Preparation and Characterization of Rh/MgSNTs Catalyst for Hydroformylation of Vinyl Acetate: The Rh0 was Obtained by Calcination
Abstract
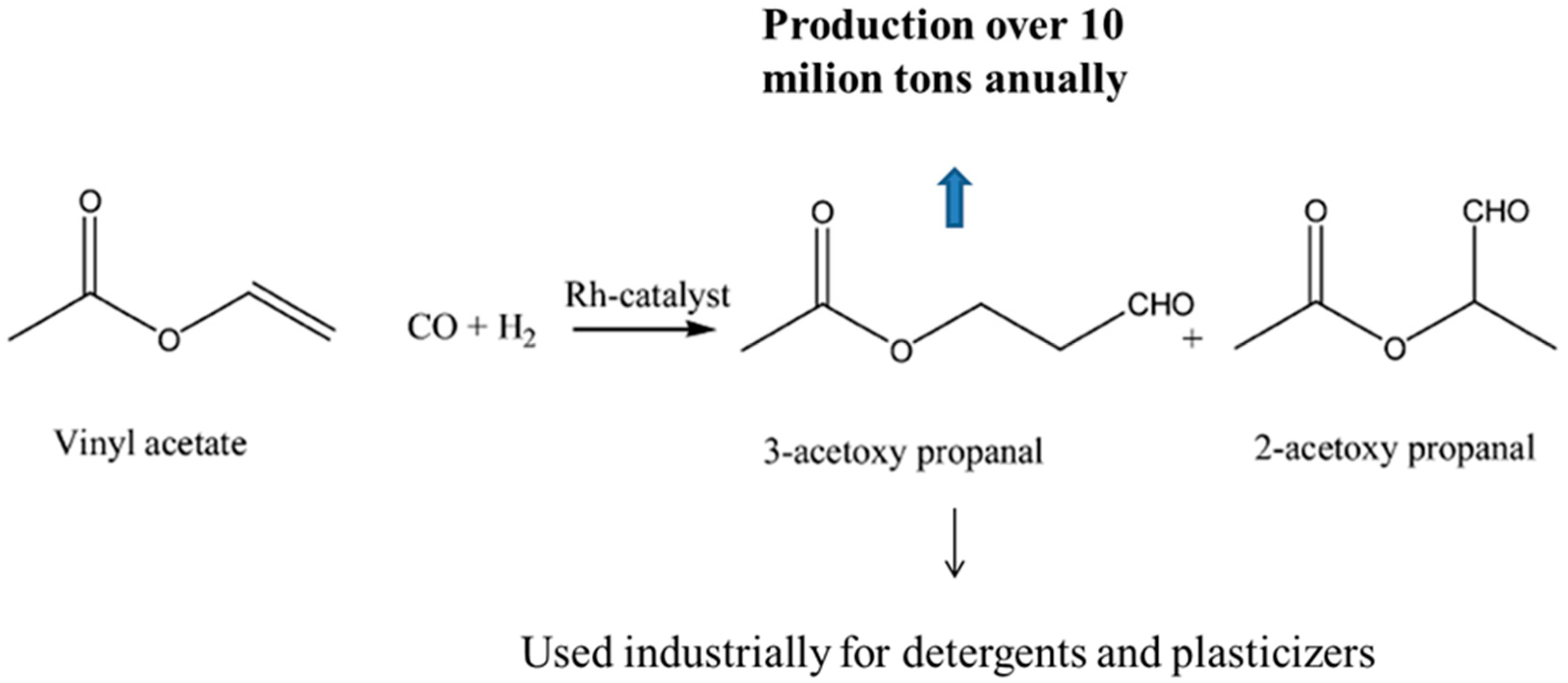
:1. Introduction
2. Materials and Methods
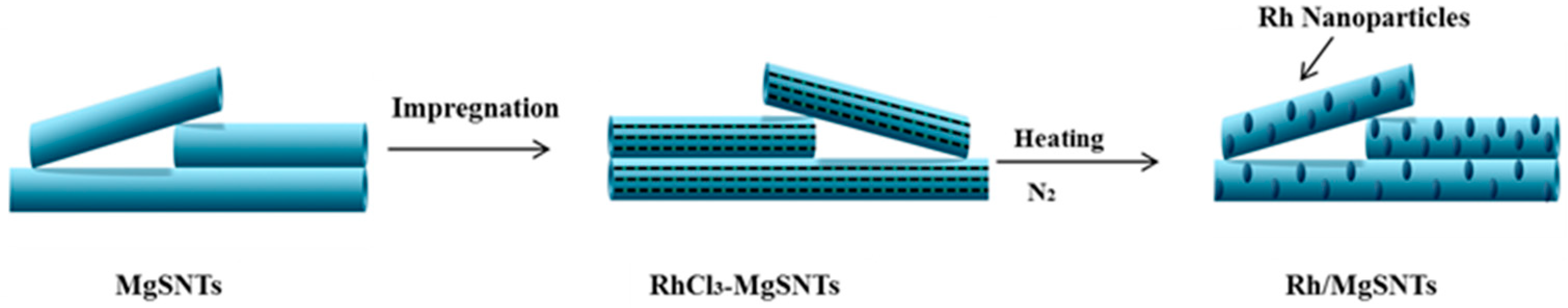
2.1. Preparation of MgSNTs Supported Rh-nanoparticle Catalysts
2.2. Evaluation of Catalytic Performance of Catalysts for Hydroformylation
2.3. Characterization
3. Results and Discussion
3.1. GC-MS Analysis
3.2. BET and ICP Analysis
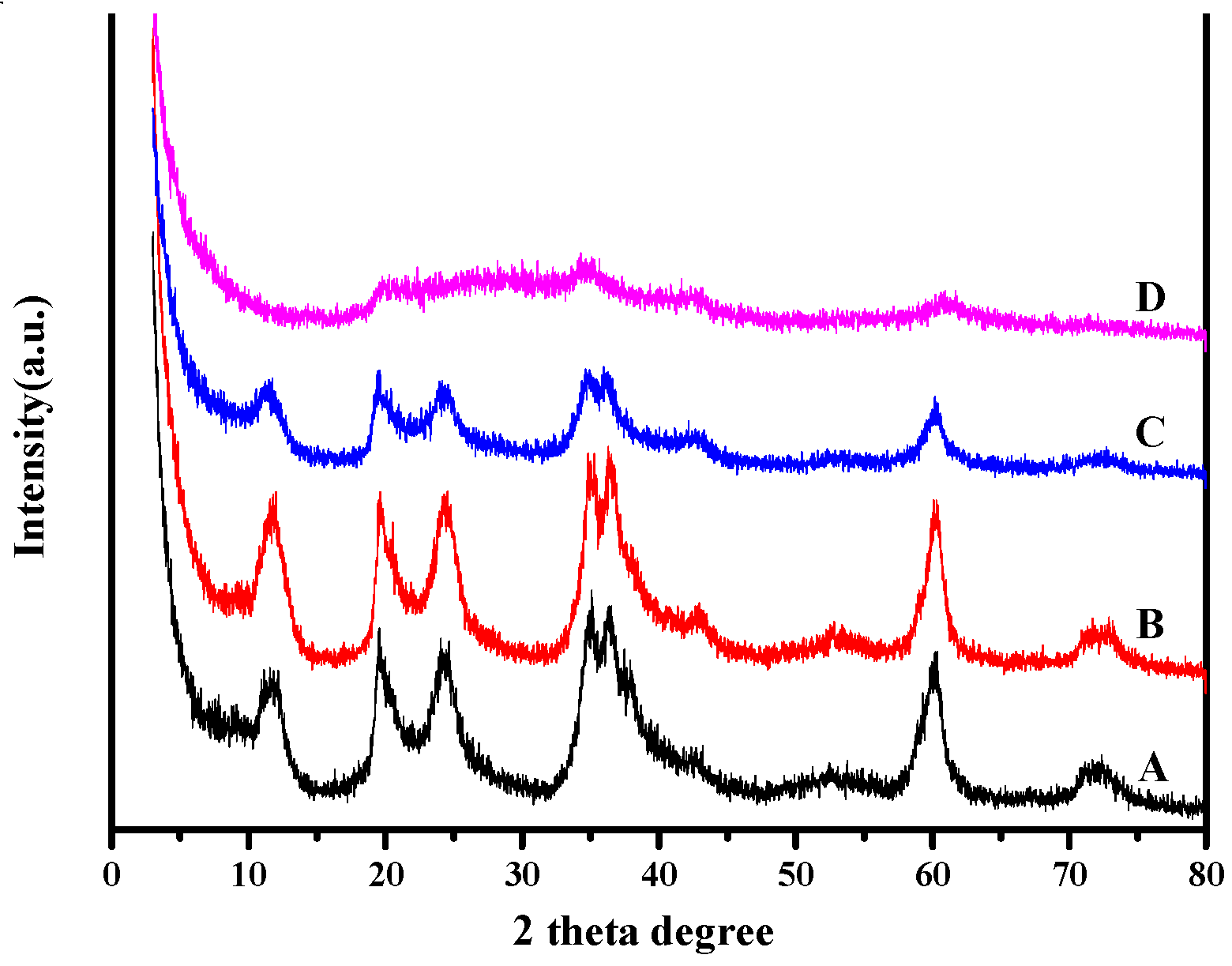
3.3. XRD Analysis
3.4. TEM Analysis
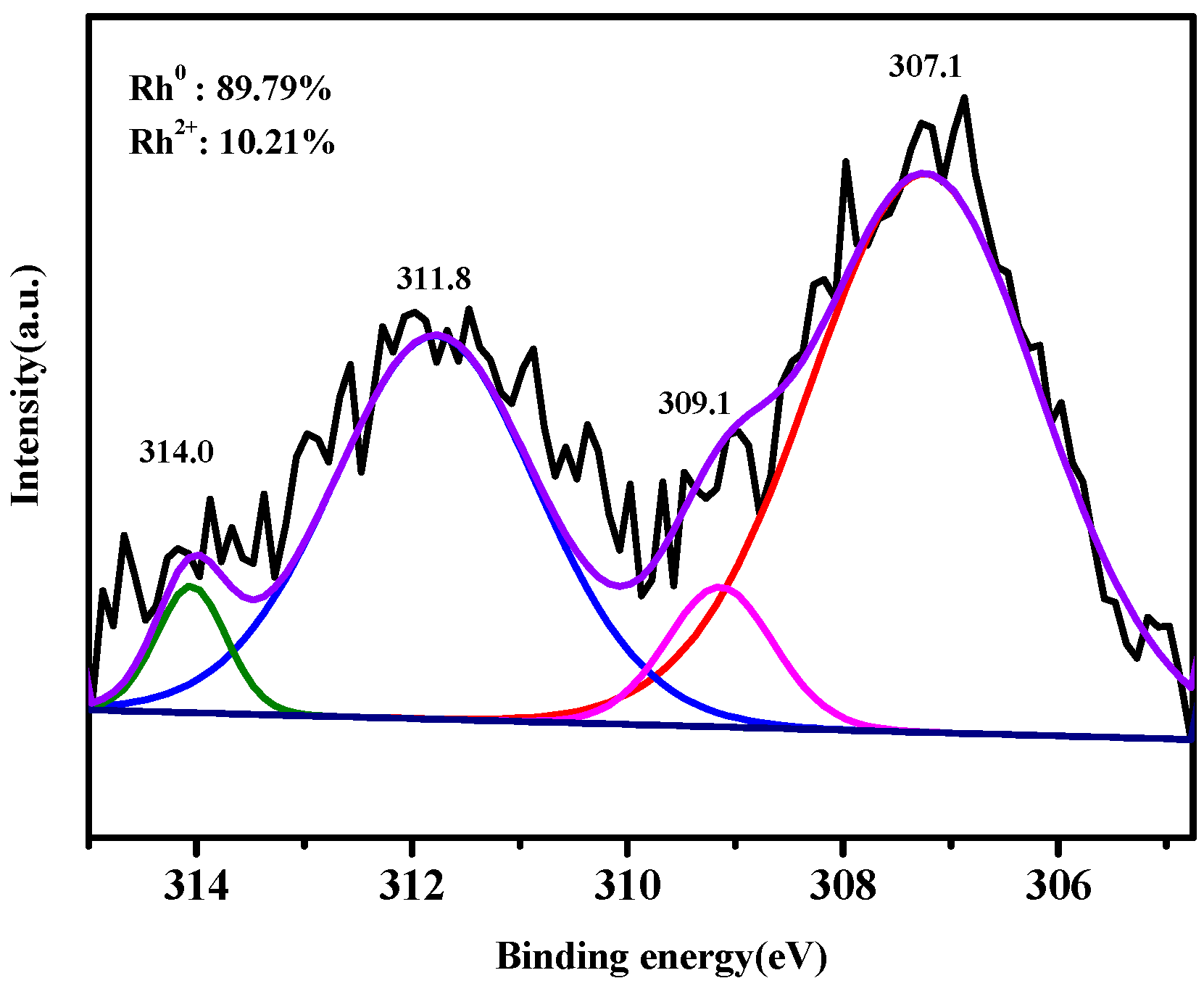
3.5. XPS Analysis
3.6. Catalytic Activity Evaluation
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Su, J.; Xie, C.L.; Chen, C.; Yu, Y.; Kennedy, G.; Somorjai, G.A.; Yang, P.D. Insights into the Mechanism of Tandem Alkene Hydroformylation over a Nanostructured Catalyst with Multiple Interfaces. J. Am. Chem. Soc. 2016, 138, 11568–11574. [Google Scholar] [CrossRef] [PubMed]
- You, C.; Li, X.X.; Yang, Y.H.; Yang, Y.S.; Tan, X.F.; Li, S.L.; Wei, B.; Lv, H.; Chung, L.W.; Zhang, X.M. Silicon-oriented regio- and enantioselective rhodium-catalyzed hydroformylation. Nat. Commun. 2018, 9, 2045. [Google Scholar] [CrossRef] [PubMed]
- Wu, Q.H.; Zhou, F.D.; Shu, X.; Jian, L.; Xu, B.; Zheng, X.L.; Yuan, M.L.; Fu, H.Y.; Li, R.X.; Chen, H. Synthesis and application of PNP pincer ligands in rhodium-catalyzed hydroformylation of cycloolefins. RSC Adv. 2016, 6, 107305–107309. [Google Scholar] [CrossRef]
