Photocatalytic Degradation of Profenofos and Triazophos Residues in the Chinese Cabbage, Brassica chinensis, Using Ce-Doped TiO2
Abstract
1. Introduction
2. Results and Discussion
2.1. Degradation Efficiency
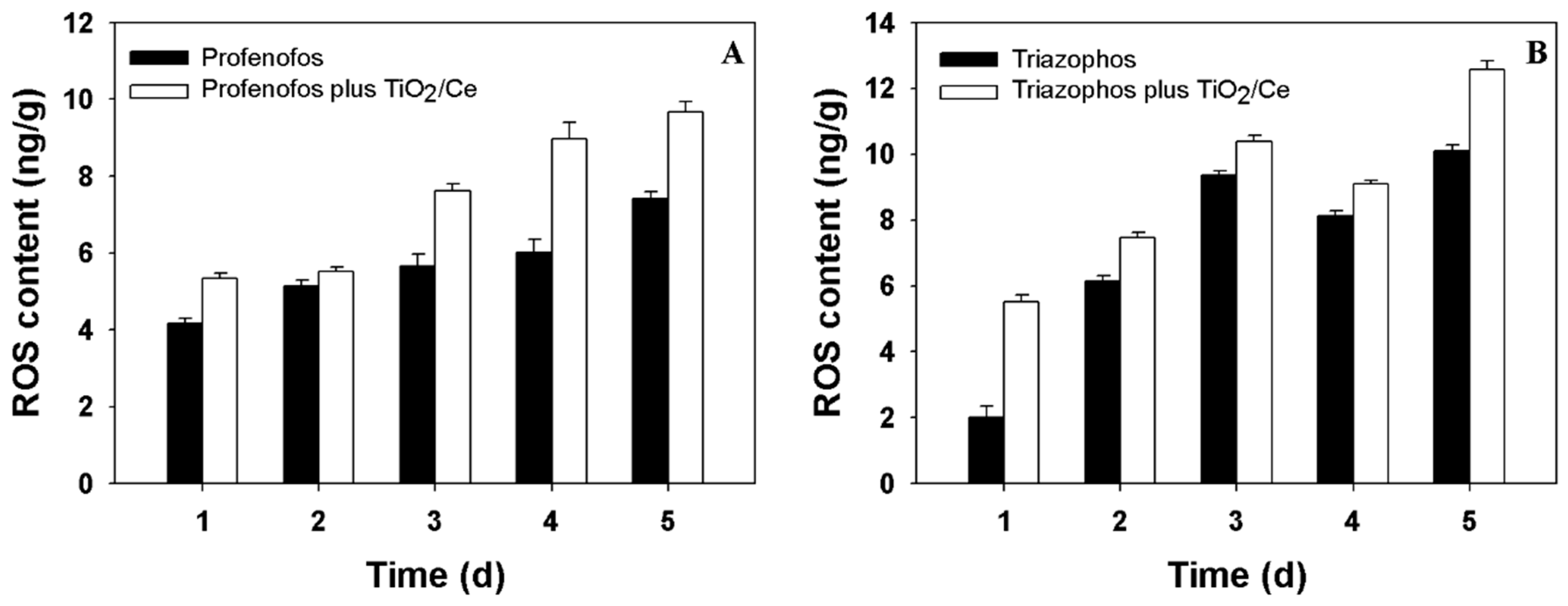
2.2. Reactive Oxygen Species (ROS) Content and Phytotoxicity
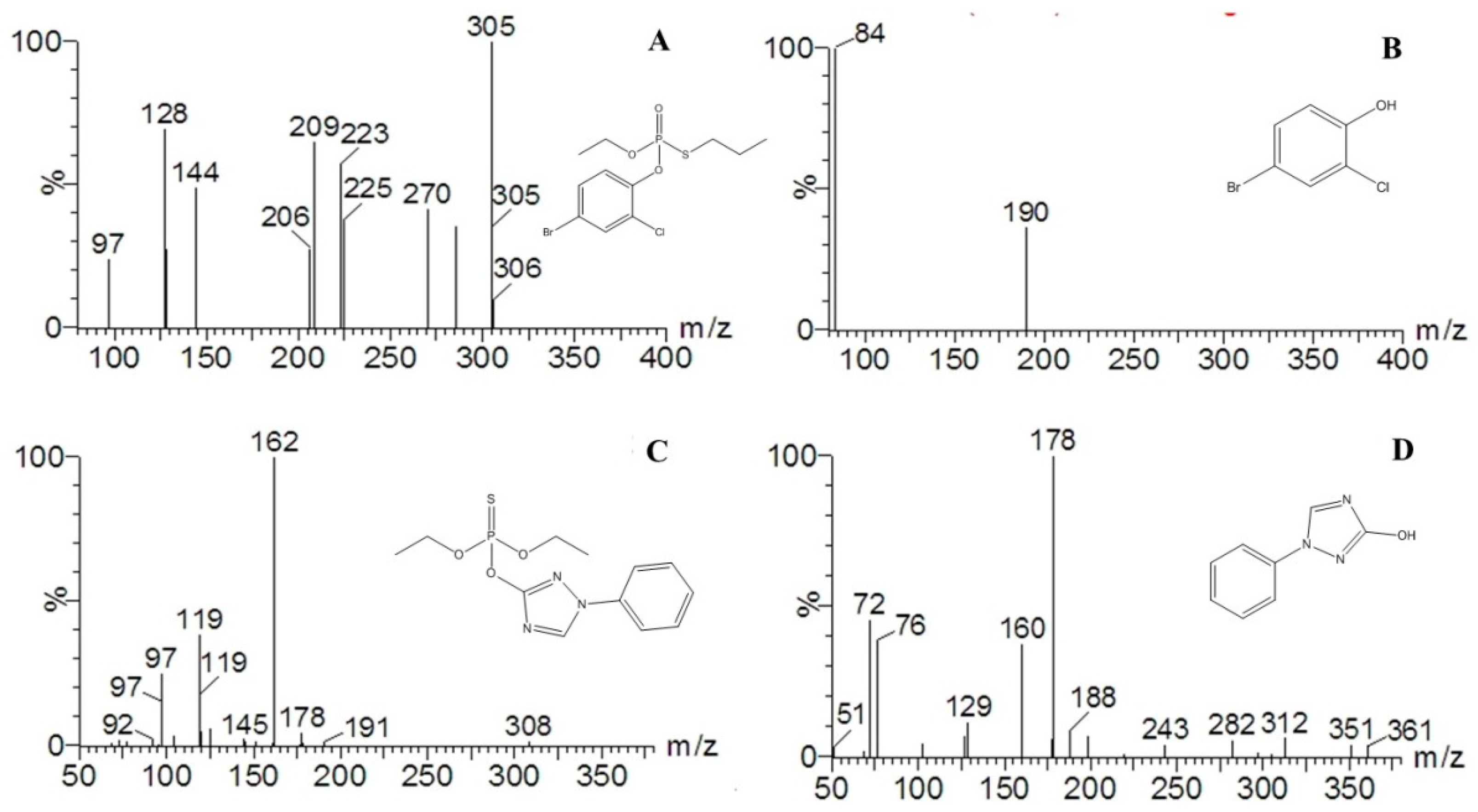
2.3. Degradation Byproducts and Molecular Structure
3. Materials and Methods
3.1. Materials
3.2. Sample Preparation
3.3. GC analysis
3.4. ROS Assay
3.5. UPLC-MS Analysis
3.6. RHF/STO-3G Calculation
3.7. Data Analysis
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ghaffar, A.; Hussain, R.; Khan, A.; Rao, Z.A. Hemato-biochemical and Genetic Damage Caused by Triazophos in Fresh Water Fish, Labeo rohita. Int. J. Agric. Biol. 2015, 17, 637–642. [Google Scholar] [CrossRef]
- Pandey, A.K.; Nagpure, N.; Trivedi, S.P.; Kumar, R.; Kushwaha, B. Profenofos induced DNA damage in freshwater fish, Channa punctatus (Bloch) using alkaline single cell gel electrophoresis. Mutat. Res.-Gen. Toxicol. Environ. Mutagenesis 2011, 726, 209–214. [Google Scholar] [CrossRef] [PubMed]
- Bajwa, U.; Sandhu, K.S. Effect of handling and processing on pesticide residues in food—A review. J. Food Sci. Technol. 2014, 51, 201–220. [Google Scholar] [CrossRef]
- Carp, O.; Huisman, C.L.; Reller, A. Photoinduced reactivity of titanium dioxide. Prog. Solid State Chem. 2004, 32, 33–177. [Google Scholar] [CrossRef]
- He, Y.; Sutton, N.B.; Rijnaarts, H.H.; Langenhoff, A.A. Degradation of pharmaceuticals in wastewater using immobilized TiO2 photocatalysis under simulated solar irradiation. Appl. Catal. B-Environ. 2016, 182, 132–141. [Google Scholar] [CrossRef]
- Khavar, A.H.C.; Moussavi, G.; Mahjoub, A.R.; Satari, M.; Abdolmaleki, P. Synthesis and visible-light photocatalytic activity of In, S-TiO2@rGO nanocomposite for degradation and detoxification of pesticide atrazine in water. Chem. Eng. J. 2018, 345, 300–311. [Google Scholar] [CrossRef]
