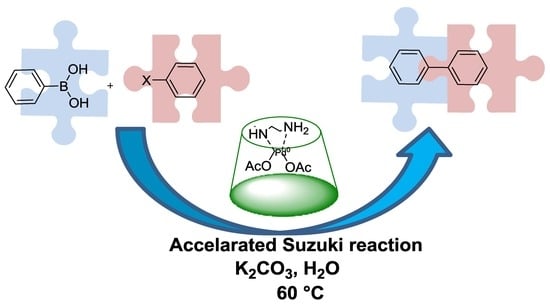
An Amino-Chain Modified β-cyclodextrin: A Supramolecular Ligand for Pd(OAc)2 Acceleration in Suzuki–Miyaura Coupling Reactions in Water
Abstract
:1. Introduction
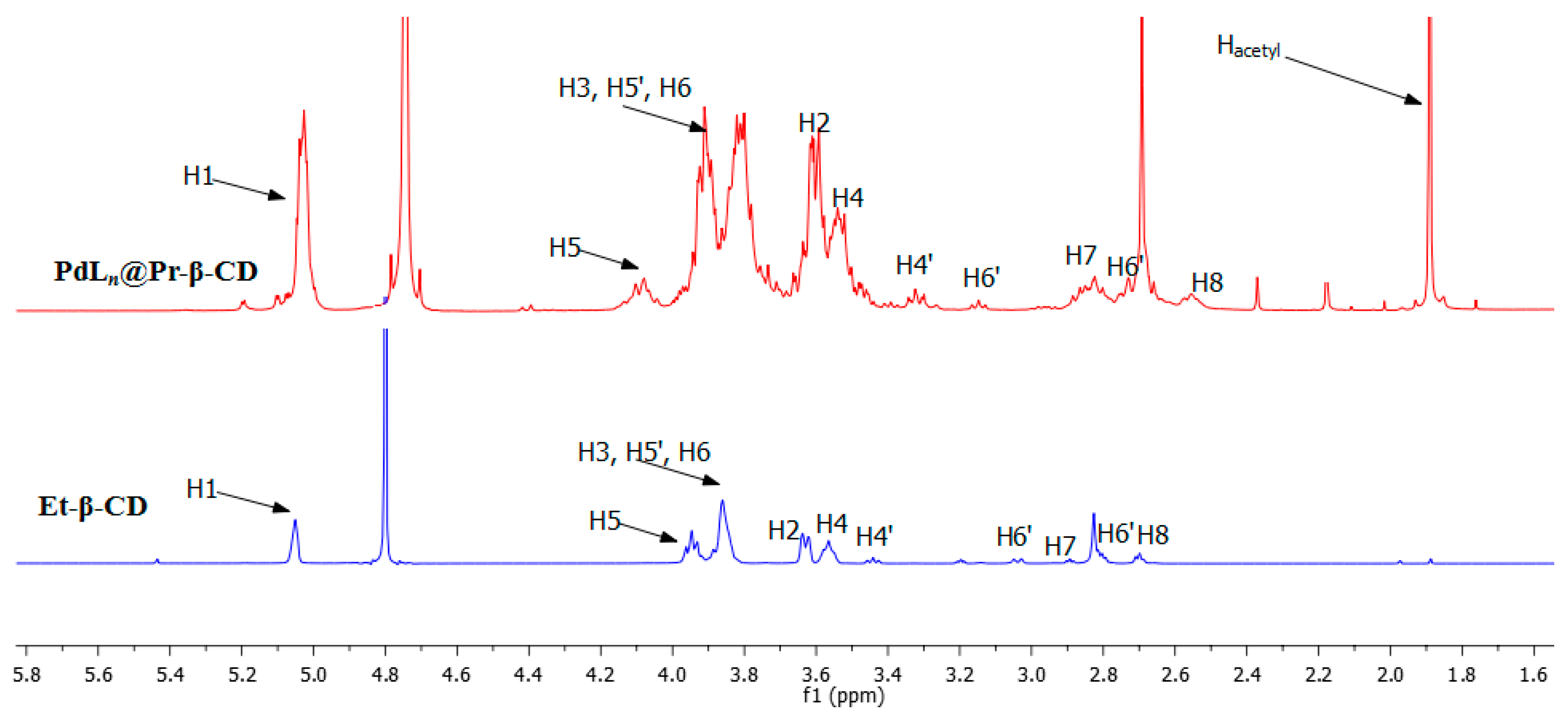
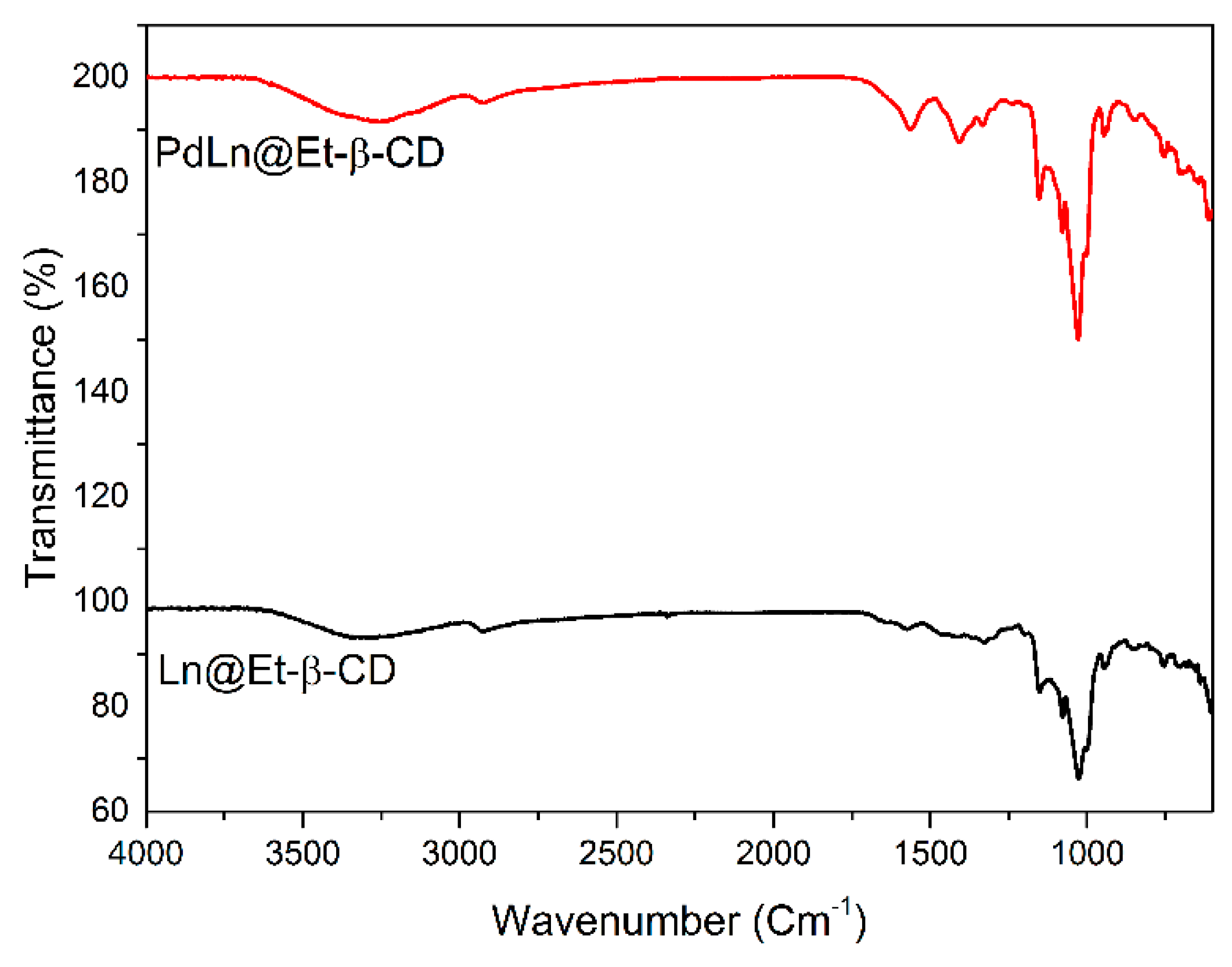
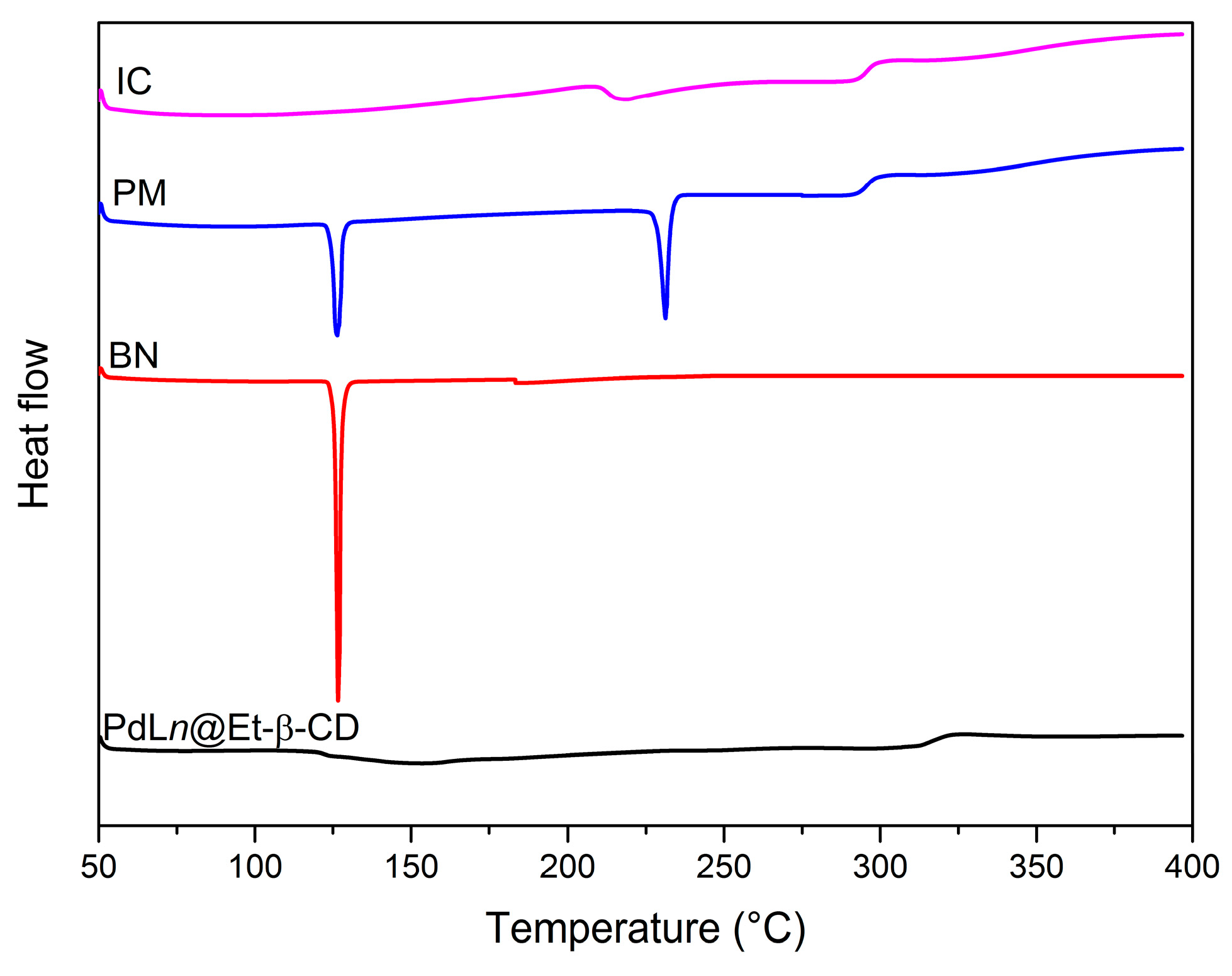
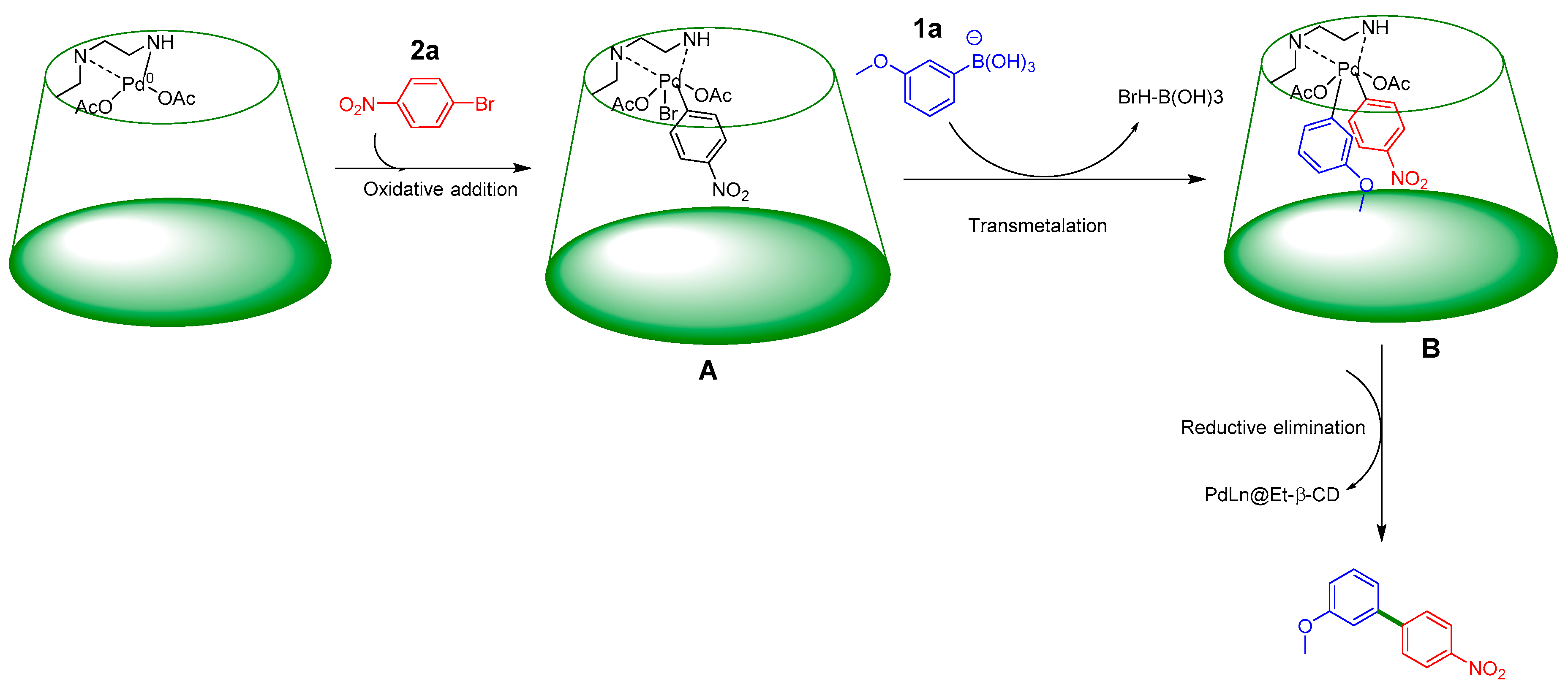
2. Results and Discussion
3. Experimental Materials
3.1. Characterization
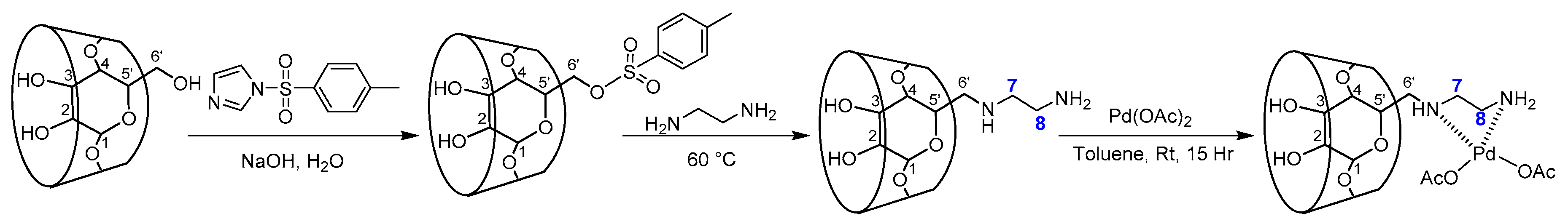
3.2. Synthesis of Mono-6-O-p-toluenesulfonyl-β-cyclodextrin (tosyl-β-CD)
3.3. Synthesis of Mono-6-deoxy-6-aminoethylamino-β-cyclodextrin (Et-β-CD) [39]
3.4. Synthesis of PdLn@Et-β-CD Catalyst
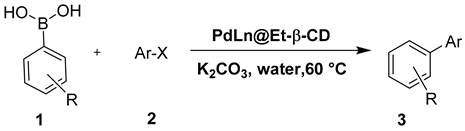
3.5. General Procedure for the Suzuki–Miyaura Coupling Reactions
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Molnár, A.R.D. Efficient, selective, and recyclable palladium catalysts in carbon–carbon coupling reactions. Chem. Rev. 2011, 111, 2251–2320. [Google Scholar] [CrossRef] [PubMed]
- Glasspoole, B.W.; Crudden, C.M. Cross-coupling: The final frontier. Nat. Chem. 2011, 3, 912–913. [Google Scholar] [CrossRef] [PubMed]
- Fihri, A.; Bouhrara, M.; Nekoueishahraki, B.; Basset, J.-M.; Polshettiwar, V. Nanocatalysts for Suzuki cross-coupling reactions. Chem. Soc. Rev. 2011, 40, 5181–5203. [Google Scholar] [CrossRef] [PubMed]
- Saito, N.; Taniguchi, T.; Hoshiya, N.; Shuto, S.; Arisawa, M.; Sato, Y. Double carbonylation of aryl iodides with amines under an atmospheric pressure of carbon monoxide using sulfur-modified Au-supported palladium. Green Chem. 2015, 17, 2358–2361. [Google Scholar] [CrossRef]
