High Active and Selective Ni/CeO2–Al2O3 and Pd–Ni/CeO2–Al2O3 Catalysts for Oxy-Steam Reforming of Methanol
Abstract
1. Introduction
2. Results and Discussion
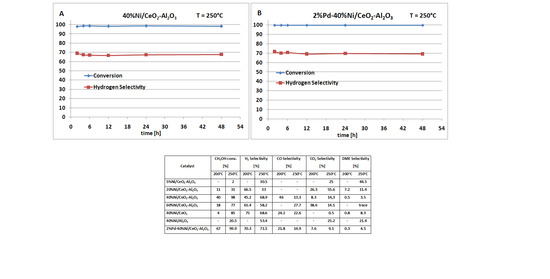
2.1. Catalytic Performance in Oxy-Steam Reforming of Methanol
2.2. Specific Surface Area and Chemisorption Measurements
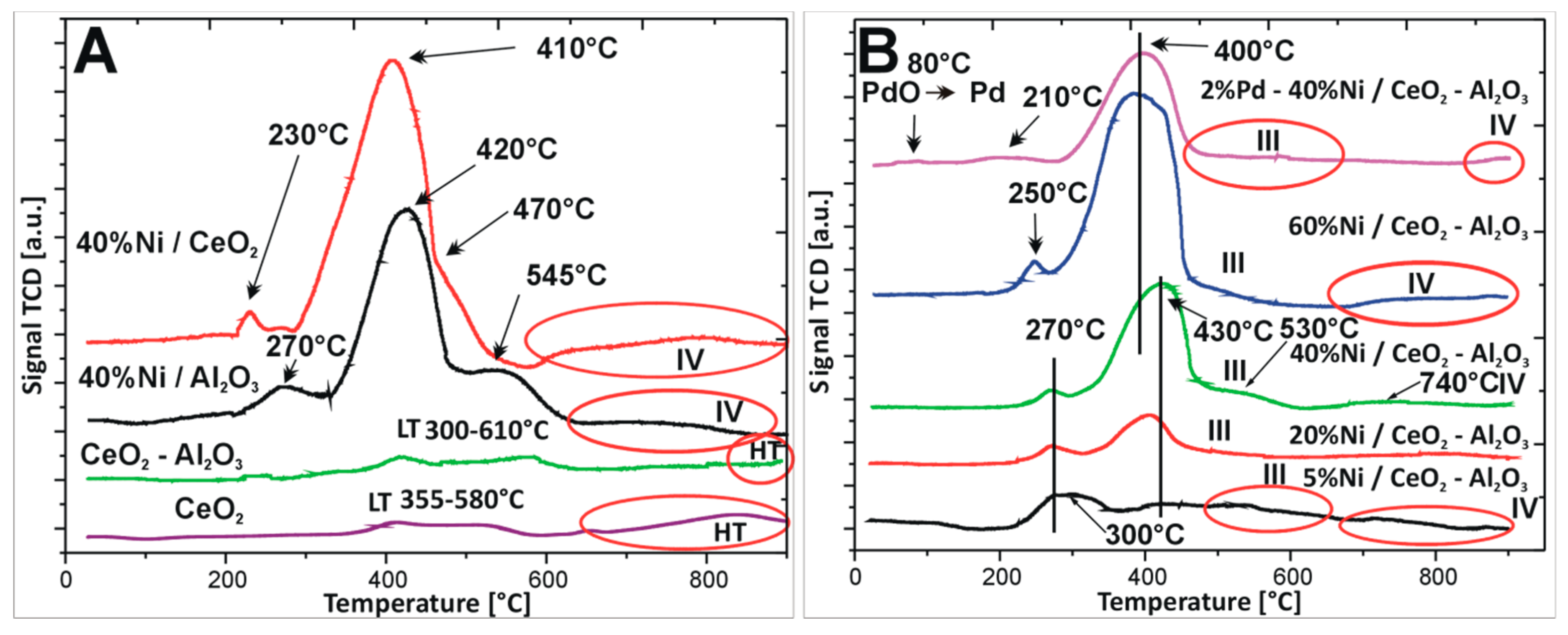
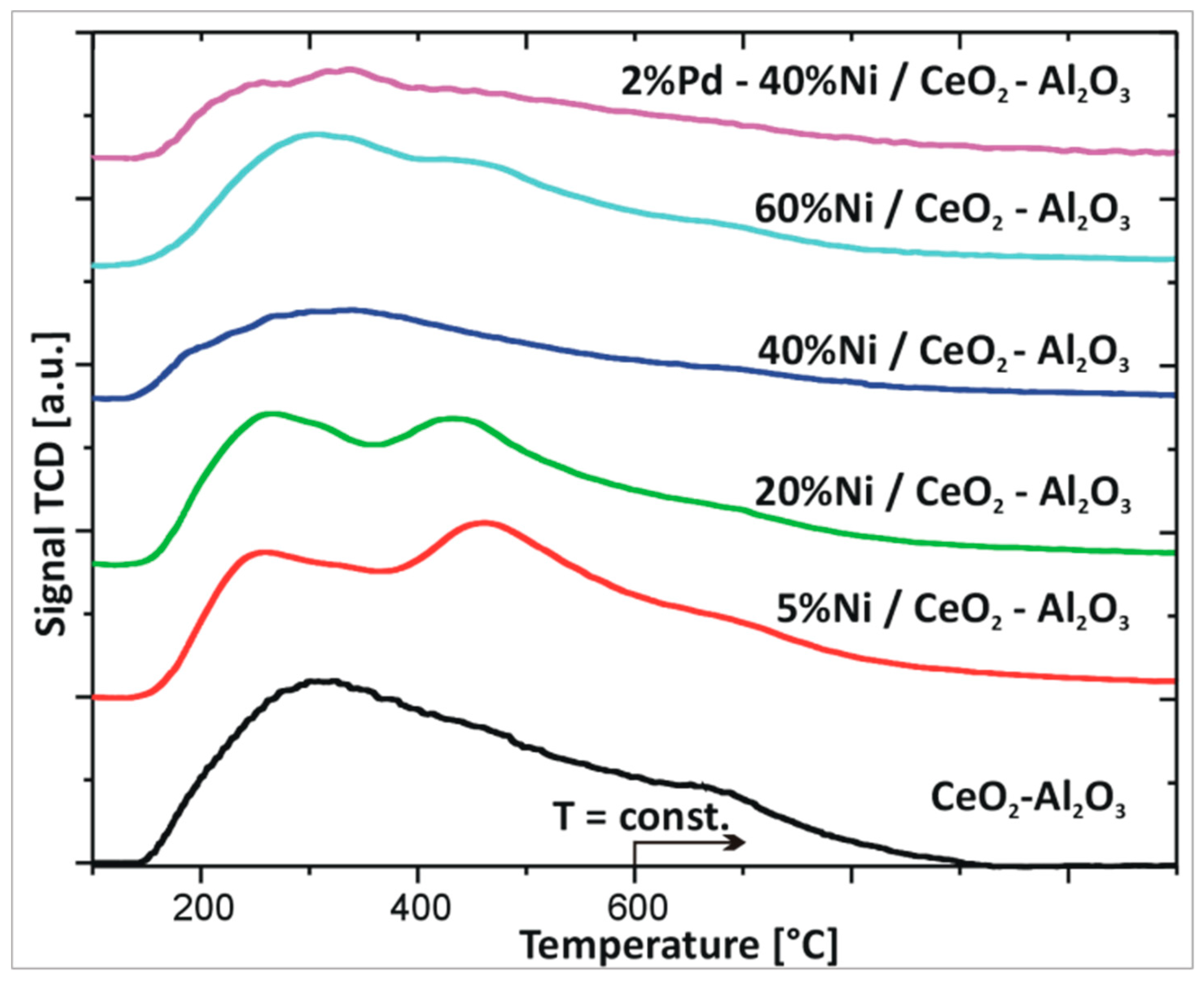
2.3. Reduction Behaviour of Monometallic Ni and Bimetallic Pd–Ni Supported Catalysts
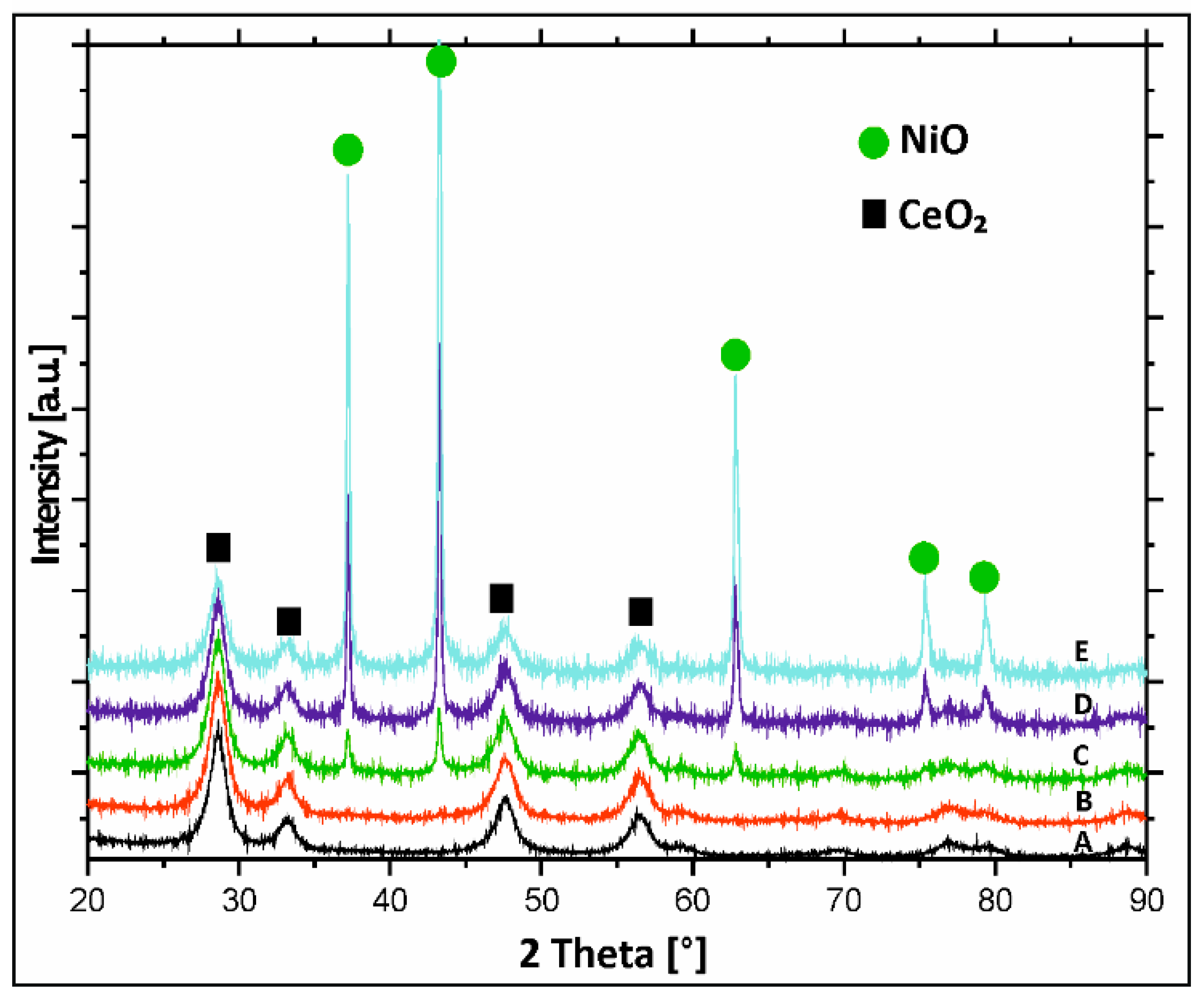
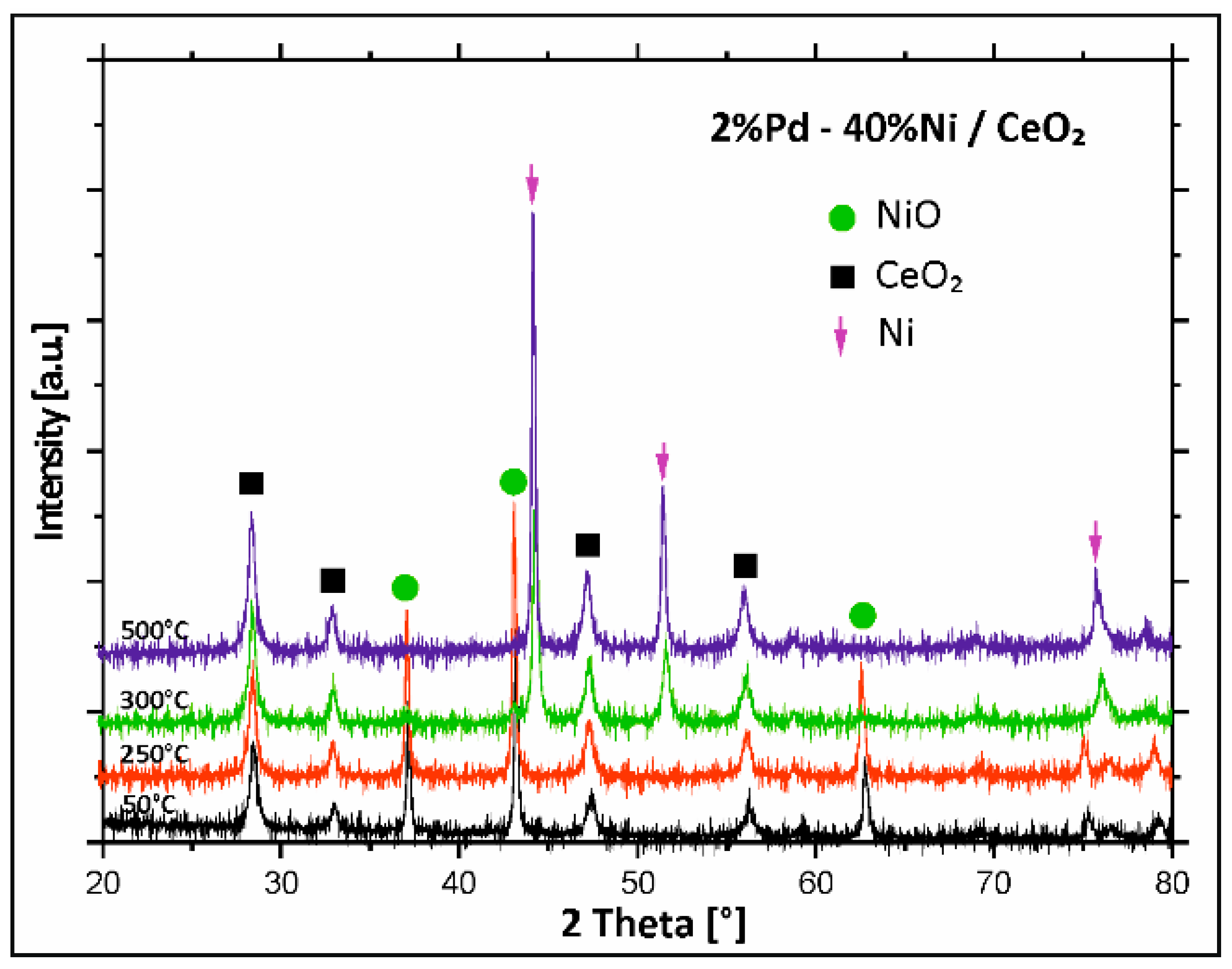
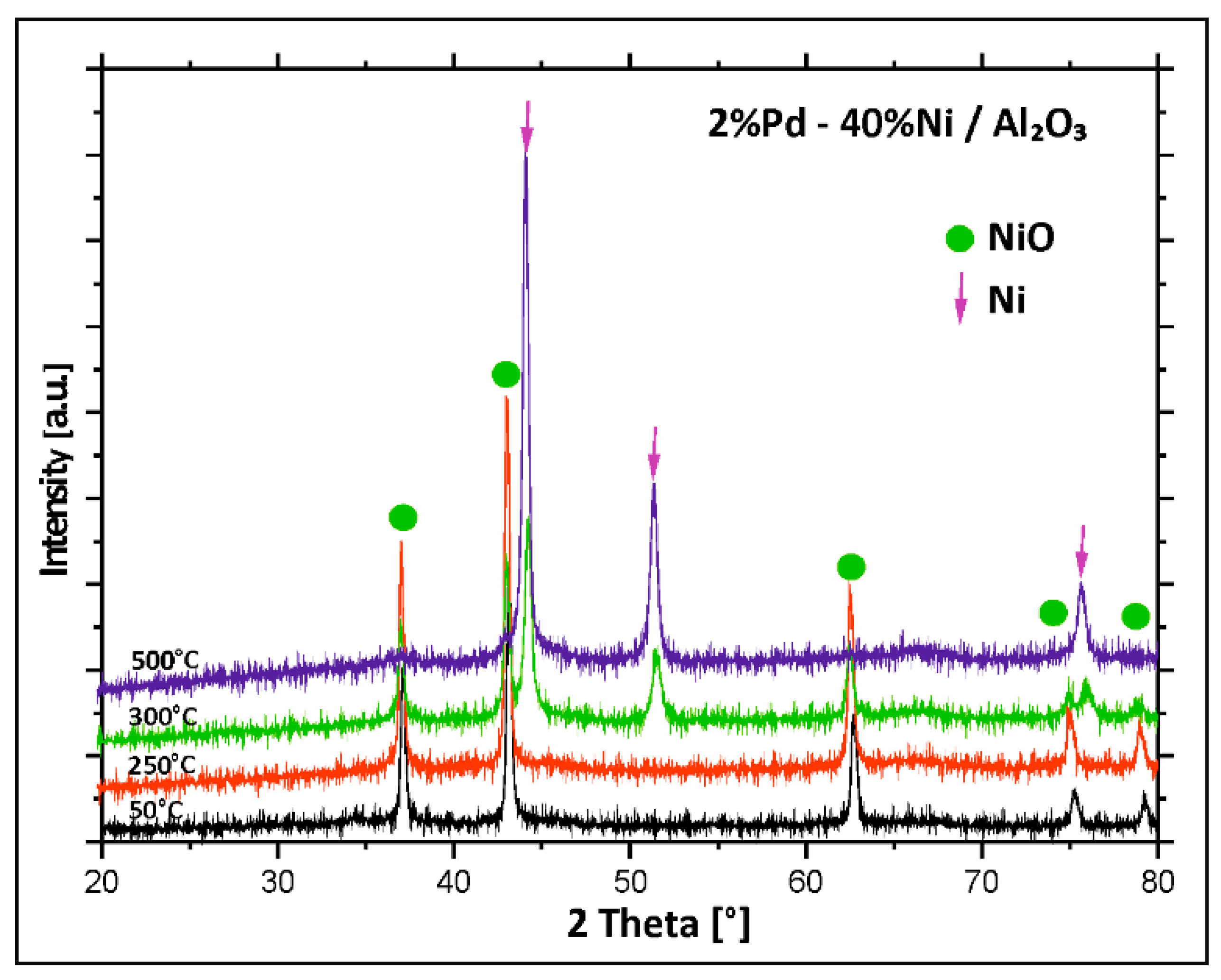
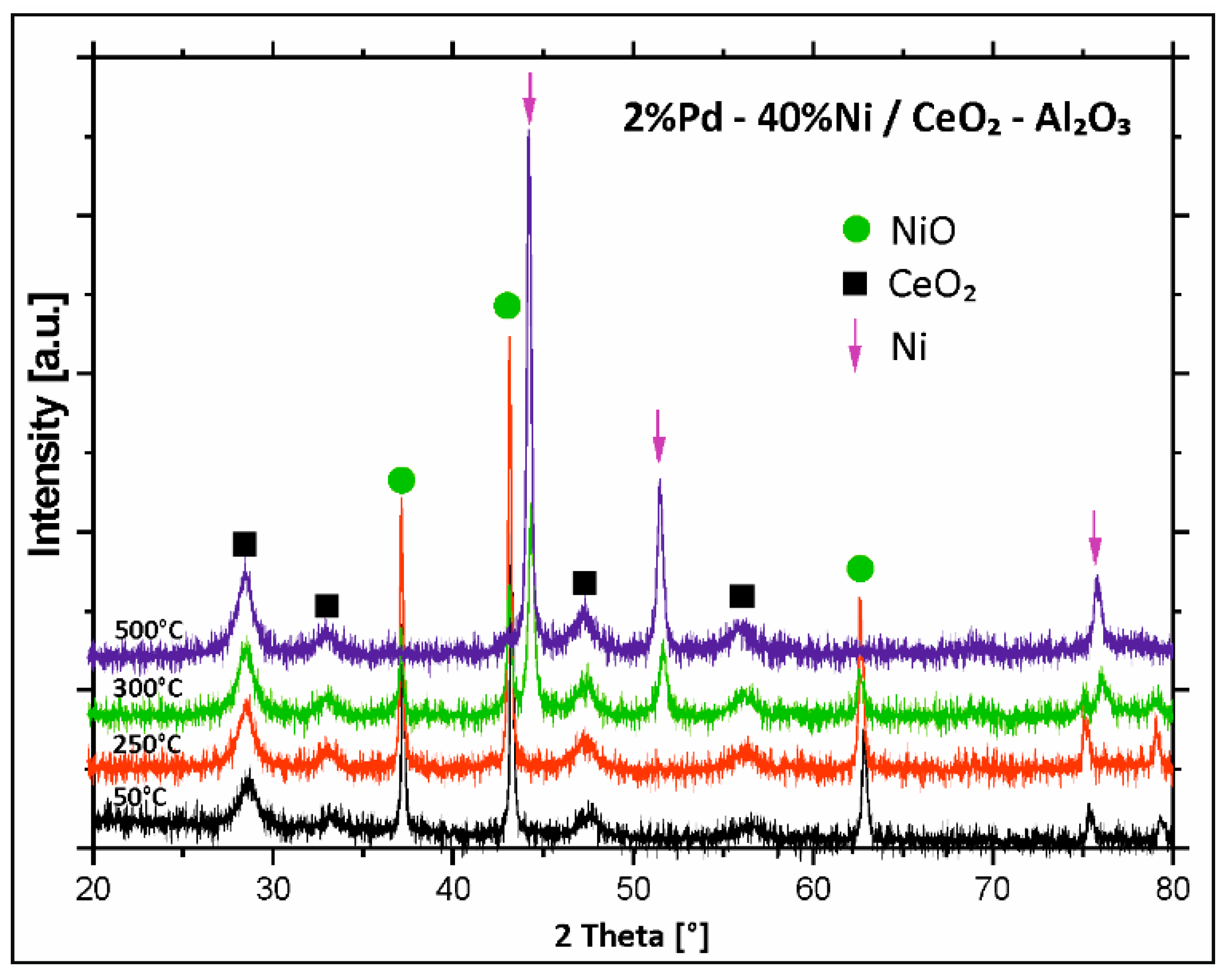
2.4. XRD Structural Analysis
2.5. Temperature Programmed Desorption of Ammonia TPD–NH3
2.6. Morphology Studies of Mono and Bimetallic Supported Catalysts Using SEM–EDS
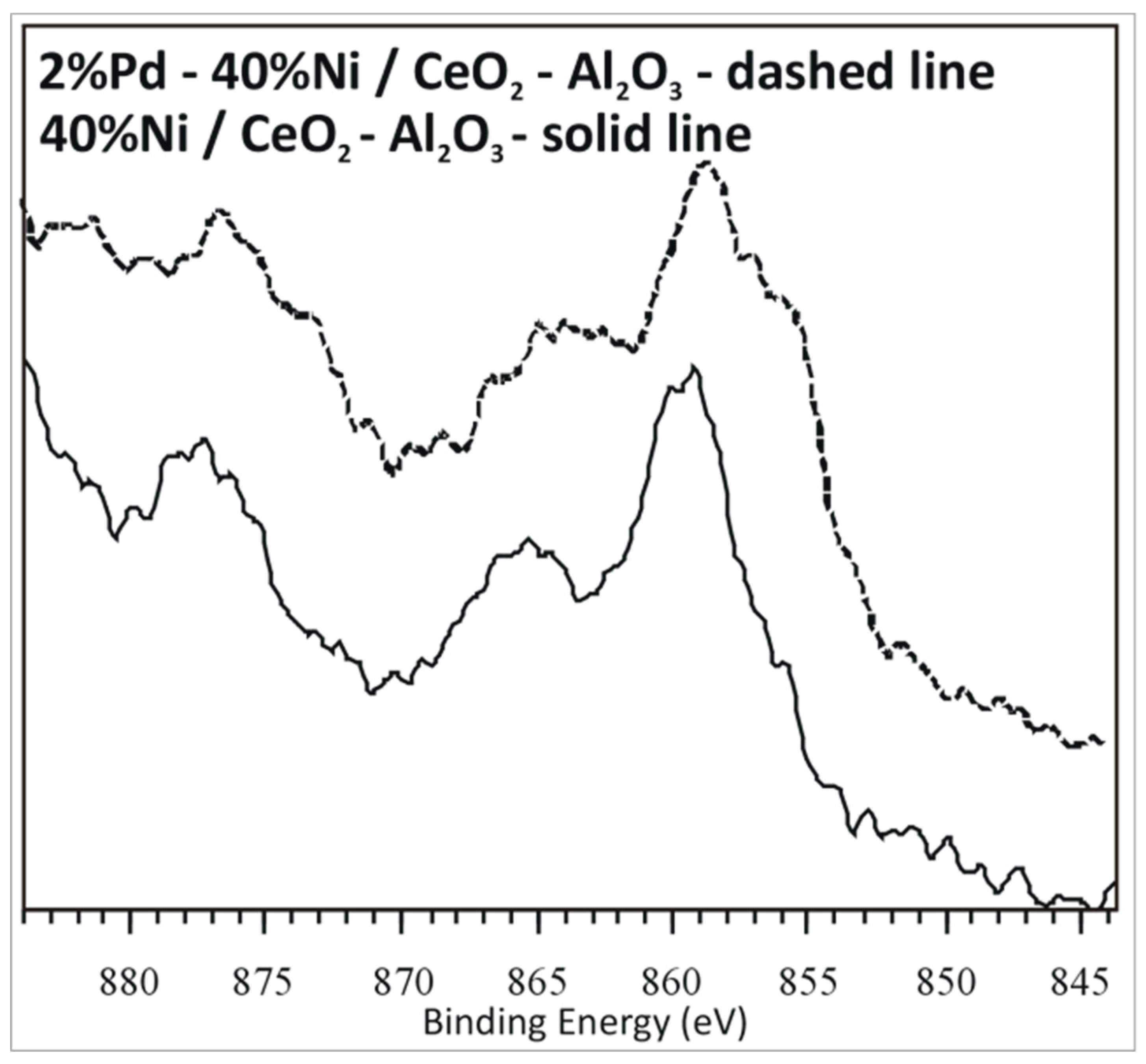
2.7. Interaction between the Active Phase Component and Support
3. Experimental
3.1. Catalytic Material Preparation
3.1.1. Support Material
3.1.2. Preparation of Supported Monometallic and Bimetallic Catalysts
3.2. Catalytic Material Characterization
3.3. Catalytic Activity Evaluation
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Mierczynski, P.; Vasilev, K.; Mierczynska, A.; Maniukiewicz, W.; Szynkowska, M.I.; Maniecki, T.P. Bimetallic Au–Cu, Au–Ni catalysts supported on MWCNTs for oxy-steam reforming of methanol. Appl. Catal. B Environ. 2016, 185, 281–294. [Google Scholar] [CrossRef]
- Voloshin, R.A.; Rodionova, M.V.; Zharmukhamedov, S.K.; Veziroglu, T.N.; Allakhverdiev, S.I. Review: Biofuel production from plant and algal biomass. Int. J. Hydrog. Energy 2016, 41, 17257–17273. [Google Scholar] [CrossRef]
- Bae, J.; Lee, S.; Kim, S.; Oh, J.; Choi, S.; Bae, M.; Kang, I.; Katikaneni, S.P. Liquid fuel processing for hydrogen production: A review. Int. J. Hydrog. Energy 2016, 41, 19990–20022. [Google Scholar] [CrossRef]
- Mierczynski, P.; Vasilev, K.; Mierczynska, A.; Ciesielski, R.; Maniukiewicz, W.; Rogowski, J.; Szynkowska, I.M.; Trifonov, A.Y.; Dubkov, S.V.; Gromov, D.; et al. The effect of gold on modern bimetallic Au-Cu/MWCNT catalysts for oxy-steam reforming of methanol. Catal. Sci. Technol. 2016, 6, 4168–4183. [Google Scholar] [CrossRef]
