Approaching Immobilization of Enzymes onto Open Porous Basotect®
Abstract
1. Introduction
2. Results
2.1. Characterization of Basotect®
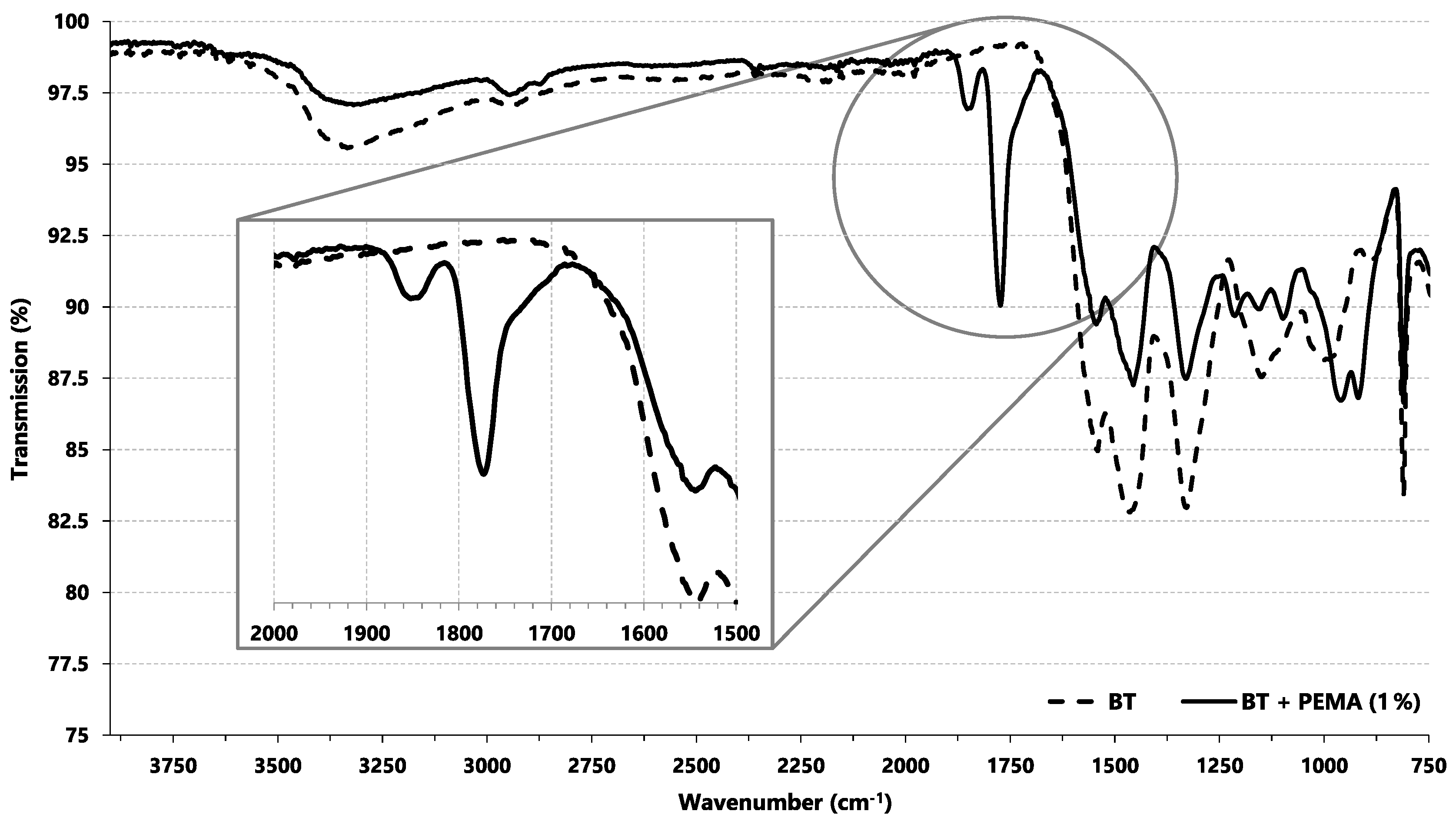
2.2. PEMA Functionalization of Basotect® and Characterization
2.3. Enzyme Immobilization
2.3.1. Adsorptive and Covalent Immobilization
2.3.2. Surface Coverage with Enzyme
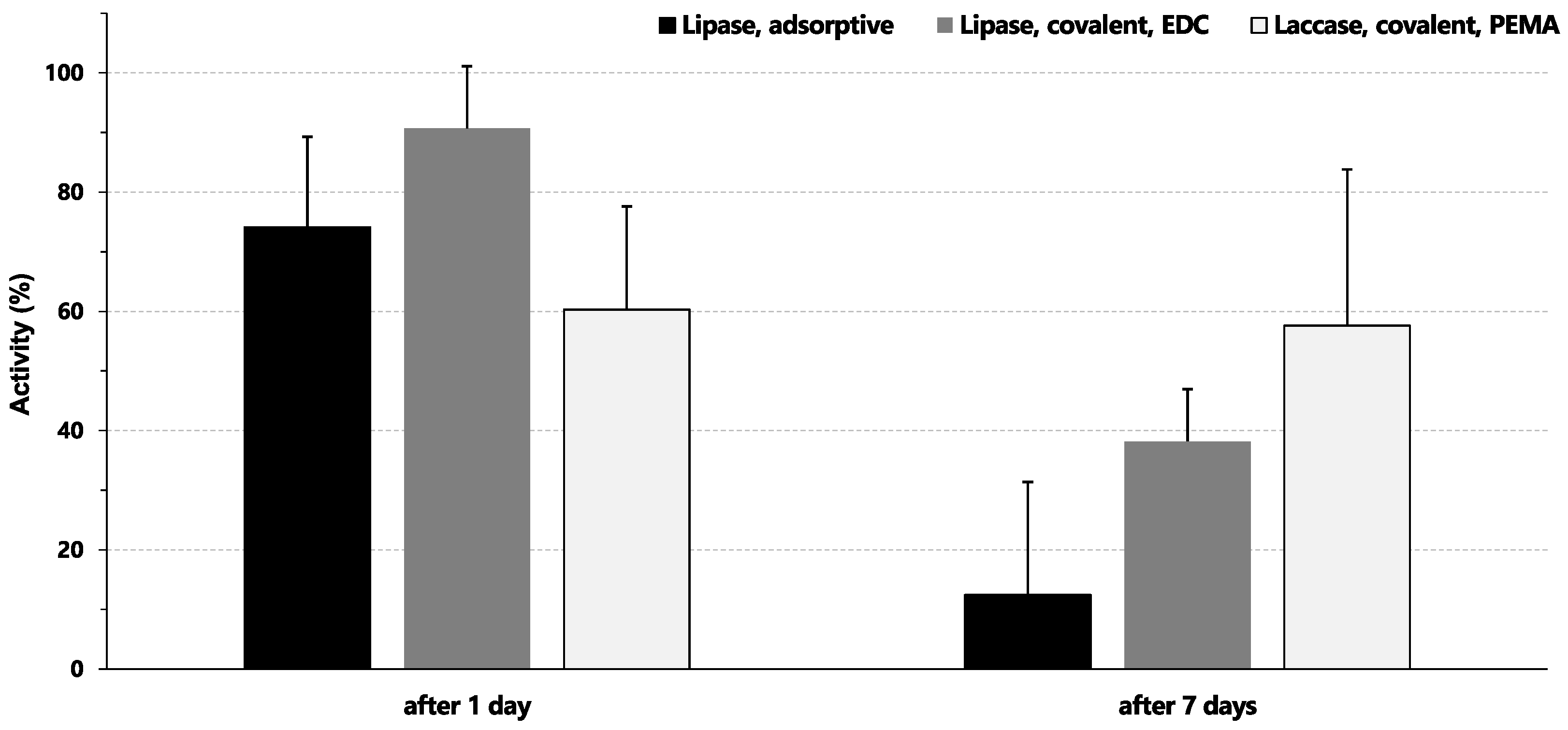
2.4. Storage Stability of Enzyme-Basotect® Interactions
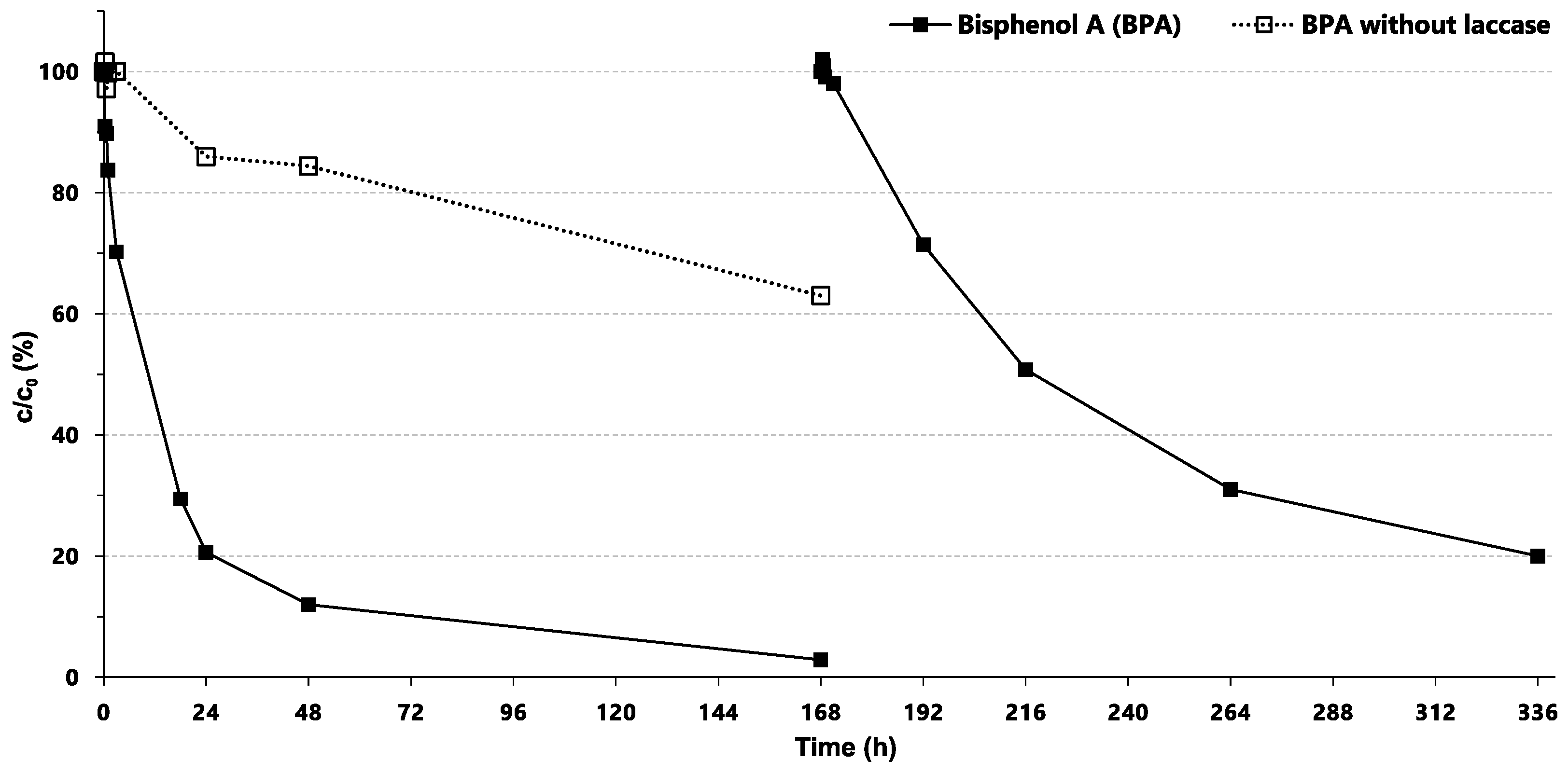
2.5. Application of Immobilized Laccase for the Elimination of Bisphenol A
3. Discussion
3.1. Characterization and Functionalization of Basotect®
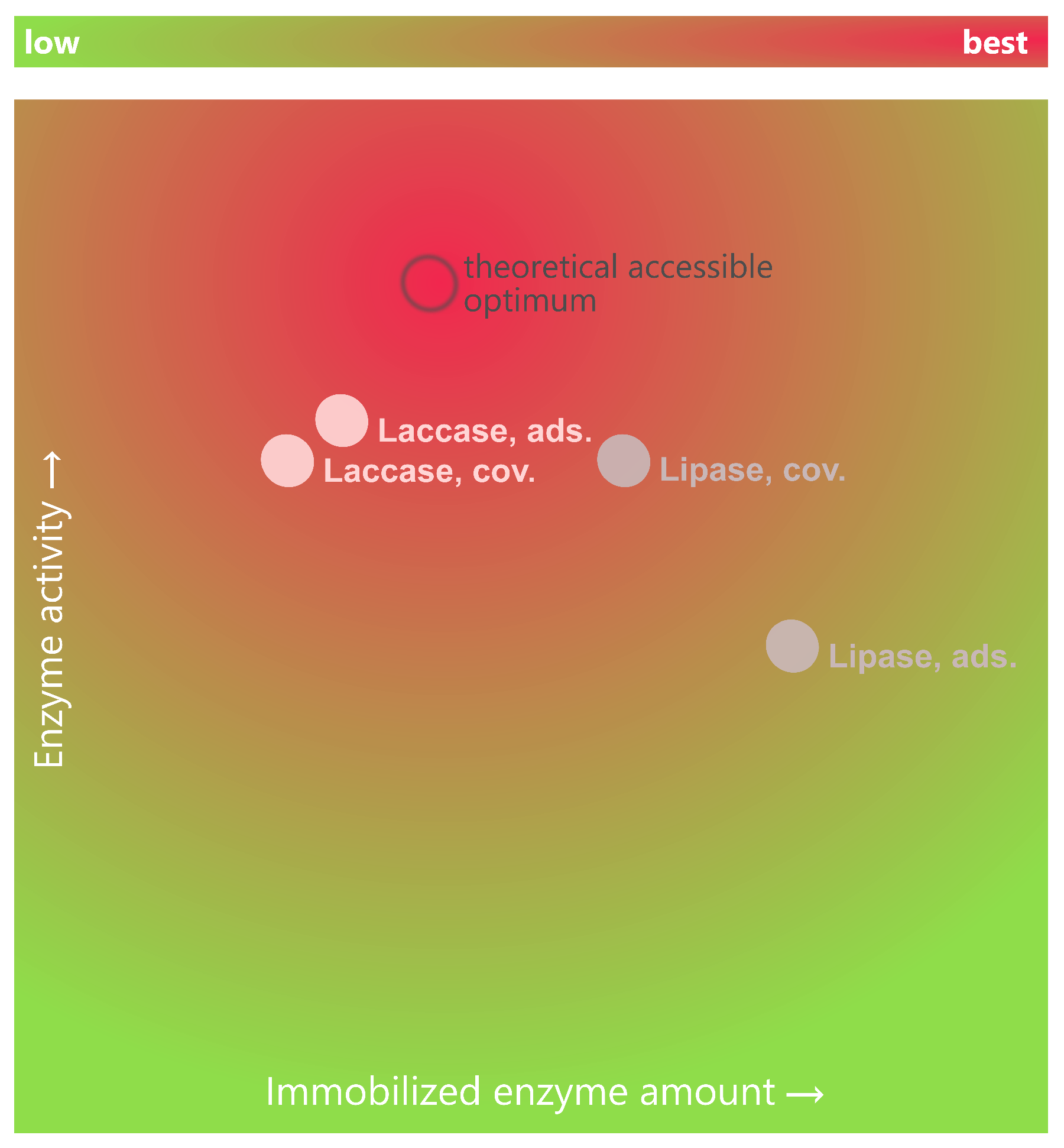
3.2. Enzyme Immobilization
3.3. Analysis of Basotect® Surface Coverage with Enzyme
3.4. Stability of Immobilized Enzymes
3.5. Prospect and Applications
4. Materials and Methods
4.1. Size and HCl Treatment of Basotect® Samples
4.2. Scanning Electron Microscopy of Basotect®
4.3. Amino Group Detection
4.3.1. ATTO-TAG FQ Fluorescence Labeling
4.3.2. Orange II Assay
4.4. PEMA Functionalization
4.4.1. ATR-FTIR Analysis
4.4.2. TBO Assay
4.5. Enzyme Immobilization
4.5.1. Adsorptive Immobilization
4.5.2. Covalent Immobilization
4.5.3. SDS Treatment
4.6. Storage Conditions
4.6.1. Laccase on PEMA-BT
4.6.2. Lipase on BT
4.7. Protein Quantification
4.8. Enzyme Activity
4.8.1. Lipase
4.8.2. Laccase
