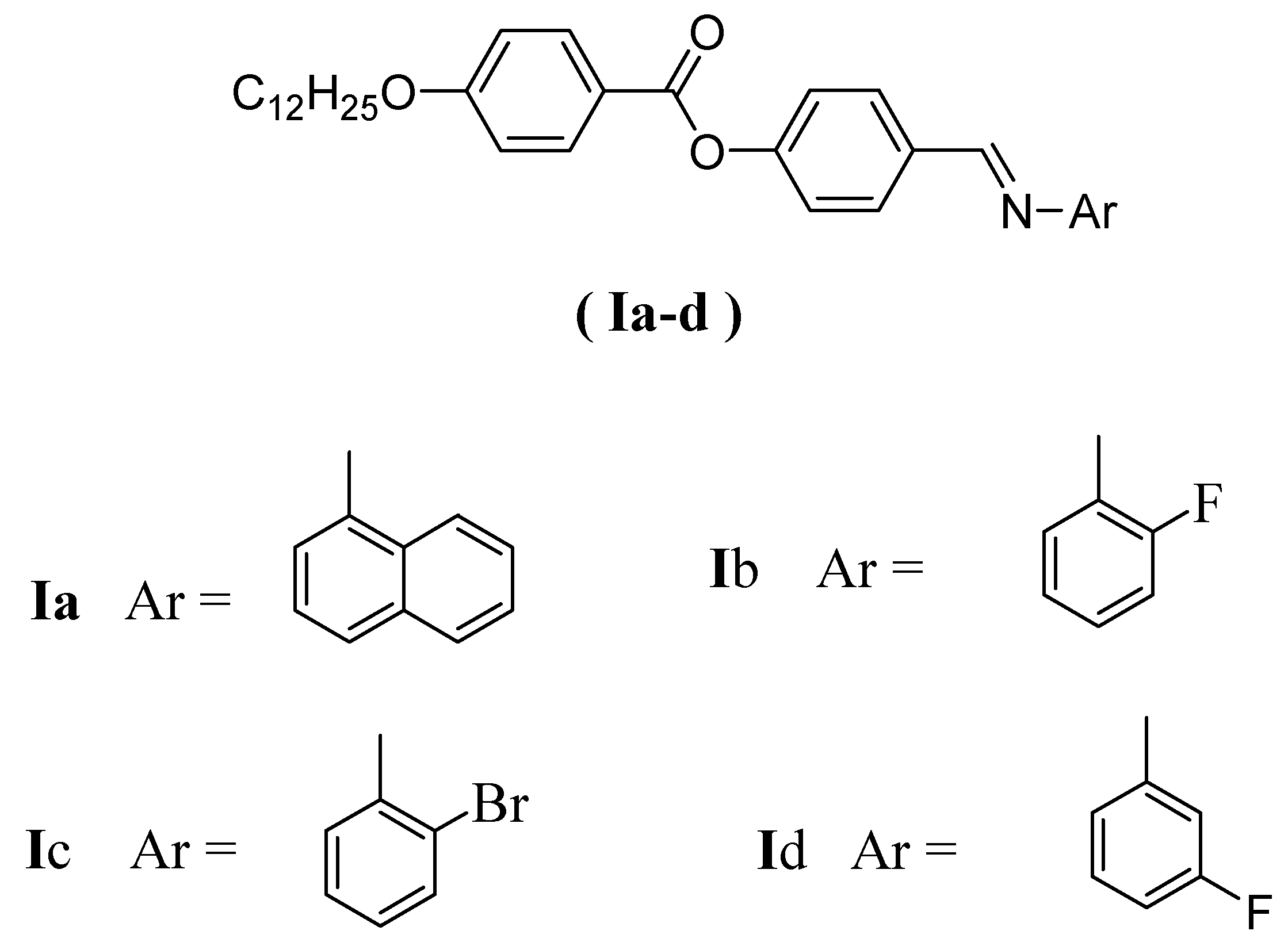
Binary Liquid Crystal Mixtures Based on Schiff Base Derivatives with Oriented Lateral Substituents
Abstract
1. Introduction
2. Experimental
2.1. Materials and Preparation of Binary Mixtures
2.2. Physical Characterization