- Lu, P.-J.; Huang, S.-C.; Chen, Y.-P.; Chiueh, L.-C.; Shih, D.Y.-C. Analysis of titanium dioxide and zinc oxide nanoparticles in cosmetics. J. Food Drug Anal. 2015, 23, 587–594. [Google Scholar] [CrossRef] [PubMed]
- Isaifan, R.J.; Samara, A.; Suwaileh, W.; Johnson, D.; Yiming, W.; Abdallah, A.A.; Aïssa, B. Improved self-cleaning properties of an efficient and easy to scale up TiO2 thin films prepared by adsorptive self-assembly. Sci. Rep.-UK 2017, 7, 9466. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.; Zhang, M.; Dong, W.; Cui, G.; Ren, Z.; Wang, W. Highly efficient photocatalytic degradation of methylene blue by PoPD/TiO2 nanocomposite. PLoS ONE 2017, 12, e0174104. [Google Scholar] [CrossRef]
- Kumari, A.; Yadav, S.K. Nanotechnology in agri-food sector. Crit. Rev. Food Sci. 2014, 54, 975–984. [Google Scholar] [CrossRef]
- Koklic, T.; Urbancic, I.; Zdovc, I.; Golob, M.; Umek, P.; Arsov, Z.; Drazic, G.; Pintaric, S.; Dobeic, M.; Strancar, J. Surface deposited one-dimensional copper-doped TiO2 nanomaterials for prevention of health care acquired infections. PLoS ONE 2018, 13, e0201490. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Sun, C.; Zhao, X.; Cui, B.; Zeng, Z.; Wang, A.; Liu, G.; Cui, H. The application of nano-TiO2 photo semiconductors in agriculture. Nanoscale Res. Lett. 2016, 11, 529. [Google Scholar] [CrossRef] [PubMed]
- Prasad, R.; Kumar, V.; Prasad, K.S. Nanotechnology in sustainable agriculture: Present concerns and future aspects. Afr. J. Biotechnol. 2014, 13, 705–713. [Google Scholar]
- Gogos, A.; Knauer, K.; Bucheli, T.D. Nanomaterials in plant protection and fertilization: Current state, foreseen applications, and research priorities. J. Agric. Food Chem. 2012, 60, 9781–9792. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.G.; Devi, L.G. Review on modified TiO2 photocatalysis under UV/visible light: Selected results and related mechanisms on interfacial charge carrier transfer dynamics. J. Phys. Chem. A 2011, 115, 13211–13241. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Mao, S.S. Titanium dioxide nanomaterials: Synthesis, properties, modifications, and applications. Chem. Rev. 2007, 107, 2891–2959. [Google Scholar] [CrossRef]
