Synthesis of Sulfur-Resistant TiO2-CeO2 Composite and Its Catalytic Performance in the Oxidation of a Soluble Organic Fraction from Diesel Exhaust
Abstract
1. Introduction
2. Results and Discussion
2.1. Sulfur Resistibility
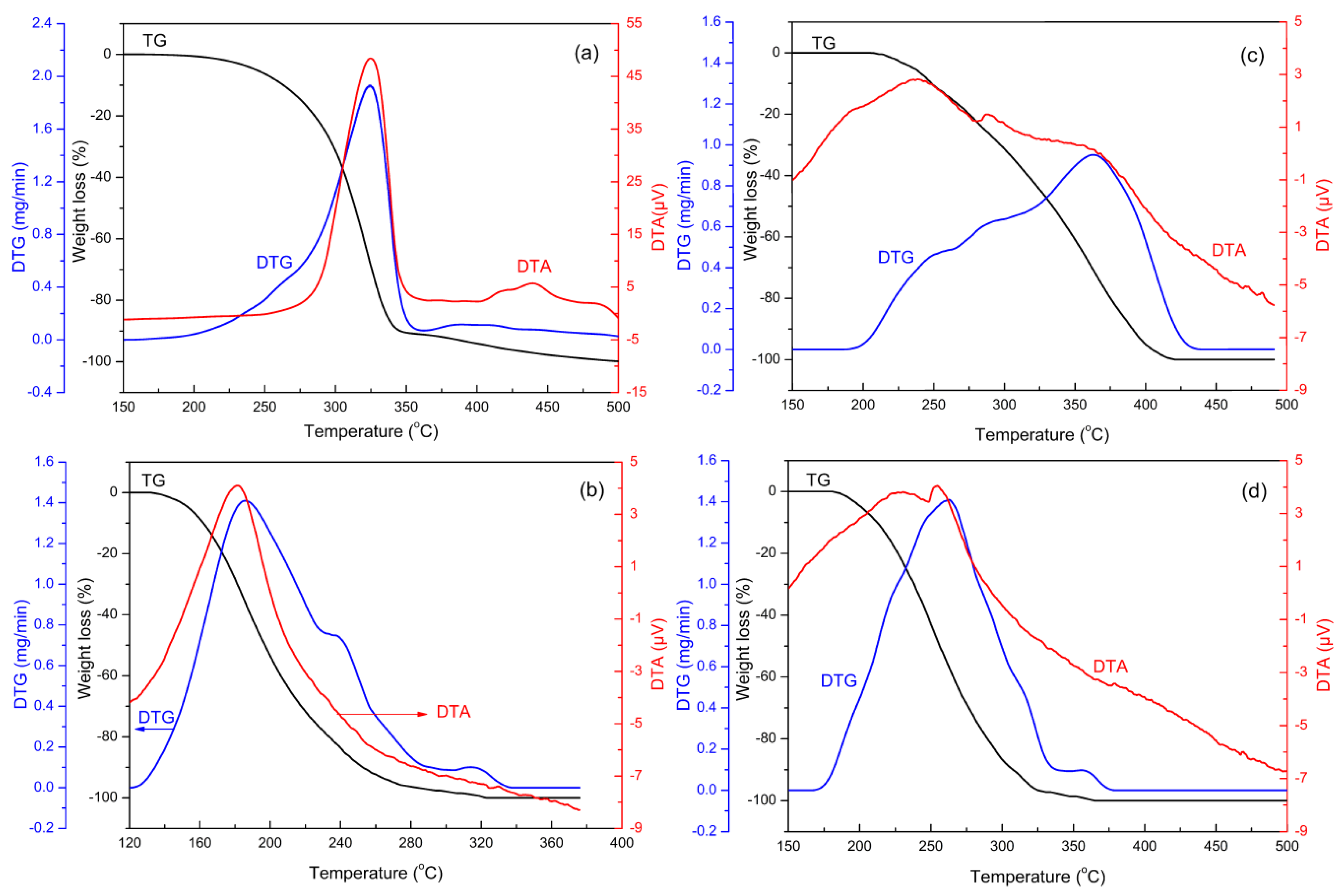
2.2. Catalytic Performance
2.3. Catalyst Characterization
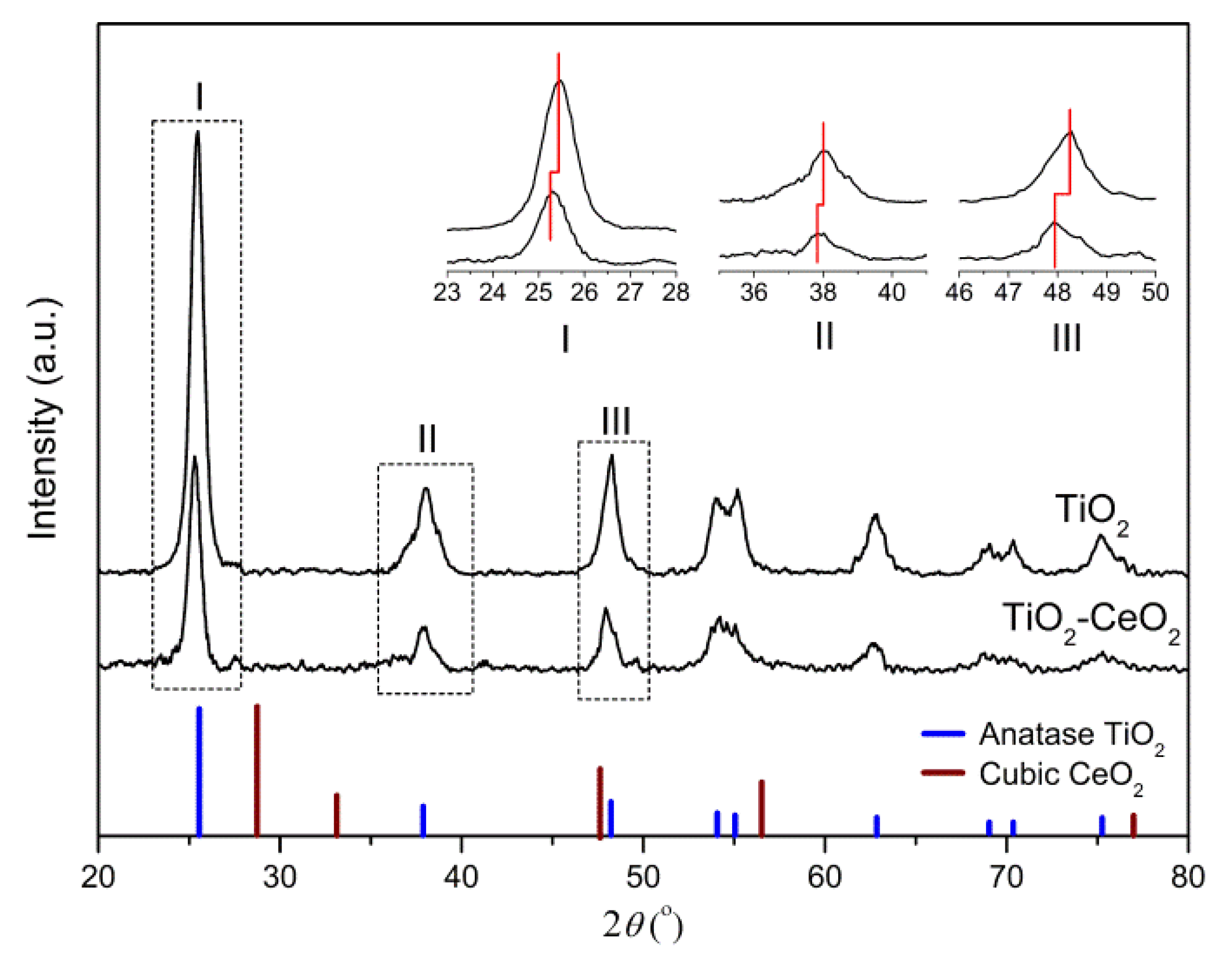
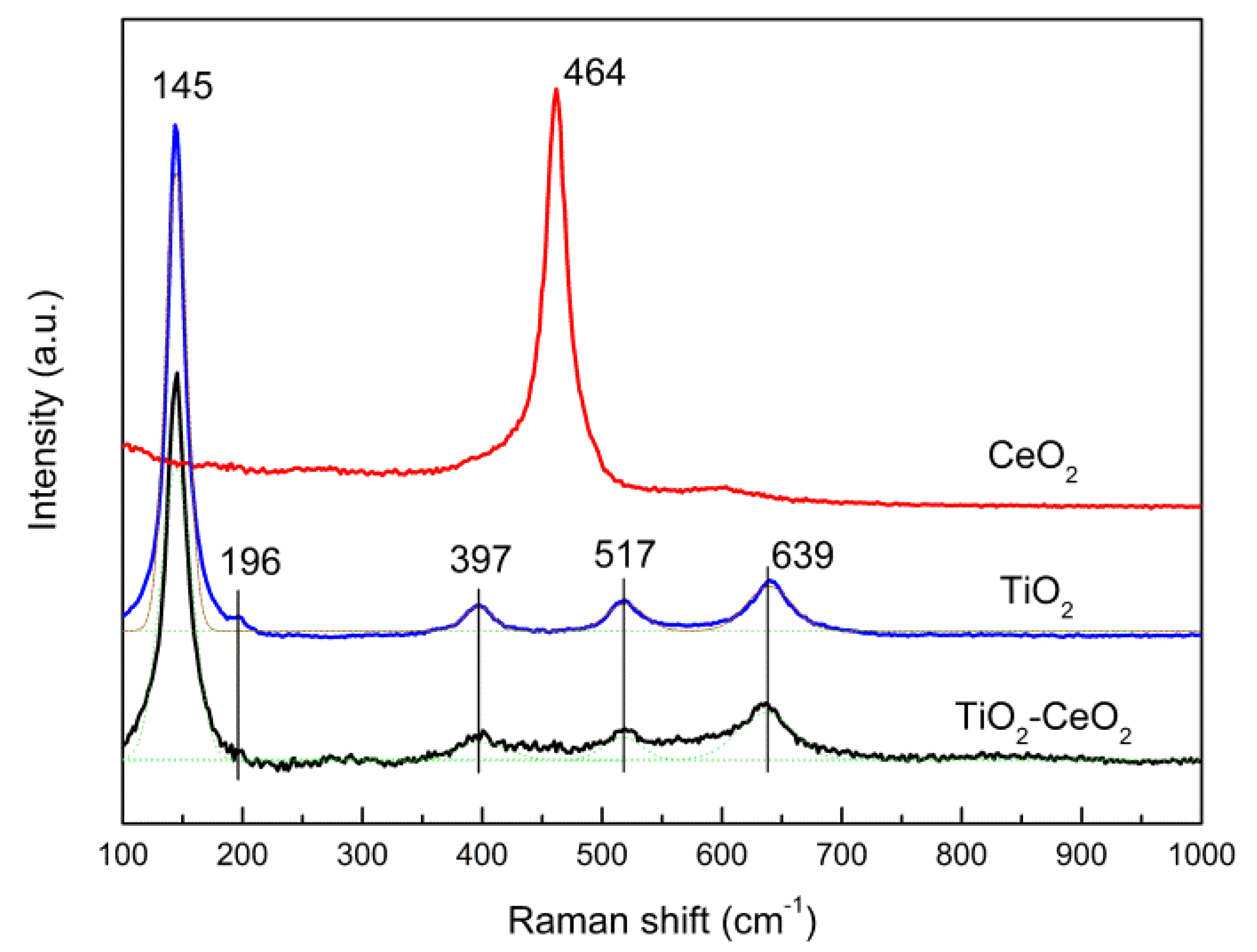
2.3.1. XRD and Raman Spectra
2.3.2. Nitrogen Adsorption-Desorption
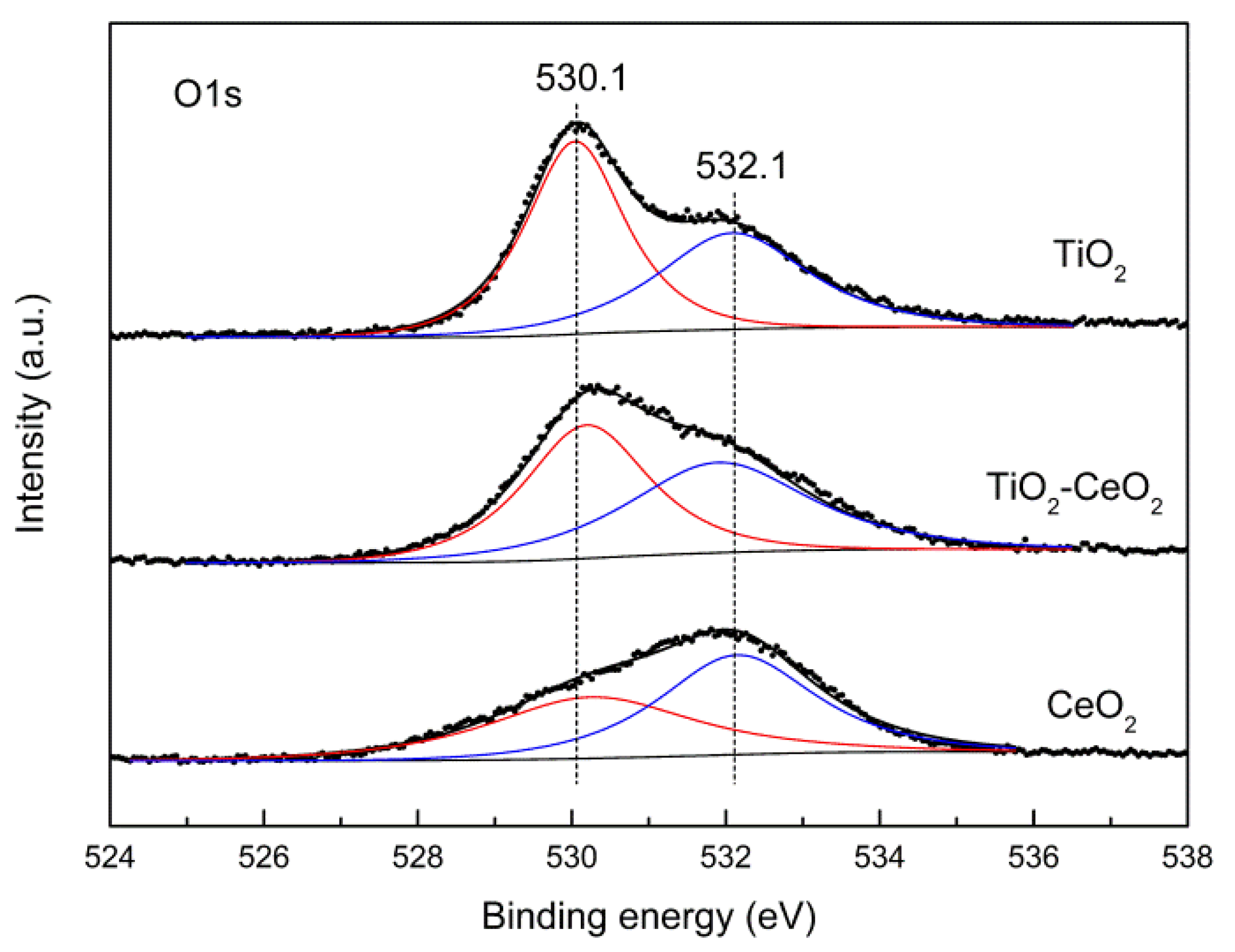
2.3.3. XPS
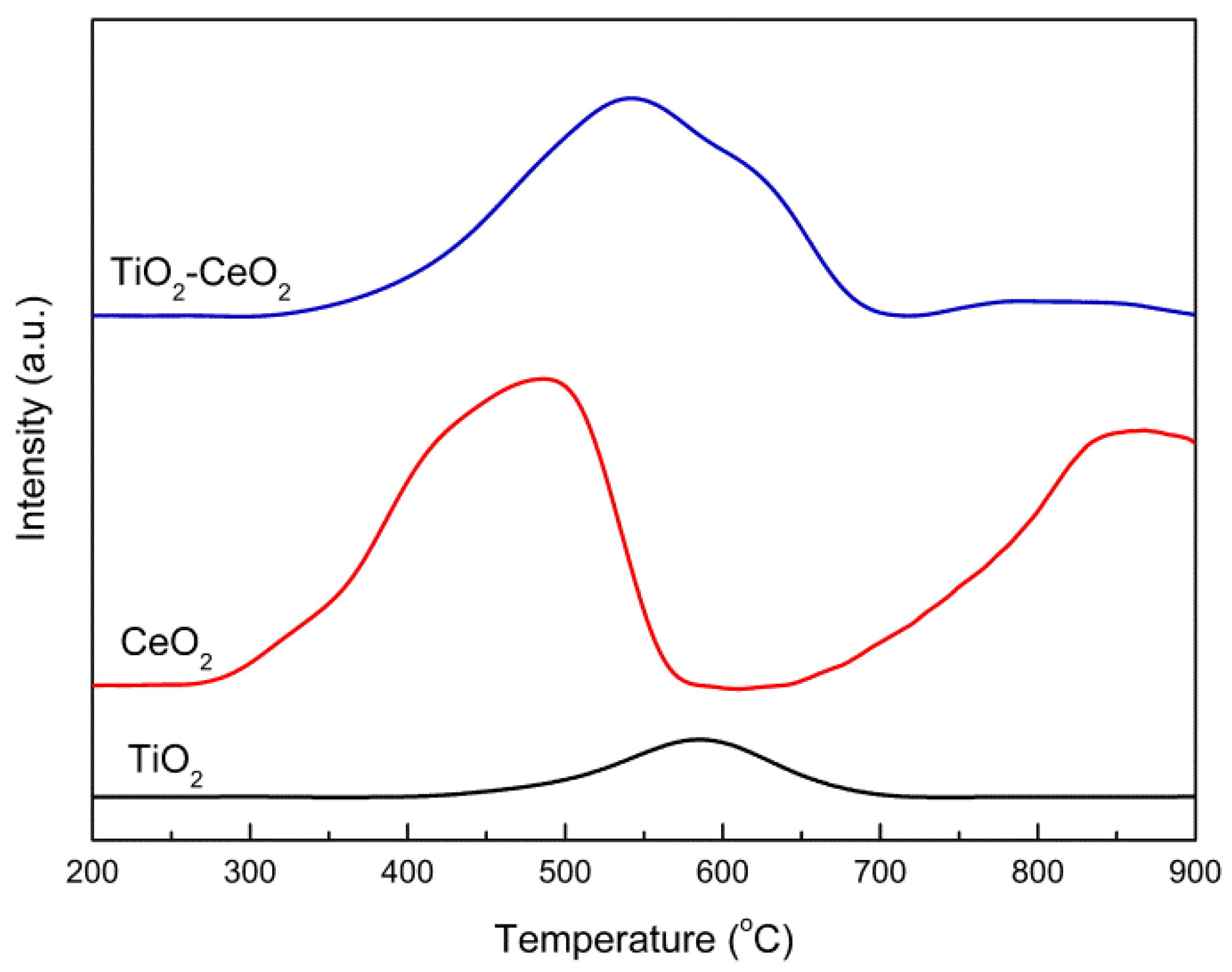
2.3.4. H2-TPR
2.3.5. OSC
3. Materials and Methods
3.1. Catalyst Preparation
3.2. Catalyst Evaluation
3.3. Catalyst Characterization
4. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Johnson, T.; Joshi, A. Review of Vehicle Efficiency and Emissions; SAE Technical Paper, 2017-01-0907; SAE: Warrendale, PA, USA, 2017. [Google Scholar]
- Wang, X.; Westermann, A.; Shi, Y.; Cai, N.; Rieu, M.; Viricelle, J.-P.; Vernoux, P. Electrochemical Removal of NOx on Ceria-Based Catalyst-Electrodes. Catalysts 2017, 7, 61. [Google Scholar] [CrossRef]
- Godoy, M.; Banús, E.; Sanz, O.; Montes, M.; Miró, E.; Milt, V. Stacked Wire Mesh Monoliths for the Simultaneous Abatement of VOCs and Diesel Soot. Catalysts 2018, 8, 16. [Google Scholar] [CrossRef]
- Williams, P.T.; Abbass, M.K.; Tarn, L.P.; Andrews, G.E.; Ng, K.L.; Bartle, K.D. A Comparison of Exhaust Pipe, Dilution Tunnel and Roadside Diesel Particulate SOF and Gaseous Hydrocarbon Emissions; SAE Technical Paper, 880351; SAE: Warrendale, PA, USA, 1988. [Google Scholar]
- Osada, H.; Aoyagi, Y.; Shimada, K.; Akiyama, K.; Goto, Y.; Suzuki, H. SOF Component of Lubricant Oil on Diesel PM in a High Boosted and Cooled EGR Engine; SAE Technical Paper, 2007-01-0123; SAE: Warrendale, PA, USA, 2007. [Google Scholar]