- Nelsen, E.R.; Brezny, A.C.; Landis, C.R. Interception and Characterization of Catalyst Species in Rhodium Bis (diazaphospholane)-Catalyzed Hydroformylation of Octene, Vinyl Acetate, Allyl Cyanide, and 1-Phenyl-1,3-butadiene. J. Am. Chem. Soc. 2015, 137, 14208–14219. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.Y.; Dong, X.Q.; Zhang, X.M. Chiral Ligands for Rhodium-Catalyzed Asymmetric Hydroformylation: A Personal Account. Chem. Rec. 2016, 16, 2674–2686. [Google Scholar] [CrossRef] [PubMed]
- Schmitz, C.; Holthusen, K.; Leitner, W.; Francio, G. Highly Regio- and Enantioselective Hydroformylation of Vinyl Esters Using BidentatePhosphine, P-Chiral Phosphorodiamidite Ligands. ACS Catal. 2016, 6, 1584–1589. [Google Scholar] [CrossRef]
- Ma, Y.B.; Qing, S.J.; Yin, D.; Mamat, X.; Zhang, J.; Gao, Z.X.; Wang, T.F.; Eli, W. Rh-based catalysts supported on MCM-41-type mesoporous silica for dicyclopentadiene hydroformylation. Catal. Today 2015, 258, 64–69. [Google Scholar] [CrossRef]
- Ma, Y.B.; Fu, J.; Gao, Z.X.; Zhang, L.B.; Li, C.Y.; Wang, T.F. Dicyclopentadiene Hydroformylation to Value-Added Fine Chemicals over Magnetically Separable Fe3O4-Supported Co-Rh Bimetallic Catalysts: Effects of Cobalt Loading. Catalysts 2017, 7, 103. [Google Scholar] [CrossRef]
- Schmidt, S.; Deglmann, P.; Hofmann, P. Density Functional Investigations of the Rh-Catalyzed Hydroformylation of 1,3-Butadiene with Bisphosphite Ligands. ACS Catal. 2014, 4, 3593–3604. [Google Scholar] [CrossRef]
- Wu, L.Q.; Liu, Q.; Spannenberg, A.; Jackstell, R.; Beller, M. Highly regioselective osmium-catalyzed hydroformylation. Chem. Commun. 2015, 51, 3080–3082. [Google Scholar] [CrossRef] [PubMed]
- Piras, I.; Jennerjahn, R.; Jackstell, R.; Spannenberg, A.; Franke, R.; Beller, M. A General and Efficient Iridium-Catalyzed Hydroformylation of Olefins. Angew. Chem. Int. Ed. 2011, 123, 280–284. [Google Scholar] [CrossRef] [PubMed]