4.9. Calculation of Theoretical and Actual Enzyme Surface Concentration
4.10. Degradation of Bisphenol A Utilizing Laccase on PEMA-BT
4.10.1. Experimental Setup
4.10.2. LC-MS/MS Detection of BPA
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Cantone, S.; Ferrario, V.; Corici, L.; Ebert, C.; Fattor, D.; Spizzo, P.; Gardossi, L. Efficient immobilisation of industrial biocatalysts: criteria and constraints for the selection of organic polymeric carriers and immobilisation methods. Chem. Soc. Rev. 2013, 42, 6262–6276. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Galan, C.; Berenguer-Murcia, N.; Fernandez-Lafuente, R.; Rodrigues, R.C. Potential of different enzyme immobilization strategies to improve enzyme performance. Adv. Synth. Catal. 2011, 353, 2885–2904. [Google Scholar] [CrossRef]
- DiCosimo, R.; McAuliffe, J.; Poulose, A.J.; Bohlmann, G. Industrial use of immobilized enzymes. Chem. Soc. Rev. 2013, 42, 6437–6474. [Google Scholar] [CrossRef] [PubMed]
- Liese, A.; Hilterhaus, L. Evaluation of immobilized enzymes for industrial applications. Chem. Soc. Rev. 2013, 42, 6236–6249. [Google Scholar] [CrossRef] [PubMed]
- Hanefeld, U.; Gardossi, L.; Magner, E. Understanding enzyme immobilisation. Chem. Soc. Rev. 2009, 38, 453–468. [Google Scholar] [CrossRef] [PubMed]
