Mixed-Phase (2H and 1T) MoS2 Catalyst for a Highly Efficient and Stable Si Photocathode
Abstract
1. Introduction
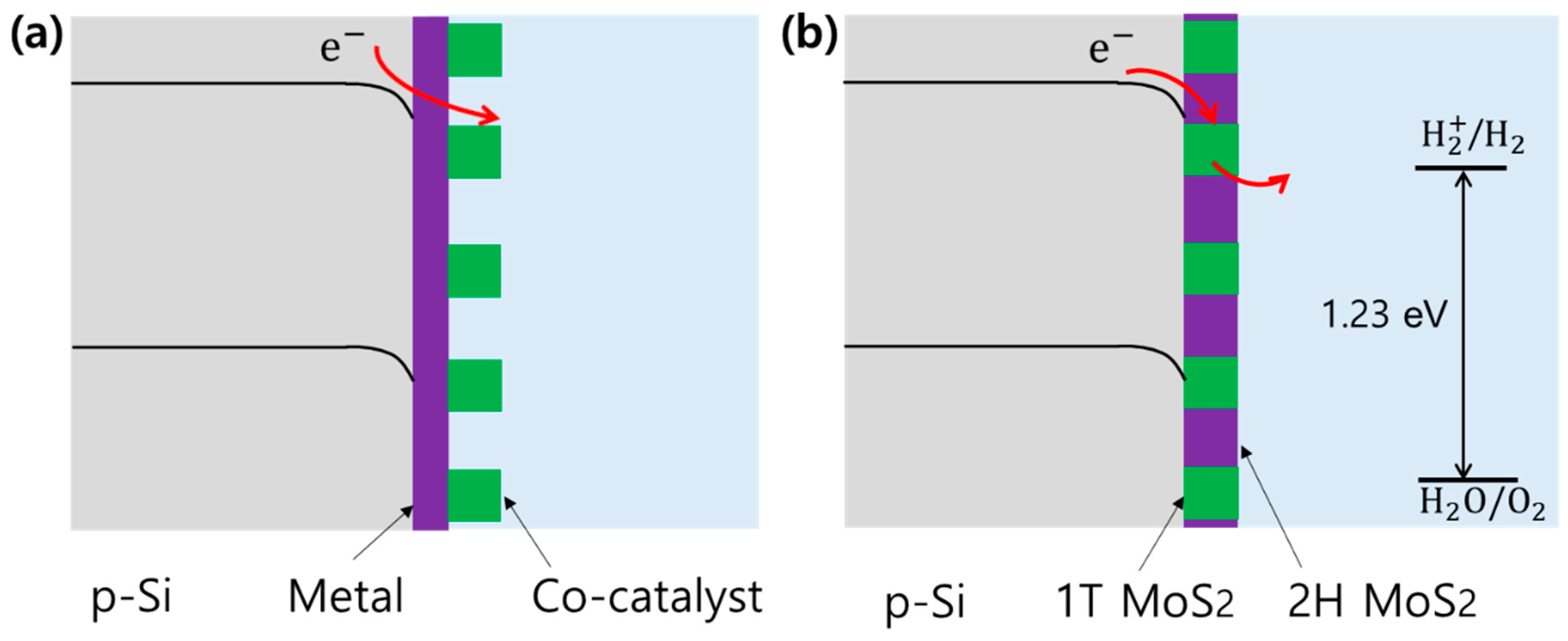
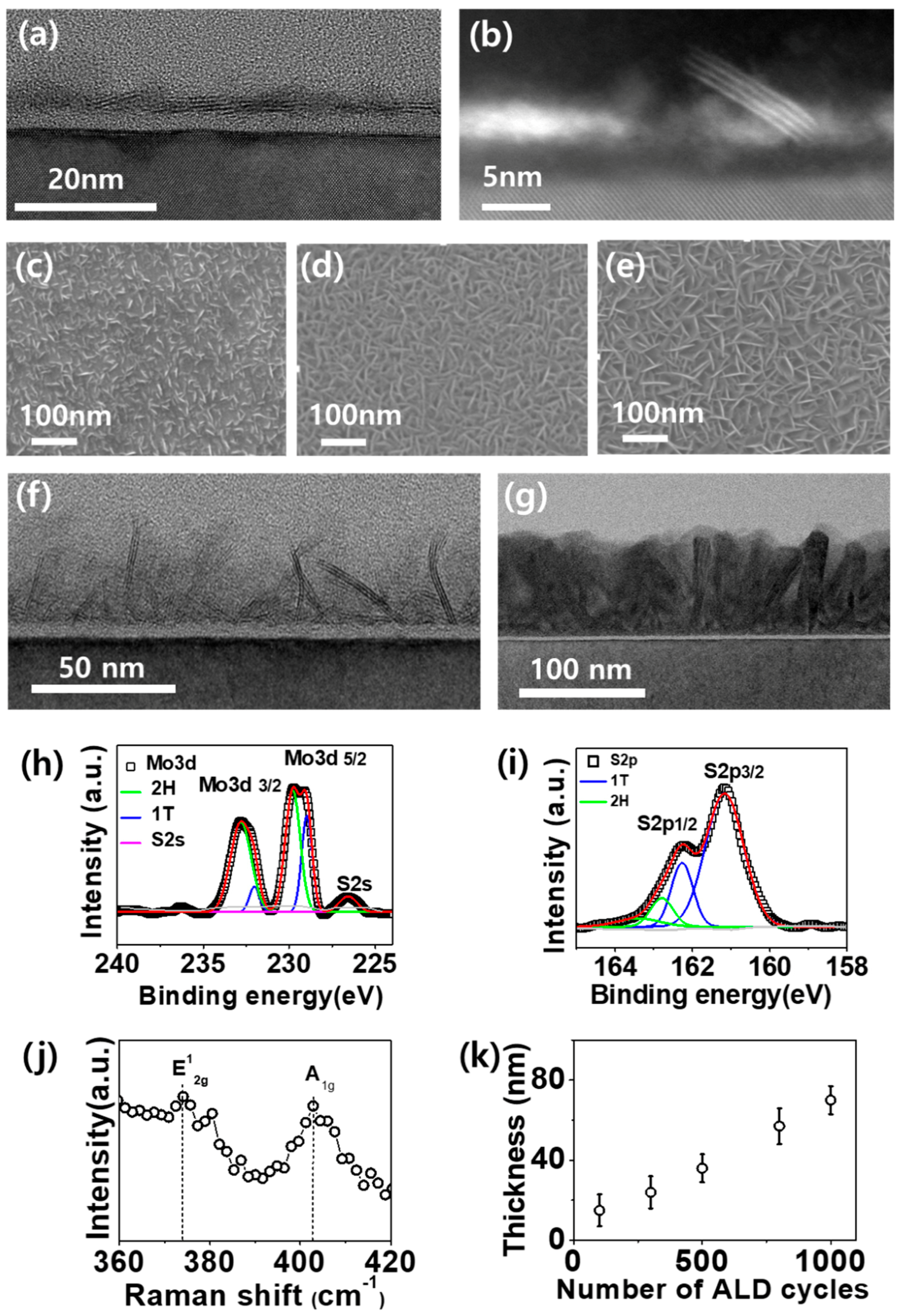
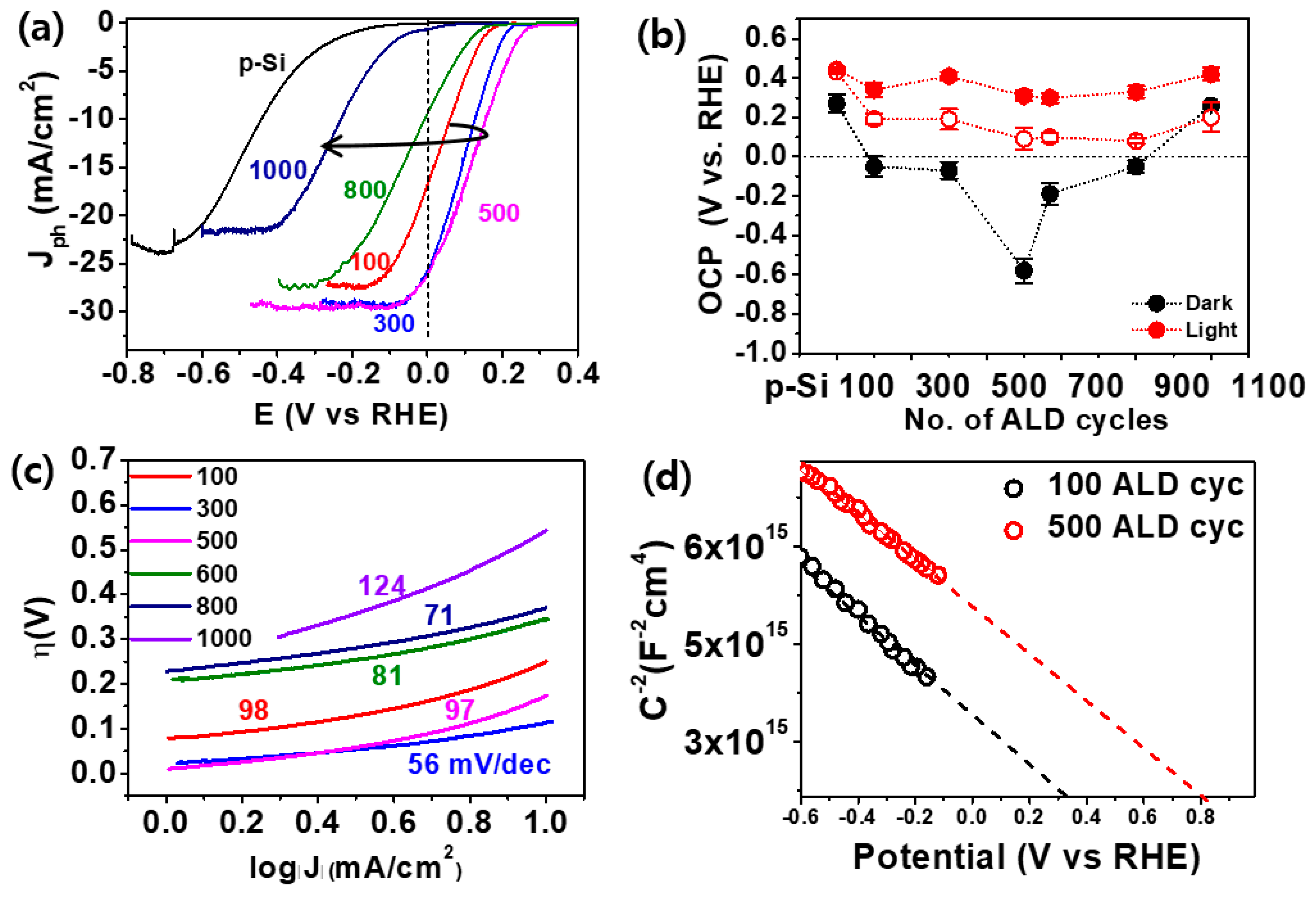
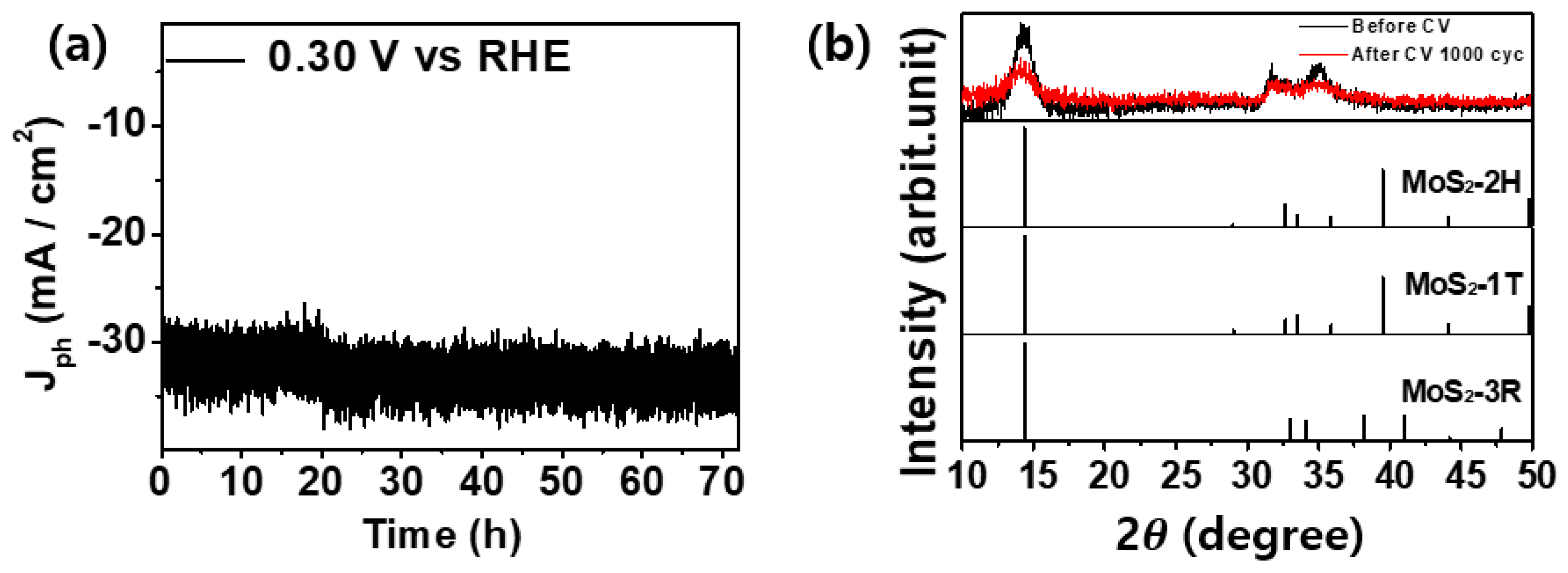
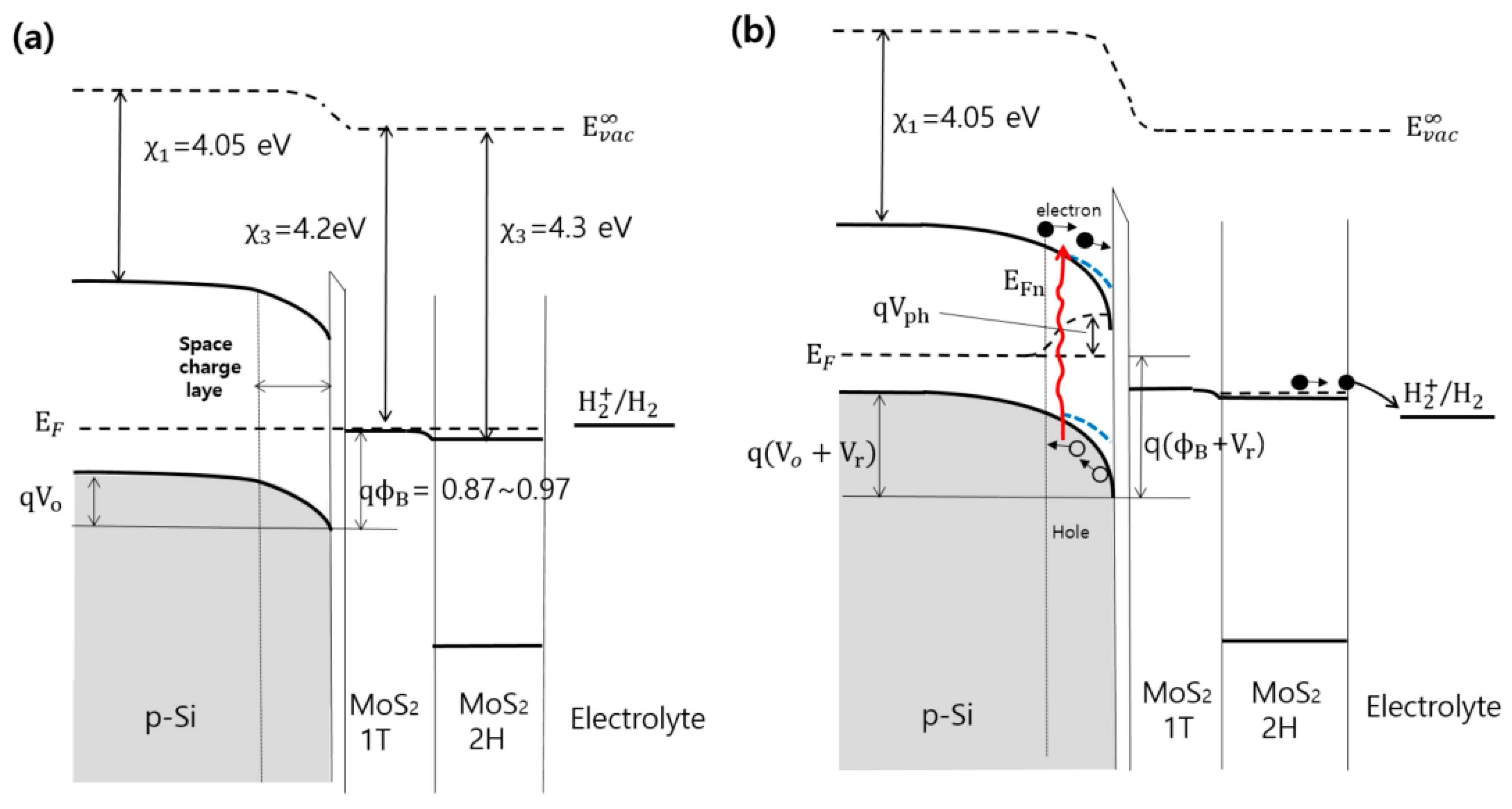
2. Results and Discussion
3. Experimental Section
3.1. Atomic Layer Deposition
3.2. Material Characterization
3.3. Electrochemical Characterization
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Sivula, K.; van de Krol, R. Semiconducting materials for photoelectrochemical energy conversion. Nat. Rev. Mater. 2016, 1, 15010. [Google Scholar] [CrossRef]
- Xing, Z.; Ren, F.; Wu, H.; Wu, L.; Wang, X.; Wang, J.; Wan, D.; Zhang, G.; Jiang, C. Enhanced PEC performance of nanoporous Si photocathodes by converting HfO2 and TiO2 passivaion layers. Sci. Rep. 2017, 7, 43901. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Jang, J.W.; Jo, Y.H.; Abdi, F.F.; Lee, Y.H.; van de Krol, R.; Lee, J.S. Hetero-type dual photoanodes for unbiased solar water splitting with extended light harvesting. Nat. Commun. 2017, 7, 13380. [Google Scholar] [CrossRef] [PubMed]
- Choi, M.J.; Jung, J.Y.; Park, M.J.; Song, J.W.; Lee, J.H.; Bang, J.H. Long-term durable silicon photocathode protected by a thin Al2O3/SiOx layer for photoelectrochemical hydrogen evolution. J. Mater. Chem. A 2014, 2, 2928–2933. [Google Scholar] [CrossRef]
- Zhao, Y.; Anderson, N.C.; Zhu, K.; Aguiar, J.A.; Seabold, J.A.; Lagemaat, J.V.D.; Oh, J. Oxidatively Stable Nanoporous Silicon Photocathodes with Enhanced Onset Voltage for Photoelectrochemical Proton Reduction. Nano Lett. 2015, 15, 2517–2525. [Google Scholar] [CrossRef] [PubMed]
- Feng, J.; Gong, M.; Kenney, M.J.; Wu, J.Z.; Zhang, B.; Li, Y.; Dai, H. Nickel-coated silicon photocathode for water splitting in alkaline electrolytes. Nano Res. 2015, 8, 1577–1583. [Google Scholar] [CrossRef]