2.3. Computational Method and Calculations
3. Results and Discussion
3.1. Binary Mixtures Studies
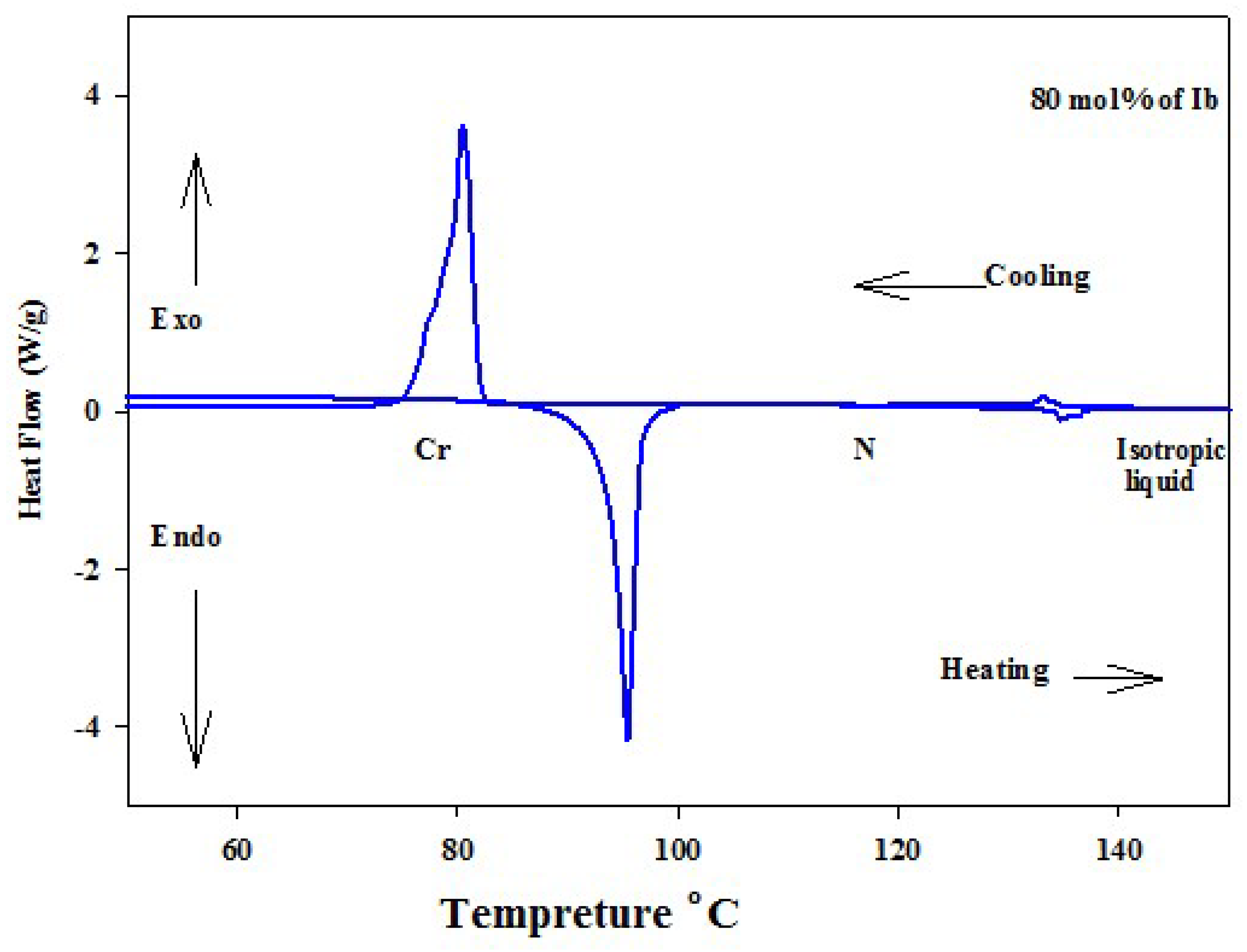
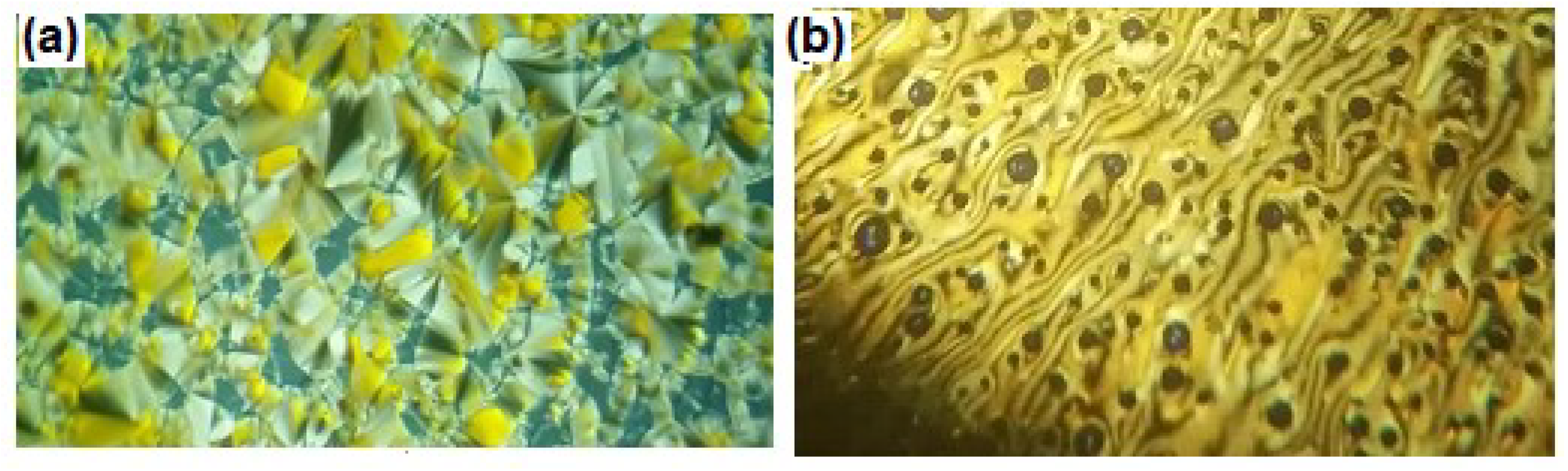
3.1.1. Binary Phase Behavior of Mixtures of Analogues of Different Rigidity
