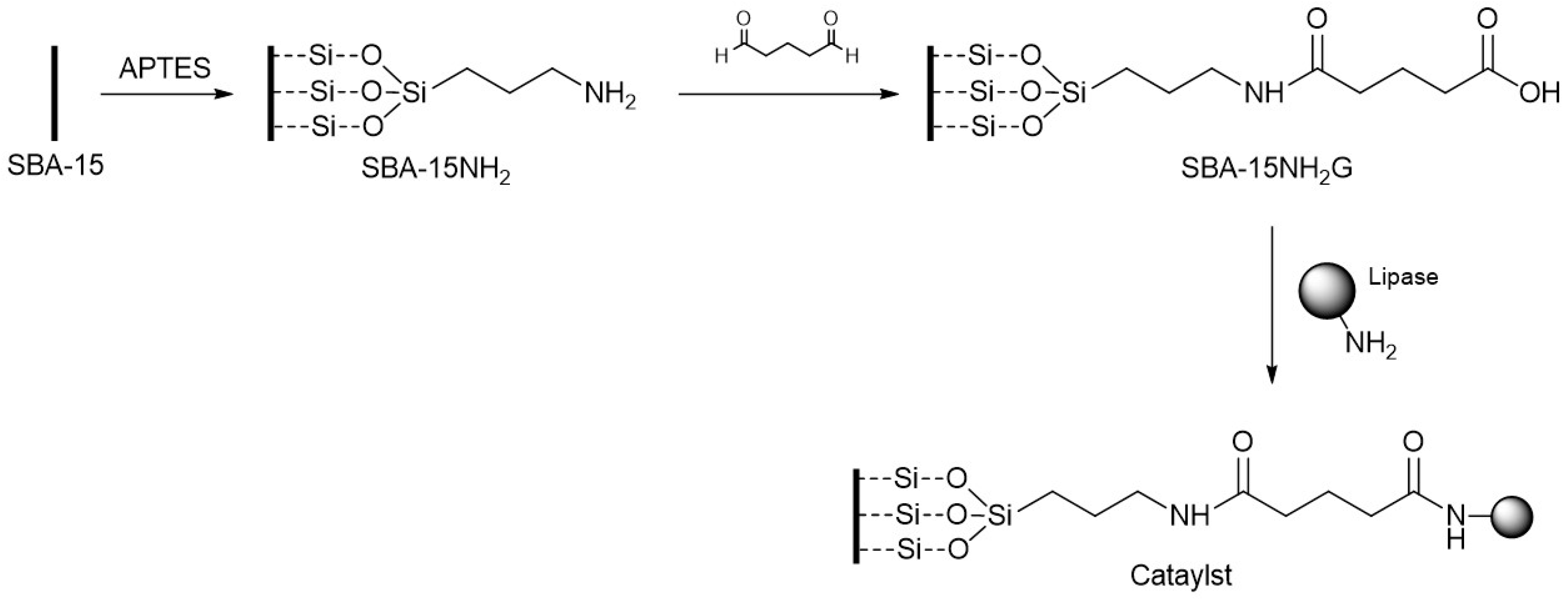
2.1. Characterization of the Synthesized Enzymatic Catalyst
This section contains the most relevant results of the synthesis of the mesoporous silica material SBA-15 as well as the modifications thereof when incorporating amino groups, glutaraldehyde and the enzymes.
The textural properties are shown in
Table 1. The pore surface and diameter were reduced slightly after functionalization of the surface with amino groups and further glutaraldehyde linkage as spacer-arm to favor the subsequent binding of the enzyme. Before functionalization, pore size of SBA-15 was 59.6 Å, and the surface area was the largest, 847.3 m
2/g, and both parameters decreased to 56.3 Å and 841.8 m
2/g, respectively, as chain length increased after amine group and glutaraldehyde introduction [
29]. However, pore volume and surface area were drastically reduced by ~70% with respect to the original SBA-15 after the introduction of enzymes.