- Collura, S.; Chaoui, N.; Azambre, B.; Finqueneisel, G.; Heintz, O.; Krzton, A.; Koch, A.; Weber, J. Influence of the soluble organic fraction on the thermal behaviour, texture and surface chemistry of diesel exhaust soot. Carbon 2005, 43, 605–613. [Google Scholar] [CrossRef]
- Atribak, I.; Bueno-López, A.; García-García, A. Uncatalysed and catalysed soot combustion under NOx + O2: Real diesel versus model soots. Combust. Flame 2010, 157, 2086–2094. [Google Scholar] [CrossRef]
- Farrauto, R.J.; Mooney, J.J. Effects of Sulfur on Performance of Catalytic Aftertreatment Devices; SAE Technical Paper, 920557; SAE: Warrendale, PA, USA, 1992. [Google Scholar]
- Spezzano, P.; Picini, P.; Cataldi, D. Gas-and particle-phase distribution of polycyclic aromatic hydrocarbons in two-stroke, 50-cm3 moped emissions. Atmos. Environ. 2009, 43, 539–545. [Google Scholar] [CrossRef]
- Stanmore, B.R.; Brilhac, J.F.; Gilot, P. The oxidation of soot: A review of experiments, mechanisms and models. Carbon 2001, 39, 2247–2268. [Google Scholar] [CrossRef]
- Clague, A.D.H.; Donnet, J.B.; Wang, T.K.; Peng, J.C.M. A comparison of diesel engine soot with carbon black1. Carbon 1999, 37, 1553–1565. [Google Scholar] [CrossRef]
- Westerholm, R.; Christensen, A.; de Serves, C.; Almén, J. Determination of Polycyclic Aromatic Hydrocarbons (PAH) in Size Fractionated Diesel Particles from a Light Duty Vehicle; SAE Technical Paper, 1999-01-3533; SAE: Warrendale, PA, USA, 1999. [Google Scholar]
- Hansen, K.F.; Jensen, M.G. Chemical and Biological Characteristics of Exhaust Emissions from a DI Diesel Engine Fuelled with Rapeseed Oil Methyl Ester (RME); SAE Technical Paper, 971689; SAE: Warrendale, PA, USA, 1997. [Google Scholar]
- Callén, M.; Iturmendi, A.; López, J.; Mastral, A. Source apportionment of the carcinogenic potential of polycyclic aromatic hydrocarbons (PAH) associated to airborne PM10 by a PMF model. Environ. Sci. Pollut. Res. 2014, 21, 2064–2076. [Google Scholar] [CrossRef] [PubMed]