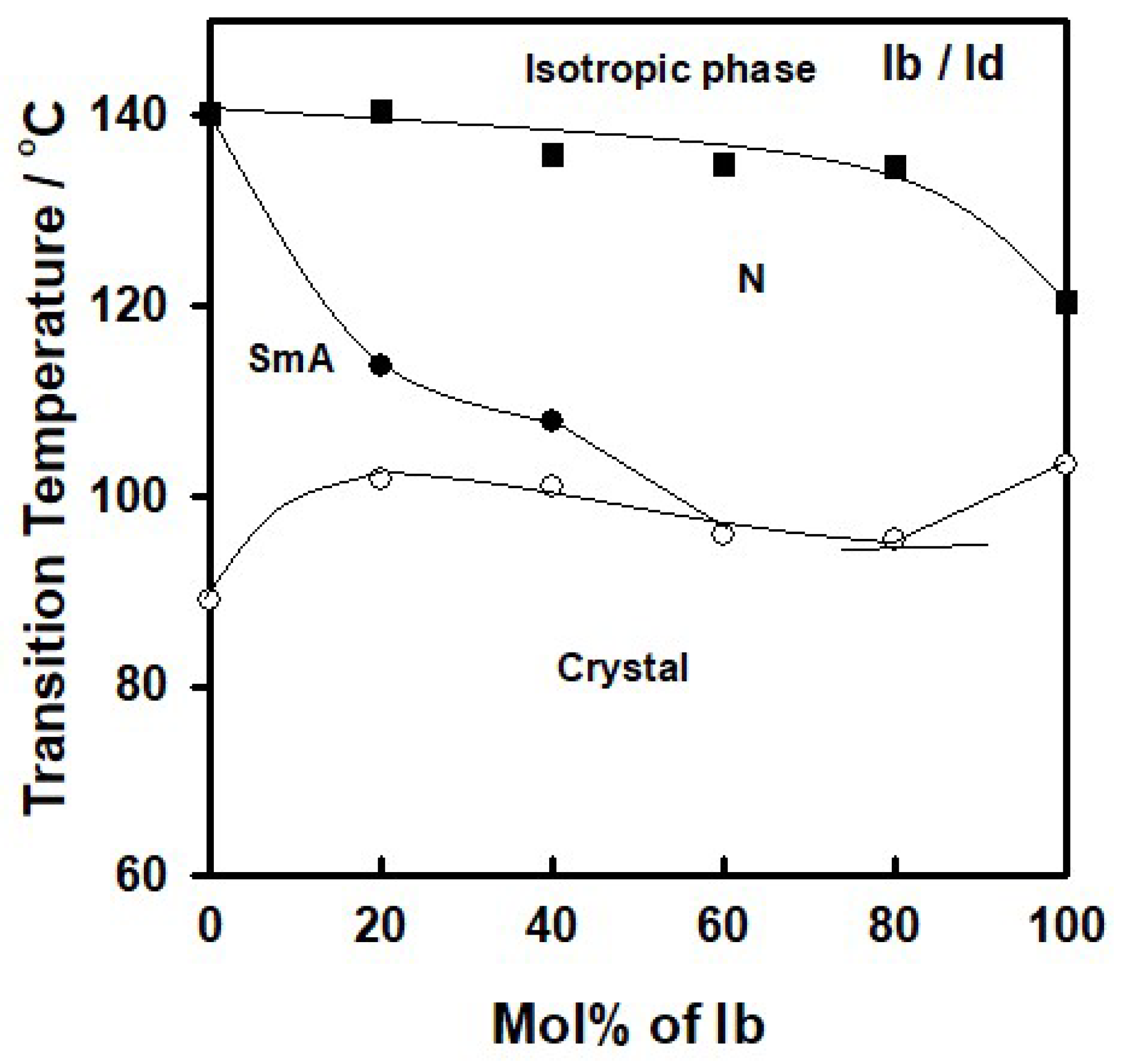
3.1.2. Binary Mixtures of the Analogues Bearing Substituent of the Same Polarity but of Different Orientation
3.1.3. Binary Mixtures of the Analogues Bearing Substituents of the Different Orientation and Different Size