- Lang, R.; Li, T.B.; Matsumura, D.J.; Miao, S.; Ren, Y.J.; Cui, Y.T.; Tan, Y.; Qiao, B.T.; Li, L.; Wang, A.Q.; et al. Hydroformylationof Olefins by a Rhodium Single-Atom Catalyst with Activity Comparable to RhCl(PPh3)3. Angew. Chem. Int. Ed. 2016, 55, 16054–16058. [Google Scholar] [CrossRef] [PubMed]
- Bocokić, V.; Kalkan, A.; Lutz, M.; Spek, A.L.; Gryko, D.T.; Reek, J.N.H. Capsule-controlled selectivity of a rhodium hydroformylation catalyst. Nat. Commun. 2013, 4, 2670. [Google Scholar] [CrossRef] [PubMed]
- Benetskiy, E.; Lühr, S.; Vilches-Herrera, M.; Selent, D.; Jiao, H.J.; Domke, L.; Dyballa, K.; Franke, R.; Börner, A. Rhodium-Catalyzed Nonisomerizing Hydroformylation of Methyl Oleate Applying Lactame-Based Phosphoramidite Ligands. ACS Catal. 2014, 4, 2130–2136. [Google Scholar] [CrossRef]
- Cardozo, A.F.; Julcour, C.; Barthe, L.; Blanco, J.F.; Chen, S.; Gayet, F.; Manoury, E.; Zhang, X.W.; Lansalot, M.; Charleux, B.; et al. Aqueous phase homogeneous catalysis using core–shell nanoreactors: Application to rhodium-catalyzed hydroformylation of 1-octene. J. Catal. 2015, 324, 1–8. [Google Scholar] [CrossRef]
- Dahl, J.A.; Maddux, B.L.S.; Hutchison, J.E. Toward Greener Nanosynthesis. Chem. Rev. 2007, 107, 2228–2269. [Google Scholar] [CrossRef] [PubMed]
- Chuai, H.Y.; Liu, X.T.; Chen, Y.; Zhu, B.L.; Zhang, S.M.; Huang, W.P. Hydroformylation of vinyl acetate and cyclohexene over TiO2 nanotube supported Rh and Ru nanoparticle catalysts. RSC Adv. 2018, 8, 12053–12059. [Google Scholar] [CrossRef]
- Chen, Y.; Su, P.H.; Liu, T.; Liu, H.C.; Zhu, B.L.; Zhang, S.M.; Huang, W.P. Titanate nanotube-supported Au-Rh bimetallic catalysts: characterization and their catalytic performances in hydroformylation of vinyl acetate. Catalysts 2018, 8, 420. [Google Scholar] [CrossRef]
