Ce and Zr Modified WO3-TiO2 Catalysts for Selective Catalytic Reduction of NOx by NH3
Abstract
:1. Introduction
2. Results and Discussion
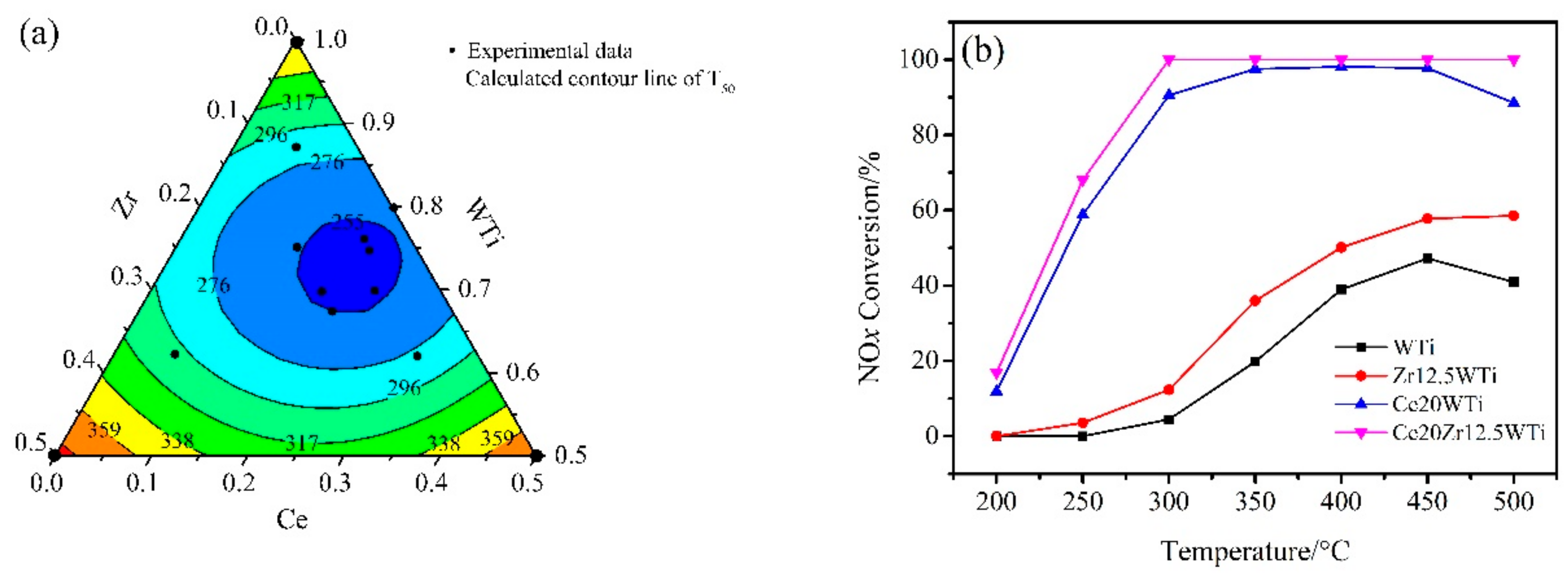
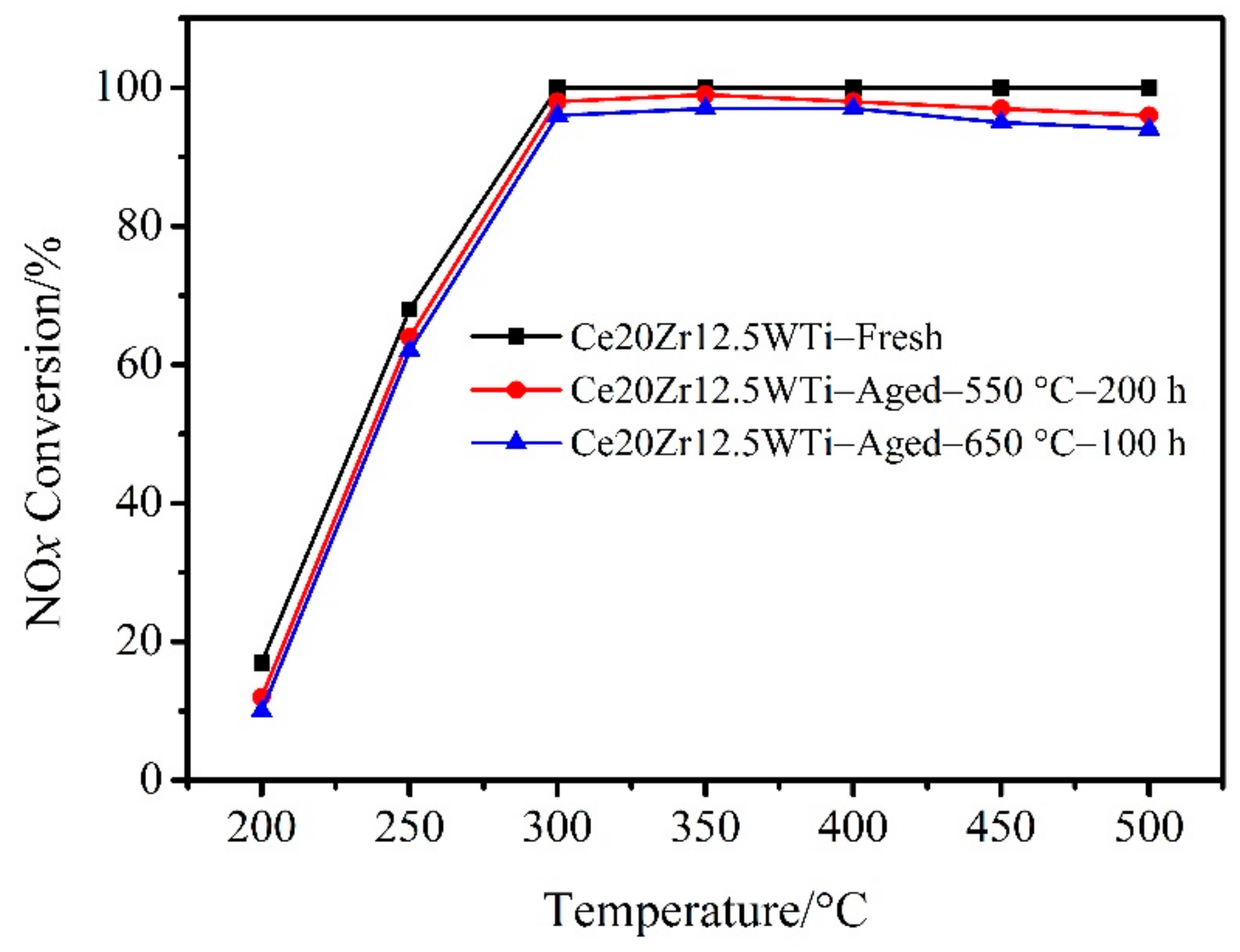
2.1. Catalytic Activity
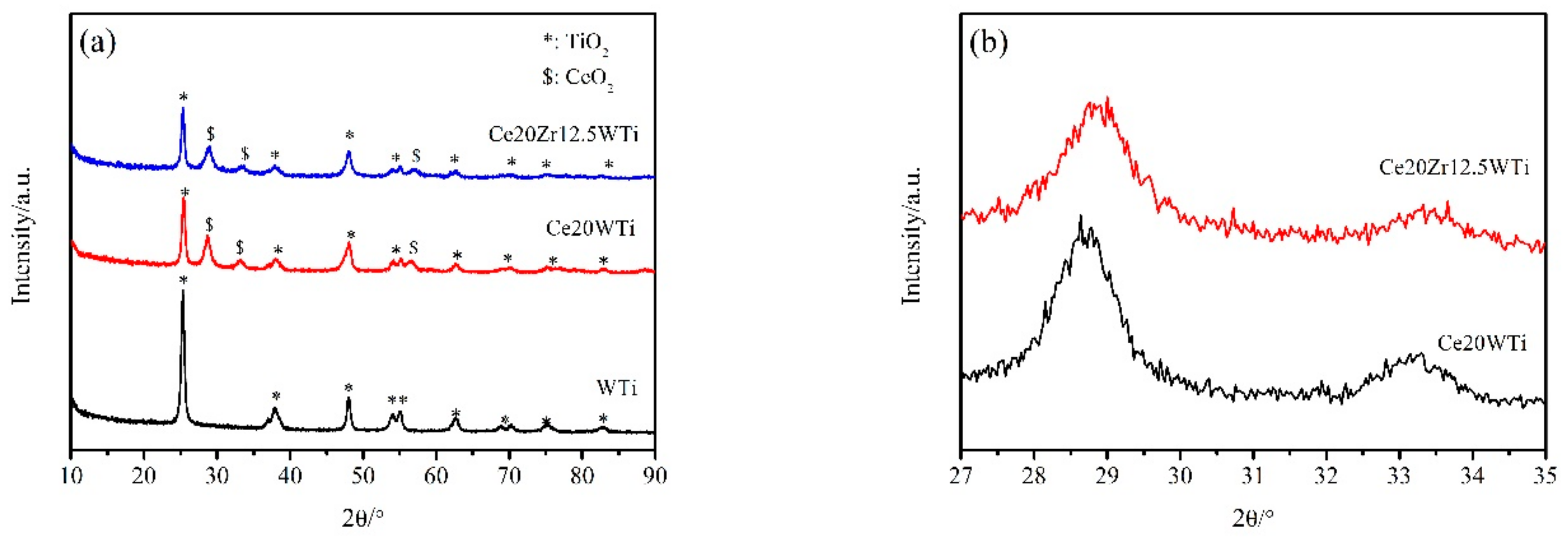
2.2. X-ray Diffraction (XRD) and Brunauer-Emmett-Teller (BET) Analysis
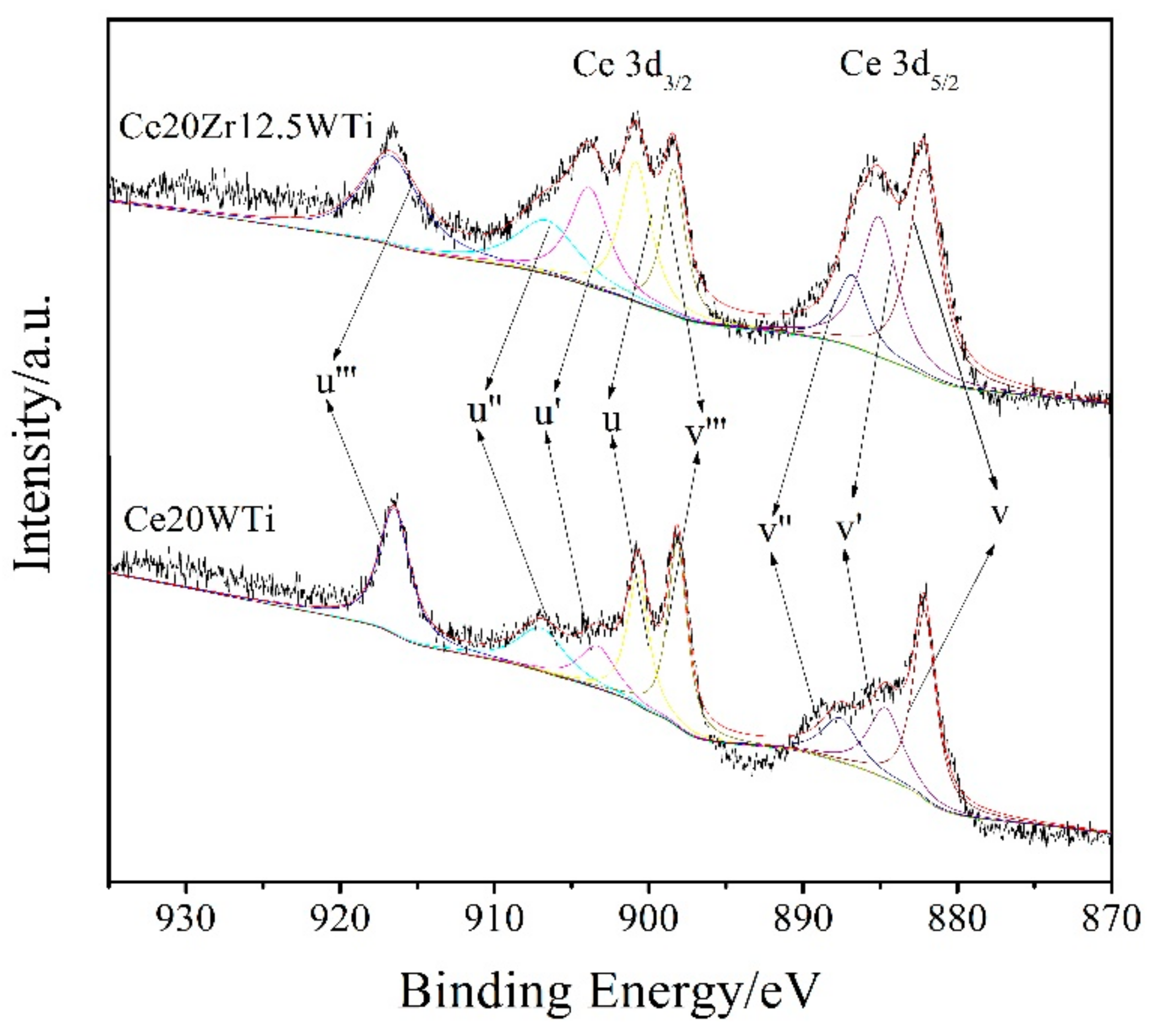
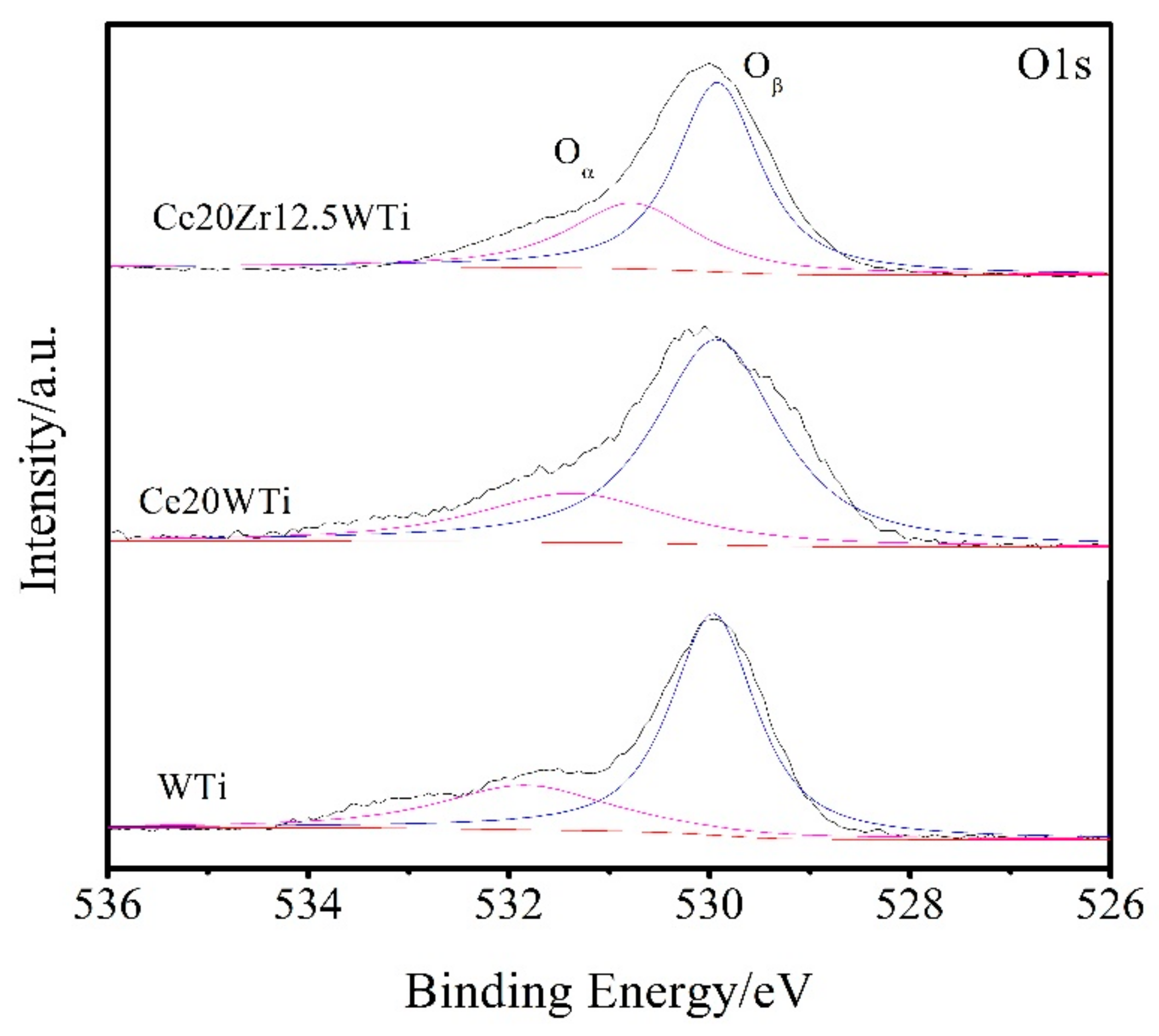
2.3. X-ray Photoelectron Spectroscopy (XPS) Results
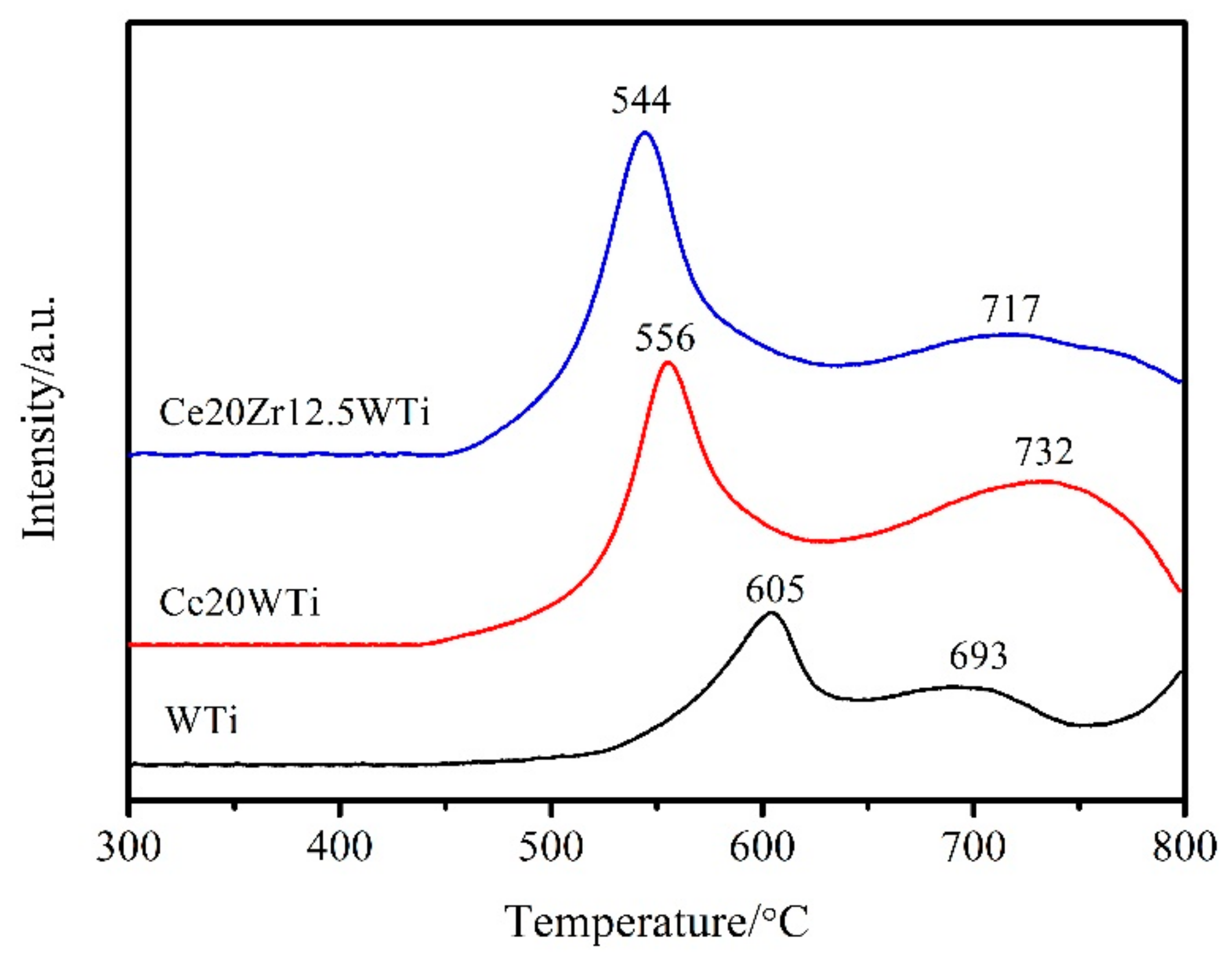
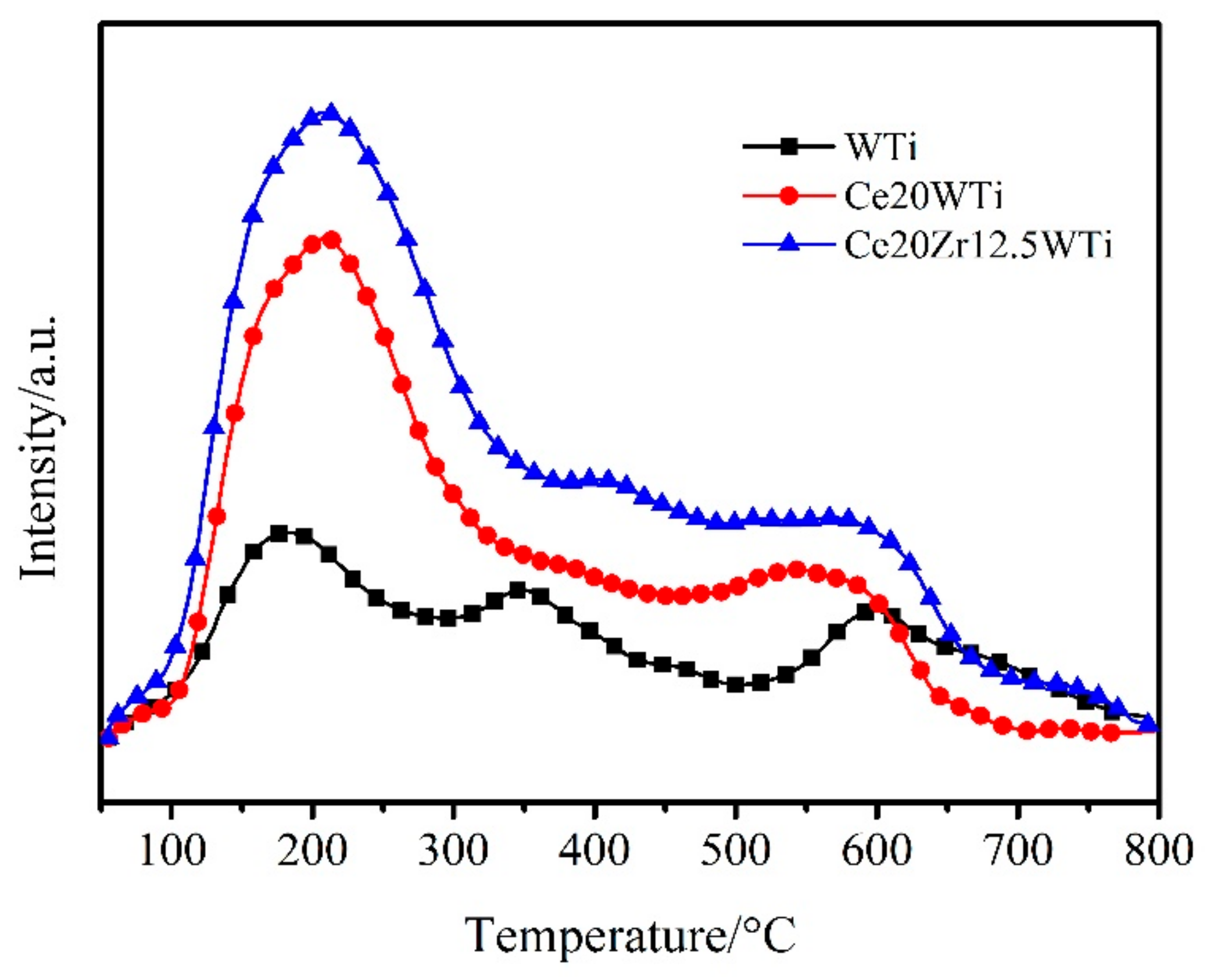
2.4.H2-Temperature Programed Reduction (H2-TPR) and NH3-Temperature Programed Desorption (NH3-TPD) Analysis
3. Materials and Methods
3.1. Catalyst Preparation
3.2. Catalyst Characterization
3.3. Activity Evaluation
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Liu, Z.; Ihl Woo, S. Recent advances in catalytic deNOx science and technology. Catal. Rev. Sci. Eng. 2006, 48, 43–89. [Google Scholar] [CrossRef]
- Roy, S.; Hegde, M.S.; Madras, G. Catalysis for NO abatement. Appl. Energy 2009, 86, 2283–2297. [Google Scholar] [CrossRef]
