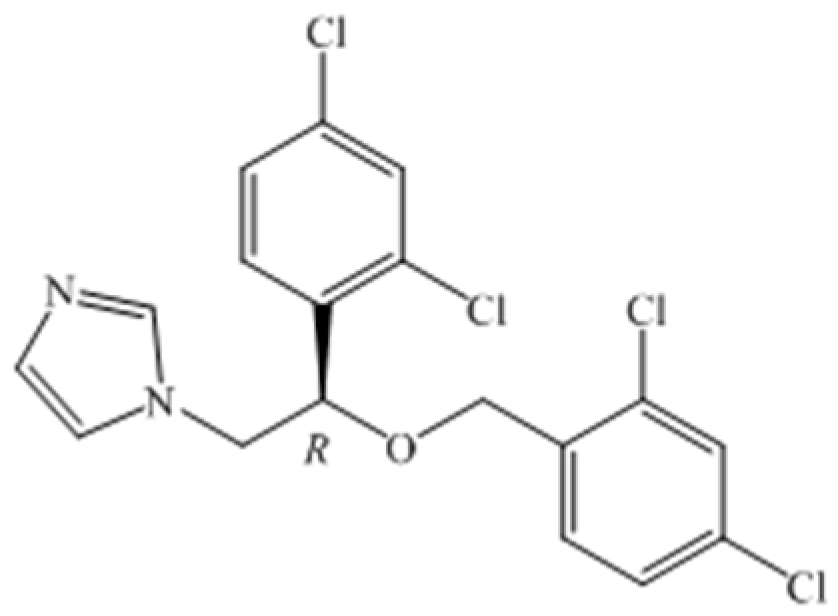
Biotransformation with a New Acinetobacter sp. Isolate for Highly Enantioselective Synthesis of a Chiral Intermediate of Miconazole
Abstract
:1. Introduction
2. Results
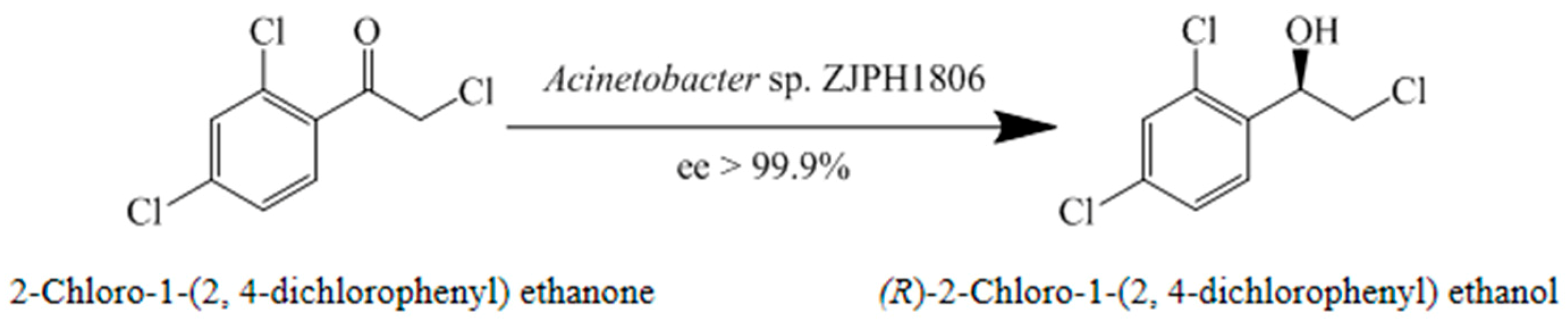
2.1. The Isolation of Strains for the Production of (R)-2-chloro-1-(2,4-dichlorophenyl) Ethanol
2.2. Identification and Characterization of Strain ZJPH1806
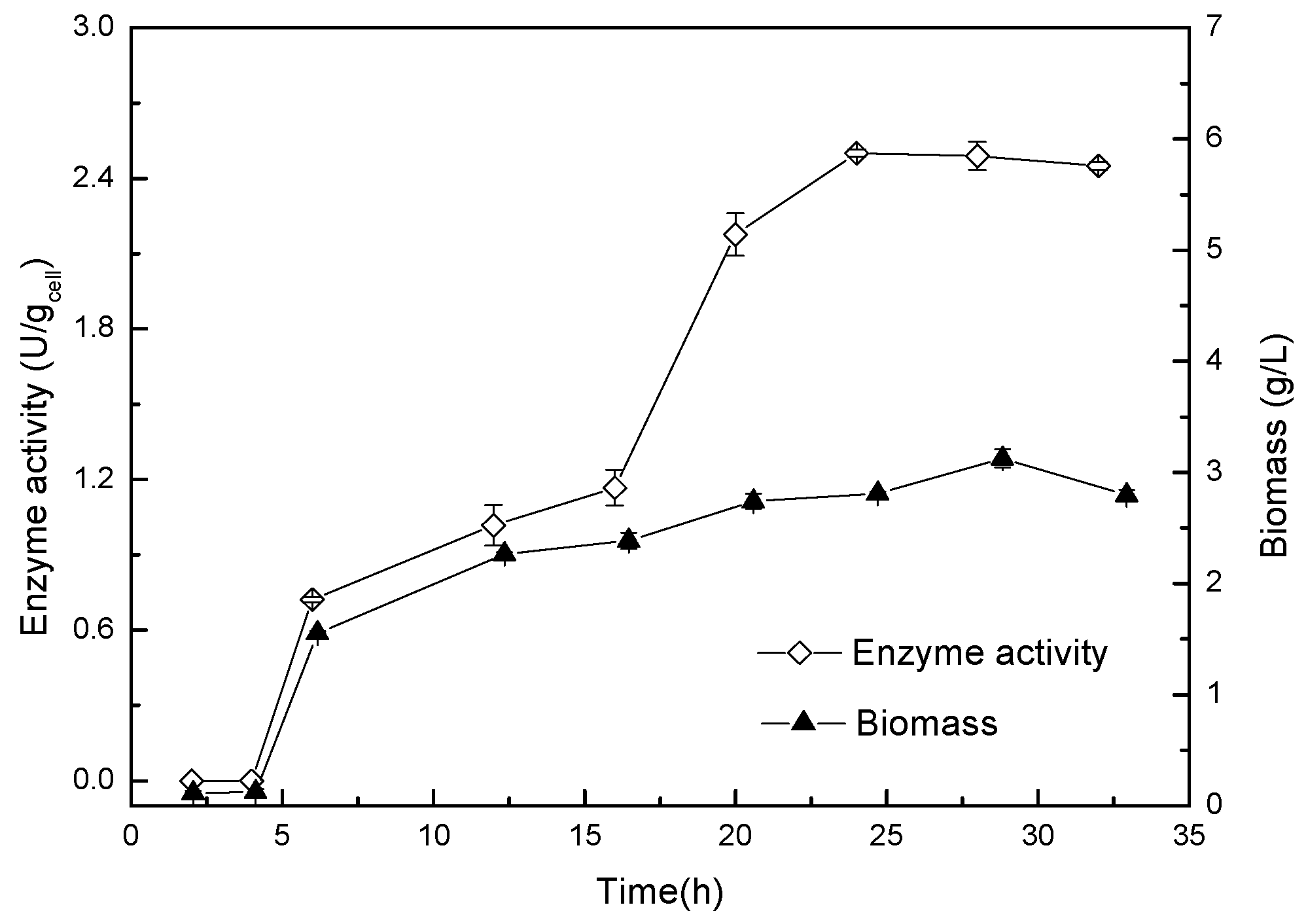
2.3. The Growth Curve of Acinetobacter sp. ZJPH1806
2.4. The Cosubstrate-Coupled System for Cofactors Regeneration
2.5. The Effects of Buffer pH and Ionic Strength on the Asymmetric Reduction of 2-chloro-1-(2,4-dichlorophenyl) Ethanone
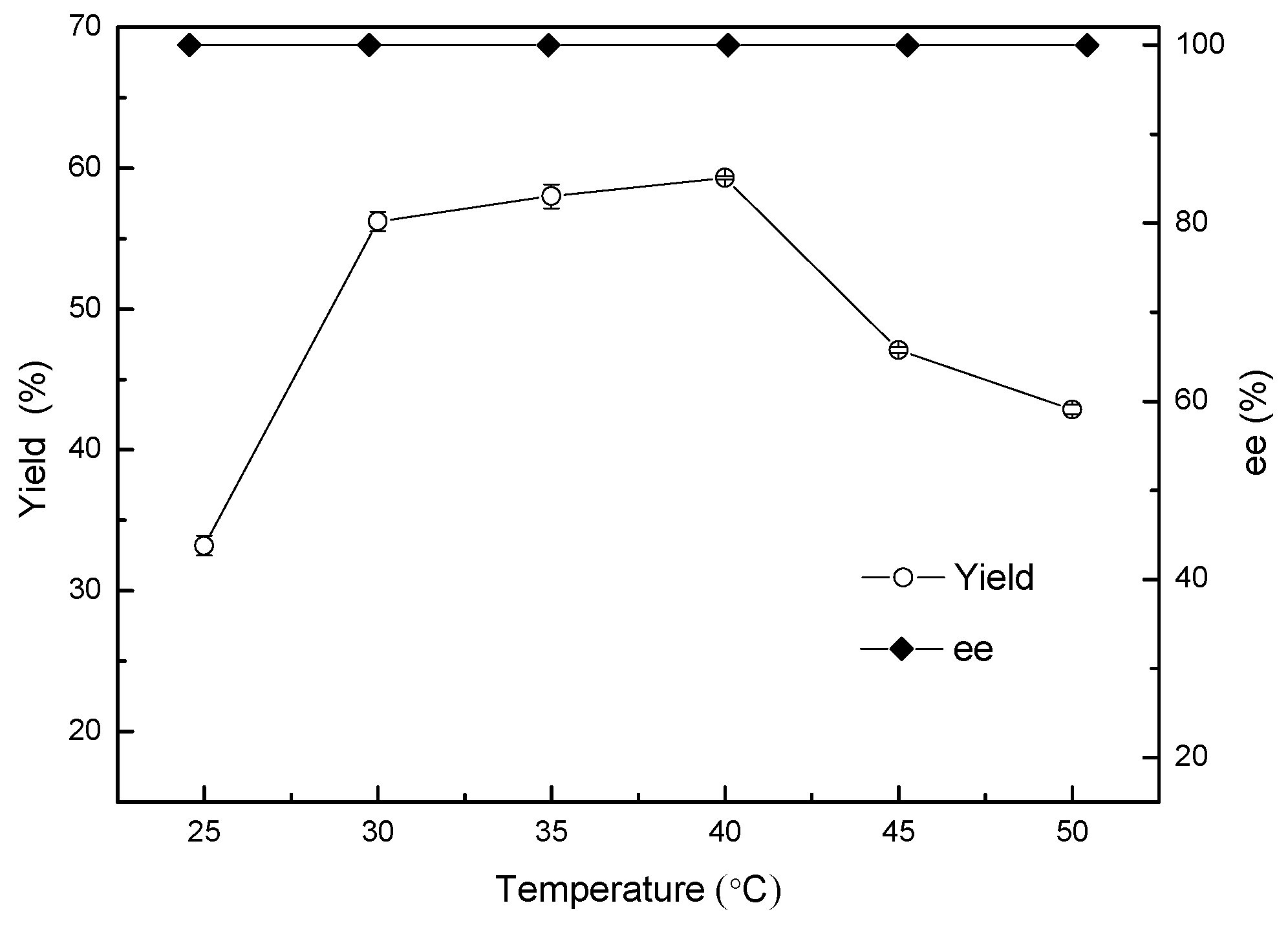
2.6. The Effect of Temperature on the Asymmetric Reduction of 2-chloro-1-(2,4-dichlorophenyl) Ethanone
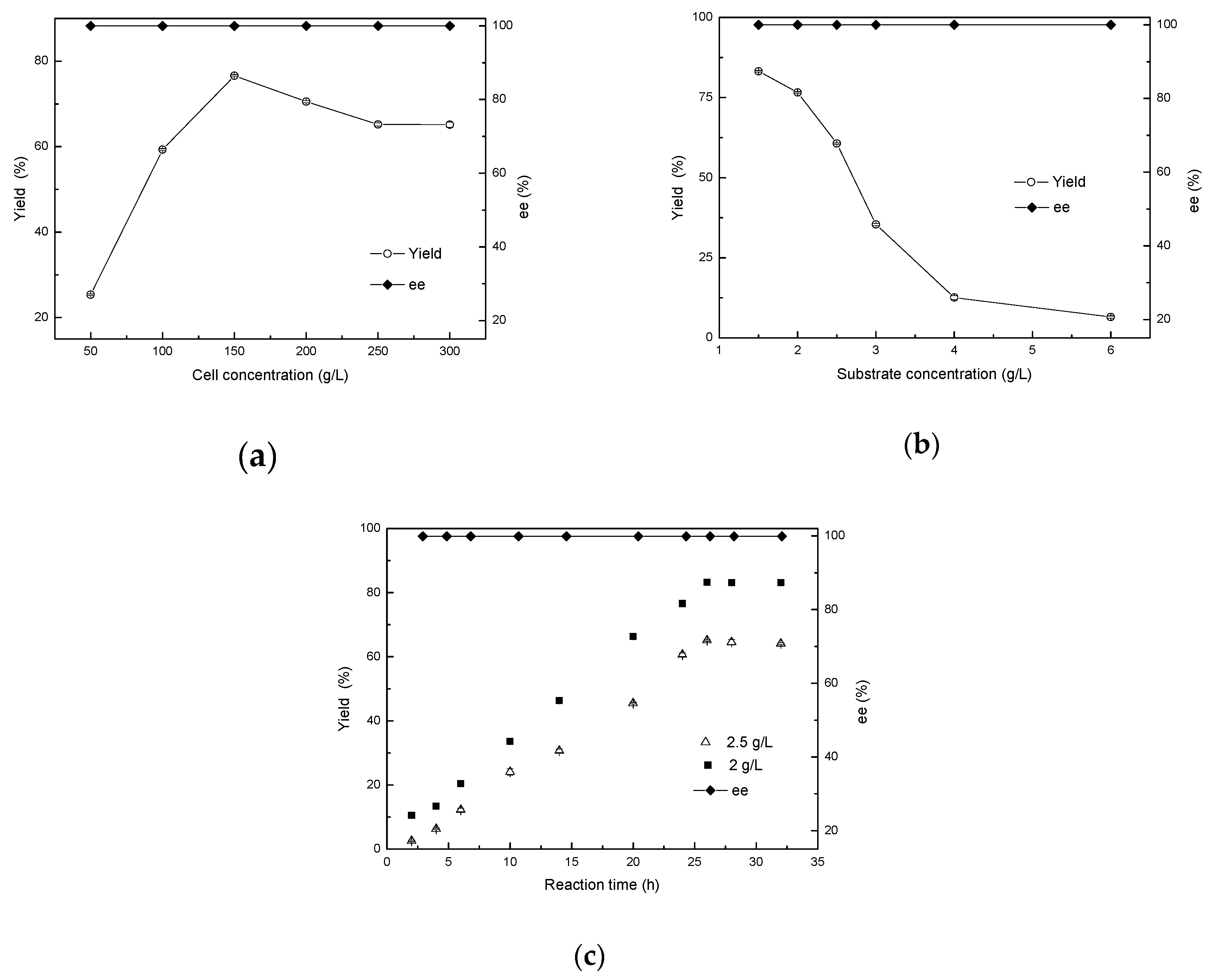
2.7. Effects of Cell Concentration, Substrate Concentration, and Reaction Time on Biocatalytic Reduction
3. Discussion
4. Materials and Methods
4.1. Materials
4.2. Screening and Cultivation of the Microorganisms
4.3. Phenotypic Characterization of Isolate ZJPH1806
4.4. 16S rDNA Sequence Determination and Phylogenetic Analysis
4.5. The Growth Curve of Acinetobacter sp. ZJPH1806
4.6. Asymmetric Bioreduction Process
4.7. Analytical Methods
4.8. The Effects of Key Variables on the Asymmetric Reduction
4.8.1. Effect of Different Cosubstrates on the Asymmetric Reduction
4.8.2. Effect of Different Glycerol Addition on the Asymmetric Reduction
