Use of Growth-Rate/Temperature-Gradient Charts for Defect Engineering in Crystal Growth from the Melt
Abstract
1. Introduction
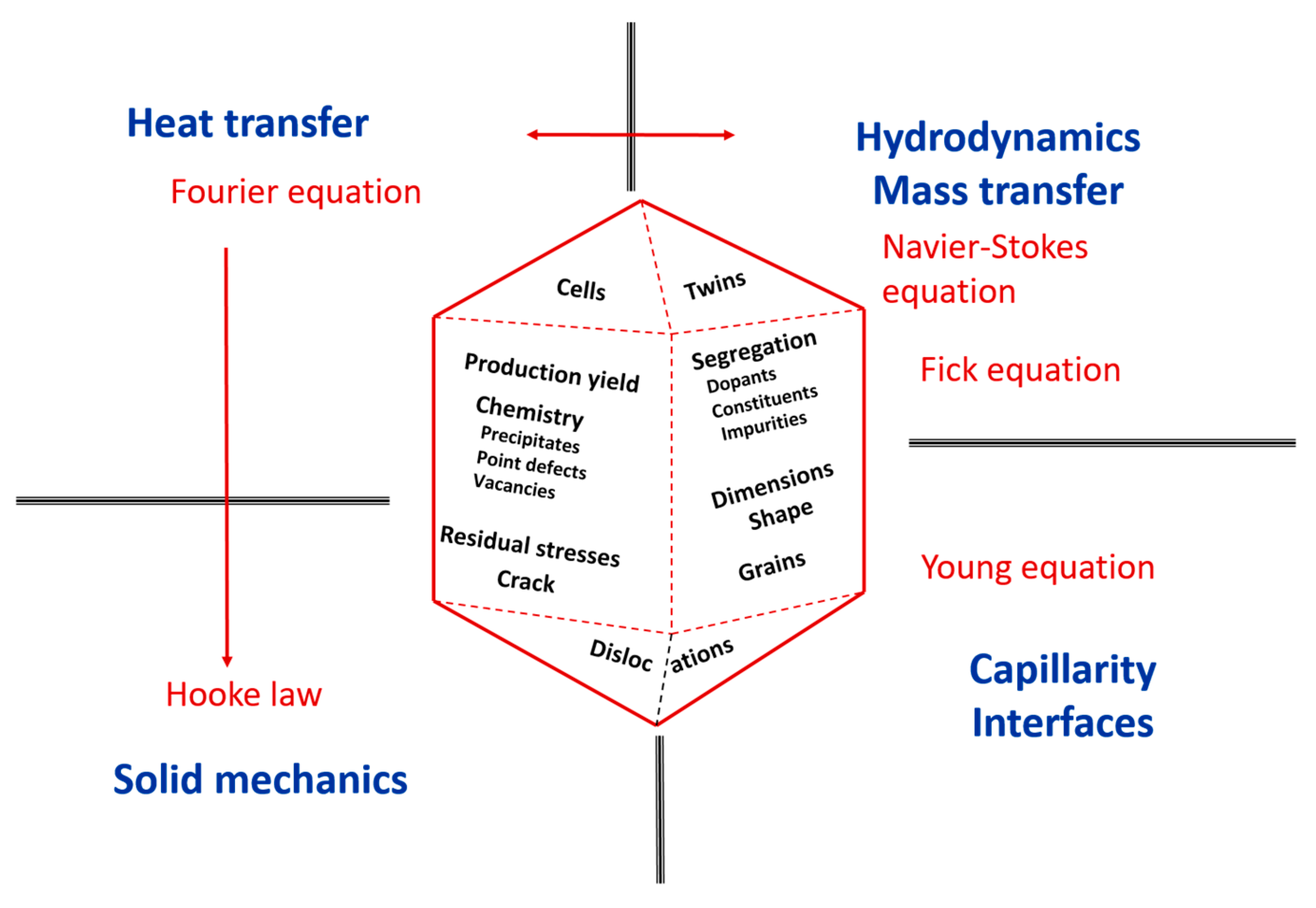
2. Classical Basic Equations Involved in Crystal Growth from the Melt
- -
- Heat transfer, modelled by Fourier’s equation, and, possibly, radiative exchanges. Temperature controls chemical reactions, i.e., with crucibles or casing. Temperature gradients generate thermo-elastic stresses in the solid and convection in the melt. Most importantly, the movement of the solid–liquid (S/L) interface is the movement of an isothermal surface, at least for pure-enough materials.