- Benbrahim-Tallaa, L.; Baan, R.A.; Grosse, Y.; Lauby-Secretan, B.; el Ghissassi, F.; Bouvard, V.; Guha, N.; Loomis, D.; Straif, K. Carcinogenicity of diesel-engine and gasoline-engine exhausts and some nitroarenes. Lancet Oncol. 2012, 13, 663–664. [Google Scholar] [CrossRef]
- Farrauto, R.J.; Voss, K.E. Monolithic diesel oxidation catalysts. Appl. Catal. B Environ. 1996, 10, 29–51. [Google Scholar] [CrossRef]
- Cao, Y.; Lan, L.; Feng, X.; Yang, Z.; Zou, S.; Xu, H.; Li, Z.; Gong, M.; Chen, Y. Cerium promotion on the hydrocarbon resistance of a Cu-SAPO-34 NH3-SCR monolith catalyst. Catal. Sci. Technol. 2015, 5, 4511–4521. [Google Scholar] [CrossRef]
- Huang, Y.-F.; Zhang, H.-L.; Yang, Z.-Z.; Zhao, M.; Huang, M.-L.; Liang, Y.-L.; Wang, J.-L.; Chen, Y.-Q. Effects of CeO2 Addition on Improved NO Oxidation Activities of Pt/SiO2-Al2O3 Diesel Oxidation Catalysts. Acta Phys.-Chim. Sin. 2017, 33, 1242–1252. [Google Scholar]
- Sudarsanam, P.; Hillary, B.; Amin, M.H.; Rockstroh, N.; Bentrup, U.; Bruckner, A.; Bhargava, S.K. Heterostructured Copper-Ceria and Iron-Ceria Nanorods: Role of Morphology, Redox, and Acid Properties in Catalytic Diesel Soot Combustion. Langmuir 2018, 34, 2663–2673. [Google Scholar] [CrossRef] [PubMed]
- Putla, S.; Amin, M.H.; Reddy, B.M.; Nafady, A.; Al Farhan, K.A.; Bhargava, S.K. MnOx Nanoparticle-Dispersed CeO2 Nanocubes: A Remarkable Heteronanostructured System with Unusual Structural Characteristics and Superior Catalytic Performance. ACS Appl. Mater. Interfaces 2015, 7, 16525–16535. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.-Z.; Yang, Y.; Zhao, M.; Gong, M.-C.; Chen, Y.-Q. Enhanced Sulfur Resistance of Pt-Pd/CeO2-ZrO2-Al2O3 Commercial Diesel Oxidation Catalyst by SiO2 Surface Cladding. Acta Phys.-Chim. Sin. 2014, 30, 1187–1193. [Google Scholar]