- Dodson, J.R.; Hunt, A.J.; Parker, H.L.; Yang, Y.; Clark, J.H. Elemental sustainability: Towards the total recovery of scarce metals. Chem. Eng. Process. 2012, 51, 69–78. [Google Scholar] [CrossRef]
- Lamblin, M.; Nassar-Hardy, L.; Hierso, J.C.; Fouquet, E.; Felpin, F.X. Recyclable Heterogeneous Palladium Catalysts in Pure Water: Sustainable Developments in Suzuki, Heck, Sonogashira and Tsuji–Trost Reactions. Adv. Synth. Catal. 2010, 352, 33–79. [Google Scholar] [CrossRef]
- Lee, D.H.; Kim, J.H.; Jun, B.H.; Kang, H.; Park, J.Y.; Lee, S. Macroporous polystyrene-supported palladium catalyst containing a bulky N-heterocyclic carbene ligand for Suzuki reaction of aryl chlorides. Org. Lett. 2008, 10, 1609–1612. [Google Scholar] [CrossRef] [PubMed]
- Sharma, P.; Singh, A.P. Phosphine free SBA-15–EDTA–Pd highly active recyclable catalyst: Synthesis characterization and application for Suzuki and Sonogashira reaction. Catal. Sci. Technol. 2014, 4, 2978–2989. [Google Scholar] [CrossRef]
- Taylor, R.H.; Felpin, F.X. Suzuki−Miyaura Reactions of Arenediazonium Salts Catalyzed by Pd(0)/C. One-Pot Chemoselective Double Cross-Coupling Reactions. Org. Lett. 2007, 9, 2911–2914. [Google Scholar] [CrossRef] [PubMed]
- Elazab, H.A.; Siamaki, A.R.; Moussa, S.; Gupton, B.F.; El-Shall, M.S. Highly efficient and magnetically recyclable graphene-supported Pd/Fe3O4 nanoparticle catalysts for Suzuki and Heck cross-coupling reactions. Appl. Catal. A Gener. 2015, 491, 58–69. [Google Scholar] [CrossRef]
- Zhang, Q.; Su, H.; Luo, J.; Wei, Y.Y. “Click” magnetic nanoparticle-supported palladium catalyst: A phosphine-free, highly efficient and magnetically recoverable catalyst for Suzuki–Miyaura coupling reactions. Catal. Sci. Technol. 2013, 3, 235–243. [Google Scholar] [CrossRef]