- Busca, G.; Lietti, L.; Ramis, G.; Berti, F. Chemical and mechanistic aspects of the selective catalytic reduction of NOx by ammonia over oxide catalysts: A review. Appl. Catal. B Environ. 1998, 18, 1–36. [Google Scholar] [CrossRef]
- Yates, M.; Martín, J.A.; Martín-Luengo, M.A.; Suárez, S.; Blanco, J. N2O formation in the ammonia oxidation and in the SCR process with V2O5-WO3 catalysts. Catal. Today 1996, 107–108, 120–125. [Google Scholar] [CrossRef]
- Reddy, B.M.; Khan, A.; Yamada, Y.; Kobayashi, T.; Loridant, S.; Volta, J.-C. Structural characterization of CeO2-TiO2 and V2O5/CeO2-TiO2 catalysts by Roman and XPS techniques. J. Phys. Chem. B 2003, 107, 5162–5167. [Google Scholar] [CrossRef]
- Guo, R.; Zhou, Y.; Pan, W.; Hong, J.; Zheng, W.; Jin, Q.; Ding, C.; Guo, S. Effect of preparation methods on the performance of CeO2/Al2O3 catalysts for selective catalytic reduction of NO with NH3. J. Ind. Eng. Chem. 2013, 19, 2022–2205. [Google Scholar] [CrossRef]
- Xu, W.; Yu, Y.; Zhang, C.; He, H. Selective catalytic reduction of NO by NH3 over a Ce/TiO2 catalyst. Catal. Commun. 2008, 9, 1453–1457. [Google Scholar] [CrossRef]
