Near-Graphite Coke Deposit on Nano-HZSM-5 Aggregates for Methanol to Propylene and Butylene Reaction
Abstract
:1. Introduction
2. Results and Discussion
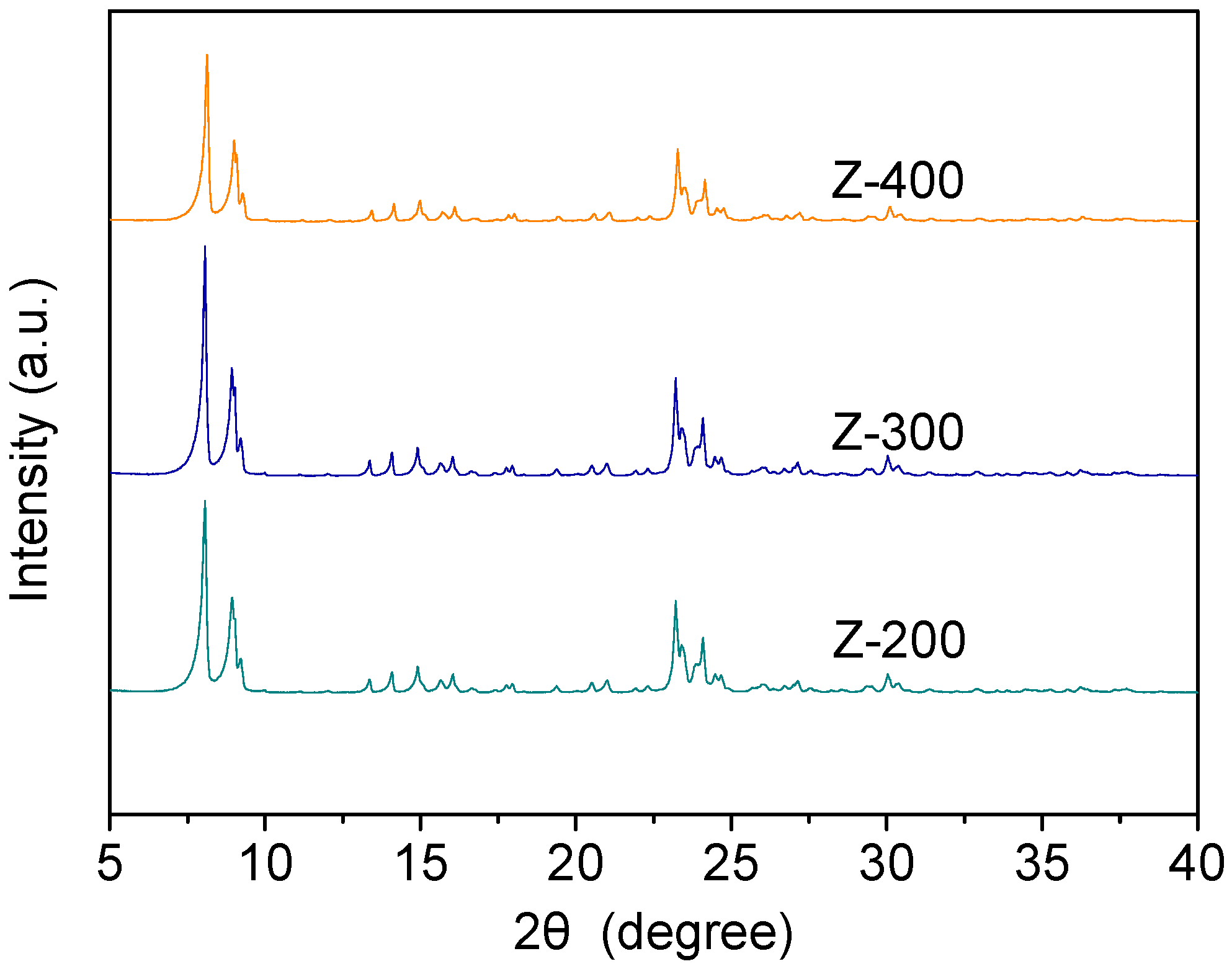
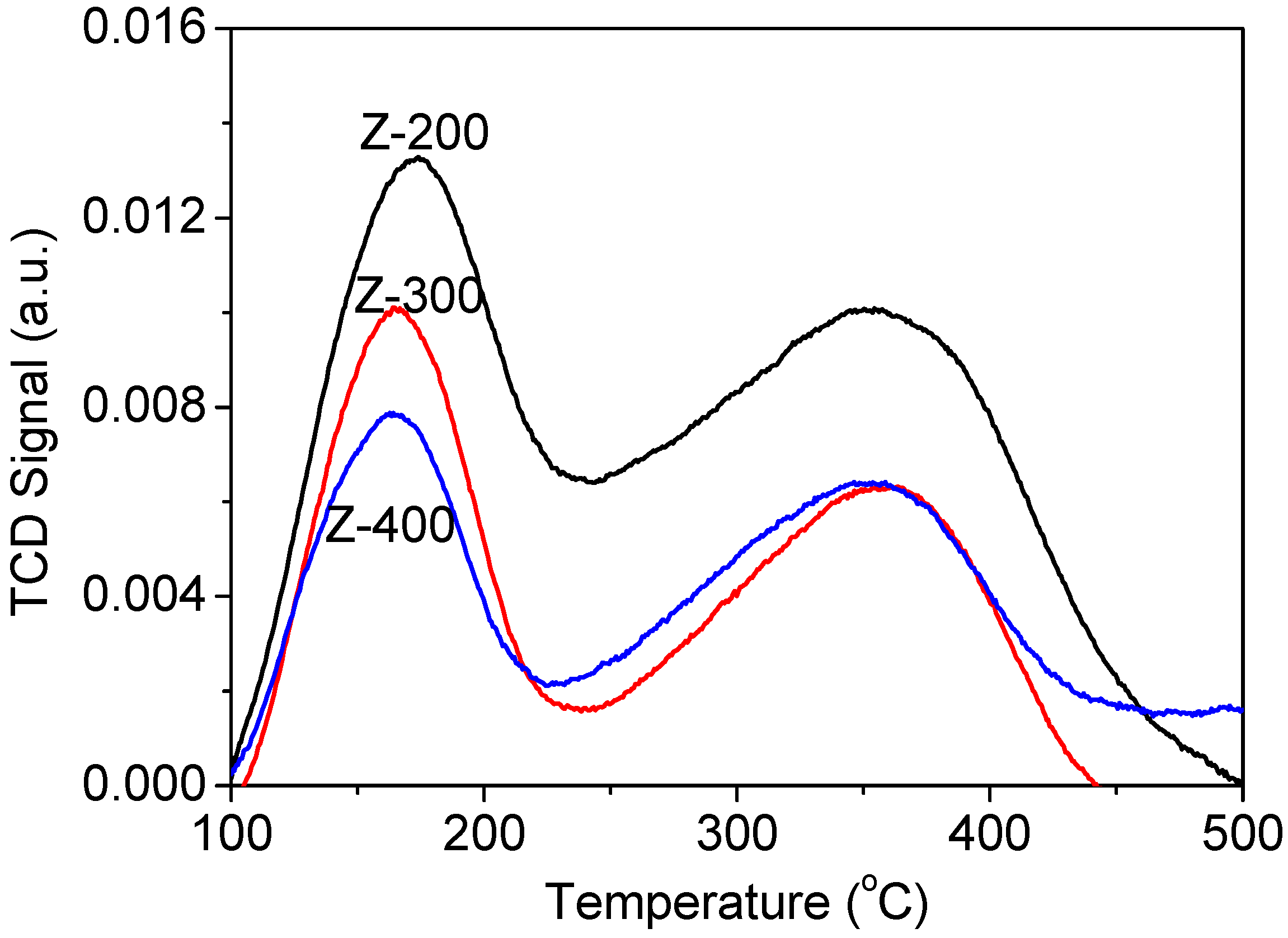
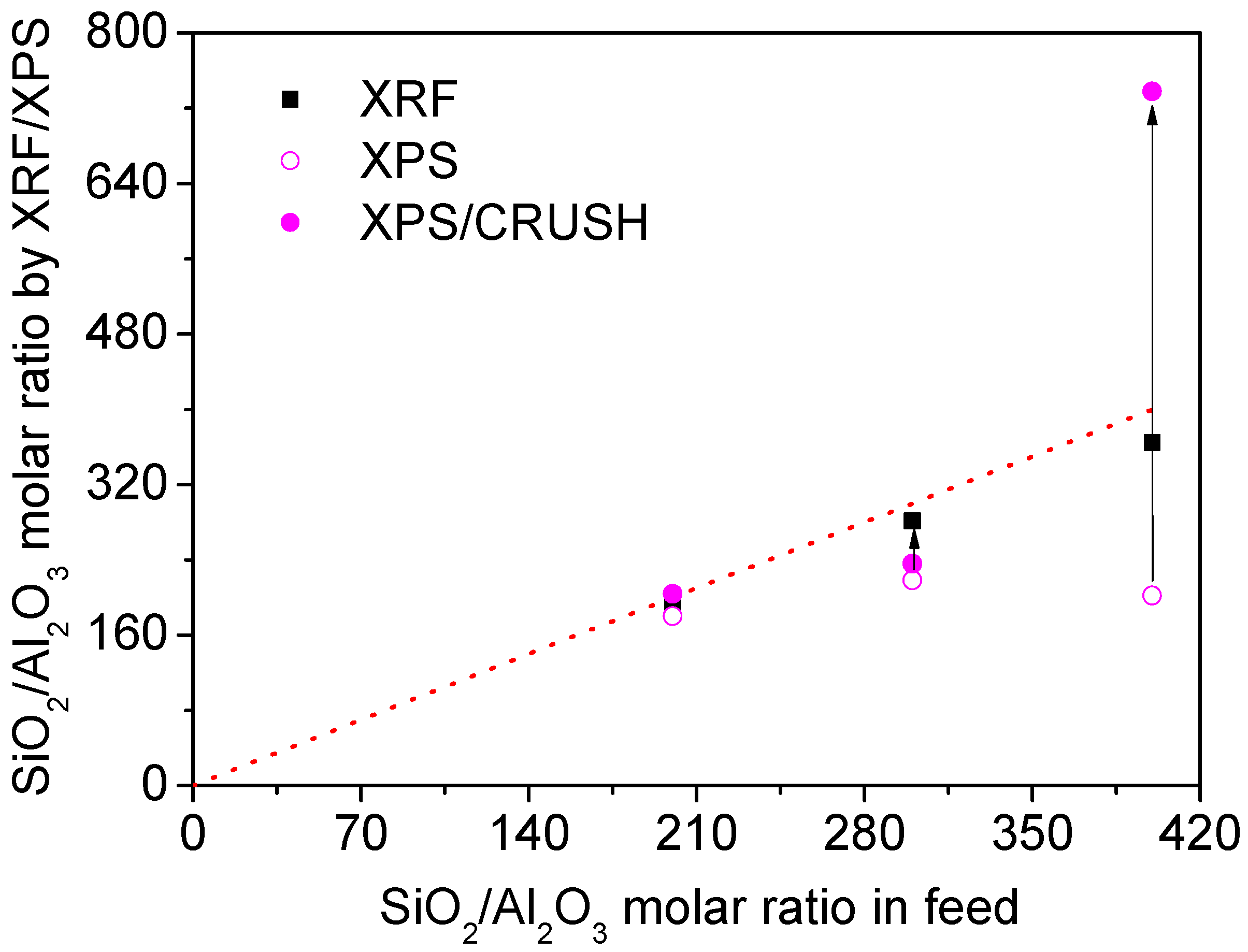
2.1. Structure and Acidity
2.2. Catalytic Performance
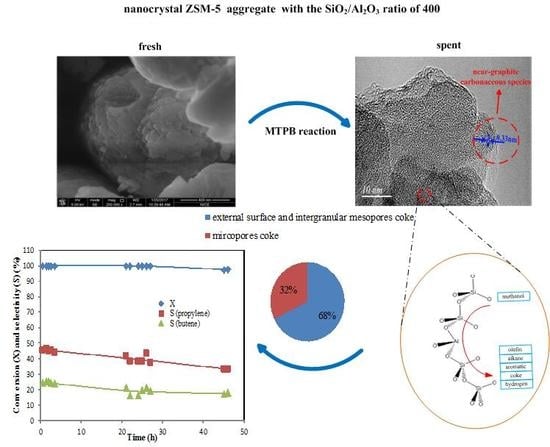
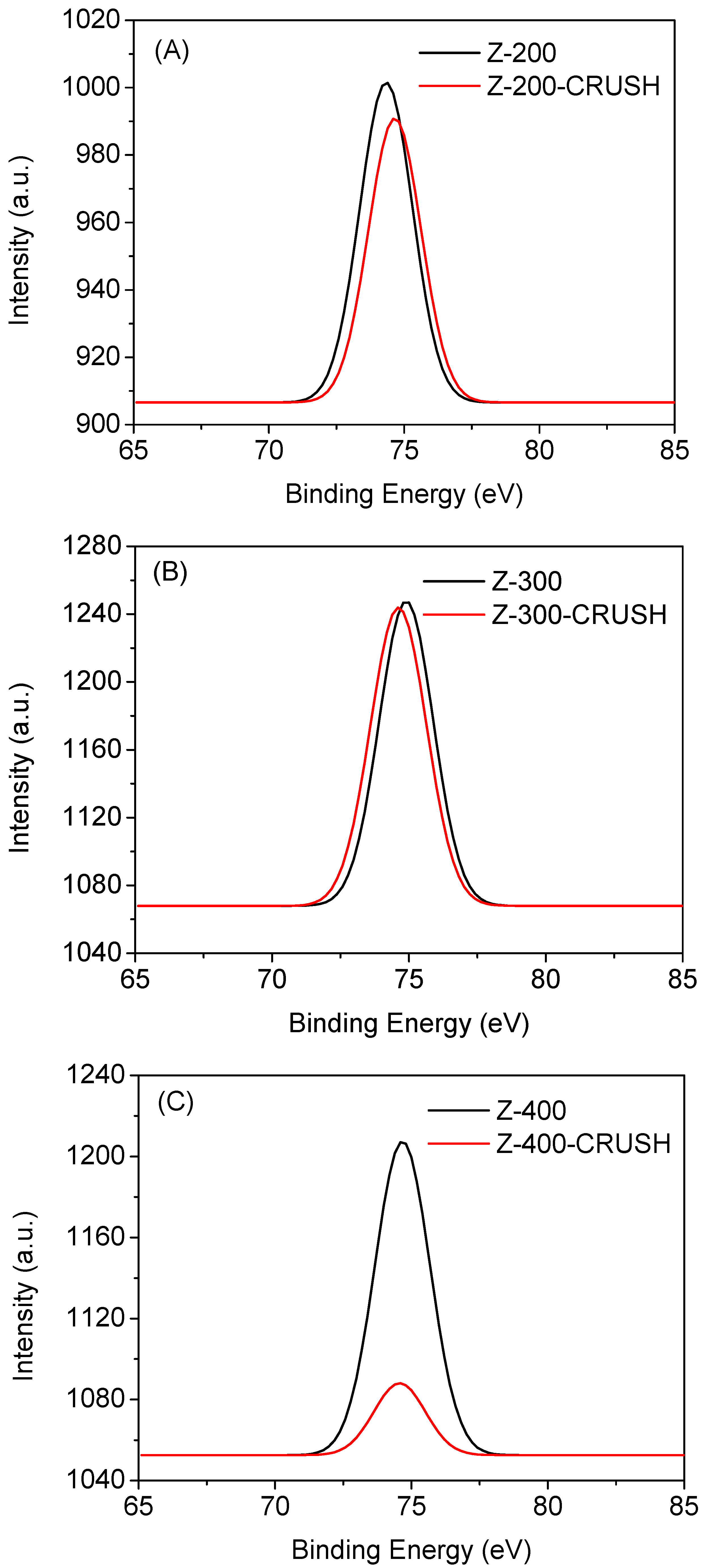
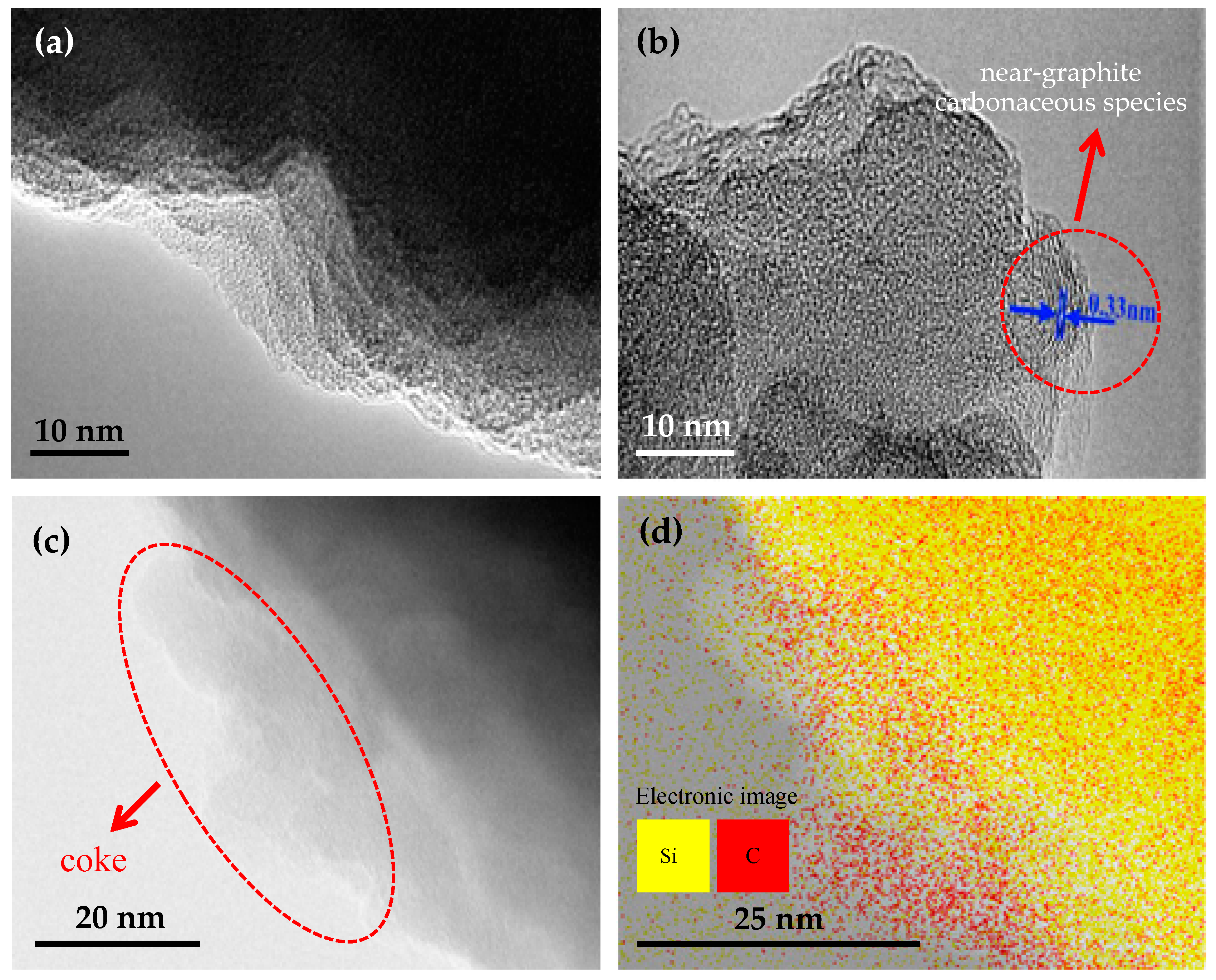
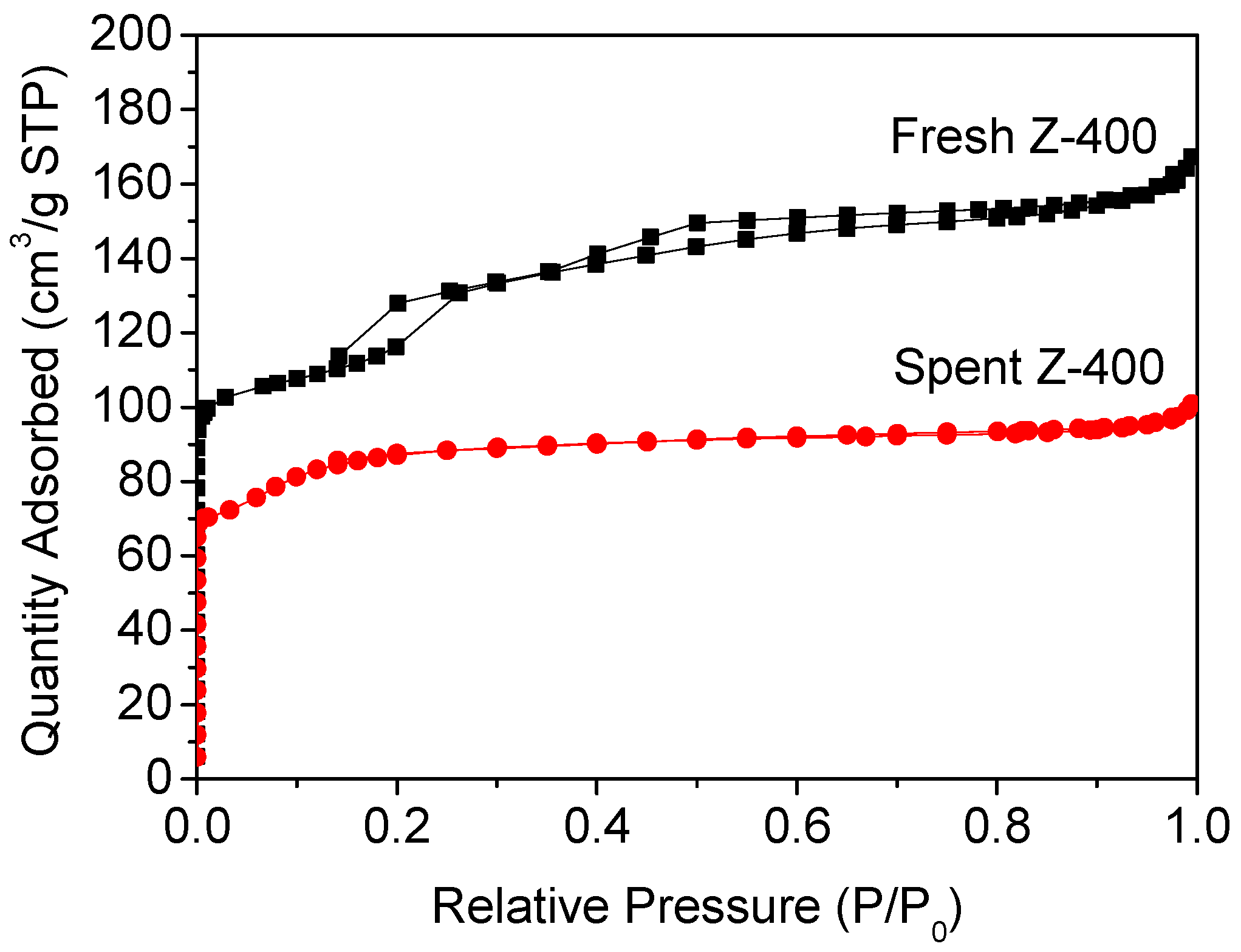
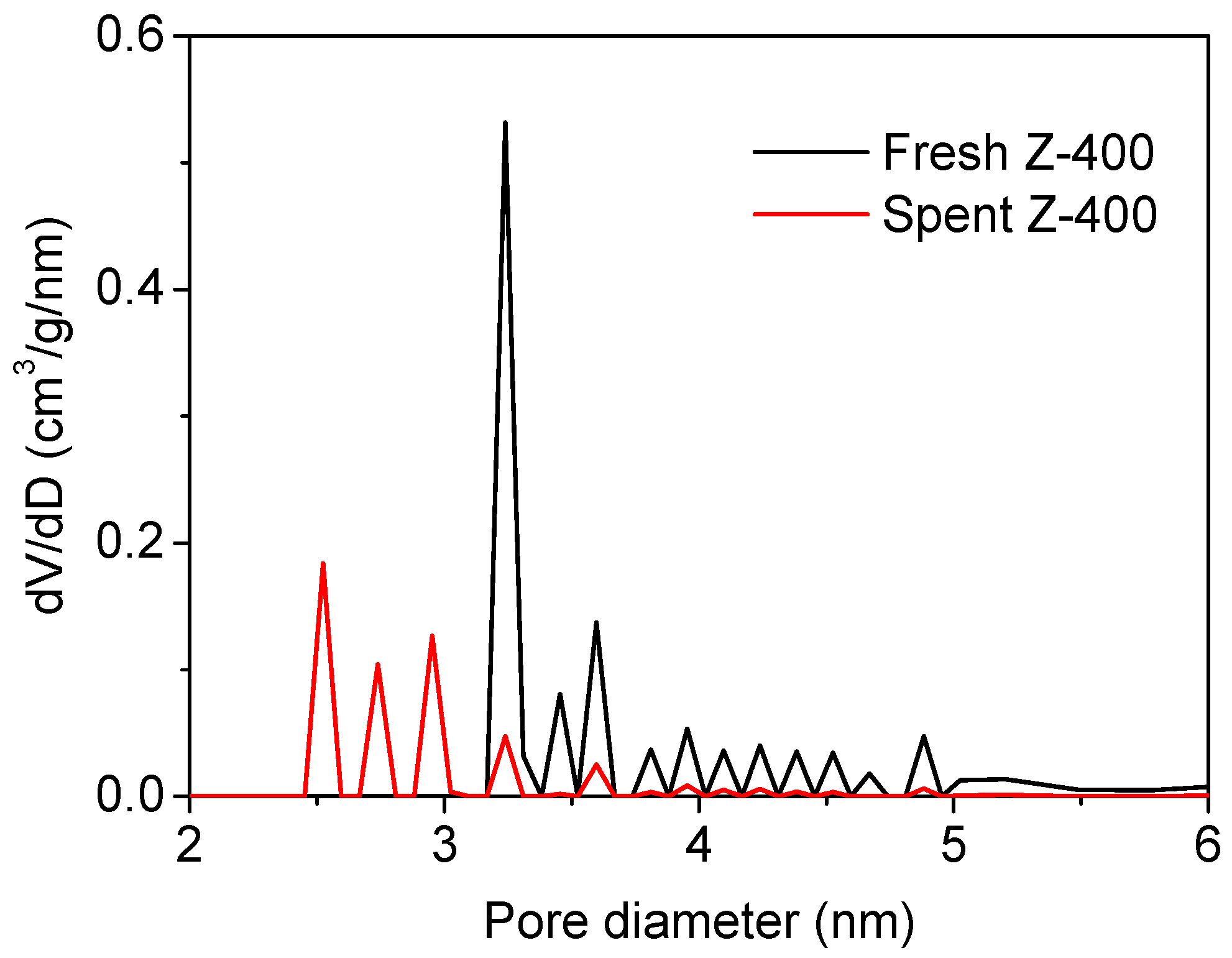
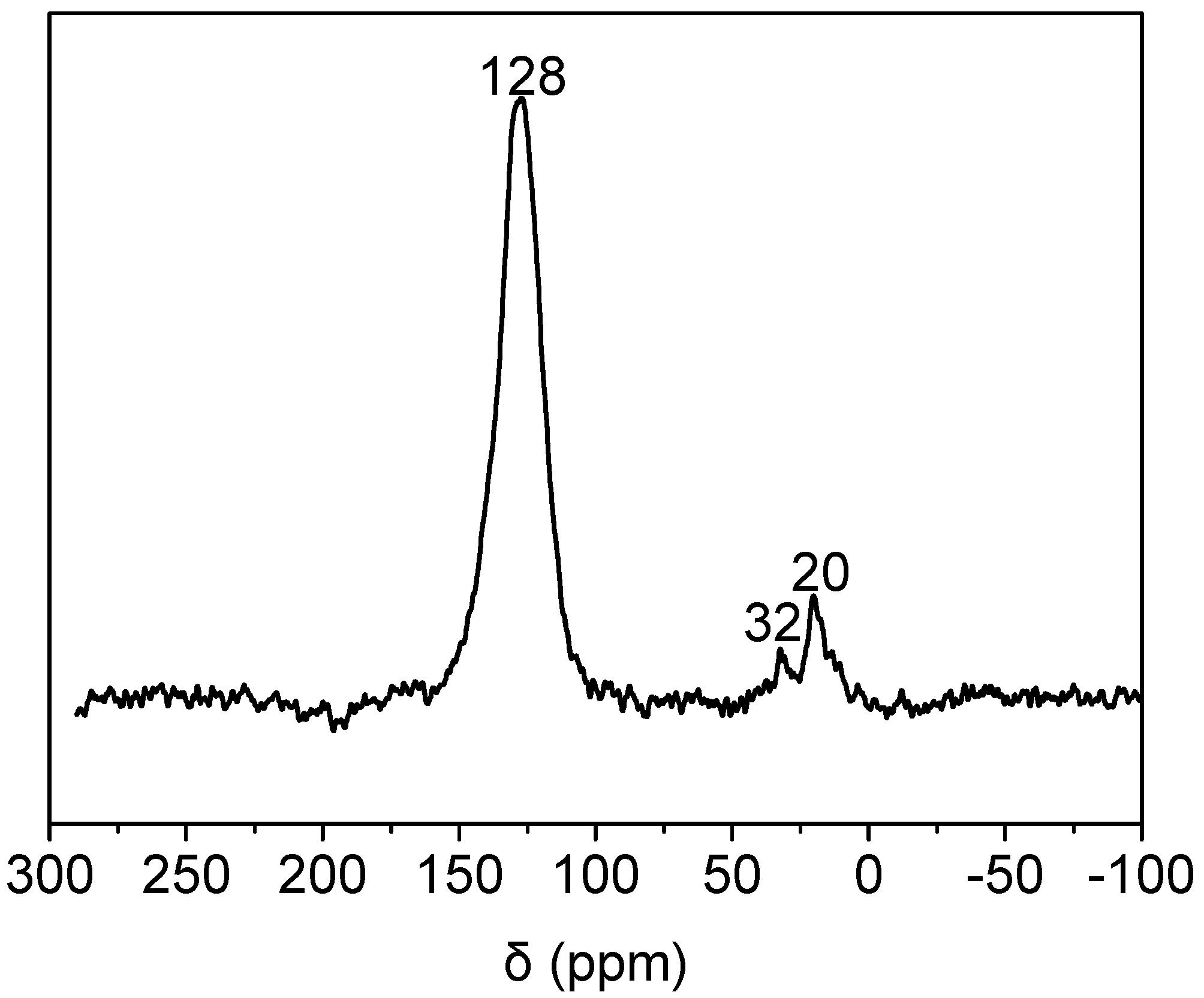
2.3. Coke Formation on HZSM-5
3. Experimental
3.1. Catalyst Preparation
3.2. Catalyst Characterization
3.3. Catalyst Evaluation
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Firoozi, M.; Baghalha, M.; Asadi, M. The effect of micro and nano particle sizes of H-ZSM-5 on the selectivity of MTP reaction. Catal. Commun. 2009, 10, 1582–1585. [Google Scholar] [CrossRef]
- Yaripour, F.; Shariatinia, Z.; Sahebdelfar, S.; Irandoukht, A. Conventional hydrothermal synthesis of nanostructured H-ZSM-5 catalysts using various templates for light olefins production from methanol. J. Nat. Gas Sci. Eng. 2015, 22, 260–269. [Google Scholar] [CrossRef]
