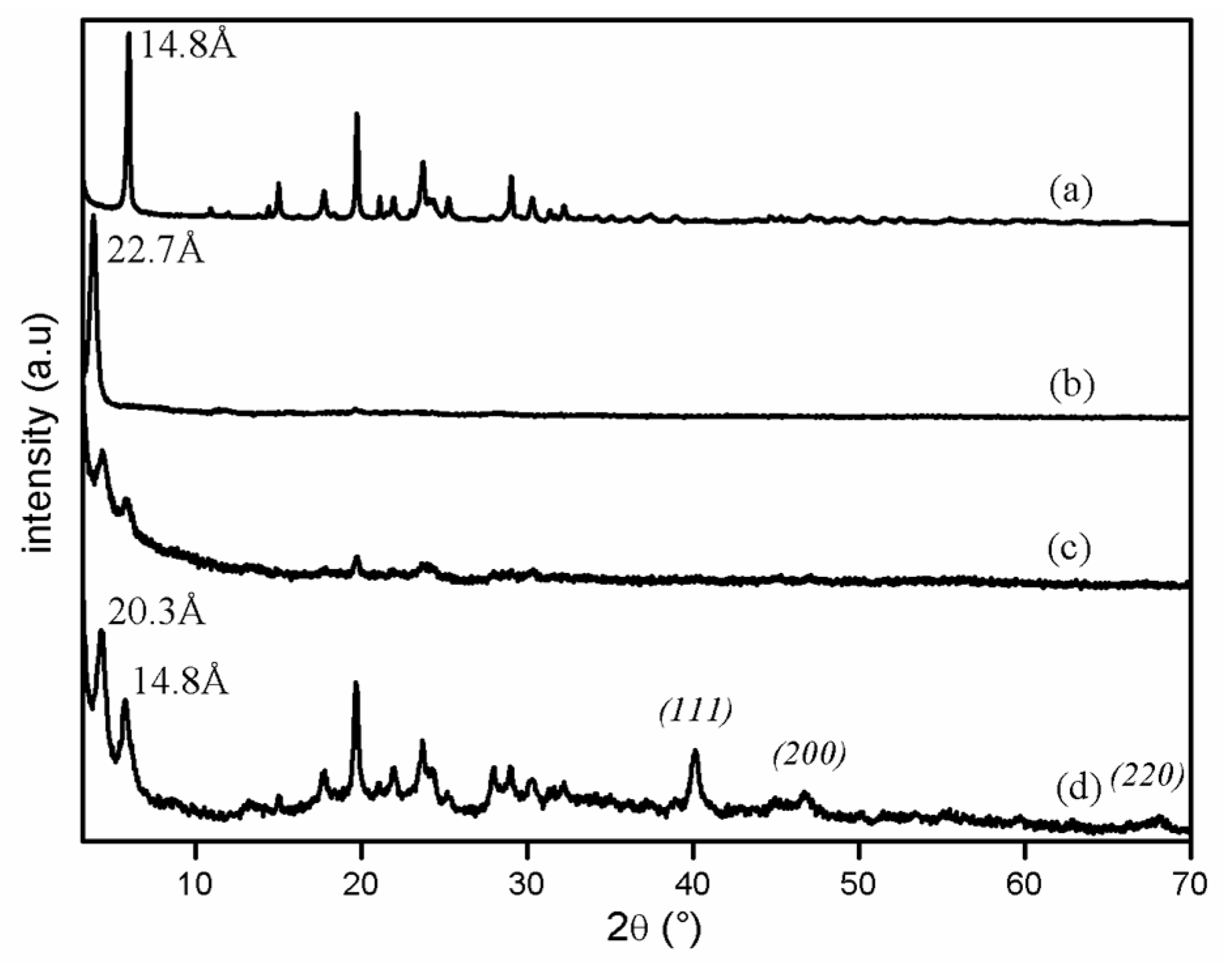
2.2.1. Suzuki–Miyaura Coupling Reaction
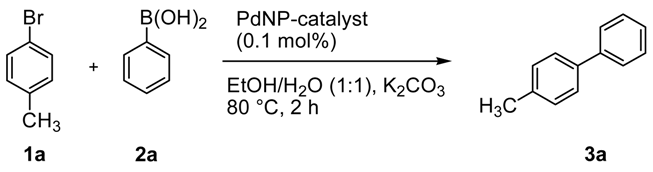
We started our study testing the activity of Pd@ZPGly catalysts in the Suzuki–Miyaura reaction of 4-bromotoluene (1a) and phenylboronic acid (2a) in EtOH/H2O (1:1) at 80 °C for 2 h.
