Raman Scattering: From Structural Biology to Medical Applications
Abstract
1. Introduction
2. Background
2.1. Spontaneous Raman Scattering
2.2. Instrumentation
2.3. Surface-Enhanced Raman Scattering
2.4. Coherent Raman Scattering
2.5. Stimulated Raman Scattering (SRS)
2.6. Coherent Anti-Stokes Raman Scattering (CARS)
2.7. Resonance Raman Scattering (RRS)
2.8. Raman Microscopy
3. Raman Scattering in Structural Biology and Cell Biophysics
3.1. RS of Lipids and Lipid Structures
3.2. RS of Proteins, Protein Interactions and Dynamics
3.3. Conformational Changes of Proteins
3.4. Pigments are Unique Proteins’ Ligands for RS Studies
3.5. CARS Imaging of Membrane Protein Crystals
4. Raman Spectroscopy in DNA Structural Investigations
SERS in Nucleic Acids Detection
5. Raman Scattering Applications for Cancer Research and Diagnostics
SERS in Cancer Research and Diagnostics
6. RS and SERS for Other Biomedical Applications
6.1. RS in Biomedicine and Diagnostics
6.2. SERS in Biomedicine and Diagnostics
6.2.1. SERS-Sensors/Nanoprobes
6.2.2. SERS-Detectors
6.2.3. Label-Free SERS Studies of Living Cells and Biological Liquids
7. Raman Scattering for Cell Imaging
8. Raman Spectroscopy of Photoactive Proteins
8.1. RS of Microbial Rhodopsins
8.2. RS of Type-2 Microbial Rhodopsins, Bilin- and Flavin-Bound Photoreceptors and Artificial Near-Infrared Rhodopsins
8.3. Optogenetics and Physiology in Terms of Raman Spectroscopy Applications
9. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| CARS | coherent anti-Stokes Raman scattering |
| DCNN | deep convolutional neural network |
| DFA | discriminant function analysis |
| EM | electron microscopy |
| LCP | lipidic cubic phase |
| PCA | principal component analysis |
| P-CARS | Polarized CARS |
| RM | Raman microscopy |
| RS | Raman spectroscopy |
| RRS | resonance Raman spectroscopy |
| SEM | scanning electron microscopy |
| SERS | surface-enhanced Raman scattering |
| SHG | second harmonic generation |
| SONICC | second order nonlinear imaging of chiral crystals |
| SRS | stimulated Raman scattering |
| TEM | transmission electron microscopy |
| TERS | tip-enhanced Raman spectroscopy |
| TPEF | two-photon excited autofluorescence |
| XRD | X-ray diffraction |
| XFEL | X-ray free electron laser |
References
- Smekal, A. Zur Quantentheorie der Dispersion. Naturwissenschaften 1923, 11, 873–875. [Google Scholar] [CrossRef]
- Raman, C.V. A change of wave-length in light scattering. Nature 1928, 121, 619. [Google Scholar] [CrossRef]
- Landsberg, G.; Mandelstam, L. Über die Lichtzerstreuung in Kristallen. Zeitschrift für Phys. 1928, 50, 769–780. [Google Scholar] [CrossRef]
- Born, M.; Bradburn, M. The theory of the Raman effect in crystals, in particular rocksalt. Proc. R. Soc. Lond. A Math. Phys. Sci. 1947, 188, 161–178. [Google Scholar] [PubMed]
- Kashima, T.; Watari, T.; Mikubo, H. Raman spectra and structure of molecular addition compound of aminopyrine and barbital in aqueous solution. Eisei Shikenjo Hokoku 1961, 79, 59–63. [Google Scholar] [PubMed]
- Tobin, M.C. Raman spectra of crystalline lysozyme, pepsin, and alpha chymotrypsin. Science 1968, 161, 68–69. [Google Scholar] [CrossRef] [PubMed]
- Lord, R.C.; Yu, N.T. Laser-excited Raman spectroscopy of biomolecules. II. Native ribonuclease and α-chymotrypsin. J. Mol. Biol. 1970, 51, 203–213. [Google Scholar] [CrossRef]
- Rimai, L.; Kilponen, R.G.; Gill, D. Resonance-enhanced raman spectra of visual pigments in intact bovine retinas at low temperatures. Biochem. Biophys. Res. Commun. 1970, 41, 492–497. [Google Scholar] [CrossRef]
- Long, T.V.; Loehr, T.M. Possible determination of iron coordination in nonheme iron proteins using laser-Raman spectroscopy. Rubredoxin. J. Am. Chem. Soc. 1970, 92, 6384–6386. [Google Scholar] [CrossRef]
- Kissick, D.J.; Wanapun, D.; Simpson, G.J. Second-order nonlinear optical imaging of chiral crystals. Annu. Rev. Anal. Chem. 2011, 4, 419–437. [Google Scholar] [CrossRef]
- Ray, L.B. Silencing neurons using optogenetics. Science 2015, 349, 598. [Google Scholar] [CrossRef]
- Shevchenko, V.; Gushchin, I.; Polovinkin, V.; Kovalev, K.; Balandin, T.; Borshchevskiy, V.; Gordeliy, V. Sodium and engineered potassium light-driven pumps. In Optogenetics: From Neuronal Function to Mapping and Disease Biology; Cambridge University Press: Cambridge, UK, 2017; pp. 79–92. ISBN 9-78110-728-1875. [Google Scholar]
- Barnett, S.C.; Perry, B.A.L.; Dalrymple-Alford, J.C.; Parr-Brownlie, L.C. Optogenetic stimulation: Understanding memory and treating deficits. Hippocampus 2018, 28, 457–470. [Google Scholar] [CrossRef] [PubMed]
- Chang, R.B. Optogenetic Control of the Peripheral Nervous System. Cold Spring Harb. Perspect. Med. 2019, 5, a034397. [Google Scholar] [CrossRef] [PubMed]
- Ostrovsky, M.A.; Kirpichnikov, M.P. Prospects of Optogenetic Prosthesis of the Degenerative Retina of the Eye. Biochemistry 2019, 84, 479–490. [Google Scholar] [CrossRef]
- Boyle, P.M.; Karathanos, T.V.; Trayanova, N.A. Cardiac Optogenetics 2018. JACC Clin. Electrophysiol. 2018, 4, 155–167. [Google Scholar] [CrossRef]
- Johnson, H.E.; Toettcher, J.E. Illuminating developmental biology with cellular optogenetics. Curr. Opin. Biotechnol. 2018, 52, 42–48. [Google Scholar] [CrossRef]
- Deiters, A. Special Issue on Optochemical and Optogenetic Control of Cellular Processes. ChemBioChem 2018, 19, 1198–1200. [Google Scholar] [CrossRef]
- Shevchenko, V.; Mager, T.; Kovalev, K.; Polovinkin, V.; Alekseev, A.; Juettner, J.; Chizhov, I.; Bamann, C.; Vavourakis, C.; Ghai, R.; et al. Inward H+ pump xenorhodopsin: Mechanism and alternative optogenetic approach. Sci. Adv. 2017, 3, e1603187. [Google Scholar] [CrossRef]
- Althaus, T.; Eisfeld, W.; Lohrmann, R.; Stockburger, M. Application of Raman Spectroscopy to Retinal Proteins. Isr. J. Chem. 1995, 35, 227–251. [Google Scholar] [CrossRef]
- Singh, N.; Kumar, P.; Riaz, U. Applications of near infrared and surface enhanced Raman scattering techniques in tumor imaging: A short review. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2019, 222, 117279. [Google Scholar] [CrossRef]
- Kast, R.E.; Serhatkulu, G.K.; Cao, A.; Pandya, A.K.; Dai, H.; Thakur, J.S.; Naik, V.M.; Naik, R.; Klein, M.D.; Auner, G.W.; et al. Raman spectroscopy can differentiate malignant tumors from normal breast tissue and detect early neoplastic changes in a mouse model. Biopolymers 2008, 89, 235–241. [Google Scholar] [CrossRef] [PubMed]
- Aljakouch, K.; Hilal, Z.; Daho, I.; Schuler, M.; Krauß, S.D.; Yosef, H.K.; Dierks, J.; Mosig, A.; Gerwert, K.; El-Mashtoly, S.F. Fast and Noninvasive Diagnosis of Cervical Cancer by Coherent Anti-Stokes Raman Scattering. Anal. Chem. 2019, 91, 13900–13906. [Google Scholar] [CrossRef] [PubMed]
- Gniadecka, M.; Philipsen, P.A.; Sigurdsson, S.; Wessel, S.; Nielsen, O.F.; Christensen, D.H.; Hercogova, J.; Rossen, K.; Thomsen, H.K.; Gniadecki, R.; et al. Melanoma Diagnosis by Raman Spectroscopy and Neural Networks: Structure Alterations in Proteins and Lipids in Intact Cancer Tissue. J. Investig. Dermatol. 2004, 122, 443–449. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.; Jha, H.C. Optical Imaging with Signal Processing for Non-Invasive Diagnosis in Gastric Cancer: Nonlinear Optical Microscopy Modalities. In Advances in Intelligent Systems and Computing; Springer: Berlin, Germany, 2019; Volume 748, pp. 609–619. [Google Scholar]
- Liang, L.; Huang, D.; Wang, H.; Li, H.; Xu, S.; Chang, Y.; Li, H.; Yang, Y.-W.; Liang, C.; Xu, W. In Situ Surface-Enhanced Raman Scattering Spectroscopy Exploring Molecular Changes of Drug-Treated Cancer Cell Nucleus. Anal. Chem. 2015, 87, 2504–2510. [Google Scholar] [CrossRef] [PubMed]
- El-Mashtoly, S.F.; Yosef, H.K.; Petersen, D.; Mavarani, L.; Maghnouj, A.; Hahn, S.; Kötting, C.; Gerwert, K. Label-Free Raman Spectroscopic Imaging Monitors the Integral Physiologically Relevant Drug Responses in Cancer Cells. Anal. Chem. 2015, 87, 7297–7304. [Google Scholar] [CrossRef] [PubMed]
- Deng, R.; Qu, H.; Liang, L.; Zhang, J.; Zhang, B.; Huang, D.; Xu, S.; Liang, C.; Xu, W. Tracing the Therapeutic Process of Targeted Aptamer/Drug Conjugate on Cancer Cells by Surface-Enhanced Raman Scattering Spectroscopy. Anal. Chem. 2017, 89, 2844–2851. [Google Scholar] [CrossRef]
- Prado, E.; Colin, A.; Servant, L.; Lecomte, S. SERS Spectra of Oligonucleotides as Fingerprints to Detect Label-Free RNA in Microfluidic Devices. J. Phys. Chem. C 2014, 118, 13965–13971. [Google Scholar] [CrossRef]
- Swierczewska, M.; Liu, G.; Lee, S.; Chen, X. High-sensitivity nanosensors for biomarker detection. Chem. Soc. Rev. 2012, 41, 2641–2655. [Google Scholar] [CrossRef]
- Kankia, B.I.; Marky, L.A. Folding of the Thrombin Aptamer into a G-Quadruplex with Sr2+: Stability, Heat, and Hydration. J. Am. Chem. Soc. 2001, 123, 10799–10804. [Google Scholar] [CrossRef]
- Aubrey, K.L.; Thomas, G.J.; Casjens, S.R. Secondary Structure and Interactions of the Packaged Dsdna Genome of Bacteriophage P22 Investigated by Raman Difference Spectroscopy. Biochemistry 1992, 31, 11835–11842. [Google Scholar] [CrossRef]
- Dong, R.; Yan, X.; Pang, X.; Liu, S. Temperature-dependent Raman spectra of collagen and DNA. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2004, 60, 557–561. [Google Scholar] [CrossRef]
- Morla-Folch, J.; Xie, H.; Alvarez-Puebla, R.A.; Guerrini, L. Fast Optical Chemical and Structural Classification of RNA. ACS Nano 2016, 10, 2834–2842. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Rico, E.; Alvarez-Puebla, R.A.; Guerrini, L. Direct surface-enhanced Raman scattering (SERS) spectroscopy of nucleic acids: From fundamental studies to real-life applications. Chem. Soc. Rev. 2018, 47, 4909–4923. [Google Scholar] [CrossRef] [PubMed]
- López-Peña, I.; Leigh, B.S.; Schlamadinger, D.E.; Kim, J.E. Insights into Protein Structure and Dynamics by Ultraviolet and Visible Resonance Raman Spectroscopy. Biochemistry 2015, 54, 4770–4783. [Google Scholar] [CrossRef] [PubMed]
- Jones, R.R.; Hooper, D.C.; Zhang, L.; Wolverson, D.; Valev, V.K. Raman Techniques: Fundamentals and Frontiers. Nanoscale Res. Lett. 2019, 14, 1–34. [Google Scholar] [CrossRef] [PubMed]
- Fang, C.; Wu, G. Temporal electronic structures of nonresonant Raman excited virtual states: A case study of ethylene thiourea. J. Raman Spectrosc. 2007, 38, 1416–1420. [Google Scholar] [CrossRef]
- Le Ru, E.C.; Blackie, E.; Meyer, M.; Etchegoin, P.G. Surface Enhanced Raman Scattering Enhancement Factors: A Comprehensive Study. J. Phys. Chem. C 2007, 111, 13794–13803. [Google Scholar] [CrossRef]
- Ding, S.Y.; You, E.M.; Tian, Z.Q.; Moskovits, M. Electromagnetic theories of surface-enhanced Raman spectroscopy. Chem. Soc. Rev. 2017, 46, 4042–4076. [Google Scholar] [CrossRef]