- Losch, P.; Boltz, M.; Louis, B.; Chavan, S.; Olsbye, U. Catalyst optimization for enhanced propylene formation in the methanol-to-olefins reaction. Comptes Rendus Chim. 2015, 18, 330–335. [Google Scholar] [CrossRef]
- Xing, A.H.; Zhang, X.F.; Suo, Y.; Feng, Q.Y.; Shi, Y.L. Comprehensive utilization and prospect of C4 hydrocarbon. Clean Coal Technol. 2015, 21, 66–71. [Google Scholar]
- Hadi, N.; Niaei, A.; Nabavi, S.R.; Farzi, A.; Shirazia, M.N. Development of a New Kinetic Model for Methanol to Propylene Process on Mn/H-ZSM-5 Catalyst. Chem. Biochem. Eng. Q. 2014, 28, 53–63. [Google Scholar]
- Mokrani, T.; Scurrell, M. Gas conversion to liquid fuels and chemicals: The methanol route-catalysis and processes development. Catal. Rev. 2009, 51, 1–145. [Google Scholar] [CrossRef]
- Yong, X.J.; Du, Z.P.; Li, Y.; Luo, C.T. Application status of Lurgi methanol to propylene technology and development of key catalyst. Technol. Dev. Chem. Ind. 2013, 42, 18–21. [Google Scholar]