- Kwon, Y.; Kim, T.Y.; Kwon, G.; Yi, J.; Lee, H. Selective Activation of Methane on Single-Atom Catalyst of Rhodium Dispersed on Zirconia for Direct Conversion. J. Am. Chem. Soc. 2017, 139, 17694–17699. [Google Scholar] [CrossRef] [PubMed]
- Chen, F.; Jiang, X.Z.; Zhang, L.L.; Lang, R.; Qiao, B.T. Single-atom catalysis: Bridging the homo- and heterogeneous catalysis Chinese. J. Catal. 2018, 39, 893–898. [Google Scholar] [CrossRef]
- Guan, E.; Gates, B.C. Stable Rhodium Pair Sites on MgO: Influence of Ligands and Rhodium Nuclearity on Catalysis of Ethylene Hydrogenation and H-D Exchange in the Reaction of H2 with D2. ACS Catal. 2018, 8, 482–487. [Google Scholar] [CrossRef]
- Vunain, E.; Ncube, P.; Jalama, K.; Meijboom, R. Confinement effect of rhodium(I) complex species on mesoporous MCM-41 and SBA-15: effect of pore size on the hydroformylation of 1-octene. J. Porous. Mater. 2018, 25, 303–320. [Google Scholar] [CrossRef]
- Ahmed, M.; Sakthivel, A. Covalent grafting of cobalt carbonyl cluster on functionalized mesoporous SBA-15 molecular sieve and its applications towards hydroformylation of 1-octene. J. Mol. Catal. A-Chem. 2016, 424, 85–90. [Google Scholar] [CrossRef]
- Wang, Y.Q.; Yan, L.; Li, C.Y.; Jiang, M.; Wang, W.L.; Ding, Y.J. Highly efficient porous organic copolymer supported Rh catalysts for heterogeneous hydroformylation of butenes. Appl. Catal. A-Gen 2018, 551, 98–105. [Google Scholar] [CrossRef]
- Shi, Y.K.; Hu, X.J.; Zhu, B.L.; Wang, S.R.; Zhang, S.M.; Huang, W.P. Synthesis and characterization of TiO2 nanotube supported Rh-nanoparticle catalysts for regioselective hydroformylation of vinyl acetate. RSC Adv. 2014, 4, 62215–62222. [Google Scholar] [CrossRef]