- Lu, J.; Li, X.; He, S.; Han, C.; Wan, G.; Lei, Y.; Chen, R.; Liu, P.; Chen, K.; Zhang, L.; et al. Hydrogen production via methanol steam reforming over Ni-based catalysts: Influences of Lanthanum (La) addition and supports. Int. J. Hydrog. Energy 2017, 42, 3647–3657. [Google Scholar] [CrossRef]
- Mierczynski, P.; Ciesielski, R.; Kedziora, A.; Nowosielska, M.; Kubicki, J.; Maniukiewicz, W.; Czylkowska, A.; Maniecki, T. Monometallic copper catalysts supported on multi-walled carbon nanotubes for the oxy-steam reforming of methanol. React. Kinet. Mech. Catal. 2016, 117, 675–691. [Google Scholar] [CrossRef]
- Liu, X.; Toyir, J.; de la Piscina, P.R.; Homs, N. Hydrogen production from methanol steam reforming over Al2O3- and ZrO2-modified CuOZnOGa2O3 catalysts. Int. J. Hydrog. Energy 2017, 42, 13704–13711. [Google Scholar] [CrossRef]
- Mierczynski, P.; Vasilev, K.; Mierczynska, A.; Maniukiewicz, W.; Maniecki, T.P. Highly selective Pd–Cu/ZnAl2O4 catalyst for hydrogen production. Appl. Catal. A Gen. 2014, 479, 26–34. [Google Scholar] [CrossRef]
- Liu, D.; Men, Y.; Wang, J.; Kolb, G.; Liu, X.; Wang, Y.; Sun, Q. Highly active and durable Pt/In2O3/Al2O3 catalysts in methanol steam reforming. Int. J. Hydrog. Energy 2016, 41, 21990–21999. [Google Scholar] [CrossRef]
- Mierczynski, P.; Vasilev, K.; Mierczynska, A.; Maniukiewicz, W.; Maniecki, T. The Effect of ZnAl2O4 on the Performance of Cu/ZnxAlyOx+1.5y Supported Catalysts in Steam Reforming of Methanol. Top. Catal. 2013, 56, 1015–1025. [Google Scholar] [CrossRef]
- Luo, Z.; Li, D.; Tang, H.; Pan, M.; Ruan, R. Degradation behavior of membrane-electrode-assembly materials in 10-cell PEMFC stack. Int. J. Hydrog. Energy 2006, 31, 1831–1837. [Google Scholar] [CrossRef]
- Xu, H.; Hou, X. Synergistic effect of CeO2 modified Pt/C electrocatalysts on the performance of PEM fuel cells. Int. J. Hydrog. Energy 2007, 32, 4397–4401. [Google Scholar] [CrossRef]
- Mierczynski, P. Comparative Studies of Bimetallic Ru–Cu, Rh–Cu, Ag–Cu, Ir–Cu Catalysts Supported on ZnO–Al2O3, ZrO2–Al2O3 Systems. Catal. Lett. 2016, 146, 1825–1837. [Google Scholar] [CrossRef]
- López, P.; Mondragón-Galicia, G.; Espinosa-Pesqueira, M.E.; Mendoza-Anaya, D.; Fernández, M.E.; Gómez-Cortés, A.; Bonifacio, J.; Martínez-Barrera, G.; Pérez-Hernández, R. Hydrogen production from oxidative steam reforming of methanol: Effect of the Cu and Ni impregnation on ZrO2 and their molecular simulation studies. Int. J. Hydrog. Energy 2012, 37, 9018–9027. [Google Scholar] [CrossRef]