- Wickramaratne, D.; Zahid, F.; Lake, R.K. Electronic and thermoelectric properties of few-layer transition metal dichalcogenides. J. Chem. Phys. 2014, 140, 124710. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Tian, H.; Ren, C.; Yu, J.; Sun, M. Electronic and optical properties of heterostructures based on transition metal dichalcogenides and graphene-like zinc oxide. Sci. Rep. 2018, 8, 12009. [Google Scholar] [CrossRef] [PubMed]
- Lang, O.; Tomm, V.; Schlaf, R.; Pettenkofer, C.; Jaegermann, W. Single crystalline GaSe/WeSe2 heterointerfaces grown by van der Waals epitaxy. ll. Junction characterization. J. Appl. Phys. 1994, 75, 7814–7820. [Google Scholar] [CrossRef]
- Andoshe, D.M.; Jin, G.; Lee, C.S.; Kim, C.; Kwon, K.C.; Choi, S.; Sohn, W.; Moon, C.W.; Lee, S.H.; Suh, J.M.; et al. Directly Assembeled 3D Molybdenum Disulfie on Silicon Wafer for Efficient Photoelectrochemical Water Reduction. Adv. Sustain. Syst. 2018, 2, 1700142–1700151. [Google Scholar] [CrossRef]
- Xiang, Q.; Yu, J.; Jaroniec, M. Synergetic effect of MoS2 and graphene as cocatalysts for enhanced photocatalytic H2 production activity of TiO2 nanoparticles. J. Am. Chem. Soc. 2012, 134, 6575–6578. [Google Scholar] [CrossRef] [PubMed]
- Chang, K.; Mei, Z.; Wang, T.; Kang, Q.; Ouyang, S.; Ye, J. MoS2/graphene cocatalyst for efficient photocatalytic H2 evolution under visible light irradiation. ACS Nano 2014, 8, 7078–7087. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Wang, H.; Peng, S. Tunable Photodeposition of MoS2 onto a Composite of Reduced Graphene Oxide and CdS for Synergic Photocatalytic Hydrogen Generation. J. Phys. Chem. C 2014, 118, 19842–19848. [Google Scholar] [CrossRef]
- Karunadasa, H.I.; Montalvo, E.; Sun, Y.; Majda, M.; Long, J.R.; Chang, C.J. A molecular MoS2 edge site mimic for catalytic hydrogen generation. Science 2012, 335, 698–702. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Meng, X.; Zhang, Z. Recent Development on MoS 2 -based Photocatalysis: A Review. J. Photochem. Photobiol. C Photochem. Rev. 2017, 25, 39–55. [Google Scholar] [CrossRef]
