Synthesis of Enaminones-Based Benzo[d]imidazole Scaffold: Characterization and Molecular Insight Structure
Abstract
1. Introduction
2. Materials and Methods
- General:
2.1. (E)-1-(1H-Benzo[d]imidazol-2-yl)-3-(dimethylamino)prop-2-en-1-one 1
2.2. (E)-3-(Dimethylamino)-1-(1-methyl-1H-benzo[d]imidazol-2-yl)prop-2-en-1-one 2
2.3. 1-(6-(1H-Benzo[d]imidazol-2-yl)-2-methylpyridin-3-yl)ethan-1-one 3
2.4. (E)-1-(6-(1H-benzo[d]imidazol-2-yl)-2-methylpyridin-3-yl)-3-(dimethylamino)prop-2-en-1-one 4
3. Results and Discussion
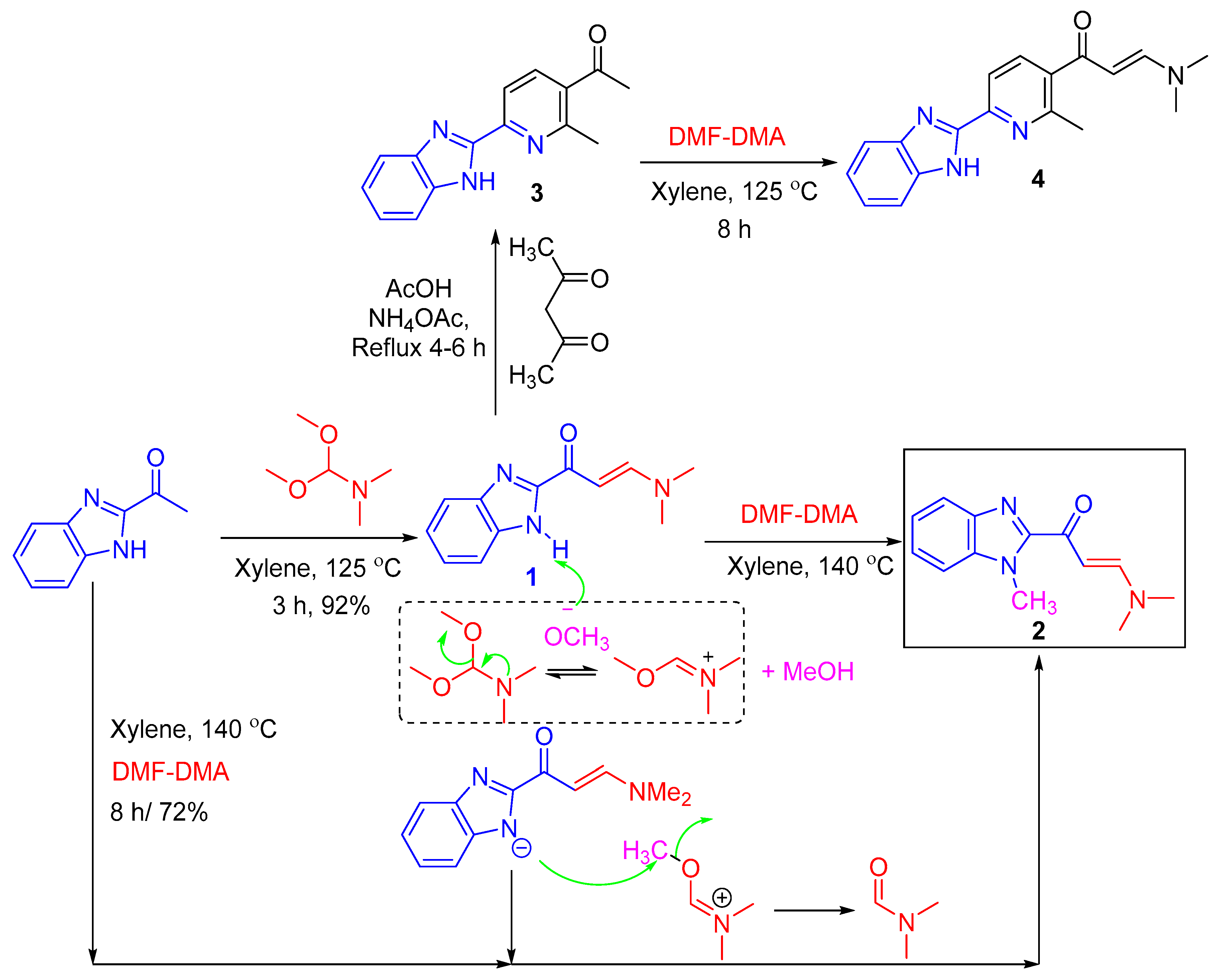
3.1. Synthesis of β-enaminones
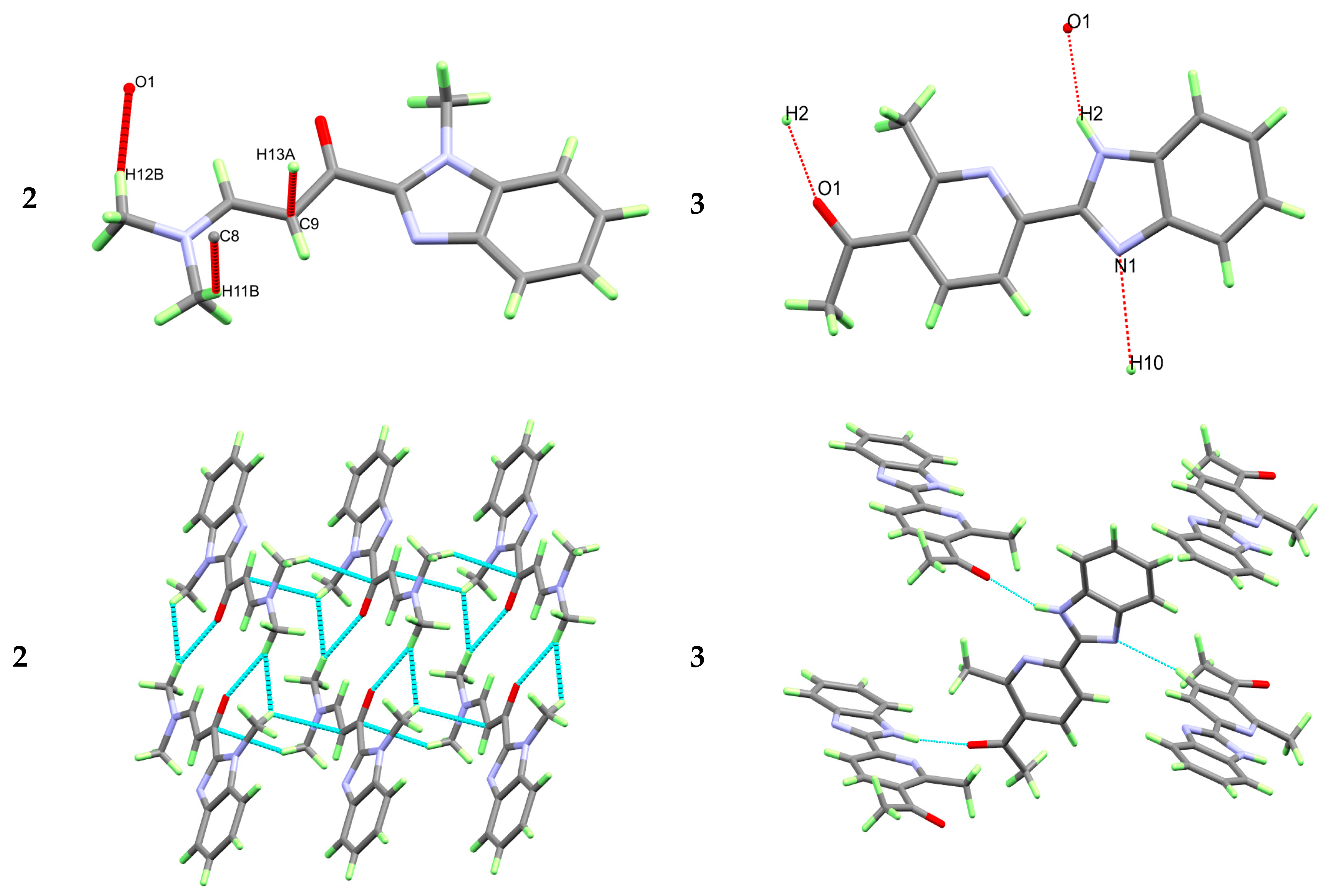
3.2. X-ray Structure
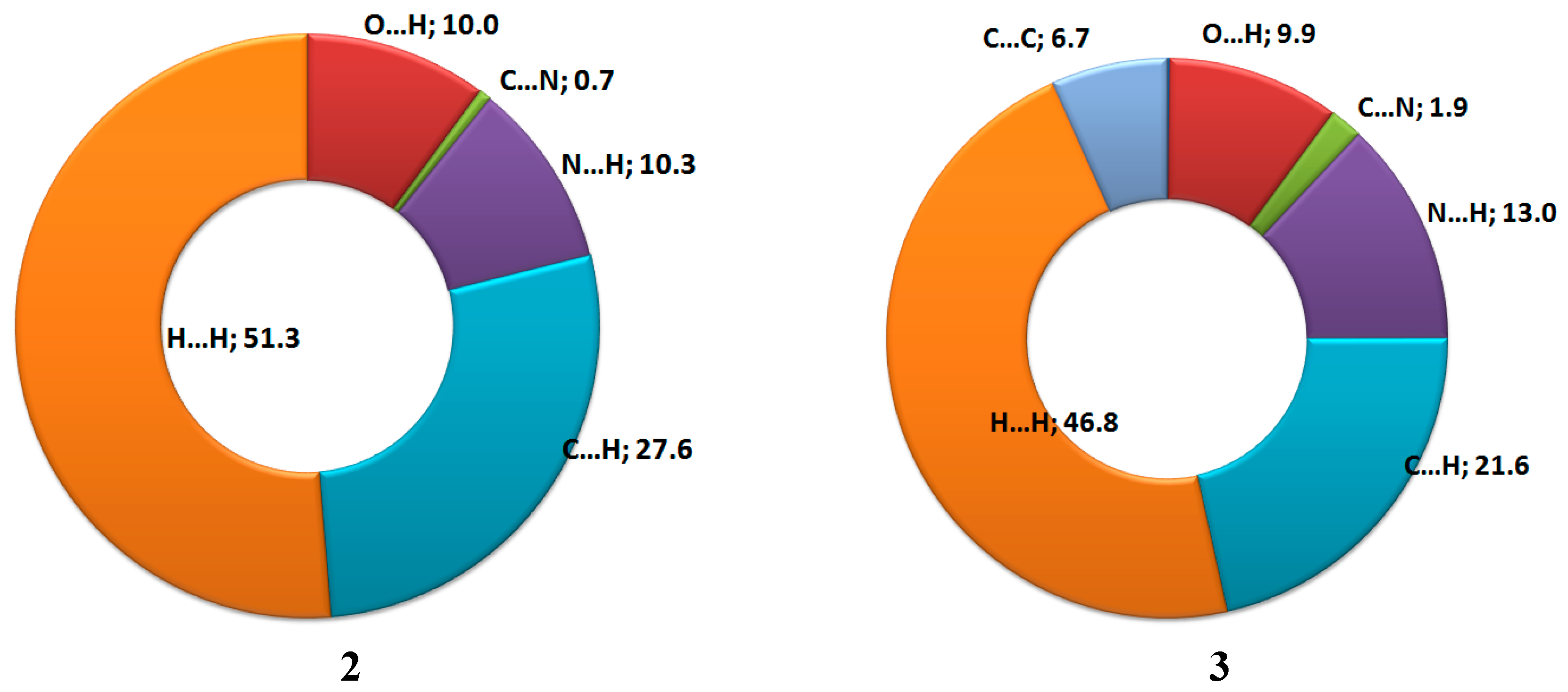
3.3. Hirshfeld Analysis
3.4. DFT Studies
3.5. Reactivity Studies
3.6. NMR Spectra
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Yadav, G.; Ganguly, S. Structure activity relationship (SAR) study of benzimidazole scaffold for different biological activities: A mini-review. Eur. J. Med. Chem. 2015, 97, 419–443. [Google Scholar] [CrossRef] [PubMed]
- Akhtar, W.; Khan, M.F.; Verma, G.; Shaquiquzzaman, M.; Rizvi, M.; Mehdi, S.H.; Akhter, M.; Alam, M.M. Therapeutic evolution of benzimidazole derivatives in the last quinquennial period. Eur. J. Med. Chem. 2017, 126, 705–753. [Google Scholar] [CrossRef] [PubMed]