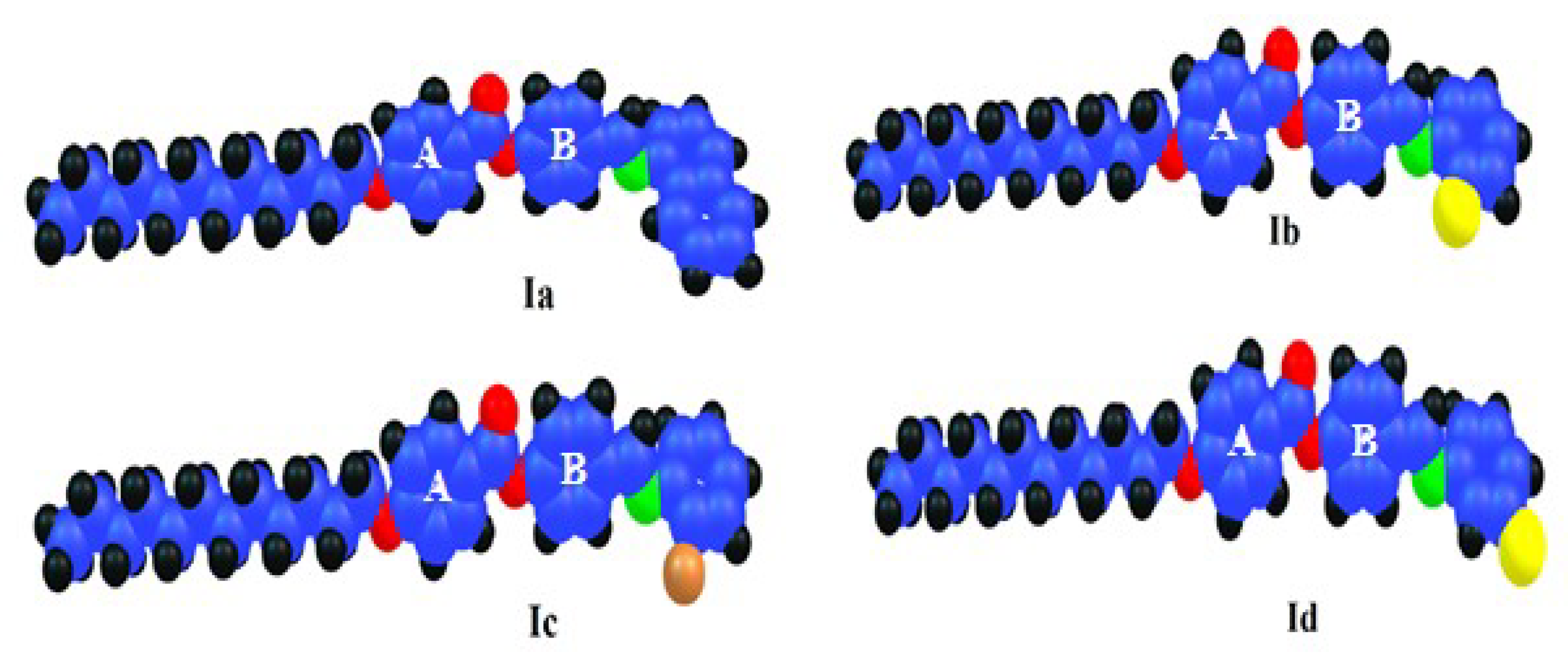
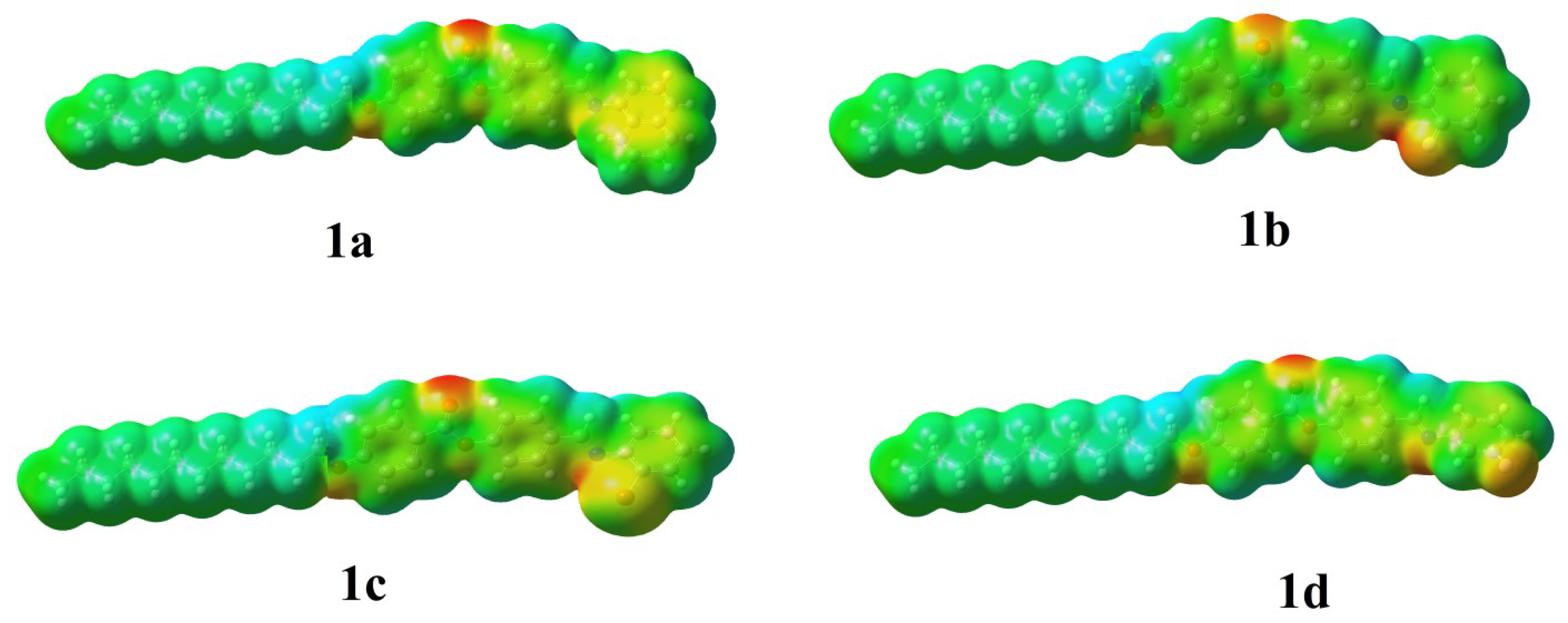
3.2. DFT Theoretical Calculations
4. Conclusions
- All prepared binary phase diagrams showed enantiotropic mesomorphic behavior.