- Hinnemann, B.; Moses, P.G.; Bonde, J.; Jørgensen, K.P.; Nielsen, J.H.; Horch, S.; Chorkendorff, I.; Nørskov, J.K. Biomimetic hydrogen evolution: MoS2 nanoparticles as catalyst for hydrogen evolution. J. Am. Chem. Soc. 2005, 127, 5308–5309. [Google Scholar] [CrossRef] [PubMed]
- Oh, S.; Kim, J.B.; Song, J.T.; Oh, J.; Kim, S.-H. Atomic layer deposited Molybdenum disulfide on Si photocathodes for highlyt efficient photoelectrochemical water redution reaction. J. Mater. Chem. A 2017, 5, 3304–3310. [Google Scholar] [CrossRef]
- Yin, Y.; Zhang, Y.; Gao, T.; Yao, T.; Zhang, X.; Han, J.; Wang, X.; Zhang, Z.; Xu, P.; Zhang, P.; et al. Synergistic Phase and Disorder Engineering in 1T-MoSe2 Nanosheets for Enhanced Hydrogen Evolution Reaction. Adv. Mater. 2017, 29, 1700311. [Google Scholar] [CrossRef] [PubMed]
- Lukowski, M.A.; Daniel, A.S.; Meng, F.; Forticaux, A.; Li, L.; Jin, S. Enhanced Hydrogen Evolution Catalysis from Chemically Exfoliated Metallic MoS2 Nanosheets. J. Am. Chem. Soc. 2013, 135, 10274–10277. [Google Scholar] [CrossRef] [PubMed]
- Voiry, D.; Salehi, M.; Silva, R.; Fujita, T.; Chen, M.; Asefa, T.; Shenoy, V.B.; Eda, G.; Chhowalla, M. Conducting MoS2 Nanosheets as Catalysts for Hydrogen Evolution Reaction. Nano Lett. 2013, 13, 6222–6227. [Google Scholar] [CrossRef] [PubMed]
- Hill, J.C.; Landers, A.T.; Switzer, J.A. An electrodeposited inhomogeneous metal–insulator–semiconductor junction for efficient photoelectrochemical water oxidation. Nat. Mater. 2015, 14, 1150–1155. [Google Scholar] [CrossRef] [PubMed]
- Thorne, J.E.; Li, S.; Du, C.; Qin, G.; Wang, D. Energetics at the Surface of Photoelectrodes and Its Influence on the Photoelectrochemical Properties. J. Phys. Chem. Lett. 2015, 6, 4083–4088. [Google Scholar] [CrossRef] [PubMed]
- Kwon, K.C.; Choi, S.; Hong, K.; Moon, C.W.; Shim, Y.-S.; Kim, D.H.; Kim, T.; Sohn, W.; Jeon, J.-M.; Lee, C.-H.; et al. Wafer-scale transferable molybdenum disulfide thin-film catalysts for photoelectrochemical hydrogen production. Energy Environ. Sci. 2016, 9, 2240–2248. [Google Scholar] [CrossRef]