- Keri, R.S.; Hiremathad, A.; Budagumpi, S.; Nagaraja, B.M. Comprehensive Review in Current Developments of Benzimidazole-Based Medicinal Chemistry. Chem. Biol. Drug Des. 2014, 86, 19–65. [Google Scholar] [CrossRef] [PubMed]
- Francesconi, V.; Cichero, E.; Schenone, S.; Naesens, L.; Tonelli, M. Synthesis and Biological Evaluation of Novel (thio)semicarbazone-Based Benzimidazoles as Antiviral Agents against Human Respiratory Viruses. Molecules 2020, 25, 1487. [Google Scholar] [CrossRef]
- Singu, P.S.; Kanugala, S.; Dhawale, S.A.; Kumar, C.G.; Kumbhare, R.M. Synthesis and Pharmacological Evaluation of Some Amide Functionalized 1 H -Benzo[ d ]imidazole-2-thiol Derivatives as Antimicrobial Agents. ChemistrySelect 2020, 5, 117–123. [Google Scholar] [CrossRef]
- Baldisserotto, A.; Demurtas, M.; Lampronti, I.; Tacchini, M.; Moi, D.; Balboni, G.; Pacifico, S.; Vertuani, S.; Manfredini, S.; Onnis, V. Synthesis and evaluation of antioxidant and antiproliferative activity of 2-arylbenzimidazoles. Bioorg. Chem. 2020, 94, 103396. [Google Scholar] [CrossRef]
- Rahim, F.; Zaman, K.; Taha, M.; Ullah, H.; Ghufran, M.; Wadood, A.; Rehman, W.; Uddin, N.; Shah, S.A.A.; Sajid, M.; et al. Synthesis, in vitro alpha-glucosidase inhibitory potential of benzimidazole bearing bis-Schiff bases and their molecular docking study. Bioorg. Chem. 2020, 94, 103394. [Google Scholar] [CrossRef]
- Son, D.-S.; Lee, E.-S.Y.; Adunyah, S.E. The Antitumor Potentials of Benzimidazole Anthelmintics as Repurposing Drugs. Immune Netw. 2020, 20, e29. [Google Scholar] [CrossRef]