- Shen, Y.; Zhu, S.; Qiu, T.; Shen, S. A novel catalyst of CeO2/Al2O3 for selective catalytic reduction of NO by NH3. Catal. Commun. 2009, 11, 20–23. [Google Scholar] [CrossRef]
- Gao, X.; Jiang, Y.; Fu, Y.; Zhong, Y.; Luo, Z.; Cen, K. Preparation and characterization of CeO2/TiO2 catalysts for selective catalytic reduction of NO with NH3. Catal. Commun. 2010, 11, 465–469. [Google Scholar] [CrossRef]
- Shan, W.; Liu, F.; He, H.; Shi, X.; Zhang, C. An environmentally-benign CeO2-TiO2 catalyst for the selective catalytic reduction of NOx with NH3 in simulated diesel exhaust. Catal. Today 2012, 184, 160–165. [Google Scholar] [CrossRef]
- Chen, L.; Li, J.; Ge, M. DRIFT Study on Cerium-tungsten/titiania catalyst for selective catalytic reduction of NOx with NH3. Environ. Sci. Technol. 2010, 44, 9590–9596. [Google Scholar] [CrossRef] [PubMed]
- Shan, W.; Liu, F.; He, H.; Shi, X.; Zhang, C. A superior Ce-W-Ti mixed oxide catalyst for the selective catalytic reduction of NOx with NH3. Appl. Catal. B Environ. 2012, 115–116, 100–106. [Google Scholar] [CrossRef]