- Seo, J.; Kim, H.J.; Pekarek, R.T.; Rose, M.J. Hybrid Organic/Inorganic Band-Edge Modulation of p-Si(111) Photoelectrodes: Effects of R, Metal Oxide, and Pt on H2 GenerationEffects of R, Metal Oxide, and Pt on H2 Generation. J. Am. Chem. Soc. 2015, 137, 3173–3176. [Google Scholar] [CrossRef] [PubMed]
- Benck, J.D.; Lee, S.C.; Fong, K.D.; Kibsgaard, J.; Sinclair, R.; Jaramillo, T.F. Designing Active and Stable Silicon Photocathodes for Solar Hydrogen Production Using Molybdenum Sulfide Nanomaterials. Adv. Energy Mater. 2014, 4, 1400739–1400747. [Google Scholar] [CrossRef]
- Digdaya, I.A.; Adhyaksa, G.W.; Trześniewski, B.J.; Garnett, E.C.; Smith, W.A. Interfacial engineering of metal-insulator-semiconductor junctions for efficient and stable photoelectrochemical water oxidation. Nat. Commun. 2017, 8, 15968. [Google Scholar] [CrossRef] [PubMed]
- Fan, R.; Mao, J.; Yin, Z.; Jie, J.; Dong, W.; Fang, L.; Zheng, F.; Shen, M. Efficient and Stable Silicon Photocathodes Coated with Vertically Staning Nano-MoS2 Films for Soalr hydrogen Production. ACS Appl. Mater. Interfaces 2017, 9, 6123–6129. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.; Battaglia, C.; Boccard, M.; Hettick, M.; Yu, Z.; Ballif, C.; Ager, J.W.; Javey, A. Amorphous Si thin film based photocathodes with high photovoltage for efficient hydrogen production. Nano Lett. 2013, 13, 5615–5618. [Google Scholar] [CrossRef] [PubMed]
- Ho, T.A.; Bae, C.; Lee, S.; Kim, M.; Montero-Moreno, J.M.; Park, J.H.; Shin, H. Edge-On MoS2 Thin Films by Atomic Layer Deposition for Understanding the Interplay between the Active Area and Hydrogen Evolution Reaction. Chem. Mater. 2017, 29, 7604–7614. [Google Scholar] [CrossRef]
- Joensen, P.; Crozier, E.; Alberding, N.; Frindt, R. A study of single-layer and restacked MoS2 by X-ray diffraction and X-ray absorption spectroscopy. J. Phys. C Solid State Phys. 1987, 20, 4043–4053. [Google Scholar] [CrossRef]
