the materials in
Figure 12. The presence of terminal polar groups was theorised by McMillan [
78] to reduce the molecular rotation around the long molecular axis, thereby allowing for a molecular torque to occur in the planes of the layers in the smectic phase, resulting in the generation of a molecular tilt and hence the formation of a potential smectic C phase. This result is also obtained for the fluorinated terphenyls in the figure, whereby oxygen being located at one end or both ends of the aromatic core unit can be used to control phase sequences, transition temperatures and other related properties.
As shown in
Figure 12, the upper two materials (a) and (b), with a single alkoxy group, show the same phase sequences and similar transition temperatures, whereas material (c) with no alkoxy groups still exhibits a smectic C phase, but at a much lower temperature. This indicates that the difluoro-substituted unit is still contributing to the induction of the molecular tilt. With respect to the upper two materials, the dielectric anisotropies are different, with the lower material (b) having the higher value, which is associated with the conjugation between the oxygen and fluorine atoms, whereas this is not the case for material (a) where the polarisation through conjugation is weak. For compounds (a) and (c), the dielectric anisotropies are similar, indicating that the major contribution to the formation of the smectic C phase is through the molecular packing, but the lower transition temperatures for (c) indicate that the contribution is less than for (a), which has a larger outboard dipole associated with oxygen. Consider now the pair of compounds (c) and (d). They are analogues of one another with the two terminal chains swapped around. They have very similar transition temperatures indicating that the positions of the alkyl chains do not greatly affect mesomorphic behaviour. Compound (e), having two terminal outboard dipoles, has the classical structure for a smectic C material. It has the highest thermal stability for the smectic C phase. Overall, the least polar materials (c) and (d) have the lowest viscosity, the lowest tilt angle, whereas (e) has the highest tilt and the highest viscosity. Therefore, formulated mixtures need to have a balance of components to give optimal properties.
Although we have shown studies with difluoro-substitution on one phenyl ring, as noted, the mesophases properties depend on which ring in terphenyl, for example, is substituted. It is possible to have fluorination on adjacent rings and also have more than two fluorine atoms attached to the aromatic core, see
Figure 11. For terphenyls, this gives a plethora of possible substitutions and associated isomers that may be prepared, and which might suit certain mesophase types and applications. Moreover, although configurational isomers can be fixed by synthesis, conformational variants have not been fully explored, in particular in relation to rotations about the phenyl–phenyl bonds in terphenyls which cause interannular twisting. The possibilities of examining conformational interactions have been examined through dielectric studies of both anisotropy and biaxiality and theoretically through computer simulations [
79]. However, to this date only a small area of the isomer landscape has been accessed; nevertheless, such materials have found practical and commercial uses in displays for example in VAN-, SSFLCD- and τ
vminLCD-mode devices. In the development of materials created via coupling reactions, Kingston Chemicals Ltd. was created in 2000 to serve small technology companies and universities with advanced organic materials, mostly based on fluorinated compounds.
3.8. Gels and Polymers
Apart from the design, synthesis and applications in low-molar-mass rod-like systems, the use of coupling reactions was important in the creation of discotics, gels, oligomers, dendrimers and polymers. These topics covered various applications in areas such as adhesives, ferroelectrics, pyroelectrics, filters, coatings, alignment and high yield strength materials. The basic science and topics are wide ranging and so we cover only a few examples here.
Crossing over from low-molar-mass materials to polymers we enter the area of gels based on the research of Hickmet [
80,
81]. We prepared gels composed of a cross-linking monomer unit and a mixture of mesomorphic materials. In the design of the formulation, the compounds were selected to be of a similar size and chemical nature and polymerised to give a desired gel network [
82,
83,
84].