- -
- Mass transfer modelled by Fick’s laws. It controls chemical homogeneity through species transport in the melt and point defect distribution in the crystal.
- -
- Hydrodynamics, modelled by Navier–Stokes equations. Liquid and gas movements impact heat and mass transfer, which, in turn, generate fluid flows.
- -
- Thermo-mechanical stresses modelled by Hooke’s law. They are likely to produce dislocations, sub-grain boundaries and, in the worst case, cracks.
- -
- Interfaces, first of all the S/L boundary, control the incorporation of constituents, dopants and impurities and, when spurious nucleation occurs, the grain or twin structure. Fluid–fluid interfaces affect the size and shape of the crystal through the fluid menisci used in many capillarity-controlled processes.
2.1. Equations Related to the Process
- -
- Heat balance. This is the most important equation in the field of solidification and is known as the “Stefan condition”, from the name of J. Stefan, who, in the 1880s, derived the sea frozen thickness as a function of the number of freezing days [3]. It is the heat balance at the S/L interface, which simply states that the heat flowing through the solid is the sum of the heat coming from the liquid and the latent heat released by the solidification of the material:
- -
- Cooling rate. It is useful for those processes where solidification is driven by the cooling of a heater, furnace, etc. It is related to other process parameters through:
- -
- Constant growth rate. While never clearly demonstrated theoretically, it is common experience among crystal growers that it is preferable to keep the growth rate constant in order to obtain a crystal of good structural quality.
- -
- Other process-related equations include temperature stability criteria in the case of the crystal shape being controlled by capillarity [4]. Additionally, in a given geometry, the temperature gradient is often constrained by a limit temperature due to evaporation, chemical reactions with the crucible or the atmosphere.