- Sim, U.; Yang, T.-Y.; Moon, J.; An, J.; Hwang, J.; Seo, J.-H.; Lee, J.; Kim, K.Y.; Lee, J.; Han, S.; et al. N-doped monolayer graphene catalyst on silicon photocathode for hydrogen production. Energy Environ. Sci. 2013, 6, 3658–3664. [Google Scholar] [CrossRef]
- Walter, M.G.; Warren, E.L.; McKone, J.R.; Boettcher, S.W.; Mi, Q.; Santori, E.A.; Lewis, N.S. Solar Water Splitting cells. Chem. Rev. 2010, 110, 6446–6473. [Google Scholar] [CrossRef] [PubMed]
- Esposito, D.V.; Levin, I.; Moffat, T.P.; Talin, A.A. H2 evolution at Si-based metal-insulator-semiconductor photoelectrodes enhanced by inversion channel charge collection and H spillover. Nat. Mater. 2013, 12, 562–568. [Google Scholar] [CrossRef] [PubMed]
- Weitering, H.H.; Ettema, A.R.H.F.; Hibma, T. Surface states and Fermi-level pinning at epitaxial Pb/Si(111) surfaces. Phys. Rev. B 1992, 45, 9126–9135. [Google Scholar] [CrossRef]
- Liu, D.; Li, L.; Gao, Y.; Wang, C.; Jiang, J.; Xiong, Y. The Nature of Photocatalytic “Water Splitting” on Silicon Nanowires. Angew. Chem. Int. Ed. 2015, 54, 2980–2985. [Google Scholar] [CrossRef] [PubMed]
- Kronik, L.; Shapira, Y. Surface photovoltage phenomena: Theory, experiment, and applications. Surf. Sci. Rep. 1999, 37, 1–206. [Google Scholar] [CrossRef]
- Benson, J.; Li, M.; Wang, S.; Wang, P.; Papakonstantinou, P. Electrocatalytic Hydrogen Evolution Reaction on Edges of a Few Layer Molybdenum Disulfide Nanodots. ACS Appl. Mater. Interfaces 2015, 7, 14113–14122. [Google Scholar] [CrossRef] [PubMed]
- Thorne, J.E.; Jang, J.W.; Liu, E.Y.; Wang, D. Understanding the origin of photoelectrode performance enhancement by probing surface kinetics. Chem. Sci. 2016, 7, 3347–3354. [Google Scholar] [CrossRef] [PubMed]
- Quickenden, T.I.; Yim, G.K. The relationship between open circuit photovoltage and light intensity in photogalvanic cells—An extension of albery and Archer’s treatment. Electrochem. Acta 1979, 24, 143–146. [Google Scholar] [CrossRef]
