Synthesis, Performance and Emission Quality Assessment of Ecodiesel from Castor Oil in Diesel/Biofuel/Alcohol Triple Blends in a Diesel Engine
Abstract
1. Introduction
2. Results
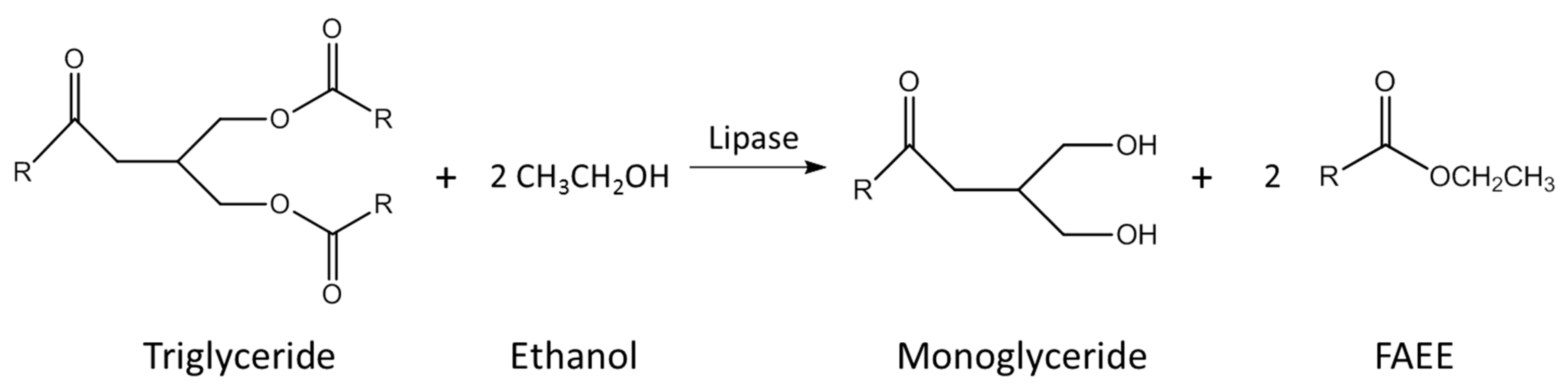
2.1. Synthesis of Ecodiesel by Kinetic Control of the Conventional Procedure of Biodiesel Production
2.2. Characterization of Biofuel Mixtures, with Fossil Diesel and Alcohols, to Allow Their Use in Conventional Compression Ignition Engines, without Any Modification
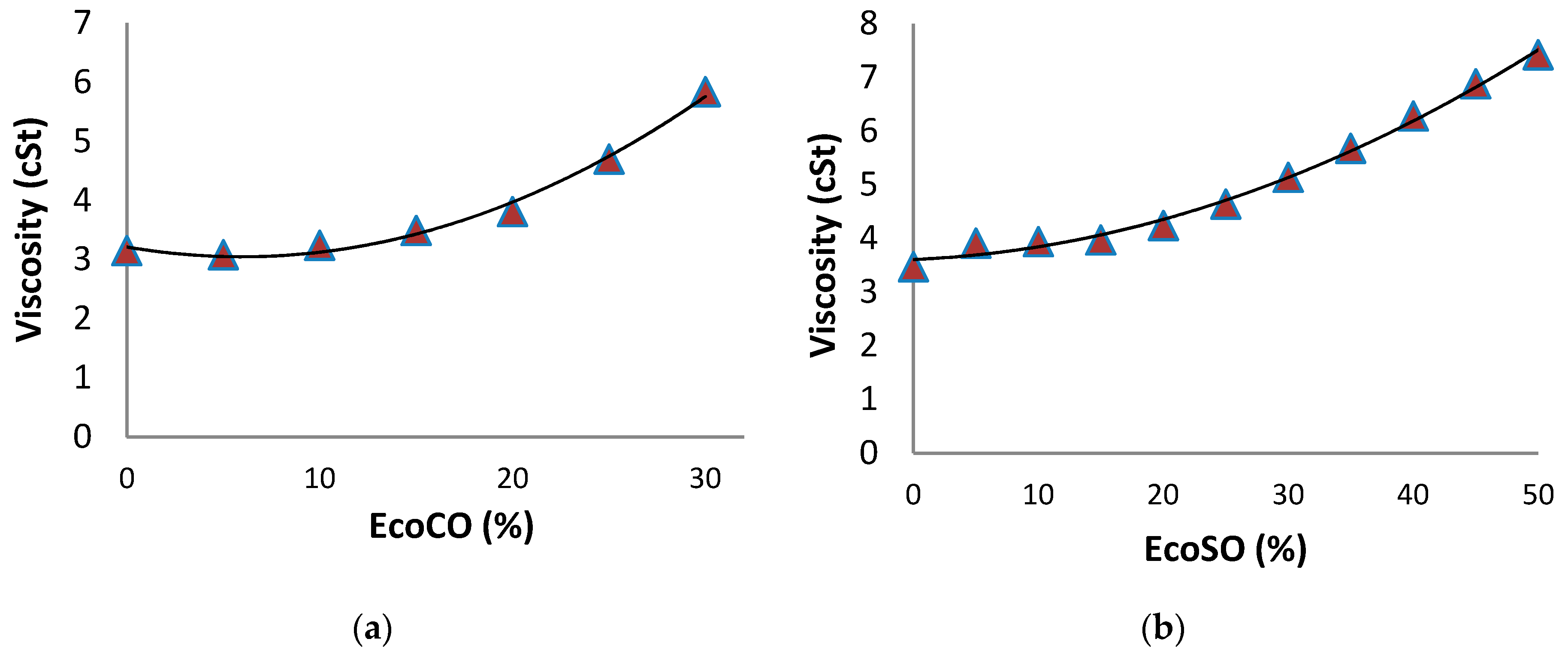
2.2.1. Double Blends: Diesel/Biofuels
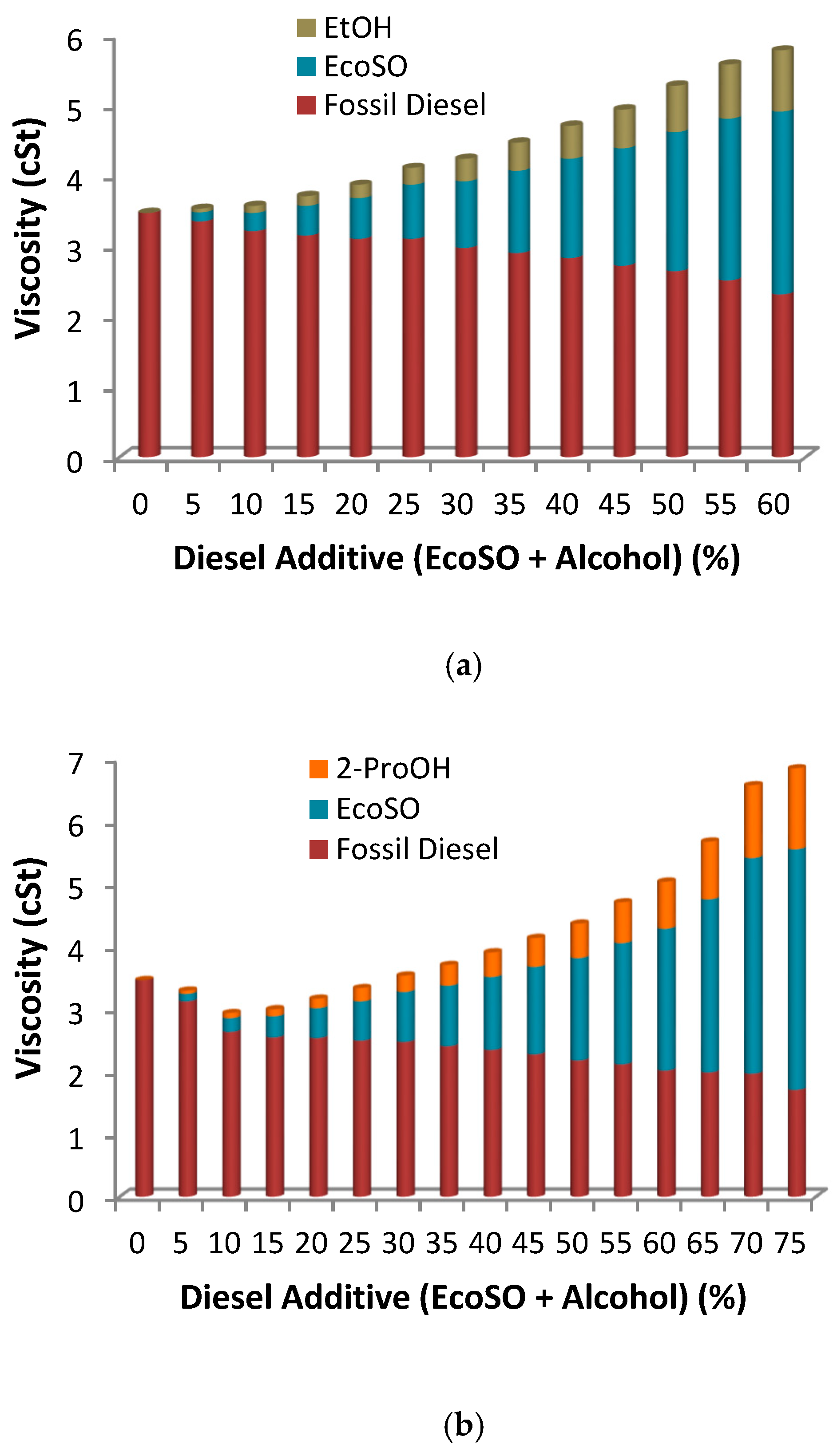
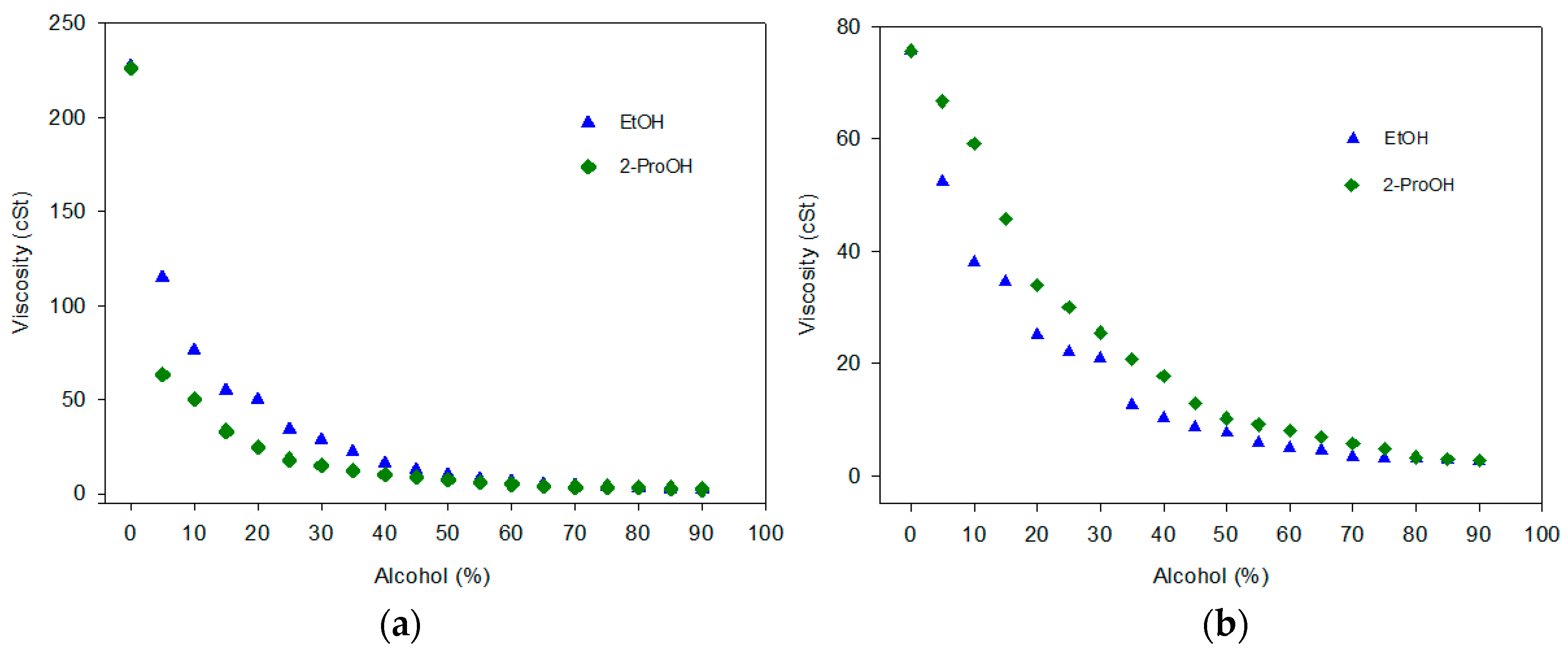
2.2.2. Triple Blends: Diesel/Biofuels/Short Chain Alcohols
2.3. Evaluation of Different Biofuels from Their Behavior in a Conventional Internal Combustion Engine
2.3.1. Double Blends Diesel/Biofuels
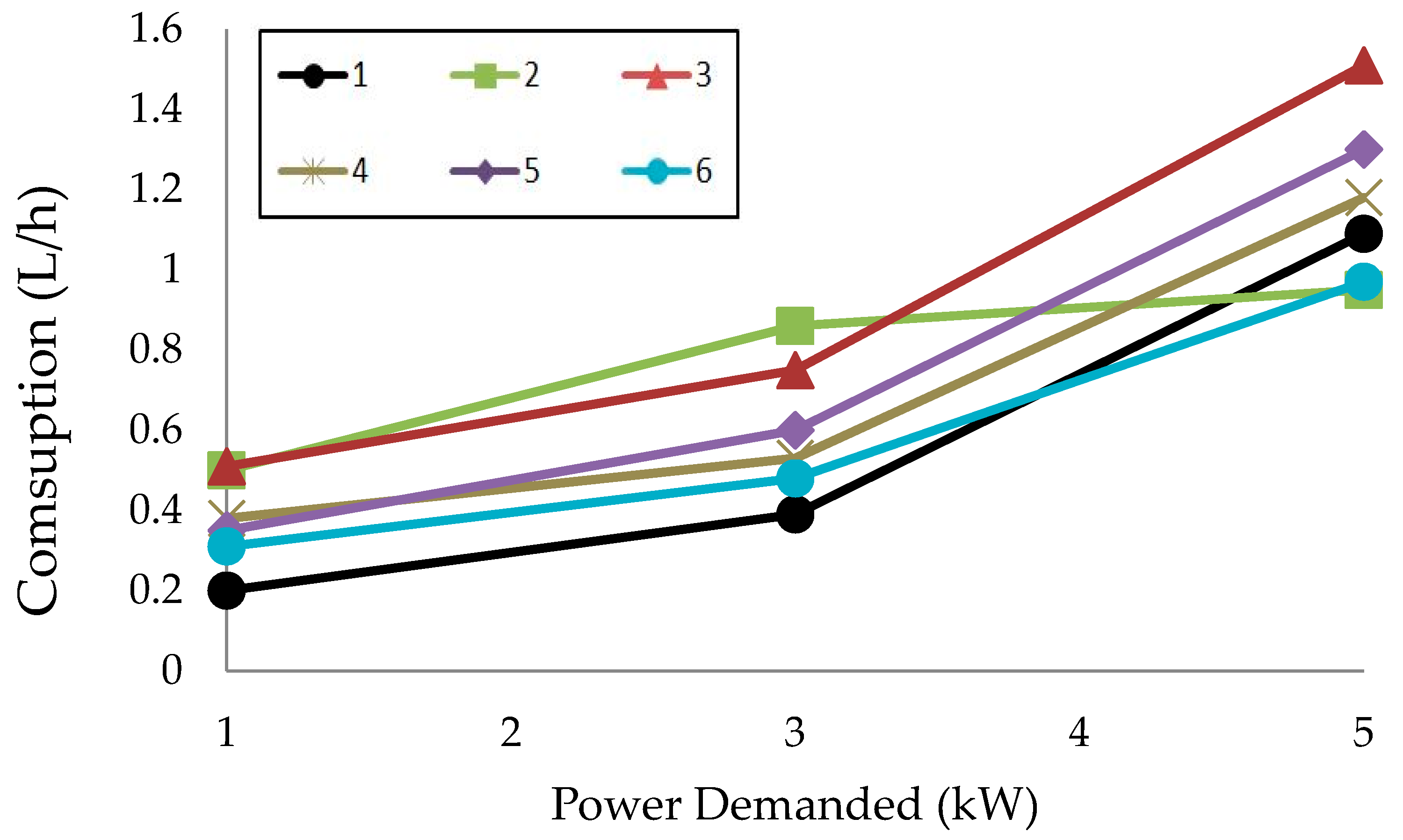
2.3.2. Triple Blends: Diesel/Biofuels/Short Chain Alcohols
3. Discussion
3.1. Synthesis of Ecodiesel by Kinetic Control of the Conventional Procedure of Biodiesel Production
3.2. Evaluation of Different Biofuels from Their Behavior in a Conventional Internal Combustion Engine
4. Materials and Methods
4.1. Evaluation of Sodium Methoxide as Homogeneous Catalyst in the Selective Alcoholysis to Obtain Ecodiesel
4.2. Determination of the Content of FAMEs, FAEEs and Glycerides (MG, DG and TG) in the Reaction Products by Gas Chromatography
4.3. Determination of Kinematic Viscosity of Biofuels
4.4. Determination of the Pour Point and Cloud Point of Biofuels
4.5. Assessment of Energy Performance and Air Pollutant Emissions in a Diesel Engine Electric Generator, Fueled with Different Blends of Biofuels
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| FAME | fatty acids methyl esters, components of conventional biodiesel |
| DMC | dimethyl carbonate |
| FAEE | Fatty Acid Ethyl Ester |
| FAE | esters of fatty acids |
| MG | monoglycerides or monoacylglycerols |
| DG | diacylglycerols |
| TG | triacylglycerols or triglycerides |
| GC | gas chromatograph |
| CO | Castor oil |
| SO | Sunflower oil |
| BCO | Biodiesel obtained from Castor oil |