2.2. Equations Related to the Crystal Defects and Structure
- -
- Interface stability. When a solute (dopant, impurity, additional constituent, etc.) is rejected into the melt, Chalmers and co-workers [5] demonstrated that there is a chemically enriched layer at the interface leading to liquid undercooling. If the temperature gradient in the liquid is not large enough, this undercooled liquid tends to solidify so that the solid–liquid interface becomes unstable. The interface remains stable and then can produce a single crystal, when:This behavior is characteristic of multicomponent melts and strongly determines the quality of such alloyed crystals.
- -
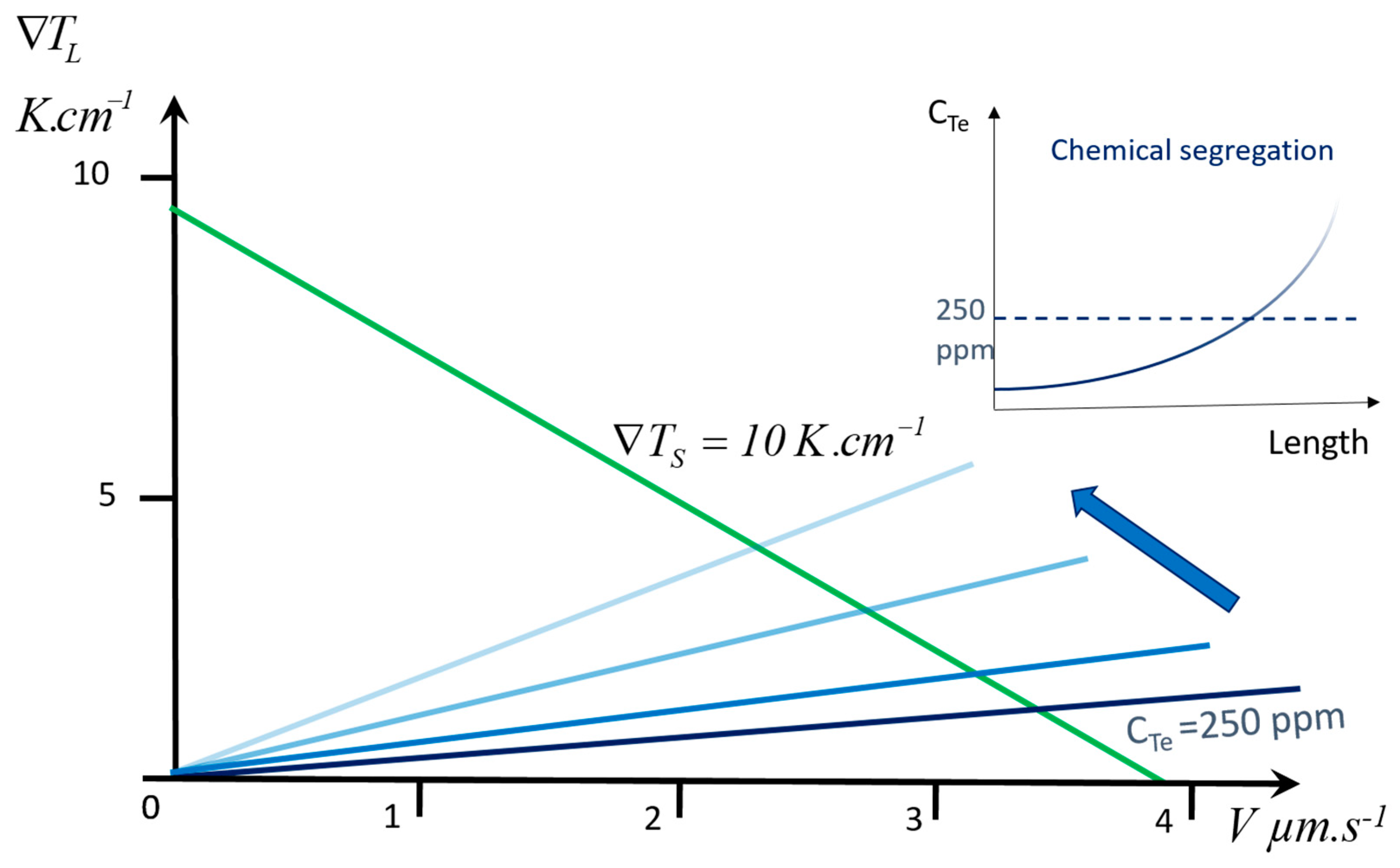
- Chemical segregation. Sheil [6] derived the equation of solute segregation in the case of a finite melt continuously homogenized by mixing. This is, in practice, applicable to most crystal growth processes. It is generally written in the form:Other equations are applicable for some specific processes. In the practical case, is replaced by an “effective” segregation coefficient, , which takes into account the growth rate and the degree of mixing of the melt.
- -
- Stresses and dislocations. Based on estimations of temperature gradients and stresses by Billig [7], the problem of dislocation generation under stresses experienced by the crystal during its growth was actively studied in the 1970s [8]. The accommodation of the deformation exceeding the yield point by an adequate number of dislocations, each providing its Burgers vector, provides a rough estimate of the resulting dislocation density:from which it follows that a crystal without dislocations can be obtained if:
- -
- The growth rate, V, imposed by the pulling shaft or by furnace cooling.