- Karimi, B.; Mansouri, F.; Vali, H. A highly water-dispersible/magnetically separable palladium catalyst based on a Fe3O4@SiO2 anchored TEG-imidazolium ionic liquid for the Suzuki–Miyaura coupling reaction in water. Green Chem. 2014, 16, 2587–2596. [Google Scholar] [CrossRef]
- Höppener, S.; Schubert, U.S.; Ignaszak, A. Electrodeposited palladium on MWCNTs as ‘semi-soluble heterogeneous’ catalyst for cross-coupling reactions. Tetrahedron Lett. 2015, 56, 4084–4087. [Google Scholar]
- Bortolotto, T.; Facchinetto, S.E.; Trindade, S.G.; Ossig, A.; Petzhold, C.L.; Vargas, J.; Rodrigues, O.E.D.; Giacomelli, C.; Schmidt, V. Polymer-coated palladium nanoparticle catalysts for Suzuki coupling reactions. J. Colloid Interface Sci. 2015, 439, 154–161. [Google Scholar] [CrossRef] [PubMed]
- Sharma, R.K.; Sharma, S.; Dutta, S.; Zborilb, R.; Gawande, M.B. Silica-nanosphere-based organic–inorganic hybrid nanomaterials: Synthesis, functionalization and applications in catalysis. Green Chem. 2015, 17, 3207–3230. [Google Scholar] [CrossRef]
- Ogasawara, S.; Kato, S. Palladium Nanoparticles Captured in Microporous Polymers: A Tailor-Made Catalyst for Heterogeneous Carbon Cross-Coupling Reactions. J. Am. Chem. Soc. 2010, 132, 4608–4613. [Google Scholar] [CrossRef] [PubMed]
- Duan, L.; Fu, R.; Xiao, Z.; Zhao, Q.; Wang, J.Q.; Chen, S.; Wan, Y. Activation of Aryl Chlorides in Water under Phase-Transfer Agent-Free and Ligand-Free Suzuki Coupling by Heterogeneous Palladium Supported on Hybrid Mesoporous Carbon. ACS Catal. 2015, 5, 575–586. [Google Scholar] [CrossRef]