- Xie, C.; Yang, S.; Shi, J.; Niu, C. Highly crystallized C-doped mesoporous anatase TiO2 with visible light photocatalytic activity. Catalysts 2016, 6, 117. [Google Scholar] [CrossRef]
- Kamaei, M.; Rashedi, H.; Dastgheib, S.; Tasharrofi, S. Comparing photocatalytic degradation of gaseous ethylbenzene using N-doped and pure TiO2 nano-catalysts coated on glass beads under both UV and visible light irradiation. Catalysts 2018, 8, 466. [Google Scholar] [CrossRef]
- Lin, Y.; Mehrvar, M. Photocatalytic treatment of an actual confectionery wastewater using Ag/TiO2/Fe2O3: Optimization of photocatalytic reactions using surface response methodology. Catalysts 2018, 8, 409. [Google Scholar] [CrossRef]
- Mikaeili, F.; Topcu, S.; Jodhani, G.; Gouma, P.-I. Flame-sprayed pure and Ce-doped TiO2 photocatalysts. Catalysts 2018, 8, 342. [Google Scholar] [CrossRef]
- Charanpahari, A.; Umare, S.; Sasikala, R. Effect of Ce, N and S multi-doping on the photocatalytic activity of TiO2. Appl. Surf. Sci. 2013, 282, 408–414. [Google Scholar] [CrossRef]
- Xie, J.; Jiang, D.; Chen, M.; Li, D.; Zhu, J.; Lü, X.; Yan, C. Preparation and characterization of monodisperse Ce-doped TiO2 microspheres with visible light photocatalytic activity. Colloid Surf. A 2010, 372, 107–114. [Google Scholar] [CrossRef]
- Liu, X.; Wang, L.; Zhou, X.; Liu, K.; Bai, L.; Zhou, X. Photocatalytic degradation of acephate in pak choi, Brassica chinensis, with Ce-doped TiO2. J. Environ. Sci. Health B. 2015, 50, 331–337. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Li, Y.; Zhou, X.; Luo, K.; Hu, L.; Liu, K.; Bai, L. Photocatalytic degradation of dimethoate in Bok choy using cerium-doped nano titanium dioxide. PLoS ONE 2018, 13, e0197560. [Google Scholar] [CrossRef] [PubMed]