| BSO | Biodiesel obtained from Sunflower oil |
| C.I. | Compression Ignition engines |
| CP | Cloud Point |
| PP | Pour point |
| CFPP | Cold filter plugging point temperature |
| POFF | Point of occlusion of the cold filter |
References
- Arutyunov, V.S.; Lisichkin, G.V. Energy resources of the 21st century: Problems and forecasts. Can renewable energy sources replace fossil fuels? Russ. Chem. Rev. 2017, 86, 777–804. [Google Scholar] [CrossRef]
- Nigam, P.S.; Singh, A. Production of liquid biofuels from renewable resources. Prog. Energy Comb. Sci. 2011, 37, 52–68. [Google Scholar] [CrossRef]
- Cheng, F.; Li, X. Preparation and Application of Biochar-Based Catalysts for Biofuel Production. Catalysts 2018, 8, 346. [Google Scholar] [CrossRef]
- Chu, S.; Majumdar, A. Opportunities and challenges for a sustainable energy future. Nature 2012, 488, 294–303. [Google Scholar] [CrossRef] [PubMed]
- Ridjan, I.; Mathiesen, B.V.; Connolly, D.; Dui, N. The feasibility of synthetic fuels in renewable energy systems. Energy 2013, 57, 76–84. [Google Scholar] [CrossRef]
- Kumar, A.A.; Sharma, S. Potential non-edible oil resources as biodiesel feedstock: An Indian perspective. Renew. Sustain. Energy Rev. 2011, 15, 1791–1800. [Google Scholar] [CrossRef]
- Rakopoulos, D.C.; Rakopoulos, C.D.; Giakoumis, E.G.; Dimaratos, A.M.; Founti, M.A. Comparative environmental behaviour of bus engine operating on blends of diesel fuel with four straight vegetable oils of Greek origin: Sunflower, cottonseed, corn and olive. Fuel 2011, 90, 3439–3446. [Google Scholar] [CrossRef]
- Lapuerta, M.; Armas, O.; Herreros, J.M. Emissions from a diesel–bioethanol blend in an automotive diesel engine. Fuel 2008, 87, 25–31. [Google Scholar] [CrossRef]
- Karabektas, M.; Hosoz, M. Performance and emission characteristics of a diesel engine using isobutanol–diesel fuel blends. Renew. Energy 2009, 34, 1554–1559. [Google Scholar] [CrossRef]
- Atmanlı, A.; Ileri, E.; Yüksel, B. Experimental investigation of engine performance and exhaust emissions of a diesel engine fueled with diesel–n-butanol–vegetable oil blends. Energy Convers. Manag. 2014, 81, 312–321. [Google Scholar] [CrossRef]