- Mierczynski, P.; Mierczynska, A.; Maniukiewicz, W.; Maniecki, T.P.; Vasilev, K. MWCNTs as a catalyst in oxy-steam reforming of methanol. RSC Adv. 2016, 6, 81408–81413. [Google Scholar] [CrossRef]
- Chang, C.-C.; Chang, C.-T.; Chiang, S.-J.; Liaw, B.-J.; Chen, Y.-Z. Oxidative steam reforming of methanol over CuO/ZnO/CeO2/ZrO2/Al2O3 catalysts. Int. J. Hydrog. Energy 2010, 35, 7675–7683. [Google Scholar] [CrossRef]
- Chang, C.-C.; Hsu, C.-C.; Chang, C.-T.; Chen, Y.-P.; Liaw, B.-J.; Chen, Y.-Z. Effect of noble metal on oxidative steam reforming of methanol over CuO/ZnO/Al2O3 catalysts. Int. J. Hydrog. Energy 2012, 37, 11176–11184. [Google Scholar] [CrossRef]
- Pérez-Hernández, R.; Gutiérrez-Martínez, A.; Gutiérrez-Wing, C.E. Effect of Cu loading on for hydrogen production by oxidative steam reforming of methanol. Int. J. Hydrog. Energy 2007, 32, 2888–2894. [Google Scholar] [CrossRef]
- Sá, S.; Silva, H.; Brandão, L.; Sousa, J.M.; Mendes, A. Catalysts for methanol steam reforming—A review. Appl. Catal. B Environ. 2010, 99, 43–57. [Google Scholar] [CrossRef]
- Pérez-Hernández, R.; Gutiérrez-Martínez, A.; Espinosa-Pesqueira, M.E.; Estanislao, M.L.; Palacios, J. Effect of the bimetallic Ni/Cu loading on the ZrO2 support for H2 production in the autothermal steam reforming of methanol. Catal. Today 2015, 250, 166–172. [Google Scholar] [CrossRef]
- Yong, S.T.; Ooi, C.W.; Chai, S.P.; Wu, X.S. Review of methanol reforming-Cu-based catalysts, surface reaction mechanisms, and reaction schemes. Int. J. Hydrog. Energy 2013, 38, 9541–9552. [Google Scholar] [CrossRef]
- Pérez-Hernández, R.; Galicia, G.M.; Anaya, D.M.; Palacios, J.; Angeles-Chavez, C.; Arenas-Alatorre, J. Synthesis and characterization of bimetallic Cu–Ni/ZrO2 nanocatalysts: H2 production by oxidative steam reforming of methanol. Int. J. Hydrog. Energy 2008, 33, 4569–4576. [Google Scholar] [CrossRef]
- Yang, H.-M.; Chan, M.-K. Steam reforming of methanol over copper–yttria catalyst supported on praseodymium–aluminum mixed oxides. Catal. Commun. 2011, 12, 1389–1395. [Google Scholar] [CrossRef]