- Feizi, H.; Rezvani Moghaddam, P.; Shahtahmassebi, N.; Fotovat, A. Impact of bulk and nanosized titanium dioxide (TiO2) on wheat seed germination and seedling growth. Biol. Trace Elem. Res. 2012, 146, 101–106. [Google Scholar] [CrossRef] [PubMed]
- Zheng, L.; Hong, F.; Lu, S.; Liu, C. Effect of nano-TiO2 on strength of naturally aged seeds and growth of spinach. Biol. Trace Elem. Res. 2005, 104, 83–91. [Google Scholar] [CrossRef]
- Affam, A.C.; Chaudhuri, M. Degradation of pesticides chlorpyrifos, cypermethrin and chlorothalonil in aqueous solution by TiO2 photocatalysis. J. Environ. Manag. 2013, 130, 160–165. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Zhou, Q.; Xie, G.; Liu, H.; Lin, H. Titanium dioxide nanotubes for solid phase extraction of benzoylurea insecticides in environmental water samples, and determination by high performance liquid chromatography with UV detection. Microch. Acta 2011, 172, 109–115. [Google Scholar] [CrossRef]
- Geng, H.R.; Miao, S.S.; Jin, S.F.; Yang, H. A newly developed molecularly imprinted polymer on the surface of TiO2 for selective extraction of triazine herbicides residues in maize, water, and soil. Anal. Bioanal. Chem. 2015, 407, 8803–8812. [Google Scholar] [CrossRef] [PubMed]
- Owolade, O.; Ogunleti, D. Effects of titanium dioxide on the diseases, development and yield of edible cowpea. J. Plant Prot. Res. 2008, 48, 329–336. [Google Scholar] [CrossRef]
- Zeng, R.; Wang, J.; Cui, J.; Hu, L.; Mu, K. Photocatalytic degradation of pesticide residues with RE3+-doped nano-TiO2. J. Rare Earths 2010, 28, 353–356. [Google Scholar] [CrossRef]
- Rabindranathan, S.; Devipriya, S.; Yesodharan, S. Photocatalytic degradation of phosphamidon on semiconductor oxides. J. Hazard. Mater. 2003, 102, 217–229. [Google Scholar] [CrossRef]
- Apel, K.; Hirt, H. Reactive oxygen species: Metabolism, oxidative stress, and signal transduction. Annu. Rev. Plant Biol. 2004, 55, 373–399. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Qiu, C.B.; Zhou, Y.; Jin, Z.P.; Yang, H. Bioaccumulation and degradation of pesticide fluroxypyr are associated with toxic tolerance in green alga Chlamydomonas reinhardtii. Ecotoxicology 2011, 20, 337–347. [Google Scholar] [CrossRef] [PubMed]
