Experimental and Theoretical Studies of Carboxylic Polymers with Low Molecular Weight as Inhibitors for Calcium Carbonate Scale
Abstract
1. Introduction
2. Materials and Instruments
2.1. Materials and Instruments
2.2. Synthesis of Polyacrylic Acid
2.3. Static Experiment
2.4. Dispersion Experiment
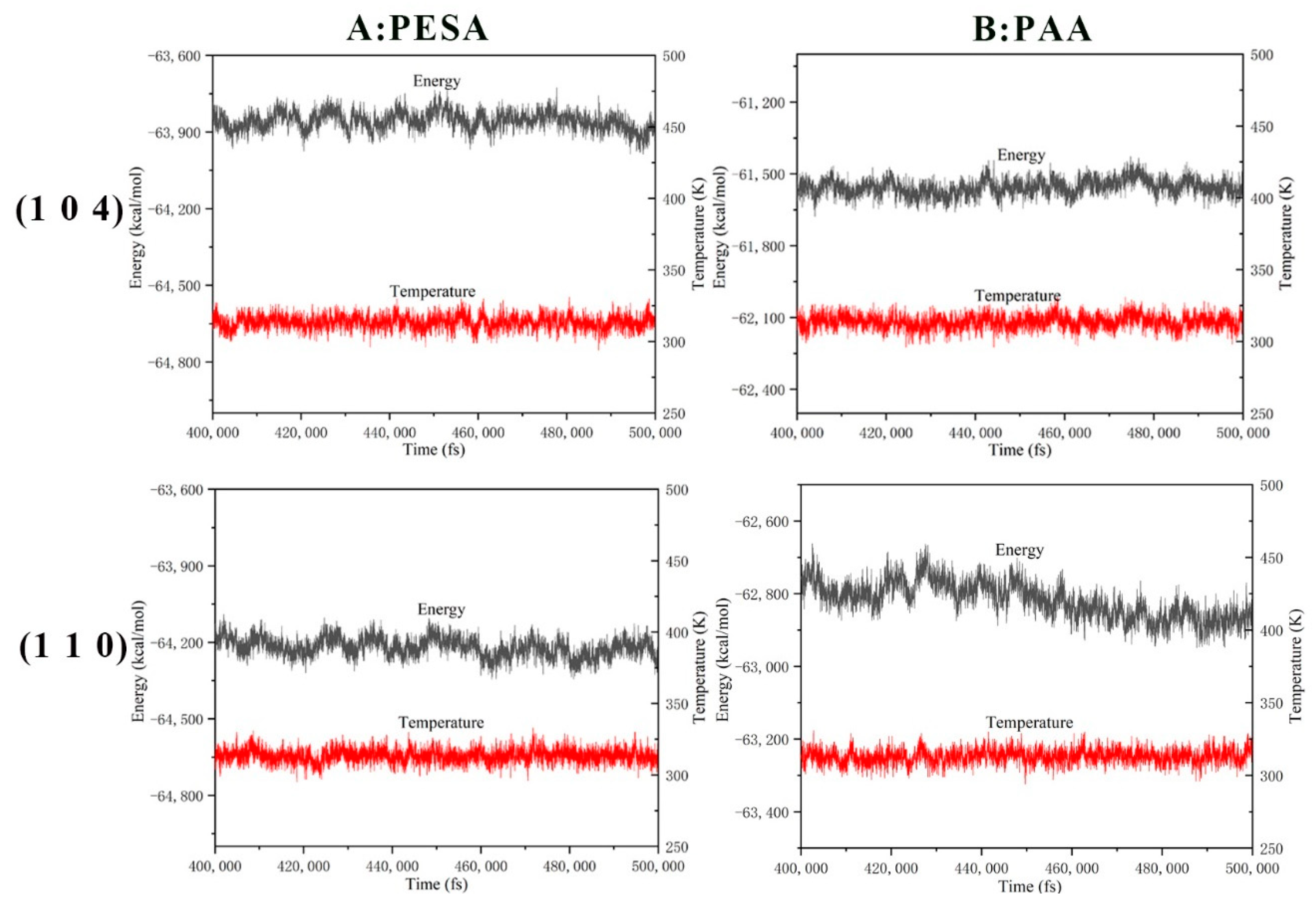
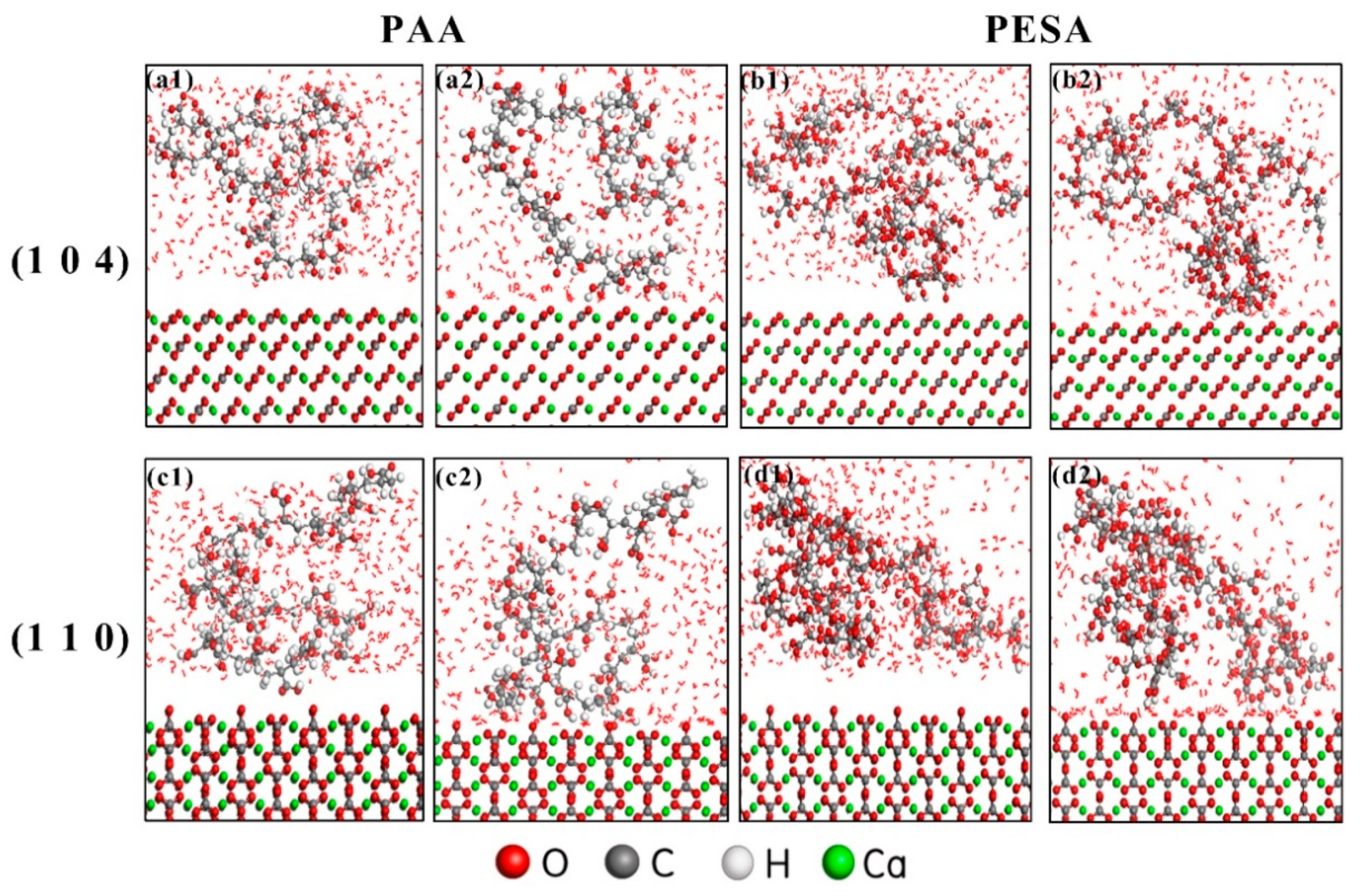
2.5. Molecular Dynamics Simulation
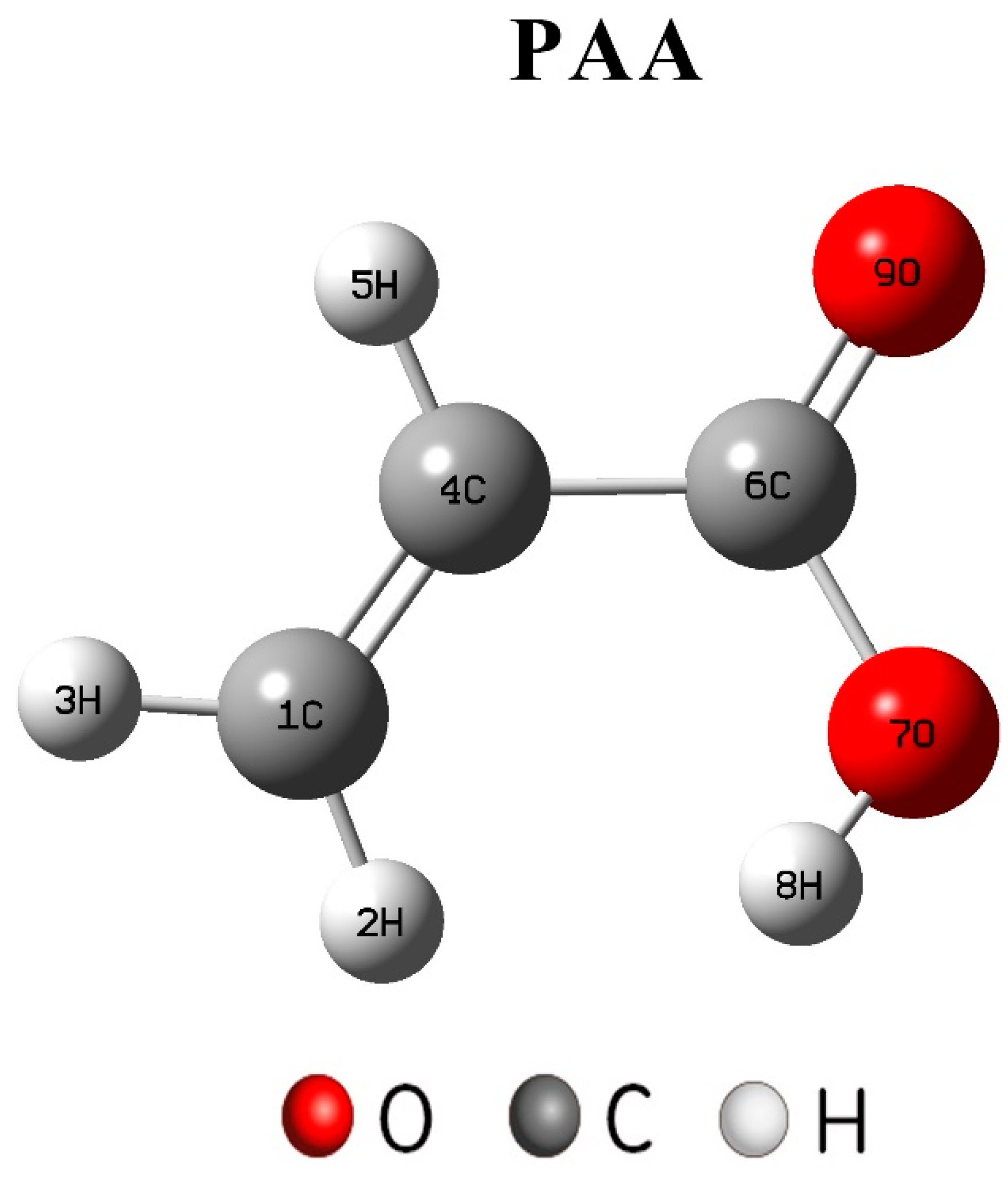
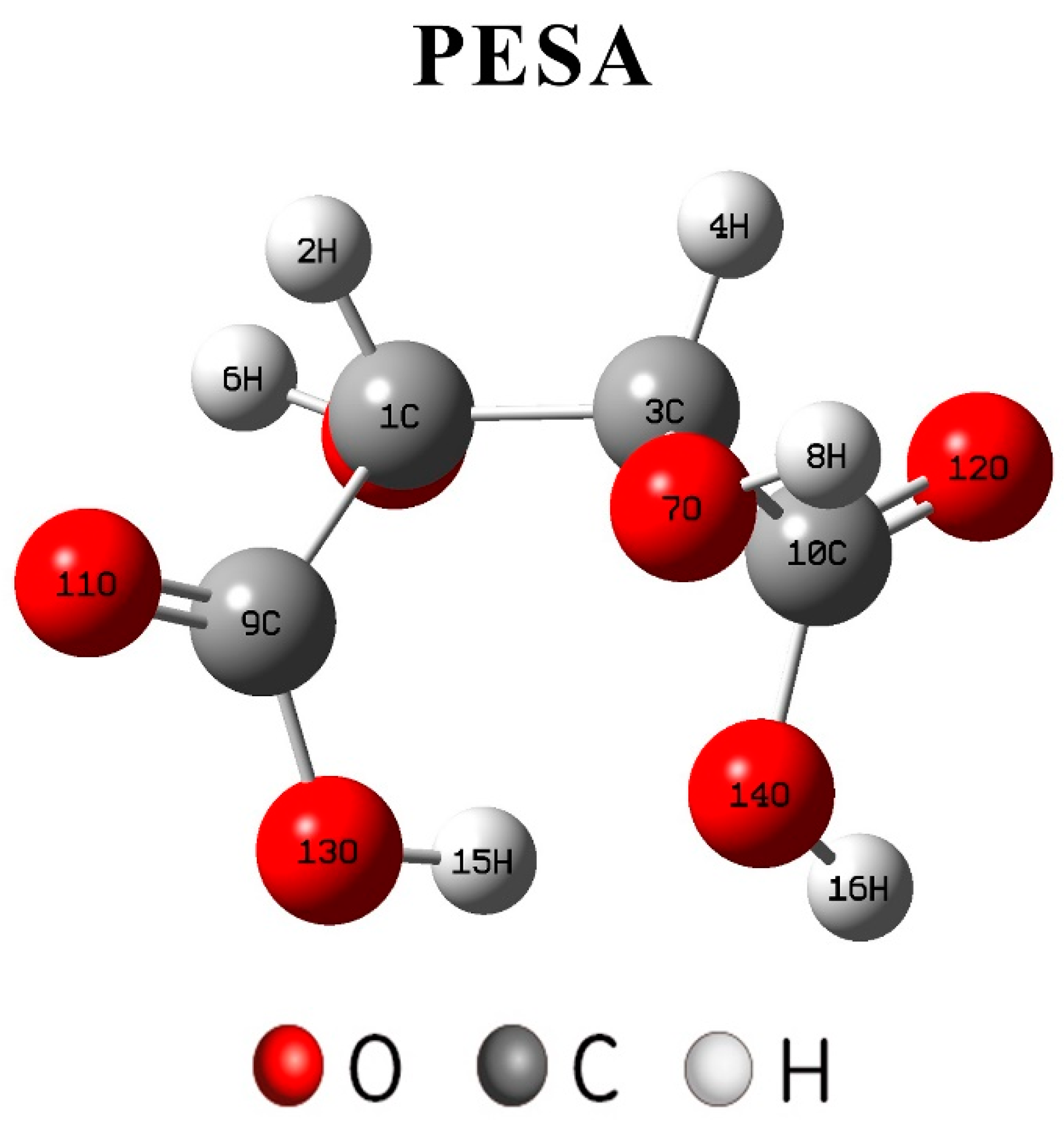