- -
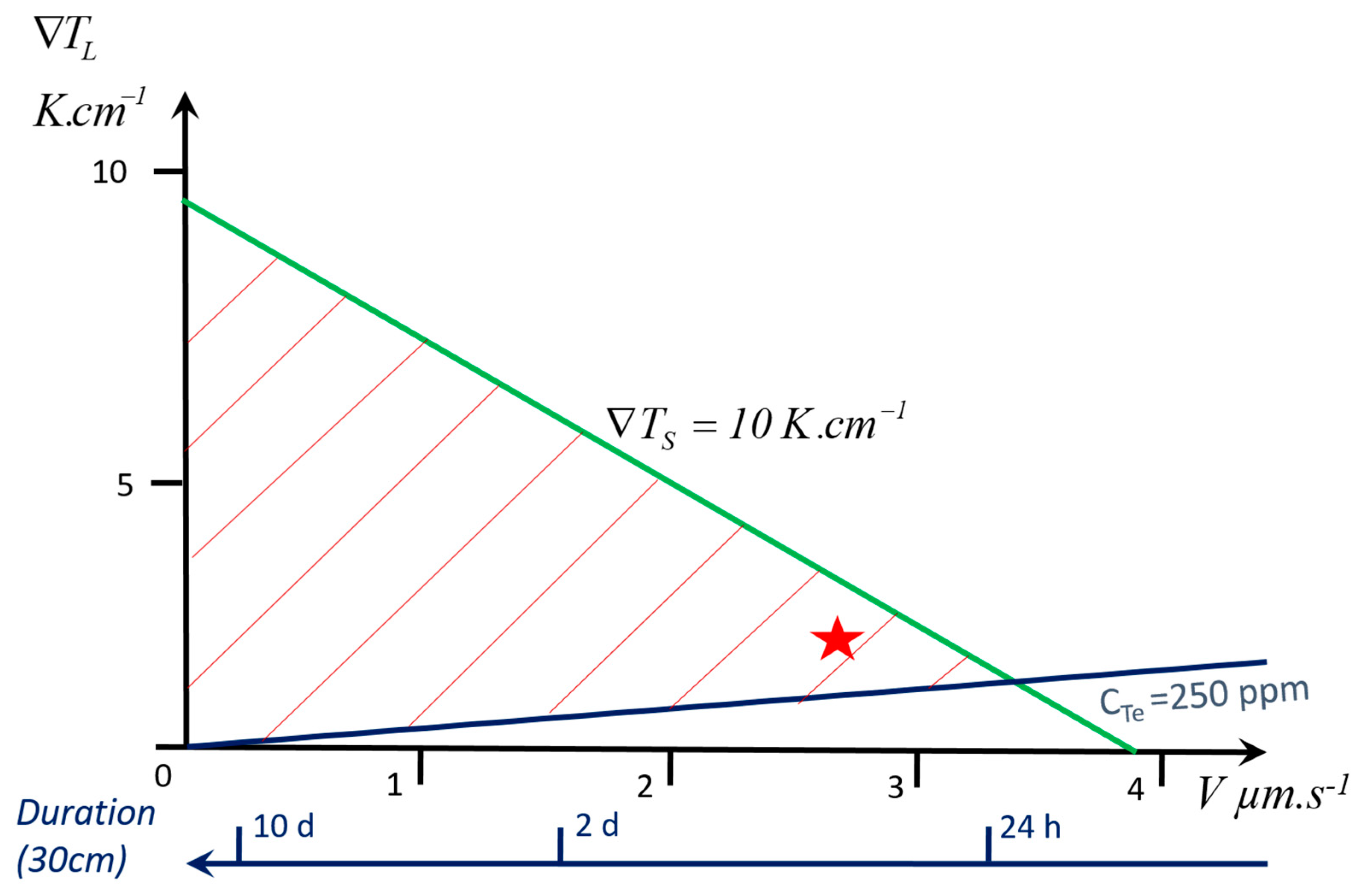
- One of the temperature gradients in the liquid or solid, or , as they are both linked by V in Equation (1). The importance of this parameter is largely underestimated: very few papers on experimental crystal growth provide gradient values, which makes difficult the analysis of the described results. Additionally, they are generally unknown in industrial processes. This is certainly due to the fact that it is very difficult to measure the temperature field in the crystal or in the liquid, which generally requires mockup materials, when available. Alternatively, a numerical simulation of heat transfer in the process should be developed.
- -
- The chemical composition of the material, C0, which is not a free parameter, as it is imposed by the crystal application.
3. Case Study: Growth of GaSb in a Czochralski Setup
4. Development of Chart for Grain Control in Photovoltaic Si Directional Solidification
- -
- Large grains, called “columnar”, as they grow perpendicularly to the S/L interface and extend along the ingot height.