- Kolli, T.; Kanerva, T.; Lappalainen, P.; Huuhtanen, M.; Vippola, M.; Kinnunen, T.; Kallinen, K.; Lepisto, T.; Lahtinen, J.; Keiski, R.L. The Effect of SO2 and H2O on the Activity of Pd/CeO2 and Pd/Zr-CeO2 Diesel Oxidation Catalysts. Top. Catal. 2009, 52, 2025–2028. [Google Scholar] [CrossRef]
- Luo, T.; Gorte, R.J. Characterization of SO2-poisoned ceria-zirconia mixed oxides. Appl. Catal. B Environ. 2004, 53, 77–85. [Google Scholar] [CrossRef]
- Deshmukh, S.S.; Zhang, M.; Kovalchuk, V.I.; d’Itri, J.L. Effect of SO2 on CO and C3H6 oxidation over CeO2 and Ce0.75Zr0.25O2. Appl. Catal. B Environ. 2003, 45, 135–145. [Google Scholar] [CrossRef]
- Xia, Z.; Fu, J.; Duan, A.; Han, L.; Wu, H.; Zhao, Z.; Xu, C.; Wang, D.; Wang, B.; Meng, Q. Post Synthesis of Aluminum Modified Mesoporous TUD-1 Materials and Their Application for FCC Diesel Hydrodesulfurization Catalysts. Catalysts 2017, 7, 141. [Google Scholar] [CrossRef]
- Zhang, M.H.; Fan, J.Y.; Chi, K.; Duan, A.J.; Zhao, Z.; Meng, X.L.; Zhang, H.L. Synthesis, characterization, and catalytic performance of NiMo catalysts supported on different crystal alumina materials in the hydrodesulfurization of diesel. Fuel Process. Technol. 2017, 156, 446–453. [Google Scholar] [CrossRef]
- Zhang, K.; Hu, J.; Gao, S.; Liu, Y.; Huang, X.; Bao, X. Sulfur content of gasoline and diesel fuels in northern China. Energy Policy 2010, 38, 2934–2940. [Google Scholar] [CrossRef]
- Corro, G. Sulfur impact on diesel emission control—A review. React. Kinet. Catal Lett. 2002, 75, 89–106. [Google Scholar] [CrossRef]
- Zelenka, P.; Ostgathe, K.; Lox, E. Reduction of Diesel Exhaust Emissions by Using Oxidation Catalysts; SAE Technical Paper, 902111; SAE: Warrendale, PA, USA, 1990. [Google Scholar]
- Li, J.; Kumar, A.; Chen, X.; Currier, N.; Yezerets, A. Impact of Different Forms of Sulfur Poisoning on Diesel Oxidation Catalyst Performance; SAE Technical Paper, 2013-01-0514; SAE: Warrendale, PA, USA, 2013. [Google Scholar]