All of the catalysts showed high activity, but only with Pd@ZPGly-1 and Pd@ZPGly-15 catalysts a complete conversion was observed after 2 h and for three consecutive run (
Table 2, Entries 1 and 3). For each reuse, the catalysts were recovered by centrifugation, washed with EtOH/H
2O (1:1) and reused in the next cycle. Pd@ZPGly-7, Pd@ZPGly-1R and 7R showed a lower activity that also decreased after recovery and reuse (
Table 2, Entries 2, 5 and 6). This could be ascribable to the distribution of the Pd nanoparticle sizes on the surface of the supports. Indeed, in these samples, the particles having mean dimensions of 10–20 nm are highly represented, and after reduction, also a relevant aggregation phenomenon occurred. The ICP-OES analysis of crude products, obtained by usual work-up procedure (filtration, extraction with ethyl acetate, distillation of solvent under reduced pressure), showed a high content of palladium (20–31 ppm,
Table 2).
It is known that both semicrystalline and microcrystalline zirconium phosphate (α-ZrP) undergo hydrolysis to some extent in basic media, especially at high temperatures [
41]. The pH of aqueous K
2CO
3 reaction medium is about 12, and therefore, the hydrolysis process of the support could be also responsible for the high palladium leaching that exceeded the allowed upper limit value [
42].
The efficiency of these novel Pd@ZPGly catalysts has been compared to Pd/α-ZrPK catalyst (palladium nanoparticles on layered potassium zirconium phosphate), a similar catalytic system recently reported by our group [
15] and with Pd/C 10 wt %. By using the same reaction conditions, Pd/α-ZrPK showed a significantly lower efficiency (
Table 2, Entry 6), while when the reaction was performed in the presence of Pd/C, the catalytic activity was comparable with that of Pd@ZPGly catalysts, but the amount of Pd in the product is extremely high (192 ppm,
Table 2, Entry 8).
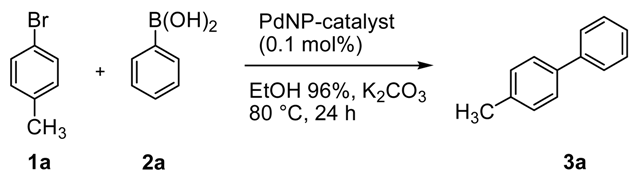
In order to simplify the procedure of the work-up and also reduce the leaching of the metal into the product, we investigated the use of the heterogeneous K
2CO
3/aq. EtOH azeotrope (EtOH 96%) system [
43]. K
2CO
3 is in fact insoluble in aqueous ethanol azeotrope, and therefore, it can be removed together with the catalyst by simple filtration making possible the separation of the pure product, after the distillation and recovery of the reaction medium, therefore reducing the waste of the process. In addition, the heterogeneous conditions can significantly protect the support avoiding its hydrolysis under basic aqueous conditions.
Pd@ZPGly catalysts were tested in the Suzuki–Miyaura reaction of 4-bromotoluene (
1a) and phenylboronic acid (
2a) in aq. EtOH azeotrope at 80 °C (
Table 3). The reaction time was longer (24 vs. 2 h), and high conversion was obtained by using Pd@ZPGly-1, -7, -15 (97–98%,
Table 3, Entries 1 and 3), while lower conversions were observed with Pd@ZPGly-1R and -7R (78% and 85%, respectively,
Table 3, Entries 4 and 5), again presumably due to the aggregation extent of Pd nanoparticles. The comparable Pd/α-ZrPK catalyst [
15] and Pd/C 10 wt % showed a much lower efficiency (
Table 3, Entries 6 and 7).
The ICP-OES analysis of crude products revealed that by using the ethanol azeotrope, a much lower content of palladium was obtained than when EtOH/H
2O (1:1) was used as the reaction medium (9–17 ppm vs. 20–31 ppm). This is consistent with the milder pH conditions achieved when the heterogeneous K
2CO
3/aq. EtOH azeotrope system is used, resulting in higher stability of the support. When Pd/C 10 wt % was used, a very high level of Pd content in the product was observed again (184 ppm,
Table 3, Entry 7). Pd@ZPGly-15 proved to be the best catalyst in terms of activity and leaching of palladium (
Table 3, Entry 3).