2.6. Quantum Chemical Method
3. Results and Discussion
3.1. Molecular Weight
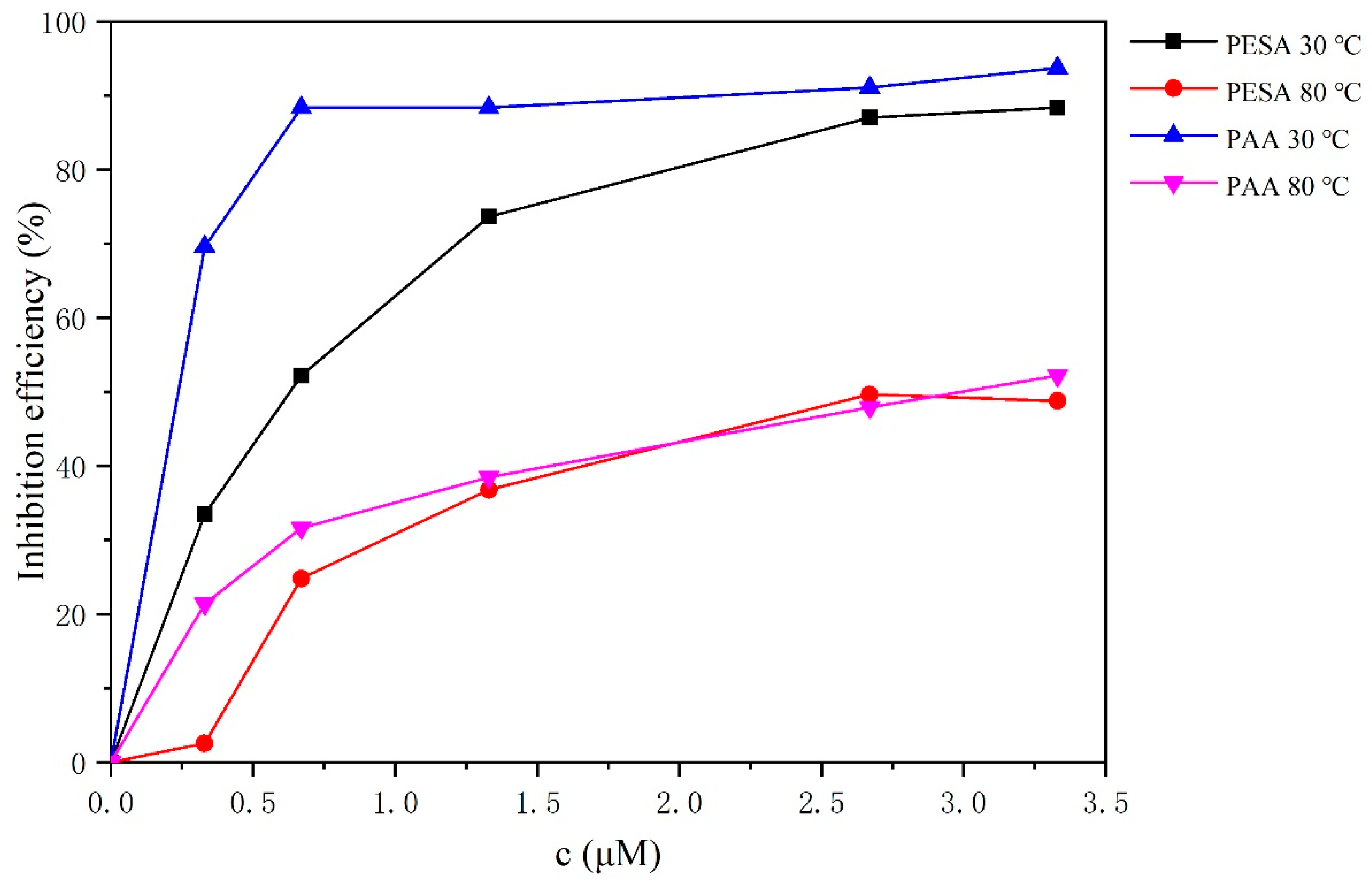
3.2. Static Scale Inhibition Analysis
3.3. Analysis of Theoretical Calculations
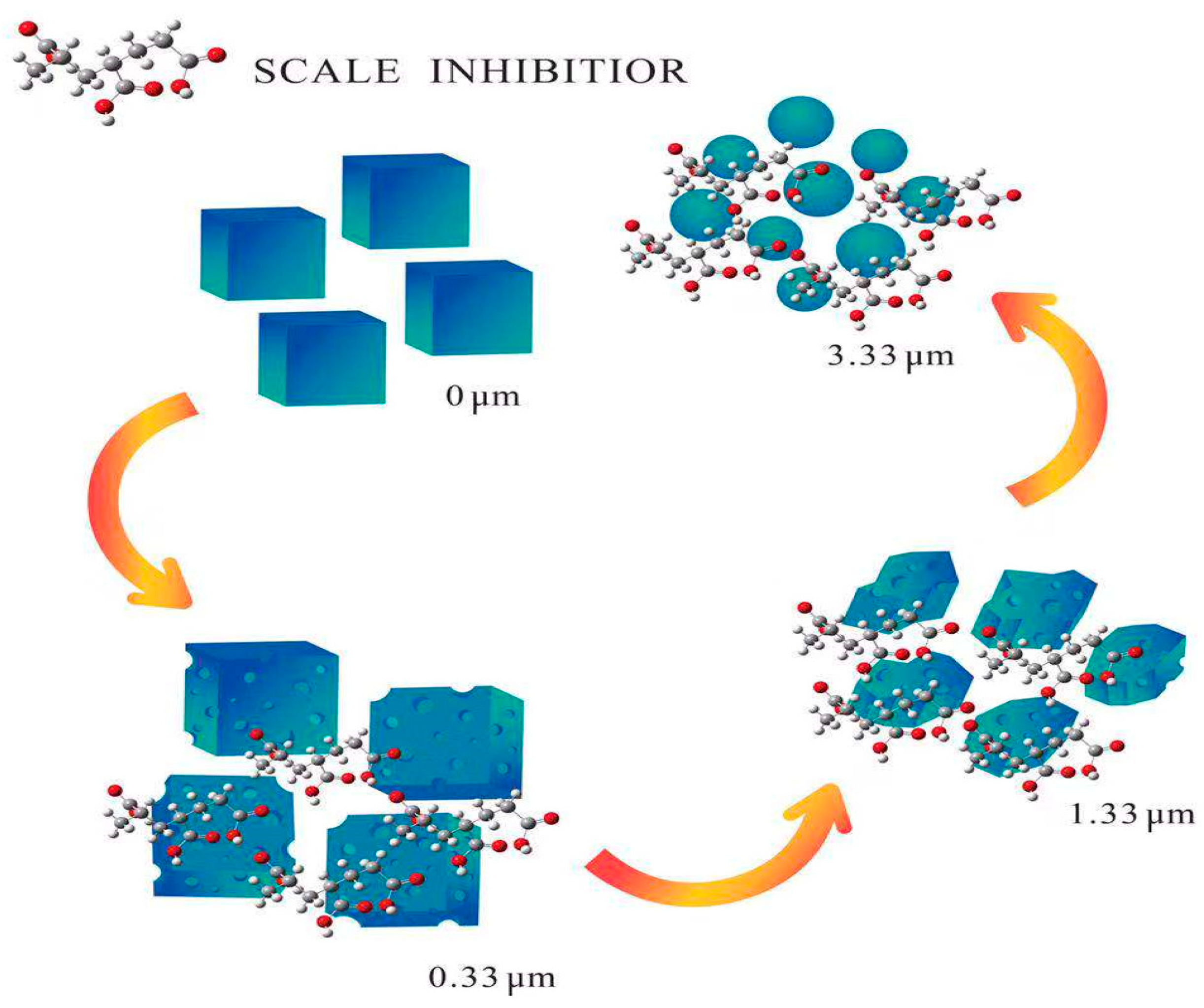
3.4. Scaling Inhibition Mechanism
4. Conclusions
- The static experiments revealed that at normal temperatures, PAA exhibited a better inhibition performance than PESA at low molecular weights, while in elevated temperatures, the behavior of both additives was more similar.