- Salvador, P.; Hidalgo, M.G.; Zaban, A.; Bisquert, J. Illumination intensity dependence of the photovoltage in nanostructured TiO2 dye-sensitized solar cells. J. Phys. Chem. B 2005, 109, 15915–15926. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Zhang, Y. Electrical characterization of TiO2/CH3NH3PbI3 heterojunction solar cells. J. Mater. Chem. A 2014, 2, 10244–10249. [Google Scholar] [CrossRef]
- Kirchartz, T.; Gong, W.; Hawks, S.A.; Agostinelli, T.; MacKenzie, R.C.I.; Yang, Y.; Nelson, J. Sensitivity of the Mott–Schottky Analysis in Organic Solar Cells. J. Phys. Chem. C 2012, 116, 7672–7680. [Google Scholar] [CrossRef]
- Kenney, M.J.; Gong, M.; Li, Y.; Wu, J.Z.; Feng, J.; Lanza, M.; Dai, H. High-performance silicon photoanodes passivated with ultrathin nickel films for water oxidation. Science 2013, 342, 836–840. [Google Scholar] [CrossRef] [PubMed]
- Fujii, K.; Ono, M.; Iwaki, Y.; Sato, K.; Ohkawa, K.; Yao, T. Photoelectrochemical Properties of the p−n Junction in and near the Surface Depletion Region of n-Type GaN. J. Phys. Chem. C 2010, 114, 22727–22735. [Google Scholar] [CrossRef]
- Ding, Q.; Meng, F.; English, C.R.; Cabán-Acevedo, M.; Shearer, M.J.; Liang, D.; Daniel, A.S.; Hamers, R.J.; Jin, S. Efficient photoelectrochemical hydrogen generation using heterostructures of Si and chemically exfoliated metallic MoS2. J. Am. Chem. Soc. 2014, 136, 8504–8507. [Google Scholar] [CrossRef] [PubMed]
- Du, P.; Zhu, Y.; Zhang, J.; Xu, D.A.; Peng, W.; Zhang, G.; Zhang, F.; Fan, X. Metallic 1T phase MoS2 nanosheets as a highly efficient co-catalyst for the photocatalytic hydrogen evolution of CdS nanorods. RSC Adv. 2016, 6, 74394–74399. [Google Scholar] [CrossRef]
- Bai, H.F.; Xu, L.C.; Di, M.Y.; Hao, L.Y.; Yang, Z.; Liu, R.P.; Li, X.Y. The intrinsic interface properties of the top and edge 1T/2H MoS2 contact: A first-principles study. J. Appl. Phys. 2018, 123, 95301–95306. [Google Scholar] [CrossRef]
- Reichman, J. The current-voltage characteristics of semiconductor-electrolyte junction photovoltaic cells. Appl. Phys. Lett. 1980, 36, 574–577. [Google Scholar] [CrossRef]