- Shan, W.; Liu, F.; He, H.; Shi, X.; Zhang, C. Novel cerium-tungsten mixed oxide catalyst for the selective catalytic reduction of NOx with NH3. Chem. Commun. 2011, 47, 8046–8048. [Google Scholar] [CrossRef] [PubMed]
- Ma, Z.; Weng, D.; Wu, X.; Si, Z. Effects of WOx modification on the activity, adsorption and redox properties of CeO2 catalyst for NOx reduction with ammonia. J. Environ. Sci. 2012, 24, 1305–1316. [Google Scholar] [CrossRef]
- Liu, Z.; Zhang, S.; Li, J.; Ma, L. Promoting effect of MoO3 on the NOx reduction by NH3 over CeO2/TiO2 catalyst studied with in situ DRIFTS. Appl. Catal. B Environ. 2014, 144, 90–95. [Google Scholar] [CrossRef]
- Daturi, M.; Finocchio, E.; Binet, C.; Lavalley, J.C.; Fally, F.; Perrichon, V. Study of bulk and surface reduction by hydrogen of CexZr1-xO2 mixed oxides followed by FTIR spectroscopy and magnetic balance. J. Phys. Chem. B 1999, 103, 4884–4891. [Google Scholar] [CrossRef]
- Daturi, M.; Bion, N.; Saussey, J.; Lavalley, J.-C.; Hedouin, C.; Seguelong, T.; Blanchard, G. Evidence of a lacunar mechanism for de NOx activity in ceria-based catalysts. Phys. Chem. Chem. Phys. 2001, 3, 252–255. [Google Scholar] [CrossRef]