- Li, H.S.; He, S.C.; Ma, K.; Wu, Q.; Jiao, Q.Z.; Sun, K.N. Micro-mesoporous composite molecular sieves H-ZSM-5/MCM-41 for methanol dehydration to dimethyl ether: Effect of SiO2/Al2O3 ratio in H-ZSM-5. Appl. Catal. A 2013, 450, 152–159. [Google Scholar] [CrossRef]
- Mores, D.; Stavitski, E.; Kox, M.H.F.; Kornatowski, J.; Olsbye, U.; Weckhuysen, B.M. Space- and Time-Resolved In-situ Spectroscopy on the Coke Formation in Molecular Sieves: Methanol-to-Olefin Conversion over H-ZSM-5 and H-SAPO-34. Chem. Eur. J. 2008, 14, 11320–11327. [Google Scholar] [CrossRef] [PubMed]
- Bibby, D.M.; Howe, R.F.; McLellan, G.D. Coke formation in high-silica zeolites. Appl. Catal. A 1992, 93, 1–34. [Google Scholar] [CrossRef]
- Rownaghi, A.A.; Rezaei, F.; Hedlund, J. Uniform mesoporous ZSM-5 single crystals catalyst with high resistance to coke formation for methanol deoxygenation. Microporous Mesoporous Mater. 2012, 151, 26–33. [Google Scholar] [CrossRef]
- Bibby, D.M.; Milestone, N.B.; Patterson, J.E.; Aldridge, L.P. Coke formation in zeolite ZSM-5. J. Catal. 1986, 97, 493–502. [Google Scholar] [CrossRef]
- Sexton, B.A.; Hughes, A.E.; Bibby, D.M. An XPS study of coke distribution on ZSM-5. J. Catal. 1988, 109, 126–131. [Google Scholar] [CrossRef]