- Lombardi, J.R.; Birke, R.L. A Unified View of Surface-Enhanced Raman Scattering. Acc. Chem. Res. 2009, 42, 734–742. [Google Scholar] [CrossRef]
- Cardinal, M.F.; Vander Ende, E.; Hackler, R.A.; McAnally, M.O.; Stair, P.C.; Schatz, G.C.; Van Duyne, R.P. Expanding applications of SERS through versatile nanomaterials engineering. Chem. Soc. Rev. 2017, 46, 3886–3903. [Google Scholar] [CrossRef]
- Pilot, R.; Signorini, R.; Fabris, L. Surface-enhanced Raman spectroscopy: Principles, substrates, and applications. In Metal Nanoparticles and Clusters: Advances in Synthesis, Properties and Applications; Springer International Publishing: Berlin, Germany, 2017; pp. 89–164. ISBN 9-78331-968-0538. [Google Scholar]
- Muniz-Miranda, M.; Muniz-Miranda, F.; Pedone, A. Spectroscopic and computational studies on ligand-capped metal nanoparticles and clusters. In Metal Nanoparticles and Clusters: Advances in Synthesis, Properties and Applications; Springer International Publishing: Berlin, Germany, 2017; pp. 55–87. ISBN 9-78331-968-0538. [Google Scholar]
- Sonntag, M.D.; Pozzi, E.A.; Jiang, N.; Hersam, M.C.; Van Duyne, R.P. Recent Advances in Tip-Enhanced Raman Spectroscopy. J. Phys. Chem. Lett. 2014, 5, 3125–3130. [Google Scholar] [CrossRef] [PubMed]
- Shao, F.; Zenobi, R. Tip-enhanced Raman spectroscopy: Principles, practice, and applications to nanospectroscopic imaging of 2D materials. Anal. Bioanal. Chem. 2019, 411, 37–61. [Google Scholar] [CrossRef] [PubMed]
- Cui, M.; Bachler, B.R.; Nichols, S.R.; Ogilvie, J.P. Comparing coherent and spontaneous Raman scattering under biological imaging conditions. Opt. Lett. 2009, 34, 773–775. [Google Scholar] [CrossRef] [PubMed]
- Boudon, V.; Bermejo, D.; Martínez, R.Z. High-resolution stimulated Raman spectroscopy and analysis of the ν1, 2ν1—ν1, ν2, 2ν2, and 3ν2—ν2 bands of CF4. J. Raman Spectrosc. 2013, 44, 731–738. [Google Scholar] [CrossRef]
- Nandakumar, P.; Kovalev, A.; Volkmer, A. Vibrational imaging Based on stimulated Raman scattering microscopy. New J. Phys. 2009, 11, 033026. [Google Scholar] [CrossRef]
- Volkmer, A.; Cheng, J.-X.; Sunney Xie, X. Vibrational Imaging with High Sensitivity via Epidetected Coherent Anti-Stokes Raman Scattering Microscopy. Phys. Rev. Lett. 2001, 87, 023901. [Google Scholar] [CrossRef]
- Lu, F.; Zheng, W.; Huang, Z. Coherent anti-Stokes Raman scattering microscopy using tightly focused radially polarized light. Opt. Lett. 2009, 34, 1870. [Google Scholar] [CrossRef]
- Parekh, S.H.; Lee, Y.J.; Aamer, K.A.; Cicerone, M.T. Label-free cellular imaging by Broadband coherent anti-stokes raman scattering microscopy. Biophys. J. 2010, 99, 2695–2704. [Google Scholar] [CrossRef]
- Ren, L.; Hurwitz, I.; Raanan, D.; Oulevey, P.; Oron, D.; Silberberg, Y. Terahertz coherent anti-Stokes Raman scattering microscopy. Optica 2019, 6, 52. [Google Scholar] [CrossRef]
- Shen, Y.; Wang, J.; Wang, K.; Sokolov, A.V.; Scully, M.O. Wide-field coherent anti-Stokes Raman scattering microscopy based on picosecond supercontinuum source. APL Photonics 2018, 3, 116104. [Google Scholar] [CrossRef]
- Tipping, W.J.; Lee, M.; Serrels, A.; Brunton, V.G.; Hulme, A.N. Stimulated Raman scattering microscopy: An emerging tool for drug discovery. Chem. Soc. Rev. 2016, 45, 2075–2089. [Google Scholar] [CrossRef] [PubMed]
- Potma, E.O.; Xie, X.S. CARS Microscopy for Biology and Medicine. Opt. Photonics News 2004, 15, 40. [Google Scholar] [CrossRef]
- Ichimura, T.; Hayazawa, N.; Hashimoto, M.; Inouye, Y.; Kawata, S. Local enhancement of coherent anti-Stokes Raman scattering by isolated gold nanoparticles. J. Raman Spectrosc. 2003, 34, 651–654. [Google Scholar] [CrossRef]
- Steuwe, C.; Kaminski, C.F.; Baumberg, J.J.; Mahajan, S. Surface Enhanced Coherent Anti-Stokes Raman Scattering on Nanostructured Gold Surfaces. Nano Lett. 2011, 11, 5339–5343. [Google Scholar] [CrossRef]
- Frontiera, R.R.; Henry, A.I.; Gruenke, N.L.; Van Duyne, R.P. Surface-enhanced femtosecond stimulated Raman spectroscopy. J. Phys. Chem. Lett. 2011, 2, 1199–1203. [Google Scholar] [CrossRef]
- Prince, R.C.; Frontiera, R.R.; Potma, E.O. Stimulated Raman scattering: From bulk to nano. Chem. Rev. 2017, 117, 5070–5094. [Google Scholar] [CrossRef]
- Robert, B. Resonance Raman spectroscopy. Photosynth. Res. 2009, 101, 147–155. [Google Scholar] [CrossRef]
- Antonio, K.A.; Schultz, Z.D. Advances in biomedical raman microscopy. Anal. Chem. 2014, 86, 30–46. [Google Scholar] [CrossRef]
- Freudiger, C.W.; Min, W.; Saar, B.G.; Lu, S.; Holtom, G.R.; He, C.; Tsai, J.C.; Kang, J.X.; Xie, X.S. Label-free biomedical imaging with high sensitivity by stimulated raman scattering microscopy. Science 2008, 322, 1857–1861. [Google Scholar] [CrossRef]
- Evans, C.L.; Xie, X.S. Coherent Anti-Stokes Raman Scattering Microscopy: Chemical Imaging for Biology and Medicine. Annu. Rev. Anal. Chem. 2008, 1, 883–909. [Google Scholar] [CrossRef]
- Zheng, X.S.; Jahn, I.J.; Weber, K.; Cialla-May, D.; Popp, J. Label-free SERS in biological and biomedical applications: Recent progress, current challenges and opportunities. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2018, 197, 56–77. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Huang, S.C.; Huang, T.X.; Su, H.S.; Zhong, J.H.; Zeng, Z.C.; Li, M.H.; Ren, B. Tip-enhanced Raman spectroscopy for surfaces and interfaces. Chem. Soc. Rev. 2017, 46, 4020–4041. [Google Scholar] [CrossRef] [PubMed]
- Rostron, P.; Gaber, S.; Gaber, D. Raman spectroscopy, review. Laser 2016, 21, 24. [Google Scholar]
- Krafft, C.; Neudert, L.; Simat, T.; Salzer, R. Near infrared Raman spectra of human brain lipids. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2005, 61, 1529–1535. [Google Scholar] [CrossRef] [PubMed]
- Brazhe, N.A.; Nikelshparg, E.I.; Prats, C.; Dela, F.; Sosnovtseva, O. Raman probing of lipids, proteins, and mitochondria in skeletal myocytes: A case study on obesity. J. Raman Spectrosc. 2017, 48, 1158–1165. [Google Scholar] [CrossRef]
- Wu, H.; Volponi, J.V.; Oliver, A.E.; Parikh, A.N.; Simmons, B.A.; Singh, S. In vivo lipidomics using single-cell Raman spectroscopy. Proc. Natl. Acad. Sci. USA 2011, 108, 3809–3814. [Google Scholar] [CrossRef]
- Wampler, R.D.; Kissick, D.J.; Dehen, C.J.; Gualtieri, E.J.; Grey, J.L.; Wang, H.F.; Thompson, D.H.; Cheng, J.X.; Simpson, G.J. Selective detection of protein crystals by second harmonic microscopy. J. Am. Chem. Soc. 2008, 130, 14076–14077. [Google Scholar] [CrossRef]
- Müller, M.; Schins, J.M. Imaging the thermodynamic state of lipid membranes with multiplex CARS microscopy. J. Phys. Chem. B 2002, 106, 3715–3723. [Google Scholar] [CrossRef]
- Wurpel, G.W.H.; Schins, J.M.; Müller, M. Direct measurement of chain order in single phospholipid mono- and bilayers with multiplex CARS. J. Phys. Chem. B 2004, 108, 3400–3403. [Google Scholar] [CrossRef]
- Cheng, J.X.; Volkmer, A.; Book, L.D.; Xie, X.S. Multiplex coherent anti-stokes Raman scattering microspectroscopy and study of lipid vesicles. J. Phys. Chem. B 2002, 106, 8493–8498. [Google Scholar] [CrossRef]
- Rinia, H.A.; Bonn, M.; Müller, M.; Vartiainen, E.M. Quantitative CARS spectroscopy using the maximum entropy method: The main lipid phase transition. ChemPhysChem 2007, 8, 279–287. [Google Scholar] [CrossRef] [PubMed]
- Spiro, T.G.; Strekas, T.C. Resonance Raman spectra of hemoglobin and cytochrome c: Inverse polarization and vibronic scattering. Proc. Natl. Acad. Sci. USA 1972, 69, 2622–2626. [Google Scholar] [CrossRef] [PubMed]
- Spiro, T.G.; Strekas, T.C. Resonance Raman Spectra of Heme Proteins. Effects of Oxidation and Spin State. J. Am. Chem. Soc. 1974, 96, 338–345. [Google Scholar] [CrossRef] [PubMed]
- Torres Filho, I.P.; Terner, J.; Pittman, R.N.; Proffitt, E.; Ward, K.R. Measurement of hemoglobin oxygen saturation using Raman microspectroscopy and 532-nm excitation. J. Appl. Physiol. 2008, 104, 1809–1817. [Google Scholar] [CrossRef] [PubMed]
- Torres Filho, I.P.; Nguyen, N.M.; Jivani, R.; Terner, J.; Romfh, P.; Vakhshoori, D.; Ward, K.R. Oxygen saturation monitoring using resonance Raman spectroscopy. J. Surg. Res. 2016, 201, 425–431. [Google Scholar] [CrossRef]
- Ward, K.R.; Barbee, R.W.; Reynolds, P.S.; Torres Filho, I.P.; Tiba, M.H.; Torres, L.; Pittman, R.N.; Terner, J. Oxygenation Monitoring of Tissue Vasculature by Resonance Raman Spectroscopy. Anal. Chem. 2007, 79, 1514–1518. [Google Scholar] [CrossRef]
- Brazhe, N.A.; Thomsen, K.; Lønstrup, M.; Brazhe, A.R.; Nikelshparg, E.I.; Maksimov, G.V.; Lauritzen, M.; Sosnovtseva, O. Monitoring of blood oxygenation in brain by resonance Raman spectroscopy. J. Biophotonics 2018, 11, e201700311. [Google Scholar] [CrossRef]
- Wood, B.R.; Caspers, P.; Puppels, G.J.; Pandiancherri, S.; McNaughton, D. Resonance Raman spectroscopy of red blood cells using near-infrared laser excitation. Anal. Bioanal. Chem. 2007, 387, 1691–1703. [Google Scholar] [CrossRef]
- Chowdhury, A.; Dasgupta, R.; Majumder, S.K. Changes in hemoglobin–oxygen affinity with shape variations of red blood cells. J. Biomed. Opt. 2017, 22, 1. [Google Scholar] [CrossRef]
- Rusciano, G.; De Luca, A.; Pesce, G.; Sasso, A. Raman Tweezers as a Diagnostic Tool of Hemoglobin-Related Blood Disorders. Sensors 2008, 8, 7818–7832. [Google Scholar] [CrossRef]
- Atkins, C.G.; Buckley, K.; Blades, M.W.; Turner, R.F.B. Raman Spectroscopy of Blood and Blood Components. Appl. Spectrosc. 2017, 71, 767–793. [Google Scholar] [CrossRef] [PubMed]
- Van Manen, H.J.; Kraan, Y.M.; Roos, D.; Otto, C. Intracellular chemical imaging of heme-containing enzymes involved in innate immunity using resonance Raman microscopy. J. Phys. Chem. B 2004, 108, 18762–18771. [Google Scholar] [CrossRef]
- Okada, M.; Smith, N.I.; Palonpon, A.F.; Endo, H.; Kawata, S.; Sodeoka, M.; Fujita, K. Label-free Raman observation of cytochrome c dynamics during apoptosis. Proc. Natl. Acad. Sci. USA 2012, 109, 28–32. [Google Scholar] [CrossRef] [PubMed]
- Kakita, M.; Kaliaperumal, V.; Hamaguchi, H.O. Resonance Raman quantification of the redox state of cytochromes b and c in-vivo and in-vitro. J. Biophotonics 2012, 5, 20–24. [Google Scholar] [CrossRef]
- Yasuda, M.; Takeshita, N.; Shigeto, S. Inhomogeneous Molecular Distributions and Cytochrome Types and Redox States in Fungal Cells Revealed by Raman Hyperspectral Imaging Using Multivariate Curve Resolution–Alternating Least Squares. Anal. Chem. 2019, 91, 12501–12508. [Google Scholar] [CrossRef]
- Brazhe, N.A.; Treiman, M.; Brazhe, A.R.; Find, N.L.; Maksimov, G.V.; Sosnovtseva, O.V. Mapping of Redox State of Mitochondrial Cytochromes in Live Cardiomyocytes Using Raman Microspectroscopy. PLoS ONE 2012, 7, e41990. [Google Scholar] [CrossRef]
- Brazhe, N.A.; Treiman, M.; Faricelli, B.; Vestergaard, J.H.; Sosnovtseva, O. In Situ Raman Study of Redox State Changes of Mitochondrial Cytochromes in a Perfused Rat Heart. PLoS ONE 2013, 8, e70488. [Google Scholar] [CrossRef]