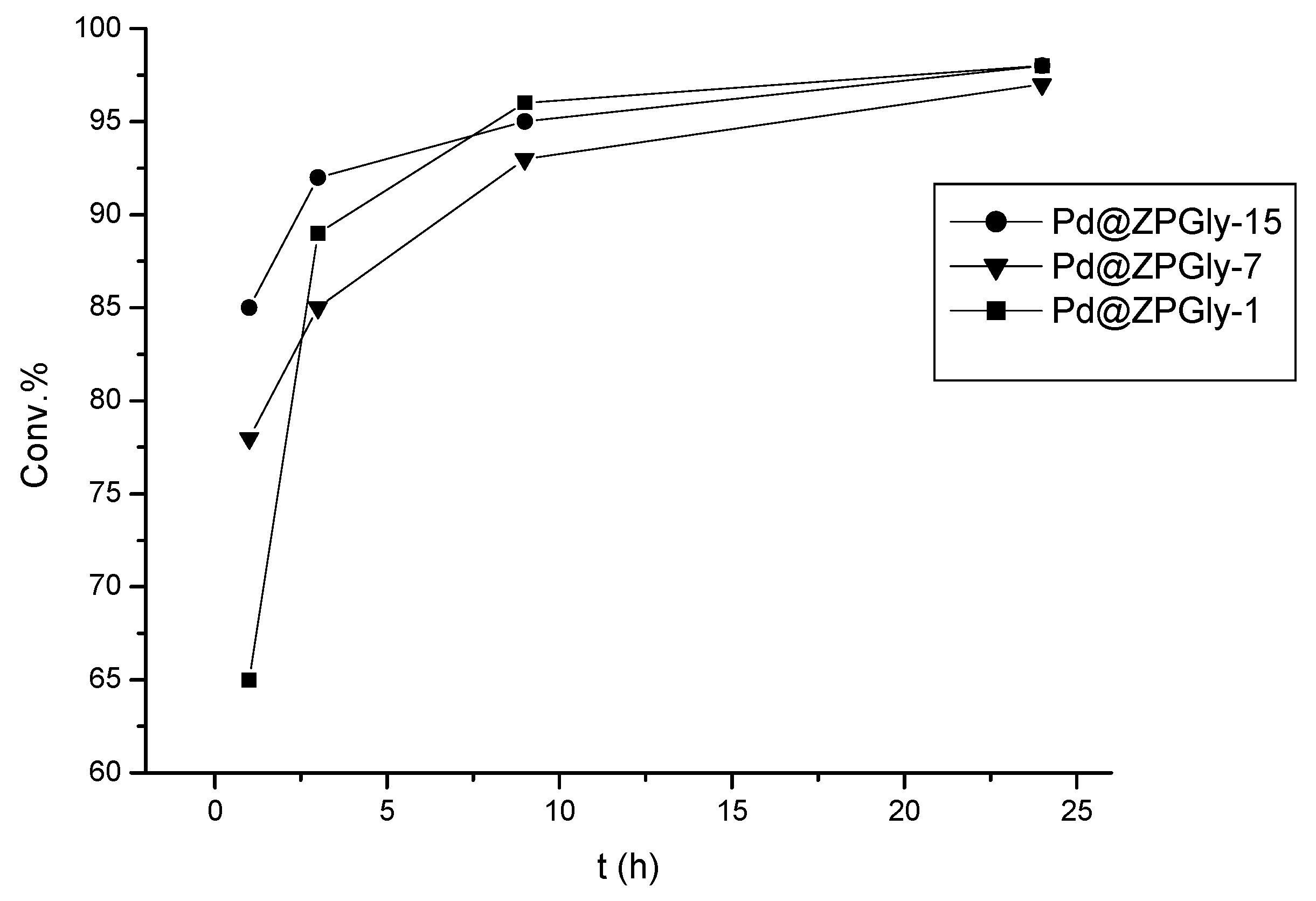
The kinetics of the Suzuki–Miyaura reaction of 4-bromotoluene (
1a) and phenylboronic acid (
2a) in aq. EtOH azeotrope in the presence of Pd@ZPGly-1, -7, -15 was further studied (
Figure 3). After 1 h at 80 °C, the difference in reactivity was significant. Pd@ZPGly-1, which mainly contains Pd(II) species, provided a lower conversion (63%) as compared to Pd@ZPGly-7 and -15 (78% and 85%, respectively) that also contained Pd(0) species. During the reaction, the reduction of Pd(II) occurs at the expense of EtOH [
15,
44,
45], and after 9 h, the conversion was comparable (93–96%). At the end of the reactions, all of the recovered catalysts were black, supporting the more complete presence of Pd(0) species. Pd@ZPGly-15 showed the highest activity and also the best stability. The Pd content (2.11 mmol/g) was very high, and also, these features can be useful in view of a large-scale flow protocol, allowing the use of the minimal quantity of catalytic material to be packed into a flow reactor.
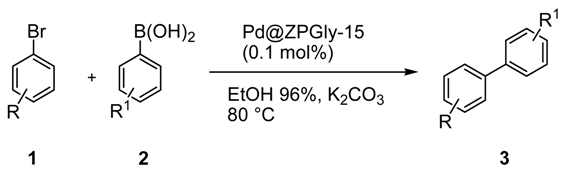
To confirm the efficiency of Pd@ZPGly-15 in combination with the K
2CO
3/aq. EtOH azeotrope system, the protocol has been extended to the reactions of a series of aryl bromides
1a–
n with phenyl boronic acids
2a,
b (
Table 4). The reactions were carried at 80 °C, in the presence of 0.1 mol % of Pd@ZPGly-15, obtaining from high to quantitative conversion of
1 and excellent yields of isolated products
3a–
n. Bromobenzene and aryl bromide with electron-donating groups (
1a,
b,
d) showed as expected a lower reactivity (reaction time 24 h) with respect to 4-substituted aryl bromide with electron-withdrawing groups (
1c,
e,
f,
g,
l) (reaction time of 3–9 h), while 2- and 3-CHO aryl bromide (
1h,
i) reacted in a longer reaction time (24 h) maybe due to steric hindrance (2-CHO) or non-conjugative meta substitution (3-CHO). 2-CN aryl bromide reacted with 4-formyl phenyl boronic acids (
2b) to furnish in very high isolated yield the biphenyl
3n, a useful intermediate for the synthesis of valsartan [
46].