- Perez-Hernandez, R.; Mondragon-Galicia, G.; Maravilla, A.A.; Palacios, J. Nano-dimensional CeO2 nanorods for high Ni loading catalysts: H2 production by autothermal steam reforming of methanol reaction. Phys. Chem. Chem. Phys. 2013, 15, 12702–12708. [Google Scholar] [CrossRef] [PubMed]
- Turco, M.; Bagnasco, G.; Cammarano, C.; Senese, P.; Costantino, U.; Sisani, M. Cu/ZnO/Al2O3 catalysts for oxidative steam reforming of methanol: The role of Cu and the dispersing oxide matrix. Appl. Catal. B Environ. 2007, 77, 46–57. [Google Scholar] [CrossRef]
- Pojanavaraphan, C.; Satitthai, U.; Luengnaruemitchai, A.; Gulari, E. Activity and stability of Au/CeO2–Fe2O3 catalysts for the hydrogen production via oxidative steam reforming of methanol. J. Ind. Eng. Chem. 2015, 22, 41–52. [Google Scholar] [CrossRef]
- Turco, M.; Bagnasco, G.; Costantino, U.; Marmottini, F.; Montanari, T.; Ramis, G.; Busca, G. Production of hydrogen from oxidative steam reforming of methanol: I. Preparation and characterization of Cu/ZnO/Al2O3 catalysts from a hydrotalcite-like LDH precursor. J. Catal. 2004, 228, 43–55. [Google Scholar] [CrossRef]
- Pojanavaraphan, C.; Luengnaruemitchai, A.; Gulari, E. Catalytic activity of Au-Cu/CeO2-ZrO2 catalysts in steam reforming of methanol. Appl. Catal. A Gen. 2013, 456, 135–143. [Google Scholar] [CrossRef]
- Yong-Feng, L.; Xin-Fa, D.; Wei-Ming, L. Effects of ZrO2-promoter on catalytic performance of CuZnAlO catalysts for production of hydrogen by steam reforming of methanol. Int. J. Hydrog. Energy 2004, 29, 1617–1621. [Google Scholar] [CrossRef]
- Santo, V.D.; Gallo, A.; Naldoni, A.; Guidotti, M.; Psaro, R. Bimetallic heterogeneous catalysts for hydrogen production. Catal. Today 2012, 197, 190–205. [Google Scholar] [CrossRef]
- Rad, S.J.H.; Haghighi, M.; Eslami, A.A.; Rahmani, F.; Rahemi, N. Sol–gel vs. impregnation preparation of MgO and CeO2 doped Ni/Al2O3 nanocatalysts used in dry reforming of methane: Effect of process conditions, synthesis method and support composition. Int. J. Hydrog. Energy 2016, 41, 5335–5350. [Google Scholar]
- Li, H.; Xu, H.; Wang, J. Methane reforming with CO2 to syngas over CeO2-promoted Ni/Al2O3-ZrO2 catalysts prepared via a direct sol-gel process. J. Nat. Gas Chem. 2011, 20, 1–8. [Google Scholar] [CrossRef]
- Ahmed, W.; Awadallah, A.E.; Aboul-Enein, A.A. Ni/CeO2–Al2O3 catalysts for methane thermo-catalytic decomposition to COx-free H2 production. Int. J. Hydrog. Energy 2016, 41, 18484–18493. [Google Scholar] [CrossRef]