- Induced SmA phase was observed for the Ia/Ib system and covered all mixing compositions.
- As usual, all binary systems showed depression in the melting temperature
- The binary phase mixtures Ia/Ib and Ib/Id each possessed eutectic behavior, but the third Ic/Id mixture showed no eutectic compositions.
- Regular decreases in nematic-to-isotropic liquid dependency were exhibited in all binary phase diagrams.
- The estimated aspect ratios for the lateral components increased in the following order: Ia < Ic < Ib < Id.
- The polarizability was highly impacted by the dimensions and the type of lateral group.
- Id showed the highest dipole moment value (6 Debye), thus it has a high degree of lateral interactions, which impact the mesophase behavior.
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Fujikake, H.; Sato, H.; Murashige, T. Polymer-stabilized ferroelectric liquid crystal for flexible displays. Displays 2004, 25, 3–8. [Google Scholar] [CrossRef]
- Ahmed, H.; Hagar, M.; Alhaddad, O. New chair shaped supramolecular complexes-based aryl nicotinate derivative; mesomorphic properties and DFT molecular geometry. RSC Adv. 2019, 9, 16366–16374. [Google Scholar] [CrossRef]
- Chen, K.-Y. Crystal Structure, Hydrogen-Bonding Properties, and DFT Studies of 2-((2-(2-Hydroxyphenyl) benzo [d] thiazol-6-yl) methylene) malononitrile. Mol. Cryst. Liq. Cryst. 2015, 623, 285–296. [Google Scholar] [CrossRef]
- Shoji, M.; Tanaka, F. Theoretical study of hydrogen-bonded supramolecular liquid crystals. Macromolecules 2002, 35, 7460–7472. [Google Scholar] [CrossRef]
- Sundaram, S.; Jayaprakasam, R.; Dhandapani, M.; Senthil, T.; Vijayakumar, V. Theoretical (DFT) and experimental studies on multiple hydrogen bonded liquid crystals comprising between aliphatic and aromatic acids. J. Mol. Liq. 2017, 243, 14–21. [Google Scholar] [CrossRef]
- Al-Mutabagani, L.; Alshabanah, L.A.; Ahmed, H.; Hagar, M.; Al-Ola, K.A.A. New Symmetrical U- and Wavy- shaped Supramolecular H-bonded Systems; Geometrical and Mesomorphic Approaches. Molecules 2020, 25, 1420. [Google Scholar] [CrossRef]
- Hagar, M.; Ahmed, H.; Saad, G. New calamitic thermotropic liquid crystals of 2-hydroxypyridine ester mesogenic core: Mesophase behaviour and DFT calculations. Liq. Cryst. 2020, 47, 114–124. [Google Scholar] [CrossRef]
- Ahmed, N.H.; Saad, G.R.; Ahmed, H.A.; Hagar, M. New wide-stability four-ring azo/ester/Schiff base liquid crystals: Synthesis, mesomorphic, photophysical, and DFT approaches. RSC Adv. 2020, 10, 9643–9656. [Google Scholar] [CrossRef]
- Alhaddad, O.; Ahmed, H.; Hagar, M. Experimental and Theoretical Approaches of New Nematogenic Chair Architectures of Supramolecular H-Bonded Liquid Crystals. Molecules 2020, 25, 365. [Google Scholar] [CrossRef]
- Nafee, S.S.; Ahmed, H.A.; Hagar, M. New architectures of supramolecular H-bonded liquid crystal complexes based on dipyridine derivatives. Liq. Cryst. 2020, 1–14. [Google Scholar] [CrossRef]
- Nafee, S.S.; Hagar, M.; Ahmed, H.A.; El-Shishtawy, R.M.; Raffah, B.M. The synthesis of new thermal stable schiff base/ester liquid crystals: A computational, mesomorphic, and optical study. Molecules 2019, 24, 3032. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, H.; Hagar, M.; Saad, G. Impact of the proportionation of dialkoxy chain length on the mesophase behaviour of Schiff base/ester liquid crystals; experimental and theoretical study. Liq. Cryst. 2019, 46, 1611–1620. [Google Scholar] [CrossRef]
- Ahmed, H.A.; Hagar, M.; Alhaddad, O.A. Phase behavior and DFT calculations of laterally methyl supramolecular hydrogen-bonding complexes. Crystals 2019, 9, 133. [Google Scholar] [CrossRef]