- Vidal, H.; Kaspar, J.; Pijolat, M.; Colon, G.; Bernal, S.; Cordon, A.; Perrichon, V.; Fally, F. Redox behavior of CeO2-ZrO2 mixed oxides: I. Influence of redox treatments on high surface area catalysts. Appl. Catal. B Environ. 2000, 27, 49–63. [Google Scholar] [CrossRef]
- Gao, X.; Jiang, Y.; Zhong, Y.; Luo, Z.; Cen, K. The activity and characterization of CeO2-TiO2 catalysts prepared by the sol-gel method for selective catalytic reduction of NO with NH3. J. Hazard. Mater. 2010, 174, 734–739. [Google Scholar] [CrossRef] [PubMed]
- Burroughs, P.; Hamnett, A.; Orchard, A.F.; Thornton, G. Satellite structure in the X-ray photoelectron spectra of some binary and mixed oxides of lanthanum and cerium. J. Chem. Soc. Dalton Trans. 1976, 17, 1686–1698. [Google Scholar] [CrossRef]
- Liu, C.; Chen, L.; Chang, H.; Ma, L.; Peng, Y.; Arandiyan, H.; Li, J. Characterization of CeO2-WO3 catalysts prepared by different methods for selective catalytic reduction of NOx with NH3. Catal. Commun. 2013, 40, 145–148. [Google Scholar] [CrossRef]