- Du, X.J.; Fu, N.H.; Zhang, S.L.; Chen, C.; Wang, D.S.; Li, Y.D. Au/CuSiO3 nanotubes: High-performance robust catalysts for selective oxidation of ethanol to acetaldehyde. Nano Res. 2016, 9, 2681–2686. [Google Scholar] [CrossRef]
- Su, P.H.; Zhen, L.A.; Chen, Y.; Liu, X.T.; Zhu, B.L.; Zhang, S.M.; Huang, W.P. Catalytic performance for hydroformylation of cyclohexene of MgSNTs-supported amorphous Co-B catalyst. Chinese J. Inorg. Chem. 2018, 34, 2197–2204. [Google Scholar] [CrossRef]
- Khan, S.R.; Bhanage, B.M. Regioselective hydroformylation of vinyl esters catalyzed by Rh(acac)(CO)2 with simple and efficient diphosphinite ligands. Catal. Commun. 2014, 46, 109–112. [Google Scholar] [CrossRef]
- Simagina, V.I.; Netskina, O.V.; Komova, O.V.; Odegova, G.V.; Kochubei, D.I.; Ishchenko, A.V. Activity of Rh/TiO2 Catalysts in NaBH4 Hydrolysis: The Effect of the Interaction between RhCl3 and the Anatase Surface during Heat Treatment. Kinet. Catal. 2008, 49, 568–573. [Google Scholar] [CrossRef]
- Su, P.H.; Liu, X.T.; Chen, Y.; Liu, H.C.; Zhu, B.L.; Zhang, S.M.; Huang, W.P. Synthesis and characterization of Rh/B-TNTs as a recyclable catalyst for hydroformylation of olefin containing -CN functional group. Nanomaterials 2018, 8, 755. [Google Scholar] [CrossRef] [PubMed]
- Shi, Y.K.; Hu, X.J.; Chen, L.; Lu, Y.; Zhu, B.L.; Zhang, S.M.; Huang, W.P. Boron modified TiO2 nanotubes supported Rh-nanoparticle catalysts for highly efficient hydroformylation of styrene. New J. Chem. 2017, 41, 6120. [Google Scholar] [CrossRef]
- Simagina, V.I.; Storozhenko, P.A.; Netskina, O.V.; Komova, O.V.; Odegova, G.V.; Larichev, Y.V.; Ishchenko, A.V.; Ozerova, A.M. Development of catalysts for hydrogen generation from hydride compounds. Catal. Today 2008, 138, 253–259. [Google Scholar] [CrossRef]