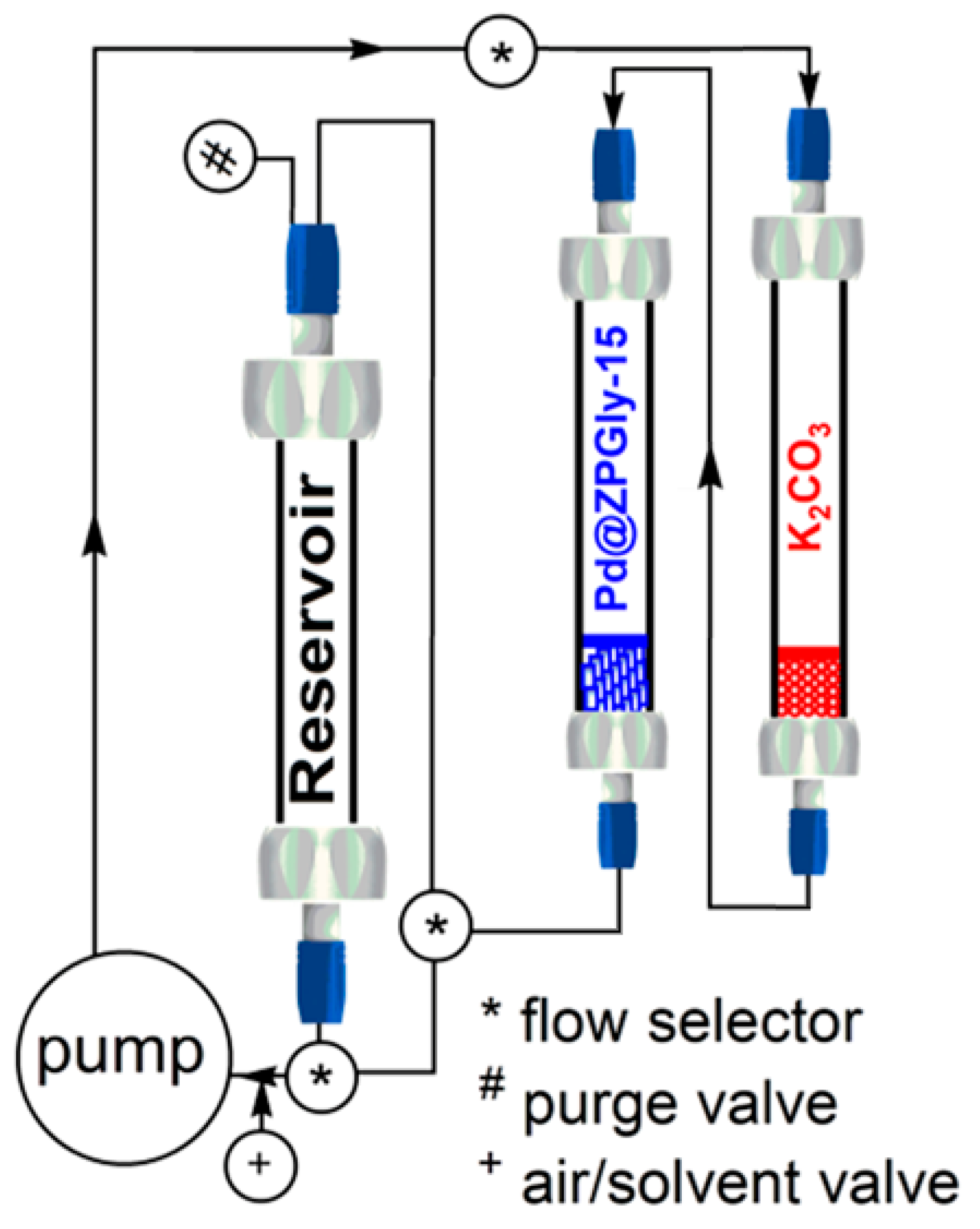
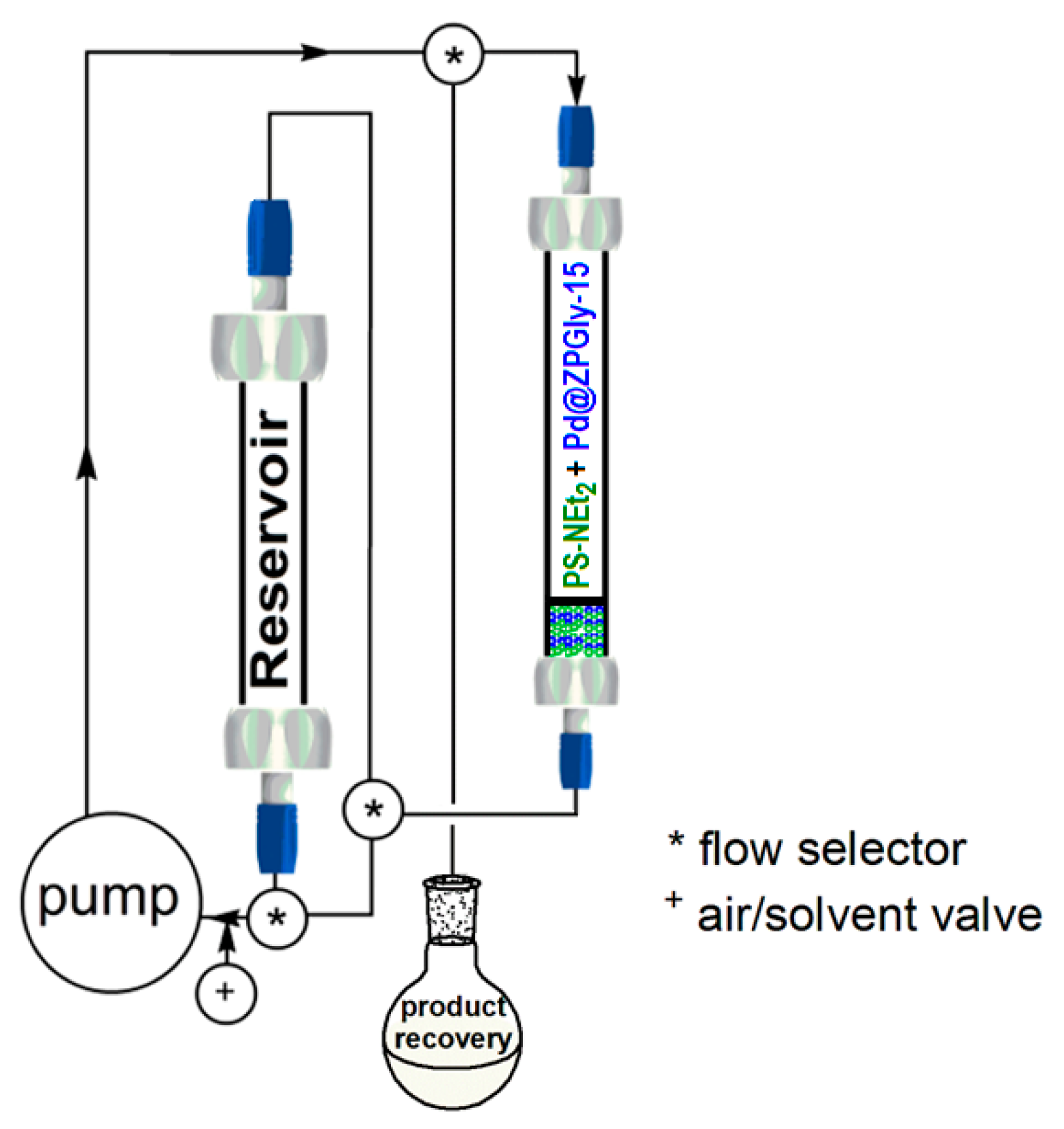
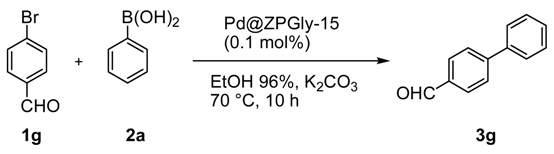
Continuing with our project and taking into account the optimized batch protocol, we have investigated the catalytic performance of Pd@ZPGly-15 in the Suzuki–Miyaura coupling of phenyl boronic acid (2a) with 4-bromo benzaldehyde (1g) in a flow protocol to assess the ability of ZPGly nanosheets to stabilize PdNPs and evaluate the catalytic activity of the recovered catalyst over time. The flow approach is also strategic to preserve the physical features of solid catalyst throughout the process and allows its simple, efficient and reproducible recovery and reuse.
To this aim, the reactor system was set up as depicted in
Scheme 1, where the base K
2CO
3 (48 mmol) and the catalyst Pd@ZPGly-15 (0.1 mmol %) were both dispersed in solid glass beads (1 mm diameter) and respectively charged into two separate glass column. Reactants (40 mmol of 4-bromo benzaldehyde (
1g) and 44 mmol of phenyl boronic acid (
2a)) dissolved in aq. EtOH azeotrope (0.33 M) were charged into a third glass column, used as the reservoir. The columns were connected to a pump by using the appropriate valves and installed into a thermostated box at 70 °C (not shown in
Scheme 1 for clarity).
The reaction mixture was pushed at a 1.5-mL/min flow rate through the base and the catalyst columns for 10 h until complete conversion was obtained (monitored by Gas–Liquid Chromatography (GLC) analyses). After this time and only the removal of the solvent by distillation (90% of solvent recovered), the product
3g was obtained as a white solid (98% yield,
Table 5).
The reusability of the catalyst was investigated, and this process was repeated for three consecutive runs with the same catalyst to obtain always the corresponding product 3g in a 98% yield.
Hot filtration test has been performed on the Suzuki reaction in aq. EtOH azeotrope with Pd@ZPGly-15. The reaction mixture was stirred at 80 °C for 20 min (conversion 71%), and the hot filtration was performed. K
2CO
3 was added again and the reaction mixture stirred at 80 °C. After a total reaction time of 1 h, the conversion was 74%, and the same conversion was observed after 24 h. This suggests that the homogeneous palladium released in solution (see
Table 3, Entry 3) does not effectively promote the process. In agreement, the reaction performed with homogeneous Pd catalyst (Pd(OAc)
2) did not give a satisfactory result as for Pd@ZPGly-15 (see the
Supplementary Materials).