- Campos-Fernández, J.; Arnal, J.M.; Gómez, J.; Dorado, M.P. A comparison of performance of higher alcohols/diesel fuel blends in a diesel engine. Appl. Energy 2012, 95, 267–275. [Google Scholar] [CrossRef]
- Navaneethakrishnan, P.; Vasudevan, D. Experimental Study on Performance and Exhaust Emission Characteristics of a C.I. Engine Fueled with Tri Compound Oxygenated Diesel Fuel Blends. Indian J. Sci. Technol. 2015, 8, 96–102. [Google Scholar] [CrossRef]
- Yilmaz, N. Comparative analysis of biodiesel-ethanol-diesel and biodiesel-methanol-diesel blends in a diesel engine. Energy 2012, 40, 210–213. [Google Scholar] [CrossRef]
- Yilmaz, N.; Vigil, F.M.; Donaldson, A.B.; Darabseh, T. Investigation of CI engine emissions in biodiesel–ethanol–diesel blends as a function of ethanol concentration. Fuel 2014, 115, 790–793. [Google Scholar] [CrossRef]
- Shahir, S.A.; Masjuki, H.H.; Kalam, M.A.; Imran, A.; Ashraful, A.M. Performance and emission assessment of diesel–biodiesel–ethanol/bioethanol blend as a fuel in diesel engines: A review. Renew. Sustain. Energy Rev. 2015, 48, 62–78. [Google Scholar] [CrossRef]
- Aydın, F.; Ogüt, H. Effects of using ethanol-biodiesel-diesel fuel in single cylinder diesel engine to engine performance and emissions. Renew. Energy 2017, 103, 688–694. [Google Scholar] [CrossRef]
- Ghobadian, B.; Yusaf, T.; Najafi, G.; Khatamifar, M. Diesterol: An environment-friendly IC engine fuel. Renew. Energy 2009, 34, 335–342. [Google Scholar] [CrossRef]
- Noorollahi, Y.; Azadbakht, M.; Ghobadian, B.; Noorollahi, Y. The effect of different diesterol (diesel–biodiesel–ethanol) blends on small air-cooled diesel engine performance and its exhaust gases. Energy 2018, 142, 196–200. [Google Scholar] [CrossRef]
- Quispe, C.A.G.; Coronado, C.R.; Carvalho, J.A., Jr. Glycerol: Production, consumption, prices, characterization and new trends in combustion. Renew. Sustain. Energy Rev. 2013, 27, 475–493. [Google Scholar] [CrossRef]
- Oh, P.P.; Lau, H.L.N.; Chen, J.; Chong, M.F.; Choo, Y.M. A review on conventional technologies and emerging process intensification (PI) methods for biodiesel production. Renew. Sustain. Energy Rev. 2012, 16, 5131–5145. [Google Scholar] [CrossRef]