- Zhang, L.; Peng, X.-M.; Damu, G.L.V.; Geng, R.-X.; Zhou, C.-H. Comprehensive Review in Current Developments of Imidazole-Based Medicinal Chemistry. Med. Res. Rev. 2013, 34, 340–437. [Google Scholar] [CrossRef]
- Boiani, M. Imidazole and Benzimidazole Derivatives as Chemotherapeutic Agents. Mini-Rev. Med. Chem. 2005, 5, 409–424. [Google Scholar] [CrossRef]
- Refaat, H.M. Synthesis and anticancer activity of some novel 2-substituted benzimidazole derivatives. Eur. J. Med. Chem. 2010, 45, 2949–2956. [Google Scholar] [CrossRef] [PubMed]
- Taha, M.; Mosaddik, A.; Rahim, F.; Ali, S.; Ibrahim, M.; Almandil, N.B. Synthesis, antiglycation and antioxidant potentials of benzimidazole derivatives. J. King Saud Univ. Sci. 2020, 32, 191–194. [Google Scholar] [CrossRef]
- Vasava, M.S.; Bhoi, M.N.; Rathwa, S.K.; Jethava, D.J.; Acharya, P.T.; Patel, D.B.; Patel, H.D. Benzimidazole: A Milestone in the Field of Medicinal Chemistry. Mini-Rev. Med. Chem. 2020, 20, 532–565. [Google Scholar] [CrossRef] [PubMed]
- Fahim, A.M.; Farag, A.; Shaaban, M.; Ragab, E. Microwave Assisted Synthesis of Pyrazolo[1, 5-a]pyrimidine, Triazolo[1,5-a]pyrimidine, Pyrimido[1,2-a]benzimdazole, Triazolo[5,1-c][1,2,4]triazine and Imidazo[2,1-c][1,2,4]triazine. Curr. Microw. Chem. 2019, 5, 111–119. [Google Scholar] [CrossRef]