- Paulson, T.; Moss, B.; Todd, B.; Eckstein, C.; Wise, B.; Singleton, D.; Zemskova, S.; Silver, R. New Developments in Diesel Oxidation Catalysts; SAE Technical Paper, 2008-01-2638; SAE: Warrendale, PA, USA, 2008. [Google Scholar]
- Kanno, Y.; Hihara, T.; Watanabe, T.; Katoh, K. Low Sulfate Generation Diesel Oxidation Catalyst; SAE Technical Paper, 2004-01-1427; SAE: Warrendale, PA, USA, 2004. [Google Scholar]
- Ueno, H.; Furutani, T.; Nagami, T.; Aono, N.; Goshima, H.; Kasahara, K. Development of Catalyst for Diesel Engine; SAE Technical Paper, 980195; SAE: Warrendale, PA, USA, 1998. [Google Scholar]
- Yang, Z.; Li, J.; Zhang, H.; Yang, Y.; Gong, M.; Chen, Y. Size-dependent CO and propylene oxidation activities of platinum nanoparticles on the monolithic Pt/TiO2-YOx diesel oxidation catalyst under simulative diesel exhaust conditions. Catal. Sci. Technol. 2015, 5, 2358–2365. [Google Scholar] [CrossRef]
- Yang, Z.; Zhang, N.; Cao, Y.; Gong, M.; Zhao, M.; Chen, Y. Effect of yttria in Pt/TiO2 on sulfur resistance diesel oxidation catalysts: Enhancement of low-temperature activity and stability. Catal. Sci. Technol. 2014, 4, 3032–3043. [Google Scholar] [CrossRef]
- Yang, Z.; Chen, Y.; Zhao, M.; Zhou, J.; Gong, M.; Chen, Y. Preparation and Properties of Pt/ZrxTi1−xO2 Catalysts with Low-Level SO2 Oxidation Activity for Diesel Oxidation, Cuihua Xuebao. Chin. J. Catal. 2012, 33, 819–826. (In Chinese) [Google Scholar]
- Yang, Z.; Zhang, N.; Cao, Y.; Li, Y.; Liao, Y.; Li, Y.; Gong, M.; Chen, Y. Promotional effect of lanthana on the high-temperature thermal stability of Pt/TiO2 sulfur-resistant diesel oxidation catalysts. RSC Adv. 2017, 7, 19318–19329. [Google Scholar] [CrossRef]
- Koranne, M.M.; Pryor, J.N. Sulfur Tolerant Alumina Catalyst Support. U.S. Patent 8,158,257, 17 April 2012. [Google Scholar]
- Zhang, Z.; Zhang, Y.; Mu, Z.; Yu, P.; Ni, X.; Wang, S.; Zheng, L. Synthesis and catalytic properties of Ce0.6Zr0.4O2 solid solutions in the oxidation of soluble organic fraction from diesel engines. Appl. Catal. B Environ. 2007, 76, 335–347. [Google Scholar] [CrossRef]