- Tischer, W.; Wedekind, F. Immobilized Enzymes: Methods and Applications. In Biocatalysis—From Discovery to Application; Springer: Berlin/Heidelberg, Germany, 1999; pp. 95–126. [Google Scholar]
- Rodrigues, R.C.; Berenguer-Murcia, N.; Fernandez-Lafuente, R. Coupling chemical modification and immobilization to improve the catalytic performance of enzymes. Adv. Synth. Catal. 2011, 353, 2216–2238. [Google Scholar] [CrossRef]
- Balcão, V.M.; Vila, M.M. Structural and functional stabilization of protein entities: State-of-the-art. Adv. Drug Deliv. Rev. 2015, 93, 25–41. [Google Scholar] [CrossRef] [PubMed]
- Stepankova, V.; Bidmanova, S.; Koudelakova, T.; Prokop, Z.; Chaloupkova, R.; Damborsky, J. Strategies for stabilization of enzymes in organic solvents. ACS Catal. 2013, 3, 2823–2836. [Google Scholar] [CrossRef]
- Brady, D.; Jordaan, J. Advances in enzyme immobilisation. Biotechnol. Lett. 2009, 31, 1639–1650. [Google Scholar] [CrossRef] [PubMed]
- Bayramoglu, G.; Kacar, Y.; Denizli, A.; Yakup Arica, M. Covalent immobilization of lipase onto hydrophobic group incorporated poly(2-hydroxyethyl methacrylate) based hydrophilic membrane matrix. J. Food Eng. 2002, 52, 367–374. [Google Scholar] [CrossRef]
- Min, K.; Yoo, Y.J. Recent progress in nanobiocatalysis for enzyme immobilization and its application. Biotechnol. Bioprocess Eng. 2014, 19, 553–567. [Google Scholar] [CrossRef]