- Nishiki-Muranishi, N.; Harada, Y.; Minamikawa, T.; Yamaoka, Y.; Dai, P.; Yaku, H.; Takamatsu, T. Label-Free Evaluation of Myocardial Infarction and Its Repair by Spontaneous Raman Spectroscopy. Anal. Chem. 2014, 86, 6903–6910. [Google Scholar] [CrossRef]
- Ohira, S.; Tanaka, H.; Harada, Y.; Minamikawa, T.; Kumamoto, Y.; Matoba, S.; Yaku, H.; Takamatsu, T. Label-free detection of myocardial ischaemia in the perfused rat heart by spontaneous Raman spectroscopy. Sci. Rep. 2017, 7, 42401. [Google Scholar] [CrossRef]
- Perry, D.A.; Salvin, J.W.; Romfh, P.; Chen, P.; Krishnamurthy, K.; Thomson, L.M.; Polizzotti, B.D.; McGowan, F.X.; Vakhshoori, D.; Kheir, J.N. Responsive monitoring of mitochondrial redox states in heart muscle predicts impending cardiac arrest. Sci. Transl. Med. 2017, 9, eaan0117. [Google Scholar] [CrossRef]
- Yamamoto, T.; Minamikawa, T.; Harada, Y.; Yamaoka, Y.; Tanaka, H.; Yaku, H.; Takamatsu, T. Label-free Evaluation of Myocardial Infarct in Surgically Excised Ventricular Myocardium by Raman Spectroscopy. Sci. Rep. 2018, 8, 14671. [Google Scholar] [CrossRef] [PubMed]
- Costas, C.; López-Puente, V.; Bodelón, G.; González-Bello, C.; Pérez-Juste, J.; Pastoriza-Santos, I.; Liz-Marzán, L.M. Using Surface Enhanced Raman Scattering to Analyze the Interactions of Protein Receptors with Bacterial Quorum Sensing Modulators. ACS Nano 2015, 9, 5567–5576. [Google Scholar] [CrossRef] [PubMed]
- Christophersen, P.C.; Birch, D.; Saarinen, J.; Isomäki, A.; Nielsen, H.M.; Yang, M.; Strachan, C.J.; Mu, H. Investigation of protein distribution in solid lipid particles and its impact on protein release using coherent anti-Stokes Raman scattering microscopy. J. Control. Release 2015, 197, 111–120. [Google Scholar] [CrossRef] [PubMed]
- Rygula, A.; Majzner, K.; Marzec, K.M.; Kaczor, A.; Pilarczyk, M.; Baranska, M. Raman spectroscopy of proteins: A review. J. Raman Spectrosc. 2013, 44, 1061–1076. [Google Scholar] [CrossRef]
- De Gelder, J.; De Gussem, K.; Vandenabeele, P.; Moens, L. Reference database of Raman spectra of biological molecules. J. Raman Spectrosc. 2007, 38, 1133–1147. [Google Scholar] [CrossRef]
- Bandekar, J. Amide modes and protein conformation. Biochim. Biophys. Acta BBA Protein Struct. Mol. 1992, 1120, 123–143. [Google Scholar] [CrossRef]
- Yu, N.T.; Jo, B.H. Comparison of protein structure in crystals and in solution by laser Raman scattering. I. Lysozyme. Arch. Biochem. Biophys. 1973, 156, 469–474. [Google Scholar] [CrossRef]
- Frontzek, A.V.; Paccou, L.; Guinet, Y.; Hédoux, A. Study of the phase transition in lysozyme crystals by Raman spectroscopy. Biochim. Biophys. Acta Gen. Subj. 2016, 1860, 412–423. [Google Scholar] [CrossRef][Green Version]
- Zabelskii, D.V.; Vlasov, A.V.; Ryzhykau, Y.L.; Murugova, T.N.; Brennich, M.; Soloviov, D.V.; Ivankov, O.I.; Borshchevskiy, V.I.; Mishin, A.V.; Rogachev, A.V.; et al. Ambiguities and completeness of SAS data analysis: Investigations of apoferritin by SAXS/SANS EID and SEC-SAXS methods. J. Phys. Conf. Ser. 2018, 994, 012017. [Google Scholar] [CrossRef]
- Vlasov, A.V.; Murugova, T.N.; Gruner, S.M.; Ivankov, O.I.; Soloviov, D.V.; Rogachev, A.V.; Round, A.; Ryzhykau, Y.L.; Mishin, A.; Balandin, T.; et al. Protein structure and structural ordering versus concentration dependence. FEBS J. 2014, 281, 593–594. [Google Scholar]
- Vlasov, A.V.; Ryzhykau, Y.L.; Gordeliy, V.I.; Kuklin, A.I. Spinach ATP-synthases form dimers in nanodiscs. Small-angle X-ray and neutron scattering investigations. FEBS J. 2017, 284, 87. [Google Scholar]
- Kuklin, A.I.; Rogachev, A.V.; Soloviov, D.V.; Ivankov, O.I.; Murugova, T.N.; Chupin, V.V.; Rulev, M.I.; Skoi, V.V.; Kučerka, N.; Vlasov, A.V.; et al. SANS investigations of biological objects on a YuMO spectrometer: Results and possibilities. J. Bioenerg. Biomembr. 2018, 50, 555. [Google Scholar]
- Murugova, T.N.; Vlasov, A.V.; Ivankov, O.I.; Rogachev, A.V.; Soloviov, D.V.; Zhigunov, A.; Kovalev, Y.S.; Ryzhykau, Y.L.; Zinovev, E.V.; Round, A.; et al. Low resolution structural studies of apoferritin via SANS and SAXS: The Effect of concentration. J. Optoelectron. Adv. Mater. 2015, 17, 1397–1402. [Google Scholar]
- Vlasova, A.D.; Khramtsov, Y.V.; Vlasov, A.V.; Kuklin, A.I.; Gordeliy, V.I.; Sobolev, A.S. Low-resolution structure of modular nanotransporters obtained by small-angle X-ray scattering method. J. Bioenerg. Biomembr. 2018, 50, 593. [Google Scholar]
- Kazantsev, A.S.; Vlasov, A.V.; Ryzhykau, Y.L.; Zabelskii, D.V.; Murugova, T.N.; Ivankov, O.I.; Rogachev, A.V.; Zinovev, E.V.; Kurbatov, N.M.; Gordeliy, V.I.; et al. SAXS studies of apoferritin in different pH with consideration of dimers. J. Bioenerg. Biomembr. 2018, 50, 548. [Google Scholar]
- Das, S.; Pal, U.; Chatterjee, M.; Pramanik, S.K.; Banerji, B.; Maiti, N.C. Envisaging Structural Insight of a Terminally Protected Proline Dipeptide by Raman Spectroscopy and Density Functional Theory Analyses. J. Phys. Chem. A 2016, 120, 9829–9840. [Google Scholar] [CrossRef]
- Bito, K.; Okuno, M.; Kano, H.; Tokuhara, S.; Naito, S.; Masukawa, Y.; Leproux, P.; Couderc, V.; Hamaguchi, H.O. Protein secondary structure imaging with ultrabroadband multiplex coherent anti-Stokes Raman scattering (CARS) microspectroscopy. J. Phys. Chem. B 2012, 116, 1452–1457. [Google Scholar] [CrossRef]
- Asamoto, D.A.K.; Kim, J.E. UV Resonance Raman Spectroscopy as a Tool to Probe Membrane Protein Structure and Dynamics. In Methods in Molecular Biology; Humana Press Inc.: Totowa, NJ, USA, 2019; Volume 2003, pp. 327–349. [Google Scholar]
- Lippert, J.L.; Tyminski, D.; Desmeules, P.J. Determination of the Secondary Structure of Proteins by Laser Raman Spectroscopy. J. Am. Chem. Soc. 1976, 98, 7075–7080. [Google Scholar] [CrossRef]
- Sane, S.U.; Cramer, S.M.; Przybycien, T.M. A holistic approach to protein secondary structure characterization using amide I band Raman spectroscopy. Anal. Biochem. 1999, 269, 255–272. [Google Scholar] [CrossRef]
- Masic, A.; Bertinetti, L.; Schuetz, R.; Galvis, L.; Timofeeva, N.; Dunlop, J.W.C.; Seto, J.; Hartmann, M.A.; Fratzl, P. Observations of Multiscale, Stress-Induced Changes of Collagen Orientation in Tendon by Polarized Raman Spectroscopy. Biomacromolecules 2011, 12, 3989–3996. [Google Scholar] [CrossRef]
- Van Gulick, L.; Saby, C.; Morjani, H.; Beljebbar, A. Age-related changes in molecular organization of type I collagen in tendon as probed by polarized SHG and Raman microspectroscopy. Sci. Rep. 2019, 9, 7280. [Google Scholar] [CrossRef] [PubMed]
- Silva, D.F.T.; Gomes, A.S.L.; de Campos Vidal, B.; Ribeiro, M.S. Birefringence and Second Harmonic Generation on Tendon Collagen Following Red Linearly Polarized Laser Irradiation. Ann. Biomed. Eng. 2013, 41, 752–762. [Google Scholar] [CrossRef] [PubMed]
- Frushour, B.G.; Koenig, J.L. Raman scattering of collagen, gelatin, and elastin. Biopolymers 1975, 14, 379–391. [Google Scholar] [CrossRef] [PubMed]
- Makowski, A.J.; Granke, M.; Uppuganti, S.; Mahadevan-Jansen, A.; Nyman, J.S. Bone Tissue Heterogeneity is Associated with Fracture Toughness: A Polarization Raman Spectroscopy Study. In Photonic Therapeutics and Diagnostics XI; Choi, B., Kollias, N., Zeng, H., Kang, H.W., Wong, B.J.F., Ilgner, J.F., Nuttal, A., Richter, C.-P., Skala, M.C., Dewhirst, M.W., et al., Eds.; SPIE: Washington, DC, USA, 2015; p. 930341. [Google Scholar]
- Galvis, L.; Dunlop, J.W.C.; Duda, G.; Fratzl, P.; Masic, A. Polarized Raman Anisotropic Response of Collagen in Tendon: Towards 3D Orientation Mapping of Collagen in Tissues. PLoS ONE 2013, 8, e63518. [Google Scholar] [CrossRef] [PubMed]
- Balakrishnan, G.; Weeks, C.L.; Ibrahim, M.; Soldatova, A.V.; Spiro, T.G. Protein dynamics from time resolved UV Raman spectroscopy. Curr. Opin. Struct. Biol. 2008, 18, 623–629. [Google Scholar] [CrossRef] [PubMed]
- Tuma, R. Raman spectroscopy of proteins: From peptides to large assemblies. J. Raman Spectrosc. 2005, 36, 307–319. [Google Scholar] [CrossRef]
- Balakhnina, I.A.; Brandt, N.N.; Chikishev, A.Y.; Mankova, A.A.; Shpachenko, I.G. Low-frequency vibrational spectroscopy of proteins with different secondary structures. J. Biomed. Opt. 2017, 22, 091509. [Google Scholar] [CrossRef]
- Ishigaki, M.; Hashimoto, K.; Sato, H.; Ozaki, Y. Non-destructive monitoring of mouse embryo development and its qualitative evaluation at the molecular level using Raman spectroscopy. Sci. Rep. 2017, 7, 43942. [Google Scholar] [CrossRef]
- McColl, I.H.; Blanch, E.W.; Hecht, L.; Barron, L.D. A Study of α-Helix Hydration in Polypeptides, Proteins, and Viruses Using Vibrational Raman Optical Activity. J. Am. Chem. Soc. 2004, 126, 8181–8188. [Google Scholar] [CrossRef]
- Berezhna, S.; Wohlrab, H.; Champion, P.M. Resonance Raman Investigations of Cytochrome c Conformational Change upon Interaction with the Membranes of Intact and Ca2+-Exposed Mitochondria. Biochemistry 2003, 42, 6149–6158. [Google Scholar] [CrossRef]
- Sun, Y.; Benabbas, A.; Zeng, W.; Kleingardner, J.G.; Bren, K.L.; Champion, P.M. Investigations of heme distortion, low-frequency vibrational excitations, and electron transfer in cytochrome c. Proc. Natl. Acad. Sci. USA 2014, 111, 6570–6575. [Google Scholar] [CrossRef] [PubMed]
- Chertkova, R.V.; Brazhe, N.A.; Bryantseva, T.V.; Nekrasov, A.N.; Dolgikh, D.A.; Yusipovich, A.I.; Sosnovtseva, O.; Maksimov, G.V.; Rubin, A.B.; Kirpichnikov, M.P. New insight into the mechanism of mitochondrial cytochrome c function. PLoS ONE 2017, 12, e0178280. [Google Scholar] [CrossRef] [PubMed]
- Varco-Merth, B.; Fromme, R.; Wang, M.; Fromme, P. Crystallization of the c14-rotor of the chloroplast ATP synthase reveals that it contains pigments. Biochim. Biophys. Acta Bioenerg. 2008, 1777, 605–612. [Google Scholar] [CrossRef] [PubMed]
- Malone, L.A.; Qian, P.; Mayneord, G.E.; Hitchcock, A.; Farmer, D.A.; Thompson, R.F.; Swainsbury, D.J.K.; Ranson, N.A.; Hunter, C.N.; Johnson, M.P. Cryo-EM structure of the spinach cytochrome b6 f complex at 3.6 Å resolution. Nature 2019, 575, 535–539. [Google Scholar] [CrossRef] [PubMed]
- Vlasov, A.V.; Kovalev, K.V.; Marx, S.-H.; Round, E.S.; Gushchin, I.Y.; Polovinkin, V.A.; Tsoy, N.M.; Okhrimenko, I.S.; Borshchevskiy, V.I.; Büldt, G.D.; et al. Unusual features of the c-ring of F1FO ATP synthases. Sci. Rep. 2019, 9, 18547. [Google Scholar] [CrossRef]
- Nazarenko, V.V.; Remeeva, A.; Yudenko, A.; Kovalev, K.; Dubenko, A.; Goncharov, I.M.; Kuzmichev, P.; Rogachev, A.V.; Buslaev, P.; Borshchevskiy, V.; et al. A thermostable flavin-based fluorescent protein from Chloroflexus aggregans: A framework for ultra-high resolution structural studies. Photochem. Photobiol. Sci. 2019, 18, 1793–1805. [Google Scholar] [CrossRef]