- Yan, Z.-Q.; Wang, D.-D.; Ding, L.; Cui, H.-Y.; Jin, H.; Yang, X.-Y.; Yang, J.-S.; Qin, B. Mechanism of artemisinin phytotoxicity action: Induction of reactive oxygen species and cell death in lettuce seedlings. Plant Physiol. Biochem. 2015, 88, 53–59. [Google Scholar] [CrossRef]
- Sirons, G.J.; Frank, R.; Sawyer, T. Residues of atrazine, cyanazine, and their phytotoxic metabolites in a clay loam soil. J. Agric. Food Chem. 1973, 21, 1016–1020. [Google Scholar] [CrossRef] [PubMed]
- Burkhard, N.; Guth, J.A. Photolysis of organophosphorus insecticides on soil surfaces. Pestic. Sci. 1979, 10, 313–319. [Google Scholar] [CrossRef]
- Capps, T.M.; Barringer, V.M.; Eberle, W.J.; Brown, D.R.; Sanson, D.R. Identification of a unique glucosylsulfate conjugate metabolite of profenofos in cotton. J. Agric. Food Chem. 1996, 44, 2408–2411. [Google Scholar] [CrossRef]
- Talwar, M.P.; Ninnekar, H.Z. Biodegradation of pesticide profenofos by the free and immobilized cells of Pseudoxanthomonas suwonensis strain HNM. J. Basic Microb. 2015, 55, 1094–1103. [Google Scholar] [CrossRef] [PubMed]
- Salunkhe, V.P.; Sawant, I.S.; Banerjee, K.; Rajguru, Y.R.; Wadkar, P.N.; Oulkar, D.P.; Naik, D.G.; Sawant, S.D. Biodegradation of profenofos by Bacillus subtilis isolated from grapevines (Vitis vinifera). J. Agric. Food Chem. 2013, 61, 7195–7202. [Google Scholar] [CrossRef]
- Amalraj, A.; Suryaprabha, T.; Rajeswari, A.; Pius, A. Photocatalytic degradation of quinalphos and profenofos pesticides using UV irradiated TiO2 nanoparticles—A kinetic study. Mater. Focus 2016, 5, 377–384. [Google Scholar] [CrossRef]
- Lin, K.; Yuan, D.; Chen, M.; Deng, Y. Kinetics and products of photo-Fenton degradation of triazophos. J. Agric. Food Chem. 2004, 52, 7614–7620. [Google Scholar] [CrossRef]