- Yang, Y.; Yang, Z.Z.; Xu, H.D.; Xu, B.Q.; Zhang, Y.H.; Gong, M.C.; Chen, Y.Q. Influence of La on CeO2-ZrO2 Catalyst for Oxidation of Soluble Organic Fraction from Diesel Exhaust. Acta Phys.-Chim. Sin. 2015, 31, 2358–2365. [Google Scholar]
- Chen, Y.-D.; Wang, L.; Guan, X.-X.; Liu, Y.-B.; Gong, M.-C.; Chen, Y.-Q. Catalytic Oxidation of Soluble Organic Fraction in Diesel Exhausts Using Composite Oxides (CeO2)x(La-Al2O3)1−x. Acta Phys.-Chim. Sin. 2013, 29, 1048–1054. [Google Scholar]
- Fornasiero, P.; Balducci, G.; di Monte, R.; Kašpar, J.; Sergo, V.; Gubitosa, G.; Ferrero, A.; Graziani, M. Modification of the Redox Behaviour of CeO2 Induced by Structural Doping with ZrO2. J. Catal. 1996, 164, 173–183. [Google Scholar] [CrossRef]
- Balachandran, U.; Eror, N.G. Raman spectra of titanium dioxide. J. Solid State Chem. 1982, 42, 276–282. [Google Scholar] [CrossRef]
- Sellick, D.R.; Aranda, A.; García, T.; López, J.M.; Solsona, B.; Mastral, A.M.; Morgan, D.J.; Carley, A.F.; Taylor, S.H. Influence of the preparation method on the activity of ceria zirconia mixed oxides for naphthalene total oxidation. Appl. Catal. B Environ. 2013, 132–133, 98–106. [Google Scholar] [CrossRef]
- Xu, H.; Cao, Y.; Wang, Y.; Fang, Z.; Lin, T.; Gong, M.; Chen, Y. The influence of molar ratios of Ce/Zr on the selective catalytic reduction of NOx with NH3 over Fe2O3-WO3/CexZr1−xO2 (0 ≤ x ≤ 1) monolith catalyst. Chin. Sci. Bull. 2014, 59, 3956–3965. [Google Scholar] [CrossRef]
- Wu, Z.; Jin, R.; Liu, Y.; Wang, H. Ceria modified MnOx/TiO2 as a superior catalyst for NO reduction with NH3 at low-temperature. Catal. Commun. 2008, 9, 2217–2220. [Google Scholar] [CrossRef]
- Zhu, H.; Qin, Z.; Shan, W.; Shen, W.; Wang, J. Pd/CeO2–TiO2 catalyst for CO oxidation at low temperature: A TPR study with H2 and CO as reducing agents. J. Catal. 2004, 225, 267–277. [Google Scholar] [CrossRef]