- Fahim, A.M.; Shalaby, M.A. Synthesis, biological evaluation, molecular docking and DFT calculations of novel benzenesulfonamide derivatives. J. Mol. Struct. 2019, 1176, 408–421. [Google Scholar] [CrossRef]
- Weng, Y.; Lv, W.; Yu, J.; Ge, B.; Cheng, G. Preparation of 2,4,5-Trisubstituted Oxazoles through Iodine-mediated Aerobic Oxidative Cyclization of Enaminones. Org. Lett. 2018, 20, 1853–1856. [Google Scholar] [CrossRef]
- Thombal, R.S.; Lee, Y.R. Synergistic Indium and Silver Dual Catalysis: A Regioselective [2 + 2 + 1]-Oxidative N-Annulation Approach for the Diverse and Polyfunctionalized N-Arylpyrazoles. Org. Lett. 2018, 20, 4681–4685. [Google Scholar] [CrossRef]
- Apraku, J.; Okoro, C.O. Design, synthesis and anticonvulsant evaluation of fluorinated benzyl amino enaminones. Bioorg. Med. Chem. 2019, 27, 161–166. [Google Scholar] [CrossRef]
- Baldwin, A.G.; Bevan, J.; Brough, D.; Ledder, R.G.; Freeman, S. Synthesis and antibacterial activities of enamine derivatives of dehydroacetic acid. Med. Chem. Res. 2017, 27, 884–889. [Google Scholar] [CrossRef]
- Ali, M.; Barakat, A.; El-Faham, A.; Al-Rasheed, H.H.; Dahlous, K.; Al-Majid, A.M.; Sharma, A.; Yousuf, S.; Sanam, M.; Ul-Haq, Z.; et al. Synthesis and characterisation of thiobarbituric acid enamine derivatives, and evaluation of their α-glucosidase inhibitory and anti-glycation activity. J. Enzym. Inhib. Med. Chem. 2020, 35, 692–701. [Google Scholar] [CrossRef]