- Gorbunov, D.; Safronova, D.; Kardasheva, Y.; Maximov, A.; Rosenberg, E.; Karakhanov, E. New heterogeneous Rh-Containing catalysts immobilized on a hybrid organic−inorganic surface for hydroformylation of unsaturated compounds. ACS Appl. Mater. Interfaces 2018, 10, 26566–26575. [Google Scholar] [CrossRef] [PubMed]


| Entry | Catalyst | SSA (m2/g) | Rh Content (wt%) |
|---|---|---|---|
| 1 | MgSNTs-300 | 250.6 | - |
| 2 | Rh/MgSNTs-300(a1) | 238.2 | 0.30 |
| 3 | Rh/MgSNTs-300(a2) | 236.7 | 0.41 |
| 4 | Rh/MgSNTs-300(a3) | 229.6 | 0.72 |
| 5 | Rh/MgSNTs-300(b1) | 236.5 | 0.42 |
| 6 | Rh/MgSNTs-300(b2) | 218.6 | 0.43 |
| 7 | Rh/MgSNTs-300(b3) | 211.2 | 0.42 |
| Catalyst | Theoretical Load ICP | Rh Content (wt%) | Conversion (%) | TOF b (h−1) | Aldehyde (%) | b:l c |
|---|---|---|---|---|---|---|
| Rh/MgSNTs-300(a1) | 0.35 | 0.16 | 74 | 82 | 69 | 96:4 |
| Rh/MgSNTs-300(a2) | 0.5 | 0.31 | 97 | 161 | 74 | 91:9 |
| Rh/MgSNTs-300(a3) | 1.0 | 0.53 | 99 | 181 | 75 | 95:5 |
| Catalyst | Temperature (°C) | Conversion (%) | By Products (%) | TOF b (h−1) | Aldehyde (%) | b:l c | |
|---|---|---|---|---|---|---|---|
| Propanal | Ethyl Acetate | ||||||
| Rh/MgSNTs-300(a2) | 60 | 22 | 22 | 13 | 29 | 65 | 99:1 |
| Rh/MgSNTs-300(a2) | 80 | 43 | 23 | 15 | 68 | 62 | 98:2 |
| Rh/MgSNTs-300(a2) | 110 | 90 | 24 | 11 | 161 | 68 | 91:9 |
| Rh/MgSNTs-300(a2) | 120 | 92 | 25 | 11 | 179 | 64 | 95:5 |
| Catalyst | Calcination Temperature (°C) | Conversion (%) | TOF b (h−1) | Aldehyde (%) | b:l c |
|---|---|---|---|---|---|
| Rh/MgSNTs-300(b1) | 200 | 68 | 101 | 45 | 99:1 |
| Rh/MgSNTs-300(a2) | 300 | 97 | 161 | 68 | 91:9 |
| Rh/MgSNTs-300(b2) | 400 | 91 | 165 | 69 | 92:8 |
| Rh/MgSNTs-300(b3) | 500 | 75 | 132 | 56 | 98:2 |