4.8.3. Effects of Buffer pH and Ionic Strength on the Asymmetric Reduction
4.8.4. Effect of Temperature on the Asymmetric Reduction
4.8.5. Optimization of Cell Concentration, Substrate Concentration, and Reaction Time on the Asymmetric Reduction
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Giraud, F.; Loge, C.; Pagniez, F.; Crepin, D.; Pape, P.L.; Borgne, M.L. Design, synthesis, and evaluation of 1-(N-benzylamino)-2-phenyl-3-(1H-1,2,4-triazol-1-yl)propan-2-ols as antifungal agents. Bioorg. Med. Chem. Lett. 2008, 18, 1820–1824. [Google Scholar] [CrossRef] [PubMed]
- Mangas-Sanchez, J.; Busto, E.; Gotor-Fernandez, V.; Malpartida, F.; Gotor, V. Asymmetric chemoenzymatic synthesis of miconazole and econazole enantiomers. The importance of chirality in their biological evaluation. J. Org. Chem. 2011, 76, 2115–2122. [Google Scholar] [CrossRef] [PubMed]
- Barasch, A.; Griffin, A.V. Miconazole revisited: New evidence of antifungal efficacy from laboratory and clinical trials. Future Microbiol. 2008, 3, 265–269. [Google Scholar] [CrossRef] [PubMed]
- Fonseca, T.S.; Lima, L.D.; Oliveira, M.C.F.; Lemos, T.L.G.; Zampieri, D.; Molinari, F.; Mattos, M.C. Chemoenzymatic synthesis of luliconazole mediated by lipases. Eur. J. Org. Chem. 2018, 2018, 2110–2116. [Google Scholar] [CrossRef]
- Zhang, X. Industrial Production Method of Chiral 2-Chloro-1-(2,4-dichlorophenyl)ethanol. C.N. Patent 106008166 A, 12 October 2016. [Google Scholar]