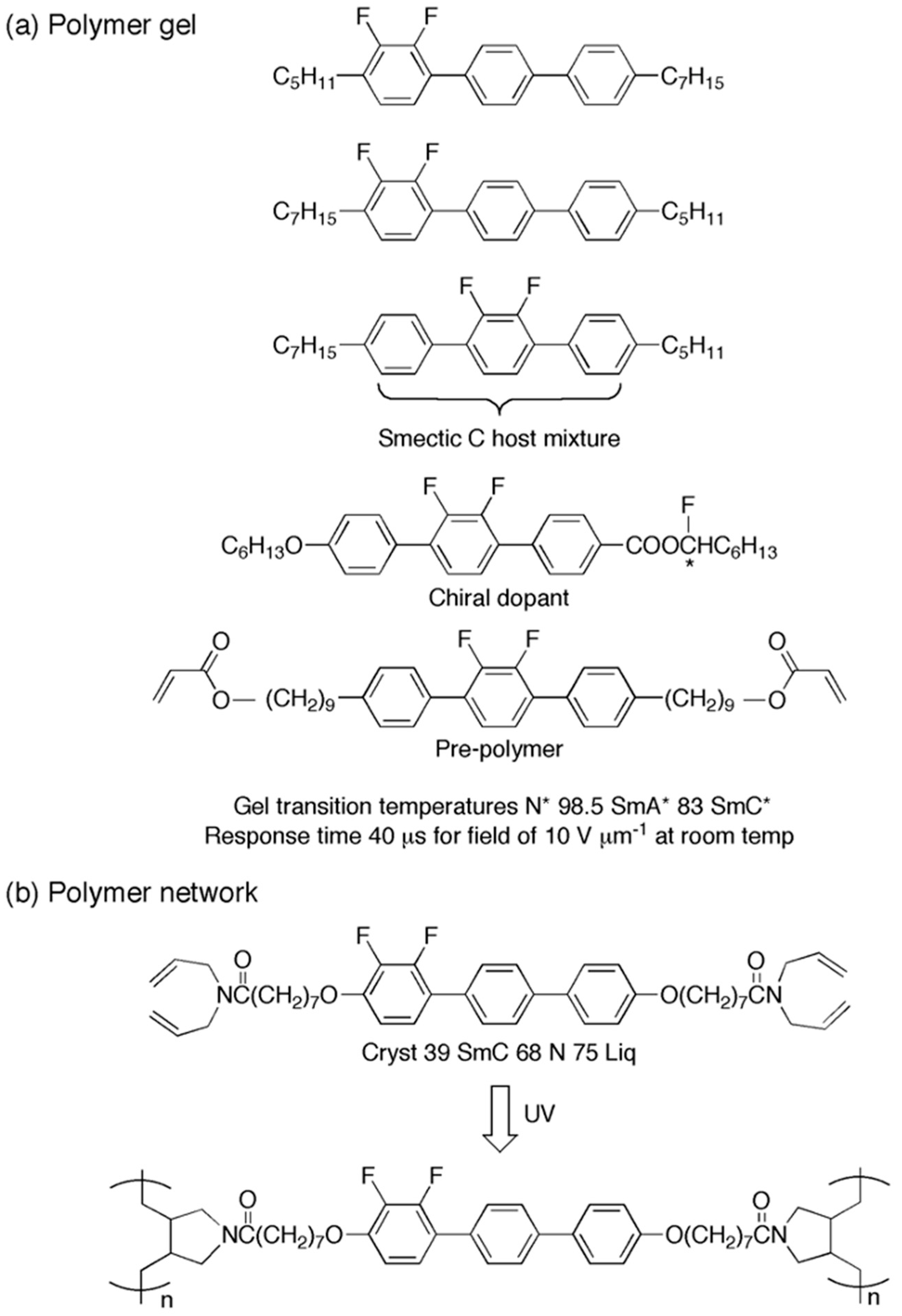
Figure 13a shows a typical formulation for a gelating mixture of liquid crystals based on an achiral host mixture of difluoroterphenyls, a chiral dopant to impart ferroelectricity, and 10 wt % of a cross-linking monomer to produce a gel [
85]. When subjected to photopolymerisation, a gel existed in a nematic phase above 98.5 °C; on cooling, a smectic A* phase was formed followed by a ferroelectric smectic C* phase at 83.0 °C. Of course, the gel did not crystallise at low temperatures below −20 °C before starting to show semblances of glassifying. At room temperature, the response time in the ferroelectric phase was just 40 ms in an electric field of 10 V mm
−1, i.e., a response time much faster than for the host/dopant mixture without polymerisation. Therefore, we move from conventional low-molar-mass materials to gelated forms that can be manipulated in many different ways.