- Rodrigues, R.C.; Ortiz, C.; Berenguer-Murcia, A.; Torres, R.; Fernandez-Lafuente, R. Modifying enzyme activity and selectivity by immobilization. Chem. Soc. Rev. 2013, 42, 6290–6307. [Google Scholar] [CrossRef] [PubMed]
- Pompe, T.; Zschoche, S.; Herold, N.; Salchert, K.; Gouzy, M.F.; Sperling, C.; Werner, C. Maleic anhydride copolymersa versatile platform for molecular biosurface engineering. Biomacromolecules 2003, 4, 1072–1079. [Google Scholar] [CrossRef] [PubMed]
- Wetzel, S.; Allertz, P.J.; Koschnicke, S.; Tasso, M.; Salchert, K. Immobilized enzymes—Valuable tools for the indication of temperature events. Biocatal. Biotransform. 2013, 31, 100–113. [Google Scholar] [CrossRef]
- Pompe, T.; Renner, L.; Grimmer, M.; Herold, N.; Werner, C. Functional films of maleic anhydride copolymers under physiological conditions. Macromol. Biosci. 2005, 5, 890–895. [Google Scholar] [CrossRef] [PubMed]
- Fernandez-Lafuente, R. Lipase from Thermomyces lanuginosus: Uses and prospects as an industrial biocatalyst. J. Mol. Catal. B Enzym. 2010, 62, 197–212. [Google Scholar] [CrossRef]
- Bezerra, R.M.; Neto, D.M.A.; Galvão, W.S.; Rios, N.S.; de Carvalho, A.C.L.M.; Correa, M.A.; Bohn, F.; Fernandez-Lafuente, R.; Fechine, P.B.; de Mattos, M.C.; et al. Design of a lipase-nano particle biocatalysts and its use in the kinetic resolution of medicament precursors. Biochem. Eng. J. 2017, 125, 104–115. [Google Scholar] [CrossRef]
