Bifunctional Electrocatalyst of Low-Symmetry Mesoporous Titanium Dioxide Modified with Cobalt Oxide for Oxygen Evolution and Reduction Reactions
Abstract
1. Introduction
2. Results and Discussion
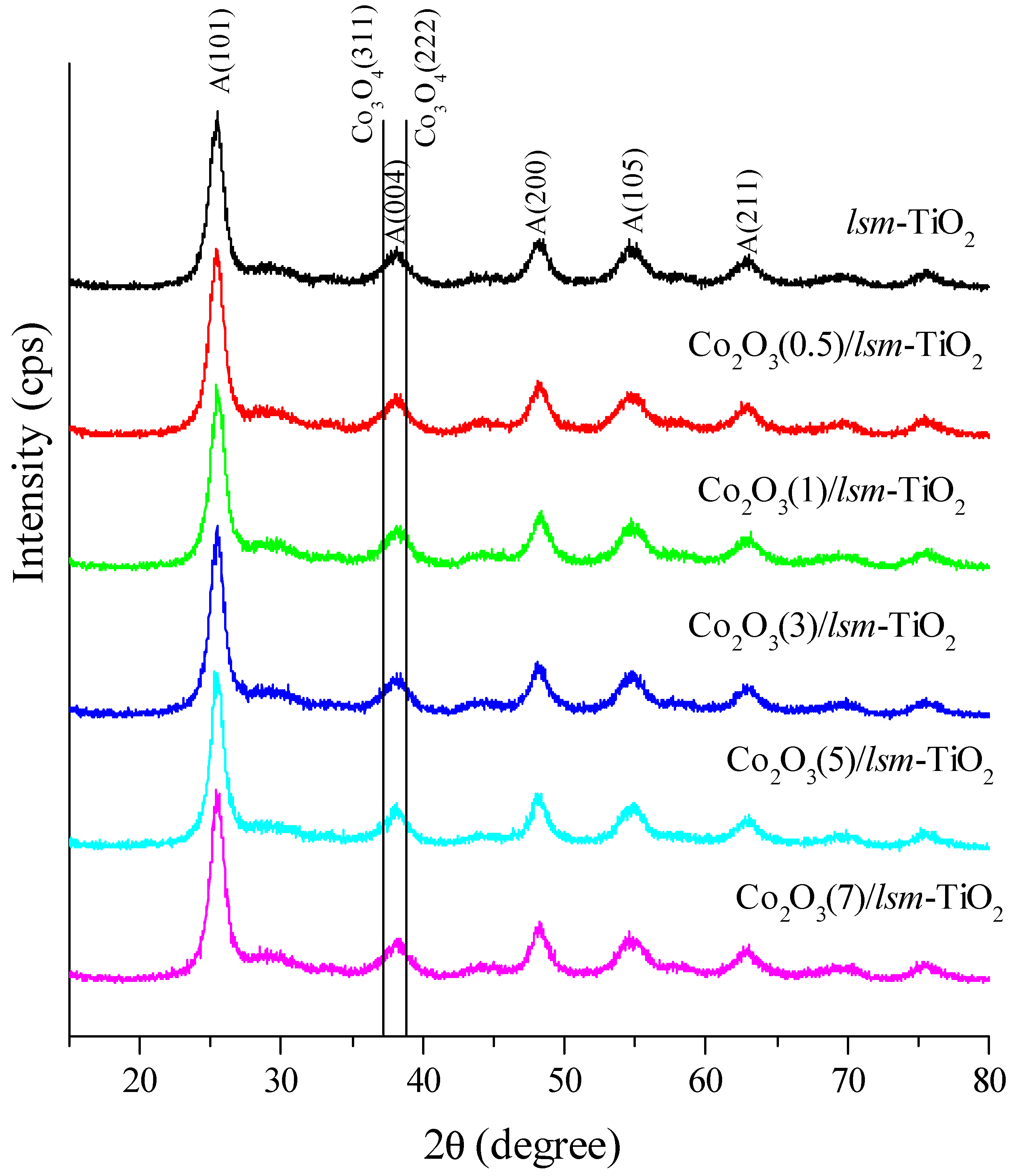
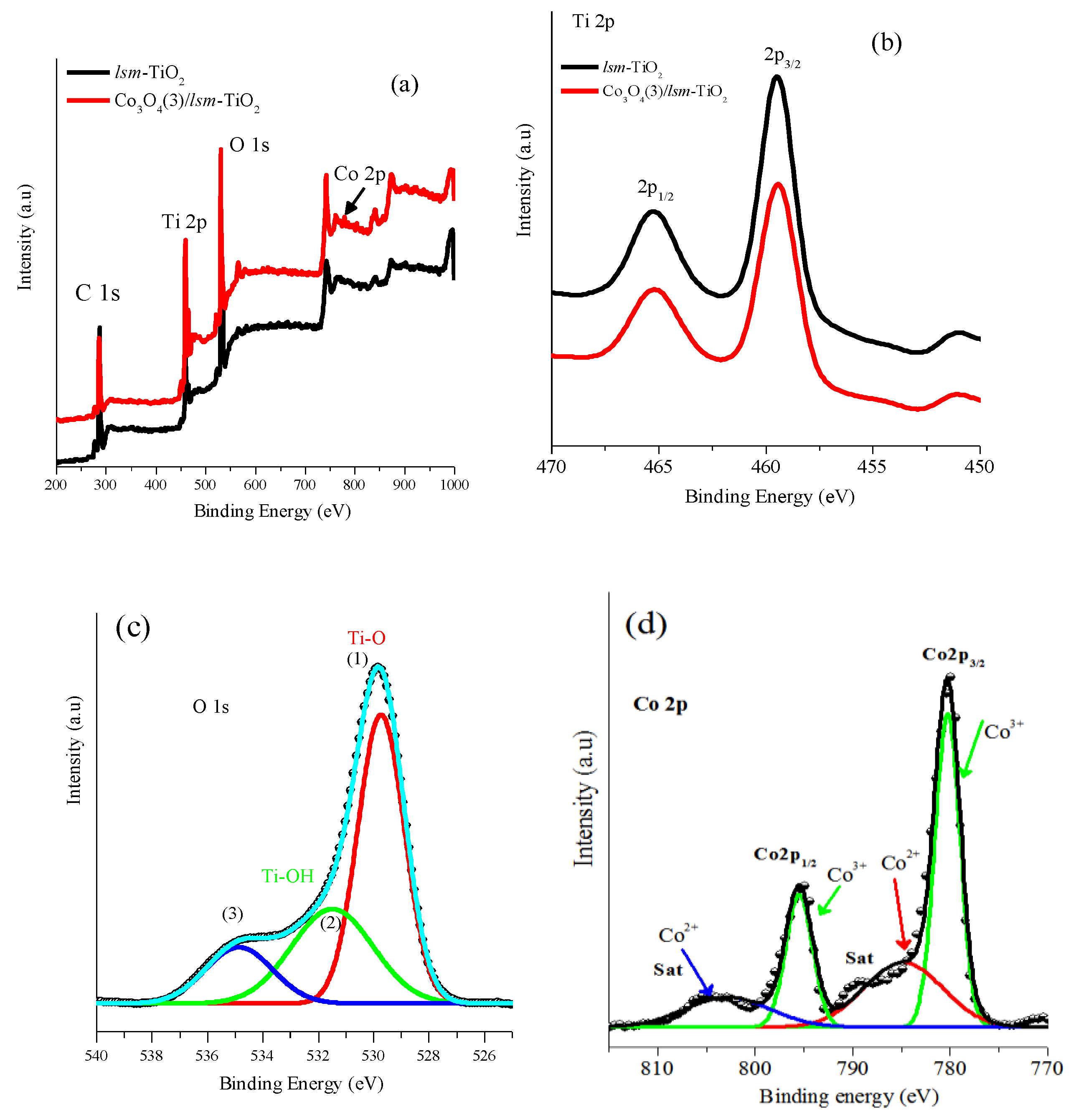
2.1. Morphological and Physicochemical Characterization of the Cobalt Oxide-Modified Mesoporous TiO2
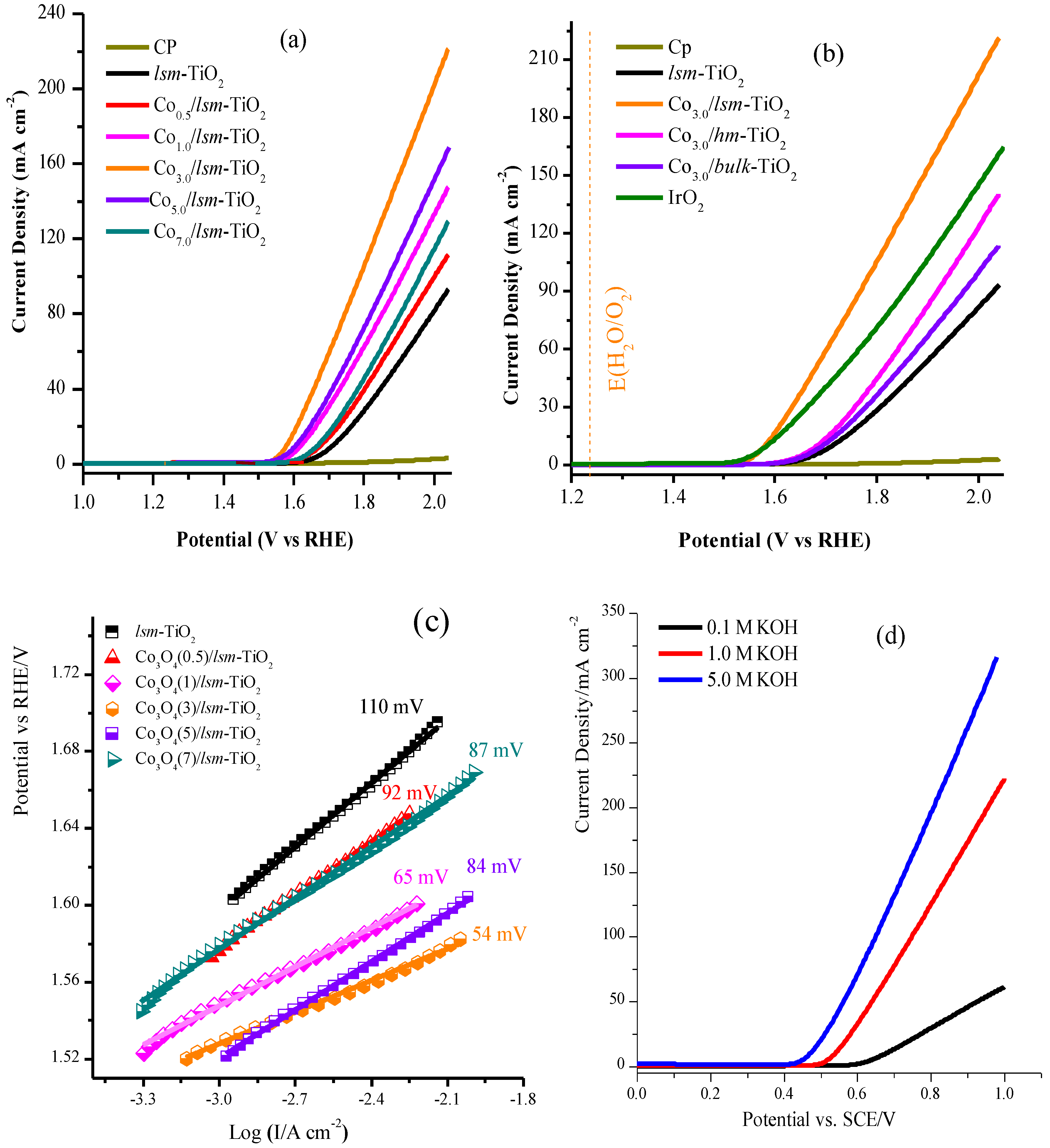
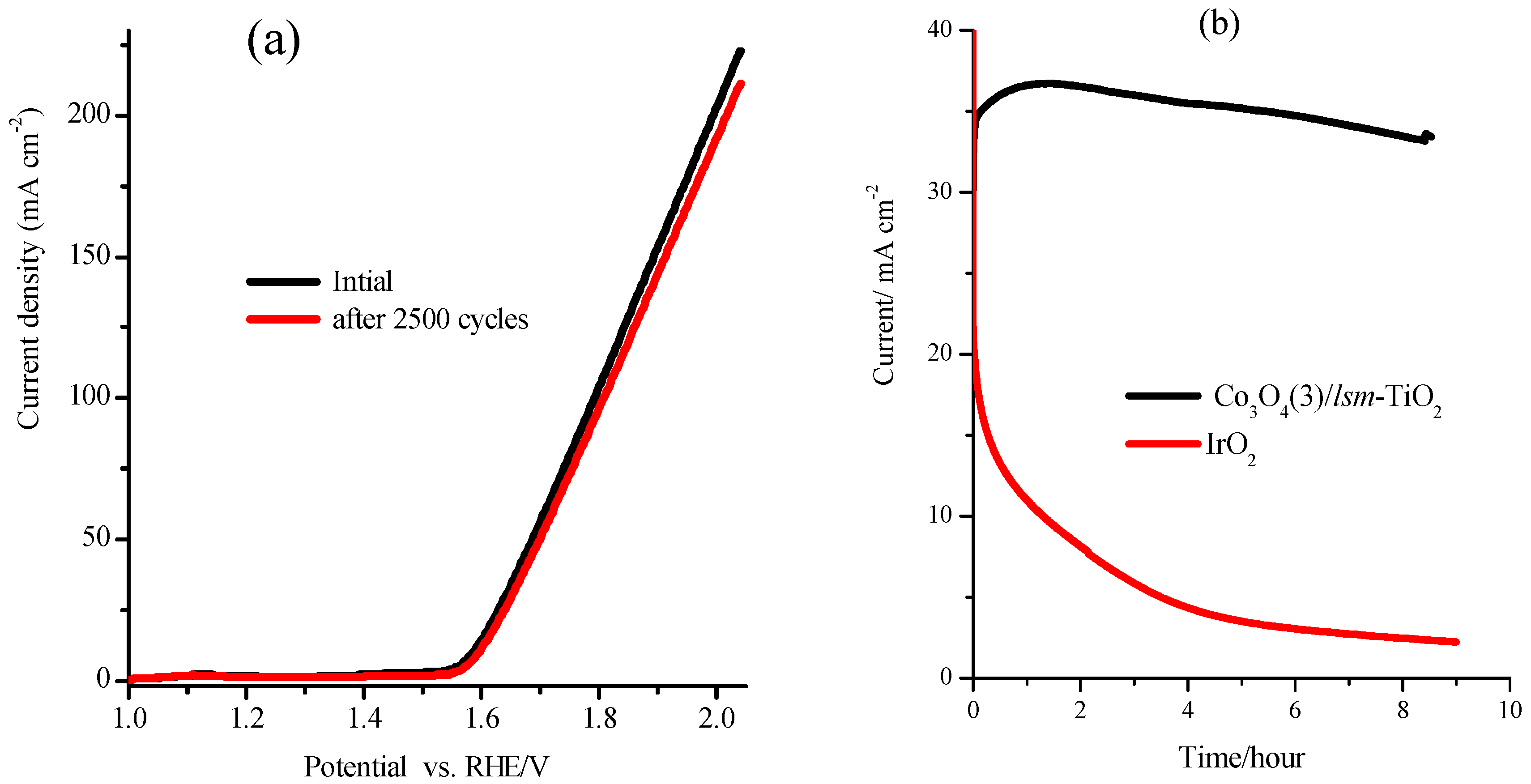
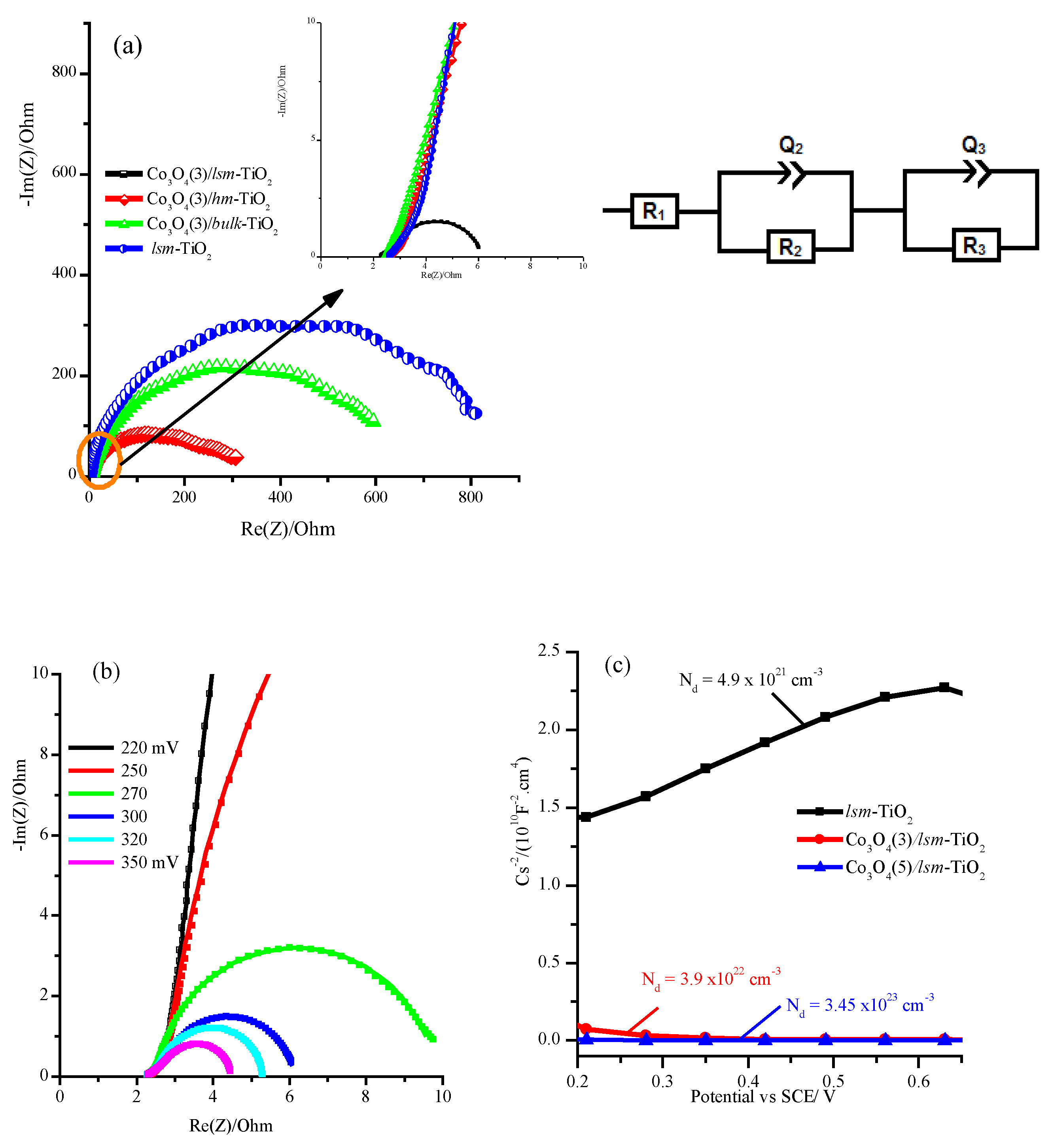
2.2. Electrochemical Performance of the Mesoporous TiO2 Modified with Cobalt Oxide Nanoparticles for OER
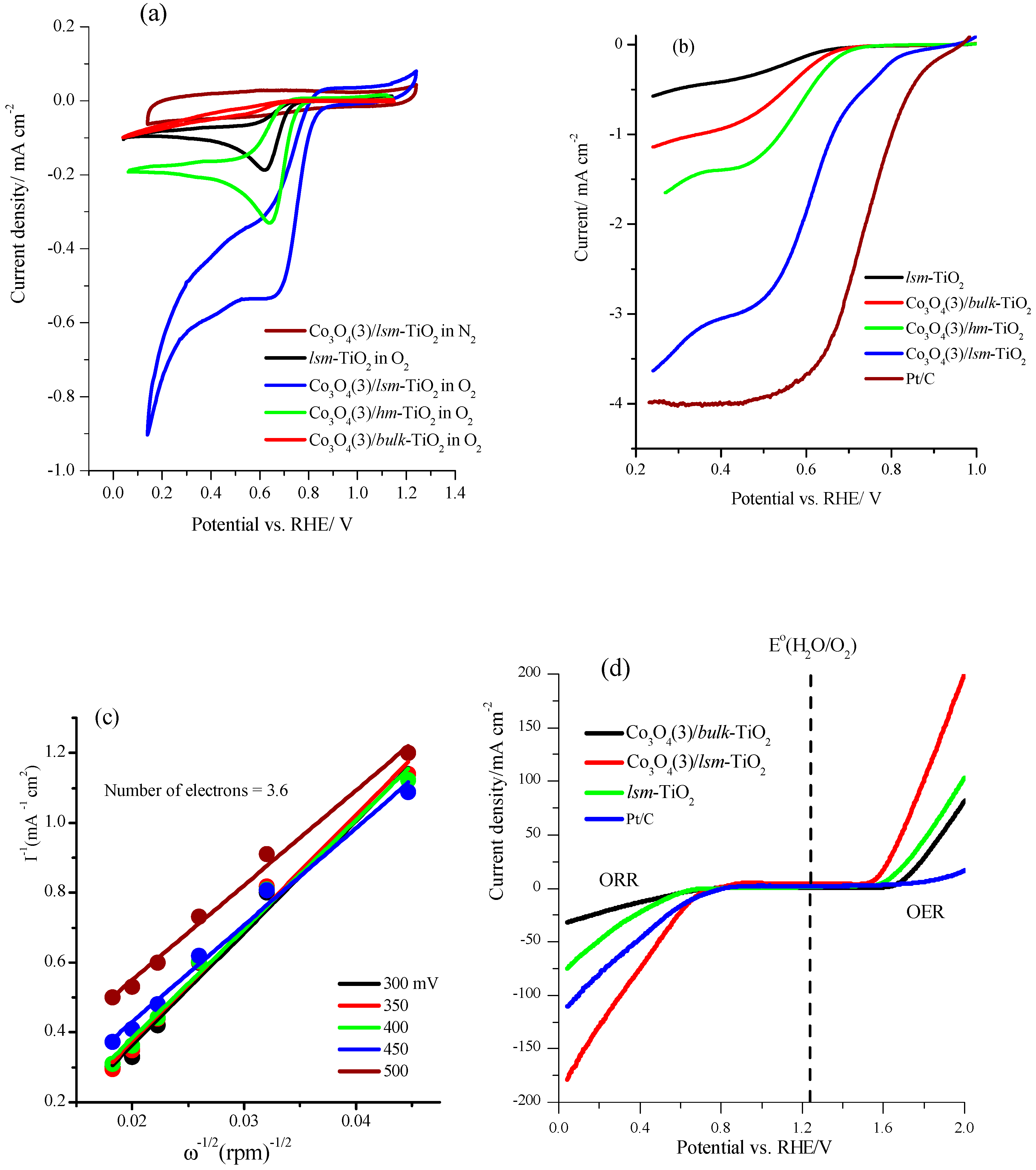
2.3. Electrochemical Performance of the Mesoporous TiO2 Modified with Cobalt Oxide Nanoparticles for ORR
3. Materials and Methods
3.1. Materials
3.2. Synthesis of Cobalt Oxide/Mesoporous TiO2 Materials
3.3. Material Characterization
3.4. Electrochemical Measurements
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hong, W.T.; Risch, M.; Stoerzinger, K.A.; Grimaud, A.; Suntivich, J.; Shao-Horn, Y. Toward the rational design of non-precious transition metal oxides for oxygen electrocatalysis. Energy Environ. Sci. 2015, 8, 1404–1427. [Google Scholar] [CrossRef]
- Qian, Y.; Hu, Z.; Ge, X.; Yang, S.; Peng, Y.; Kang, Z.; Liu, Z.; Lee, J.Y.; Zhao, N.Y. A metal-free ORR/OER bifunctional electrocatalyst derived from metal-organic frameworks for rechargeable Zn-Air batteries. Carbon 2017, 111, 641–650. [Google Scholar] [CrossRef]
- Girishkumar, G.; McCloskey, B.; Luntz, A.C.; Swanson, S.; Wilcke, W. Lithium−air battery: Promise and challenges. J. Phys. Chem. Lett. 2010, 1, 2193–2203. [Google Scholar] [CrossRef]
- Stambouli, A.B.; Traversa, E. Solid oxide fuel cells (SOFCs): A review of an environmentally clean and efficient source of energy, Renewable Sustainable. Energy Rev. 2002, 6, 433–455. [Google Scholar] [CrossRef]
- Sivanantham, A.; Ganesan, P.; Shanmugam, S. Hierarchical NiCo2S4 Nanowire Arrays Supported on Ni Foam: An Efficient and Durable Bifunctional Electrocatalyst for Oxygen and Hydrogen Evolution Reactions. Adv. Funct. Mater. 2016, 26, 4661–4672. [Google Scholar] [CrossRef]
- Liu, Q.; Xie, L.; Liu, Z.; Du, G.; Asiri, A.M.; Sun, X. Zn-doped Ni3S2 nanosheets array as a high-performance electrochemical water oxidation catalyst in alkaline solution. Chem. Commun. 2017, 53, 12446–12449. [Google Scholar] [CrossRef]
- Grimaud, A.; Diaz-Morales, O.; Han, B.; Hong, W.T.; Lee, Y.L.; Giordano, L.; Stoerzinger, K.A.; Koper, M.T.M.; Shao-Horn, Y. Activating lattice oxygen redox reactions in metal oxides to catalyse oxygen evolution. Nat. Chem. 2017, 9, 457–465. [Google Scholar] [CrossRef]