- Mabkhot, Y.N.; Barakat, A.; Yousuf, S.; Choudhary, M.I.; Frey, W.; Ben Hadda, T.; Mubarak, M. Substituted thieno[2,3- b ]thiophenes and related congeners: Synthesis, β-glucuronidase inhibition activity, crystal structure, and POM analyses. Bioorg. Med. Chem. 2014, 22, 6715–6725. [Google Scholar] [CrossRef] [PubMed]
- Mabkhot, Y.N.; Aldawsari, F.D.; Al-Showiman, S.S.; Barakat, A.; Soliman, S.M.; Choudhary, M.I.; Yousuf, S.; Mubarak, M.S.; Ben Hadda, T. Novel enaminone derived from thieno [2,3-b] thiene: Synthesis, x-ray crystal structure, HOMO, LUMO, NBO analyses and biological activity. Chem. Cent. J. 2015, 9, 1–11. [Google Scholar] [CrossRef]
- Sims, G.K.; Sommers, L.E. Biodegradation of pyridine derivatives in soil suspensions. Environ. Toxicol. Chem. 1986, 5, 503–509. [Google Scholar] [CrossRef]
- Lungu, C.; Bratanovici, B.I.; Grigore, M.M.; Antoci, V.; Mangalagiu, I.I.; Mirabela, G.M. Hybrid Imidazole-Pyridine Derivatives: An Approach to Novel Anticancer DNA Intercalators. Curr. Med. Chem. 2020, 27, 154–169. [Google Scholar] [CrossRef] [PubMed]
- Gad, E.M.; Nafie, M.S.; Eltamany, E.H.; Hammad, M.S.A.G.; Barakat, A.; Boraei, A.T. Discovery of New Apoptosis-Inducing Agents for Breast Cancer Based on Ethyl 2-Amino-4,5,6,7-Tetra Hydrobenzo[b]Thiophene-3-Carboxylate: Synthesis, In Vitro, and In Vivo Activity Evaluation. Molecules 2020, 25, 2523. [Google Scholar] [CrossRef] [PubMed]