- Liang, B.; Yang, C.; Gong, M.; Zhao, Y.; Zhang, J.; Zhu, C.; Jiang, J.; Li, S. Adsorption and degradation of triazophos, chlorpyrifos and their main hydrolytic metabolites in paddy soil from Chaohu Lake, China. J. Environ. Manag. 2011, 92, 2229–2234. [Google Scholar] [CrossRef] [PubMed]
- Lin, K.; Yuan, D.; Deng, Y.; Chen, M. Hydrolytic products and kinetics of triazophos in buffered and alkaline solutions with different values of pH. J. Agric. Food Chem. 2004, 52, 5404–5411. [Google Scholar]
- Wang, L.; Zhang, L.; Chen, H.; Tian, Q.; Zhu, G. Isolation of a triazophos-degrading strain Klebsiella sp. E6 effectively utilizing triazophos as sole nitrogen source. FEMS Microbiol. Lett. 2005, 253, 259–265. [Google Scholar] [CrossRef]
- Yang, C.; Li, R.; Song, Y.; Chen, K.; Li, S.; Jiang, J. Identification of the biochemical degradation pathway of triazophos and its intermediate in Diaphorobacter sp. TPD-1. Curr. Microbiol. 2011, 62, 1294–1301. [Google Scholar] [CrossRef] [PubMed]
- Aungpradit, T.; Sutthivaiyakit, P.; Martens, D.; Sutthivaiyakit, S.; Kettrup, A.A. Photocatalytic degradation of triazophos in aqueous titanium dioxide suspension: Identification of intermediates and degradation pathways. J. Hazard. Mater. 2007, 146, 204–213. [Google Scholar] [CrossRef] [PubMed]
- Zhu, X.; Yuan, C.; Bao, Y.; Yang, J.; Wu, Y. Photocatalytic degradation of pesticide pyridaben on TiO2 particles. J. Mol. Catal. A Chem. 2005, 229, 95–105. [Google Scholar] [CrossRef]
- Grisenti, R.; Schöllkopf, W.; Toennies, J.; Hegerfeldt, G.; Köhler, T.; Stoll, M. Determination of the bond length and binding energy of the helium dimer by diffraction from a transmission grating. Phys. Rev. Lett. 2000, 85, 2284. [Google Scholar] [CrossRef]
- Saez, J.S.; Garraleta, M.H.; Anton, P.C.; Alonso, M.F. Identification of 4-bromo-2-chlorophenol as a contaminant responsible for organoleptic taint in melons. Food Addit. Contam. 1991, 8, 627–631. [Google Scholar] [CrossRef]
- Liu, B.; McConnell, L.; Torrents, A. Hydrolysis of chlorpyrifos in natural waters of the Chesapeake Bay. Chemosphere 2001, 44, 1315–1323. [Google Scholar] [CrossRef]