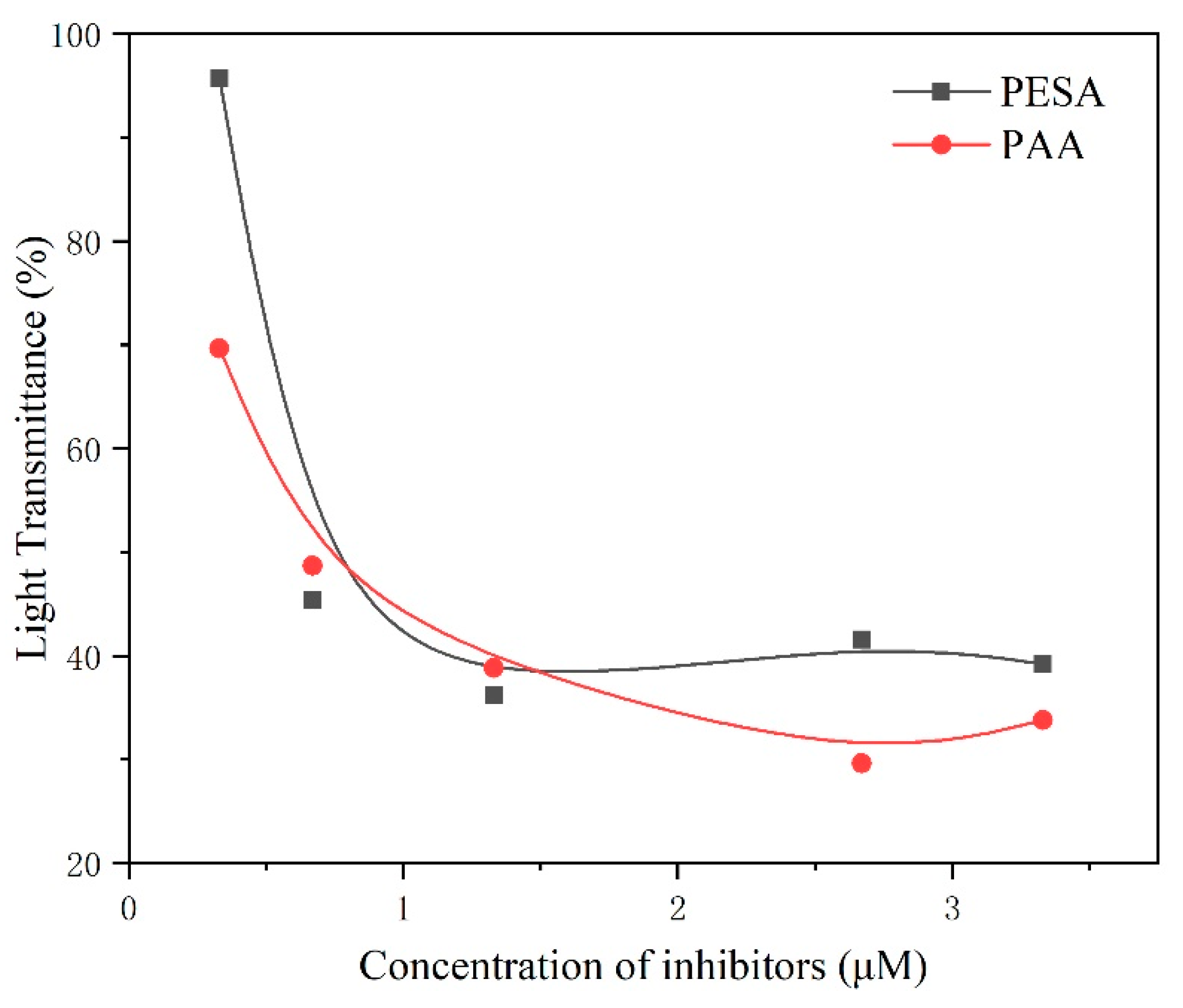
- PAA was demonstrated to exhibit better dispersion performance than PESA within this investigated molecular weight range.
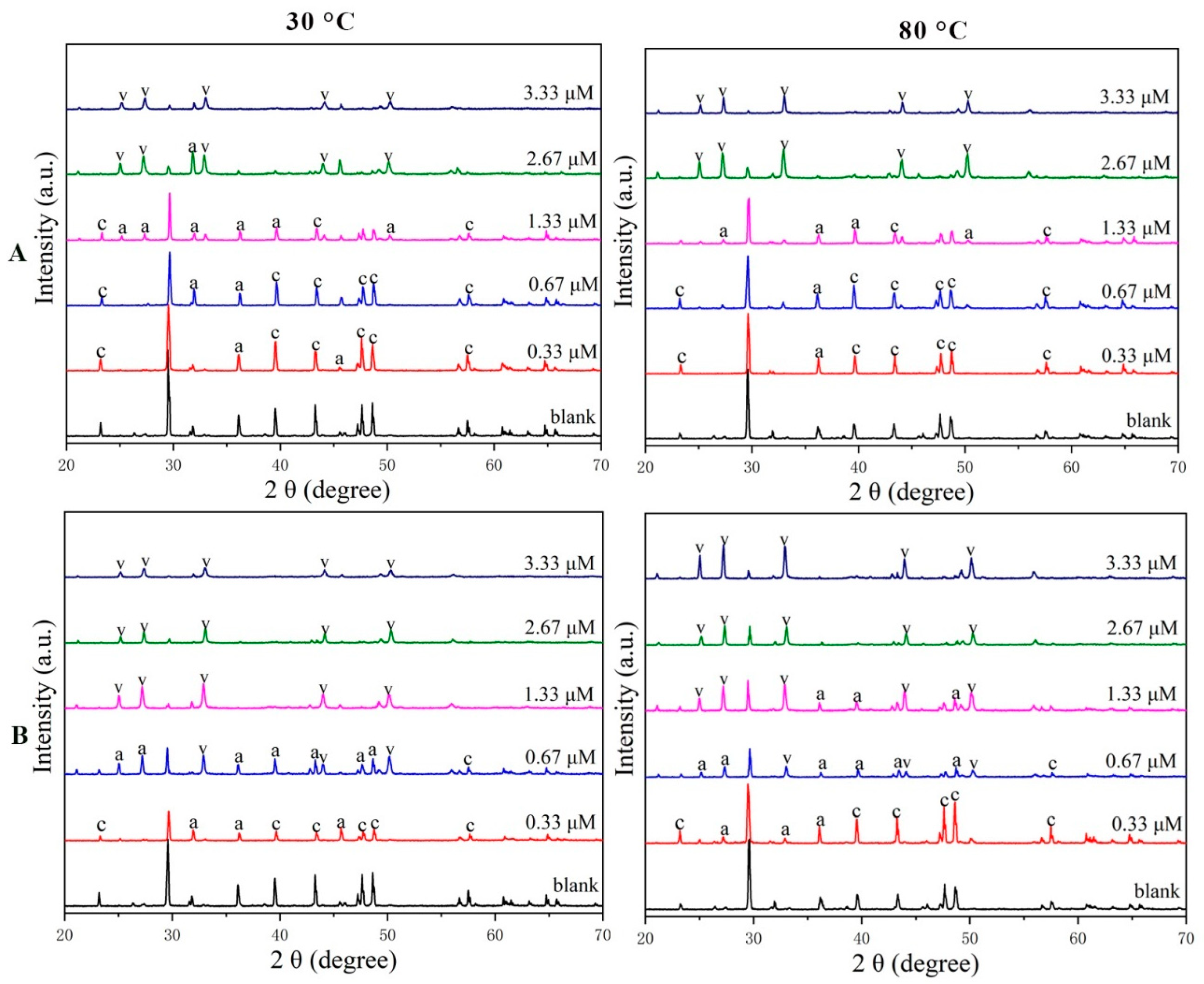
- Results of the XRD and SEM revealed that PAA and PESA inhibited the growth of the calcite phase at the (1 0 4) and (1 1 0) crystal planes, and favored the nucleation of vaterite.
- The stronger binding energies of PAA rendered the interaction of PAA with (1 0 4) and (1 1 0) calcite crystal surfaces stronger than that of PESA.
- Due to the different number of free carboxylic groups of PAA and PESA, PAA had more carboxylic acid groups, which led to a better scale inhibition efficiency than that of PESA.
Author Contributions
Funding
Conflicts of Interest
References
- Yang, L.; Yang, W.; Xu, B.; Yin, X.; Chen, Y.; Liu, Y.; Ji, Y.; Huan, Y. Synthesis and scale inhibition performance of a novel environmental friendly and hydrophilic terpolymer inhibitor. Desalination 2017, 416, 166–174. [Google Scholar] [CrossRef]
- Zhao, Y.; Xu, Z.; Wang, B.; He, J. Scale inhibition performance of sodium carboxymethyl cellulose on heat transfer surface at various temperatures: Experiments and molecular dynamics simulation. Int. J. Heat Mass Transf. 2019, 141, 457–463. [Google Scholar] [CrossRef]
- Okumura, M.; Kitano, Y. Coprecipitation of alkali metal ions with calcium carbonate. Geochimica et Cosmochimica Acta 1986, 50, 49–58. [Google Scholar] [CrossRef]
- Liu, G.; Xue, M.; Yang, H. Polyether copolymer as an environmentally friendly scale and corrosion inhibitor in seawater. Desalination 2017, 419, 133–140. [Google Scholar] [CrossRef]
- Zhang, S.; Qu, H.; Yang, Z.; Fu, C.-E.; Tian, Z.; Yang, W. Scale inhibition performance and mechanism of sulfamic/amino acids modified polyaspartic acid against calcium sulfate. Desalination 2017, 419, 152–159. [Google Scholar] [CrossRef]
- Shen, C.; Xu, X.; Hou, X.-Y.; Wu, D.-X.; Yin, J.-H. Molecular weight effect on PAA antiscale performance in LT-MED desalination system: Static experiment and MD simulation. Desalination 2018, 445, 1–5. [Google Scholar] [CrossRef]
- Yu, W.; Wang, Y.; Li, A.; Yang, H. Evaluation of the structural morphology of starch-graft-poly(acrylic acid) on its scale-inhibition efficiency. Water Res. 2018, 141, 86–95. [Google Scholar] [CrossRef]
- Zheng, T.; Zhang, X.; Yi, H. Spherical vaterite microspheres of calcium carbonate synthesized with poly (acrylic acid) and sodium dodecyl benzene sulfonate. J. Cryst. Growth 2019, 528, 125275. [Google Scholar] [CrossRef]
- Reddy, M.M.; Hoch, A.R. Calcite Crystal Growth Rate Inhibition by Polycarboxylic Acids. J. Colloid Interface Sci. 2001, 235, 365–370. [Google Scholar] [CrossRef]
- Chauhan, K.; Kumar, R.; Kumar, M.; Sharma, P.; Chauhan, G.S. Modified pectin-based polymers as green antiscalants for calcium sulfate scale inhibition. Desalination 2012, 305, 31–37. [Google Scholar] [CrossRef]
- Chen, J.; Xu, L.; Han, J.; Su, M.; Wu, Q. Synthesis of modified polyaspartic acid and evaluation of its scale inhibition and dispersion capacity. Desalination 2015, 358, 42–48. [Google Scholar] [CrossRef]