- Gómez-Martínez, M.; Buxaderas, E.; Pastor, I.M.; Alonso, D.A. Palladium nanoparticles supported on graphene and reduced graphene oxide as efficient recyclable catalyst for the Suzuki–Miyaura reaction of potassium aryltrifluoroborates. J. Mol. Catal. A 2015, 404–405, 1–7. [Google Scholar] [CrossRef]
- Putta, C.; Sharavath, V.; Sarkara, S.; Ghosh, S. Palladium nanoparticles on b-cyclodextrin functionalised graphene nanosheets: A supramolecular based heterogeneous catalyst for C–C coupling reactions under green reaction conditions. RSC Adv. 2015, 5, 6652–6660. [Google Scholar] [CrossRef]
- Qi, M.; Tan, P.Z.; Xue, F.; Malhi, H.S.; Zhang, Z.X.; Young, D.J.; Andy Hor, T.S. A supramolecular recyclable catalyst for aqueous Suzuki–Miyaura coupling. RSC Adv. 2015, 5, 3590–3596. [Google Scholar] [CrossRef]
- Guitet, M.; Marcelo, F.; Beaumais, S.A.; Zhang, Y.; Jiménez-Barbero, J.; Tilloy, S.; Monflier, E.; Ménand, M.; Sollogoub, M. Diametrically Opposed Carbenes on anα-Cyclodextrin: Synthesis, Characterization of Organometallic Complexes and Suzuki–Miyaura Couplingin Ethanol and in Water. Eur. J. Org. Chem. 2013, 3691–3696. [Google Scholar] [CrossRef]
- Cassez, A.; Ponchel, A.; Hapiot, F.; Monflier, E. Unexpected Multifunctional Effects of Methylated Cyclodextrins in a Palladium Charcoal-Catalyzed Suzuki−Miyaura Reaction. Org. Lett. 2006, 8, 4823–4826. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Li, J.; Shi, X.; Liu, Y.; Xie, K.; Liu, Y.; Jiang, Y.; Yang, B.; Yang, R. Cyclodextrin-supported palladium complex: A highly active and recoverable catalyst for Suzuki–Miyaura cross-coupling reaction in aqueous medium. Appl. Organometal. Chem. 2017, 31, e3592. [Google Scholar] [CrossRef]