- Calero, J.; Luna, D.; Sancho, E.D.; Luna, C.; Bautista, F.M.; Romero, A.A.; Posadillo, A.; Berbel, J.; Verdugo-Escamilla, C. An overview on glycerol-free processes for the production of renewable liquid biofuels, applicable in diesel engines. Renew. Sustain. Energy Rev. 2015, 42, 1437–1452. [Google Scholar] [CrossRef]
- Calero, J.; Luna, D.; Sancho, E.D.; Luna, C.; Posadillo, A.; Bautista, F.M.; Romero, A.A.; Berbel, J.; Verdugo, C. Technological challenges for the production of biodiesel in arid lands. J. Arid Environ. 2014, 102, 127–138. [Google Scholar] [CrossRef]
- Verdugo, C.; Luque, R.; Luna, D.; Hidalgo, J.M.; Posadillo, A.; Sancho, E.D.; Rodriguez, S.; Ferreira-Dias, S.; Bautista, F.; Romero, A.A. A comprehensive study of reaction parameters in the enzymatic production of novel biofuels integrating glycerol into their composition. Bioresour. Technol. 2010, 101, 6657–6662. [Google Scholar] [CrossRef] [PubMed]
- Luna, C.; Sancho, E.; Luna, D.; Caballero, V.; Calero, J.; Posadillo, A.; Verdugo, C.; Bautista, F.M.; Romero, A.A. Biofuel that keeps glycerol as monoglyceride by 1,3-selective ethanolysis with pig pancreatic lipase covalently immobilized on AlPO4 support. Energies 2013, 6, 3879–3900. [Google Scholar] [CrossRef]
- Luna, C.; Verdugo, C.; Sancho, E.D.; Luna, D.; Calero, J.; Posadillo, A.; Bautista, F.M.; Romero, A.A. Production of a biodiesel-like biofuel without glycerol generation, by using Novozym 435, an immobilized Candida antarctica lipase. Bioresour. Bioprocess. 2014, 1, 11. [Google Scholar] [CrossRef]
- Luna, C.; Verdugo, C.; Sancho, E.D.; Luna, D.; Calero, J.; Posadillo, A.; Bautista, F.M.; Romero, A.A. Biocatalytic behaviour of immobilized Rhizopus oryzae lipase in the 1,3-selective ethanolysis of sunflower oil to obtain a biofuel similar to biodiesel. Molecules 2014, 19, 11419–11439. [Google Scholar] [CrossRef] [PubMed]
- Luna, C.; Luna, D.; Bautista, F.M.; Estevez, R.; Calero, J.; Posadillo, A.; Romero, A.A.; Sancho, E.D. Application of Enzymatic Extracts from a CALB Standard Strain as Biocatalyst within the Context of Conventional Biodiesel Production Optimization. Molecules 2017, 22, 2025. [Google Scholar] [CrossRef]