- Ji, Y.; Chen, Y.; Le, J.; Qian, M.; Huan, Y.; Yang, W.; Yin, X.; Liu, Y.; Wang, X.; Chen, Y. Highly effective scale inhibition performance of amino trimethylenephosphonic acid on calcium carbonate. Desalination 2017, 422, 165–173. [Google Scholar] [CrossRef]
- Zhao, Y.; Jia, L.; Liu, K.; Gao, P.; Ge, H.; Fu, L. Inhibition of calcium sulfate scale by poly (citric acid). Desalination 2016, 392, 1–7. [Google Scholar] [CrossRef]
- Abdel-Aal, N.; Sawada, K. Inhibition of adhesion and precipitation of CaCO3 by aminopolyphosphonate. J. Cryst. Growth 2003, 256, 188–200. [Google Scholar] [CrossRef]
- Mady, M.F.; Malmin, H.; Kelland, M.A. Sulfonated Nonpolymeric Aminophosphonate Scale Inhibitors—Improving the Compatibility and Biodegradability. Energy Fuels 2019, 33, 6197–6204. [Google Scholar] [CrossRef]
- Zhang, H.-P.; Luo, X.-G.; Lin, X.-Y.; Tang, P.-P.; Lu, X.; Yang, M.-J.; Tang, Y. Biodegradable carboxymethyl inulin as a scale inhibitor for calcite crystal growth: Molecular level understanding. Desalination 2016, 381, 1–7. [Google Scholar] [CrossRef]
- Menzri, R.; Ghizellaoui, S.; Tlili, M. Calcium carbonate inhibition by green inhibitors: Thiamine and Pyridoxine. Desalination 2017, 404, 147–154. [Google Scholar] [CrossRef]
- Zhang, W.; Gongwei, L.; Jin, F.; Huo, Y.; Sun, T.; Li, C. Synthesis and characterization of an ionic liquid-carboxylic acid copolymer scale inhibitor and its scale inhibition performance. Water Supply 2019, 19, 1463–1472. [Google Scholar] [CrossRef]
- Liu, D.; Dong, W.; Li, F.; Hui, F.; Lédion, J. Comparative performance of polyepoxysuccinic acid and polyaspartic acid on scaling inhibition by static and rapid controlled precipitation methods. Desalination 2012, 304, 1–10. [Google Scholar] [CrossRef]
- Sonobe, Y.; Watamura, H.; Hirasawa, I. Polymorphism, Size and Shape Control of Calcium Carbonate Crystals in the Presence of a Polyelectrolyte. Chem. Eng. Technol. 2015, 38, 1053–1058. [Google Scholar] [CrossRef]
- Rabizadeh, T.; Morgan, D.J.; Peacock, C.L.; Benning, L.G. Effectiveness of Green Additives vs Poly(acrylic acid) in Inhibiting Calcium Sulfate Dihydrate Crystallization. Ind. Eng. Chem. Res. 2019, 58, 1561–1569. [Google Scholar] [CrossRef]
- Al-Hamzah, A.A.; East, C.P.; Doherty, W.O.S.; Fellows, C.M. Inhibition of homogenous formation of calcium carbonate by poly (acrylic acid). The effect of molar mass and end-group functionality. Desalination 2014, 338, 93–105. [Google Scholar] [CrossRef]
- Laukala, T.; Kronlund, D.; Heiskanen, I.; Backfolk, K. The effect of polyacrylic acid and reaction conditions on nanocluster formation of precipitated calcium carbonate on microcellulose. Cellulose 2017, 88, 2409–2826. [Google Scholar] [CrossRef]
- Doherty, W.; Fellows, C.; Gorjian, S.; Senogles, E.; Cheung, A. Inhibition of calcium oxalate monohydrate by poly(acrylic acid)s with different end groups. J. Appl. Polym. Sci. 2003, 91, 2035–2041. [Google Scholar] [CrossRef]
- Rehman, A.U.; Khan, A.; Humayun, M.; Bibi, G.; Shi, K.; Raziq, F. Effect of biocides and anionic homopolymeric inhibitors on the precipitation behavior of calcium fluoride. Bull. Chem. Soc. Ethiop. 2017, 31, 115. [Google Scholar] [CrossRef][Green Version]