- Pacia, M.Z.; Turnau, K.; Baranska, M.; Kaczor, A. Interplay between carotenoids, hemoproteins and the “life band” origin studied in live Rhodotorula mucilaginosa cells by means of Raman microimaging. Analyst 2015, 140, 1809–1813. [Google Scholar] [CrossRef]
- Nishida, T.; Kaino, T.; Ikarashi, R.; Nakata, D.; Terao, K.; Ando, M.; Hamaguchi, H.O.; Kawamukai, M.; Yamamoto, T. The effect of coenzyme Q10 included by γ-cyclodextrin on the growth of fission yeast studied by microscope Raman spectroscopy. J. Mol. Struct. 2013, 1048, 375–381. [Google Scholar] [CrossRef]
- Slepkov, A.D.; Barlow, A.M.; Ridsdale, A.; McGinn, P.J.; Stolow, A. In vivo hyperspectral CARS and FWM microscopy of carotenoid accumulation in H. Pluvialis. In Proceedings of the Multimodal Biomedical Imaging IX, San Francisco, CA, USA, 1–6 February 2014; SPIE: San Francisco, CA, USA, 2014; Volume 8937, p. 893709. [Google Scholar]
- Røhr, Å.K.; Hersleth, H.P.; Kristoffer Andersson, K. Tracking flavin conformations in protein crystal structures with raman spectroscopy and QM/MM calculations. Angew. Chem. Int. Ed. 2010, 49, 2324–2327. [Google Scholar] [CrossRef]
- Altose, M.D.; Zheng, Y.; Dong, J.; Palfey, B.A.; Carey, P.R. Comparing protein-ligand interactions in solution and single crystals by Raman spectroscopy. Proc. Natl. Acad. Sci. USA 2001, 98, 3006–3011. [Google Scholar] [CrossRef]
- Mishin, A.; Gusach, A.; Luginina, A.; Marin, E.; Borshchevskiy, V.; Cherezov, V. An outlook on using serial femtosecond crystallography in drug discovery. Expert Opin. Drug Discov. 2019, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Baltoumas, F.A.; Theodoropoulou, M.C.; Hamodrakas, S.J. Interactions of the α-subunits of heterotrimeric G-proteins with GPCRs, effectors and RGS proteins: A critical review and analysis of interacting surfaces, conformational shifts, structural diversity and electrostatic potentials. J. Struct. Biol. 2013, 182, 209–218. [Google Scholar] [CrossRef] [PubMed]
- Gusach, A.; Luginina, A.; Marin, E.; Brouillette, R.L.; Besserer-Offroy, É.; Longpré, J.; Ishchenko, A.; Popov, P.; Patel, N.; Fujimoto, T.; et al. Structural basis of ligand selectivity and disease mutations in cysteinyl leukotriene receptors. Nat. Commun. 2019, 10, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Luginina, A.; Gusach, A.; Marin, E.; Mishin, A.; Brouillette, R.; Popov, P.; Shiriaeva, A.; Besserer-Offroy, É.; Longpré, J.M.; Lyapina, E.; et al. Structure-based mechanism of cysteinyl leukotriene receptor inhibition by antiasthmatic drugs. Sci. Adv. 2019, 5, eaax2518. [Google Scholar] [CrossRef] [PubMed]
- Cherezov, V. Lipidic cubic phase technologies for membrane protein structural studies. Curr. Opin. Struct. Biol. 2011, 21, 559–566. [Google Scholar] [CrossRef] [PubMed]
- Ishchenko, A.; Peng, L.; Zinovev, E.; Vlasov, A.; Lee, S.C.; Kuklin, A.; Mishin, A.; Borshchevskiy, V.; Zhang, Q.; Cherezov, V. Chemically Stable Lipids for Membrane Protein Crystallization. Cryst. Growth Des. 2017, 17, 3502–3511. [Google Scholar] [CrossRef] [PubMed]
- Bogorodskiy, A.; Frolov, F.; Mishin, A.; Round, E.; Polovinkin, V.; Cherezov, V.; Gordeliy, V.; Büldt, G.; Gensch, T.; Borshchevskiy, V. Nucleation and Growth of Membrane Protein Crystals in Meso—A Fluorescence Microscopy Study. Cryst. Growth Des. 2015, 15, 5656–5660. [Google Scholar] [CrossRef]
- Newman, J.A.; Simpson, G.J. Nonlinear optical characterization of membrane proteinmicrocrystals and nanocrystals. Adv. Exp. Med. Biol. 2016, 922, 91–103. [Google Scholar]
- Closser, R.G.; Gualtieri, E.J.; Newman, J.A.; Simpson, G.J. Characterization of salt interferences in second-harmonic generation detection of protein crystals. J. Appl. Crystallogr. 2013, 46, 1903–1906. [Google Scholar] [CrossRef]
- Arzumanyan, G.; Doroshkevich, N.V.; Mamatkulov, K.; Shashkov, S.; Zinovev, E.V.; Vlasov, A.; Round, E.; Gordeliy, V. Highly Sensitive Coherent anti-Stokes Raman Scattering Imaging of Protein Crystals. J. Am. Chem. Soc. 2016, 138, 13457–13460. [Google Scholar] [CrossRef]
- Razumas, V.; Talaikyte, Z.; Barauskas, J.; Larsson, K.; Miezis, Y.; Nylander, T. Effects of distearoylphosphatidylglycerol and lysozyme on the structure of the monoolein-water cubic phase: X-ray diffraction and Raman scattering studies. Chem. Phys. Lipids 1996, 84, 123–138. [Google Scholar] [CrossRef]
- Lord, R.C.; Thomas, G.J. Raman spectral studies of nucleic acids and related molecules-I Ribonucleic acid derivatives. Spectrochim. Acta Part A Mol. Spectrosc. 1967, 23, 2551–2591. [Google Scholar] [CrossRef]
- Thomas, G.J.; Wang, A.H.-J. Laser Raman Spectroscopy of Nucleic Acids. In Nucleic Acids and Molecular Biology; Springer: Berlin, Germany, 1988; pp. 1–30. [Google Scholar]
- Benevides, J.M.; Thomas, G.J. Characterization of DNA structures by Raman spectroscopy: High-salt and low-salt forms of double helical poly(dG-dC) in H2O and D2O solutions and application to B, Z and A-DNA. Nucleic Acids Res. 1983, 11, 5747–5761. [Google Scholar] [CrossRef] [PubMed]
- Nishimura, Y.; Tsuboi, M.; Sato, T.; Aoki, K. Conformation-sensitive Raman lines of mononucleotides and their use in a structure analysis of polynucleotides: Guanine and cytosine nucleotides. J. Mol. Struct. 1986, 146, 123–153. [Google Scholar] [CrossRef]
- Serban, D.; Benevides, J.M.; Thomas, G.J. DNA secondary structure and Raman markers of supercoiling in Escherichia coli plasmid pUC19. Biochemistry 2002, 41, 847–853. [Google Scholar] [CrossRef]
- Deng, H.; Bloomfield, V.A.; Benevides, J.M.; Thomas, G.J. Dependence of the raman signature of genomic B-DNA on nucleotide base sequence. Biopolymers 1999, 50, 656–666. [Google Scholar] [CrossRef]
- Pagba, C.V.; Lane, S.M.; Wachsmann-Hogiu, S. Conformational changes in quadruplex oligonucleotide structures probed by Raman spectroscopy. Biomed. Opt. Express 2011, 2, 207. [Google Scholar] [CrossRef]
- Thorogood, H.; Waters, T.R.; Parker, A.W.; Wharton, C.W.; Connolly, B.A. Resonance Raman spectroscopy of 4-thiothymidine and oligodeoxynucleotides containing this base both free in solution and bound to the restriction endonuclease EcoRV. Biochemistry 1996, 35, 8723–8733. [Google Scholar] [CrossRef]
- Toyama, A.; Miyagawa, Y.; Yoshimura, A.; Fujimoto, N.; Takeuchi, H. Characterization of individual adenine residues in DNA by a combination of site-selective C8-deuteration and UV resonance raman difference spectroscopy. J. Mol. Struct. 2001, 598, 85–91. [Google Scholar] [CrossRef]
- Tsuboi, M.; Kubo, Y.; Ikeda, T.; Overman, S.A.; Osman, O.; Thomas, G.J. Protein and DNA residue orientations in the filamentous virus Pf1 determined by polarized Raman and polarized FTIR spectroscopy. Biochemistry 2003, 42, 940–950. [Google Scholar] [CrossRef]
- Benevides, J.M.; Overman, S.A.; Thomas, G.J. Raman, polarized Raman and ultraviolet resonance Raman spectroscopy of nucleic acids and their complexes. J. Raman Spectrosc. 2005, 36, 279–299. [Google Scholar] [CrossRef]
- Thomas, G.J.; Benevides, J.M.; Overman, S.A.; Ueda, T.; Ushizawa, K.; Saitoh, M.; Tsuboi, M. Polarized Raman spectra of oriented fibers of A DNA and B DNA: Anisotropic and isotropic local Raman tensors of base and backbone vibrations. Biophys. J. 1995, 68, 1073–1088. [Google Scholar] [CrossRef]
- Tsuboi, M.; Benevides, J.M.; Thomas, G.J. The complex of ethidium bromide with genomic DNA: Structure analysis by polarized Raman spectroscopy. Biophys. J. 2007, 92, 928–934. [Google Scholar] [CrossRef] [PubMed]
- Friedman, S.J.; Terentis, A.C. Analysis of G-quadruplex conformations using Raman and polarized Raman spectroscopy. J. Raman Spectrosc. 2016, 47, 259–268. [Google Scholar] [CrossRef]
- Wu, C.Y.; Lo, W.Y.; Chiu, C.R.; Yang, T.S. Surface enhanced Raman spectra of oligonucleotides induced by spermine. J. Raman Spectrosc. 2006, 37, 799–807. [Google Scholar] [CrossRef]
- Chen, C.; Li, Y.; Kerman, S.; Neutens, P.; Willems, K.; Cornelissen, S.; Lagae, L.; Stakenborg, T.; Van Dorpe, P. High spatial resolution nanoslit SERS for single-molecule nucleobase sensing. Nat. Commun. 2018, 9, 1733. [Google Scholar] [CrossRef]
- Yang, J.M.; Jin, L.; Pan, Z.Q.; Zhou, Y.; Liu, H.L.; Ji, L.N.; Xia, X.H.; Wang, K. Surface-Enhanced Raman Scattering Probing the Translocation of DNA and Amino Acid through Plasmonic Nanopores. Anal. Chem. 2019, 91, 6275–6280. [Google Scholar] [CrossRef]
- Li, Y.; Gao, T.; Xu, G.; Xiang, X.; Zhao, B.; Han, X.X.; Guo, X. Direct Approach toward Label-Free DNA Detection by Surface-Enhanced Raman Spectroscopy: Discrimination of a Single-Base Mutation in 50 Base-Paired Double Helixes. Anal. Chem. 2019, 91, 7980–7984. [Google Scholar] [CrossRef]
- Chen, C.; Liu, W.; Tian, S.; Hong, T. Novel surface-enhanced raman spectroscopy techniques for DNA, protein and drug detection. Sensors 2019, 19, 1712. [Google Scholar] [CrossRef]
- Masetti, M.; Xie, H.; Krpetić, Ž.; Recanatini, M.; Alvarez-Puebla, R.A.; Guerrini, L. Revealing DNA Interactions with Exogenous Agents by Surface-Enhanced Raman Scattering. J. Am. Chem. Soc. 2015, 137, 469–476. [Google Scholar] [CrossRef]
- Guerrini, L.; Krpetić, Ž.; van Lierop, D.; Alvarez-Puebla, R.A.; Graham, D. Direct Surface-Enhanced Raman Scattering Analysis of DNA Duplexes. Angew. Chem. Int. Ed. 2015, 54, 1144–1148. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Han, X.; Zhou, S.; Yan, Y.; Xiang, X.; Zhao, B.; Guo, X. Structural Features of DNA G-Quadruplexes Revealed by Surface-Enhanced Raman Spectroscopy. J. Phys. Chem. Lett. 2018, 9, 3245–3252. [Google Scholar] [CrossRef] [PubMed]
- Qian, Y.; Fan, T.; Yao, Y.; Shi, X.; Liao, X.; Zhou, F.; Gao, F. Label-free and Raman dyes-free surface-enhanced Raman spectroscopy for detection of DNA. Sens. Actuators B Chem. 2018, 254, 483–489. [Google Scholar] [CrossRef]
- Lin, D.; Wu, Q.; Qiu, S.; Chen, G.; Feng, S.; Chen, R.; Zeng, H. Label-free liquid biopsy based on blood circulating DNA detection using SERS-based nanotechnology for nasopharyngeal cancer screening. Nanomed. Nanotechnol. Biol. Med. 2019, 22, 102100. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Yang, T.; Li, C.S.; Song, Y.; Lou, H.; Guan, D.; Jin, L. Surface Enhanced Raman Spectroscopy (SERS) for the multiplex detection of Braf, Kras, and Pik3ca mutations in plasma of colorectal cancer patients. Theranostics 2018, 8, 1678–1689. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Jeon, J.; Choi, N.; Lee, J.O.; Kim, Y.P.; Choo, J. SERS-based genetic assay for amplification-free detection of prostate cancer specific PCA3 mimic DNA. Sens. Actuators B Chem. 2017, 251, 302–309. [Google Scholar] [CrossRef]
- Korshoj, L.E.; Nagpal, P. Diagnostic Optical Sequencing. ACS Appl. Mater. Interfaces 2019, 11, 35587–35596. [Google Scholar] [CrossRef]
- van Lierop, D.; Faulds, K.; Graham, D. Separation Free DNA Detection Using Surface Enhanced Raman Scattering. Anal. Chem. 2011, 83, 5817–5821. [Google Scholar] [CrossRef]