- Liu, Z.; Yi, Y.; Li, J.; lhl Woo, S.; Wang, B.; Cao, X.; Li, Z. A superior catalyst with dual redox cycles for the selective reduction of NOx by ammonia. Chem. Commun. 2013, 49, 7726–7728. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Wu, C.-Y.; Li, Y.; Zhang, J. CeO2-TiO2 catalysts for catalytic oxidation of elemental mercury in low-rank coal combustion flue gas. Environ. Sci. Technol. 2011, 45, 7394–7400. [Google Scholar] [CrossRef] [PubMed]
- Cai, S.; Zhang, D.; Zhang, L.; Huang, L.; Li, H.; Gao, R.; Shi, L.; Zhang, J. Comparative study of 3D ordered macroporous Ce0.75Zr0.2M0.05O2-δ (M = Fe, Cu, Mn, Co) for selective catalytic reduction of NO with NH3. Catal. Sci. Technol. 2014, 4, 93–101. [Google Scholar] [CrossRef]
- Wu, Z.; Jin, R.; Liu, Y.; Wang, H. Ceria modified MnOx/TiO2 as a superior catalyst for NO reduction with NH3 at a low-temperature. Catal. Commun. 2008, 9, 2217–2220. [Google Scholar] [CrossRef]
- Chen, L.; Li, J.; Ge, M. Promotional effect of Ce-doped V2O5-WO3/TiO2 with low vanadium loadings for selective catalytic reduction of NOx by NH3. J. Phys. Chem. C 2009, 113, 21177–21184. [Google Scholar] [CrossRef]