- Guisnet, M.; Magnoux, P. Organic chemistry of coke formation. Appl. Catal. A 2001, 212, 83–96. [Google Scholar] [CrossRef]
- Kim, J.; Choi, M.; Ryoo, R. Effect of mesoporosity against the deactivation of MFI zeolite catalyst during the methanol-to-hydrocarbon conversion process. J. Catal. 2010, 269, 219–228. [Google Scholar] [CrossRef]
- Hu, Z.; Zhang, H.; Wang, L.; Zhang, H.; Zhang, Y.; Xu, H.; Shen, W.; Tang, Y. Highly stable boron-modified hierarchical nanocrystalline ZSM-5 zeolite for the methanol to propylene reaction. Catal. Sci. Technol. 2014, 4, 2891–2895. [Google Scholar] [CrossRef]
- Ahmadpour, J.; Taghizadeh, M. Catalytic conversion of methanol to propylene over high-silica mesoporous ZSM-5 zeolites prepared by different combinations of mesogenous templates. J. Nat. Gas Sci. Eng. 2015, 23, 184–194. [Google Scholar] [CrossRef]
- Kim, Y.; Kim, J.C.; Jo, C.; Kim, T.W.; Kim, C.U.; Jeong, S.Y.; Chae, H.J. Structural and physicochemical effects of MFI zeolite nanosheets for the selective synthesis of propylene from methanol. Microporous Mesoporous Mater. 2016, 222, 1–8. [Google Scholar] [CrossRef]