- Das, D.; Llorca, J.; Dominguez, M.; Colussi, S.; Trovarelli, A.; Gayen, A. Methanol steam reforming behavior of copper impregnated over CeO2–ZrO2 derived from a surfactant assisted coprecipitation route. Int. J. Hydrog. Energy 2015, 40, 10463–10479. [Google Scholar] [CrossRef]
- Mierczynski, P.; Ciesielski, R.; Kedziora, A.; Shtyka, O.; Maniecki, T.P. Methanol Synthesis Using Copper Catalysts Supported on CeO2−Al2O3 Mixed Oxide. Fibre Chem. 2017, 48, 271–275. [Google Scholar] [CrossRef]
- Li, G.; Hu, L.; Hill, J.M. Comparison of reducibility and stability of alumina-supported Ni catalysts prepared by impregnation and co-precipitation. Appl. Catal. A Gen. 2006, 301, 16–24. [Google Scholar] [CrossRef]
- Guo, R.-T.; Zhou, Y.; Pan, W.-G.; Hong, J.-N.; Zhen, W.-L.; Jin, Q.; Ding, C.-G.; Guo, S.-Y. Effect of preparation methods on the performance of CeO2/Al2O3 catalysts for selective catalytic reduction of NO with NH3. J. Ind. Eng. Chem. 2013, 19, 2022–2025. [Google Scholar] [CrossRef]
- Ding, M.-Y.; Tu, J.-Y.; Wang, T.-J.; Ma, L.-L.; Wang, C.-G.; Chen, L.-G. Bio-syngas methanation towards synthetic natural gas (SNG) over highly active Al2O3–CeO2 supported Ni catalyst. Fuel Process. Technol. 2015, 134, 480–486. [Google Scholar] [CrossRef]
- Kapoor, M.P.; Ichihashi, Y.; Kuraoka, K.; Shen, W.-J.; Matsumura, Y. Catalytic Methanol Decomposition over Palladium Deposited on Mesoporous Cerium Oxide. Catal. Lett. 2003, 88, 83–87. [Google Scholar] [CrossRef]
- Kapoor, M.P.; Ichihashi, Y.; Kuraoka, K.; Matsumura, Y. Catalytic methanol decomposition over palladium deposited on thermally stable mesoporous titanium oxide. J. Mol. Catal. A Chem. 2003, 198, 303–308. [Google Scholar] [CrossRef]
- Lenarda, M.; Moretti, E.; Storaro, L.; Patrono, P.; Pinzari, F.; Rodríguez-Castellón, E.; Jiménez-López, A.; Busca, G.; Finocchio, E.; Montanari, T.; et al. Finely dispersed Pd-Zn catalyst supported on an organized mesoporous alumina for hydrogen production by methanol steam reforming. Appl. Catal. A Gen. 2006, 312, 220–228. [Google Scholar] [CrossRef]
- Chin, Y.-H.; Dagle, R.; Hu, J.; Dohnalkova, A.C.; Wang, Y. Steam reforming of methanol over highly active Pd/ZnO catalyst. Catal. Today 2002, 77, 79–88. [Google Scholar] [CrossRef]