- Karkmaz, M.; Puzenat, E.; Guillard, C.; Herrmann, J. Photocatalytic degradation of the alimentary azo dye amaranth: Mineralization of the azo group to nitrogen. Appl. Catal. B-Environ. 2004, 51, 183–194. [Google Scholar] [CrossRef]


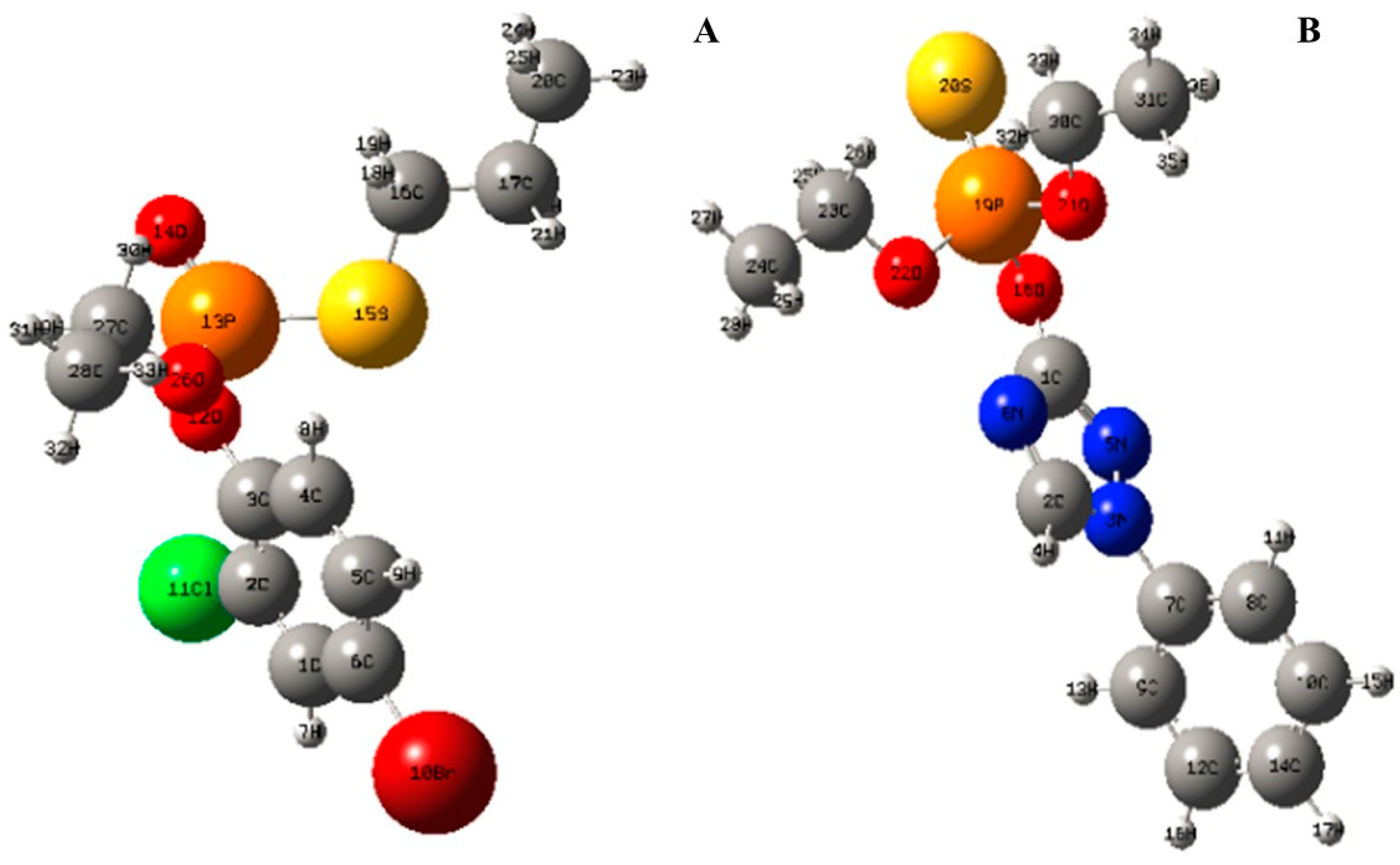
| Pesticide | Bond | Bond Length (×10−10 m) | Bond | Bond Length (×10−10 m) |
|---|---|---|---|---|
| Profenofos | P13-O14 | 1.45200 | C16-C17 | 1.50715 |
| P13-O26 | 1.76000 | C17-C20 | 1.50713 | |
| P13-O12 | 1.76000 | O26-C27 | 1.43000 | |
| P13-S15 | 2.11000 | C27-C28 | 1.50025 | |
| C16-S15 | 1.78000 | O12-C3 | 1.43000 | |
| C3-C4 | 1.39543 | C1-C2 | 1.39516 | |
| C4-C5 | 1.39483 | C2-Cl11 | 1.76000 | |
| C5-C6 | 1.39514 | C2-C3 | 1.39471 | |
| C6-Br10 | 1.91000 | C6-C1 | 1.39483 | |
| Triazophos | P19-S20 | 1.85680 | N3-C7 | 1.47000 |
| P19-O18 | 1.76000 | C7-C8 | 1.39516 | |
| P19-O21 | 1.76000 | C7-C9 | 1.39483 | |
| P19-O22 | 1.76000 | C8-C10 | 1.39471 | |
| O18-C1 | 1.43000 | C9-C12 | 1.39514 | |
| C1-N5 | 1.40175 | C10-C14 | 1.39543 | |
| C1-N6 | 1.43487 | C12-C14 | 1.39483 | |
| C2-N6 | 1.40185 | O21-C30 | 1.43000 | |
| C2-N3 | 1.39180 | O22-C23 | 1.43000 | |
| N3-N5 | 1.39180 | C30-C31 | 1.50025 | |
| C23-C24 | 1.50025 |
| Pesticide | Atom | Charge | Atom | Charge |
|---|---|---|---|---|
| Profenofos | C1 | −0.050417 | P13 | 1.020008 |
| C2 | 0.035499 | O14 | −0.505145 | |
| C3 | 0.114263 | S15 | 0.021148 | |
| C4 | −0.084254 | C16 | −0.191632 | |
| C5 | −0.050410 | C17 | −0.096471 | |
| C6 | −0.049715 | C20 | −0.177921 | |
| Br10 | 0.015273 | O26 | −0.381770 | |
| Cl11 | −0.139687 | C27 | 0.000156 | |
| O12 | −0.363450 | C28 | −0.180574 | |
| Triazophos | C1 | 0.249223 | C14 | −0.06174 |
| C2 | 0.138768 | O18 | −0.35249 | |
| N3 | −0.17545 | P19 | 1.018974 | |
| N5 | −0.16349 | S20 | −0.33818 | |
| N6 | −0.27804 | O21 | −0.36477 | |
| C7 | 0.116977 | O22 | −0.36329 | |
| C8 | −0.07132 | C23 | 0.002962 | |
| C9 | −0.06147 | C24 | −0.17913 | |
| C10 | −0.05342 | C30 | 0.00403 | |