- Luo, J.Y.; Xu, X.B.; Guo, P. New Synthetic Method for Key Chiral Intermediates of Luliconazole. C.N. Patent 108299156 A, 20 January 2018. [Google Scholar]
- Shang, Y.P.; Chen, Q.; Kong, X.D.; Zhang, Y.J.; Xu, J.H.; Yu, H.L. Efficient synthesis of (R)-2-chloro-1-(2,4-dichlorophenyl) ethanol with a ketoreductase from Scheffersomyces stipitis CBS 6045. Adv. Synth. Catal. 2017, 359, 426–431. [Google Scholar] [CrossRef]
- Grynkiewicz, G.; Borowiecki, P. New applications of biotechnology in the field of pharmaceutical syntheses. Przem. Chem. 2019, 98, 434–441. [Google Scholar]
- Xie, Y.; Xu, J.H.; Xu, Y. Isolation of a Bacillus strain producing ketone reductase with high substrate tolerance. Bioresour. Technol. 2010, 101, 1054–1059. [Google Scholar] [CrossRef]
- Lin, H.; Chen, Y.Z.; Xu, X.Y.; Xia, S.W.; Wang, L.X. Preparation of key intermediates of adrenergic receptor agonists: Highly enantioselective production of (R)-α-halohydrins with Saccharomyces cerevisiae CGMCC 2.396. J. Mol. Catal. B Enzym. 2009, 57, 1–5. [Google Scholar] [CrossRef]
- Xie, Y.; Xu, J.H.; Lu, W.Y.; Lin, G.Q. Adzuki bean: A new resource of biocatalyst for asymmetric reduction of aromatic ketones with high stereoselectivity and substrate tolerance. Bioresour. Technol. 2009, 100, 2463–2468. [Google Scholar] [CrossRef]
- Tang, Y.P.; Ding, G.F.; Yu, F.M.; Yang, Z.S.; Huang, F.F. A Method for Biosynthesis of Intermediates of Miconazole. C.N. Patent 108396040 A, 14 August 2018. [Google Scholar]