- Cubeiro, M.L.; Fierro, J.L.G. Selective Production of Hydrogen by Partial Oxidation of Methanol over ZnO-Supported Palladium Catalysts. J. Catal. 1998, 179, 150–162. [Google Scholar] [CrossRef]
- Rynkowski, J.M.; Paryjczak, T.; Lewicki, A.; Szynkowska, M.I.; Maniecki, T.P.; Jóźwiak, W.K. Characterization of Ru/CeO2-Al2O3 catalysts and their Performance in CO2 Methanation. React. Kinet. Catal. Lett. 2000, 71, 55–64. [Google Scholar] [CrossRef]
- Mierczynski, P.; Maniukiewicz, W.; Maniecki, T. Comparative studies of Pd, Ru, Ni, Cu/ZnAl2O4 catalysts for the water gas shift reaction (vol 11, pg 912, 2013). Cent. Eur. J. Chem. 2013, 11, 1255. [Google Scholar]
- Zheng, W.; Zhang, J.; Ge, Q.; Xu, H.; Li, W. Effects of CeO2 addition on Ni/Al2O3 catalysts for the reaction of ammonia decomposition to hydrogen. Appl. Catal. B Environ. 2008, 80, 98–105. [Google Scholar] [CrossRef]
- Mierczynski, P.; Maniukiewicz, W.; Maniecki, T. Comparative studies of Pd, Ru, Ni, Cu/ZnAl2O4 catalysts for the water gas shift reaction. Cent. Eur. J. Chem. 2013, 11, 912–919. [Google Scholar]
- Maniecki, T.; Bawolak-Olczak, K.; Mierczynski, P.; Maniukiewicz, W.; Jozwiak, W. Effect of the chemical composition of (MgO)(x)(Al2O3)(y) support on the catalytic performance of Ni and Ni-Au catalysts for the partial oxidation of methane. Chem. Eng. J. 2009, 154, 142–148. [Google Scholar] [CrossRef]
- Maniecki, T.; Stadnichenko, A.; Maniukiewicz, W.; Bawolak, K.; Mierczynski, P.; Boronin, A.; Jozwiak, W. An active phase transformation on surface of Ni-Au/Al2O3 catalyst during partial oxidation of methane to synthesis gas. Kinet. Catal. 2010, 51, 573–578. [Google Scholar] [CrossRef]
- Ding, M.; Tu, J.; Zhang, Q.; Wang, M.; Tsubaki, N.; Wang, T.; Ma, L. Enhancement of methanation of bio-syngas over CeO2-modified Ni/Al2O3 catalysts. Biomass Bioenergy 2016, 85, 12–17. [Google Scholar] [CrossRef]
- Wu, H.; Pantaleo, G.; la Parola, V.; Venezia, A.M.; Collard, X.; Aprile, C.; Liotta, L.F. Bi- and trimetallic Ni catalysts over Al2O3 and Al2O3-MOx (M = Ce or Mg) oxides for methane dry reforming: Au and Pt additive effects. Appl. Catal. B Environ. 2014, 156–157, 350–361. [Google Scholar] [CrossRef]
- Osorio-Vargas, P.; Flores-González, N.A.; Navarro, R.M.; Fierro, J.L.G.; Campos, C.H.; Reyes, P. Improved stability of Ni/Al2O3 catalysts by effect of promoters (La2O3, CeO2) for ethanol steam-reforming reaction. Catal. Today 2016, 259, 27–38. [Google Scholar] [CrossRef]