- Yin, Q.; Tan, J.M.; Besson, C.; Geletii, Y.V.; Musaev, D.G.; Kuznetsov, A.E.; Luo, Z.; Hardcastle, K.I.; Hill, C.L. A fast soluble carbon-free molecular water oxidation catalyst based on abundant metals. Science 2010, 328, 342–345. [Google Scholar] [CrossRef]
- Liu, Y.; Wang, H.; Lin, D.; Liu, C.; Hsu, P.C.; Liu, W.; Chen, W.; Cui, Y. Electrochemical tuning of olivine-type lithium transition-metal phosphates as efficient water oxidation catalysts. Energy Environ. Sci. 2015, 8, 1719–1724. [Google Scholar] [CrossRef]
- McCrory, C.C.L.; Jung, S.; Ferrer, I.M.; Chatman, S.M.; Peters, J.C.; Jaramillo, T.F. Benchmarking hydrogen evolving reaction and oxygen evolving reaction electrocatalysts for solar water splitting devices. J. Am. Chem. Soc. 2015, 137, 4347–4357. [Google Scholar] [CrossRef]
- Caro, C.; Thirunavukkarasu, K.; Anilkumar, M.; Shiju, N.R.; Rothenberg, G. Selective Autooxidation of Ethanol over Titania-Supported Molybdenum Oxide Catalysts: Structure and Reactivity. Adv. Synth. Catal. 2012, 354, 1327–1336. [Google Scholar] [CrossRef] [PubMed]
- Caro, C.; Gámez, F.; Sayagues, M.J.; Polvillo, R.; Royo, J.L. AgACTiO2 nanoparticles with microbiocide properties under visible light. Mater. Res. Express 2015, 2, 055002. [Google Scholar] [CrossRef]
- Zhang, W.; Innocenti, G.; Ferbinteanu, M.; Ramos-Fernandez, E.V.; Sepulveda-Escribano, A.; Wu, H.; Cavani, F.; Rothenberg, G.; Shiju, N.R. Understanding the oxidative dehydrogenation of ethyl lactate to ethyl pyruvate over vanadia/titania. Catal. Sci. Technol. 2018, 8, 3737–3747. [Google Scholar] [CrossRef]
- Lee, Y.; Suntivich, J.; May, K.J.; Perry, E.E.; Shao-Horn, Y. Synthesis and activities of rutile IrO2 and RuO2 nanoparticles for oxygen evolution in acid and alkaline solutions. J. Phys. Chem. Lett. 2012, 3, 399–404. [Google Scholar] [CrossRef] [PubMed]
- Ghanem, M.A.; Arunachalam, P.; Almayouf, A.; Weller, M.T. Efficient Bi-Functional Electrocatalysts of Strontium Iron Oxy-Halides for Oxygen Evolution and Reduction Reactions in Alkaline Media. J. Electrochem. Soc. 2016, 163, H450–H458. [Google Scholar] [CrossRef]
- Deng, X.; Tüysüz, H. Cobalt-Oxide-Based Materials as Water Oxidation Catalyst: Recent Progress and Challenges. ACS Catal. 2014, 4, 3701–3714. [Google Scholar] [CrossRef]
- Zhang, G.; Xia, B.Y.; Wang, X.; Lou, X.W. Strongly coupled NiCo2O4-rGO hybrid nanosheets as a methanol-tolerant electrocatalyst for the oxygen reduction reaction. Adv. Mater. 2014, 26, 2408–2412. [Google Scholar] [CrossRef]
- Wang, Y.; Zhang, Y.; Liu, Z.; Xie, C.; Feng, S.; Liu, D.; Shao, M.; Wang, S. Layered double hydroxide nanosheets with multiple vacancies obtained by dry exfoliation as highly efficient oxygen evolution electrocatalysts. Angew. Chem. Int. Ed. 2017, 56, 5867–5871. [Google Scholar] [CrossRef]