- Calero, J.; Cumplido, G.; Luna, D.; Sancho, E.D.; Luna, C.; Posadillo, A.; Bautista, F.M.; Romero, A.A.; Verdugo-Escamilla, C. Production of a Biofuel that Keeps the Glycerol as a Monoglyceride by Using Supported KF as Heterogeneous Catalyst. Energies 2014, 7, 3764–3780. [Google Scholar] [CrossRef]
- Calero, J.; Luna, D.; Sancho, E.D.; Luna, C.; Bautista, F.M.; Romero, A.A.; Posadillo, A.; Verdugo, C. develoment of a new biodiesel that integrates glycerol, by using CaO as heterogeneous catalyst, in the partial methanolysis of sunflower oil. Fuel 2014, 7, 94–102. [Google Scholar] [CrossRef]
- Dias, J.M.; Araújo, J.M.; Costa, J.F.; Alvim-Ferraz, M.C.M.; Almeida, M.F. Biodiesel production from raw castor oil. Energy 2013, 53, 58–69. [Google Scholar] [CrossRef]
- Kim, K.H.; Lee, O.K.; Lee, E.Y. Nano-Immobilized Biocatalysts for Biodiesel Production from Renewable and Sustainable Resources. A review. Catalysts 2018, 8, 68. [Google Scholar] [CrossRef]
- Bello, E.I.; Makanju, A. Production, Characterization and Evaluation of Castor oil Biodiesel as Alternative Fuel for Diesel Engines. J. Emerg. Trends Eng. Appl. Sci. 2011, 2, 525–530. [Google Scholar]
- Scholz, V.; Silva, J.N. Prospects and risks of the use of castor oil as a fuel. A review. Biomass Bioenergy 2008, 32, 95–100. [Google Scholar] [CrossRef]
- Patel, V.R.; Dumancas, G.G.; Viswanath, L.C.K.; Maples, R.; Subong, B.J.J. Castor Oil: Properties, Uses, and Optimization of Processing Parameters in Commercial Production. Lipid Insights 2016, 9, 1–12. [Google Scholar] [CrossRef]
- Knothe, G. “Designer” Biodiesel: Optimizing Fatty Ester Composition to Improve Fuel Properties. Energy Fuels 2008, 22, 1358–1364. [Google Scholar] [CrossRef]
- Berman, P.; Nizri, S.; Wiesman, Z. Castor oil biodiesel and its blends as alternative fuel. Biomass Bioenergy 2011, 35, 2861–2866. [Google Scholar] [CrossRef]
- Panwar, N.L.; Shrirame, H.Y.; Rathore, N.S.; Jindal, S.; Kurchania, A.K. Performance evaluation of a diesel engine fueled with methyl ester of castor seed oil. Appl. Therm. Eng. 2010, 30, 245–249. [Google Scholar] [CrossRef]