- Hu, S.; Shan, J.; Zhang, Q.; Wang, Y.; Liu, Y.S.; Gong, Y.J.; Wu, Z.J.; Dou, T. Selective formation of propylene from methanol over high-silica nanosheets of MFI zeolite. Appl. Catal. A 2012, 445, 215–220. [Google Scholar] [CrossRef]
- Han, W.; Jia, Y.X.; Xiong, G.X.; Yang, W.S. Hydrothermal Stability of Meso-microporous Composites and Their Catalytic Cracking Performance. Chin. J. Catal. 2011, 32, 418–427. [Google Scholar] [CrossRef]
- Sang, Y.; Jiao, Q.; Li, H.; Wu, Q.; Zhao, Y.; Sun, K. HZSM-5/MCM-41 composite molecular sieves for the catalytic cracking of endothermic hydrocarbon fuels: Nano-ZSM-5 zeolites as the source. J. Nanopart. Res. 2014, 16, 1–11. [Google Scholar] [CrossRef]
- Rownaghi, A.A.; Hedlund, J. Methanol to gasoline-range hydrocarbons: Influence of nanocrystal size and mesoporosity on catalytic performance and product distribution of ZSM-5. Ind. Eng. Chem. Res. 2011, 50, 11872–11878. [Google Scholar] [CrossRef]
- Hughes, A.E.; Wilshier, K.G.; Sexton, B.A.; Smart, P. Aluminum distribution in ZSM-5 as determined by X-ray photoelectron spectroscopy. J. Catal. 1983, 80, 221–227. [Google Scholar] [CrossRef]