- Duarte, M.F.P.; Rocha, I.M.; Figueiredo, J.L.; Freire, C.; Pereira, M.F.R. CoMn-LDH@carbon nanotube composites: Bifunctional electrocatalysts for oxygen reactions. Catal. Today 2018, 301, 17–24. [Google Scholar] [CrossRef]
- Wang, Y.; Hu, T.; Liu, Q.; Zhang, L. CoMn2O4 embedded in MnOOH nanorods as a bifunctional catalyst for oxygen reduction and oxygen evolution reactions. Chem. Commun. 2018, 54, 4005–4008. [Google Scholar] [CrossRef]
- Liang, Y.; Li, Y.; Wang, H.; Zhou, J.; Wang, J.; Regier, T.; Dai, H. Co3O4 nanocrystals on graphene as a synergistic catalyst for oxygen reduction reaction. Nat. Mater. 2011, 10, 780–786. [Google Scholar] [CrossRef] [PubMed]
- Oh, T.; Kim, K.; Kim, J. Controllable active sites and facile synthesis of cobalt nanoparticle embedded in nitrogen and sulfur co-doped carbon nanotubes as efficient bifunctional electrocatalysts for oxygen reduction and evolution reactions. J. Energy Chem. 2019, 38, 60–67. [Google Scholar] [CrossRef]
- Kuang, M.; Zheng, G. Nanostructured bifunctional redox electrocatalysts. Small 2016, 12, 5656–5675. [Google Scholar] [CrossRef] [PubMed]
- Huang, Z.F.; Wang, J.; Peng, Y.; Jung, C.Y.; Fisher, A.; Wang, X. Design of efficient bifunctional oxygen reduction/evolution electrocatalyst: Recent advances and perspectives. Adv. Energy Mater. 2017, 7, 1700544. [Google Scholar] [CrossRef]
- Shang, L.; Yu, H.; Huang, X.; Bian, T.; Shi, R.; Zhao, Y.; Waterhouse, G.I.N.; Wu, L.Z.; Tung, C.H.; Zhang, T. Well-dispersed ZIF-derived Co, N-Co-doped carbon nanoframes through mesoporous-silica-protected calcination as efficient oxygen reduction electrocatalysts. Adv. Mater. 2016, 28, 1668–1674. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; Liu, Y.; Sun, Y. Mesoporous Colloidal Superparticles of Platinum-Group Nanocrystals with Surfactant-Free Surfaces and Enhanced Heterogeneous Catalysis. Adv. Funct. Mater. 2015, 25, 1638–1647. [Google Scholar] [CrossRef]
- Wu, Z.; Zhao, D. Ordered mesoporous materials as adsorbents. Chem. Commun. 2011, 47, 3332–3338. [Google Scholar] [CrossRef]
- Knossalla, J.; Mezzavilla, S.; Schüth, F. Continuous synthesis of nanostructured silica-based materials in a gas-liquid segmented flow tubular reactor. New J. Chem. 2016, 40, 4361–4366. [Google Scholar] [CrossRef]
- Song, W.; Ren, Z.; Chen, S.Y.; Meng, Y.; Biswas, S.; Nandi, P.; Elsen, H.A.; Gao, P.X.; Suib, S.L. Ni- and Mn-promoted mesoporous Co3O4: A stable bifunctional catalyst with surface-structure-dependent activity for oxygen reduction reaction and oxygen evolution reaction. ACS Appl. Mater. Interfaces 2016, 8, 20802–20813. [Google Scholar] [CrossRef]
- Sa, Y.J.; Kwon, K.; Cheon, J.Y.; Kleitz, F.; Joo, S.H. Ordered mesoporous Co3O4 spinels as stable, bifunctional, noble metal-free oxygen electrocatalysts. J. Mater. Chem. A 2013, 1, 9992–10001. [Google Scholar] [CrossRef]