- Köken, N. Polymers containing amino bis(methylene phosphonic acid) groups for scale inhibition. Pigment Resin Technol. 2019, 48, 73–83. [Google Scholar] [CrossRef]
- Moulay, S.; Boukherissa, M.; Abdoune, F.; Benabdelmoumene, F. Low Molecular Weight Poly(acrylic acid) as a Salt Scaling Inhibitor in Oilfield Operations. J. Iran. Chem. Soc. 2005, 2, 212–219. [Google Scholar] [CrossRef]
- Alhamzah, A.; Smith, E.; Fellows, C. Inhibition of Homogeneous Formation of Magnesium Hydroxide by Low Molar Mass Poly(Acrylic Acid) with Different End-Groups. Ind. Eng. Chem. Res. 2015, 54, 150203192126006. [Google Scholar]
- Shi, W.-Y.; Ding, C.; Yan, J.-L.; Han, X.-Y.; Lv, Z.-M.; Lei, W.; Xia, M.-Z.; Wang, F.-Y. Molecular dynamics simulation for interaction of PESA and acrylic copolymers with calcite crystal surfaces. Desalination 2012, 291, 8–14. [Google Scholar] [CrossRef]
- Zhang, B.; Sun, P.; Chen, F.; Li, F. Synergistic inhibition effect of polyaminoamide dendrimers and polyepoxysuccinic acid on silica polymerization. Colloids Surf. A Physicochem. Eng. Asp. 2012, 410, 159–169. [Google Scholar] [CrossRef]
- Zhu, B.; Xu, X.; Tang, R. Hydration layer structures on calcite facets and their roles in selective adsorptions of biomolecules: A molecular dynamics study. J. Chem. Phys. 2013, 139, 234705. [Google Scholar] [CrossRef] [PubMed]
- Yuan, P.-Q.; Cheng, Z.-M.; Zhou, Z.-M.; Yuan, W.-K.; Semiat, R. Zeta potential on the anti-scalant modified sub-micro calcite surface. Colloids Surf. A Physicochem. Eng. Asp. 2008, 328, 60–66. [Google Scholar] [CrossRef]
- Prabhakar, S.; Melnik, R. Influence of Mg2+, SO42− and Na+ ions of sea water in crude oil recovery: DFT and ab initio molecular dynamics simulations. Colloids Surf. A Physicochem. Eng. Asp. 2018, 539, 53–58. [Google Scholar] [CrossRef]
- Zhang, R.; Wang, X.; Zhou, L.; Liu, Z.; Crump, D. The impact of dissolved oxygen on sulfate radical-induced oxidation of organic micro-pollutants: A theoretical study. Water Res. 2018, 135, 144–154. [Google Scholar] [CrossRef] [PubMed]
- Kavitha, A.L.; Vasudevan, T.; Prabu, H.G. Evaluation of synthesized antiscalants for cooling water system application. Desalination 2011, 268, 38–45. [Google Scholar] [CrossRef]
- Yuan, X.; Dong, S.; Zheng, Q.; Yang, W.; Huang, T. Novel and efficient curcumin based fluorescent polymer for scale and corrosion inhibition. Chem. Eng. J. 2020, 389, 124296. [Google Scholar] [CrossRef]
- Suo, G.; Xie, L.; Xu, S.; Feng, L.; Dong, T.; Shao, X. Study on inhibitors’ performance under the condition of high concentration ratio in MED system. Desalination 2018, 437, 100–107. [Google Scholar] [CrossRef]
- Ylikantola, A.; Linnanto, J.; Knuutinen, J.; Oravilahti, A.; Toivakka, M. Molecular modeling studies of interactions between sodium polyacrylate polymer and calcite surface. Appl. Surf. Sci. 2013, 276, 43–52. [Google Scholar] [CrossRef]
- Chen, C.; Lei, W.; Xia, M.; Wang, F.; Gong, X. Molecular modeling of several phosphonates onto the stepped calcite (0 1 1) surface. Desalination 2013, 309, 208–212. [Google Scholar] [CrossRef]
- Liu, Q.; Wang, Q.; Xiang, L. Influence of poly acrylic acid on the dispersion of calcite nano-particles. Appl. Surf. Sci. 2008, 254, 7104–7108. [Google Scholar] [CrossRef]