- Legrand, F.; Ménand, M.; Sollogoub, M.; Tilloy, S.; Monflier, E. An N-heterocyclic carbene ligand based on a β-cyclodextrin–imidazolium salt: Synthesis, characterization of organometallic complexes and Suzuki coupling. New J. Chem. 2011, 35, 2061–2065. [Google Scholar] [CrossRef]
- Yang, R.; Chen, J.B.; Xiao, C.F.; Liu, Z.C.; Gao, Z.Y.; Yan, S.J.; Zhang, J.H.; Zhang, H.B.; Lin, J. Inclusion complex of GA-13316 with -cyclodextrin: Preparation, characterization, molecular modeling, and in vitro evaluation. Carbohydr. Polym. 2014, 111, 655–662. [Google Scholar] [CrossRef]
- Wang, F.; Yang, B.; Zhao, Y.L.; Liao, X.L.; Gao, C.Z.; Jiang, R.J.; Han, B.; Yang, J.; Liu, M.; Zhou, R.G. Host-guest inclusion system of scutellarein with 2-hydroxypropyl-beta-cyclodextrin: Preparation, characterization, and anticancer activity. J. Biomater. Sci. Polym. E 2014, 6, 594–607. [Google Scholar] [CrossRef] [PubMed]
- Thombal, R.S.; Jadhav, V.H. Biomass derived -cyclodextrin-SO3H carbonaceous solid acid catalyst for catalytic conversion of carbohydrates to 5-hydroxymethylfurfural. Appl. Catal. A 2015, 499, 213–216. [Google Scholar] [CrossRef]
- Khana, R.I.; Pitchumani, K. A pyridinium modified β-cyclodextrin: An ionic supramolecular ligand for palladium acetate in C–C coupling reactions in water. Green Chem. 2016, 18, 5518–5528. [Google Scholar] [CrossRef]
- Dindulkar, S.D.; Jeong, D.; Kim, H.; Jung, S. Functionalized β-cyclodextrin as supramolecular ligand and their Pd(OAc)2 complex: Highly efficient and reusable catalyst for Mizoroki–Heck cross-coupling reactions in aqueous medium. Carbohydr. Res. 2016, 430, 85–94. [Google Scholar] [CrossRef]
- Bellia, F.; La Mendola, D.; Pedone, C.; Rizzarelli, E.; Saviano, M.; Vecchio, G. Selectively functionalized cyclodextrins and their metal complexes. Chem. Soc. Rev. 2009, 38, 2756–2781. [Google Scholar] [CrossRef]