It is of course possible to take monomers such as those employed in making gels and to polymerise them on their own in order to form networks. In the example below, shown in
Figure 13b, we use a novel prepolymeric system based on diallylamine to create a network. The mesogenic unit is again based on a difluoroterphenyl and exhibits smectic C and nematic phases in which photopolymerisation can take place. Preorganisation of the monomer species can retain to some degree its organisation in the photopolymerised nematic or smectic C phases. Such organised networks are of use in preparing polarisers, colour filters, and optical compensators. Without the use of a mesogenic unit between the diallylamine groups, materials can be created that are of use in coatings, adhesives, ionic liquids, etc. Two spin-off companies were formed by DERA to exploit various possible applications, one called NPS in 2002 and the other IPS in 2005.
3.9. Nematic Disc-like Materials
In the late 1970s, Hull became involved with the synthesis of disc-shaped molecules based on hexa-substituted benzenes and triphenylenes. This area of research became revisited at a similar time as research that was ongoing on terphenyls because there were some analogies in the synthetic methodologies. Research thus went into the synthesis of bowl-like molecules and oblong-shaped coordination complexes, for columnar ferroelectricity and nematic biaxiality, respectfully. However, by the mid-1990s, research had switched to a search for nematic discotic triphenylenes possessing negative birefringencies and potentially negative dielectric properties with the possibility of searching for biaxial nematic phases.
The first method used in 1978 to produce the 2,3,6,7,10,11-hexamethoxytriphenylene involved the oxidative trimerisation of veratrole with a third of an amount of chloranil. The process was tedious and produced low yields. A second method involved the cyclisation of veratrole in the presence of iron (III) chloride, which also gave low yields. However, synthetic refinements learned through collaborations with UEA in the consortium gave almost quantitative yields of the hexa-methoxytriphenylene. Demethylation with boron tribromide gave suitable yields of 2,3,6,7,10,11-hexahydroxytriphenylene, which could be derivatised to give discotic liquid crystals. Some of the more important materials that were prepared are shown in
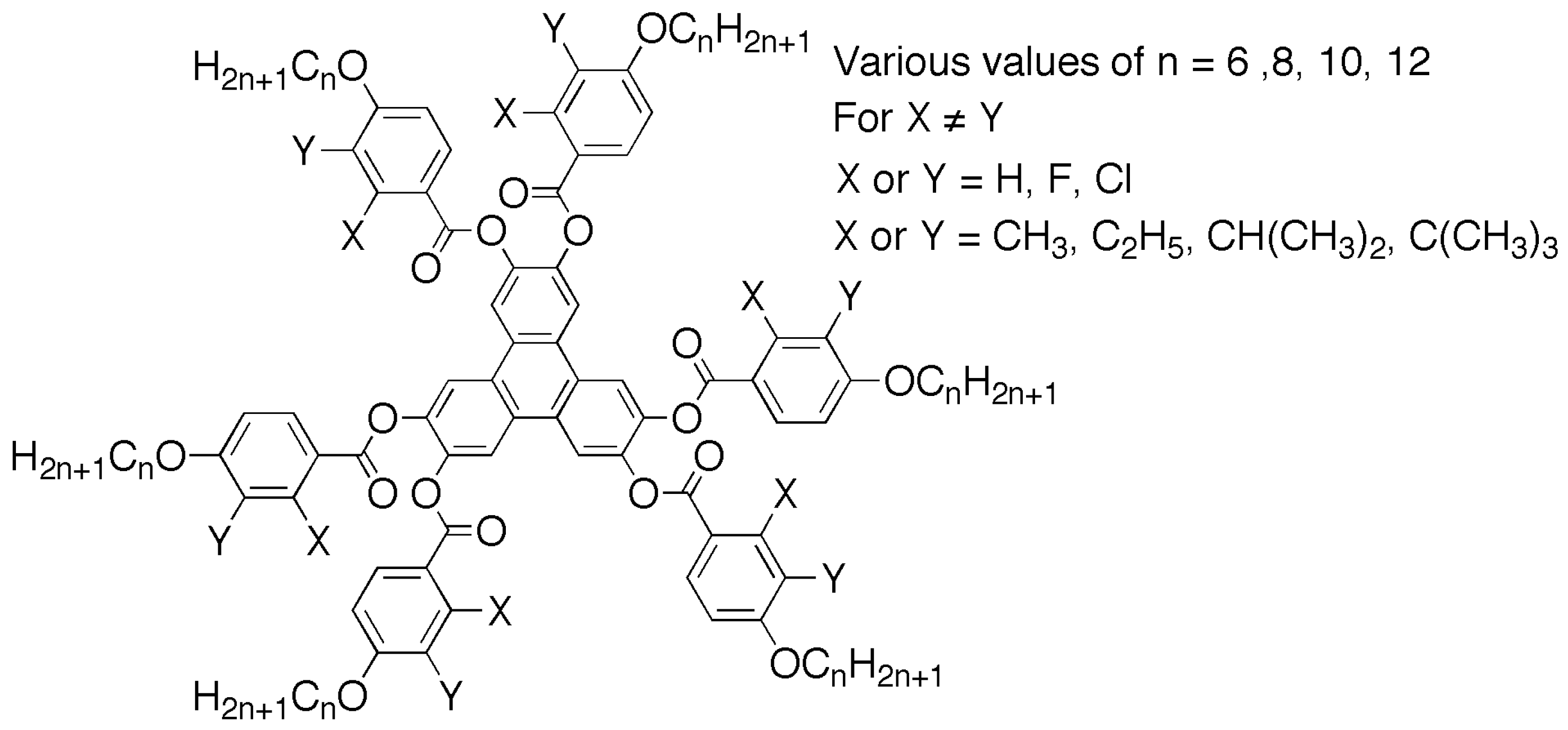
