Characteristics of Flakes Stacked Cr2N with Many Domains in Super Duplex Stainless Steel
Abstract
1. Introductions
2. Materials and Methods
3. Results and Discussion
3.1. Cr2N Transformation at 600 °C under 7 h Holding in SAF 2507
3.2. The Detailed Substructure of Cr2N
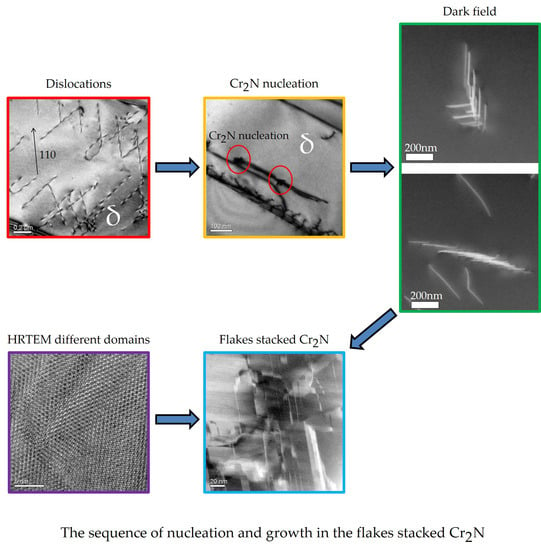
3.3. Nucleation and Growth Mechanism of Cr2N at 600 °C
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Davision, R.M.; Redmond, J.D. Practical guide to using duplex stainless steels. Mater. Perform. 1990, 29, 57–62. [Google Scholar]
- Kiesheyer, H. Proceedings of the International Conference on Stainless Steels’91; The Iron and Steel Institute of Japan: Chiba, Japan, 1991; p. 1148. [Google Scholar]
- Fruytier, D.J. Industrial experiences with duplex stainless steel related to their specific properties. Duplex Stainl. Steels 1991, 1, 497–509. [Google Scholar]
- Foct, J.; Akdut, N.; Gottstein, G. Why are “duplex” microstructures easier to form than expected? Scr. Metall. Mater. 1992, 27, 1033–1038. [Google Scholar] [CrossRef]
- Sridhar, N.; Kolts, J.; Flasche, L.H. A duplex stainless steel for chloride environments. JOM 1985, 37, 31–35. [Google Scholar] [CrossRef]
- Magnin, T.; Lardon, J.M. Cyclic deformation mechanisms of a two-phase stainless steel in various environmental conditions. Mater. Sci. Eng. A 1988, 104, 21–28. [Google Scholar] [CrossRef]
- Bernhardsson, S.; Oredsson, J.; Martenson, C. Duplex Stainless Steels; American Society for Metals: Metals Park, OH, USA, 1983; p. 267. [Google Scholar]
- Solomon, H.D.; Devine, T.M., Jr. Duplex Stainless Steels—A Tale of Two Phases; American Society for Metals: Metals Park, OH, USA, 1982; pp. 693–756. [Google Scholar]
- Örnek, C.; Zhong, X.; Engelberg, D.L. Low-Temperature environmentally assisted cracking of grade 2205 duplex stainless steel beneath a MgCl2: FeCl3 salt droplet. Corrosion 2016, 72, 384–399. [Google Scholar] [CrossRef]
- Nilsson, J.O. Super duplex stainless steels. Mater. Sci. Technol. 1992, 8, 685–700. [Google Scholar] [CrossRef]
- Ohmori, Y.; Nakai, K.; Ohtsubo, H.; Isshiki, Y. Mechanism of Widmanstätten austenite formation in a δ/γ duplex phase stainless steel. ISIJ Int. 1995, 35, 969–975. [Google Scholar] [CrossRef]
- Shek, C.H.; Shen, G.J.; Lai, J.K.L.; Duggan, B.J. Early stages of decomposition of ferrite in duplex stainless steel. Mater. Sci. Technol. 1994, 10, 306–311. [Google Scholar] [CrossRef]
- Chen, T.H.; Yang, J.R. Effects of solution treatment and continuous cooling on σ-phase precipitation in a 2205 duplex stainless steel. Mater. Sci. Eng. A 2001, 311, 28–41. [Google Scholar] [CrossRef]
- Redjaimia, A.; Ruterana, P.; Metauer, G.; Gantois, M. Identification and characterization of a novel intermetallic compound in a Fe-22 wt% Cr-5 wt% Ni-3 wt% Mo-0.03 wt% C duplex stainless steel. Philos. Mag. A 1993, 67, 1277–1286. [Google Scholar] [CrossRef]
- Redjaïmia, A.; Morniroli, J.P.; Donnadieu, P.; Metauer, G. Microstructural and analytical study of heavily faulted Frank-Kasper R-phase precipitates in the ferrite of a duplex stainless steel. J. Mater. Sci. 2002, 37, 4079–4091. [Google Scholar] [CrossRef]
- Martins, M.; Casteletti, L.C. Sigma phase morphologies in cast and aged super duplex stainless steel. Mater. Charact. 2009, 60, 792–795. [Google Scholar] [CrossRef]
- Ghosh, S.K.; Mondal, S. High temperature ageing behaviour of a duplex stainless steel. Mater. Charact. 2008, 59, 1776–1783. [Google Scholar] [CrossRef]
- Escriba, D.M.; Materna-Morris, E.; Plaut, R.L.; Padilha, A.F. Chi-phase precipitation in a duplex stainless steel. Mater. Charact. 2009, 60, 1214–1219. [Google Scholar] [CrossRef]
- Andrews, K.W.; Brookes, P.E. Chi Phase in Alloy Steels. Met. Treat. Drop Forg. 1951, 18, 301. [Google Scholar]
- McMullin, J.G.; Reiter, S.F.; Ebeling, D.G. Equilibrium Structures in Fe-Cr-Mo Alloys. Trans. Am. Soc. Met. 1954, 46, 799–811. [Google Scholar]