- Mahadevan-Jansen, A.; Richards-Kortum, R. Raman spectroscopy for cancer detection: A review. In Proceedings of the 19th Annual International Conference of the IEEE Engineering in Medicine and Biology Society. ‘Magnificent Milestones and Emerging Opportunities in Medical Engineering’, Chicago, IL, USA, 30 October–2 November 1997; Volume 6, pp. 2722–2728. [Google Scholar]
- Liu, C.H.; Das, B.B.; Glassman, W.L.S.; Tang, G.C.; Yoo, K.M.; Zhu, H.R.; Akins, D.L.; Lubicz, S.S.; Cleary, J.; Prudente, R.; et al. Raman, fluorescence, and time-resolved light scattering as optical diagnostic techniques to separate diseased and normal biomedical media. J. Photochem. Photobiol. B Biol. 1992, 16, 187–209. [Google Scholar] [CrossRef]
- Kallaway, C.; Almond, L.M.; Barr, H.; Wood, J.; Hutchings, J.; Kendall, C.; Stone, N. Advances in the clinical application of Raman spectroscopy for cancer diagnostics. Photodiagn. Photodyn. Ther. 2013, 10, 207–219. [Google Scholar] [CrossRef]
- Almond, L.M.; Hutchings, J.; Shepherd, N.; Barr, H.; Stone, N.; Kendall, C. Raman spectroscopy: A potential tool for early objective diagnosis of neoplasia in the oesophagus. J. Biophotonics 2011, 4, 685–695. [Google Scholar] [CrossRef] [PubMed]
- Old, O.J.; Fullwood, L.M.; Scott, R.; Lloyd, G.R.; Almond, L.M.; Shepherd, N.A.; Stone, N.; Barr, H.; Kendall, C. Vibrational spectroscopy for cancer diagnostics. Anal. Methods 2014, 6, 3901–3917. [Google Scholar] [CrossRef]
- Frank, C.J.; McCreery, R.L.; Redd, D.C.B. Raman Spectroscopy of Normal and Diseased Human Breast Tissues. Anal. Chem. 1995, 67, 777–783. [Google Scholar] [CrossRef] [PubMed]
- Haka, A.S.; Shafer-Peltier, K.E.; Fitzmaurice, M.; Crowe, J.; Dasari, R.R.; Feld, M.S. Diagnosing breast cancer by using Raman spectroscopy. Proc. Natl. Acad. Sci. USA 2005, 102, 12371–12376. [Google Scholar] [CrossRef] [PubMed]
- Palombo, F.; Madami, M.; Fioretto, D.; Nallala, J.; Barr, H.; David, A.; Stone, N. Chemico-mechanical imaging of Barrett’s oesophagus. J. Biophotonics 2016, 9, 694–700. [Google Scholar] [CrossRef] [PubMed]
- Kast, R.E.; Tucker, S.C.; Killian, K.; Trexler, M.; Honn, K.V.; Auner, G.W. Emerging technology: Applications of Raman spectroscopy for prostate cancer. Cancer Metastasis Rev. 2014, 33, 673–693. [Google Scholar] [CrossRef]
- Devpura, S.; Thakur, J.S.; Sarkar, F.H.; Sakr, W.A.; Naik, V.M.; Naik, R. Detection of benign epithelia, prostatic intraepithelial neoplasia, and cancer regions in radical prostatectomy tissues using Raman spectroscopy. Vib. Spectrosc. 2010, 53, 227–232. [Google Scholar] [CrossRef]
- Crow, P.; Molckovsky, A.; Stone, N.; Uff, J.; Wilson, B.; Wongkeesong, L.M. Assessment of fiberoptic near-infrared raman spectroscopy for diagnosis of bladder and prostate cancer. Urology 2005, 65, 1126–1130. [Google Scholar] [CrossRef]
- Huang, Z.; McWilliams, A.; Lui, H.; McLean, D.I.; Lam, S.; Zeng, H. Near-infrared Raman spectroscopy for optical diagnosis of lung cancer. Int. J. Cancer 2003, 107, 1047–1052. [Google Scholar] [CrossRef]
- Xu, M.; Ma, J.; Qu, Y.; Mao, W.; Zheng, R. Recognition of gastric cancer by Raman spectroscopy. In Proceedings of the 8th International Conference on Photonics and Imaging in Biology and Medicine (PIBM 2009), Wuhan, China, 8–10 August 2009; SPIE: San Francisco, CA, USA, 2009; Volume 7519, p. 75191H. [Google Scholar]
- Taketani, A.; Andriana, B.B.; Matsuyoshi, H.; Sato, H. Raman endoscopy for monitoring the anticancer drug treatment of colorectal tumors in live mice. Analyst 2017, 142, 3680–3688. [Google Scholar] [CrossRef]
- Laing, S.; Hernandez-Santana, A.; Sassmannshausen, J.; Asquith, D.L.; McInnes, I.B.; Faulds, K.; Graham, D. Quantitative Detection of Human Tumor Necrosis Factor α by a Resonance Raman Enzyme-Linked Immunosorbent Assay. Anal. Chem. 2011, 83, 297–302. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Fan, Y.; He, M.; Ma, X.; Song, Y.; Liu, M.; Xu, J. Accuracy of Raman spectroscopy in differentiating brain tumor from normal brain tissue. Oncotarget 2017, 8, 36824. [Google Scholar] [CrossRef] [PubMed]
- Garzon-Muvdi, T.; Kut, C.; Li, X.; Chaichana, K.L. Intraoperative imaging techniques for glioma surgery. Future Oncol. 2017, 13, 1731–1745. [Google Scholar] [CrossRef] [PubMed]
- Hollon, T.; Lewis, S.; Freudiger, C.W.; Xie, X.S.; Orringer, D.A. Improving the accuracy of brain tumor surgery via Raman-based technology. J. Neurosurg. 2016, 40, E9. [Google Scholar] [CrossRef]
- Jermyn, M.; Mok, K.; Mercier, J.; Desroches, J.; Pichette, J.; Saint-Arnaud, K.; Bernstein, L.; Guiot, M.C.; Petrecca, K.; Leblond, F. Intraoperative brain cancer detection with Raman spectroscopy in humans. Sci. Transl. Med. 2015, 7, 274ra19. [Google Scholar] [CrossRef]
- Jermyn, M.; Desroches, J.; Mercier, J.; Tremblay, M.-A.; St-Arnaud, K.; Guiot, M.-C.; Petrecca, K.; Leblond, F. Neural networks improve brain cancer detection with Raman spectroscopy in the presence of operating room light artifacts. J. Biomed. Opt. 2016, 21, 094002. [Google Scholar] [CrossRef]
- Desroches, J.; Jermyn, M.; Pinto, M.; Picot, F.; Tremblay, M.A.; Obaid, S.; Marple, E.; Urmey, K.; Trudel, D.; Soulez, G.; et al. A new method using Raman spectroscopy for in vivo targeted brain cancer tissue biopsy. Sci. Rep. 2018, 8, 1792. [Google Scholar] [CrossRef]
- Krafft, C.; Sobottka, S.B.; Schackert, G.; Salzer, R. Raman and infrared spectroscopic mapping of human primary intracranial tumors: A comparative study. J. Raman Spectrosc. 2006, 37, 367–375. [Google Scholar] [CrossRef]
- Krafft, C.; Bergner, N.; Romeike, B.; Reichart, R.; Kalff, R.; Geiger, K.; Kirsch, M.; Schackert, G.; Popp, J. Raman spectroscopic imaging as complementary tool for histopathologic assessment of brain tumors. In Proceedings of the Photonic Therapeutics and Diagnostics VIII, San Francisco, CA, USA, 21–26 January 2012; SPIE: San Francisco, CA, USA, 2012; Volume 8207, p. 82074I. [Google Scholar]
- Koljenović, S.; Choo-Smith, L.P.; Schut, T.C.B.; Kros, J.M.; Van den Berge, H.J.; Puppels, G.J. Discriminating vital tumor from necrotic tissue in human glioblastoma tissue samples by Raman spectroscopy. Lab. Investig. 2002, 82, 1265–1277. [Google Scholar] [CrossRef]
- Managò, S.; Valente, C.; Mirabelli, P.; Circolo, D.; Basile, F.; Corda, D.; De Luca, A.C. A reliable Raman-spectroscopy-based approach for diagnosis, classification and follow-up of B-cell acute lymphoblastic leukemia. Sci. Rep. 2016, 6, 24821. [Google Scholar] [CrossRef]
- Fabris, L. SERS Tags: The Next Promising Tool for Personalized Cancer Detection? ChemNanoMat 2016, 2, 249–258. [Google Scholar] [CrossRef]
- Cialla-May, D.; Zheng, X.S.; Weber, K.; Popp, J. Recent progress in surface-enhanced Raman spectroscopy for biological and biomedical applications: From cells to clinics. Chem. Soc. Rev. 2017, 46, 3945–3961. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Schlücker, S. ISERS microscopy for tissue-based cancer diagnostics with SERS nanotags. In Springer Series in Surface Sciences; Springer: Berlin, Germany, 2018; Volume 66, pp. 347–379. [Google Scholar]
- Guerrini, L.; Alvarez-Puebla, R.A. Surface-Enhanced Raman Spectroscopy in Cancer Diagnosis, Prognosis and Monitoring. Cancers 2019, 11, 748. [Google Scholar] [CrossRef] [PubMed]
- Schlücker, S.; Küstner, B.; Punge, A.; Bonfig, R.; Marx, A.; Ströbel, P. Immuno-Raman microspectroscopy: In situ detection of antigens in tissue specimens by surface-enhanced Raman scattering. J. Raman Spectrosc. 2006, 37, 719–721. [Google Scholar] [CrossRef]
- Wang, X.P.; Zhang, Y.; König, M.; Papadopoulou, E.; Walkenfort, B.; Kasimir-Bauer, S.; Bankfalvi, A.; Schlücker, S. ISERS microscopy guided by wide field immunofluorescence: Analysis of HER2 expression on normal and breast cancer FFPE tissue sections. Analyst 2016, 141, 5113–5119. [Google Scholar] [CrossRef]
- Stepula, E.; König, M.; Wang, X.; Levermann, J.; Schimming, T.; Kasimir-Bauer, S.; Schilling, B.; Schlücker, S. Localization of PD-L1 on single cancer cells by iSERS microscopy with Au/Au core/satellite nanoparticles. J. Biophotonics 2019. [Google Scholar] [CrossRef]
- Lee, S.; Chon, H.; Yoon, S.Y.; Lee, E.K.; Chang, S.I.; Lim, D.W.; Choo, J. Fabrication of SERS-fluorescence dual modal nanoprobes and application to multiplex cancer cell imaging. Nanoscale 2012, 4, 124–129. [Google Scholar] [CrossRef]
- Zavaleta, C.L.; Smith, B.R.; Walton, I.; Doering, W.; Davis, G.; Shojaei, B.; Natan, M.J.; Gambhir, S.S. Multiplexed imaging of surface enhanced Raman scattering nanotags in living mice using noninvasive Raman spectroscopy. Proc. Natl. Acad. Sci. USA 2009, 106, 13511–13516. [Google Scholar] [CrossRef]
- Zeng, Y.; Koo, K.M.; Trau, M.; Shen, A.G.; Hu, J.M. Watching SERS glow for multiplex biomolecular analysis in the clinic: A review. Appl. Mater. Today 2019, 15, 431–444. [Google Scholar] [CrossRef]
- Pal, S.; Ray, A.; Andreou, C.; Zhou, Y.; Rakshit, T.; Wlodarczyk, M.; Maeda, M.; Toledo-Crow, R.; Berisha, N.; Yang, J.; et al. DNA-enabled rational design of fluorescence-Raman bimodal nanoprobes for cancer imaging and therapy. Nat. Commun. 2019, 10. [Google Scholar] [CrossRef]
- Zavaleta, C.L.; Garai, E.; Liu, J.T.C.; Sensarn, S.; Mandella, M.J.; Van De Sompel, D.; Friedland, S.; Van Dam, J.; Contag, C.H.; Gambhira, S.S. A Raman-based endoscopic strategy for multiplexed molecular imaging. Proc. Natl. Acad. Sci. USA 2013, 110, E2288–E2297. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.W.; Kang, S.; Khan, A.; Bao, P.Q.; Liu, J.T.C. In vivo multiplexed molecular imaging of esophageal cancer via spectral endoscopy of topically applied SERS nanoparticles. Biomed. Opt. Express 2015, 6, 3714. [Google Scholar] [CrossRef] [PubMed]
- Jeong, S.; Kim, Y.I.; Kang, H.; Kim, G.; Cha, M.G.; Chang, H.; Jung, K.O.; Kim, Y.H.; Jun, B.H.; Hwang, D.W.; et al. Fluorescence-Raman Dual Modal Endoscopic System for Multiplexed Molecular Diagnostics. Sci. Rep. 2015, 5, 9455. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Ashton, J.R.; Moding, E.J.; Yuan, H.; Register, J.K.; Fales, A.M.; Choi, J.; Whitley, M.J.; Zhao, X.; Qi, Y.; et al. A plasmonic gold nanostar theranostic probe for in vivo tumor imaging and photothermal therapy. Theranostics 2015, 5, 946–960. [Google Scholar] [CrossRef] [PubMed]
- Qiu, Y.; Lin, M.; Chen, G.; Fan, C.; Li, M.; Gu, X.; Cong, S.; Zhao, Z.; Fu, L.; Fang, X.; et al. Photodegradable CuS SERS Probes for Intraoperative Residual Tumor Detection, Ablation, and Self-Clearance. ACS Appl. Mater. Interfaces 2019, 11, 23436–23444. [Google Scholar] [CrossRef] [PubMed]
- Miao, K.; Wei, L. Live-Cell Imaging and Quantification of PolyQ Aggregates by Stimulated Raman Scattering of Selective Deuterium Labeling. bioRxiv 2019. bioRxiv:820217. [Google Scholar]