- -
- Some regions where small grains appear. They are called “equiaxed”, as their shape is rather spherical. They generally occur in C-contaminated raw materials, at the top of the ingot, where the solute is likely to be segregated (see Equation (4)).
- -
- Some grains are striped by parallel lines, which, in fact, are a sign of intense twinning.
- -
- This occurs in the faceted zone of the diagram, where the mean temperature of the S/L interface is lower than the Si melting point (as can be seen in Figure 6c): there is an undercooled liquid layer in contact with the S/L interface.
- -
- The carbon is segregated ahead of the S/L interface and, when the solubility limit is reached, precipitates as SiC.
- -
- These SiC particles act as nucleation centers for the undercooled Si just above the S/L interface [15]. Therefore, equiaxed, facetted grains of Si appear and grow ahead of the main, columnar interface.
- -
- There is competition between the equiaxed and the columnar grains, which decides the local equiaxed or columnar structure of the ingot. This was modelled in the same way as equiaxed growth in metallurgy [16], with the difference that both equiaxed and columnar grains are facetted in the case of Si.
5. Conclusions and Perspectives
Funding
Acknowledgments
Conflicts of Interest
Nomenclature
| b | Burgers vector length (m) |
| C0 | Initial concentration of the liquid (mol·m−3) |
| CS | Concentration in the solid (mol·m−3) |
| DL | Diffusion coefficient of a solute in the liquid (m2·s−1) |
| E | Young’s modulus (Pa) |
| f | Solidified fraction |
| k | Segregation coefficient of the solute |
| keff | Effective segregation coefficient |
| kL, ks | Thermal conductivity of liquid or solid, respectively (W·m−1.K−1) |
| mL | Liquidus line slope (K·mol−1·m3) |
| ND | Dislocation density (m·2) |
| R | Crystal radius (m) |
| TDie | Temperature of the die in the EFG process (K) |
| Tm | Melting temperature (K) |
| Cooling rate (K·s−1) | |
| V | Growth rate (m·s−1) |
| ΔT | S/L interface undercooling due to facets (K) |
| ΔHm | Latent heat of solidification (J·kg−1) |
| ∇TL,∇Ts | Temperature gradient in the liquid or solid, respectively (K·m−1) |
| α | Coefficient of thermal expansion (K−1) |
| σCRSS | Yield stress of the crystal (Pa) |
| ρ | Specific mass of liquid (kg·m−3) |
References
- Trivedi, R.; Kurz, W. Microstructure and phase selection diagrams for directionally solidified binary alloys. In Proceedings of the Intelligence Processing Materials, Proceedings Symposium, Fall Meeting of the Minerals, Metals & Materials Society, Indianapolis, Indiana, 2–5 October 1989; Wadley, H.N.G., Eckhart, E.W., Jr., Eds.; 1990; pp. 177–193. [Google Scholar]
- Kurz, W. Solidification microstructure-processing maps: Theory and application. Adv. Eng. Mater. 2001, 3, 443–452. [Google Scholar] [CrossRef]
- Stefan, J. Über die Theorie der Eisbildung. Ann. Phys. 1891, 278, 269–286. [Google Scholar] [CrossRef]
- Duffar, T. Crystal Growth Processes Based on Capillarity: Czochralski, Floating Zone, Shaping and Crucible Techniques; John Wiley & Sons: Chichester, UK, 2010; p. 566. ISBN 978-0-470-71244-3. [Google Scholar]
- Tiller, W.A.; Jackson, K.A.; Rutter, J.W.; Chalmers, B. The redistribution of solute atoms during the solidification of metals. Acta Metall. 1953, 1, 428–437. [Google Scholar] [CrossRef]