- Hapiot, F.; Ponchel, A.; Tilloy, S.; Monflier, E. Cyclodextrins and their applications in aqueous-phase metal-catalyzed reactions. C. R. Chim. 2011, 14, 149–166. [Google Scholar] [CrossRef]
- Xue, C.; Palaniappan, K.; Arumugam, G.; Hackney, S.A.; Liu, J.; Liu, H. Sonogashira reactions catalyzed by water-soluble, b-cyclodextrin-capped palladium nanoparticles. Catal. Lett. 2007, 116, 94–100. [Google Scholar] [CrossRef]
- Saha, S.; Roy, A.; Roy, K.; Roy, M.N. Study to explore the mechanism to form inclusion complexes of β-cyclodextrin with vitamin molecules. Sci. Rep. 2016, 6, 35764. [Google Scholar] [CrossRef]
- Patel, R.; Patel, M. Solid-state characterization and in vitro dissolution behavior of lorazepam: Hydroxypropyl-β-cyclodextrin inclusion complex. Drug Discov. Ther. 2010, 4, 442–452. [Google Scholar]
- de Araujo, D.R.; Tsuneda, S.S.; Cereda, C.M.; Carvalho, F.D.G.; Preté, P.S.; Fernandes, S.A.; Yokaichiya, F.; Franco, M.K.; Mazzaro, I.; Fraceto, L.F.; et al. Development and pharmacological evaluation of ropivacaine-2-hydroxypropyl-beta-cyclodextrin inclusion complex. Eur. J. Pharm. Sci. 2008, 33, 60–71. [Google Scholar] [CrossRef]
- Kamphorst, A.O.; de Sá, I.M.; Faria, A.M.C.; Sinisterra, R.D. Association complexes between ovalbumin and cyclodextrins have no effect on the immunological properties of ovalbumin. Eur. J. Pharm. Biopharm. 2004, 57, 199–205. [Google Scholar] [CrossRef]
- Schmidt, A.F.; Kurokhtina, A.A. Distinguishing between the homogeneous and heterogeneous mechanisms of catalysis in the Mizoroki-Heck and Suzuki-Miyaura reactions: Problems and prospects. Kinet. Catal. 2012, 53, 714–730. [Google Scholar] [CrossRef]