- Reddy, B.M.; Rao, K.N.; Reddy, G.K.; Khan, A.; Park, S.-E. Structural Characterization and Oxidehydrogenation Activity of CeO2/Al2O3 and V2O5/CeO2/Al2O3 Catalysts. J. Phys. Chem. C 2007, 111, 18751–18758. [Google Scholar] [CrossRef]
- Jiao, Y.; Zhang, J.; Du, Y.; Li, F.; Li, C.; Lu, J.; Wang, J.; Chen, Y. Hydrogen production by catalytic steam reforming of hydrocarbon fuels over Ni/Ce–Al2O3 bifunctional catalysts: Effects of SrO addition. Int. J. Hydrog. Energy 2016, 41, 13436–13447. [Google Scholar] [CrossRef]
- Luisetto, I.; Tuti, S.; Battocchio, C.; Mastro, S.L.; Sodo, A. Ni/CeO2–Al2O3 catalysts for the dry reforming of methane: The effect of CeAlO3 content and nickel crystallite size on catalytic activity and coke resistance. Appl. Catal. A Gen. 2015, 500, 12–22. [Google Scholar] [CrossRef]
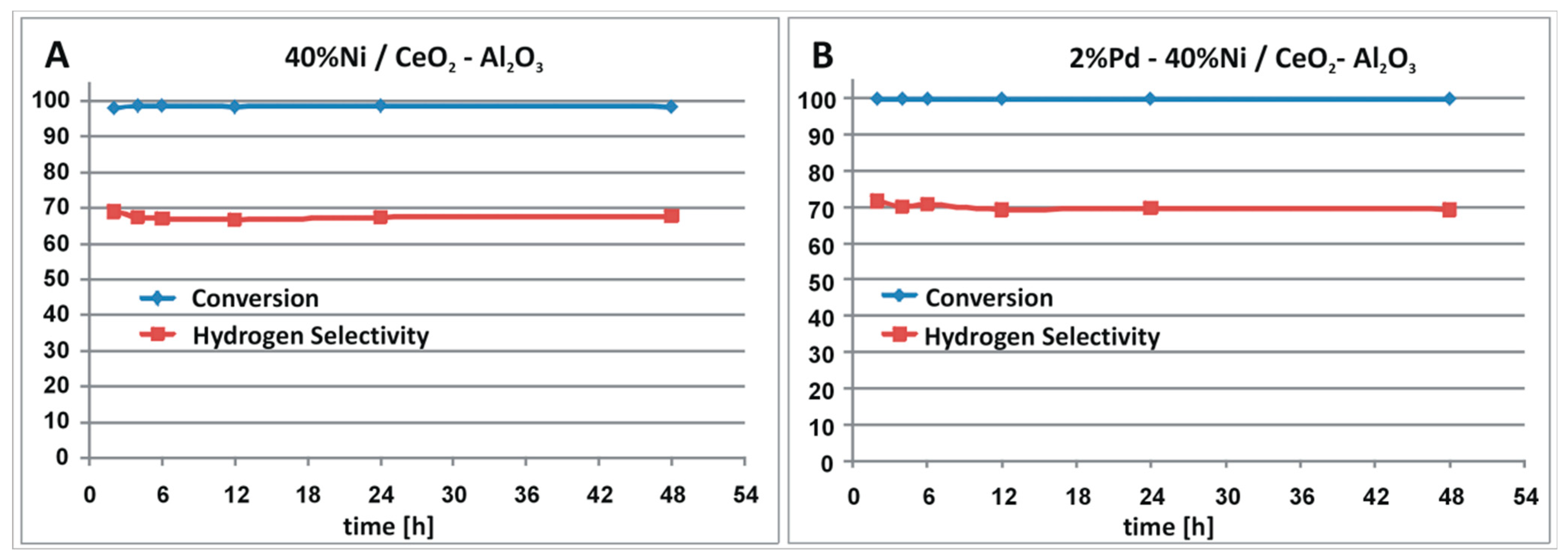
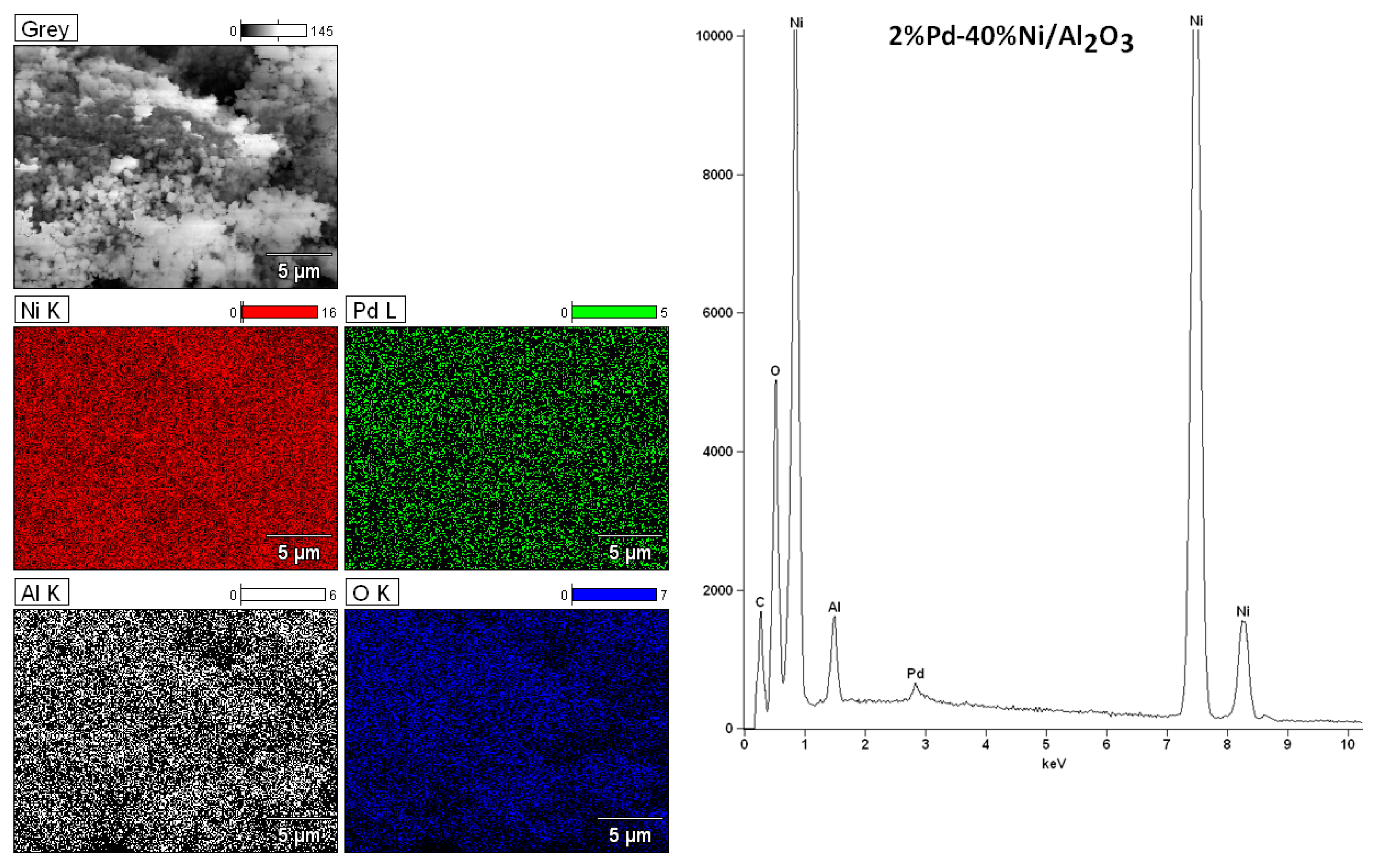
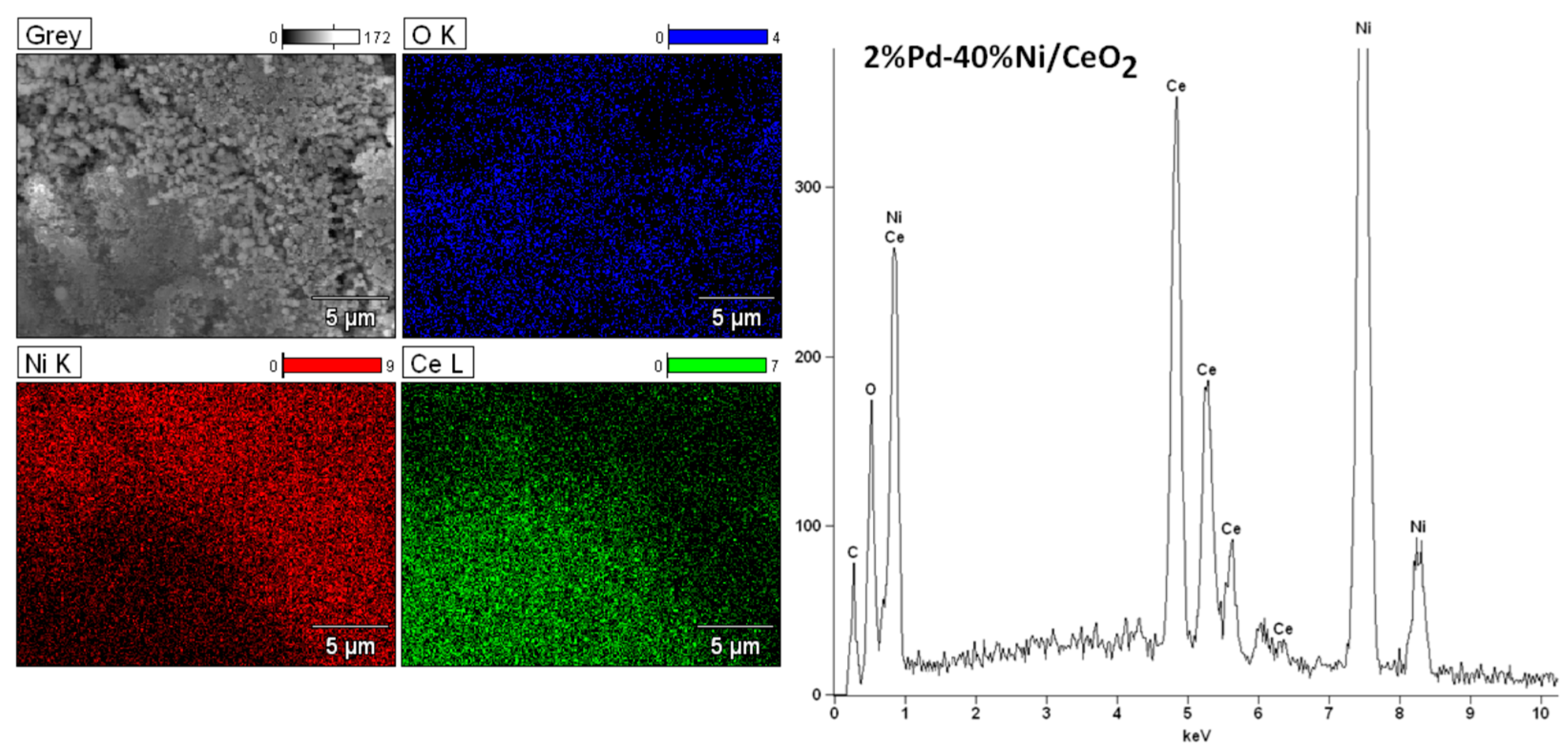
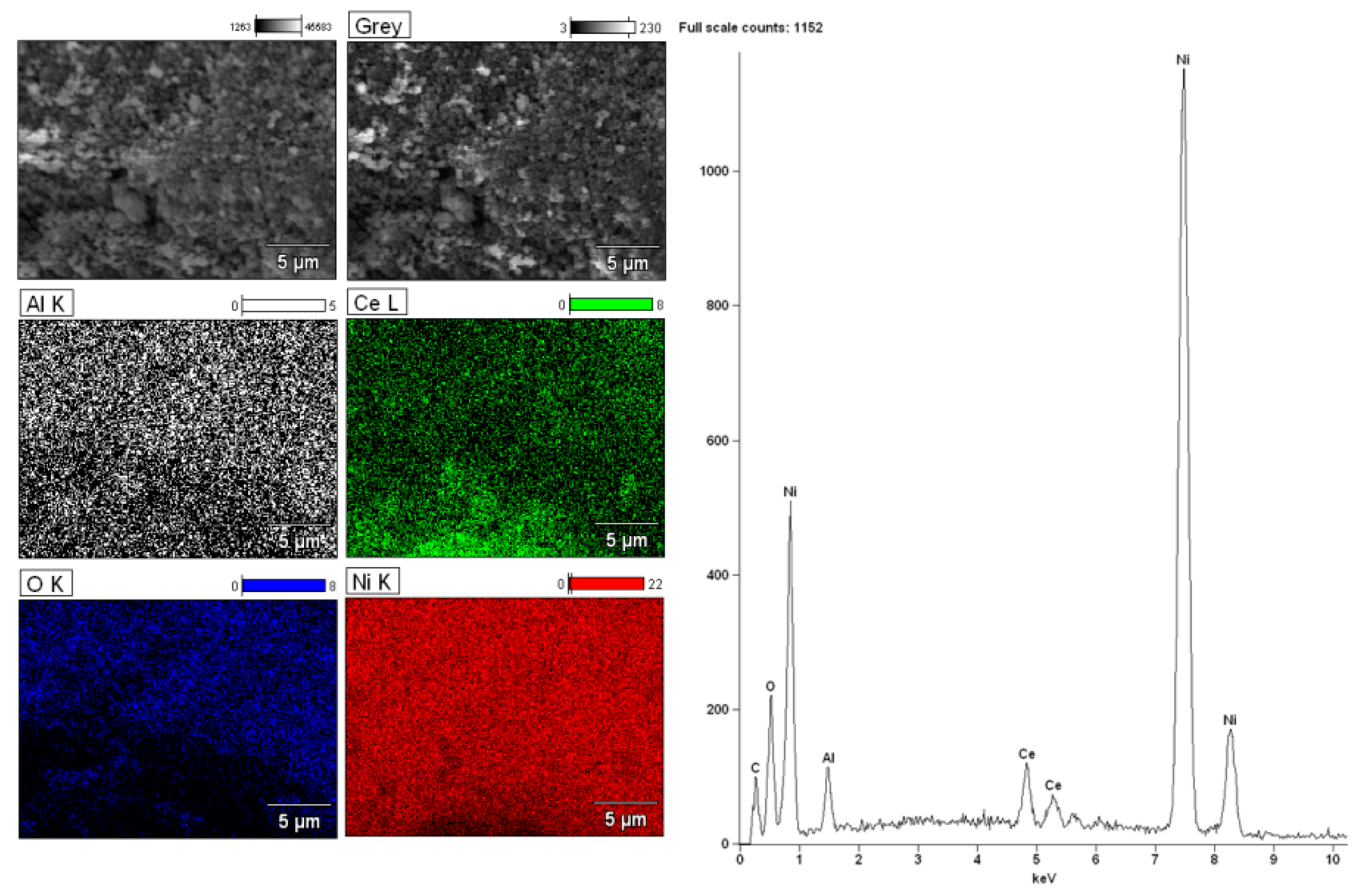
| Catalyst | CH3OH Conv. (%) | H2 Selectivity (%) | CO Selectivity (%) | CO2 Selectivity (%) | DME Selectivity (%) | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| 200 °C | 250 °C | 200 °C | 250 °C | 200 °C | 250 °C | 200 °C | 250 °C | 200 °C | 250 °C | |
| 5%Ni/CeO2–Al2O3 | - | 2 | - | 30.5 | - | - | - | 25 | - | 44.5 |
| 20%Ni/CeO2–Al2O3 | 11 | 31 | 66.5 | 33 | - | - | 26.3 | 55.6 | 7.2 | 11.4 |
| 40%Ni/CeO2–Al2O3 | 40 | 98 | 45.2 | 68.9 | 46 | 13.3 | 8.3 | 14.3 | 0.5 | 3.5 |
| 60%Ni/CeO2–Al2O3 | 18 | 77 | 61.4 | 58.2 | - | 27.7 | 38.6 | 14.1 | - | trace |
| 40%Ni/CeO2 | 4 | 85 | 75 | 68.6 | 24.2 | 22.6 | - | 0.5 | 0.8 | 8.3 |
| 40%Ni/Al2O3 | - | 20.5 | - | 53.4 | - | - | - | 25.2 | - | 21.4 |
| 2%Pd–40%Ni/CeO2–Al2O3 | 67 | 99.9 | 70.3 | 71.5 | 21.8 | 14.9 | 7.6 | 9.1 | 0.3 | 4.5 |
| Catalytic Material | BET Surface Area (m2/g) | Monolayer Capacity (cm3/g) | Metal Dispersion (%) | Metal Area (m2g−1 of Metal) | Crystallite Size (nm) | H2 Sorption (μmolH2gcat−1) | X a (%) |
|---|---|---|---|---|---|---|---|
| Al2O3 | 210 | 48.2 | - | - | - | - | - |
| CeO2 | 60 | 13.8 | - | - | - | - | - |
| eO2-Al2O3 | 145 | 33.2 | - | - | - | - | - |
| 5%Ni/CeO2–Al2O3 | 132 | 30.3 | 1.06 | 7.09 | 95.0 | 0.102 | 2 |