- Scheil, E. Bemerkungen zur Schichtkristallbildung. Z. Met. 1942, 34, 70–72. [Google Scholar]
- Billig, E. Some defects in crystals grown from the melt—1. Defect caused by thermal stresses. Proc. R. Soc. Lond. 1956, 235, 37–55. [Google Scholar]
- Indenbom, V.L. Stresses and dislocations in crystal growth. Izv. Nauk Akad. SSR Ser. Fiz. 1973, 37, 2258–2267. [Google Scholar]
- Drosd, R.; Washburn, J. Some observations on the amorphous to crystalline transformation in silicon. J. Appl. Phys. 1982, 53, 397–403. [Google Scholar] [CrossRef][Green Version]
- Duffar, T.; Nadri, A. On the twinning occurrence in bulk semiconductor crystal growth. Scr. Mater. 2010, 62, 955–960. [Google Scholar] [CrossRef]
- Beatty, K.M.; Jackson, K.A. Monte Carlo modeling of silicon crystal growth. J. Cryst. Growth 2000, 211, 13–17. [Google Scholar] [CrossRef]
- Duffar, T.; Nadri, A. The grain–grain–liquid triple phase line during solidification of multi-crystalline silicon. Comptes Rendus Phys. 2013, 14, 185–191. [Google Scholar] [CrossRef]
- Mangelinck-Noël, N.; Duffar, T. Modelling of the transition from a planar faceted front to equiaxed growth: Application to photovoltaic polycrystalline silicon. J. Cryst. Growth 2008, 311, 20–25. [Google Scholar] [CrossRef]
- Beaudhuin, M.; Duffar, T.; Lemiti, M.; Zaidat, K. One-dimensional model of the equiaxed grain formation in multi-crystalline silicon. J. Cryst. Growth 2011, 319, 106–113. [Google Scholar] [CrossRef]
- Beaudhuin, M.; Chichignoud, G.; Bertho, P.; Duffar, T.; Lemiti, M.; Zaidat, K. Carbon reaction with levitated silicon—Experimental and thermodynamic approaches. Mater. Chem. Phys. 2012, 133, 284–288. [Google Scholar] [CrossRef]
- Lipton, J.; Glicksman, M.E.; Kurz, W. Equiaxed dendrite growth in alloys at small undercooling. Metall. Mater. Trans. A 1987, 18A, 341–345. [Google Scholar] [CrossRef]
- Voigt, A.; Wolf, E.; Strunk, H.P. Grain orientation and grain boundaries in cast multicrystalline silicon. Mat. Sci. Eng. 1998, B54, 202–206. [Google Scholar]
- Carroz, L.; Duffar, T. Tuning the sapphire EFG process to the growth of Al2O3/YAG/ZrO2:Y eutectic. J. Cryst. Growth 2018, 489, 5–10. [Google Scholar] [CrossRef]




| α | 7 × 10−6 K−1 |
| E | 8.7 × 104 MPa |
| σCRSS | 0.5 MPa |
| kL | 10.2 W·m−1·K−1 |
| kS | 6.4 W·m−1·K−1 |
| ΔH | 0.2 × 106 J·kg−1 |
| ρ | 6 × 103 kg·m−3 |
| k | 0.3 |
| DL | 0.8 × 10−8 m2·s−1 |
| mL | −2 × 10−4 K·(ppm)−1 |
© 2020 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Duffar, T. Use of Growth-Rate/Temperature-Gradient Charts for Defect Engineering in Crystal Growth from the Melt. Crystals 2020, 10, 909. https://doi.org/10.3390/cryst10100909
Duffar T. Use of Growth-Rate/Temperature-Gradient Charts for Defect Engineering in Crystal Growth from the Melt. Crystals. 2020; 10(10):909. https://doi.org/10.3390/cryst10100909
Chicago/Turabian StyleDuffar, Thierry. 2020. "Use of Growth-Rate/Temperature-Gradient Charts for Defect Engineering in Crystal Growth from the Melt" Crystals 10, no. 10: 909. https://doi.org/10.3390/cryst10100909
APA StyleDuffar, T. (2020). Use of Growth-Rate/Temperature-Gradient Charts for Defect Engineering in Crystal Growth from the Melt. Crystals, 10(10), 909. https://doi.org/10.3390/cryst10100909