- Amer, M.S.; Ghanem, M.A.; Al-Mayouf, A.M.; Arunachalam, P.; Khdary, N. Low-loading of oxidized platinum nanoparticles into mesoporous titanium dioxide for effective and durable hydrogen evolution in acidic media. Arab. J. Chem. 2018. [Google Scholar] [CrossRef]
- Mosa, I.M.; Biswas, S.; El-Sawy, A.M.; Botu, V.; Guild, C.; Song, W.; Ramprasad, R.; Rusling, J.F.; Suib, S.L. Tunable mesoporous manganese oxide for high-performance oxygen reduction and evolution reactions. J. Mater. Chem. A 2015, 4, 620–631. [Google Scholar] [CrossRef]
- Zhou, W.; Li, W.; Wang, J.Q.; Qu, Y.; Yang, Y.; Xie, Y.; Zhang, K.; Wang, L.; Fu, H.; Zhao, D. Ordered mesoporous black TiO2 as highly efficient hydrogen evolution photocatalyst. J. Am. Chem. Soc. 2014, 136, 9280–9283. [Google Scholar] [CrossRef] [PubMed]
- Hartmann, P.; Lee, D.K.; Smarsly, B.M.; Janek, J. Mesoporous TiO2: Comparison of classical sol−gel and nanoparticle-based photoelectrodes for the water splitting reaction. ACS Nano 2010, 4, 3147–3154. [Google Scholar] [CrossRef] [PubMed]
- Amer, M.S.; Ghanem, M.A.; Al-Mayouf, A.M.; Arunachalam, P. Low-symmetry mesoporous titanium dioxide (lsm-TiO2) electrocatalyst for efficient and durable oxygen evolution in aqueous alkali. J. Electrochem. Soc. 2018, 165, H300–H309. [Google Scholar] [CrossRef]
- Ghanem, M.A.; Arunachalam, P.; Amer, M.S.; Al-Mayouf, A.M. Mesoporous titanium dioxide photoanodes decorated with gold nanoparticles for boosting the photoelectrochemical alkali water oxidation. Mater. Chem. Phys. 2018, 213, 56–66. [Google Scholar] [CrossRef]
- Liu, B.; Chen, H.M.; Liu, C.; Andrews, S.C.; Hahn, C.; Yang, P. Large-scale synthesis of transition-metal-doped TiO2 nanowires with controllable overpotential. J. Am. Chem. Soc. 2013, 135, 9995–9998. [Google Scholar] [CrossRef] [PubMed]
- Jang, D.M.; Kwak, I.H.; Kwon, E.L.; Jung, C.S.; Im, H.S.; Park, K.; Park, J. Transition-metal doping of oxide nanocrystals for enhanced catalytic oxygen evolution. J. Phys. Chem. C 2015, 119, 1921–1927. [Google Scholar] [CrossRef]
- Cai, L.; Cho, I.S.; Logar, M.; Mehta, A.; He, J.; Lee, C.H.; Rao, P.M.; Feng, Y.; Wilcox, J.; Prinz, F.B.; et al. Sol-flame synthesis of cobalt-doped TiO2 nanowires with enhanced electrocatalytic activity for oxygen evolution reaction. Phys. Chem. Chem. Phys. 2014, 16, 12299–12306. [Google Scholar] [CrossRef]
- Roy, N.; Sohn, Y.; Leung, K.T.; Pradhan, D. Engineered electronic states of transition metal doped TiO2 nanocrystals for low overpotential oxygen evolution reaction. J. Phys. Chem. C 2014, 118, 29499–29506. [Google Scholar] [CrossRef]