- Barton, D.G.; Soled, S.L.; Meitzner, G.D.; Fuentes, G.A.; Iglesia, E. Structural and catalytic characterization of solid acids based on zirconia modified by tungsten oxide. J. Catal. 1999, 181, 57–72. [Google Scholar] [CrossRef]
- Ma, Z.; Weng, D.; Wu, X.; Si, Z.; Wang, B. A novel Nb-Ce/WOx-TiO2 catalyst with high NH3-SCR activity and stability. Catal. Commun. 2012, 27, 97–100. [Google Scholar] [CrossRef]
- Geng, Y.; He, H.; Chen, X.; Ding, H.; Yang, S.; Liu, F.; Shan, W. The effect of Ce on a high-efficiency CeO2/WO3-TiO2 catalyst for the selective catalytic reduction of NOx with NH3. RSC Adv. 2016, 6, 64803–64810. [Google Scholar] [CrossRef]
- Zhao, W.; Tang, Y.; Wan, Y.; Li, L.; Yao, S.; Li, X.; Gu, J.; Li, Y.; Shi, J. Promotion effects of SiO2 or/and Al2O3 doped CeO2/TiO2 catalysts for selective catalytic reduction of NO by NH3. J Hazard. Mater. 2014, 278, 350–359. [Google Scholar] [CrossRef] [PubMed]
| Catalysts | BET Surface Area (m2·g−1) | Total Pore Volume (cm3·g−1) | Average Pore Diameter (nm) |
|---|---|---|---|
| WTi | 80.32 | 0.2874 | 125.3 |
| Ce20WTi | 46.93 | 0.1685 | 109.0 |
| Ce20Zr12.5WTi | 58.34 | 0.1513 | 86.04 |
| Catalysts | Ce3+/(Ce3+ + Ce4+) | Oα/(Oα + Oβ) |
|---|---|---|