When the reactions were performed under flow conditions, although the catalyst and the base were loaded into two separate columns, an excellent catalytic activity was observed. A “release and catch” mechanism has been hypothesized in which soluble active palladium species are released from the solid catalyst during the oxidative addition of the halide and then redeposited on the support as a consequence of the reductive elimination step when the new carbon–carbon bond is formed [
43,
47].
The catalytic system ultimately operates as an efficient heterogeneous system, and Pd content in the final product, determined by ICP analyses, is very low (3–5 ppm,
Table 5), proving that the ZPGly system is able to efficiently re-capture palladium. Moreover, the recovered catalytic system showed the same activity.
The flow system, with a separate column for the base K2CO3 and the catalyst Pd@ZPGly-15, allows optimizing the recovery and reuse of the solid catalyst and keeps its physical and chemical integrity over it use. In fact, the palladium leaching was much lower than that observed in batch conditions (3–5 vs. 11 ppm).
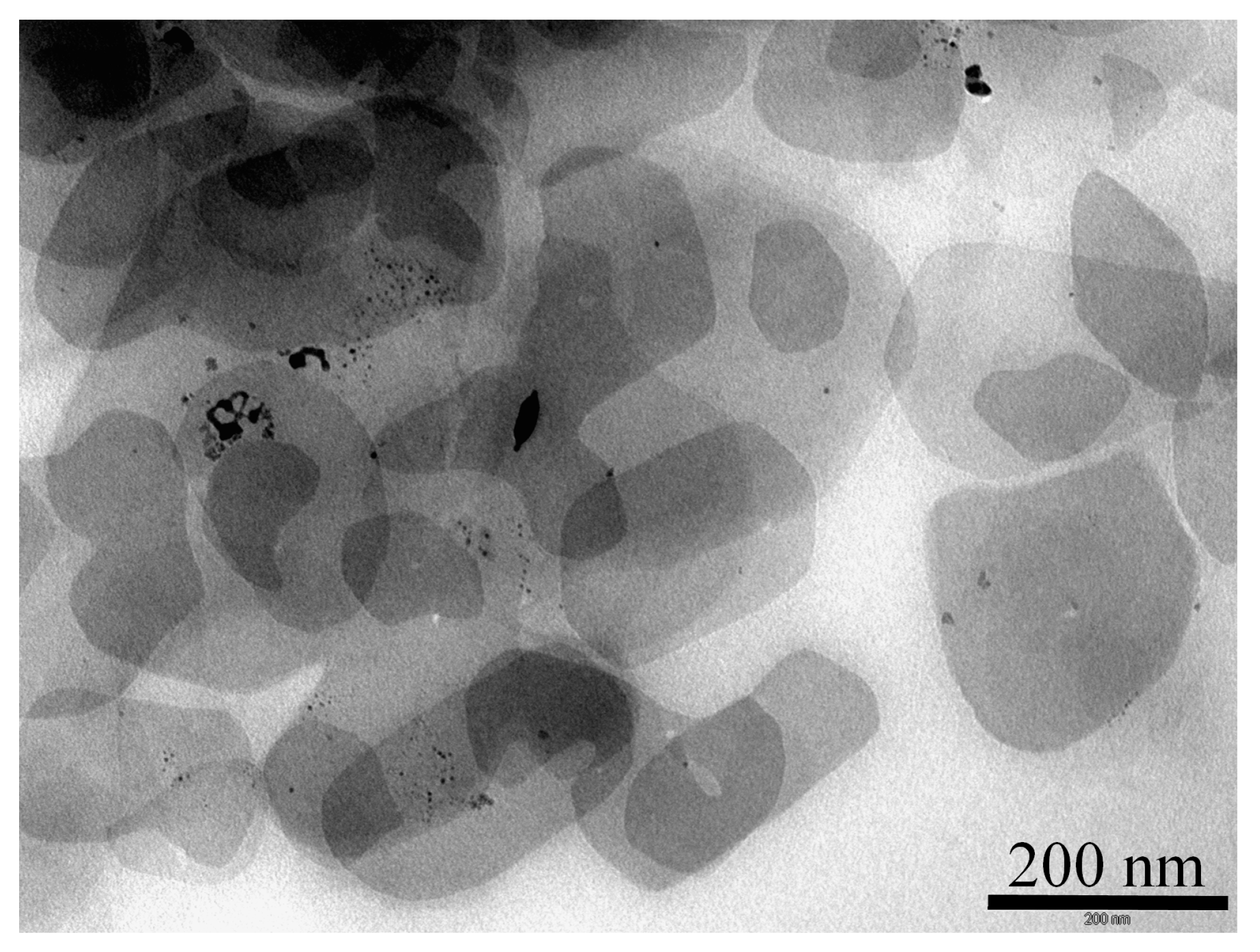
The TEM images of recovered Pd@ZPGly-15 after three catalytic cycles show that the ZPGly nanosheets’ structure and the distribution of PdNPs on the layers are maintained (
Figure 4).