- García-Mota, M.; Vojvodic, A.; Metiu, H.; Man, I.C.; Su, H.Y.; Rossmeisl, J.; Nørskov, J.K. Tailoring the activity for oxygen evolution electrocatalysis on rutile TiO2(110) by transition-metal substitution. ChemCatChem 2011, 3, 1607–1611. [Google Scholar] [CrossRef]
- Han, L.N.; Lv, L.B.; Zhu, Q.C.; Wei, X.; Li, X.H.; Chen, J.S. Ultra-durable two-electrode Zn-air secondary batteries based on bifunctional titania nanocatalysts: Co2+ dopant boosts the electrochemical activity. J. Mater. Chem. A 2016, 4, 7841–7847. [Google Scholar] [CrossRef]
- Yang, Y.; Kao, L.C.; Liu, Y.; Sun, K.; Yu, H.; Guo, J.; Liou, S.Y.H.; Hoffmann, M.R. Modification of mesoporous titanium dioxide with cobalt oxide electrocatalyst for enhanced oxygen evolution reaction. ACS Catal. 2018, 8, 4278–4287. [Google Scholar] [CrossRef] [PubMed]
- Wu, Q.L.; Rankin, S.E. Tuning the mesopore size of titania thin films using a polymeric swelling agent. J. Phys. Chem. C 2011, 115, 11925–11933. [Google Scholar] [CrossRef]
- Crepaldi, E.L.; Soler-Illia, G.J.D.A.; Grosso, D.; Cagnol, F.; Ribot, F.; Sanchez, C. Controlled formation of highly organized mesoporous titania thin films: From mesostructured hybrids to mesoporous nanoanatase TiO2. J. Am. Chem. Soc. 2003, 125, 9770–9786. [Google Scholar] [CrossRef] [PubMed]
- Alberius, P.C.; Frindell, K.L.; Hayward, R.C.; Kramer, E.J.; Stucky, G.D.; Chmelka, B.F. General predictive syntheses of cubic, hexagonal, and lamellar silica and titania mesostructured thin films. Chem. Mater. 2002, 14, 3284–3294. [Google Scholar] [CrossRef]
- Wang, T.; Meng, X.; Liu, G.; Chang, K.; Li, P.; Kang, Q.; Liu, L.; Li, M.; Ouyang, S.; Ye, J. In situ synthesis of ordered mesoporous Co-doped TiO2 and its enhanced photocatalytic activity and selectivity for the reduction of CO2. J. Mater. Chem. A 2015, 3, 9491–9501. [Google Scholar] [CrossRef]
- Papageorgiou, A.C.; Cabailh, G.; Chen, Q.; Resta, A.; Lundgren, E.; Andersen, J.N.; Thornton, G. Growth and reactivity of titanium oxide ultrathin films on Ni(110). J. Phys. Chem. C 2007, 111, 7704–7710. [Google Scholar] [CrossRef]
- Wang, G.M.; Wang, H.; Ling, Y.; Tang, Y.; Yang, X.; Fitzmorris, R.C.; Wang, C.; Zhang, J.Z.; Li, Y. Hydrogen-Treated TiO2 Nanowire Arrays for Photoelectrochemical Water Splitting. Nano Lett. 2011, 11, 3026–3033. [Google Scholar] [CrossRef]
- Liu, D.; Zhang, Y.; Xiao, P.; Garcia, B.B.; Zhang, Q.; Zhou, X.; Cao, G. TiO2 nanotube arrays annealed in CO exhibiting high performance for lithium ion intercalation. Tech. Proc. 2009, 3, 50–57. [Google Scholar] [CrossRef]
- Chen, C.J.; Wen, Y.W.; Hu, X.L.; Ji, X.L.; Yan, M.Y.; Mai, L.Q.; Hu, P.; Shan, B.; Huang, Y.H. Na+ intercalation pseudocapacitance in graphene-coupled titanium oxide enabling ultra-fast sodium storage and long-term cycling. Nat. Commun. 2015, 6, 69297. [Google Scholar] [CrossRef] [PubMed]