- Wang, C.F.; Zhang, Q.; Zhu, Y.F.; Zhang, D.K.; Chen, J.Y.; Chiang, F.K. p-Xylene selectivity enhancement in methanol toluene alkylation byseparation of catalysis function and shape-selective function. Mol. Catal. 2017, 433, 242–249. [Google Scholar] [CrossRef]
- Wang, Q.Y.; Xu, S.T.; Chen, J.R.; Wei, Y.X.; Li, J.Z.; Fan, D.; Yu, Z.X.; Qi, Y.; He, Y.L.; Xu, S.L.; et al. Synthesis of mesoporous ZSM-5 catalysts using different mesogenous templates and their application in methanol conversion for enhanced catalyst lifespan. RSC Adv. 2014, 4, 21479–21491. [Google Scholar] [CrossRef]
- Li, J.H.; Wang, Y.N.; Jia, W.Z.; Xi, Z.W.; Chen, H.H.; Zhu, Z.R.; Hu, Z.H. Effect of external surface of HZSM-5 zeolite on product distribution in the conversion of methanol to hydrocarbons. J. Energy Chem. 2014, 23, 771–780. [Google Scholar] [CrossRef]
- Choi, M.; Na, K.; Kim, J.; Sakamoto, Y.; Terasaki, O.; Ryoo, R. Stable single-unit-cell nanosheets of zeolite MFI as active and long-lived catalysts. Nature 2009, 461, 246–249. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Jiang, Y.J.; Hunger, M. Mechanistic investigations of the methanol-to-olefin (MTO) process on acidic zeolite catalysts by in situ solid-state NMR spectroscopy. Catal. Today 2006, 113, 102–114. [Google Scholar] [CrossRef]


| Product Selectivity (%) a | Z-200 | Z-300 | Z-400 |
|---|---|---|---|
| C3H6 | 29.5 | 37.6 | 42.9 |
| C4H8 | 14.5 | 19.6 | 21.7 |
| C5H10 | 4.6 | 5.2 | 6.6 |
| C6+ | 7.2 | 7.7 | 8.4 |
| C2H4 | 10.0 | 8.6 | 7.2 |
| C2H6 | 0.11 | 0.08 | 0.07 |
| C3H8 | 1.2 | 1.0 | 0.77 |
| C4H10 | 4.6 | 4.3 | 3.3 |
| Lifetime (h) b | 20 | 28 | 32 |
| Methanol/catalyst ratio c | 60 | 84 | 96 |
| Samples | SBET (m2/g) | Smicro (m2/g) | Sexter (m2/g) | Vtot (cm3/g) | Vmicro (cm3/g) | Vmeso (cm3/g) |
|---|---|---|---|---|---|---|
| Fresh Z-200 | 451.7 | 300.8 | 150.9 | 0.275 | 0.119 | 0.156 |
| Coke deposited Z-200 | 299.2 | 68.0 | 231.2 | 0.173 | 0.030 | 0.143 |
| Fresh Z-300 | 431.2 | 268.7 | 162.5 | 0.305 | 0.109 | 0.196 |
| Coke deposited Z-300 | 311.8 | 26.6 | 285.2 | 0.171 | 0.027 | 0.144 |
| Fresh Z-400 | 435.9 | 262.6 | 173.3 | 0.259 | 0.103 | 0.156 |
| Coke deposited Z-400 | 303.4 | 177.7 | 125.7 | 0.156 | 0.080 | 0.076 |