- Hagar, M.; Ahmed, H.; El-Sayed, T.; Alnoman, R. Mesophase behavior and DFT conformational analysis of new symmetrical diester chalcone liquid crystals. J. Mol. Liq. 2019, 285, 96–105. [Google Scholar] [CrossRef]
- Alnoman, R.; Ahmed, H.A.; Hagar, M. Synthesis, optical, and geometrical approaches of new natural fatty acids’ esters/Schiff base liquid crystals. Molecules 2019, 24, 4293. [Google Scholar] [CrossRef] [PubMed]
- Hagar, M.; Ahmed, H.; Alhaddad, O. New azobenzene-based natural fatty acid liquid crystals with low melting point: Synthesis, DFT calculations and binary mixtures. Liq. Cryst. 2019, 46, 2223–2234. [Google Scholar] [CrossRef]
- Ahmed, H.; Hagar, M.; Alhaddad, O. Mesomorphic and geometrical orientation study of the relative position of fluorine atom in some thermotropic liquid crystal systems. Liq. Cryst. 2019, 1–10. [Google Scholar] [CrossRef]
- Zaki, A.A.; Ahmed, H.; Hagar, M. Impact of fluorine orientation on the optical properties of difluorophenylazophenyl benzoates liquid crystal. Mater. Chem. Phys. 2018, 216, 316–324. [Google Scholar] [CrossRef]
- Weissflog, W.; Demus, D. Compounds with lateral long-chain substituents—A new molecule structure concept for thermotropic liquid crystals. Cryst. Res. Technol. 1983, 18, K21–K24. [Google Scholar] [CrossRef]
- Weissflog, W.; Demus, D. Thermotropic liquid crystalline compounds with lateral long-chain substituents (II): Synthesis and liquid crystalline properties of 1, 4-Bis [4-substituted-benzoyloxy]-2-n-alkylbenzenes. Cryst. Res. Technol. 1984, 19, 55–64. [Google Scholar] [CrossRef]
- Weissflog, W.; Demus, D. New lateral long-chain substituted liquid crystals. Mol. Cryst. Liq. Cryst. 1985, 129, 235–243. [Google Scholar] [CrossRef]
- Ahmed, H.; Hagar, M.; Aljuhani, A. Mesophase behavior of new linear supramolecular hydrogen-bonding complexes. RSC Adv. 2018, 8, 34937–34946. [Google Scholar] [CrossRef]
- Hagar, M.; Ahmed, H.; Saad, G. Mesophase stability of new Schiff base ester liquid crystals with different polar substituents. Liq. Cryst. 2018, 45, 1324–1332. [Google Scholar] [CrossRef]
- Dave, J.; Patel, P.; Vasanth, K. Mixed mesomorphism in binary systems forming smectic-nematic phases. Mol. Cryst. 1969, 8, 93–100. [Google Scholar] [CrossRef]
- Dave, J.S.; Menon, M.R.; Patel, P.R. Chiral phases induced by doping nonmesogenic component into mesogenic esters. Mol. Cryst. Liq. Cryst. 2003, 392, 83–95. [Google Scholar] [CrossRef]
- Vora, R.; Gupta, R.; Patel, K. Exhibition of induced mesophases in the binary systems where one or both the components are non-mesogenic. Mol. Cryst. Liq. Cryst. 1991, 209, 251–263. [Google Scholar] [CrossRef]
- Fujimura, S.; Yamamura, Y.; Hishida, M.; Nagatomo, S.; Saito, K. Reentrant nematic phase in 4-alkyl-4′-cyanobiphenyl (n CB) binary mixtures. Liq. Cryst. 2014, 41, 927–932. [Google Scholar] [CrossRef]
- Prasad, A.; Das, M.K. Determination of elastic constants of a binary system (7CPB+ 9. CN) showing nematic, induced smectic Ad and re-entrant nematic phases. Liq. Cryst. 2014, 41, 1261–1268. [Google Scholar] [CrossRef]
- Salud, J.; Lopez, D.; Diez-Berart, S.; de la Fuente, M. Tests of the tricritical point in the SmA-to-N phase transition of binary mixtures of butyloxybenzylidene octylaniline and hexyloxybenzylidene octylaniline. Liq. Cryst. 2013, 40, 293–304. [Google Scholar] [CrossRef]
- Prajapati, A.K.; Patel, N.S.; Lad, V.G. Induction of chirality by doping mesogens with non-mesogenic chiral dopant. Mol. Cryst. Liq. Cryst. Sci. Technol. Sect. A. Mol. Cryst. Liq. Cryst. 2000, 348, 41–51. [Google Scholar] [CrossRef]