- Ferreira-Leitão, V.S.; Cammarota, M.C.; Aguieiras, E.C.G.; de Sá, L.R.V.; Fernandez-Lafuente, R.; Freire, D.M.G. The protagonism of biocatalysis in green chemistry and its environmental benefits. Catalysts 2017, 7, 9. [Google Scholar]
- Hollmann, F.; Arends, I.W.C.E.; Holtmann, D. Enzymatic reductions for the chemist. Green Chem. 2011, 13, 2285–2313. [Google Scholar] [CrossRef]
- Sahin, E.; Serencam, H.; Dertli, E. Whole cell application of Lactobacillus paracasei BD101 to produce enantiomerically pure (S)-cyclohexyl(phenyl) methanol. Chirality 2019, 31, 211–218. [Google Scholar] [CrossRef]
- Vitale, P.; Perna, F.M.; Agrimi, G.; Scilimati, A.; Salomone, A.; Cardellicchioe, C.; Capriati, V. Asymmetric chemoenzymatic synthesis of 1,3-diols and 2,4-disubstituted aryloxetanes by using whole cell biocatalysts. Org. Biomol. Chem. 2016, 14, 11438–11445. [Google Scholar] [CrossRef]
- Kara, S.; Schrittwieser, J.H.; Hollmann, F.; Ansorge-Schumacher, M.B. Recent trends and novel concepts in cofactor-dependent biotransformations. Appl. Microbiol. Biotechnol. 2014, 98, 1517–1529. [Google Scholar] [CrossRef]
- Gu, Y.L.; Jerome, F. Glycerol as a sustainable solvent for green chemistry. Green Chem. 2010, 12, 1127–1138. [Google Scholar] [CrossRef]
- Asami, K.; Machida, T.; Jung, S.; Hanaya, K.; Shoji, M.; Sugai, T. Synthesis of (R)-bambuterol based on asymmetric reduction of 1-[3,5-bis(dimethylcarbamoyloxy)phenyl]-2-chloroethanone with incubated whole cells of Williopsis californica JCM 3600. J. Mol. Catal. B Enzym. 2013, 97, 106–109. [Google Scholar] [CrossRef]