- Boraei, A.T.; Sarhan, A.A.M.; Yousuf, S.; Barakat, A. Synthesis of a New Series of Nitrogen/Sulfur Heterocycles by Linking Four Rings: Indole; 1,2,4-Triazole; Pyridazine; and Quinoxaline. Molecules 2020, 25, 450. [Google Scholar] [CrossRef]
- Boraei, A.T.; Ghabbour, H.A.; Gomaa, M.S.; El Ashry, E.S.H.; Barakat, A. Synthesis and Anti-Proliferative Assessment of Triazolo-Thiadiazepine and Triazolo-Thiadiazine Scaffolds. Molecules 2019, 24, 4471. [Google Scholar] [CrossRef]
- Al-Majid, A.M.; Islam, M.S.; Atef, S.; El-Senduny, F.F.; Badria, F.; Elshaier, Y.A.; Ali, M.; Barakat, A.; Rahman, A.F.M.M. Synthesis of Pyridine-Dicarboxamide-Cyclohexanone Derivatives: Anticancer and α-Glucosidase Inhibitory Activities and In Silico Study. Molecules 2019, 24, 1332. [Google Scholar] [CrossRef]
- Soliman, S.M.; Barakat, A.; Islam, M.S.; Ghabbour, H.A. Synthesis, Crystal Structure and DFT Studies of a New Dinuclear Ag(I)-Malonamide Complex. Molecules 2018, 23, 888. [Google Scholar] [CrossRef]
- El Ashry, E.S.H.; El Kilany, Y.; Nahas, N.M.; Barakat, A.; Al-Qurashi, N.; Ghabbour, H.A.; Fun, H.-K. Synthesis and Crystal Structures of Benzimidazole-2-thione Derivatives by Alkylation Reactions. Molecules 2015, 21, 12. [Google Scholar] [CrossRef]
- Turner, M.J.; McKinnon, J.J.; Wolff, S.K.; Grimwood, D.J.; Spackman, P.R.; Jayatilaka, D.; Spackman, M.A. Crystal Explorer17 (2017) University of Western Australia. Available online: http://hirshfeldsurface.net (accessed on 12 June 2017).