- Valente, O.S.; Silva, M.J.; Pasa, V.M.D.; Belchior, C.R.P.; Sodré, J.R. Fuel consumption and emissions from a diesel power generator fuelled with castor oil and soybean biodiesel. Fuel 2010, 89, 3637–3642. [Google Scholar] [CrossRef]
- Nabi, M.N.; Akhter, M.S.; Shahadat, M.M.Z. Improvement of engine emissions with conventional diesel fuel and diesel–biodiesel blends. Bioresour. Technol. 2006, 97, 372–378. [Google Scholar] [CrossRef]
- Atabani, A.E.; Silitonga, A.S.; Ong, H.C.; Mahlia, T.M.I.; Masjuki, H.H. Non-edible vegetable oils: A critical evaluation of oil extraction, fatty acid compositions, biodiesel production, characteristics, engine performance and emissions production. Renew. Sustain. Energy Rev. 2013, 18, 211–245. [Google Scholar] [CrossRef]
- Gokdogan, O.; Eryilmaz, T.; Yesilyurt, M.K. Thermophysical properties of castor oil (Ricinus communis L.) biodıesel and its blends. CT&F 2015, 6, 95–128. [Google Scholar]
- Akanaw, T.T.; Moges, H.G.; Babu, R.; Bisrat, D. Castor Seed from Melkasa Agricultural Research Centre, East Showa, Ethiopia and it’s biodiesel performance in Four Stroke Diesel Engine. Int. J. Renew. Energy Dev. 2014, 3, 3–99. [Google Scholar] [CrossRef]
- Mobarak, H.M.; Mohamad, E.N.; Masjuki, H.H.; Kalam, M.A.; Mahmud, K.A.H.A.; Habibullah, M.; Ashraful, A.M. The prospects of biolubricants as alternatives in automotive applications. Renew. Sustain. Energy Rev. 2014, 33, 34–43. [Google Scholar] [CrossRef]
- Posadillo, A. ptimization of Experimental Conditions for the Production of Second Generation Biofuels that Integrate Glycerol. Universidad de Córdoba, 2016. Available online: https://helvia.uco.es/handle/10396/13237 (accessed on 1 October 2015).
- Lee, W.J.; Liu, Y.-C.; Mwangi, F.K.; Chen, W.H.; Lin, S.-L.; Fukushima, Y.; Liao, C.-N.; Wang, L.C. Assessment of energy performance and air pollutant emissions in a diesel engine generator fueled with water-containing ethanol–biodiesel–diesel blend of fuels. Energy 2011, 36, 5591–5599. [Google Scholar] [CrossRef]