- Byun, H.S.; Zhong, N.; Bittman, R. 6A-O-p-Toluenesulfonyl-β-Cyclodextrin. Org. Synth. 2000, 77, 225–232. [Google Scholar]
- Bonomo, R.P.; Cucinotta, V.; Allessandro, F.D.; Impellizzeri, G.; Maccarrone, G.; Rizzarelli, E.; Vecchio, G. Coordination properties of 6-deoxy-6-[1-(2-amino) ethylamino]-β-cyclodextrin and the ability of itscopper(II) complex to recognize and separate amino acid enantiomeric pairs. J. Incl. Phenom. Mol. Recognit. Chem. 1993, 15, 167–180. [Google Scholar] [CrossRef]
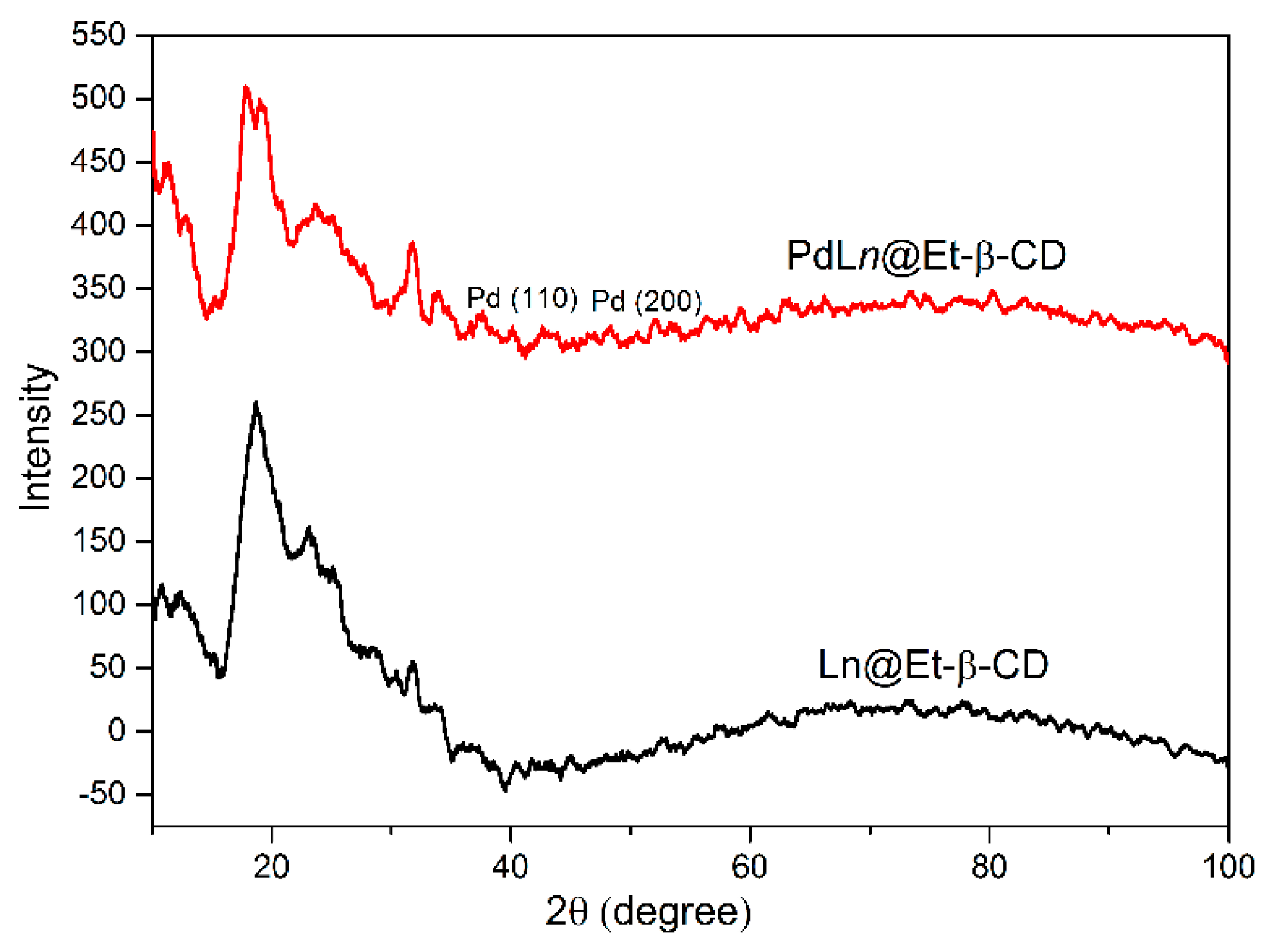
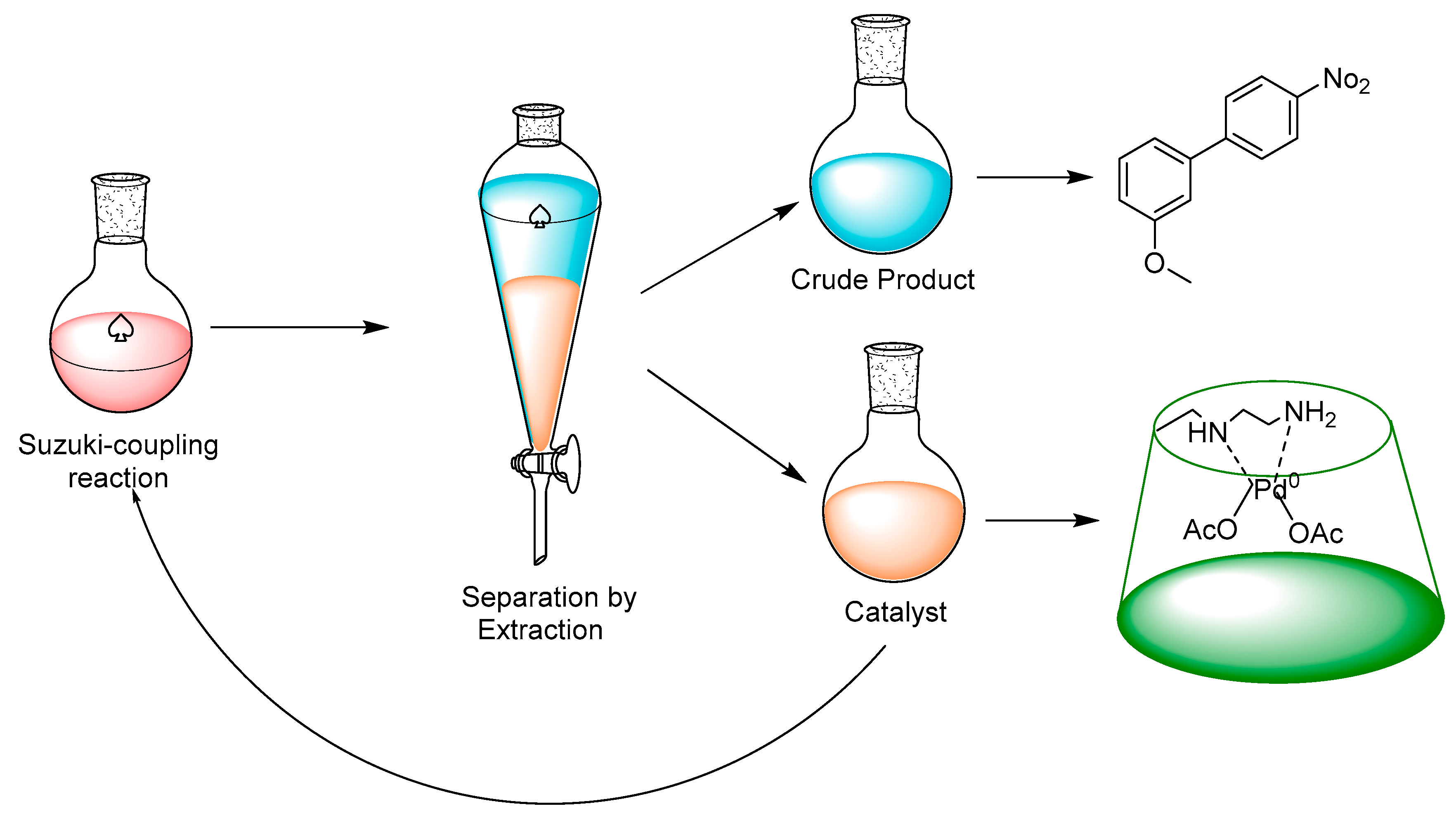
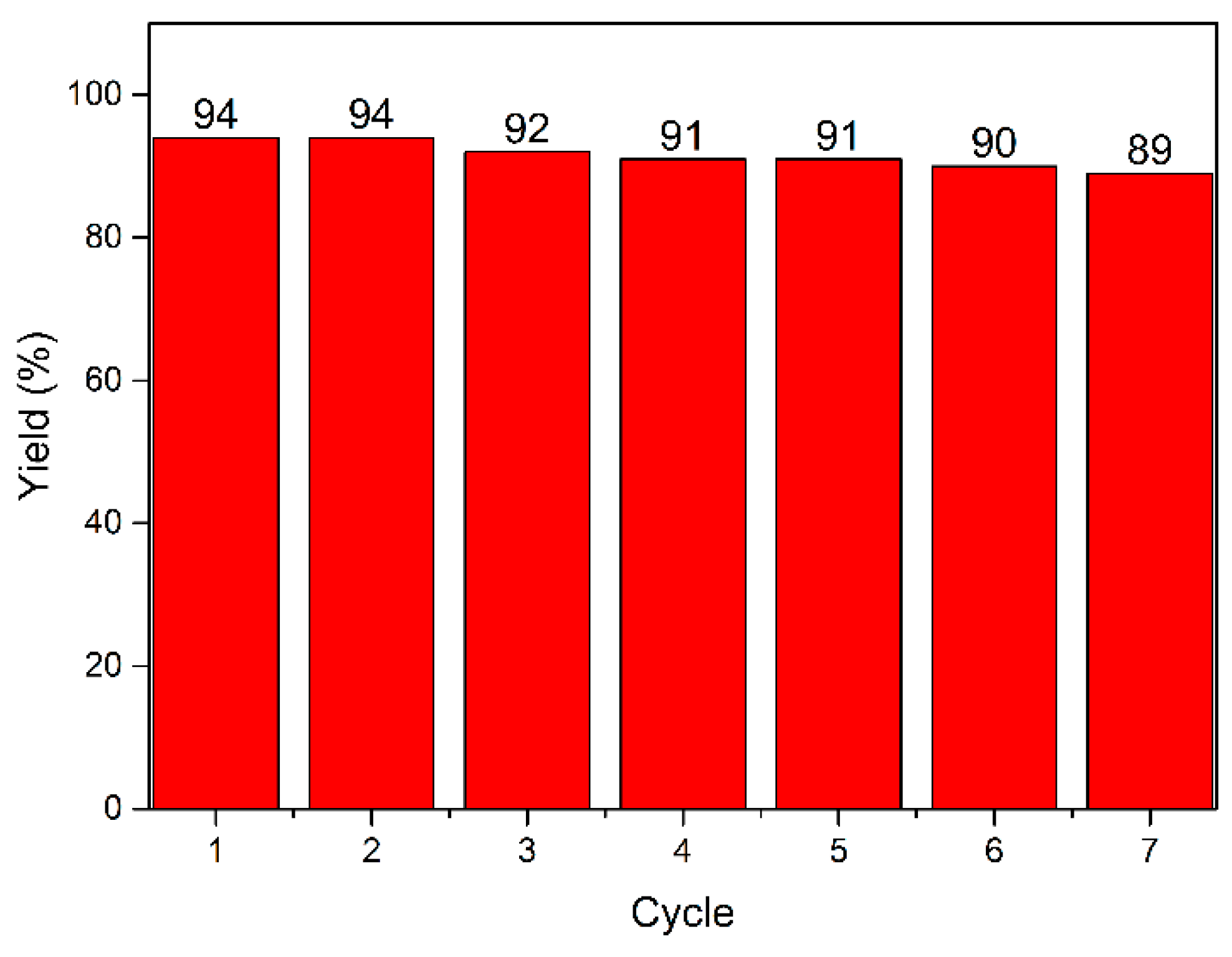
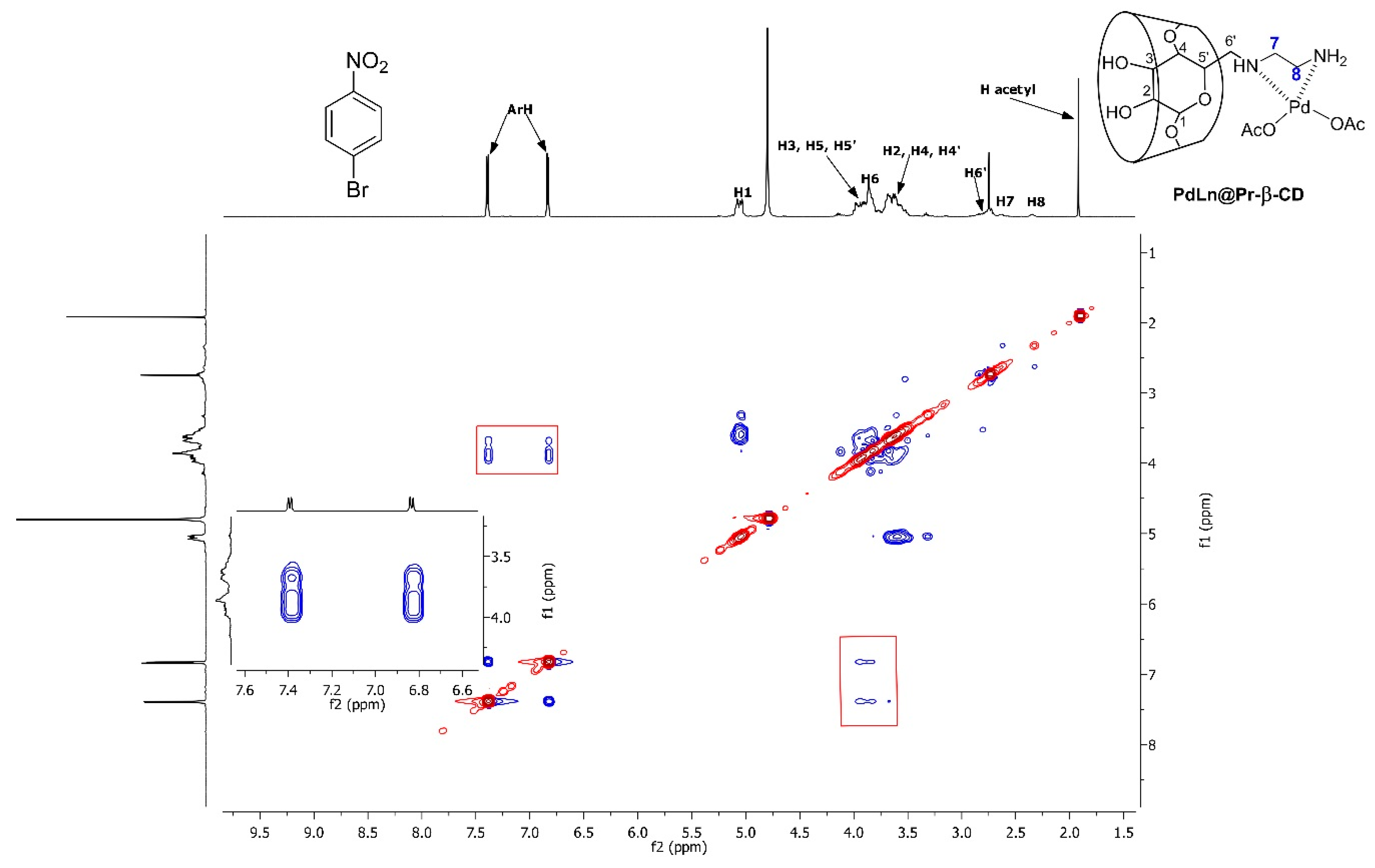


| Entry | Catalyst (mol%) | Base | Temperature (°C) | Time (h) | Yield (%) b |
|---|---|---|---|---|---|
| 1 | PdLn@Et-β-CD (1) | Na2CO3 | 60 | 2 | 71 |
| 2 | PdLn@Et-β-CD (1) | Cs2CO3 | 60 | 2 | 76 |
| 3 | PdLn@Et-β-CD (1) | K3PO4 | 60 | 3 | 68 |
| 4 | PdLn@Et-β-CD (1) | NaHCO3 | 60 | 3 | 59 |
| 5 | PdLn@Et-β-CD (1) | K2CO3 | 60 | 1 | 90 |
| 6 | PdLn@Et-β-CD (1) | - | 60 | 14 | - |
| 7 | PdLn@Et-β-CD (1.5) | K2CO3 | 60 | 1 | 90 |
| 8 | PdLn@Et-β-CD (2) | K2CO3 | 60 | 1 | 94 |
| 9 | PdLn@Et-β-CD (2.5) | K2CO3 | 60 | 1 | 94 |
| 10 | PdLn@Et-β-CD (3) | K2CO3 | 60 | 1 | 93 |
| 11 | PdLn@Et-β-CD (2) | K2CO3 | 40 | 6 | 72 |
| 12 | PdLn@Et-β-CD (2) | K2CO3 | 50 | 4 | 84 |
| 13 | PdLn@Et-β-CD (2) | K2CO3 | 70 | 1 | 94 |
| 14 | PdLn@Et-β-CD (2) | K2CO3 | 90 | 1 | 94 |
| 15 | PdLn@Et-β-CD (2) | K2CO3 | 100 | 1 | 94 |
| 16 | β-CD (5)/Pd(OAc)2 (2.5) | K2CO3 | 60 | 15 | 11 |
| Entry | R | Ar | X | Compound | Yield (%) b |
|---|---|---|---|---|---|
| 1 | 4-CH3O-C6H4 | 4-NO2 | Br | 3a | 94 |
| 2 | Ph | 4-NO2 | Br | 3b | 98 |