- Frisch, M.J.; Trucks, G.W.; Schlegel, H.B.; Scuseria, G.E.; Robb, M.A.; Cheeseman, J.R.; Scalmani, G.; Barone, V.; Mennucci, B.; Petersson, G.A.; et al. GAUSSIAN 09; Revision A02; Gaussian Inc.: Wallingford, CT, USA, 2009. [Google Scholar]
- Dennington, R., II; Keith, T.; Millam, J. (Eds.) GaussView; Version 4.1; Semichem Inc.: Shawnee Mission, KS, USA, 2007. [Google Scholar]
- Reed, A.E.; Curtiss, L.A.; Weinhold, F. Intermolecular interactions from a natural bond orbital, donor-acceptor viewpoint. Chem. Rev. 1988, 88, 899–926. [Google Scholar] [CrossRef]
- Marten, B.; Kim, K.; Cortis, C.; Friesner, R.A.; Murphy, R.B.; Ringnalda, M.N.; Sitkoff, D.; Honig, B. New Model for Calculation of Solvation Free Energies: Correction of Self-Consistent Reaction Field Continuum Dielectric Theory for Short-Range Hydrogen-Bonding Effects. J. Phys. Chem. 1996, 100, 11775–11788. [Google Scholar] [CrossRef]
- Tannor, D.J.; Marten, B.; Murphy, R.; Friesner, R.A.; Sitkoff, D.; Nicholls, A.; Honig, B.; Ringnalda, M.; Iii, W.A.G. Accurate First Principles Calculation of Molecular Charge Distributions and Solvation Energies from Ab Initio Quantum Mechanics and Continuum Dielectric Theory. J. Am. Chem. Soc. 1994, 116, 11875–11882. [Google Scholar] [CrossRef]
- Cheeseman, J.R.; Trucks, G.W.; Keith, T.A.; Frisch, M.J. A comparison of models for calculating nuclear magnetic resonance shielding tensors. J. Chem. Phys. 1996, 104, 5497–5509. [Google Scholar] [CrossRef]
- Determann, R.; Dreher, J.; Baumann, K.; Preu, L.; Jones, P.G.; Totzke, F.; Schächtele, C.; Kubbutat, M.H.G.; Kunick, C. 2-Anilino-4-(benzimidazol-2-yl)pyrimidines—A multikinase inhibitor scaffold with antiproliferative activity toward cancer cell lines. Eur. J. Med. Chem. 2012, 53, 254–263. [Google Scholar] [CrossRef]
- Saleh, T.S.; Al-Omar, M.A.; Abdel-Aziz, H.A. One-Pot Synthesis of Enaminones Using Golds Reagent. Lett. Org. Chem. 2010, 7, 483–486. [Google Scholar] [CrossRef]
- Shaaban, M.R.; Saleh, T.S.; Osman, F.H.; Farag, A.M. Regioselective Synthesis of Some Novel Pyrazoles, Isoxazoles, Pyrazolo[3,4-d]pyridazines and Isoxazolo[3,4-d]pyridazines Pendant to Benzimidazole. J. Heterocycl. Chem. 2007, 44, 177. [Google Scholar] [CrossRef]
- Stanovnik, B.; Tisler, M.; Hribar, A.; Barlin, G.; Brown, D. Methylation of heterocyclic compounds containing NH, SH and/or OH groups by means of N,N-dimethylformamide dimethyl acetal. Aust. J. Chem. 1981, 34, 1729–1738. [Google Scholar] [CrossRef]
- Kumar, K.; Hemasunder, G.; Naidu, A.; Dubey, P.K. Alkylation studies on pyrazolyl and isoxazolyl benzimidazoles. Heterocycl. Chem. 2011, 20, 393–398. [Google Scholar]
- Foresman, J.B.; Frisch, Æ. Exploring Chemistry with Electronic Structure Methods, 2nd ed.; Gaussian: Pittsburgh, PA, USA, 1996. [Google Scholar]
- Chang, R. Chemistry, 7th ed.; McGraw-Hill: New York, NY, USA, 2001. [Google Scholar]
- Kosar, B.; Albayrak, C. Spectroscopic investigations and quantum chemical computational study of (E)-4-methoxy-2-[(p-tolylimino)methyl]phenol. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2011, 78, 160–167. [Google Scholar] [CrossRef] [PubMed]
- Koopmans, T.A. Ordering of wave functions and eigenenergies to the individual electrons of an atom. Physica 1933, 1, 104–113. [Google Scholar] [CrossRef]