- Chaves, L.I.; de Souza, S.N.M.; Rosa, H.A.; Bariccatti, R.A.; Nogueira, C.E.C.; Secco, D.; Wazilewski, W.T.; Avaci, A.B.; Brenneisen, P.J.; da Silva, M.J. Comparative performance of a diesel cycle engine using blends of diesel and biodiesel. Afr. J. Biotechnol. 2012, 11, 14578–14585. [Google Scholar] [CrossRef]
- Blanco, M.; Coello, J.; Maspoch, S.; Puigdomènech, A.; Peralta, X.; González, J.M.; Torres, J. Correlating Bacharach opacity in fuel oil exhaust. prediction of the operating parameters that reduce it. Oil Gas Sci. Technol. IFP 2000, 55, 533–541. [Google Scholar]
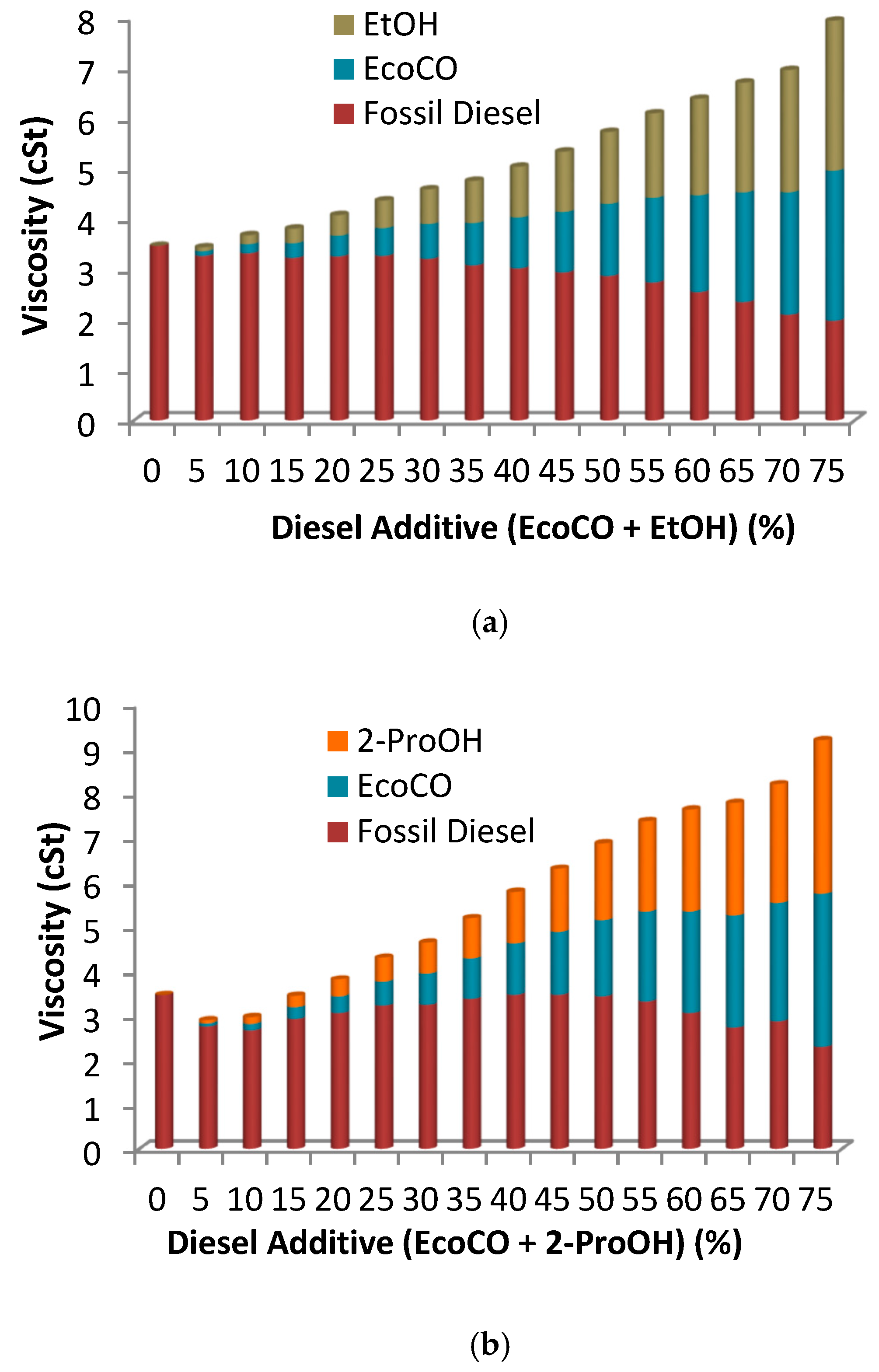
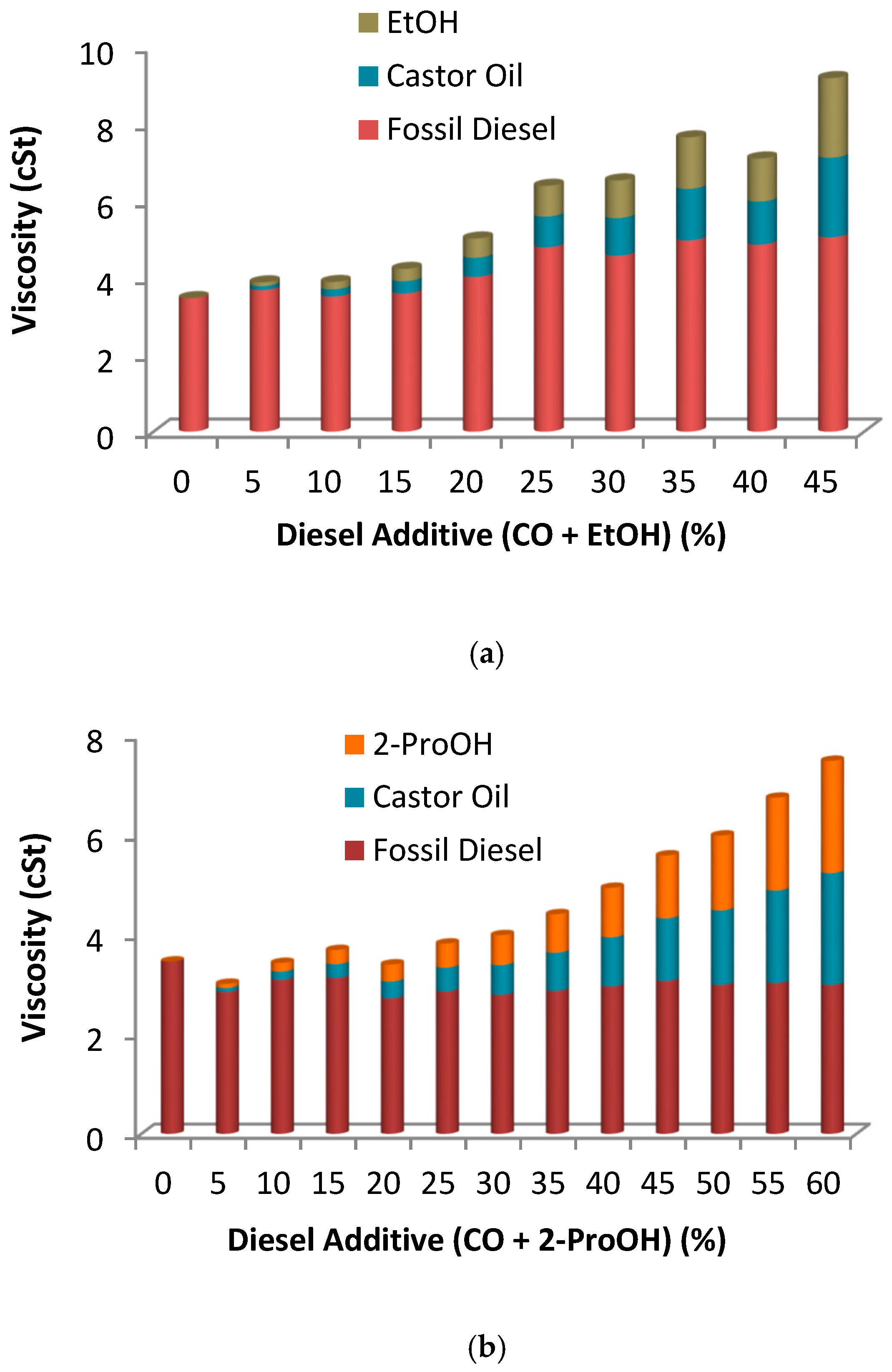

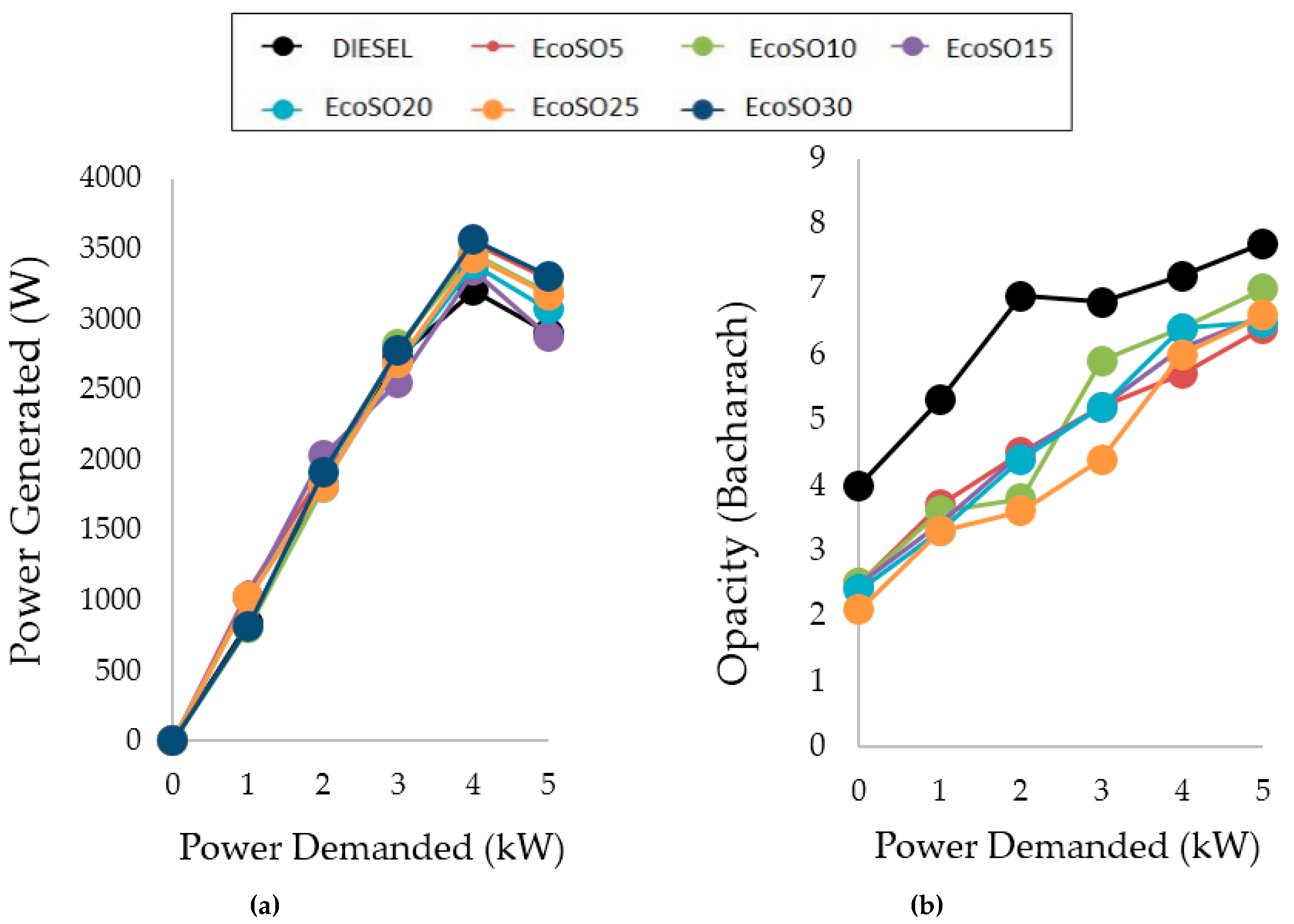
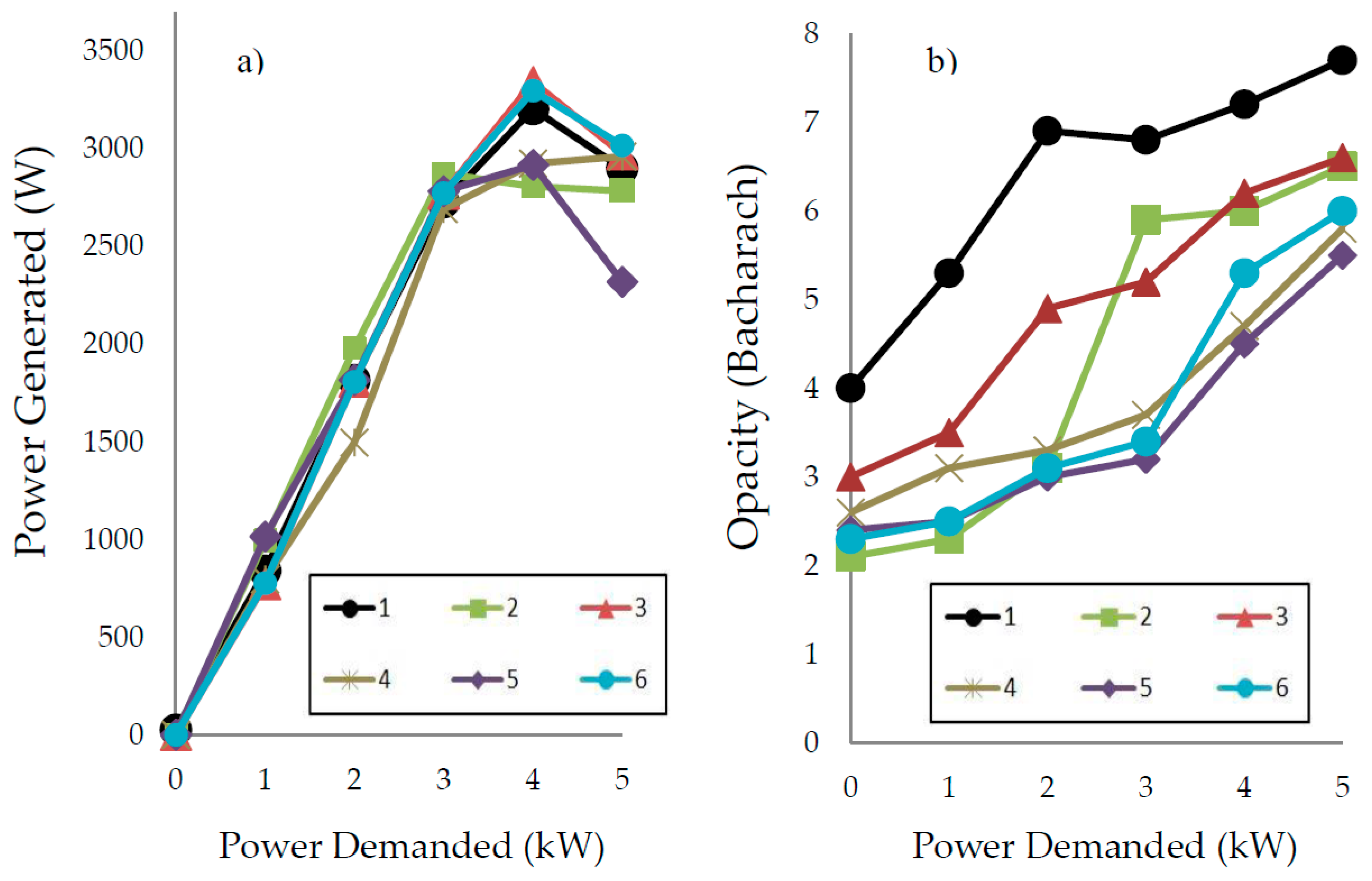


| Vegetable Oil | Oil Viscosity (cSt) | Selectivity (%) | FAME (%) | MG (%) | DG (%) | Biodiesel Viscosity (cSt) |
|---|---|---|---|---|---|---|
| Sunflower | 37.37 | 91.9 | 85.3 | 6.6 | 8.1 | 4 |
| Castor | 227.0 | 100 | 90.0 | 10.0 | 0.0 | 11 |
| Nº | EtOH (mL) | MeOH (mL) | NaOMe (mL) | Visc. (cSt) | Sel. (%) | FAE (%) | MG (%) | DG (%) |
|---|---|---|---|---|---|---|---|---|
| 1 | 25 | 35 | 10 | 6.60 | 89.1 | 64.5 | 24.6 | 10.9 |
| 2 | 25 | 25 | 10 | 10.95 | 86.9 | 67.6 | 19.3 | 13.1 |
| 3 | 20 | 20 | 7 | 13.18 | 80.7 | 64.3 | 15.4 | 19.3 |
| 4 | 30 | 30 | 5 | 9.43 | 77.1 | 63.4 | 13.7 | 22.9 |
| 5 | 25 | 25 | 5 | 13.15 | 80.0 | 64.9 | 15.1 | 20.0 |
| 6 | 30 | 25 | 5 | 11.77 | 77.8 | 58.5 | 19.3 | 22.2 |
| Time (min) | Conv. (%) | Sel. (%) | FAME (%) | MG (%) | DG (%) | TG (%) | Ecodiesel Visc. (cSt) |
|---|---|---|---|---|---|---|---|
| 15 | 67.3 | 44.0 | 34.1 | 9.9 | 23.3 | 32.7 | 93 |
| 30 | 71.1 | 42.7 | 30.4 | 12.3 | 28.4 | 28.9 | 106 |
| 45 | 73.3 | 52.0 | 40.9 | 11.1 | 21.3 | 26.7 | 64 |
| 60 | 78.0 | 50.8 | 40.3 | 10.5 | 27.2 | 22.0 | 76 |
| 15 * | 58.9 | 23.2 | 19.6 | 3.6 | 35.7 | 41.1 | 12 |
| 30 * | 86.7 | 38.9 | 32.4 | 6.5 | 47.8 | 13.3 | 12 |
| 45 * | 100 | 84.3 | 70.8 | 13.5 | 15.7 | 0.0 | 11 |
| 60 * | 100 | 66.6 | 54.4 | 12.2 | 33.4 | 0.0 | 11 |
| Blends | Biofuel in Blend (%) | Viscosity (cSt) | Pour Point (°C) | Cloud Point (°C) |
|---|---|---|---|---|
| BSO10 | 10 | 3.17 | −15 | −10 |