The amount of enzyme loaded in each biocatalyst was determined by mass balance measuring the enzyme concentration in the supernatant solution using the Bradford’s assay. The immobilization values were 35 and 33 mg/g for SBA-15-NH
2G-PC and SBA-15-NH
2G-CalB, respectively, similar to those obtained by Bautista et al. [
4] (36.1 mg/g) and slightly lower than that obtained by Serra et al. [
30] (44 mg/g), both using CalB as enzyme. The slightly higher incorporation of enzyme in the case of PC may be due to the fact that its size is somewhat smaller (3 × 3.2 × 6.6 nm) [
31] and can enter into the pores more easily than CalB (3 × 4 × 6 nm) [
32].
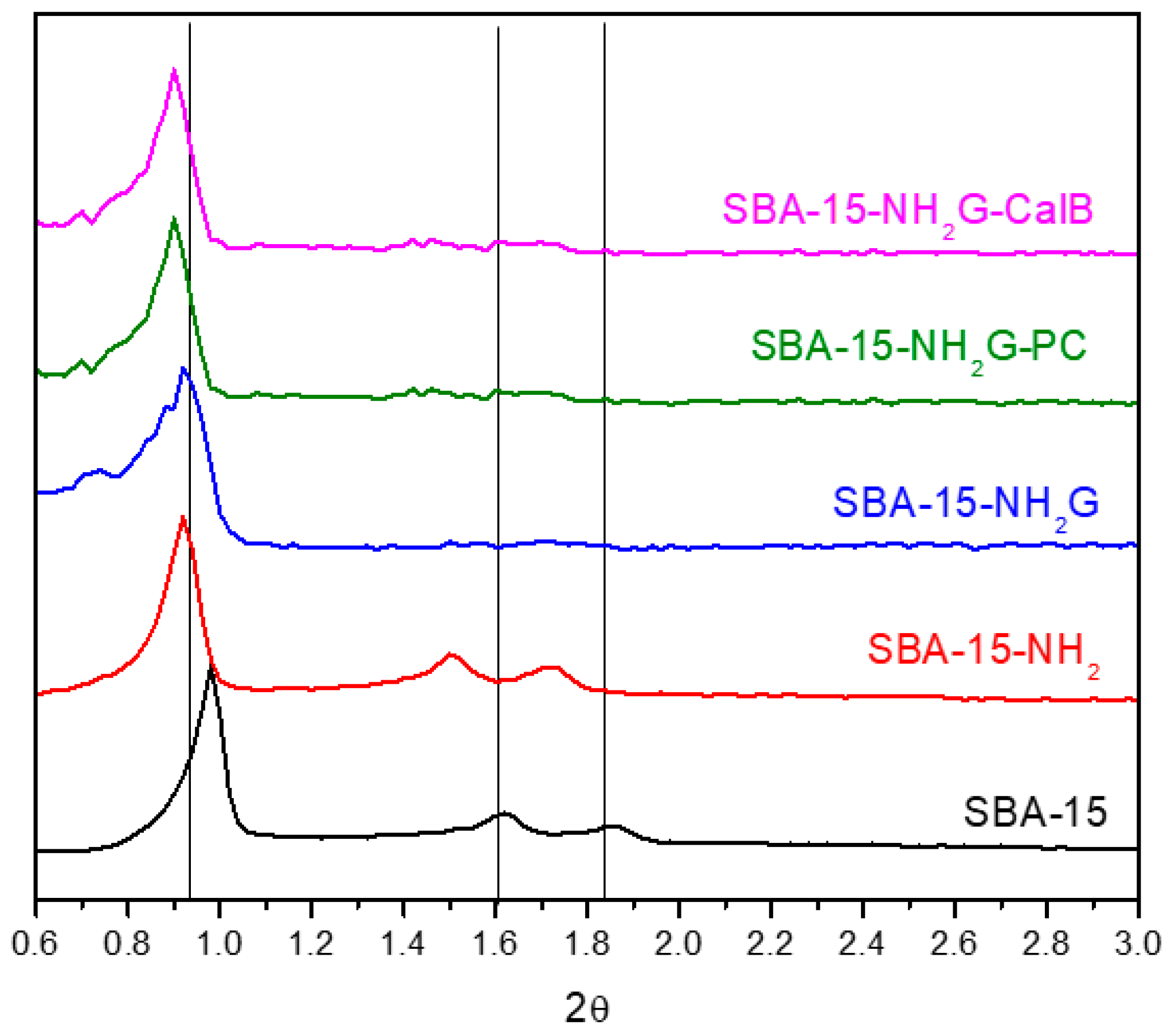
Regarding the structure, XRD (
Figure 1) shows three reflection peaks at 2
θ = 0.97°, 1.6° and 1.9° corresponding to
p6mm mesoporous hexagonal symmetry (planes (100), (110) and (200), respectively) [
33]. Based on the results obtained, the immobilization of enzymes covalently bound to the material did not modify its structure [
10].