- Tacias-Pascacio, V.G.; Virgen-OrtÃz, J.J.; Jiménez-Pérez, M.; Yates, M.; Torrestiana-Sanchez, B.; Rosales-Quintero, A.; Fernandez-Lafuente, R. Evaluation of different lipase biocatalysts in the production of biodiesel from used cooking oil: Critical role of the immobilization support. Fuel 2017, 200, 1–10. [Google Scholar] [CrossRef]
- Rodríguez Couto, S.; Toca Herrera, J.L. Industrial and biotechnological applications of laccases: A review. Biotechnol. Adv. 2006, 24, 500–513. [Google Scholar] [CrossRef] [PubMed]
- Gamallo, M.; Moldes-Diz, Y.; Eibes, G.; Feijoo, G.; Lema, J.M.; Moreira, M.T. Sequential reactors for the removal of endocrine disrupting chemicals by laccase immobilized onto fumed silica microparticles. Biocatal. Biotransform. 2017, 1–11. [Google Scholar] [CrossRef]
- Dai, Y.; Yao, J.; Song, Y.; Liu, X.; Wang, S.; Yuan, Y. Enhanced performance of immobilized laccase in electrospun fibrous membranes by carbon nanotubes modification and its application for bisphenol A removal from water. J. Hazard. Mater. 2016, 317, 485–493. [Google Scholar] [CrossRef] [PubMed]
- Torrinha, L.; Montenegro, M.C.B.S.M.; AraÃjo, A.N. Implementation of a simple nanostructured bio-electrode with immobilized Rhus vernicifera laccase for oxygen sensing applications. Electroanalysis 2017, 29, 1566–1572. [Google Scholar] [CrossRef]