- Lazarus, M.S.; Sham, T.K. X-ray photoelectron spectroscopy (XPS) studies of hydrogen reduced rutile (TiO2-x) surfaces. Chem. Phys. Lett. 1982, 92, 670–674. [Google Scholar] [CrossRef]
- McCafferty, E.; Wightman, J.P. Determination of the concentration of surface hydroxyl groups on metal oxide films by a quantitative XPS method. Surf. Interface Anal. 1998, 26, 549–564. [Google Scholar] [CrossRef]
- Feng, D.; Gao, T.N.; Fan, M.; Li, A.; Li, K.; Wang, T.; Huo, Q.; Qiao, Z.A. A general ligand-assisted self-assembly approach to crystalline mesoporous metal oxides. NPG Asia Mater. 2018, 10, 800–809. [Google Scholar] [CrossRef]
- Kruk, M.; Jaroniec, M.; Guan, S.; Inagaki, S. Gas Adsorption Characterization of Ordered Organic−Inorganic Nanocomposite Materials. Chem. Mater. 2001, 13, 3169–3183. [Google Scholar] [CrossRef]
- Sing, K.S.W.; Everet, D.H.; Haul, R.A.W. Reporting physisorption data for gas-solid systems with special reference to the determination of surface-area and porosity. Pure Appl. Chem. 1985, 57, 603–619. [Google Scholar] [CrossRef]
- Lu, W.; Liu, T.; Xie, L.; Tang, C.; Liu, D.; Hao, S.; Qu, F.; Du, G.; Ma, Y.; Asiri, A.M.; et al. In situ derived Co-B nanoarray: A high-efficiency and durable 3d bifunctional electrocatalyst for overall alkaline water splitting. Small 2017, 13, 1700805. [Google Scholar] [CrossRef]
- Gao, M.; Sheng, W.; Zhuang, Z.; Fang, Q.; Gu, S.; Jiang, J.; Yan, Y. Efficient water oxidation using nanostructured α-nickel-hydroxide as an electrocatalyst. J. Am. Chem. Soc. 2014, 136, 7077–7084. [Google Scholar] [CrossRef]
- Thenuwara, A.C.; Shumlas, S.L.; Attanayake, N.H.; Aulin, Y.V.; McKendry, I.G.; Qiao, Q.; Zhu, Y.; Borguet, E.; Zdilla, M.J.; Strongin, D.R. Intercalation of cobalt into the interlayer of birnessite improves oxygen evolution catalysis. ACS Catal. 2016, 6, 7739–7743. [Google Scholar] [CrossRef]
- Zhou, T.; Cao, Z.; Zhang, P.; Ma, H.; Gao, Z.; Wang, H.; Lu, Y.; He, J.; Zhao, Y. Transition metal ions regulated oxygen evolution reaction performance of Ni-based hydroxides hierarchical nanoarrays. Sci. Rep. 2017, 7, 46154. [Google Scholar] [CrossRef]
- Wang, X.; Zhuang, L.; He, T.; Jia, Y.; Zhang, L.; Yan, X.; Gao, M.; Du, A.; Zhu, Z.; Yao, X.; et al. Grafting cobalt diselenide on defective graphene for enhanced oxygen evolution reaction. iScience 2018, 7, 145–153. [Google Scholar] [CrossRef] [PubMed]
- Hall, D.E. Alkaline Water Electrolysis Anode Materials. J. Electrochem. Soc. 1985, 132, 41C–48C. [Google Scholar] [CrossRef]
- Wang, Y.; Zhou, T.; Jiang, K.; Da, P.; Peng, Z.; Tang, J.; Kong, B.; Cai, W.B.; Yang, Z.; Zheng, G. Reduced mesoporous Co3O4 nanowires as efficient water oxidation electrocatalysts and supercapacitor electrodes. Adv. Energy Mater. 2014, 4, 1400696. [Google Scholar] [CrossRef]