| Sample | Total Coke (%) | Coke Inside Micropore (gcoke/gcat) | Coke on External Surface (gcoke/gcat) |
|---|---|---|---|
| Z-200 | 11.1 | 0.109 | 0.002 |
| Z-300 | 11.1 | 0.100 | 0.011 |
| Z-400 | 8.7 | 0.028 | 0.059 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sang, Y.; Xing, A.; Wang, C.; Han, Z.; Wu, Y. Near-Graphite Coke Deposit on Nano-HZSM-5 Aggregates for Methanol to Propylene and Butylene Reaction. Catalysts 2017, 7, 171. https://doi.org/10.3390/catal7060171
Sang Y, Xing A, Wang C, Han Z, Wu Y. Near-Graphite Coke Deposit on Nano-HZSM-5 Aggregates for Methanol to Propylene and Butylene Reaction. Catalysts. 2017; 7(6):171. https://doi.org/10.3390/catal7060171
Chicago/Turabian StyleSang, Yu, Aihua Xing, Chuanfu Wang, Zhihua Han, and Yulong Wu. 2017. "Near-Graphite Coke Deposit on Nano-HZSM-5 Aggregates for Methanol to Propylene and Butylene Reaction" Catalysts 7, no. 6: 171. https://doi.org/10.3390/catal7060171
APA StyleSang, Y., Xing, A., Wang, C., Han, Z., & Wu, Y. (2017). Near-Graphite Coke Deposit on Nano-HZSM-5 Aggregates for Methanol to Propylene and Butylene Reaction. Catalysts, 7(6), 171. https://doi.org/10.3390/catal7060171