- Sun, J.; Huang, J.; Ding, X.Z.; Wang, P. Efficient enantioselective biocatalytic production of a chiral intermediate of sitagliptin by a newly filamentous fungus isolate. Appl. Biochem. Biotechnol. 2016, 180, 695–706. [Google Scholar] [CrossRef]
- Guo, Z.W.; Chen, Y.J.; Goswami, A.; Hanson, R.L.; Patel, R.N. Synthesis of ethyl and t-butyl (3R,5S)-dihydroxy-6-benzyloxy hexanoates via diastereo- and enantioselective microbial reduction. Terahedron Asymmetry 2006, 17, 1589–1602. [Google Scholar] [CrossRef]
- Zhang, Y.J.; Chen, J.L.; Chen, C.S.; Wu, S.J. Isolation of a Bacillus Aryabhattai strain for the resolution of (R,S)-ethyl indoline-2-carboxylate to produce (S)-indoline-2-carboxylic acid. Catalysts 2019, 9, 206. [Google Scholar] [CrossRef]
- Wang, S.S.; Xu, Y.; Zhang, R.Z.; Zhang, B.T.; Xiao, R. Improvement of (R)-carbonyl reductase-mediated biosynthesis of (R)-1-phenyl-1,2-ethanediol by a novel dual-cosubstrate-coupled system for NADH recycling. Process Biochem. 2012, 47, 1060–1065. [Google Scholar] [CrossRef]
- Simon, R.C.; Mutti, F.G.; Kroutil, W. Biocatalytic synthesis of enantiopure building blocks for pharmaceuticals. Drug. Discov. Today Technol. 2013, 10, e37–e44. [Google Scholar] [CrossRef]
- Zhu, D.M.; Hyatt, B.A.; Hua, L. Enzymatic hydrogen transfer reduction of α-chloro aromatic ketones catalyzed by a hyperthermophilic alcohol dehydrogenase. J. Mol. Catal. B Enzym. 2009, 56, 272–276. [Google Scholar] [CrossRef]