Figure 14. These materials are the hexa-benzoate derivatives of hexa-hydroxytriphenylene, where the benzoate esters could be designed to incorporate lateral groups, which might be polar (halogens) or apolar (aliphatic chains).
Stacking of triphenylene units together is more likely to form columns of molecules, and therefore, the incorporation of benzoates seemed a likely way to generate discotic nematic phases. To achieve this, substitution that prises apart the molecular discs was postulated, for which a wide variety of substitutions were tested, as shown in
Figure 15. It was found that if the substituents in the benzoate were too large (C
2H
5, etc.) or too polar (F, etc.), nematic phases were not achieved. A methyl substituent, in the two or three positions of the outer benzoate moieties, were the only material types that would support nematic phase formation.
Figure 15 shows the aromatic region of benzoate-substituted triphenylenes. The left-hand model has no substituent in the external benzoate ring, whereas the right-hand model has a large tert-butyl substituent; in this way, the separation of adjacent triphenylene rings can be illustrated. The large tert-butyl moiety sterically hinders the internal packing of the discs and twists the benzoate rings out of the plane of the triphenylene core, the benzoate rings then prising apart the molecules, thereby preventing columnar formation (and in this particular case actually preventing nematic phases forming). It appears that the methyl substituent is the right size to partially prise the discs apart allowing for the formation of the nematic phase.
Table 3 shows the structures of triphenylene-2,3,6,7,10,11-hexahexayl hexakis(4-alkoxy-2 or 3-methylbenzoate(s) where the methyl group is either pointing inwards towards the triphenylene core (inner) or away from the core (outer), where the exterior alkoxy chain is also varied in length. All of the examples shown exhibit discotic nematic phases, with melting points around 100 °C and clearing points for the most part beginning around 200 °C. These temperatures are much lower than those of the nonmethyl-substituted analogues, which have greater tendencies in also forming columnar mesophases. These property–structure correlations show that lateral substitution in external benzoate rings is of practical use in the design of nematic-discotic materials for a variety of applications [
87,
88,
89,
90].