- Ryzhikova, E.; Kazakov, O.; Halamkova, L.; Celmins, D.; Malone, P.; Molho, E.; Zimmerman, E.A.; Lednev, I.K. Raman spectroscopy of blood serum for Alzheimer’s disease diagnostics: Specificity relative to other types of dementia. J. Biophotonics 2015, 8, 584–596. [Google Scholar] [CrossRef]
- Noothalapati, H.; Sasaki, T.; Kaino, T.; Kawamukai, M.; Ando, M.; Hamaguchi, H.O.; Yamamoto, T. Label-free Chemical Imaging of Fungal Spore Walls by Raman Microscopy and Multivariate Curve Resolution Analysis. Sci. Rep. 2016, 6, 27789. [Google Scholar] [CrossRef]
- Schweikhard, V.; Baral, A.; Krishnamachari, V.; Hay, W.C.; Fuhrmann, M. Label-free characterization of Amyloid-β-plaques and associated lipids in brain tissues using stimulated Raman scattering microscopy. bioRxiv 2019. bioRxiv:789248. [Google Scholar]
- Krasnoslobodtsev, A.V.; Deckert-Gaudig, T.; Zhang, Y.; Deckert, V.; Lyubchenko, Y.L. Polymorphism of amyloid fibrils formed by a peptide from the yeast prion protein Sup35: AFM and Tip-Enhanced Raman Scattering studies. Ultramicroscopy 2016, 165, 26–33. [Google Scholar] [CrossRef]
- Kurouski, D.; Deckert-Gaudig, T.; Deckert, V.; Lednev, I.K. Structure and Composition of Insulin Fibril Surfaces Probed by TERS. J. Am. Chem. Soc. 2012, 134, 13323–13329. [Google Scholar] [CrossRef] [PubMed]
- Bae, K.; Zheng, W.; Huang, Z. Quantitative assessment of spinal cord injury using circularly polarized coherent anti-Stokes Raman scattering microscopy. Appl. Phys. Lett. 2017, 111, 063704. [Google Scholar] [CrossRef]
- Yang, Y.C.; Chang, W.T.; Huang, S.K.; Liau, I. Characterization of the pharmaceutical effect of drugs on atherosclerotic lesions in vivo using integrated fluorescence imaging and raman spectral measurements. Anal. Chem. 2014, 86, 3863–3868. [Google Scholar] [CrossRef] [PubMed]
- Miyaoka, R.; Hosokawa, M.; Ando, M.; Mori, T.; Hamaguchi, H.O.; Takeyama, H. In situ detection of antibiotic amphotericin B produced in Streptomyces nodosus using Raman microspectroscopy. Mar. Drugs 2014, 12, 2827–2839. [Google Scholar] [CrossRef] [PubMed]
- Altunbek, M.; Çetin, D.; Suludere, Z.; Çulha, M. Surface-enhanced Raman spectroscopy based 3D spheroid culture for drug discovery studies. Talanta 2019, 191, 390–399. [Google Scholar] [CrossRef]
- Olaetxea, I.; Lopez, E.; Valero, A.; Seifert, A. Determination of physiological lactate and pH by Raman spectroscopy*. In Proceedings of the 41st Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Berlin, Germany, 23–27 July 2019; pp. 475–481. [Google Scholar]
- Noothalapati, H.; Uemura, S.; Ohshima, N.; Kinoshita, Y.; Ando, M.; Hamaguchi, H.O.; Yamamoto, T. Towards the development of a non-bioptic diagnostic technique for eosinophilic esophagitis using Raman spectroscopy. Vib. Spectrosc. 2016, 85, 7–10. [Google Scholar] [CrossRef]
- Hirose, K.; Aoki, T.; Furukawa, T.; Fukushima, S.; Niioka, H.; Deguchi, S.; Hashimoto, M. Coherent anti-Stokes Raman scattering rigid endoscope toward robot-assisted surgery. Biomed. Opt. Express 2018, 9, 387. [Google Scholar] [CrossRef]
- Makowski, A.J.; Pence, I.J.; Uppuganti, S.; Zein-Sabatto, A.; Huszagh, M.C.; Mahadevan-Jansen, A.; Nyman, J.S. Polarization in Raman spectroscopy helps explain bone brittleness in genetic mouse models. J. Biomed. Opt. 2014, 19, 117008. [Google Scholar] [CrossRef]
- Virkler, K.; Lednev, I.K. Raman spectroscopy offers great potential for the nondestructive confirmatory identification of body fluids. Forensic Sci. Int. 2008, 181, e1–e5. [Google Scholar] [CrossRef]
- Muro, C.K.; Doty, K.C.; de Souza Fernandes, L.; Lednev, I.K. Forensic body fluid identification and differentiation by Raman spectroscopy. Forensic Chem. 2016, 1, 31–38. [Google Scholar] [CrossRef]
- Mistek, E.; Halámková, L.; Doty, K.C.; Muro, C.K.; Lednev, I.K. Race Differentiation by Raman Spectroscopy of a Bloodstain for Forensic Purposes. Anal. Chem. 2016, 88, 7453–7456. [Google Scholar] [CrossRef] [PubMed]
- Sikirzhytskaya, A.; Sikirzhytski, V.; Lednev, I.K. Determining Gender by Raman Spectroscopy of a Bloodstain. Anal. Chem. 2017, 89, 1486–1492. [Google Scholar] [CrossRef] [PubMed]
- McLaughlin, G.; Doty, K.C.; Lednev, I.K. Raman Spectroscopy of Blood for Species Identification. Anal. Chem. 2014, 86, 11628–11633. [Google Scholar] [CrossRef] [PubMed]
- Doty, K.C.; McLaughlin, G.; Lednev, I.K. A Raman “spectroscopic clock” for bloodstain age determination: The first week after deposition. Anal. Bioanal. Chem. 2016, 408, 3993–4001. [Google Scholar] [CrossRef]
- Langer, J.; Novikov, S.M.; Liz-Marzán, L.M. Sensing using plasmonic nanostructures and nanoparticles. Nanotechnology 2015, 26, 322001. [Google Scholar] [CrossRef]
- Eremina, O.E.; Semenova, A.A.; Sergeeva, E.A.; Brazhe, N.A.; Maksimov, G.V.; Shekhovtsova, T.N.; Goodilin, E.A.; Veselova, I.A. Surface-enhanced Raman spectroscopy in modern chemical analysis: Advances and prospects. Russ. Chem. Rev. 2018, 87, 741–770. [Google Scholar] [CrossRef]
- Fikiet, M.A.; Khandasammy, S.R.; Mistek, E.; Ahmed, Y.; Halámková, L.; Bueno, J.; Lednev, I.K. Surface enhanced Raman spectroscopy: A review of recent applications in forensic science. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2018, 197, 255–260. [Google Scholar] [CrossRef]
- Laing, S.; Jamieson, L.E.; Faulds, K.; Graham, D. Surface-enhanced Raman spectroscopy for in vivo biosensing. Nat. Rev. Chem. 2017, 1, 0060. [Google Scholar] [CrossRef]
- Talley, C.E.; Jusinski, L.; Hollars, C.W.; Lane, S.M.; Huser, T. Intracellular pH Sensors Based on Surface-Enhanced Raman Scattering. Anal. Chem. 2004, 76, 7064–7068. [Google Scholar] [CrossRef]
- Kneipp, J.; Kneipp, H.; Wittig, B.; Kneipp, K. One- and Two-Photon Excited Optical pH Probing for Cells Using Surface-Enhanced Raman and Hyper-Raman Nanosensors. Nano Lett. 2007, 7, 2819–2823. [Google Scholar] [CrossRef]
- Wang, J.; Geng, Y.; Shen, Y.; Shi, W.; Xu, W.; Xu, S. SERS-active fiber tip for intracellular and extracellular pH sensing in living single cells. Sens. Actuators B Chem. 2019, 290, 527–534. [Google Scholar] [CrossRef]
- Lyandres, O.; Yuen, J.M.; Shah, N.C.; VanDuyne, R.P.; Walsh, J.T.; Glucksberg, M.R. Progress toward an in vivo surface-enhanced Raman spectroscopy glucose sensor. Diabetes Technol. Ther. 2008, 10, 257–265. [Google Scholar] [CrossRef] [PubMed]
- Qi, G.; Wang, Y.; Zhang, B.; Sun, D.; Fu, C.; Xu, W.; Xu, S. Glucose oxidase probe as a surface-enhanced Raman scattering sensor for glucose. Anal. Bioanal. Chem. 2016, 408, 7513–7520. [Google Scholar] [CrossRef] [PubMed]
- Fu, C.; Jin, S.; Oh, J.; Xu, S.; Jung, Y.M. Facile detection of glucose in human serum employing silver-ion-guided surface-enhanced Raman spectroscopy signal amplification. Analyst 2017, 142, 2887–2891. [Google Scholar] [CrossRef] [PubMed]
- Qu, L.-L.; Li, D.-W.; Qin, L.-X.; Mu, J.; Fossey, J.S.; Long, Y.-T. Selective and Sensitive Detection of Intracellular O 2 •− Using Au NPs/Cytochrome c as SERS Nanosensors. Anal. Chem. 2013, 85, 9549–9555. [Google Scholar] [CrossRef] [PubMed]
- Zorov, D.B.; Juhaszova, M.; Sollott, S.J. Mitochondrial reactive oxygen species (ROS) and ROS-induced ROS release. Physiol. Rev. 2014, 94, 909–950. [Google Scholar] [CrossRef]
- Lenzi, E.; Jimenez de Aberasturi, D.; Liz-Marzán, L.M. Surface-Enhanced Raman Scattering Tags for Three-Dimensional Bioimaging and Biomarker Detection. ACS Sens. 2019, 4, 1126–1137. [Google Scholar] [CrossRef]
- Mosier-Boss, P. Review on SERS of Bacteria. Biosensors 2017, 7, 51. [Google Scholar] [CrossRef]
- Pang, Y.; Wan, N.; Shi, L.; Wang, C.; Sun, Z.; Xiao, R.; Wang, S. Dual-recognition surface-enhanced Raman scattering(SERS)biosensor for pathogenic bacteria detection by using vancomycin-SERS tags and aptamer-Fe3O4@Au. Anal. Chim. Acta 2019, 1077, 288–296. [Google Scholar] [CrossRef]
- Yuan, K.; Zheng, J.; Yang, D.; Jurado Sánchez, B.; Liu, X.; Guo, X.; Liu, C.; Dina, N.E.; Jian, J.; Bao, Z.; et al. Self-Assembly of Au@Ag Nanoparticles on Mussel Shell To Form Large-Scale 3D Supercrystals as Natural SERS Substrates for the Detection of Pathogenic Bacteria. ACS Omega 2018, 3, 2855–2864. [Google Scholar] [CrossRef]
- Liu, T.Y.; Tsai, K.T.; Wang, H.H.; Chen, Y.; Chen, Y.H.; Chao, Y.C.; Chang, H.H.; Lin, C.H.; Wang, J.K.; Wang, Y.L. Functionalized arrays of Raman-enhancing nanoparticles for capture and culture-free analysis of bacteria in human blood. Nat. Commun. 2011, 2, 538. [Google Scholar] [CrossRef] [PubMed]
- Chisanga, M.; Linton, D.; Muhamadali, H.; Ellis, D.I.; Kimber, R.L.; Mironov, A.; Goodacre, R. Rapid differentiation of Campylobacter jejuni cell wall mutants using Raman spectroscopy, SERS and mass spectrometry combined with chemometrics. Analyst 2020. [Google Scholar] [CrossRef] [PubMed]
- Gracie, K.; Correa, E.; Mabbott, S.; Dougan, J.A.; Graham, D.; Goodacre, R.; Faulds, K. Simultaneous detection and quantification of three bacterial meningitis pathogens by SERS. Chem. Sci. 2014, 5, 1030–1040. [Google Scholar] [CrossRef]
- Lussier, F.; Brulé, T.; Vishwakarma, M.; Das, T.; Spatz, J.P.; Masson, J.F. Dynamic-SERS optophysiology: A nanosensor for monitoring cell secretion events. Nano Lett. 2016, 16, 3866–3871. [Google Scholar] [CrossRef]
- Lussier, F.; Missirlis, D.; Spatz, J.P.; Masson, J.-F. Machine-Learning-Driven Surface-Enhanced Raman Scattering Optophysiology Reveals Multiplexed Metabolite Gradients Near Cells. ACS Nano 2019, 13, 1403–1411. [Google Scholar] [CrossRef]
- Zhu, H.; Lussier, F.; Ducrot, C.; Bourque, M.-J.; Spatz, J.P.; Cui, W.; Yu, L.; Peng, W.; Trudeau, L.-É.; Bazuin, C.G.; et al. Block Copolymer Brush Layer-Templated Gold Nanoparticles on Nanofibers for Surface-Enhanced Raman Scattering Optophysiology. ACS Appl. Mater. Interfaces 2019, 11, 4373–4384. [Google Scholar] [CrossRef]
- Vitol, E.A.; Orynbayeva, Z.; Bouchard, M.J.; Azizkhan-Clifford, J.; Friedman, G.; Gogotsi, Y. In Situ Intracellular Spectroscopy with Surface Enhanced Raman Spectroscopy (SERS)-Enabled Nanopipettes. ACS Nano 2009, 3, 3529–3536. [Google Scholar] [CrossRef]
- Vitol, E.A.; Brailoiu, E.; Orynbayeva, Z.; Dun, N.J.; Friedman, G.; Gogotsi, Y. Surface-Enhanced Raman Spectroscopy as a Tool for Detecting Ca 2+ Mobilizing Second Messengers in Cell Extracts. Anal. Chem. 2010, 82, 6770–6774. [Google Scholar] [CrossRef]
- Ryzhikova, E.; Ralbovsky, N.M.; Halámková, L.; Celmins, D.; Malone, P.; Molho, E.; Quinn, J.; Zimmerman, E.A.; Lednev, I.K. Multivariate Statistical Analysis of Surface Enhanced Raman Spectra of Human Serum for Alzheimer’s Disease Diagnosis. Appl. Sci. 2019, 9, 3256. [Google Scholar] [CrossRef]
- Brazhe, N.A.; Abdali, S.; Brazhe, A.R.; Luneva, O.G.; Bryzgalova, N.Y.; Parshina, E.Y.; Sosnovtseva, O.V.; Maksimov, G.V. New insight into erythrocyte through in vivo surface-enhanced Raman spectroscopy. Biophys. J. 2009, 97, 3206–3214. [Google Scholar] [CrossRef]