- Wachtmeister, J.; Rother, D. Recent advances in whole cell biocatalysis techniques bridging from investigative to industrial scale. Curr. Opin. Biotechnol. 2016, 42, 169–177. [Google Scholar] [CrossRef]
- Goldberg, S.; Guo, Z.W.; Chen, S.; Goswami, A.; Patel, R.N. Synthesis of ethyl-(3R,5S)-dihydroxy-6-benzyloxyhexanoates via diastereo- and enantioselective microbial reduction: Cloning and expression of ketoreductase III from Acinetobacter sp. SC 13874. Enzym. Microb. Technol. 2008, 43, 544–549. [Google Scholar] [CrossRef]
- Wu, X.R.; Liu, N.; He, Y.M.; Chen, Y.J. Cloning, expression, and characterization of a novel diketoreductase from Acinetobacter baylyi. Acta Biochim. Biophys. Sin. 2009, 41, 163–170. [Google Scholar] [CrossRef]
- Atalah, J.; Cáceres-Moreno, P.; Espina, G.; Blamey, J.M. Thermophiles and the applications of their enzymes as new biocatalysts. Bioresour. Technol. 2019, 280, 478–488. [Google Scholar] [CrossRef]

| Strain | Yield (%) | ee (%) | Stereoselectivity |
|---|---|---|---|
| NB1-2 | 17.9 | 57.1 | S |
| CP2-3 | 23.4 | 79.6 | S |
| SX4-7 | 15.8 | 99.9 | S |
| BJ-1 | 9.0 | 72.1 | S |
| AF-4 | 20.1 | 54.3 | S |
| JX1-3 | 23.9 | 59.0 | R |
| HZ1-6 | 12.1 | 99.9 | R |
| XM1-1 | 15.0 | 99.9 | R |
| ZJPH1806 | 28.6 | 99.9 | R |
| Cosubstrate | Yield (%) | ee (%) |
|---|---|---|
| Glucose | 49.4 | 99.9 |
| Sucrose | 40.6 | 99.9 |
| Maltose | 48.6 | 99.9 |
| Glycerol | 50.1 | 99.9 |