| 3 | 1-naphthalene | 4-NO2 | Br | 3c | 92 |
| 4 | 2,4-difluoro-C6H3 | 4-NO2 | Br | 3d | 90 |
| 5 | 2,4-difluoro-C6H3 | 4-OH | Br | 3e | 91 |
| 6 | 2,4-(CF3)2-C6H3 | 4-NO2 | Br | 3f | 90 |
| 7 | Ph | 3-Me | I | 3g | 95 |
| 8 | Ph | Ph | I | 3h | 98 |
| 9 | 1-naphthalene | 5-indole | Br | 3i | 90 |
| 10 | Ph | 2-phenylpropan-2-ol | Br | 3j | 93 |
| 11 | Ph | 4-OMe | Br | 3k | 96 |
| 12 | Ph | 2-Me | I | 3l | 97 |
| 13 | Ph | 4-Me | I | 3m | 96 |
| 14 | Ph | 2-CHO | Br | 3n | 90 |
| 15c | 3-CH3O-C6H4 | 4-OH | Br | 3o | 95 |
| 16 | 2,4-(CF3)2-C6H3 | 4-OH | Br | 3p | 91 |
| 17 | Ph | 2-Py | I | 3q | 92 |
| 18 | 4-Cl-C6H4 | 4-OMe | Br | 3r | 95 |
| 19 | Ph | 4-NO2 | Cl | 3s | 54 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shinde, V.V.; Jeong, D.; Jung, S. An Amino-Chain Modified β-cyclodextrin: A Supramolecular Ligand for Pd(OAc)2 Acceleration in Suzuki–Miyaura Coupling Reactions in Water. Catalysts 2019, 9, 111. https://doi.org/10.3390/catal9020111
Shinde VV, Jeong D, Jung S. An Amino-Chain Modified β-cyclodextrin: A Supramolecular Ligand for Pd(OAc)2 Acceleration in Suzuki–Miyaura Coupling Reactions in Water. Catalysts. 2019; 9(2):111. https://doi.org/10.3390/catal9020111
Chicago/Turabian StyleShinde, Vijay Vilas, Daham Jeong, and Seunho Jung. 2019. "An Amino-Chain Modified β-cyclodextrin: A Supramolecular Ligand for Pd(OAc)2 Acceleration in Suzuki–Miyaura Coupling Reactions in Water" Catalysts 9, no. 2: 111. https://doi.org/10.3390/catal9020111
APA StyleShinde, V. V., Jeong, D., & Jung, S. (2019). An Amino-Chain Modified β-cyclodextrin: A Supramolecular Ligand for Pd(OAc)2 Acceleration in Suzuki–Miyaura Coupling Reactions in Water. Catalysts, 9(2), 111. https://doi.org/10.3390/catal9020111