| C12 | −0.0536 | C31 | −0.17975 |
| Analysis Conditions | Profenofos | Triazophos |
|---|---|---|
| Injector temperature | 250 °C | 220 °C |
| Detector temperature | Electron capture detector (ECD) 330 °C | Flame photometric detector (FPD) 250 °C |
| Injection volume | 1 µL | 1 µL |
| Split ratio | 5 | 5 |
| Carrier gas | Nitrogen | Nitrogen |
| Separation column | RTX-5 capillary column (30 m × 0.25 mm × 0.25 µm) | RTX-5 capillary column (30 m × 0.25 mm × 0.25 µm) |
| Column volume | 1.5 mL/min | 1.5 mL/min |
| Column temperature | Programmed at 150 °C, kept for 1 min, then increased from 150 °C to 240 °C at 10 °C/min, and kept at 240 °C for 5 min. | Programmed at 120 °C, kept for 1 min, next increased from 120 °C to 200 °C at 20 °C/min, kept for 6 min, then from 200 °C to 240 °C at 20 °C/min, and kept at 240 °C for 6 min. |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, X.; Zhan, Y.; Zhang, Z.; Pan, L.; Hu, L.; Liu, K.; Zhou, X.; Bai, L. Photocatalytic Degradation of Profenofos and Triazophos Residues in the Chinese Cabbage, Brassica chinensis, Using Ce-Doped TiO2. Catalysts 2019, 9, 294. https://doi.org/10.3390/catal9030294
Liu X, Zhan Y, Zhang Z, Pan L, Hu L, Liu K, Zhou X, Bai L. Photocatalytic Degradation of Profenofos and Triazophos Residues in the Chinese Cabbage, Brassica chinensis, Using Ce-Doped TiO2. Catalysts. 2019; 9(3):294. https://doi.org/10.3390/catal9030294
Chicago/Turabian StyleLiu, Xiangying, You Zhan, Zhongqin Zhang, Lang Pan, Lifeng Hu, Kailin Liu, Xuguo Zhou, and Lianyang Bai. 2019. "Photocatalytic Degradation of Profenofos and Triazophos Residues in the Chinese Cabbage, Brassica chinensis, Using Ce-Doped TiO2" Catalysts 9, no. 3: 294. https://doi.org/10.3390/catal9030294
APA StyleLiu, X., Zhan, Y., Zhang, Z., Pan, L., Hu, L., Liu, K., Zhou, X., & Bai, L. (2019). Photocatalytic Degradation of Profenofos and Triazophos Residues in the Chinese Cabbage, Brassica chinensis, Using Ce-Doped TiO2. Catalysts, 9(3), 294. https://doi.org/10.3390/catal9030294