- Shi, W.; Xia, M.; Lei, W.; Wang, F. Molecular dynamics study of polyether polyamino methylene phosphonates as an inhibitor of anhydrite crystal. Desalination 2013, 322, 137–143. [Google Scholar] [CrossRef]
- Dubis, A.; Zamaraeva, M.V.; Siergiejczyk, L.; Charishnikova, O.; Shlyonsky, V. Ferutinin as a Ca(2+) complexone: Lipid bilayers, conductometry, FT-IR, NMR studies and DFT-B3LYP calculations. Dalton Trans. 2015, 44, 16506–16515. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Yang, F.; Zhao, X.; Li, Y. Effects of nitro- and amino-group on the antioxidant activity of genistein: A theoretical study. Food Chem. 2019, 275, 339–345. [Google Scholar] [CrossRef]
- Zhang, K.; Yang, W.; Xu, B.; Chen, Y.; Yin, X.; Liu, Y.; Zuo, H. Inhibitory effect of konjac glucomanan on pitting corrosion of AA5052 aluminium alloy in NaCl solution. J. Colloid Interface Sci. 2018, 517, 52–60. [Google Scholar] [CrossRef] [PubMed]
- Elliot, M.N. Scale control by threshold treatment. Desalination 1970, 8, 221–236. [Google Scholar] [CrossRef]
- Zhang, K.; Xu, B.; Yang, W.; Yin, X.; Liu, Y.; Chen, Y. Halogen-substituted imidazoline derivatives as corrosion inhibitors for mild steel in hydrochloric acid solution. Corros. Sci. 2015, 90, 284–295. [Google Scholar] [CrossRef]
- Tang, Y.; Yang, X.; Yang, W.; Wan, R.; Chen, Y.; Yin, X. A preliminary investigation of corrosion inhibition of mild steel in 0.5M H2SO4 by 2-amino-5-(n-pyridyl)-1,3,4-thiadiazole: Polarization, EIS and molecular dynamics simulations. Corros. Sci. 2010, 52, 1801–1808. [Google Scholar] [CrossRef]
- Weber, E.; Levy, D.; Sasson, M.B.; Fitch, A.N.; Pokroy, B. Structural analysis of metal-doped calcium oxalate. RSC Adv. 2015, 5, 98626–98633. [Google Scholar] [CrossRef]

| Surfaces | Molecules | Einter | Eb |
|---|---|---|---|
| (1 0 4) | PESA | −69.698 | 69.698 |
| PAA | −106.229 | 106.229 | |
| (1 1 0) | PESA | −86.212 | 86.212 |
| PAA | −141.308 | 141.308 |
| Inhibitors | O(–H) | (C=)O |
|---|---|---|
| PESA | −0.554 | −0.389 |
| PAA | −0.610 | −0.438 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zuo, Y.; Yang, W.; Zhang, K.; Chen, Y.; Yin, X.; Liu, Y. Experimental and Theoretical Studies of Carboxylic Polymers with Low Molecular Weight as Inhibitors for Calcium Carbonate Scale. Crystals 2020, 10, 406. https://doi.org/10.3390/cryst10050406
Zuo Y, Yang W, Zhang K, Chen Y, Yin X, Liu Y. Experimental and Theoretical Studies of Carboxylic Polymers with Low Molecular Weight as Inhibitors for Calcium Carbonate Scale. Crystals. 2020; 10(5):406. https://doi.org/10.3390/cryst10050406
Chicago/Turabian StyleZuo, Yuwei, Wenzhong Yang, Kegui Zhang, Yun Chen, Xiaoshuang Yin, and Ying Liu. 2020. "Experimental and Theoretical Studies of Carboxylic Polymers with Low Molecular Weight as Inhibitors for Calcium Carbonate Scale" Crystals 10, no. 5: 406. https://doi.org/10.3390/cryst10050406
APA StyleZuo, Y., Yang, W., Zhang, K., Chen, Y., Yin, X., & Liu, Y. (2020). Experimental and Theoretical Studies of Carboxylic Polymers with Low Molecular Weight as Inhibitors for Calcium Carbonate Scale. Crystals, 10(5), 406. https://doi.org/10.3390/cryst10050406