- Semenova, A.A.; Goodilin, E.A.; Brazhe, N.A.; Ivanov, V.K.; Baranchikov, A.E.; Lebedev, V.A.; Goldt, A.E.; Sosnovtseva, O.V.; Savilov, S.V.; Egorov, A.V.; et al. Planar SERS nanostructures with stochastic silver ring morphology for biosensor chips. J. Mater. Chem. 2012, 22, 24530–24544. [Google Scholar] [CrossRef]
- Brazhe, N.A.; Parshina, E.Y.; Khabatova, V.V.; Semenova, A.A.; Brazhe, A.R.; Yusipovich, A.I.; Sarycheva, A.S.; Churin, A.A.; Goodilin, E.A.; Maksimov, G.V.; et al. Tuning SERS for living erythrocytes: Focus on nanoparticle size and plasmon resonance position. J. Raman Spectrosc. 2013, 44, 686–694. [Google Scholar] [CrossRef]
- Sarycheva, A.S.; Brazhe, N.A.; Baizhumanov, A.A.; Nikelshparg, E.I.; Semenova, A.A.; Garshev, A.V.; Baranchikov, A.E.; Ivanov, V.K.; Maksimov, G.V.; Sosnovtseva, O.; et al. New nanocomposites for SERS studies of living cells and mitochondria. J. Mater. Chem. B 2016, 4, 539–546. [Google Scholar] [CrossRef]
- Luneva, O.G.; Brazhe, N.A.; Maksimova, N.V.; Rodnenkov, O.V.; Parshina, E.Y.; Bryzgalova, N.Y.; Maksimov, G.V.; Rubin, A.B.; Orlov, S.N.; Chazov, E.I. Ion transport, membrane fluidity and haemoglobin conformation in erythrocyte from patients with cardiovascular diseases: Role of augmented plasma cholesterol. Pathophysiology 2007, 14, 41–46. [Google Scholar] [CrossRef] [PubMed]
- Brazhe, N.A.; Evlyukhin, A.B.; Goodilin, E.A.; Semenova, A.A.; Novikov, S.M.; Bozhevolnyi, S.I.; Chichkov, B.N.; Sarycheva, A.S.; Baizhumanov, A.A.; Nikelshparg, E.I.; et al. Probing cytochrome c in living mitochondria with surface-enhanced Raman spectroscopy. Sci. Rep. 2015, 5, 13793. [Google Scholar] [CrossRef]
- Stone, N.; Kerssens, M.; Lloyd, G.R.; Faulds, K.; Graham, D.; Matousek, P. Surface enhanced spatially offset Raman spectroscopic (SESORS) imaging—The next dimension. Chem. Sci. 2011, 2, 776–780. [Google Scholar] [CrossRef]
- Stuart, D.A.; Yuen, J.M.; Shah, N.; Lyandres, O.; Yonzon, C.R.; Glucksberg, M.R.; Walsh, J.T.; Van Duyne, R.P. In Vivo Glucose Measurement by Surface-Enhanced Raman Spectroscopy. Anal. Chem. 2006, 78, 7211–7215. [Google Scholar] [CrossRef]
- Camp, C.H.; Lee, Y.J.; Heddleston, J.M.; Hartshorn, C.M.; Walker, A.R.H.; Rich, J.N.; Lathia, J.D.; Cicerone, M.T. High-speed coherent Raman fingerprint imaging of biological tissues. Nat. Photonics 2014, 8, 627–634. [Google Scholar] [CrossRef]
- Liao, C.S.; Slipchenko, M.N.; Wang, P.; Li, J.; Lee, S.Y.; Oglesbee, R.A.; Cheng, J.X. Microsecond scale vibrational spectroscopic imaging by multiplex stimulated Raman scattering microscopy. Light Sci. Appl. 2015, 4, e265. [Google Scholar] [CrossRef]
- Yamakoshi, H.; Dodo, K.; Palonpon, A.; Ando, J.; Fujita, K.; Kawata, S.; Sodeoka, M. Alkyne-tag Raman imaging for visualization of mobile small molecules in live cells. J. Am. Chem. Soc. 2012, 134, 20681–20689. [Google Scholar] [CrossRef]
- Wei, L.; Hu, F.; Shen, Y.; Chen, Z.; Yu, Y.; Lin, C.C.; Wang, M.C.; Min, W. Live-cell imaging of alkyne-tagged small biomolecules by stimulated Raman scattering. Nat. Methods 2014, 11, 410–412. [Google Scholar] [CrossRef] [PubMed]
- Hosokawa, M.; Ando, M.; Mukai, S.; Osada, K.; Yoshino, T.; Hamaguchi, H.O.; Tanaka, T. In vivo live cell imaging for the quantitative monitoring of lipids by using raman microspectroscopy. Anal. Chem. 2014, 86, 8224–8230. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.J.; Zhang, W.; Zhang, D.; Yang, Y.; Liu, B.; Barker, E.L.; Buhman, K.K.; Slipchenko, L.V.; Dai, M.; Cheng, J.X. Assessing Cholesterol Storage in Live Cells and C. elegans by Stimulated Raman Scattering Imaging of Phenyl-Diyne Cholesterol. Sci. Rep. 2015, 5, 7930. [Google Scholar] [CrossRef] [PubMed]
- Tian, F.; Yang, W.; Mordes, D.A.; Wang, J.Y.; Salameh, J.S.; Mok, J.; Chew, J.; Sharma, A.; Leno-Duran, E.; Suzuki-Uematsu, S.; et al. Monitoring peripheral nerve degeneration in ALS by label-free stimulated Raman scattering imaging. Nat. Commun. 2016, 7, 13283. [Google Scholar] [CrossRef] [PubMed]
- Shen, Y.; Xu, F.; Wei, L.; Hu, F.; Min, W. Live-cell quantitative imaging of proteome degradation by stimulated raman scattering. Angew. Chem. Int. Ed. 2014, 53, 5596–5599. [Google Scholar] [CrossRef]
- Hu, F.; Wei, L.; Zheng, C.; Shen, Y.; Min, W. Live-cell vibrational imaging of choline metabolites by stimulated Raman scattering coupled with isotope-based metabolic labeling. Analyst 2014, 139, 2312–2317. [Google Scholar] [CrossRef]
- Zhang, D.; Slipchenko, M.N.; Cheng, J.X. Highly sensitive vibrational imaging by femtosecond pulse stimulated raman loss. J. Phys. Chem. Lett. 2011, 2, 1248–1253. [Google Scholar] [CrossRef]
- Hong, W.; Karanja, C.W.; Abutaleb, N.S.; Younis, W.; Zhang, X.; Seleem, M.N.; Cheng, J.X. Antibiotic Susceptibility Determination within One Cell Cycle at Single-Bacterium Level by Stimulated Raman Metabolic Imaging. Anal. Chem. 2018, 90, 3737–3743. [Google Scholar] [CrossRef]
- Karanja, C.W.; Hong, W.; Younis, W.; Eldesouky, H.E.; Seleem, M.N.; Cheng, J.X. Stimulated Raman Imaging Reveals Aberrant Lipogenesis as a Metabolic Marker for Azole-Resistant Candida albicans. Anal. Chem. 2017, 89, 9822–9829. [Google Scholar] [CrossRef]
- Palonpon, A.F.; Sodeoka, M.; Fujita, K. Molecular imaging of live cells by Raman microscopy. Curr. Opin. Chem. Biol. 2013, 17, 708–715. [Google Scholar] [CrossRef]
- Klein, K.; Gigler, A.M.; Aschenbrenner, T.; Monetti, R.; Bunk, W.; Jamitzky, F.; Morfill, G.; Stark, R.W.; Schlegel, J. Label-free live-cell imaging with confocal Raman microscopy. Biophys. J. 2012, 102, 360–368. [Google Scholar] [CrossRef] [PubMed]
- Harada, Y.; Dai, P.; Yamaoka, Y.; Ogawa, M.; Tanaka, H.; Nosaka, K.; Akaji, K.; Takamatsu, T. Intracellular dynamics of topoisomerase i inhibitor, CPT-11, by slit-scanning confocal Raman microscopy. Histochem. Cell Biol. 2009, 132, 39–46. [Google Scholar] [CrossRef] [PubMed]
- El-Mashtoly, S.F.; Petersen, D.; Yosef, H.K.; Mosig, A.; Reinacher-Schick, A.; Kötting, C.; Gerwert, K. Label-free imaging of drug distribution and metabolism in colon cancer cells by Raman microscopy. Analyst 2014, 139, 1155–1161. [Google Scholar] [CrossRef] [PubMed]
- Harkness, L.; Novikov, S.M.; Beermann, J.; Bozhevolnyi, S.I.; Kassem, M. Identification of Abnormal Stem Cells Using Raman Spectroscopy. Stem Cells Dev. 2012, 21, 2152–2159. [Google Scholar] [CrossRef] [PubMed]
- Vanna, R.; Ronchi, P.; Lenferink, A.T.M.; Tresoldi, C.; Morasso, C.; Mehn, D.; Bedoni, M.; Picciolini, S.; Terstappen, L.W.M.M.; Ciceri, F.; et al. Label-free imaging and identification of typical cells of acute myeloid leukaemia and myelodysplastic syndrome by Raman microspectroscopy. Analyst 2015, 140, 1054–1064. [Google Scholar] [CrossRef]
- Kaliaperumal, V.; Hamaguchi, H.O. Casting new physicochemical light on the fundamental biological processes in single living cells by using Raman microspectroscopy. Chem. Rec. 2012, 12, 567–580. [Google Scholar] [CrossRef]
- Kottke, T.; Xie, A.; Larsen, D.S.; Hoff, W.D. Photoreceptors Take Charge: Emerging Principles for Light Sensing. Annu. Rev. Biophys. 2018, 47, 291–313. [Google Scholar] [CrossRef]
- Boyden, E.S.; Zhang, F.; Bamberg, E.; Nagel, G.; Deisseroth, K. Millisecond-timescale, genetically targeted optical control of neural activity. Nat. Neurosci. 2005, 8, 1263–1268. [Google Scholar] [CrossRef]
- Ernst, O.P.; Lodowski, D.T.; Elstner, M.; Hegemann, P.; Brown, L.S.; Kandori, H. Microbial and Animal Rhodopsins: Structures, Functions, and Molecular Mechanisms. Chem. Rev. 2014, 114, 126–163. [Google Scholar] [CrossRef]
- Gushchin, I.; Gordeliy, V. Microbial rhodopsins. In Subcellular Biochemistry; Springer: New York, NY, USA, 2018; Volume 87, pp. 19–56. [Google Scholar]
- Bratanov, D.; Kovalev, K.; Machtens, J.P.; Astashkin, R.; Chizhov, I.; Soloviov, D.; Volkov, D.; Polovinkin, V.; Zabelskii, D.; Mager, T.; et al. Unique structure and function of viral rhodopsins. Nat. Commun. 2019, 10, 1–13. [Google Scholar] [CrossRef]
- Kovalev, K.; Polovinkin, V.; Gushchin, I.; Alekseev, A.; Shevchenko, V.; Borshchevskiy, V.; Astashkin, R.; Balandin, T.; Bratanov, D.; Vaganova, S.; et al. Structure and mechanisms of sodium-pumping KR2 rhodopsin. Sci. Adv. 2019, 5, eaav2671. [Google Scholar] [CrossRef] [PubMed]
- Kovalev, K.; Volkov, D.; Astashkin, R.; Alekseev, A.; Gushchin, I.; Haro-Moreno, J.M.; Rogachev, A.; Balandin, T.; Borshchevskiy, V.; Popov, A.; et al. High Resolution Structural Insights into Heliorhodopsin Family. bioRxiv 2019. bioRxiv:767665. [Google Scholar]
- Ishchenko, A.; Round, E.; Borshchevskiy, V.; Grudinin, S.; Gushchin, I.; Klare, J.P.; Remeeva, A.; Polovinkin, V.; Utrobin, P.; Balandin, T.; et al. New Insights on Signal Propagation by Sensory Rhodopsin II/Transducer Complex. Sci. Rep. 2017, 7, 41811. [Google Scholar] [CrossRef]
- Gushchin, I.; Shevchenko, V.; Polovinkin, V.; Borshchevskiy, V.; Buslaev, P.; Bamberg, E.; Gordeliy, V. Structure of the light-driven sodium pump KR2 and its implications for optogenetics. FEBS J. 2016, 283, 1232–1238. [Google Scholar] [CrossRef] [PubMed]
- Gushchin, I.; Shevchenko, V.; Polovinkin, V.; Kovalev, K.; Alekseev, A.; Round, E.; Borshchevskiy, V.; Balandin, T.; Popov, A.; Gensch, T.; et al. Crystal structure of a light-driven sodium pump. Nat. Struct. Mol. Biol. 2015, 22, 390–395. [Google Scholar] [CrossRef] [PubMed]
- Borshchevskiy, V.; Round, E.; Erofeev, I.; Weik, M.; Ishchenko, A.; Gushchin, I.; Mishin, A.; Willbold, D.; Büldt, G.; Gordeliy, V. Low-dose X-ray radiation induces structural alterations in proteins. Acta Crystallogr. Sect. D Biol. Crystallogr. 2014, 70, 2675–2685. [Google Scholar] [CrossRef] [PubMed]
- Yi, A.; Mamaeva, N.; Li, H.; Spudich, J.L.; Rothschild, K.J. Resonance Raman Study of an Anion Channelrhodopsin: Effects of Mutations near the Retinylidene Schiff Base. Biochemistry 2016, 55, 2371–2380. [Google Scholar] [CrossRef]
- Stoeckenius, W.; Bogomolni, R.A. Bacteriorhodopsin and Related Pigments of Halo Bacteria. Ann. Rev. Biochem. 1982, 51, 587–616. [Google Scholar] [CrossRef]
- Dencher, N.; Wilms, M. Flash photometric experiments on the photochemical cycle of bacteriorhodopsin. Biophys. Struct. Mech. 1975, 1, 259–271. [Google Scholar] [CrossRef]
- Lozier, R.H.; Bogomolni, R.A.; Stoeckenius, W. Bacteriorhodopsin: A light-driven proton pump in Halobacterium Halobium. Biophys. J. 1975, 15, 955–962. [Google Scholar] [CrossRef]
- Smith, S.O.; Lugtenburg, J.; Mathies, R.A. Determination of retinal chromophore structure in bacteriorhodopsin with resonance Raman spectroscopy. J. Membr. Biol. 1985, 85, 95–109. [Google Scholar] [CrossRef] [PubMed]
- Lewis, A.; Spoonhower, J.; Bogomolni, R.A.; Lozier, R.H.; Stoeckenius, W. Tunable Laser Resonance Raman Spectroscopy of Bacteriorhodopsin. Proc. Natl. Acad. Sci. USA 1974, 71, 4462–4466. [Google Scholar] [CrossRef] [PubMed]