- Parr, R.G.; Yang, W. Density Functional Theory of Atoms and Molecules, 1st ed.; Oxford University Press: Oxford, UK, 1989. [Google Scholar]
- Parr, R.G.; Szentpály, L.V.; Liu, S. Electrophilicity Index. J. Am. Chem. Soc. 1999, 121, 1922–1924. [Google Scholar] [CrossRef]
- Singh, R.; Kumar, A.; Tiwari, R.; Rawat, P.; Gupta, V. A combined experimental and quantum chemical (DFT and AIM) study on molecular structure, spectroscopic properties, NBO and multiple interaction analysis in a novel ethyl 4-[2-(carbamoyl)hydrazinylidene]-3,5-dimethyl-1H-pyrrole-2-carboxylate and its dimer. J. Mol. Struct. 2013, 1035, 427–440. [Google Scholar] [CrossRef]








| Compound | 2 | 3 |
|---|---|---|
| CCDC code | 1999582 | 2032100 |
| Empirical formula | C13H15N3O | C15H13N3O |
| Formula weight | 229.28 | 251.28 |
| Temperature/K | 104(2) | 120(2) |
| Radiation | CuKα (λ = 1.54178) | CuKα (λ = 1.54178) |
| Crystal system | monoclinic | Orthorhombic |
| Space group | P21/n | P212121 |
| a/Å | 9.8953(3) | 6.82950(10) |
| b/Å | 5.7545(2) | 8.00540(10) |
| c/Å | 21.7891(7) | 22.4779(2) |
| β/° | 100.627(2) | |
| Volume/Å3 | 1219.45(7) | 1228.93(3) |
| Z | 4 | 4 |
| Crystal size/mm3 | 0.170 × 0.140 × 0.080 | 0.271 × 0.203 × 0.089 |
| ρcalcg/cm3 | 1.249 | 1.358 |
| μ/mm−1 | 0.657 | 0.710 |
| F(000) | 488 | 528 |
| 2Θ range for data collection/° | 4.13 to 68.20 | 3.933 to 77.074. |
| Index ranges | −11 ≤ h ≤ 11, −6 ≤ k ≤ 6, −25 ≤ l ≤ 26 | −8 ≤ h ≤ 8, −10 ≤ k ≤ 10, −28 ≤ l ≤ 28 |
| Reflections collected | 8164 | 29119 |
| Goodness-of-fit on F2 | 1.057 | 1.072 |
| Data/restraints/parameters | 2209/0/157 | 2597/0/178 |
| Independent reflections | 2209 [Rint = 0.0334] | 2597 [Rint = 0.0313] |
| Final R indexes [all data] | R1 = 0.0555, wR2 = 0.1352 | R1 = 0.0283, wR2 = 0.0742 |
| Final R indexes [I ≥ 2σ (I)] | R1 = 0.0464, wR2 = 0.1268 | R1 = 0.0288, wR2 = 0.0747 |
| Largest diff. peak/hole/e Å−3 | 0.18/–0.20 | 0.137/–0.217 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alshahrani, S.; Soliman, S.M.; Alamary, A.S.; Al-Majid, A.M.; Haukka, M.; Yousuf, S.; Barakat, A. Synthesis of Enaminones-Based Benzo[d]imidazole Scaffold: Characterization and Molecular Insight Structure. Crystals 2020, 10, 955. https://doi.org/10.3390/cryst10100955
Alshahrani S, Soliman SM, Alamary AS, Al-Majid AM, Haukka M, Yousuf S, Barakat A. Synthesis of Enaminones-Based Benzo[d]imidazole Scaffold: Characterization and Molecular Insight Structure. Crystals. 2020; 10(10):955. https://doi.org/10.3390/cryst10100955
Chicago/Turabian StyleAlshahrani, Saeed, Saied M. Soliman, Abdullah Saleh Alamary, Abdullah Mohammed Al-Majid, Matti Haukka, Sammer Yousuf, and Assem Barakat. 2020. "Synthesis of Enaminones-Based Benzo[d]imidazole Scaffold: Characterization and Molecular Insight Structure" Crystals 10, no. 10: 955. https://doi.org/10.3390/cryst10100955
APA StyleAlshahrani, S., Soliman, S. M., Alamary, A. S., Al-Majid, A. M., Haukka, M., Yousuf, S., & Barakat, A. (2020). Synthesis of Enaminones-Based Benzo[d]imidazole Scaffold: Characterization and Molecular Insight Structure. Crystals, 10(10), 955. https://doi.org/10.3390/cryst10100955