| BSO20 | 20 | 3.30 | −10 | −12 |
| BSO30 | 30 | 3.50 | −10 | −12 |
| BSO40 | 40 | 3.83 | −9 | −11 |
| BSO50 | 50 | 4.16 | −9 | −8 |
| EcoSO10 | 10 | 3.93 | −11 | −10 |
| EcoSO20 | 20 | 4.42 | −10 | −9 |
| EcoSO30 | 30 | 5.15 | −9 | −8 |
| EcoSO40 | 40 | 6.86 | −9 | −7 |
| EcoSO50 | 50 | 7.13 | −8 | −6 |
| Entry | Diesel/Biofuel/Alcohol (%) | Viscosity (cSt) | Pour Point (°C) | Cloud Point (°C) |
|---|---|---|---|---|
| 1 | Diesel (100) | 3.47 | −19 | −4 |
| 2 | Diesel/EcoSO/EtOH (50/40/10) | 4.72 | −9 | −7 |
| 3 | Diesel/EcoSO/2-ProOH (35/50/15) | 5.20 | −12 | −9 |
| 4 | Diesel/EcoCO/2-ProOH (50/25/25) | 5.60 | −13 | −11 |
| 5 | Diesel/CO/EtOH (50/25/25) | 5.93 | −7 | −6 |
| 6 | Diesel/CO/2-ProOH (50/25/25) | 5.81 | −8 | −5 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hurtado, B.; Posadillo, A.; Luna, D.; Bautista, F.M.; Hidalgo, J.M.; Luna, C.; Calero, J.; Romero, A.A.; Estevez, R. Synthesis, Performance and Emission Quality Assessment of Ecodiesel from Castor Oil in Diesel/Biofuel/Alcohol Triple Blends in a Diesel Engine. Catalysts 2019, 9, 40. https://doi.org/10.3390/catal9010040
Hurtado B, Posadillo A, Luna D, Bautista FM, Hidalgo JM, Luna C, Calero J, Romero AA, Estevez R. Synthesis, Performance and Emission Quality Assessment of Ecodiesel from Castor Oil in Diesel/Biofuel/Alcohol Triple Blends in a Diesel Engine. Catalysts. 2019; 9(1):40. https://doi.org/10.3390/catal9010040
Chicago/Turabian StyleHurtado, Beatriz, Alejandro Posadillo, Diego Luna, Felipa M. Bautista, Jose M. Hidalgo, Carlos Luna, Juan Calero, Antonio A. Romero, and Rafael Estevez. 2019. "Synthesis, Performance and Emission Quality Assessment of Ecodiesel from Castor Oil in Diesel/Biofuel/Alcohol Triple Blends in a Diesel Engine" Catalysts 9, no. 1: 40. https://doi.org/10.3390/catal9010040
APA StyleHurtado, B., Posadillo, A., Luna, D., Bautista, F. M., Hidalgo, J. M., Luna, C., Calero, J., Romero, A. A., & Estevez, R. (2019). Synthesis, Performance and Emission Quality Assessment of Ecodiesel from Castor Oil in Diesel/Biofuel/Alcohol Triple Blends in a Diesel Engine. Catalysts, 9(1), 40. https://doi.org/10.3390/catal9010040