Figure 2 shows TEM images obtained from SBA-15 (a) and modified material with anchored lipases from PC (b) and CalB (c). It is clearly noted that both samples show the arrays of long-range mesopore channels, which are similar to the image of SBA-15 reported in the literature [
19]. When the SBA-15 contains enzymes partially occupying the interior of its pores, a morphology showing the different degrees of sharpness of the channels, similar to that described by Mohammadi et al. [
34], can be observed. However, as explained by Abdullah et al. [
11], the fact that the modification of the surface of the SBA-15 material was made by grafting a posteriori prevents major structural changes from taking place and only intervening in the formation of new links with Si-O-Si that are on the surface.
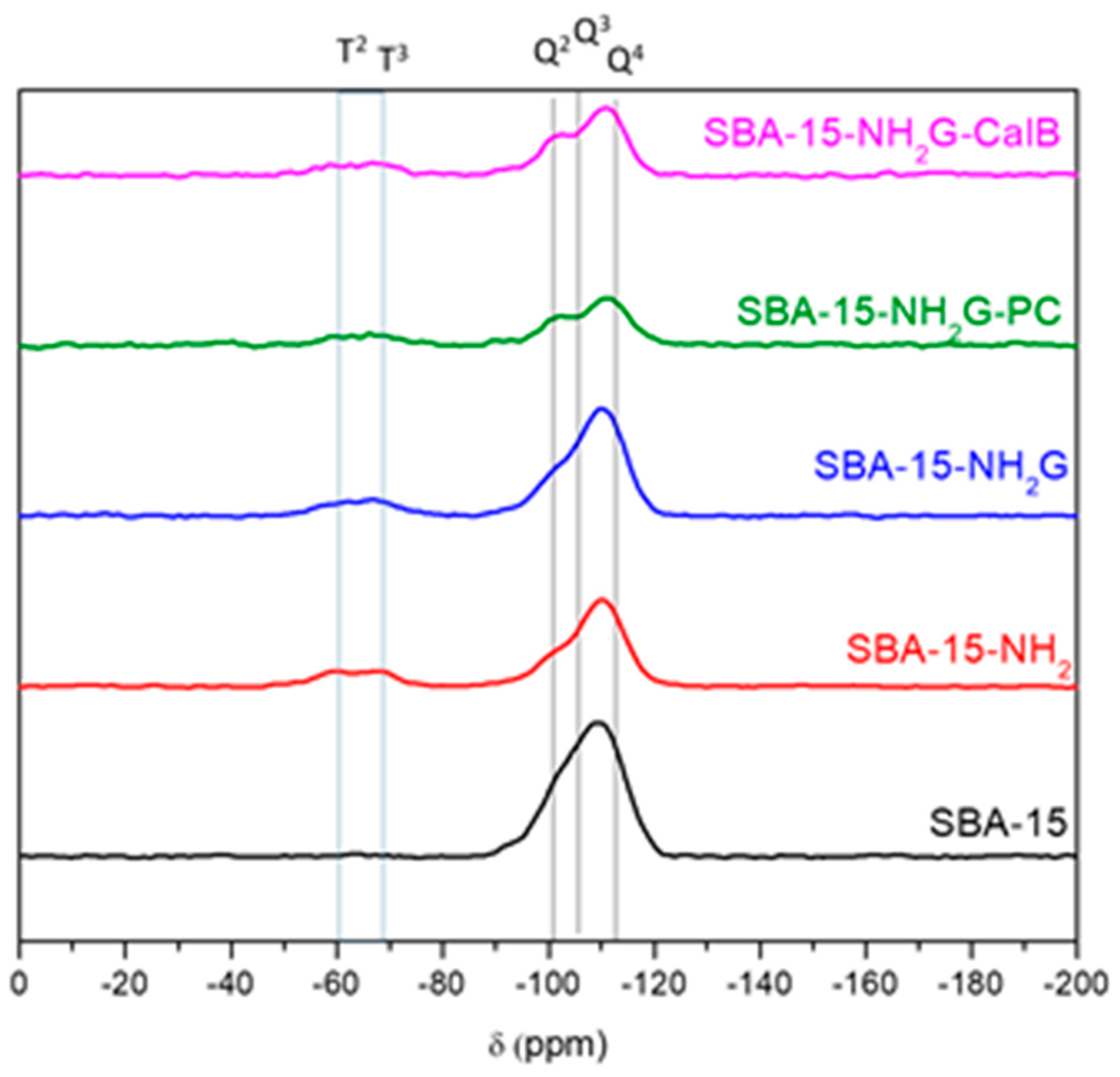
The
29Si MAS-NMR spectrum (
Figure 3) of pure silica shows one wide signal within the range −90 to −110 ppm that correspond to the Si(OSi)
4 (Q
4), HOSi(OSi)
3 (Q
3) and (HO)
2Si(OSi)
2 (Q
2) sites of the silica framework [
35]. Around a displacement −60 to −78 ppm, a broad signal can be seen that corresponds to two signals created by the Si-C links corresponding to RSi(OSi)
3 (T
3) and RSi(HO)(OSi)
2 (T
2) [
36]. These last two signals are due to the incorporation of the organic part (CH
3CH
2CH
2NH
2) by linking on the surface of the material. The peak areas were calculated after Gaussian deconvolution of the spectra considering the associated species and the respective intensities. The results of the integrations (
Table 2) show that the greatest contribution to the areas is given by the peak corresponding to a Si atom linked to four Si-O structures, i.e., corresponding to the Q
4 signal. Overlapped with this peak, two signals of smaller area corresponding to the formation of one or two Si-OH bonds, signals Q
3 and Q
2 respectively. When the amino group was incorporated by grafting on the SBA-15 surface by using 3-aminopropyltriethoxysilane (APTES), the material SBA-15-NH
2 was formed, showing a significant increase in T signals (from 0% to ~10% for both T