- Pettersson, N.H.; Lindell, D.; Lindberg, F.; Borgenstam, A. Formation of Chromium Nitride and Intragranular Austenite in a Super Duplex Stainless Steel. Metall. Mater. Trans. A 2019, 50, 5594–5601. [Google Scholar] [CrossRef]
- Muthupandi, V.; Srinivasan, P.B.; Seshadri, S.; Sundaresan, S. Effect of weld metal chemistry and heat input on the structure and properties of duplex stainless steel welds. Mater. Sci. Eng. A 2003, 358, 9–16. [Google Scholar] [CrossRef]
- Weiss, B.F.; Stickler, R. Phase instabilities during high temperature exposure of 316 austenitic stainless steel. Metall. Mater. Trans. B 1972, 3, 851–866. [Google Scholar] [CrossRef]
- Cho, H.S.; Lee, K. Effect of cold working and isothermal aging on the precipitation of sigma phase in 2205 duplex stainless steel. Mater. Charact. 2013, 75, 29–34. [Google Scholar] [CrossRef]
- Oh, Y.J.; Hong, J.H. Nitrogen effect on precipitation and sensitization in cold-worked Type 316L (N) stainless steels. J. Nucl. Mater. 2000, 278, 242–250. [Google Scholar] [CrossRef]
- Nilsson, J.O.; Wilson, A. Influence of isothermal phase transformations on toughness and pitting corrosion of super duplex stainless steel SAF 2507. Mater. Sci. Technol. 1993, 9, 545–554. [Google Scholar] [CrossRef]
- Zhang, B.; Jiang, Z.; Li, H.; Zhang, S.; Feng, H.; Li, H. Precipitation behavior and phase transformation of hyper duplex stainless steel UNS S32707 at nose temperature. Mater. Charact. 2017, 129, 31–39. [Google Scholar] [CrossRef]
- Mishra, M.K.; Rao, A.G.; Sarkar, R.; Kashyap, B.P.; Prabhu, N. Effect of preaging deformation on aging characteristics of 2507 super duplex stainless steel. J. Mater. Eng. Perform. 2016, 25, 374–381. [Google Scholar] [CrossRef]
- Nilsson, J.O.; Wilson, A.; Josefsson, B.; Thorvaldsson, T. Relationship between pitting corrosion, toughness and microstructure for isothermally heat treated super duplex stainless steel. Appl. Stainl. Steel 1992, 1, 280–289. [Google Scholar]
- Simmons, J.W. Overview: High-nitrogen alloying of stainless steels. Mater. Sci. Eng. A 1996, 207, 159–169. [Google Scholar] [CrossRef]
- Lin, K.; Shi, H.Q.; Ma, L.Q.; Ding, Y. The Analysis and Research of Secondary Phases Generated during Isothermal Aging of Duplex Stainless Steels. Appl. Mech. Mater. 2012, 193–194, 411–417. [Google Scholar] [CrossRef]
- Tsai, Y.-T.; Lin, P.-C.; Chen, Y.-W.; Wang, S.-H.; Yang, J.-R. Fatigue behavior and microstructural characteristics of a duplex stainless steel weld metal under vibration-assisted welding. Mater. Sci. Eng. A 2018, 721, 319–327. [Google Scholar] [CrossRef]
- Pettersson, N.; Pettersson, R.F.; Wessman, S. Precipitation of chromium nitrides in the super duplex stainless steel 2507. Metall. Mater. Trans. A 2015, 46, 1062–1072. [Google Scholar] [CrossRef]
- Ramirez, A.J.; Lippold, J.C.; Brandi, S.D. The relationship between chromium nitride and secondary austenite precipitation in duplex stainless steels. Metall. Mater. Trans. A 2003, 34, 1575–1597. [Google Scholar] [CrossRef]




Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lin, P.-C.; Tsai, Y.-T.; Gan, N.-H.; Yang, J.-R.; Wang, S.-H.; Chang, H.-Y.; Lin, T.-R.; Chiu, P.-K. Characteristics of Flakes Stacked Cr2N with Many Domains in Super Duplex Stainless Steel. Crystals 2020, 10, 965. https://doi.org/10.3390/cryst10110965
Lin P-C, Tsai Y-T, Gan N-H, Yang J-R, Wang S-H, Chang H-Y, Lin T-R, Chiu P-K. Characteristics of Flakes Stacked Cr2N with Many Domains in Super Duplex Stainless Steel. Crystals. 2020; 10(11):965. https://doi.org/10.3390/cryst10110965
Chicago/Turabian StyleLin, Po-Chiang, Yu-Ting Tsai, Neng-Hao Gan, Jer-Ren Yang, Shing-Hoa Wang, Horng-Yi Chang, Tzy-Rong Lin, and Po-Kai Chiu. 2020. "Characteristics of Flakes Stacked Cr2N with Many Domains in Super Duplex Stainless Steel" Crystals 10, no. 11: 965. https://doi.org/10.3390/cryst10110965
APA StyleLin, P.-C., Tsai, Y.-T., Gan, N.-H., Yang, J.-R., Wang, S.-H., Chang, H.-Y., Lin, T.-R., & Chiu, P.-K. (2020). Characteristics of Flakes Stacked Cr2N with Many Domains in Super Duplex Stainless Steel. Crystals, 10(11), 965. https://doi.org/10.3390/cryst10110965