The control over the synthetic pathways developed between Hull, UEA and Leeds Universities for such materials, and the high yields which could be achieved for their production, meant that they would be of practical use. As with the ferroelectric materials described earlier, it was possible to create polymeric networks (EU Orchis network 1989), with interest being in various areas of molecular electronics. In other areas of R&D their properties of negative birefringence of nematic discotics were of interest in optical films, in particular for applications in optical compensation films for various devices. The beautiful work at Fuji film on these films and related materials resulted in the invention and development of films for wide viewing angles in nematic displays [
91].
3.10. Chiral Materials—Liquid Crystals and Dopants
Various forms of chirality permeate throughout liquid crystals from molecules [
92] to mesophase structures [
93]. Knowing the connectivity and relationships between the left-hand and the right-hand can be invariably important. An amusing story about this was told by George Gray:
“BDH Ltd. sold commercially both Hull’s right- and left-handed compounds. Customers could choose which to use. One customer decided to do better than everyone else and to use some of each additive. Of course, the two cancelled out and the effect was zero- and he complained most bitterly that our products were no good. He had to be gently educated”.
Gray’s story is amusing given that the first chiral cyanobiphenyl was prepared in 1973, and at that time there was no relationship between stereogenic architecture and mesophase macrostructure other than the physical properties of enantiomers would be opposite to one another. However, for mesomorphic materials, there was a need for the purposes of mixture formulation to either reduce helical pitch length or expand it. In 1976, relationships between stereochemistry, molecular structure and helical twist direction were developed for materials with single stereogenic centres, with the following relationships:
where R and S are the Chan, Ingold, Prelog systematic labelling systems for asymmetric centres, o and e are the parities (odd and even) for the number of atoms the centre is removed from the central rigid molecular core, and d and l (dextro and laevo) being related to the optical rotation direction for the helical structure in a chiral nematic phase. These Gray and McDonnell rules [
55] were applied for formulations that did not necessarily have single enantiomers in their mixtures [
94].
For smectic C ferroelectric liquid crystals, they too are dependent on the relationships between stereochemistry and broken symmetries on the macroscale [
95], but in this case extra relationships are needed for the development of formulations. These include the direction of the spontaneous polarisation (Ps+ and Ps−) and the direction of the dipole at the stereogenic centre (+I, −I), thereby giving us similar rules to those of Gray and McDonnell, but with two extra terms as shown below [
96].
Many property–structure correlations were drawn up for ferroelectric smectic liquid crystals that related helical twist and spontaneous polarisation directions to molecular stereochemistry in an attempt to formulate mixtures where the values of the spontaneous polarisation and the helical pitch length were maximised. For devices, this optimised situation meant that helicity did not affect alignment, and a high polarisation reduced the switching voltage. A simple correlation between molecular architecture and properties is shown in
Figure 16, which can be applied to some degree to nematogens and smectogens that have single stereogenic centres.
3.11. Serendipity—Polar Nematics
As the consortium network was nearing to its end, York (our new location) was asked to participate in two new projects; one on low birefringent ferroelectric materials that operated at ambient temperatures and another on extremely polar nematics. The first, we have somewhat discussed earlier on in this article, and as usual, to lower the birefringence required the inclusion of alicyclic ring systems, but the downside to this approach was tilted smectic phases were not usually favoured. Using an end-group design and new synthetic methodologies, this was achieved.
For the second project, achieving extremely high polarities meant using singular or multiples of strongly polar substituents. Discussions at the start of the programme focused on the use of nitro units as the polar moieties, see
Figure 17. Therefore, to generate a high polarity meant having as high a proportion of nitro units as possible in the molecular structure of the target. A small molecule with a single nitro unit that mimicked cyanobiphenyls was considered, but it was already known from Gray’s results, shown in
Table 1, that the nitro analogues were poor nematogens. However, from Gray’s 1962 textbook [
42], it was also known that nitro-substituted methoxyterphenyls were mesomorphic. Furthermore, having previously synthesised re-entrant nematic materials, such as the DBnNO
2’s, [
97] based on the research of Hardouin et al. [
98,
99,
100,
101,
102], we additionally knew that nitro-substituted three-ring reversed and normal esters would support nematic phase formation. However, the problem with this approach was that the melting points were expected to be high and well above room temperature.