| Catalyst | Cycle Times | Conversion (%) | By Products (%) | Aldehyde (%) | b:l b | |
|---|---|---|---|---|---|---|
| Propanal | Ethyl Acetate | |||||
| Rh/MgSNTs-300(a2) | 1 | 97 | 19 | 13 | 68 | 91:9 |
| Rh/MgSNTs-300(a2) | 2 | 82 | 19 | 15 | 66 | 92:8 |
| Rh/MgSNTs-300(a2) | 3 | 74 | 23 | 19 | 58 | 95:5 |
| Rh/MgSNTs-300(a2) | 4 | 58 | 24 | 22 | 54 | 97:3 |
| Catalyst | Cycle Times | Rh (PPM) |
|---|---|---|
| Rh/MgSNTs-300 | 1 | 20.3 |
| Rh/MgSNTs-300 | 2 | 16.4 |
| Rh/MgSNTs-300 | 3 | 15.7 |
| Rh/MgSNTs-300 | 4 | 9.7 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Su, P.; Chen, Y.; Liu, X.; Chuai, H.; Liu, H.; Zhu, B.; Zhang, S.; Huang, W. Preparation and Characterization of Rh/MgSNTs Catalyst for Hydroformylation of Vinyl Acetate: The Rh0 was Obtained by Calcination. Catalysts 2019, 9, 215. https://doi.org/10.3390/catal9030215
Su P, Chen Y, Liu X, Chuai H, Liu H, Zhu B, Zhang S, Huang W. Preparation and Characterization of Rh/MgSNTs Catalyst for Hydroformylation of Vinyl Acetate: The Rh0 was Obtained by Calcination. Catalysts. 2019; 9(3):215. https://doi.org/10.3390/catal9030215
Chicago/Turabian StyleSu, Penghe, Ya Chen, Xiaotong Liu, Hongyuan Chuai, Hongchi Liu, Baolin Zhu, Shoumin Zhang, and Weiping Huang. 2019. "Preparation and Characterization of Rh/MgSNTs Catalyst for Hydroformylation of Vinyl Acetate: The Rh0 was Obtained by Calcination" Catalysts 9, no. 3: 215. https://doi.org/10.3390/catal9030215
APA StyleSu, P., Chen, Y., Liu, X., Chuai, H., Liu, H., Zhu, B., Zhang, S., & Huang, W. (2019). Preparation and Characterization of Rh/MgSNTs Catalyst for Hydroformylation of Vinyl Acetate: The Rh0 was Obtained by Calcination. Catalysts, 9(3), 215. https://doi.org/10.3390/catal9030215