- Vora, R.; Rajput, S. Binary mesogenic systems comprised of ester mesogens and non-mesogens. Mol. Cryst. Liq. Cryst. 1991, 209, 265–277. [Google Scholar] [CrossRef]
- Govindaiah, T.; Nagappa; Sathyanarayana, P.; Mahadeva, J.; Sreepad, H. Induced chiral smectic phase in mixtures of mesogenic and non-mesogenic compounds. Mol. Cryst. Liq. Cryst. 2011, 548, 55–60. [Google Scholar] [CrossRef]
- Lohar, J.; Dave, J.S., Jr. Emergence of smectic mesophase in binary mixtures of pure nematogens. Mol. Cryst. Liq. Cryst. 1983, 103, 181–192. [Google Scholar] [CrossRef]
- Sarkar, S.D.; Choudhury, B. Study of binary mixtures of two liquid crystalline samples showing induced smectic phase. Assam Univ. J. Sci. Technol. 2010, 5, 167–168. [Google Scholar]
- Ahmed, H.; Naoum, M. Mesophase behavior of binary and ternary mixtures of benzoic acids bearing terminal substituents of different polarity and chain-lengths. Thermochim. Acta 2014, 575, 122–128. [Google Scholar] [CrossRef]
- Alhaddad, O.A.; Ahmed, H.A.; Hagar, M.; Saad, G.R.; Al-Ola, A.; Khulood, A.; Naoum, M.M. Thermal and Photophysical Studies of Binary Mixtures of Liquid Crystal with Different Geometrical Mesogens. Crystals 2020, 10, 223. [Google Scholar] [CrossRef]
- Ahmed, H.; Naoum, M.; Saad, G. Mesophase behaviour of 1: 1 mixtures of 4-n-alkoxyphenylazo benzoic acids bearing terminal alkoxy groups of different chain lengths. Liq. Cryst. 2016, 43, 1259–1267. [Google Scholar] [CrossRef]
- Naoum, M.; Mohammady, S.; Ahmed, H. Lateral protrusion and mesophase behaviour in pure and mixed states of model compounds of the type 4-(4′-substituted phenylazo)-2-(or 3-) methyl phenyl-4′-alkoxy benzoates. Liq. Cryst. 2010, 37, 1245–1257. [Google Scholar] [CrossRef]
- Mohamady, S.Z.; Nessim, R.I.; Shehab, O.R.; Naoum, M.M. Effect of steric factor on mesomorphic stability, II: Binary mixtures of homologues of 4-(4′-substituted phenylazo)-1-naphthyl-4″-alkoxybenzoates. Mol. Cryst. Liq. Cryst. 2006, 451, 53–64. [Google Scholar] [CrossRef]
- Naoum, M.; Fahmi, A.; Alaasar, M.; Abdel-Aziz, M. Effect of lateral substitution of different polarity on the mesophase behaviour in pure and mixed states of 4-(4′-substituted phenylazo)-2-substituted phenyl-4″-alkoxy benzoates. Liq. Cryst. 2011, 38, 391–405. [Google Scholar] [CrossRef]
- Frisch, M.; Trucks, G.; Schlegel, H.B.; Scuseria, G.; Robb, M.; Cheeseman, J.; Scalmani, G.; Barone, V.; Mennucci, B.; Petersson, G. Gaussian 09, Revision A. 02; Gaussian Inc.: Wallingford, CT, USA, 2009; p. 200. [Google Scholar]
- Dennington, R.; Keith, T.; Millam, J. GaussView, Version 5; Semichem Inc.: Crieff, UK, 2009. [Google Scholar]
- Ahmed, H.; Hagar, M.; El-Sayed, T.B.; Alnoman, R. Schiff base/ester liquid crystals with different lateral substituents: Mesophase behaviour and DFT calculations. Liq. Cryst. 2019, 46, 1–11. [Google Scholar] [CrossRef]
- Naoum, M.M.; Saad, G.R.; Nessim, R.I.; Abdel-Aziz, T.A.; Seliger, H. Effect of molecular structure on the phase behaviour of some liquid crystalline compounds and their binary mixtures II. 4-Hexadecyloxyphenyl arylates and aryl 4-hexadecyloxy benzoates. Liq. Cryst. 1997, 23, 789–795. [Google Scholar] [CrossRef]
- Nafee, S.S.; Ahmed, H.; Hagar, M. Theoretical, experimental and optical study of new thiophene-based liquid crystals and their positional isomers. Liq. Cryst. 2020, 1–12. [Google Scholar] [CrossRef]
- Hagar, M.; Ahmed, H.; Saad, G. Synthesis and mesophase behaviour of Schiff base/ester 4-(arylideneamino) phenyl-4″-alkoxy benzoates and their binary mixtures. J. Mol. Liq. 2019, 273, 266–273. [Google Scholar] [CrossRef]