Furthermore, to assess the waste minimization for both batch and flow protocols, we calculate the E-factor values. When the reaction was performed in homogeneous K2CO3/EtOH-H2O (1:1), the use of the not easily recoverable mixture of solvents and the need for extraction work-up lead to an E-factor value of 76. Using the heterogeneous K2CO3/aq. EtOH azeotrope system, the sustainability of the batch protocol has been greatly improved since no purification step is needed (E-factor 27.7), and this value is noticeable reduced in a large-scale flow protocol process by the recovery of azeotrope reaction medium by distillation (E-factor 3.2).
2.2.2. Heck Reaction
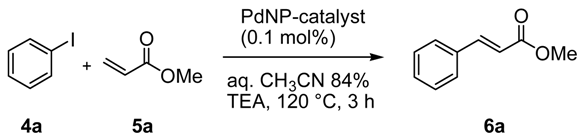
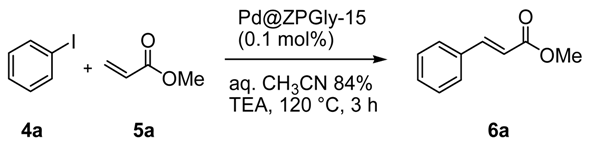
The catalytic activity of Pd@ZPGly catalysts has been investigated in the representative Heck reaction between iodobenzene (
4a) and methyl acrylate (
5a) in aqueous CH
3CN azeotrope (aq. CH
3CN 84%), an alternative to conventional polar aprotic media, as a safer and easily recoverable reaction medium [
15,
48].
In the presence of a soluble base, such us triethylamine (TEA), at 120 °C for 3 h, all of the Pd@ZPGly catalysts promoted the complete conversion (
Table 6, Entries 1–5), but after a 1-h reaction time, lower conversions were observed in the case of Pd@ZPGly-1R and -7R (
Table 6, Entries 4 and 5), again due to the presence of larger PdNPs aggregates. Pd/α-ZrPK catalyst, already reported by our group [
15], and Pd/C 10 wt% showed a comparable catalytic efficiency (
Table 6, Entry 6).
A remarkable difference was, however, observed in the Pd content determined by ICP-OES analyses of the crude products obtained by filtration and the usual extraction work-up procedure. In fact, the Pd@ZrPGly catalysts showed a Pd leaching in the products of 13–21 ppm, while in the case of Pd/α-ZrPK, the leaching was higher (34 ppm). Extremely high was the content of Pd in the product obtained by Pd/C catalysis (187 ppm,
Table 6, Entry 7). This result is very notable and proves that the presence of glycine groups on the surface of ZPGly nanosheets allows better efficiency in re-capturing palladium. As well as for the Suzuki–Miyaura reaction, the best result in terms of activity and release of Pd was obtained with Pd@ZrPGly15.
It is known that a “release and catch” mechanism is operative for a PdNP-catalyzed Heck reaction [
15,
36,
48] and that the required organic base strongly binds the soluble palladium species, facilitating their dissolution from the solid catalyst and consequently the leaching into the products. Besides, the base has the fundamental role of regenerating the active Pd(0) species [
49], with the formation of the corresponding ammonium salt.
In order to reduce the leaching of Pd and the amount of waste but also simplify the work-up procedure, we investigated the use of diethylaminomethyl-polystyrene (PS-TEA) as the heterogeneous base, which can be removed by simple filtration from the reaction mixture, and therefore, the isolation of the final product can be obtained simply by distillation of the azeotropic reaction medium. Since the polystyrene-bonded amino groups may act as capping agents for the Pd nanoparticles, a significantly reduced Pd leaching is expected [
50,
51,
52,
53].
As shown in
Table 6, when the reaction was performed in the presence of PS-TEA as the base, the catalytic activity of Pd@ZrPGly-15 was unchanged, while the Pd content in the final product was only 2 ppm (
Table 6, Entry 8 vs. Entry 3), much below the allowed limit value [
42]. The comparable Pd/α-ZrPK catalyst [
15] showed a lower efficiency to recapture Pd (6 ppm,
Table 6, Entry 9), proving that ZrPGly can act as an excellent scavenger for palladium nanoparticles.
Recycle test was performed in batch, by using homogeneous TEA as the base, to evaluate the robustness of Pd@ZPGly-15. For each cycle, the solid catalyst was separated by centrifugation, washed with aq. CH
3CN azeotrope and used again in the next cycle (
Table 7). Pd@ZPGly-15 was used six times always maintaining the same activity and achieving a TON (turnover number) of ca. 6000 and a TOF (turnover frequency) of ca. 333 h
−1. The ICP-OES analyses of crude products showed a stable palladium leaching at each cycle (10–12 ppm).
In
Figure 5, the TEM images of Pd@ZPGly-15 after six cycles are shown. The sample is almost unchanged, and the result is constituted by thin lamellae that support PdNPs with dimensions comparable to those of the fresh prepared catalyst.