- Watanabe, S.; Ma, X.; Song, C. Characterization of Structural and Surface Properties of Nanocrystalline TiO2−CeO2 Mixed Oxides by XRD, XPS, TPR, and TPD. J. Phys. Chem. C 2009, 113, 14249–14257. [Google Scholar] [CrossRef]
- Guo, J.; Shi, Z.; Wu, D.; Yin, H.; Gong, M.; Chen, Y. Effects of Nd on the properties of CeO2–ZrO2 and catalytic activities of three-way catalysts with low Pt and Rh. J. Alloy. Compd. 2015, 621, 104–115. [Google Scholar] [CrossRef]
- Andersson, J.; Antonsson, M.; Eurenius, L.; Olsson, E.; Skoglundh, M. Deactivation of diesel oxidation catalysts: Vehicle- and synthetic aging correlations. Appl. Catal. B Environ. 2007, 72, 71–81. [Google Scholar] [CrossRef]
- Voss, K.E.; Lampert, J.K.; Farrauto, R.J.; Rice, G.W.; Punke, A.; Krohn, R. Catalytic oxidation of diesel particulates with base metal oxides. Stud. Surf. Sci. Catal. 1995, 96, 499–515. [Google Scholar]
- Koranne, M.M.; Pryor, J.N.; Chapman, D.M.; Brezny, R. Sulfur Tolerant Alumina Catalyst Support. U.S. Patent 8,076,263, 13 December 2011. [Google Scholar]
- Yang, Z.; Li, Y.; Liao, Y.; Li, Y.; Zhang, N. Preparation and properties of the Pt/SiO2-Al2O3 sulfur resistance diesel oxidation catalyst. Environ. Chem. 2016, 35, 1682–1689. (In Chinese) [Google Scholar]
- Lan, L.; Chen, S.; Cao, Y.; Gong, M.; Chen, Y. New insights into the structure of a CeO2-ZrO2-Al2O3 composite and its influence on the performance of the supported Pd-only three-way catalyst. Catal. Sci. Technol. 2015, 5, 4488–4500. [Google Scholar] [CrossRef]
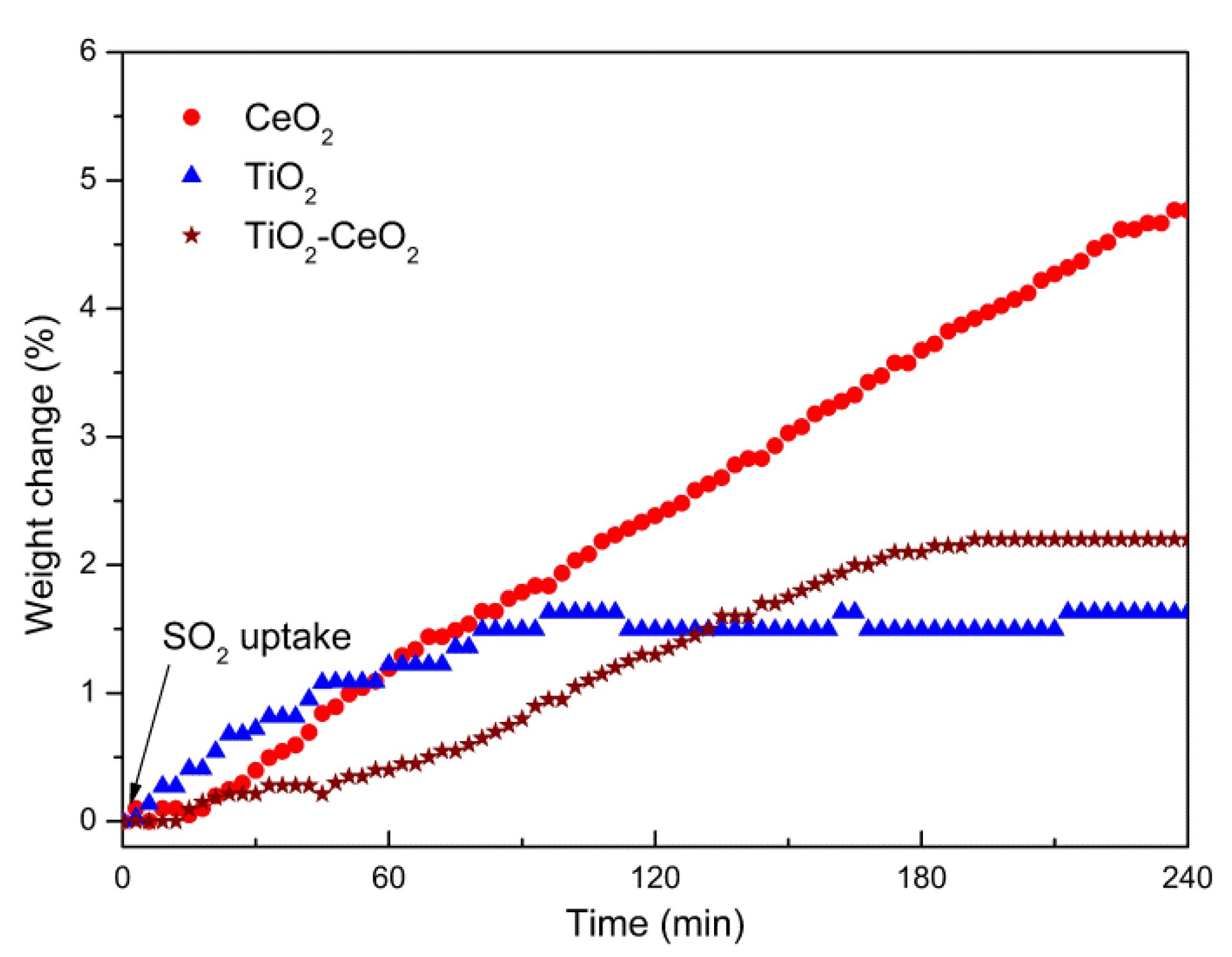

| Samples | Surface Area a (m2/g) | Sulfur Content (wt. %) | Normalized Sulfur Uptake c (µg/m2) | |
|---|---|---|---|---|
| Sulfur Uptake | EDX b | |||
| TiO2 | 98 | 1.63 | 1.02 | 166 |
| TiO2-CeO2 | 118 | 2.01 | 1.04 | 170 |
| CeO2 | 53 | 4.72 | 4.26 | 891 |