| 20%Ni/CeO2–Al2O3 | 128 | 29.5 | 1.16 | 7.71 | 87.4 | 0.442 | 31 |
| 40%Ni/CeO2–Al2O3 | 78 | 17.9 | 0.54 | 3.61 | 186 | 0.414 | 98 |
| 40%Ni/Al2O3 | 133 | 30.5 | 0.69 | 4.63 | 146 | 0.326 | 20.5 |
| 40%Ni/CeO2 | 34 | 7.8 | 0.63 | 4.18 | 146 | 0.479 | 85 |
| 60%Ni/CeO2–Al2O3 | 58 | 13.2 | 0.28 | 1.90 | 356 | 0.531 | 77 |
| 2%Pd–40%Ni/CeO2–Al2O3 | 42 | 9.6 | - | - | - | - | 99.9 |
| Catalysts/Supports | Weak Centres (mmol/g) 100–300 °C | Medium Centres (mmol/g) 300–450 °C | Strong Centres (mmol/g) 450–600 °C | Total Acidity (mmol/g) 100–600 °C |
|---|---|---|---|---|
| CeO2–Al2O3 | 0.13 | 0.19 | 0.27 | 0.59 |
| 5%Ni/CeO2–Al2O3 | 0.12 | 0.17 | 0.34 | 0.63 |
| 20%Ni/CeO2–Al2O3 | 0.12 | 0.16 | 0.28 | 0.56 |
| 40%Ni/CeO2–Al2O3 | 0.07 | 0.10 | 0.13 | 0.30 |
| 60%Ni/CeO2–Al2O3 | 0.09 | 0.14 | 0.18 | 0.41 |
| 40%Ni/CeO2 | 0.07 | 0.07 | 0.12 | 0.26 |
| 40%Ni/Al2O3 | 0.13 | 0.15 | 0.24 | 0.52 |
| 2%Pd–40%Ni/CeO2–Al2O3 | 0.06 | 0.09 | 0.13 | 0.28 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mierczynski, P.; Mierczynska, A.; Ciesielski, R.; Mosinska, M.; Nowosielska, M.; Czylkowska, A.; Maniukiewicz, W.; Szynkowska, M.I.; Vasilev, K. High Active and Selective Ni/CeO2–Al2O3 and Pd–Ni/CeO2–Al2O3 Catalysts for Oxy-Steam Reforming of Methanol. Catalysts 2018, 8, 380. https://doi.org/10.3390/catal8090380
Mierczynski P, Mierczynska A, Ciesielski R, Mosinska M, Nowosielska M, Czylkowska A, Maniukiewicz W, Szynkowska MI, Vasilev K. High Active and Selective Ni/CeO2–Al2O3 and Pd–Ni/CeO2–Al2O3 Catalysts for Oxy-Steam Reforming of Methanol. Catalysts. 2018; 8(9):380. https://doi.org/10.3390/catal8090380
Chicago/Turabian StyleMierczynski, Pawel, Agnieszka Mierczynska, Radoslaw Ciesielski, Magdalena Mosinska, Magdalena Nowosielska, Agnieszka Czylkowska, Waldemar Maniukiewicz, Malgorzata I. Szynkowska, and Krasimir Vasilev. 2018. "High Active and Selective Ni/CeO2–Al2O3 and Pd–Ni/CeO2–Al2O3 Catalysts for Oxy-Steam Reforming of Methanol" Catalysts 8, no. 9: 380. https://doi.org/10.3390/catal8090380
APA StyleMierczynski, P., Mierczynska, A., Ciesielski, R., Mosinska, M., Nowosielska, M., Czylkowska, A., Maniukiewicz, W., Szynkowska, M. I., & Vasilev, K. (2018). High Active and Selective Ni/CeO2–Al2O3 and Pd–Ni/CeO2–Al2O3 Catalysts for Oxy-Steam Reforming of Methanol. Catalysts, 8(9), 380. https://doi.org/10.3390/catal8090380