- Bard, A.; Faulkner, L. Electrochemical Methods. Fundamentals and Applications, 2nd ed.; John Wiley and Sons: New York, NY, USA, 2001. [Google Scholar]
- Claeys, C.L.; Watanabe, M.; Rai-Choudhury, P.; Stallhofer, P. High Purity Silicon; The Electrochemical Society, Inc.: Burghausen, Germany, 2002. [Google Scholar]
- Benchamekh, R.; Nesoklon, M.; Jancu, J.-M.; Voisin, P. Semiconductor Modeling Techniques; Springer: Berlin/Heidelberg, Germany, 2012. [Google Scholar]
- Schmidt, H.; Giustiniano, F.; Eda, G. Electronic transport properties of transition metal dichalcogenide field-effect devices: Surface and interface effects. Chem. Soc. Rev. 2015, 44, 7715–7736. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Zhang, C.; Rana, F. Ultrafast Dynamics of Defect-Assisted Electron–Hole Recombination in Monolayer MoS2. Nano Lett. 2015, 15, 339–345. [Google Scholar] [CrossRef] [PubMed]
- Yun, W.S.; Han, S.; Hong, S.C.; Kim, I.G.; Lee, J. Thickness and strain effects on electronic structures of transition metal dichalcogenides: 2H-MX2 (M = Mo, W; X = S, Se, Te) semiconductors. Phys. Rev. B 2012, 85, 33305–33310. [Google Scholar] [CrossRef]
- Hayden, T.; Dumesic, J.; Sherwood, R.; Baker, R. Direct Observation by Controlled Atmosphere Electron Microscopy of the Changes in Morphology of Molybdenum Oxide and Sulfide Supported on Alumina and Graphite. J. Catal. 1987, 105, 299–318. [Google Scholar] [CrossRef]
- Li, T.-L.; Lee, Y.-L.; Teng, H. High-performance quantum dot-sensitized solar cells based on sensitization with CuInS2 quantum dots/CdS heterostructure. Energy Environ. Sci. 2012, 5, 5315–5324. [Google Scholar] [CrossRef]
- Agrawal, A.; Lin, J.; Barth, M.; White, R.; Zheng, B.; Chopra, S.; Gupta, S.; Wang, K.; Gelatos, J.; Mohney, S.E.; et al. Fermi level depinning and contact resistivity reduction using a reduced titania interlayer in n-silicon metal-insulator-semiconductor ohmic contacts nanowire array electrode to construct a photocathode composed of elements abundant on the earth for hydrogen generation. Appl. Phys. Lett. 2014, 104, 112101. [Google Scholar]