2 and T
3) and a concomitant decrease in Q signals, which evidenced the formation of Si-C bonds. However, the addition of glutaraldehyde (SBA-15-NH
2G) and CalB or PC lipases (SBA-15-NH
2G-CalB and SBA-15-NH
2G-PC, respectively) did not modified the peak contribution of T and Q signals significantly, proving that these incorporations are according to the immobilization scheme shown in
Figure 4, where the silica framework was not affected thereafter. These results confirm that the structure is not modified when both organic modifiers and lipases are introduced [
37].
The elemental analysis shows how the composition of hydrogen, carbon, nitrogen and oxygen varies as new functional groups, amino and aldehyde are introduced into the structure, as well as with the incorporation of enzymes. The results shown in
Table 3 are coherent since the initial mesoporous structure contains only silicon and hydrogen. The presence of nitrogen, carbon and oxygen are due to the incorporation of the different functional groups in the SBA-15. The introduction of the amino group from APTES increases the nitrogen concentration to ~2.8%, but the incorporation of glutaraldehyde decreases the nitrogen content to 2.2%, due to the addition of carbon, hydrogen and oxygen into the structure. Similar results were reported previously for different functionalization of SBA-15 materials [
18]. Conversely, an increase in all the elements after lipase immobilization was observed in SBA-15NH
2G-PC and SBA-15NH
2G-CalB because the incorporation of the lipase molecules added N, C and H, as expected.
After water removal at T < 100 °C, thermogravimetric analysis shows a weight loss between 200 and 300 °C that corresponds to glutaraldehyde followed by the loss of the amino group at temperatures between 300 and 600 °C (
Figure 5). The results of this analysis show an organic loss, corresponding to propylamine (CH
3CH
2CH
2NH
2), of 11.8 wt% in all the materials. The nitrogen content present in the material SBA-15-NH
2 according to this test (2.8 wt%) corroborates the results of elemental analysis (
Table 3).