| Samples | T10% (°C) | Tm (°C) | Tf (°C) | Exothermal Peak Area (µV·°C/mg Lube) | Exothermal Peak Area (µV·°C/(mg Lube)/(mg Sample)) |
|---|---|---|---|---|---|
| lube | 264 | 324 | 507 | 283.9 | - |
| lube/CeO2 | 162 | 186 | 322 | 1068.1 | 119.5 |
| lube/TiO2 | 249 | - a | 420 | 901.6 | 102.6 |
| lube/TiO2-CeO2 | 212 | 261 | 362 | 1029.5 | 115.5 |
| Samples | Surface Area (m2/g) | Pore Volume (cm3/g) | Average Pore Diameter (nm) |
|---|---|---|---|
| TiO2 | 98 | 0.22 | 7.2 |
| TiO2-CeO2 | 118 | 0.26 | 8.6 |
| Samples | OSC (µmol O2/g Sample) | Normalized OSC (µmol O2/g CeO2) |
|---|---|---|
| TiO2 | 2.9 | - |
| TiO2-CeO2 a | 101 | 524 |
| CeO2 | 73 | 73 |
| Ce0.35Zr0.60Nd0.05O2 | 269 [49] | 638 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, N.; Yang, Z.; Chen, Z.; Li, Y.; Liao, Y.; Li, Y.; Gong, M.; Chen, Y. Synthesis of Sulfur-Resistant TiO2-CeO2 Composite and Its Catalytic Performance in the Oxidation of a Soluble Organic Fraction from Diesel Exhaust. Catalysts 2018, 8, 246. https://doi.org/10.3390/catal8060246
Zhang N, Yang Z, Chen Z, Li Y, Liao Y, Li Y, Gong M, Chen Y. Synthesis of Sulfur-Resistant TiO2-CeO2 Composite and Its Catalytic Performance in the Oxidation of a Soluble Organic Fraction from Diesel Exhaust. Catalysts. 2018; 8(6):246. https://doi.org/10.3390/catal8060246
Chicago/Turabian StyleZhang, Na, Zhengzheng Yang, Zhi Chen, Yunxiang Li, Yunwen Liao, Youping Li, Maochu Gong, and Yaoqiang Chen. 2018. "Synthesis of Sulfur-Resistant TiO2-CeO2 Composite and Its Catalytic Performance in the Oxidation of a Soluble Organic Fraction from Diesel Exhaust" Catalysts 8, no. 6: 246. https://doi.org/10.3390/catal8060246
APA StyleZhang, N., Yang, Z., Chen, Z., Li, Y., Liao, Y., Li, Y., Gong, M., & Chen, Y. (2018). Synthesis of Sulfur-Resistant TiO2-CeO2 Composite and Its Catalytic Performance in the Oxidation of a Soluble Organic Fraction from Diesel Exhaust. Catalysts, 8(6), 246. https://doi.org/10.3390/catal8060246