- Schlesinger, M.; Lasia, A. Modeling of Impedance of Porous Electrodes, Modeling and Numerical Simulations; Springer: New York, NY, USA, 2009; pp. 67–137. [Google Scholar]
- Bao, J.; Zhang, X.; Fan, B.; Zhang, J.; Zhou, M.; Yang, W.; Hu, X.; Wang, H.; Pan, B.; Xie, Y. Ultrathin spinel-structured nanosheets rich in oxygen deficiencies for enhanced electrocatalytic water oxidation. Angew. Chem. 2015, 127, 7507–7512. [Google Scholar] [CrossRef]
- Aijaz, A.; Masa, J.; Rösler, C.; Xia, W.; Weide, P.; Botz, A.J.R.; Fischer, R.A.; Schuhmann, W.; Muhler, M. Co@Co3O4 encapsulated in carbon nanotube-grafted nitrogen-doped carbon polyhedra as an advanced bifunctional oxygen electrode. Angew. Chem. Int. Ed. 2016, 55, 4087–4091. [Google Scholar] [CrossRef] [PubMed]
- Tang, H.; Prasad, K.; Sanjinès, R.; Schmid, P.E.; Lévy, F. Electrical and optical properties of TiO2 anatase thin films. J. Appl. Phys. 1994, 75, 2042–2047. [Google Scholar] [CrossRef]
- Wang, Q.; Hu, W.; Huang, Y. Nitrogen-doped graphene anchored cobalt oxides efficiently bi-functionally catalyze both oxygen reduction reaction and oxygen revolution reaction. Int. J. Hydrog. Energy 2017, 42, 5899–5907. [Google Scholar] [CrossRef]
- Huang, Y.B.; Zhang, M.; Liu, P.; Cheng, F.L.; Wang, L.S. Co3O4 supported on N, P-doped carbon as a bifunctional electrocatalyst for oxygen reduction and evolution reactions. Chin. J. Catal. 2016, 37, 1249–1256. [Google Scholar] [CrossRef]
- Jung, J.I.; Risch, M.; Park, S.; Kim, M.G.; Nam, G.; Jeong, H.Y.; Shao-Horn, Y.; Cho, J. Optimizing nanoparticle perovskite for bifunctional oxygen electrocatalysis. Energy Environ. Sci. 2016, 9, 176–183. [Google Scholar] [CrossRef]
- Masa, J.; Xia, W.; Sinev, I.; Zhao, A.; Sun, Z.; Grützke, S.; Weide, P.; Muhler, M.; Schuhmann, W. MnxOy/NC and CoxOy/NC nanoparticles embedded in a nitrogen-doped carbon matrix for high-performance bifunctional oxygen electrodes. Angew. Chem. Int. Ed. 2014, 53, 8508–8512. [Google Scholar] [CrossRef]

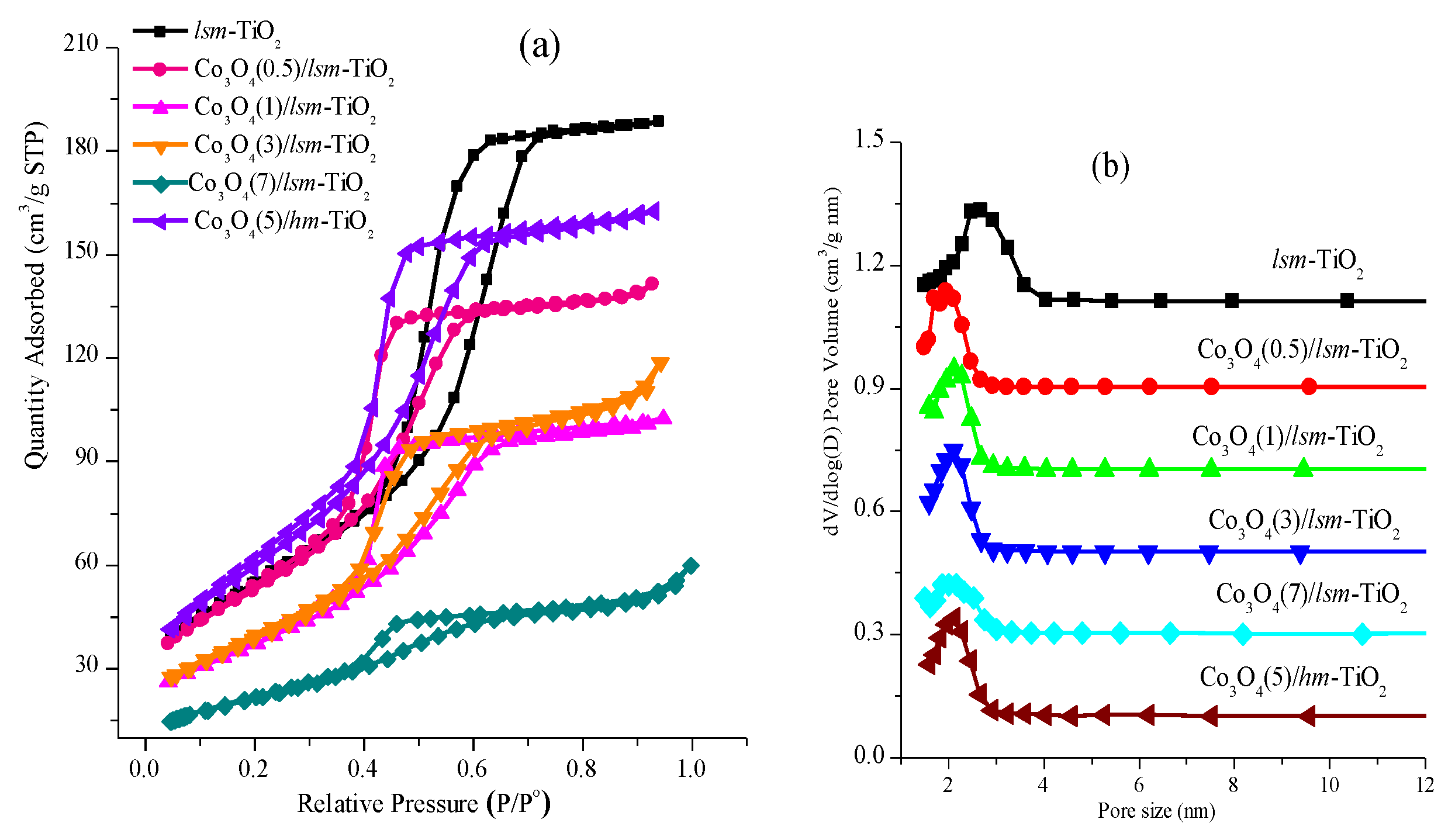
| Catalyst | BET Surface Area (m2 g−1) a | Pore Size (nm) b | Pore Volume (cm3 g−1) c |
|---|---|---|---|
| lsm-TiO2 | 200 | 2.7 | 0.290 |
| Co3O4(0.5)/lsm-TiO2 | 175 | 2.01 | 0.25 |
| Co3O4(1)/lsm-TiO2 | 158 | 2.03 | 0.17 |
| Co3O4(3)/lsm-TiO2 | 146 | 2.11 | 0.11 |
| Co3O4(5)/lsm-TiO2 | 119 | 1.95 | 0.10 |
| Co3O4(7)/lsm-TiO2 | 81 | 1.83 | 0.071 |
| Co3O4(3)/hm-TiO2 | 229 | 2.11 | 0.22 |
| Co3O4(3)/bulk-TiO2 | 42 | 1.50 | 0.025 |
| Catalysts | Onset Potential vs. RHE/V a | η (mV) at (10 mA/cm2) b | η (mV) at (20 mA/cm2) c | Current mA/cm2 at 2.0 V vs. RHE | Tafel Slope (mV/dec) d | mass Activity at 1.6 V (A g−1 ) e | TOF at η = 0.370 V (s−1 ) f |
|---|---|---|---|---|---|---|---|