- Smit, G.D.J.; Rogge, S.; Klapwijk, T.M. Scaling of nano-Schottky-diodes. Appl. Phys. Lett. 2002, 81, 3852–3854. [Google Scholar] [CrossRef]
- Rossi, R.C.; Lewis, N.S. Investigation of the Size-Scaling Behavior of Spatially Nonuniform Barrier Height Contacts to Semiconductor Surfaces Using Ordered Nanometer-Scale Nickel Arrays on Silicon Electrodes. J. Phys. Chem. B 2001, 105, 12303–12318. [Google Scholar] [CrossRef]
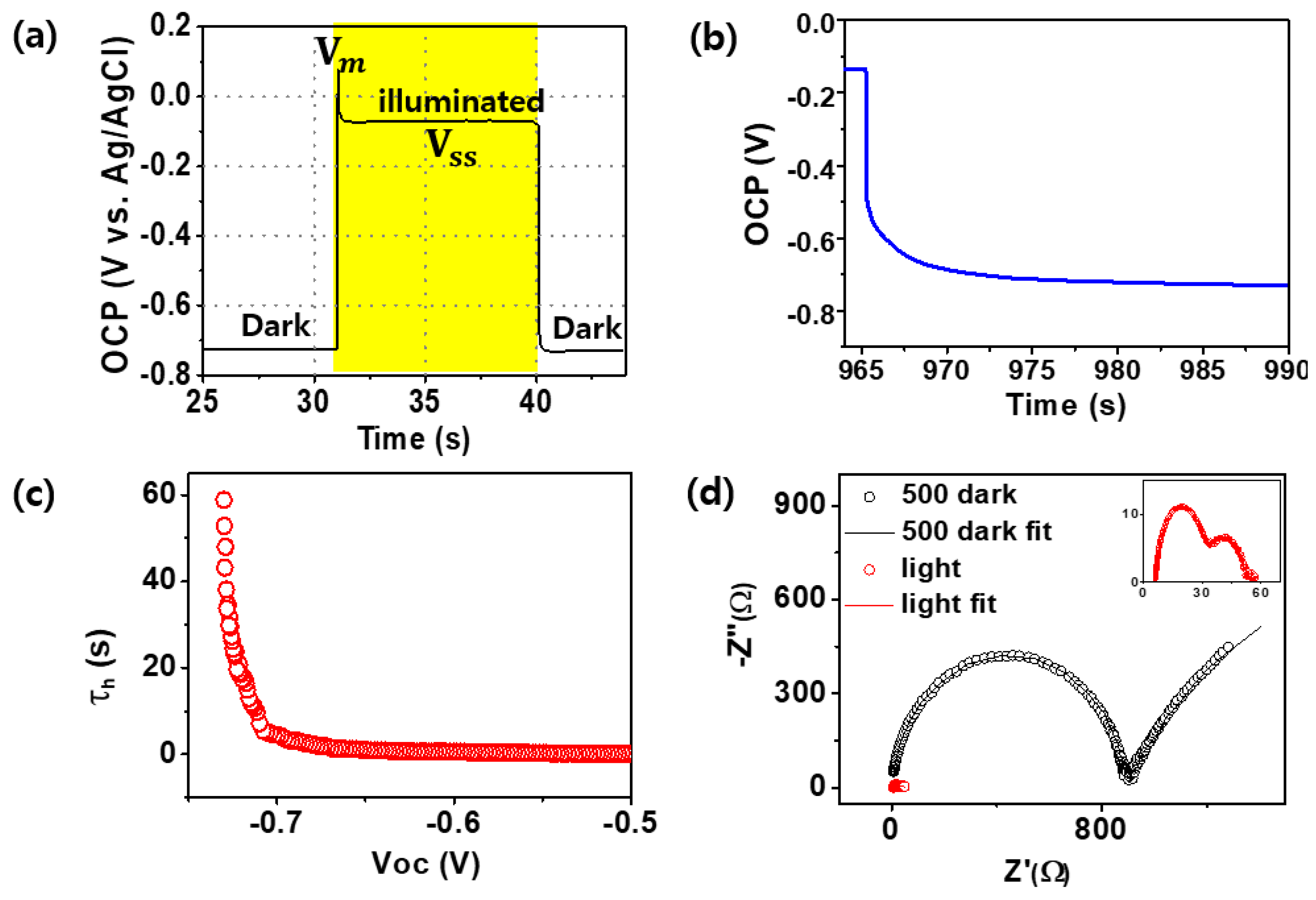
| p-Si | 100 | 300 | 500 | 600 | 800 | 1000 | |
|---|---|---|---|---|---|---|---|
| Vph | 0.11 ± 0.3 | 0.39 ± 0.04 | 0.48 ± 0.01 | 0.80 ± 0.02 | 0.49 ± 0.01 | 0.38 ± 0.01 | 0.26 ± 0.06 |
| Von | −0.12 ± 0.1 | 0.16 ± 0.02 | 0.24 ± 0.03 | 0.35 ± 0.03 | 0.27 ± 0.02 | 0.16 ± 0.01 | −0.17 ± 0.02 |
| Jsc | 0.03 ± 0.05 | 17.18 ± 2.03 | 26.50 ± 1.40 | 26.70 ± 1.13 | 17.09 ± 1.63 | 10.03 ± 0.68 | 0.67 ± 0.26 |
| Jph | 24.00 ± 1.31 | 27.00 ± 0.53 | 29.00 ± 0.62 | 30.00 ± 1.06 | 27.00 ± 2.30 | 26.00 ± 0.98 | 21.28 ± 1.08 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Joe, J.; Bae, C.; Kim, E.; Ho, T.A.; Yang, H.; Park, J.H.; Shin, H. Mixed-Phase (2H and 1T) MoS2 Catalyst for a Highly Efficient and Stable Si Photocathode. Catalysts 2018, 8, 580. https://doi.org/10.3390/catal8120580
Joe J, Bae C, Kim E, Ho TA, Yang H, Park JH, Shin H. Mixed-Phase (2H and 1T) MoS2 Catalyst for a Highly Efficient and Stable Si Photocathode. Catalysts. 2018; 8(12):580. https://doi.org/10.3390/catal8120580
Chicago/Turabian StyleJoe, Jemee, Changdeuck Bae, Eunsoo Kim, Thi Anh Ho, Heejun Yang, Jong Hyeok Park, and Hyunjung Shin. 2018. "Mixed-Phase (2H and 1T) MoS2 Catalyst for a Highly Efficient and Stable Si Photocathode" Catalysts 8, no. 12: 580. https://doi.org/10.3390/catal8120580
APA StyleJoe, J., Bae, C., Kim, E., Ho, T. A., Yang, H., Park, J. H., & Shin, H. (2018). Mixed-Phase (2H and 1T) MoS2 Catalyst for a Highly Efficient and Stable Si Photocathode. Catalysts, 8(12), 580. https://doi.org/10.3390/catal8120580