| WTi | – | 23.2% |
| Ce20WTi | 19.1% | 28.8% |
| Ce20Zr12.5WTi | 26.5% | 35.4% |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhao, W.; Li, Z.; Wang, Y.; Fan, R.; Zhang, C.; Wang, Y.; Guo, X.; Wang, R.; Zhang, S. Ce and Zr Modified WO3-TiO2 Catalysts for Selective Catalytic Reduction of NOx by NH3. Catalysts 2018, 8, 375. https://doi.org/10.3390/catal8090375
Zhao W, Li Z, Wang Y, Fan R, Zhang C, Wang Y, Guo X, Wang R, Zhang S. Ce and Zr Modified WO3-TiO2 Catalysts for Selective Catalytic Reduction of NOx by NH3. Catalysts. 2018; 8(9):375. https://doi.org/10.3390/catal8090375
Chicago/Turabian StyleZhao, Wenyi, Zhaoqiang Li, Yan Wang, Rongrong Fan, Cheng Zhang, Yu Wang, Xin Guo, Rong Wang, and Shule Zhang. 2018. "Ce and Zr Modified WO3-TiO2 Catalysts for Selective Catalytic Reduction of NOx by NH3" Catalysts 8, no. 9: 375. https://doi.org/10.3390/catal8090375
APA StyleZhao, W., Li, Z., Wang, Y., Fan, R., Zhang, C., Wang, Y., Guo, X., Wang, R., & Zhang, S. (2018). Ce and Zr Modified WO3-TiO2 Catalysts for Selective Catalytic Reduction of NOx by NH3. Catalysts, 8(9), 375. https://doi.org/10.3390/catal8090375