- Nafee, S.S.; Hagar, M.; Ahmed, H.A.; Alhaddad, O.; El-Shishtawy, R.M.; Raffah, B.M. New two rings Schiff base liquid crystals; ball mill synthesis, mesomorphic, Hammett and DFT studies. J. Mol. Liq. 2019, 299, 112161. [Google Scholar] [CrossRef]
- Kim, S.N.; Kastelic, J.R. Use of Liquid Crystal Polymer Particulates Having a High Aspect Ratio in Polymeric Molding Resins to Suppress Melt Dripping. U.S. Patent 4,439,578, 27 March 1984. [Google Scholar]
- Sengupta, A.; Tkalec, U.; Ravnik, M.; Yeomans, J.M.; Bahr, C.; Herminghaus, S. Liquid crystal microfluidics for tunable flow shaping. Phys. Rev. Lett. 2013, 110, 048303. [Google Scholar] [CrossRef]


| Compound | TCr–SmA | ΔHCr–SmA | TCr–N | ΔHCr–N | TSmA–I | ΔHSmA–I | TSmA–N | ΔHSmA–N | TN–I | ΔHN–I |
|---|---|---|---|---|---|---|---|---|---|---|
| System Ia/Ib | ||||||||||
| 0% Ia | - | - | 103.3 | 48.28 | 120.3 | 0.80 | ||||
| 20% Ia | 98.0 | 69.10 | 107.3 | 1.89 | 120.9 | 1.26 | ||||
| 40% Ia | 76.9 | 64.3 | 97.1 | 3.16 | 111.3 | 2.67 | ||||
| 60% Ia | 77.5 | 44.79 | 95.8 | 5.25 | 111.5 | 1.64 | ||||
| 80% Ia | 84.9 | 69.15 | 94.8 | 2.43 | 105.9 | 1.77 | ||||
| 100% Ia | 98.8 | 19.73 | 96.3 * | 0.18 | ||||||
| System Ib/Id | ||||||||||
| 0% Ib | 88.9 | 45.69 | 140.2 | 5.10 | ||||||
| 20% Ib | 101.8 | 58.65 | 113.5 | 0.98 | 140.4 | 1.55 | ||||
| 40% Ib | 100.9 | 69.09 | 107.7 | 0.66 | 132.4 | 1.93 | ||||
| 60% Ib | 95.9 | 94.61 | 135.8 | 2.38 | ||||||
| 80% Ib | 95.4 | 68.64 | 134.6 | 1.25 | ||||||
| 100% Ib | 103.3 | 48.28 | 120.3 | 0.80 | ||||||
| System Ic/Id | ||||||||||
| 0% Ic | 88.9 | 45.69 | 140.2 | 5.10 | ||||||
| 20% Ic | 98.5 | 66.90 | 115.6 | 0.89 | 124.8 | 1.52 | ||||
| 40% Ic | 99.4 | 73.3 | 119.6 | 1.10 | ||||||
| 60% Ic | 100.0 | 69.46 | 118.3 | 0.91 | ||||||
| 80% Ic | 100.7 | 71.84 | 112.8 | 0.85 | ||||||
| 100% Ic | 93.2 | 46.33 | 99.0 | 0.37 | ||||||
| Parameter | Ia | Ib | Ic | Id | |
|---|---|---|---|---|---|
| Polarizability α | 465.85 | 414.09 | 424.56 | 425.11 | |
| Dipole Moment | 4.35 | 4.09 | 4.25 | 6.12 | |
| Twist angle between A-B | 40 | 41 | 39 | 40 | |
| Dimensions Å | Width (D) | 9.51 | 7.89 | 8.76 | 7.82 |
| Length (L) | 36.81 | 36.65 | 36.51 | 36.56 | |
| Aspect ratio (L/D) | 3.87 | 4.65 | 4.17 | 4.68 | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alnoman, R.B.; Hagar, M.; Ahmed, H.A.; Naoum, M.M.; Sobaih, H.A.; Almshaly, J.S.; Haddad, M.M.; Alhaisoni, R.A.; Alsobhi, T.A. Binary Liquid Crystal Mixtures Based on Schiff Base Derivatives with Oriented Lateral Substituents. Crystals 2020, 10, 319. https://doi.org/10.3390/cryst10040319
Alnoman RB, Hagar M, Ahmed HA, Naoum MM, Sobaih HA, Almshaly JS, Haddad MM, Alhaisoni RA, Alsobhi TA. Binary Liquid Crystal Mixtures Based on Schiff Base Derivatives with Oriented Lateral Substituents. Crystals. 2020; 10(4):319. https://doi.org/10.3390/cryst10040319
Chicago/Turabian StyleAlnoman, Rua B., Mohamed Hagar, Hoda A. Ahmed, Magdi M. Naoum, Hanefah A. Sobaih, Jawaher S. Almshaly, Mawadh M. Haddad, Rana A. Alhaisoni, and Tahani A. Alsobhi. 2020. "Binary Liquid Crystal Mixtures Based on Schiff Base Derivatives with Oriented Lateral Substituents" Crystals 10, no. 4: 319. https://doi.org/10.3390/cryst10040319
APA StyleAlnoman, R. B., Hagar, M., Ahmed, H. A., Naoum, M. M., Sobaih, H. A., Almshaly, J. S., Haddad, M. M., Alhaisoni, R. A., & Alsobhi, T. A. (2020). Binary Liquid Crystal Mixtures Based on Schiff Base Derivatives with Oriented Lateral Substituents. Crystals, 10(4), 319. https://doi.org/10.3390/cryst10040319