- Braiman, M.; Mathies, R. Resonance Raman spectra of bacteriorhodopsin’s primary photoproduct: Evidence for a distorted 13-cis retinal chromophore. Proc. Natl. Acad. Sci. USA 1982, 79, 403–407. [Google Scholar] [CrossRef] [PubMed]
- Aton, B.; Doukas, A.G.; Callender, R.H.; Becher, B.; Ebrey, T.G. Resonance Raman studies of the purple membrane. Biochemistry 1977, 16, 2995–2999. [Google Scholar] [CrossRef] [PubMed]
- Polovinkin, V.; Balandin, T.; Volkov, O.; Round, E.; Borshchevskiy, V.; Utrobin, P.; von Stetten, D.; Royant, A.; Willbold, D.; Arzumanyan, G.; et al. Nanoparticle Surface-Enhanced Raman Scattering of Bacteriorhodopsin Stabilized by Amphipol A8-35. J. Membr. Biol. 2014, 247, 971–980. [Google Scholar] [CrossRef] [PubMed]
- Mizuno, M.; Mizutani, Y. Protein Response to Chromophore Isomerization in Microbial Rhodopsins Revealed by Picosecond Time-Resolved Ultraviolet Resonance Raman Spectroscopy: A Review. In Recent Progress in Colloid and Surface Chemistry with Biological Applications; American Chemical Society: Washington, DC, USA, 2015; pp. 329–353. [Google Scholar]
- Kralj, J.M.; Spudich, E.N.; Spudich, J.L.; Rothschild, K.J. Raman Spectroscopy Reveals Direct Chromophore Interactions in the Leu/Gln105 Spectral Tuning Switch of Proteorhodopsins. J. Phys. Chem. B 2008, 112, 11770–11776. [Google Scholar] [CrossRef]
- Kajimoto, K.; Kikukawa, T.; Nakashima, H.; Yamaryo, H.; Saito, Y.; Fujisawa, T.; Demura, M.; Unno, M. Transient Resonance Raman Spectroscopy of a Light-Driven Sodium-Ion-Pump Rhodopsin from Indibacter alkaliphilus. J. Phys. Chem. B 2017, 121, 4431–4437. [Google Scholar] [CrossRef]
- Nack, M.; Radu, I.; Bamann, C.; Bamberg, E.; Heberle, J. The retinal structure of channelrhodopsin-2 assessed by resonance Raman spectroscopy. FEBS Lett. 2009, 583, 3676–3680. [Google Scholar] [CrossRef]
- Volkov, O.; Kovalev, K.; Polovinkin, V.; Borshchevskiy, V.; Bamann, C.; Astashkin, R.; Marin, E.; Popov, A.; Balandin, T.; Willbold, D.; et al. Structural insights into ion conduction by channelrhodopsin 2. Science 2017, 358, eaan8862. [Google Scholar] [CrossRef]
- Hontani, Y.; Inoue, K.; Kloz, M.; Kato, Y.; Kandori, H.; Kennis, J.T.M. The photochemistry of sodium ion pump rhodopsin observed by watermarked femto- to submillisecond stimulated Raman spectroscopy. Phys. Chem. Chem. Phys. 2016, 18, 24729–24736. [Google Scholar] [CrossRef]
- Pushkarev, A.; Inoue, K.; Larom, S.; Flores-Uribe, J.; Singh, M.; Konno, M.; Tomida, S.; Ito, S.; Nakamura, R.; Tsunoda, S.P.; et al. A distinct abundant group of microbial rhodopsins discovered using functional metagenomics. Nature 2018, 558, 595–599. [Google Scholar] [CrossRef] [PubMed]
- Otomo, A.; Mizuno, M.; Singh, M.; Shihoya, W.; Inoue, K.; Nureki, O.; Béjà, O.; Kandori, H.; Mizutani, Y. Resonance Raman Investigation of the Chromophore Structure of Heliorhodopsins. J. Phys. Chem. Lett. 2018, 9, 6431–6436. [Google Scholar] [CrossRef]
- Gordeliy, V.I.; Labahn, J.; Moukhametzianov, R.; Efremov, R.; Granzin, J.; Schlesinger, R.; Büldt, G.; Savopol, T.; Scheidig, A.J.; Klare, J.P.; et al. Molecular basis of transmembrane signalling by sensory rhodopsin II–transducer complex. Nature 2002, 419, 484–487. [Google Scholar] [CrossRef] [PubMed]
- Mizuno, M.; Sudo, Y.; Homma, M.; Mizutani, Y. Direct Observation of the Structural Change of Tyr174 in the Primary Reaction of Sensory Rhodopsin II. Biochemistry 2011, 50, 3170–3180. [Google Scholar] [CrossRef] [PubMed]
- Roy, P.P.; Kato, Y.; Abe-Yoshizumi, R.; Pieri, E.; Ferré, N.; Kandori, H.; Buckup, T. Mapping the ultrafast vibrational dynamics of all-: Trans and 13- cis retinal isomerization in Anabaena Sensory Rhodopsin. Phys. Chem. Chem. Phys. 2018, 20, 30159–30173. [Google Scholar] [CrossRef] [PubMed]
- Roy, P.P.; Abe-Yoshizumi, R.; Kandori, H.; Buckup, T. Point Mutation of Anabaena Sensory Rhodopsin Enhances Ground-State Hydrogen Out-of-Plane Wag Raman Activity. J. Phys. Chem. Lett. 2019, 10, 1012–1017. [Google Scholar] [CrossRef] [PubMed]
- Morizumi, T.; Ou, W.L.; Van Eps, N.; Inoue, K.; Kandori, H.; Brown, L.S.; Ernst, O.P. X-ray Crystallographic Structure and Oligomerization of Gloeobacter Rhodopsin. Sci. Rep. 2019, 9, 11283. [Google Scholar] [CrossRef]
- Harris, A.; Ljumovic, M.; Bondar, A.N.; Shibata, Y.; Ito, S.; Inoue, K.; Kandori, H.; Brown, L.S. A new group of eubacterial light-driven retinal-binding proton pumps with an unusual cytoplasmic proton donor. Biochim. Biophys. Acta Bioenerg. 2015, 1847, 1518–1529. [Google Scholar] [CrossRef]
- Ogren, J.I.; Mamaev, S.; Russano, D.; Li, H.; Spudich, J.L.; Rothschild, K.J. Retinal Chromophore Structure and Schiff Base Interactions in Red-Shifted Channelrhodopsin-1 from Chlamydomonas augustae. Biochemistry 2014, 53, 3961–3970. [Google Scholar] [CrossRef]
- Muders, V.; Kerruth, S.; Lórenz-Fonfría, V.A.; Bamann, C.; Heberle, J.; Schlesinger, R. Resonance Raman and FTIR spectroscopic characterization of the closed and open states of channelrhodopsin-1. FEBS Lett. 2014, 588, 2301–2306. [Google Scholar] [CrossRef]
- Kukura, P.; McCamant, D.W.; Yoon, S.; Wandschneider, D.B.; Mathies, R.A. Chemistry: Structural observation of the primary isomerization in vision with femtosecond-stimulated Raman. Science 2005, 310, 1006–1009. [Google Scholar] [CrossRef] [PubMed]
- Popp, A.; Ujj, L.; Atkinson, G.H. Bathorhodopsin structure in the room-temperature rhodopsin photosequence: Picosecond time-resolved coherent anti-stokes Raman scattering. Proc. Natl. Acad. Sci. USA 1996, 93, 372–376. [Google Scholar] [CrossRef]
- Matysik, J.; Hildebrandt, P.; Schlamann, W.; Braslavsky, S.E.; Schaffner, K. Fourier-Transform Resonance Raman Spectroscopy of Intermediates of the Phytochrome Photocycle. Biochemistry 1995, 34, 10497–10507. [Google Scholar] [CrossRef] [PubMed]
- Velazquez Escobar, F.; von Stetten, D.; Günther-Lütkens, M.; Keidel, A.; Michael, N.; Lamparter, T.; Essen, L.-O.; Hughes, J.; Gärtner, W.; Yang, Y.; et al. Conformational heterogeneity of the Pfr chromophore in plant and cyanobacterial phytochromes. Front. Mol. Biosci. 2015, 2, 37. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Spillane, K.M.; Dasgupta, J.; Lagarias, J.C.; Mathies, R.A. Homogeneity of Phytochrome Cph1 Vibronic Absorption Revealed by Resonance Raman Intensity Analysis. J. Am. Chem. Soc. 2009, 131, 13946–13948. [Google Scholar] [CrossRef] [PubMed]
- Unno, M.; Kikuchi, S.; Masuda, S. Structural refinement of a key tryptophan residue in the BLUF photoreceptor AppA by ultraviolet resonance raman spectroscopy. Biophys. J. 2010, 98, 1949–1956. [Google Scholar] [CrossRef][Green Version]
- Hontani, Y.; Ganapathy, S.; Frehan, S.; Kloz, M.; de Grip, W.J.; Kennis, J.T.M. Strong pH-Dependent Near-Infrared Fluorescence in a Microbial Rhodopsin Reconstituted with a Red-Shifting Retinal Analogue. J. Phys. Chem. Lett. 2018, 9, 6469–6474. [Google Scholar] [CrossRef]
- Mei, G.; Mamaeva, N.; Ganapathy, S.; Wang, P.; DeGrip, W.J.; Rothschild, K.J. Raman spectroscopy of a near infrared absorbing proteorhodopsin: Similarities to the bacteriorhodopsin O photointermediate. PLoS ONE 2018, 13, e0209506. [Google Scholar] [CrossRef]
- Gonzalez-Perez, A.; Mosgaard, L.D.; Budvytyte, R.; Villagran-Vargas, E.; Jackson, A.D.; Heimburg, T. Solitary electromechanical pulses in lobster neurons. Biophys. Chem. 2016, 216, 51–59. [Google Scholar] [CrossRef]
- Graf, B.W.; Ralston, T.S.; Ko, H.-J.; Boppart, S.A. Detecting intrinsic scattering changes correlated to neuron action potentials using optical coherence imaging. Opt. Express 2009, 17, 13447. [Google Scholar] [CrossRef]
- Liu, B.; Lee, H.J.; Zhang, D.; Liao, C.S.; Ji, N.; Xia, Y.; Cheng, J.X. Label-free spectroscopic detection of membrane potential using stimulated Raman scattering. Appl. Phys. Lett. 2015, 106, 173704. [Google Scholar] [CrossRef]
- Lee, H.J.; Cheng, J.-X.; Huang, K.-C.; Mei, G.; Mamaeva, N.; de Grip, W.J.; Rothschild, K.J. Pre-resonance stimulated Raman scattering spectroscopy and imaging of membrane potential using near-infrared rhodopsins. In Proceedings of the Multiphoton Microscopy in the Biomedical Sciences XIX, San Francisco, CA, USA, 2–7 February 2019; SPIE: San Francisco, CA, USA, 2019; p. 81. [Google Scholar]
- Lee, H.J.; Jiang, Y.; Cheng, J.-X. Label-free Optical Imaging of Membrane Potential. Curr. Opin. Biomed. Eng. 2019, 12, 118–125. [Google Scholar] [CrossRef]
- Huang, W. Overproduction of proteorhodopsin enhances long-term viability of Escherichia coli. Appl. Environ. Microbiol. 2019. [Google Scholar] [CrossRef]
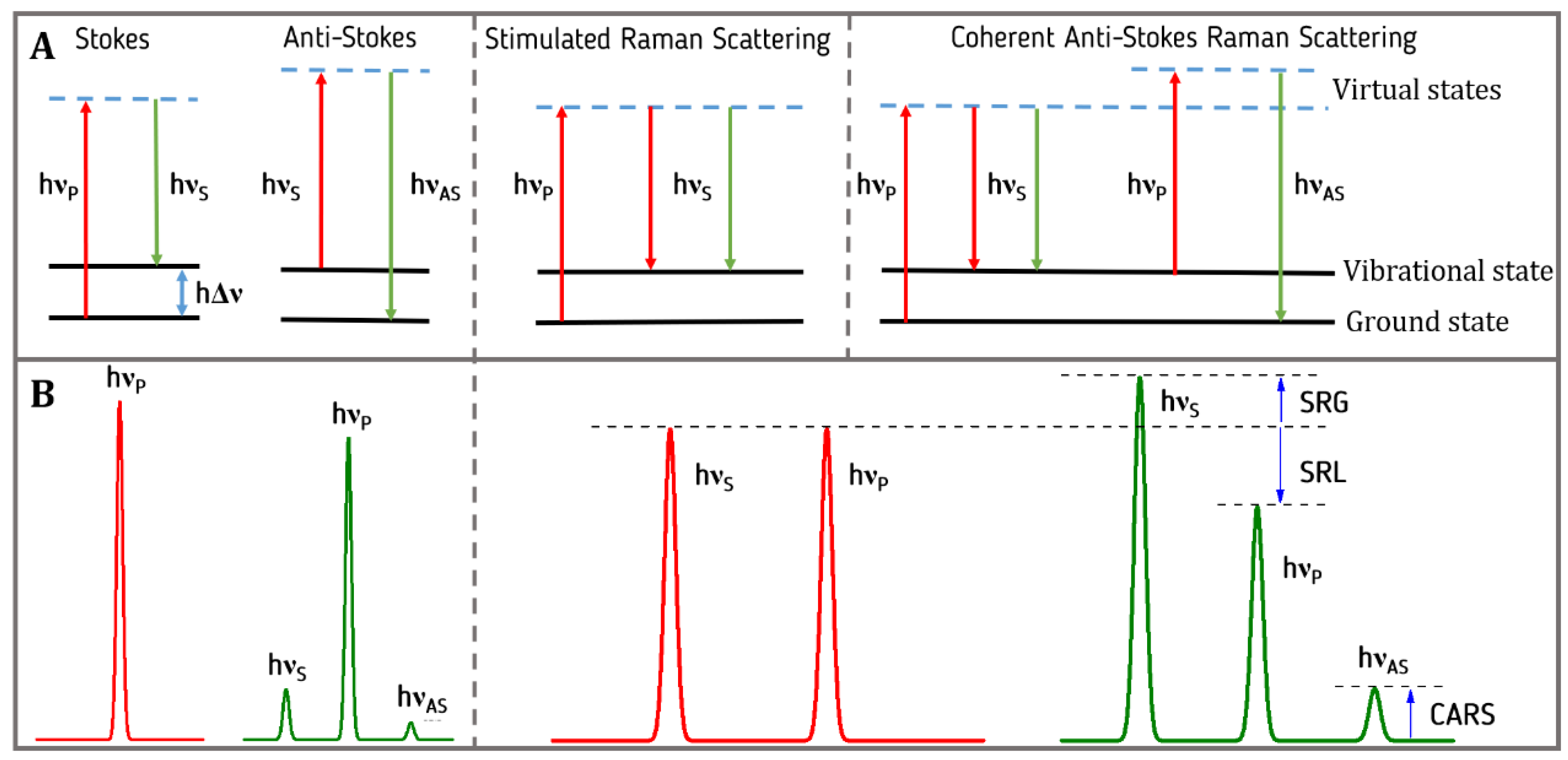
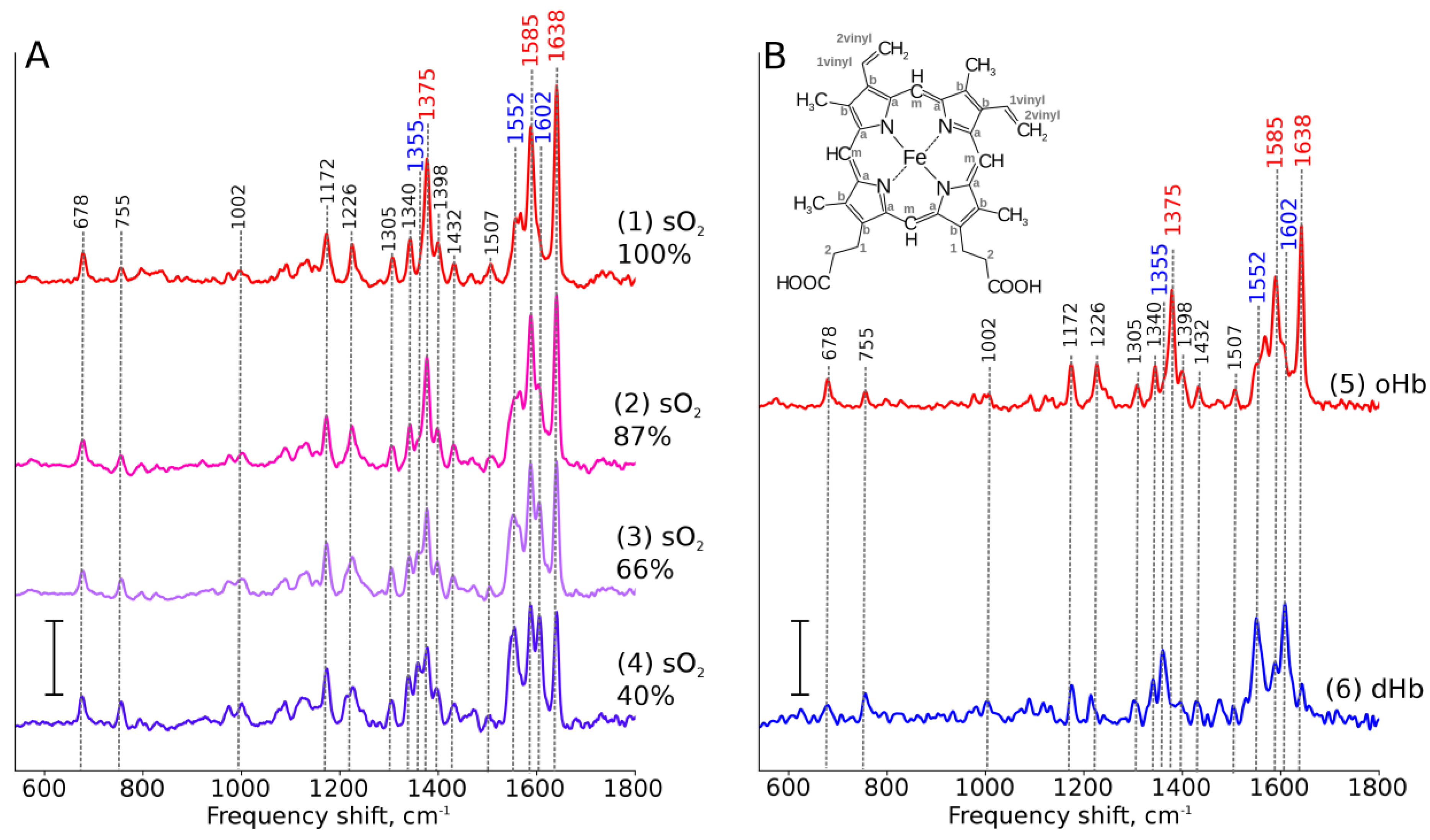
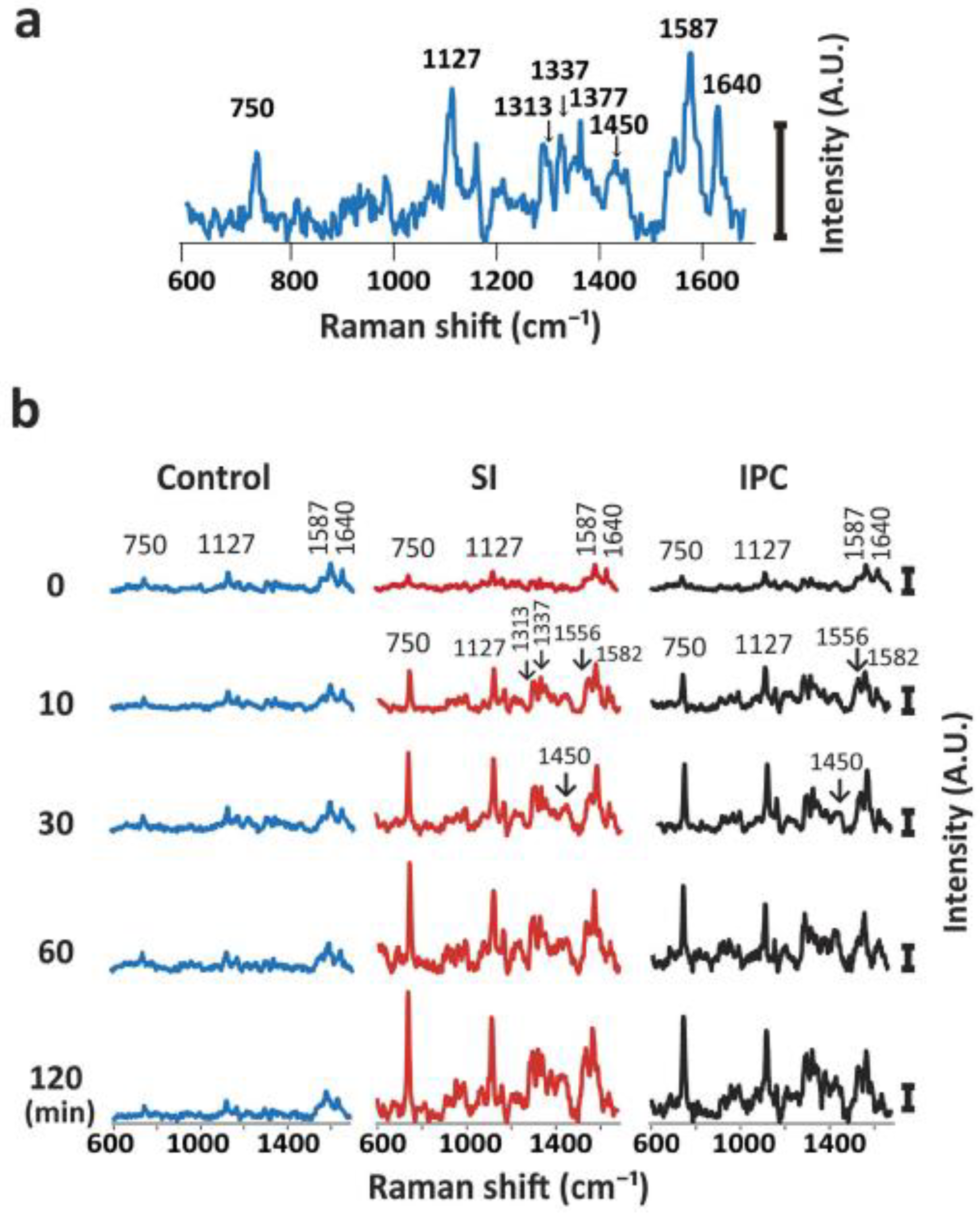
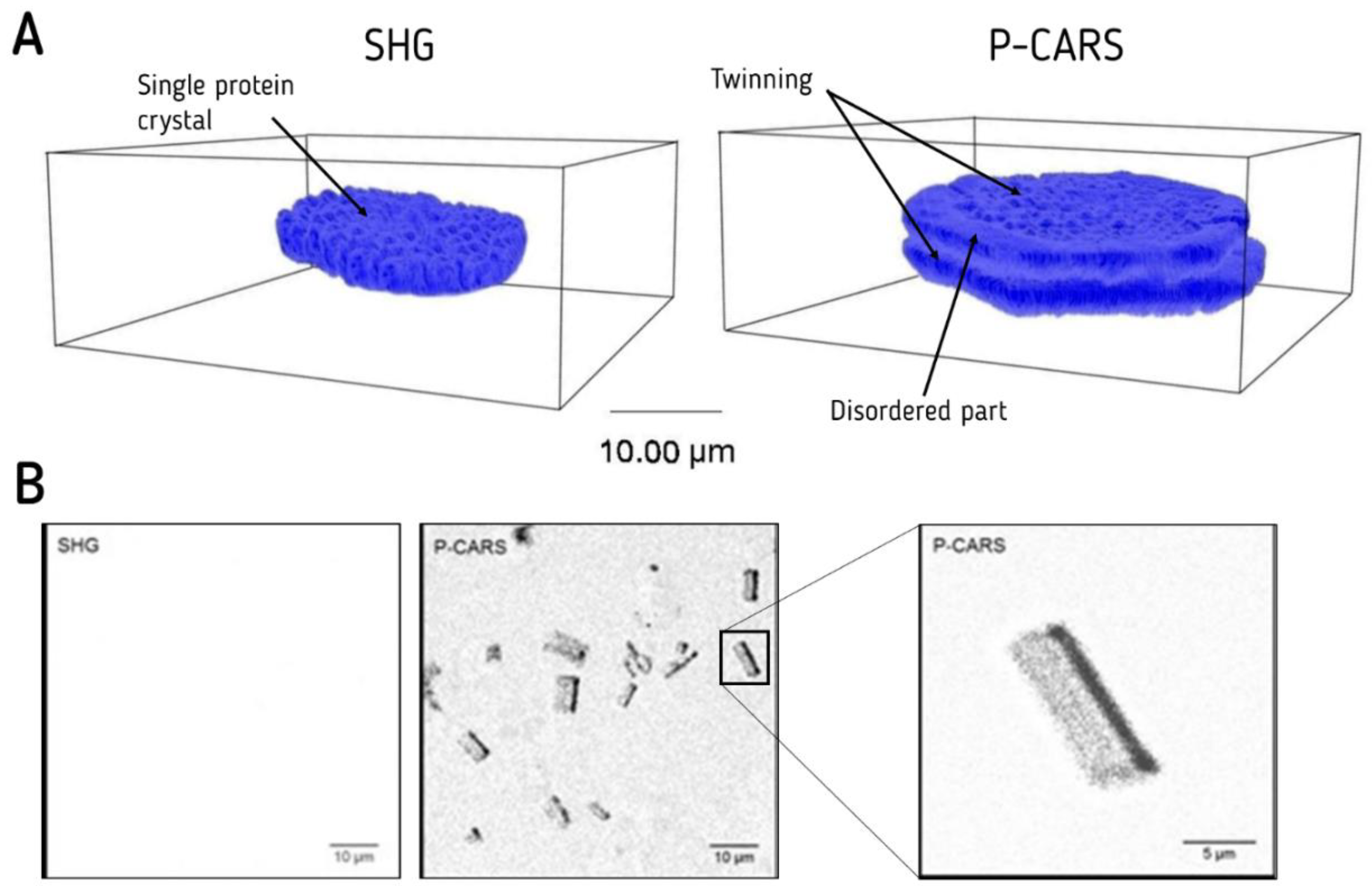
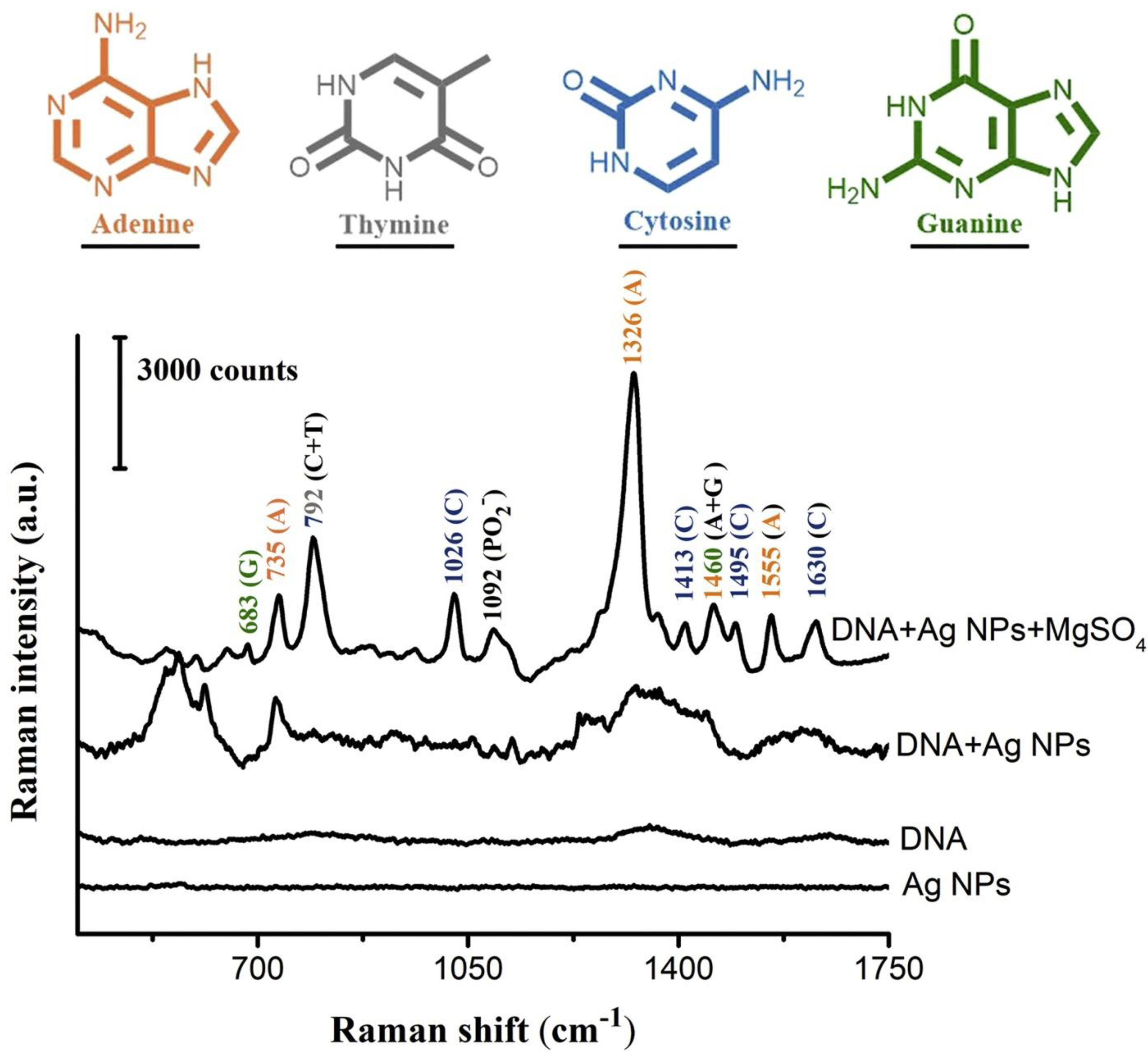

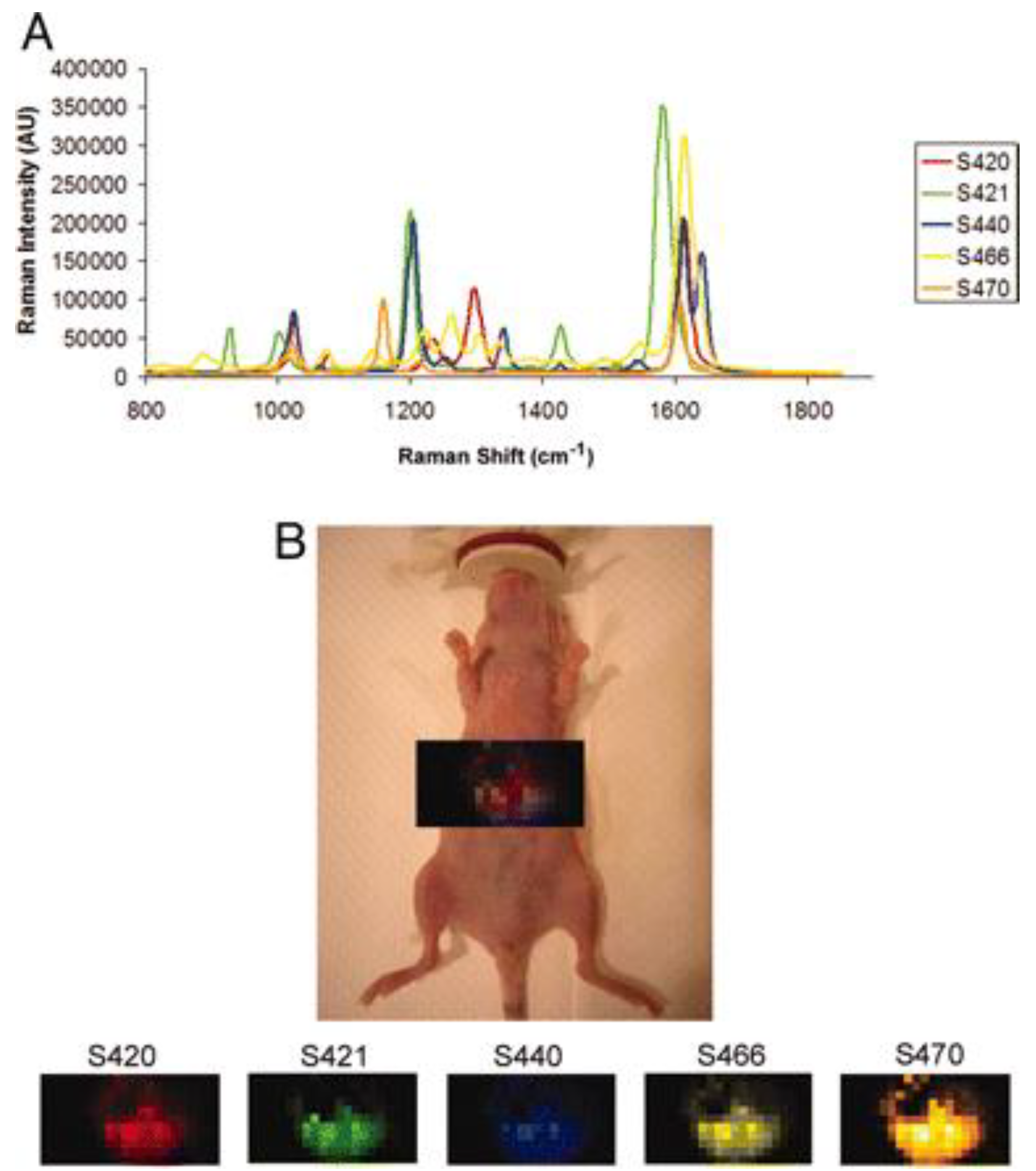
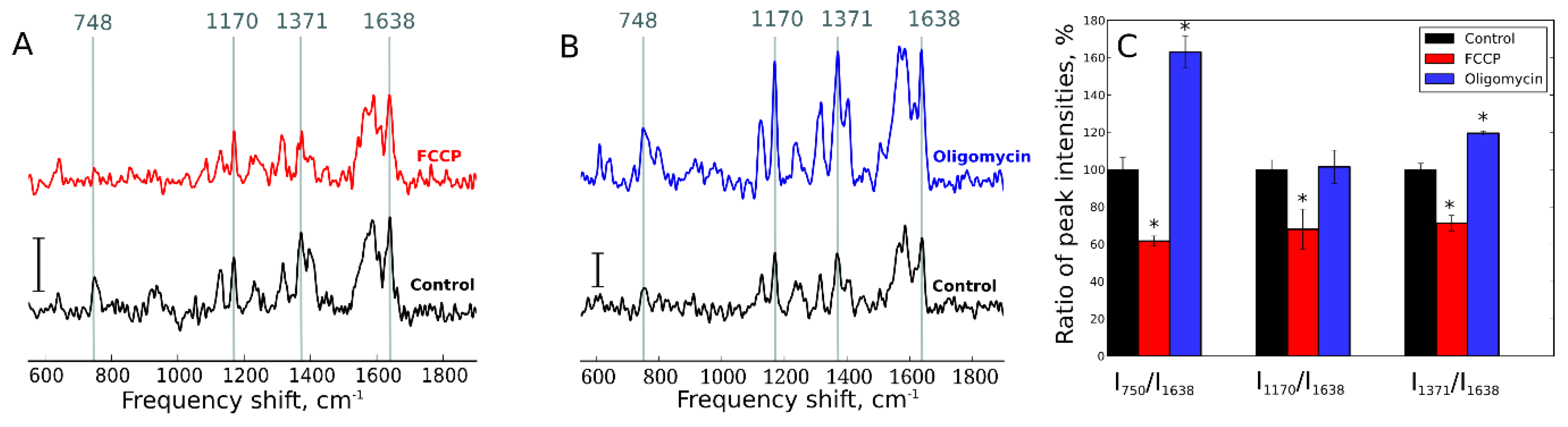
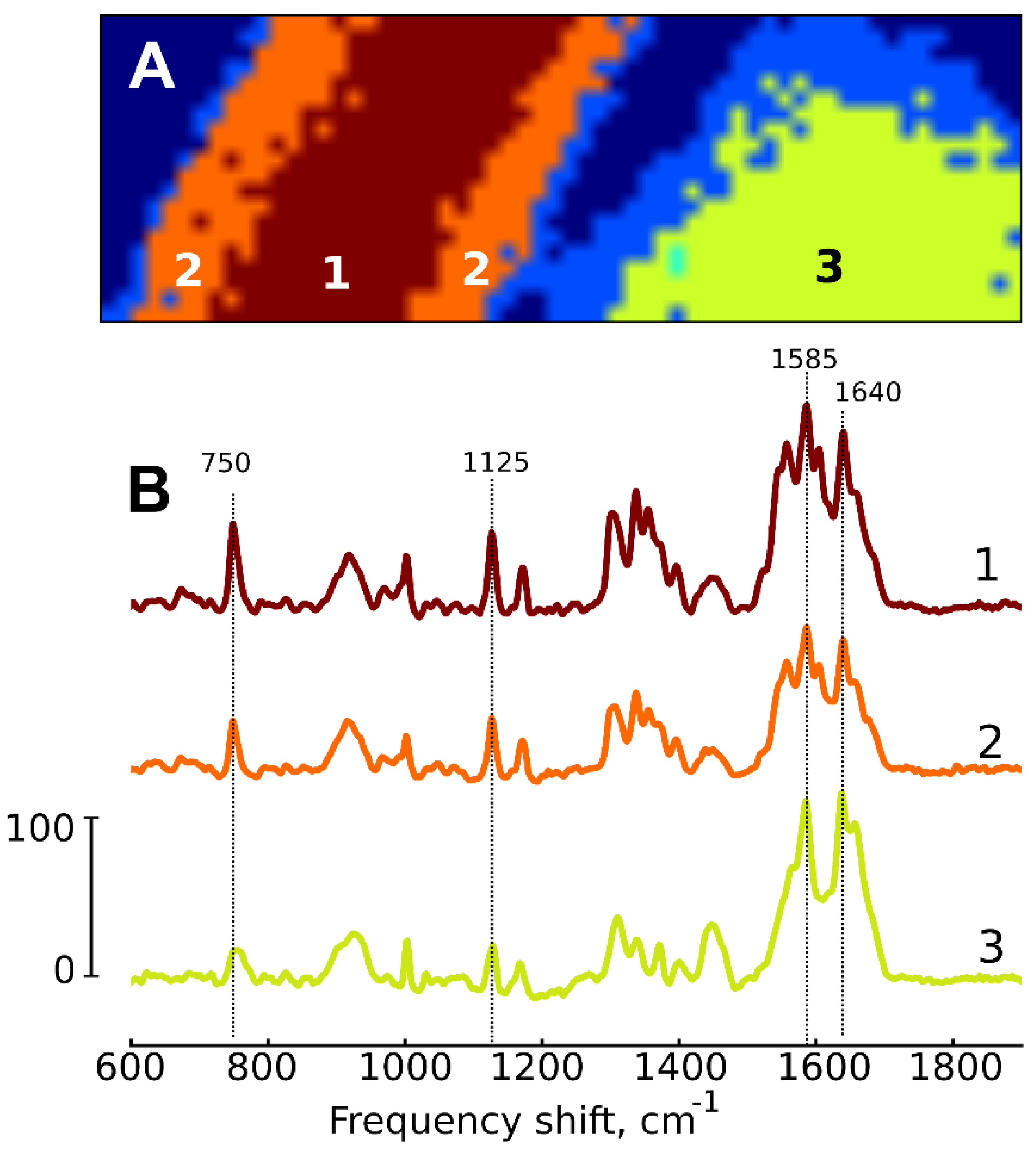
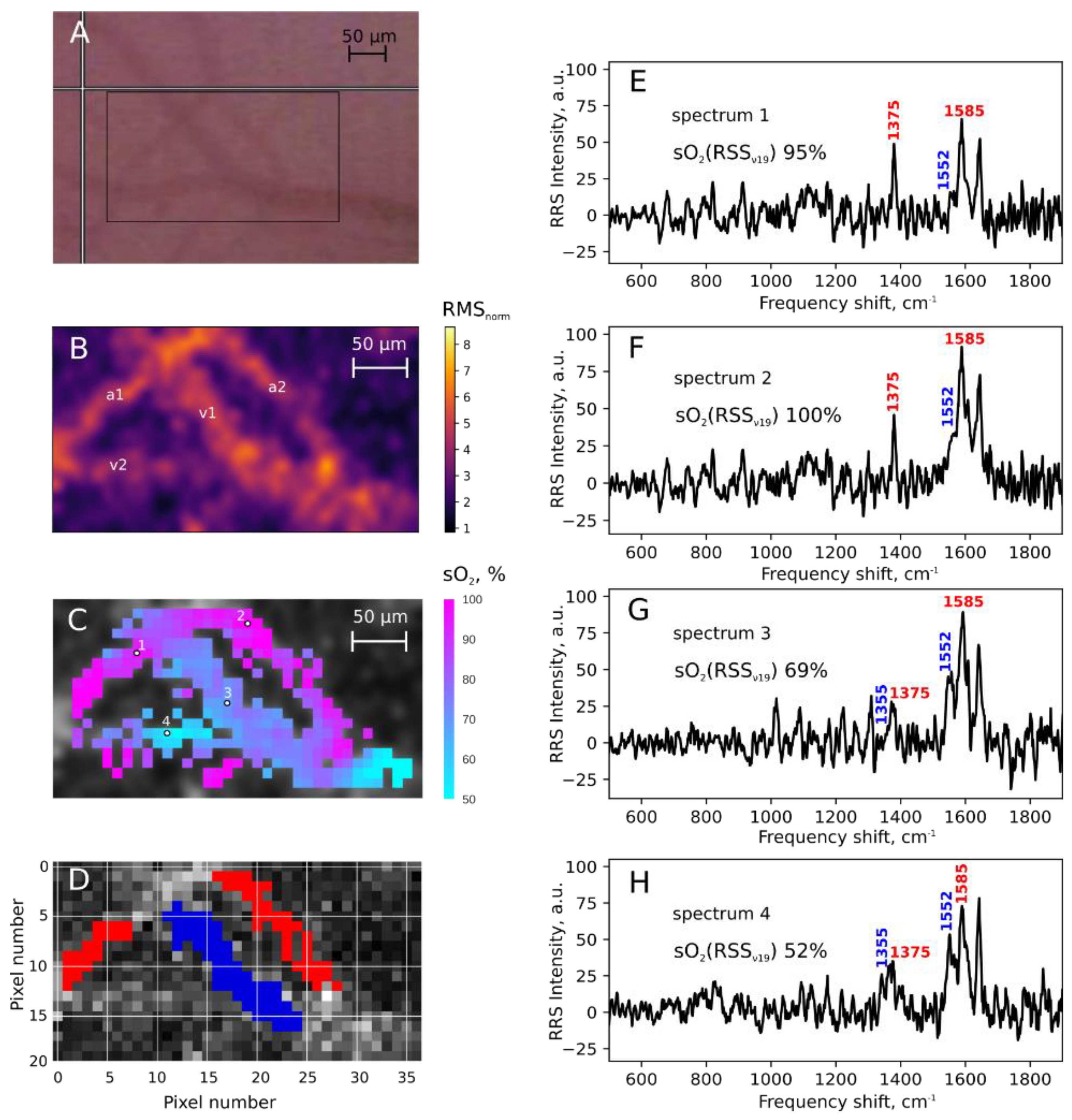
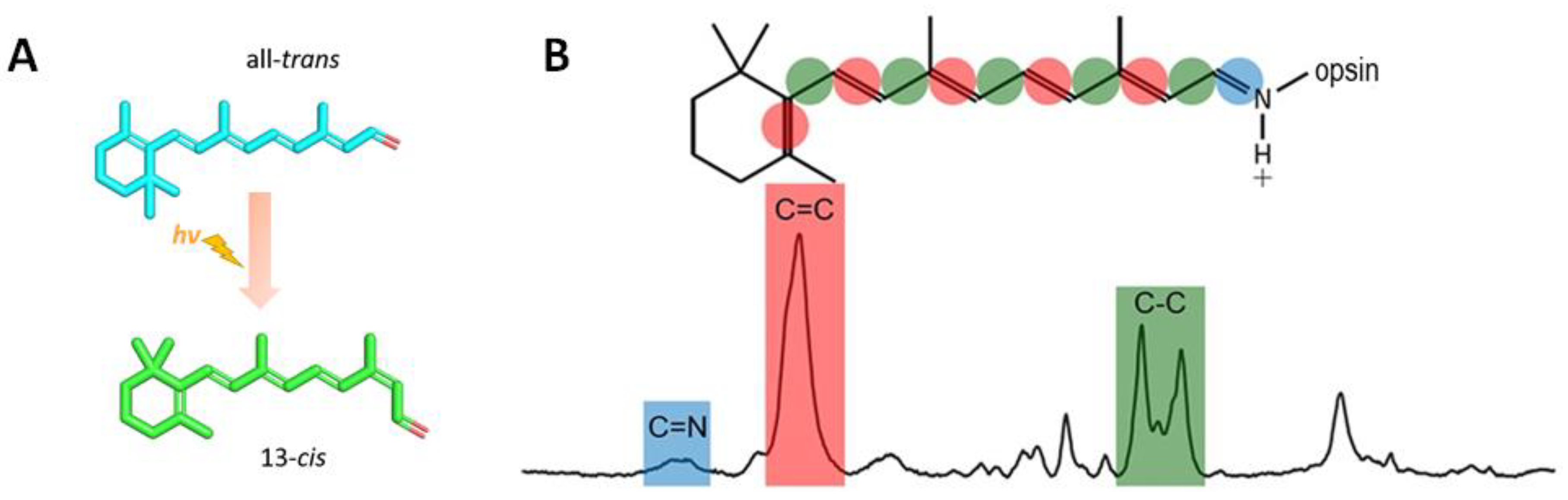
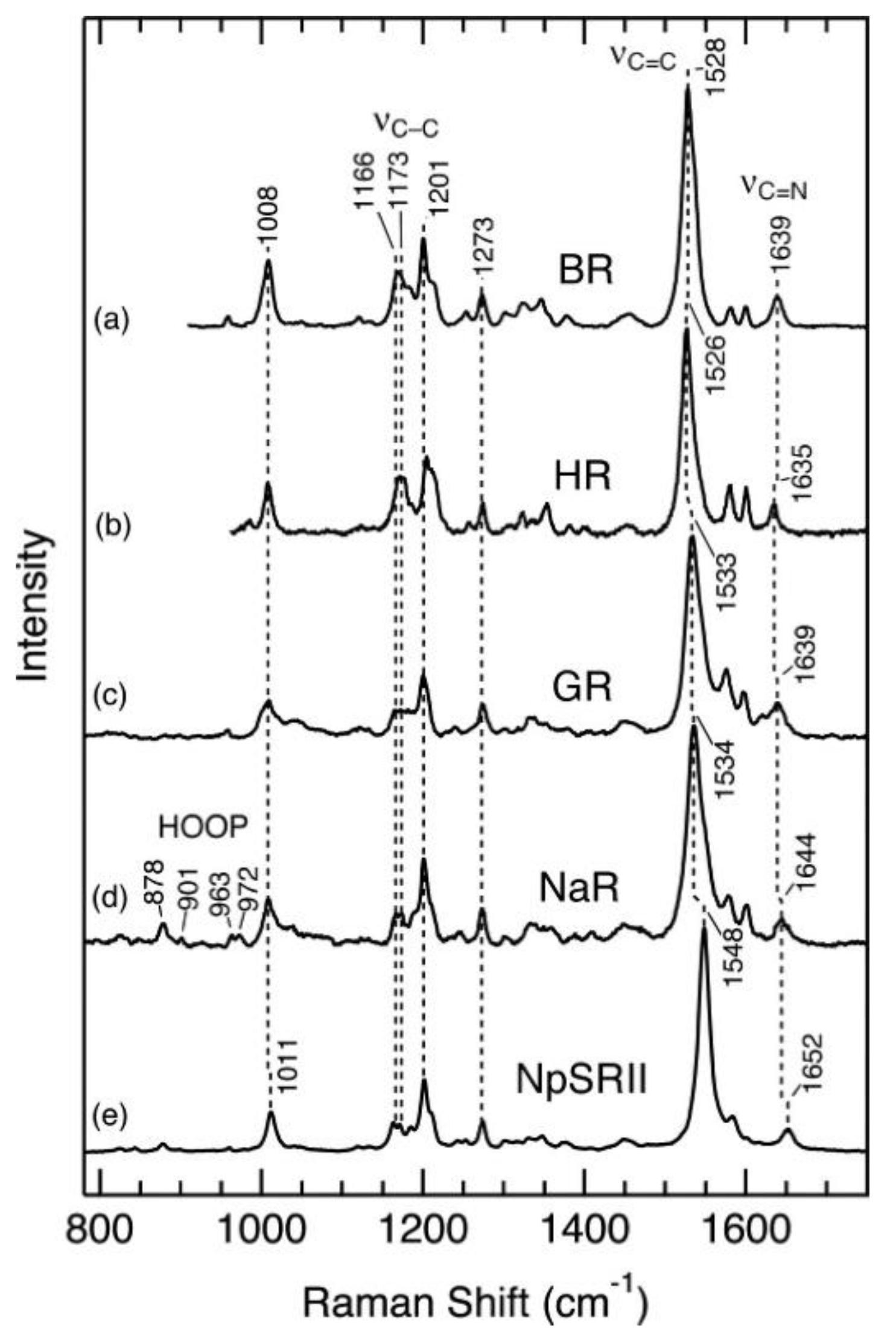
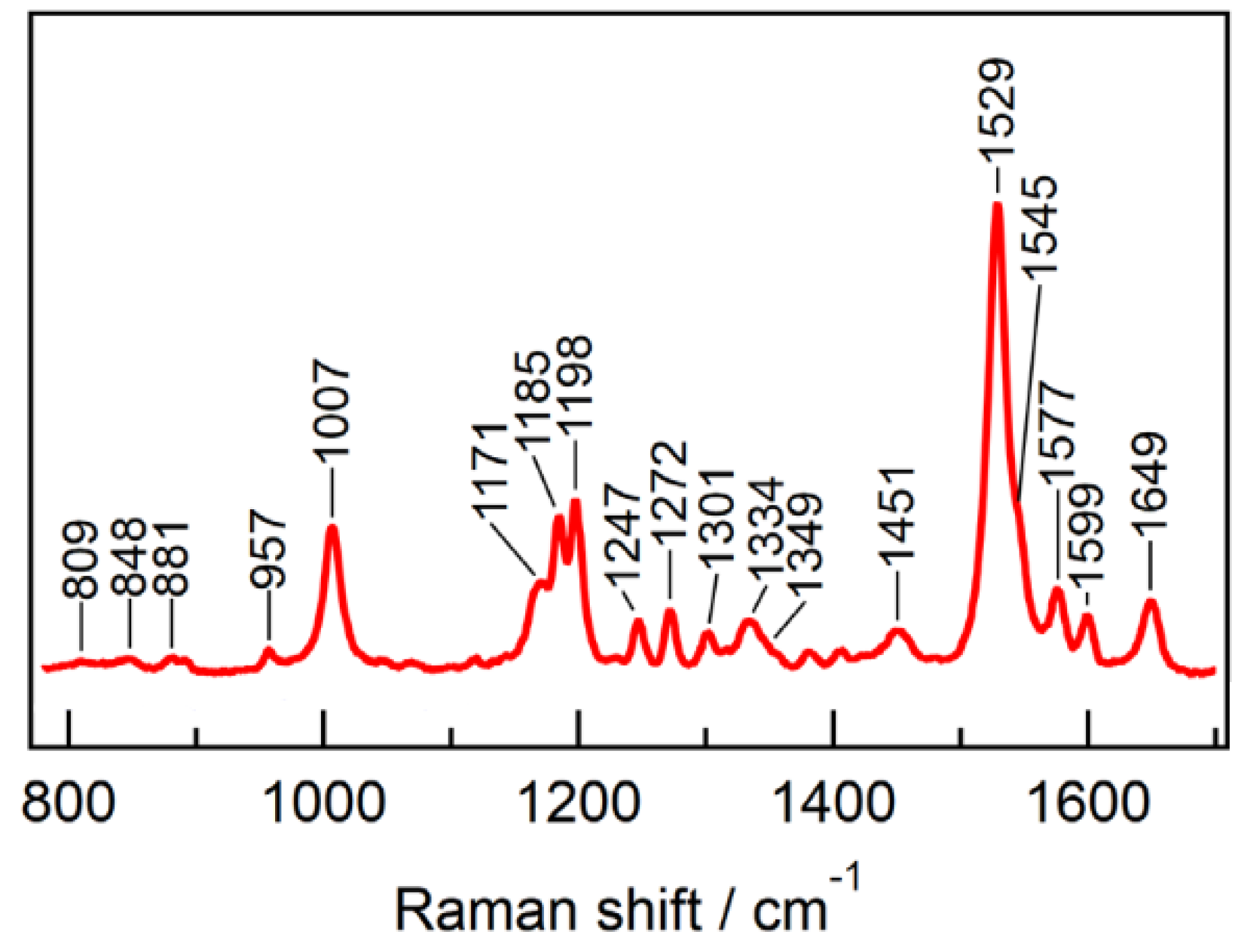
Retinal | FAD | Bilin | ||||||
|---|---|---|---|---|---|---|---|---|
| Vertebrate Rhodopsins | Channel Rhodopsins | Sensory Rhodopsins | Proteo-Rhodopsins | BR | HeRs | NaR | BLUF | Phytochrome |
| fs SRS ps TR-CARS | RRS Confocal near-IR RRS | UVRR DFWM | Near-IR RR | RS RRS SERS CARS TR-UVRR Near-IR RRS | RR | SRS Transient RRS | UVRR | Fourier transform RRS RRS |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vlasov, A.V.; Maliar, N.L.; Bazhenov, S.V.; Nikelshparg, E.I.; Brazhe, N.A.; Vlasova, A.D.; Osipov, S.D.; Sudarev, V.V.; Ryzhykau, Y.L.; Bogorodskiy, A.O.; et al. Raman Scattering: From Structural Biology to Medical Applications. Crystals 2020, 10, 38. https://doi.org/10.3390/cryst10010038
Vlasov AV, Maliar NL, Bazhenov SV, Nikelshparg EI, Brazhe NA, Vlasova AD, Osipov SD, Sudarev VV, Ryzhykau YL, Bogorodskiy AO, et al. Raman Scattering: From Structural Biology to Medical Applications. Crystals. 2020; 10(1):38. https://doi.org/10.3390/cryst10010038
Chicago/Turabian StyleVlasov, Alexey V., Nina L. Maliar, Sergey V. Bazhenov, Evelina I. Nikelshparg, Nadezda A. Brazhe, Anastasiia D. Vlasova, Stepan D. Osipov, Vsevolod V. Sudarev, Yury L. Ryzhykau, Andrey O. Bogorodskiy, and et al. 2020. "Raman Scattering: From Structural Biology to Medical Applications" Crystals 10, no. 1: 38. https://doi.org/10.3390/cryst10010038
APA StyleVlasov, A. V., Maliar, N. L., Bazhenov, S. V., Nikelshparg, E. I., Brazhe, N. A., Vlasova, A. D., Osipov, S. D., Sudarev, V. V., Ryzhykau, Y. L., Bogorodskiy, A. O., Zinovev, E. V., Rogachev, A. V., Manukhov, I. V., Borshchevskiy, V. I., Kuklin, A. I., Pokorný, J., Sosnovtseva, O., Maksimov, G. V., & Gordeliy, V. I. (2020). Raman Scattering: From Structural Biology to Medical Applications. Crystals, 10(1), 38. https://doi.org/10.3390/cryst10010038