| Alcohol | 33.3 | 99.9 |
| Isopropanol | 40.4 | 99.9 |
| Methanol | 9.6 | 99.9 |
| Control | 32.6 | 99.9 |
| Buffer Solution Condition | Yield (%) | ee (%) | |
|---|---|---|---|
| pH a | 6.0, 0.1 M | 10.4 | 99.9 |
| 6.4, 0.1 M | 37.8 | 99.9 | |
| 6.8, 0.1 M | 53.3 | 99.9 | |
| 7.2, 0.1 M | 55.3 | 99.9 | |
| 7.6, 0.1 M | 56.2 | 99.9 | |
| 8.0, 0.1 M | 50.1 | 99.9 | |
| pH 7.6 | 7.6, 0.05 M | 55.8 | 99.9 |
| 7.6, 0.2 M | 55.0 | 99.9 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Miao, Y.; Liu, Y.; He, Y.; Wang, P. Biotransformation with a New Acinetobacter sp. Isolate for Highly Enantioselective Synthesis of a Chiral Intermediate of Miconazole. Catalysts 2019, 9, 462. https://doi.org/10.3390/catal9050462
Miao Y, Liu Y, He Y, Wang P. Biotransformation with a New Acinetobacter sp. Isolate for Highly Enantioselective Synthesis of a Chiral Intermediate of Miconazole. Catalysts. 2019; 9(5):462. https://doi.org/10.3390/catal9050462
Chicago/Turabian StyleMiao, Yanfei, Yuewang Liu, Yushu He, and Pu Wang. 2019. "Biotransformation with a New Acinetobacter sp. Isolate for Highly Enantioselective Synthesis of a Chiral Intermediate of Miconazole" Catalysts 9, no. 5: 462. https://doi.org/10.3390/catal9050462
APA StyleMiao, Y., Liu, Y., He, Y., & Wang, P. (2019). Biotransformation with a New Acinetobacter sp. Isolate for Highly Enantioselective Synthesis of a Chiral Intermediate of Miconazole. Catalysts, 9(5), 462. https://doi.org/10.3390/catal9050462