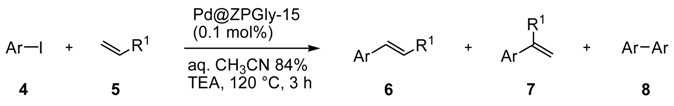
To confirm the efficiency of Pd@ZPGly-15 in the Heck coupling reaction, we extended the study to several aryl iodides
4a–
h with methyl acrylate (
5a) or with styrene (
5b) (
Table 8). The reactions of the aryl iodides (
4a–
h) with methyl acrylate (
5a) were carried at 120 °C for 3 h, in the presence of 0.1 mol % of Pd@ZPGly-15 and TEA as the base, obtaining from high to quantitative conversions of
4 and excellent yields of isolated products
6a–
h (
Table 8, Entries 1–8). For 4-nitro-iodobenzene (
4b), 2′-iodoacetophenone (
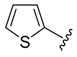
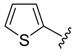
4f) and 2-iodothiophene (
4h), we observed the formation of the corresponding homocoupling products, the biphenyls
8b,
8f and
8h, and therefore, the products
6b,
6f and
6h were purified by column chromatography (
Table 8, Entries 2, 6 and 8).
When styrene (
5b) was used with iodobenzene (
4a) or 2-iodothiophene (
4h), a longer reaction time (4 h) was required to attain quantitative conversion (
Table 8, Entries 9 and 10). The formation of the
gem-products
7i–
l and, also, for 2-iodothiophene (
1h), the formation of the biphenyl
8h were observed (
Table 8, Entries 9 and 10).
Finally, we focused our attention on the definition of a flow procedure to optimize the isolation of the pure product while minimizing the waste production.
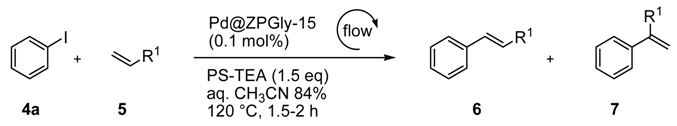
The catalytic behavior of Pd@ZPGly-15, under flow conditions, was studied in the Heck reaction of iodobenzene (4a) with methyl acrylate (5a) and styrene (5b) by using aq. CH3CN azeotrope as the reaction medium and PS-TEA as the heterogeneous base.
A mixture of the catalyst Pd@ZPGly-15 (0.1 mol %) and the supported base PS-TEA (1.5 equiv) was charged in a stainless-steel HPLC column, while an aq. CH
3CN azeotrope solution of the reactants (40 mmol iodobenzene (
4a) and 60 mmol methyl acrylate (
5a)) was charged in a second glass column, the reservoir (
Scheme 2).
The columns were connected to a pump and installed into a thermostated box at 120 °C (not shown in
Scheme 2 for clarity). The reaction mixture was pumped at a 2.0-mL/min flow rate through the columns for 1.5 h to obtain quantitative conversion of
4a to
6a (monitored by GLC,
Table 9, Entry 1).
The column that contained both the PS-TEA and catalyst was then washed with aq. CH
3CN azeotrope to clean the apparatus and completely recover the product. Finally, the product
6a was obtained in pure form, after distillation of the reaction medium and without any further purification step, in 87% yield (
Table 9, Entry 1).
The aqueous CH3CN azeotrope, recovered by distillation, was pure (GLC analysis), comparable to the starting azeotropic mixture and, therefore, fully reusable.
The reusability of the catalyst, in flow conditions, was evaluated for the reaction of iodobenzene (
4a) with methyl acrylate (
5a). The column that contained both the PS-TEA and catalyst was washed with a solution of TEA in aq. CH
3CN azeotrope to regenerate the supported ammine by removal of hydriodic acid (HI) formed during the Heck reaction and then washed with azeotrope to remove the residual TEA. After this treatment, a second batch of reactants was charged in the reservoir, and quantitative conversion was achieved again in 1.5 h (
Table 9, Entry 2). An unchanged catalytic activity was also obtained in the third run (
Table 9, Entry 3).
After three runs, iodobenzene (
4a) and styrene (
5b) were charged in the reservoir, and the reaction mixture was pumped through the column, containing the recovered PS-TEA and catalyst, to obtain after 2 h a quantitative conversion of
4a to
6i (86% yield,
Table 9, Entry 4).
To evaluate the sustainability of the processes, we calculated the E-factor value for both the batch and the flow protocols. When TEA was used as the base in the batch condition, an E-factor value of 70.4 was obtained, mainly due to the need of extraction work-up. Under flow conditions by using PS-TEA as the base, we were able to easily run the process on a larger scale (50 mmol) and to reduce this value to 4.7, since the reaction products can be recovered simply by distillation of the reaction medium that was totally recovered.
The ICP analyses of the final products revealed the presence of a very low Pd content of ca. 2–4 ppm, below the allowed limit value [
42] (
Table 9). The low residual of Pd in the products and the high activity over more runs is in agreement with the aforementioned “release and catch” mechanism [
36,
48].