A weight loss of 7.48% is observed in the material SBA-15-NH2G, which implies an incorporation of glutaraldehyde of 37 wt% with respect to the amount of available amino groups. This indicates that part of amino groups incorporated into the material did not form bonds with glutaraldehyde. In addition, this test allowed to corroborate the results of nitrogen content (2.35 wt%) with that obtained by elemental analysis (2.23 wt%).
2.2. Production of FAEEs Using Single Lipase Biocatalysts
The activity of both synthesized biocatalysts (SBA-15-NH
2G-CalB and SBA-15-NH
2G-PC) towards FAEEs production from wet and dry extracted lipids from
I. galbana was evaluated and compared with the commercial Novozym 435. The results were assessed based on the molar conversion of saponifiable lipids measured by
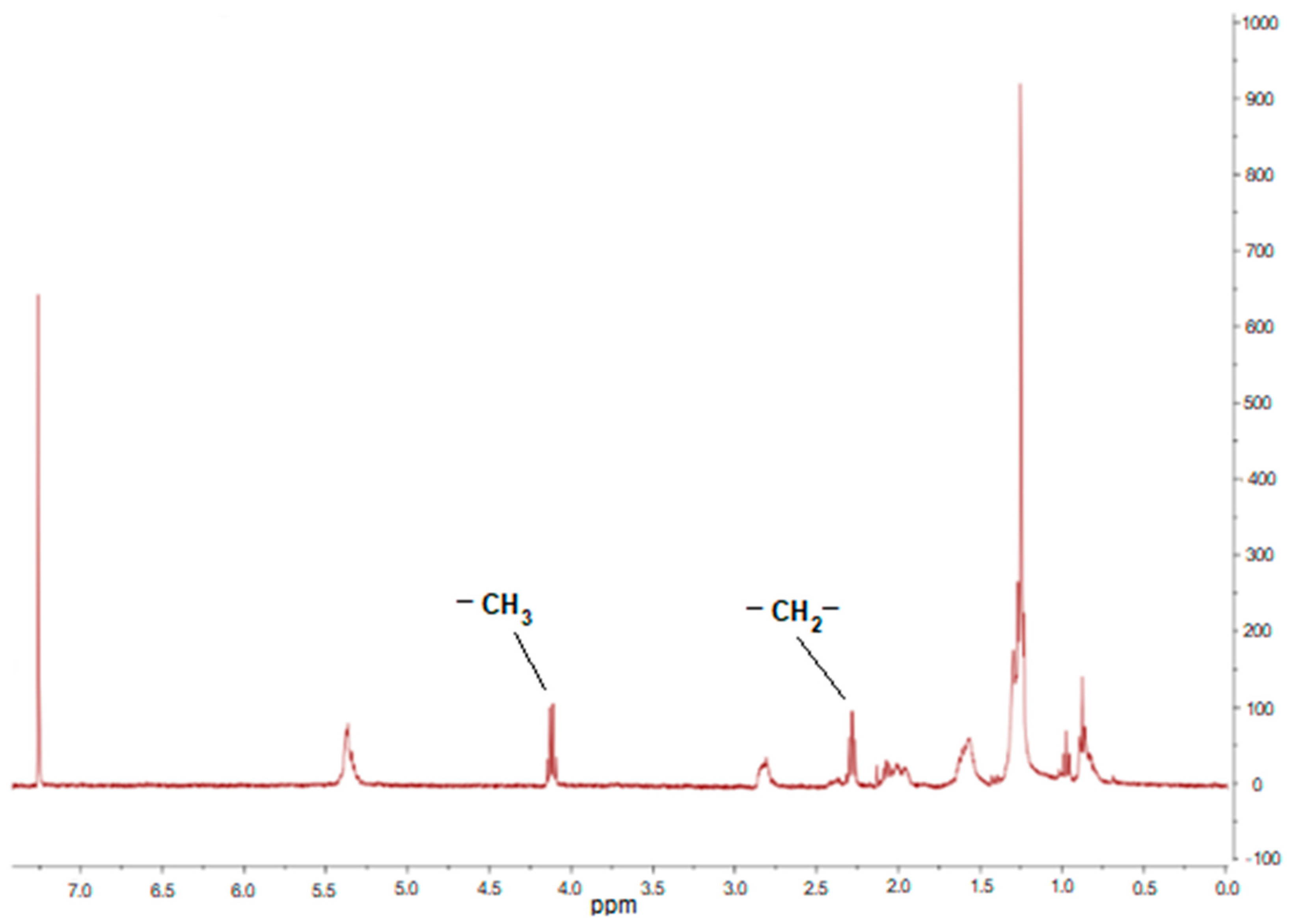
1H NMR (
Table 4). As it can be seen, the lipid extraction method had a large impact on lipase-catalyzed conversion to FAEEs. When the microalgal oil, used as feedstock, was extracted by the wet method, the results showed a reduction in FAEEs production from 97 to 85.5 mol% and from 91 to 87 mol% using CalB and PC lipase-based biocatalysts, respectively. The commercial Novozym 435 also showed a negative effect when lipids extracted by the wet method were used. Microalgal oils from both the wet and dry extraction methods did not contain water. Therefore, the decrease in conversion may be caused by the extraction of water-soluble lipase-inhibitor compounds during the wet route that are not present in the oil extracted from dry biomass, because lipase activity was negatively affected by polar compounds causing inhibition or denaturation [
38].
The highest conversion to FAEEs (97 mol%) using dry extracted lipids with ethyl acetate from I. galbana microalgae was achieved using SBA-15-NH2G-CalB as biocatalyst. For the wet extraction route, both CalB and PC lipase-based biocatalysts showed similar activity (85.5 and 87 mol%, respectively) so that SBA-15-NH2G-CalB seems to be more sensitive to the presence of water or other polar compounds likely extracted during the wet process. However, in all cases, the synthesized biocatalysts SBA-15-NH2G-CalB and SBA-15-NH2G-PC were significantly more active towards FAEEs production than the commercial Novozym 435, proving the better performance of both biocatalysts synthesized in the present work.