To lower the melting points, we had to take a number of well-known pathways in molecular design by using dimers, trimers, tetramers, etc., [
103]. Using property–structure correlations, we knew that the incorporation of lateral aliphatic chains in three- and four-ring rod-like molecules would not only depress the melting point, but would also depress smectic mesophase formation, thereby favouring nematics [
104]. By joining a lateral aliphatic chain of varying lengths to a similar rod-like molecule in order to create a lateral supermolecule, we could depress crystallisation to temperatures below 0 °C. For example, by joining two laterally substituted dimers together to give a tetramer, we had also shown that we could produce materials that were nematic with useful transition temperatures via having differing arms on dimers and tetramers [
105,
106]. For all of the materials types, there was also the possibility of improving on the number density of nitro groups. There was one problem with all of these concepts, and that was high viscosities and that switching would be slow, but this was not seen as a problem for the potential applications. Thus, a strategy was put in place for the start of the project (see
Figure 17), but it was not long before en-route novelty appeared in the presence of an extra nematic phase for compounds RM230 and RM734.
Compounds RM230 and RM734 were materials that were made during the exploratory research and which were originally thought to exhibit nematic re-entrancy [
107] after formation of a smectic C phase from a nematic phase on cooling from the liquid. It was only later that it was concluded that the materials did not have smectic C phases, and that we were on the cusp of a direct nematic-to-nematic phase transition [
108,
109], with the lower temperature nematic phase later classified as a new splay nematic phase [
110].
In the original article concerning compound RM734 [
108], the electrical field studies, using a triangular waveform and a frequency in the range of 0.1–20 Hz (ACLT property tester), gave the dielectric anisotropy in the upper temperature nematic phase to be approximately 8.5, and 6.2 in the lower phase, with Kirkwood factors of g ~0.262 and ~0.117, respectively, indicating that there was a greater antiparallel pairing in the lower temperature nematic phase. In addition, the higher temperature nematic phase also had a reasonable degree of pairing. Electrical field studies were further conducted by Clark et al. [
111] and they concluded that RM734 exhibited the first ferroelectric nematic phase. In the Physics World magazine, the discovery of a ferroelectric nematic phase was listed among the “Breakthroughs of the Year” finalists for 2020, (10th Dec issue, see citation below). At a similar time to our report, Nishikawa et al. [
112] also saw such behaviour in completely different materials. In these days of fast-moving discoveries, Merck reported on a material that exhibited a ferroelectric nematic phase on cooling from the liquid state and was still in that phase at room temperature [
113]. Thus, we seemed to be coming around full circle from the 1980s–1990s when the work at Hull focused on ferroelectrics for microdisplays.
First observation of a ferroelectric nematic liquid crystal (From Physics Today)
To Noel Clark and colleagues at the University of Colorado Boulder and the University of Utah in the US, for observing a ferroelectric nematic phase of matter in liquid crystals more than 100 years after it was predicted to exist. In this phase, all the molecules within specific patches, or domains, of the liquid crystal point in roughly the same direction—a phenomenon known as polar ordering that was first hypothesized by Peter Debye and Max Born back in the 1910s. Clark and colleagues found that when they applied a weak electric field to an organic molecule known as RM734, a striking palette of colours developed towards the edges of the cell containing the liquid crystal. In this phase, RM734 proved far more responsive to electric fields than traditional nematic liquid crystals. Although further work is required to identify materials that display the phenomenon at room temperatures, ferroelectric nematics could find applications in areas from new types of display screens to reimagined computer memory.
As George Gray would say “For the greatest effect, an invention has to be timely—again bringing in something of the element of luck or change”.