| lsm-TiO2 | 1.620 | 490 | 530 | 94 | 98 | 2 | 0.00033 |
| Co3O4(0.5)/lsm-TiO2 | 1.510 | 449 | 492 | 113 | 92 | 5 | 0.0038 |
| Co3O4(1)/lsm-TiO2 | 1.490 | 390 | 432 | 149 | 65 | 15 | 0.012 |
| Co3O4(3)/lsm-TiO2 | 1.485 | 348 | 370 | 222 | 54 | 42 | 0.035 |
| Co3O4(5)/lsm-TiO2 | 1.490 | 370 | 410 | 170 | 84 | 23 | 0.020 |
| Co3O4(7)/lsm-TiO2 | 1.530 | 437 | 481 | 131 | 87 | 5 | 0.0037 |
| Co3O4(3)/hm-TiO2 | 1.58 | 452 | 490 | 141 | 121 | 3 | 0.0028 |
| Co3O4(3)/bulk-TiO2 | 1.590 | 470 | 510 | 114 | 91 | 3 | 0.0020 |
| IrO2 | 1.475 | 354 | 398 | 164 | 81 | 34 | 0.019 |
| Electrolyte (KOH) | η (mV) at 10 mA/cm2 | η (mV) at 20 mA/cm2 | I (mA/cm2) at 1.0 V | Tafel Slope mV/dec |
|---|---|---|---|---|
| 0.1 M | 445 | 512 | 61 | 87 |
| 1.0 M | 309 | 337 | 222 | 54 |
| 5.0 M | 243 | 272 | 318 | 71 |
| Catalyst | R1 (Ω) | Q2 (µF. sn−1) | R2 (Ω) | Q3 (µF. sn−1) | R3 (Ω) |
|---|---|---|---|---|---|
| lsm-TiO2 | 2.152 | 100 | 304 | 109 | 448 |
| Co3O4(3)/lsm-TiO2 | 2.260 | 10,140 | 7.10 | 9760 | 0.480 |
| Co3O4(3)/hm-TiO2 | 2.720 | 7170 | 184 | 3230 | 85 |
| Co3O4(3)/bulk-TiO2 | 2.253 | 3870 | 723 | 190 | 110.4 |
| Overpotential (η)/mV | R1 (Ω) | Q2 (µF. sn−1) | R2 (Ω) | Q3 (µF. sn−1) | R3 (Ω) |
|---|---|---|---|---|---|
| 220 | 2.26 | 9370 | 299 | 104,000 | 0.95 |
| 250 | 2.38 | 8890 | 53 | 4260 | 1.40 |
| 280 | 2.26 | 10,140 | 7.1 | 9760 | 0.48 |
| 300 | 2.41 | 12,650 | 3.90 | -- | 3.4 |
| 320 | 2.25 | 75,750 | 2.52 | 9806 | 2.52 |
| 350 | 2.27 | 33,120 | 0.59 | 9840 | 1.69 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Amer, M.S.; Ghanem, M.A.; Arunachalam, P.; Al-Mayouf, A.M.; Hadadi, S.M. Bifunctional Electrocatalyst of Low-Symmetry Mesoporous Titanium Dioxide Modified with Cobalt Oxide for Oxygen Evolution and Reduction Reactions. Catalysts 2019, 9, 836. https://doi.org/10.3390/catal9100836
Amer MS, Ghanem MA, Arunachalam P, Al-Mayouf AM, Hadadi SM. Bifunctional Electrocatalyst of Low-Symmetry Mesoporous Titanium Dioxide Modified with Cobalt Oxide for Oxygen Evolution and Reduction Reactions. Catalysts. 2019; 9(10):836. https://doi.org/10.3390/catal9100836
Chicago/Turabian StyleAmer, Mabrook S., Mohamed A. Ghanem, Prabhakarn Arunachalam, Abdullah M. Al-Mayouf, and Sultan M. Hadadi. 2019. "Bifunctional Electrocatalyst of Low-Symmetry Mesoporous Titanium Dioxide Modified with Cobalt Oxide for Oxygen Evolution and Reduction Reactions" Catalysts 9, no. 10: 836. https://doi.org/10.3390/catal9100836
APA StyleAmer, M. S., Ghanem, M. A., Arunachalam, P., Al-Mayouf, A. M., & Hadadi, S. M. (2019). Bifunctional Electrocatalyst of Low-Symmetry Mesoporous Titanium Dioxide Modified with Cobalt Oxide for Oxygen Evolution and Reduction Reactions. Catalysts, 9(10), 836. https://doi.org/10.3390/catal9100836