2.3. Production of FAEEs Using Combi-Lipase Biocatalysts
In recent years, the search for new biocatalysts calls for the combination of different enzymes of different specificity to produce a higher production yield of FAEEs [
26,
27]. Consequently, a biocatalyst formed by the combination in different proportions of CalB and PC supported on SBA-15 modified with amino groups and glutaraldehyde is used in order to evaluate the FAEEs production in the same conditions used previously, using the lipids extracted by wet route with ethyl acetate as raw material.
Table 5 shows the synergistic effect of different combi-lipases on the conversion to FAEEs, along with those values achieves with the corresponding single-lipase biocatalysts It is observed that the combi-lipase 25:75 produced the highest conversion to FAEEs (97.2 mol%) which is more than 10% higher than those produced by the biocatalysts formed by CalB or PC alone. This is a remarkable result because it demonstrates that it is possible to design a catalyst containing an optimized mixture of different lipases that maximizes the conversion to FAEEs.
The fatty acid profile of FAEEs produced by the optimized 25:75 SBA-15-NH
2G-CalB:SBA-15-NH
2G-PC is shown in
Table 6. The major saturated fatty acids are myristic (C14:0) and palmitic (C16:0), while palmitoleic (C16:1) and oleic (C18:1) acids represented the main monounsaturated and linolenic (C18:3) the most abundant polyunsaturated fatty acid. It is important to highlight the value of linolenic acid (20.2%), a concentration about 10% higher than that regulated by EN 14,214 standard (
Table 7). This would require further actions after biodiesel production to meet the required specifications, such as mixing with biodiesel from other lipid sources whose linolenic acid content is lower than that of the microalgae used in this work.
Other key properties of the biodiesel produced by the combi-lipase biocatalyst 25:75 SBA-15-NH
2G-CalB:SBA-15-NH
2G-PC using the lipids obtained after wet extraction with ethyl acetate are shown in
Table 7. Iodine value as well as mono, di and triglyceride content and sulfur content fulfill the European standard EN 14214. However, metals and phosphorus content are higher than those regulated by the above standard, which would require a further purification stage.