- Fernandez-Lafuente, R.; Armisen, P.; Sabuquillo, P.; Fernandez-Lorente, G.; Guisan, J.M. Immobilization of lipases by selective adsorption on hydrophobic supports. Chem. Phys. Lipids 1998, 93, 185–197. [Google Scholar] [CrossRef]
- Minovska, V.; Winkelhausen, E.; Kuzmanova, S. Lipase immobilized by different techniques on various support materials applied in oil hydrolysis. J. Serbian Chem. Soc. 2005, 70, 609–624. [Google Scholar] [CrossRef]
- Kosaka, P.M.; Kawano, Y.; El Seoud, O.A.; Petri, D.F.S. Catalytic Activity of Lipase Immobilized onto Ultrathin Films of Cellulose Esters. Langmuir 2007, 23, 12167–12173. [Google Scholar] [CrossRef] [PubMed]
- Xu, R.; Si, Y.; Wu, X.; Li, F.; Zhang, B. Triclosan removal by laccase immobilized on mesoporous nanofibers: Strong adsorption and efficient degradation. Chem. Eng. J. 2014, 255, 63–70. [Google Scholar] [CrossRef]
- De Cazes, M.; Belleville, M.P.; Mougel, M.; Kellner, H.; Sanchez-Marcano, J. Characterization of laccase-grafted ceramic membranes for pharmaceuticals degradation. J. Membr. Sci. 2015, 476, 384–393. [Google Scholar] [CrossRef]
- Bautista, L.F.; Morales, G.; Sanz, R. Immobilization strategies for laccase from Trametes versicolor on mesostructured silica materials and the application to the degradation of naphthalene. Bioresour. Technol. 2010, 101, 8541–8548. [Google Scholar] [CrossRef] [PubMed]
- Nicolucci, C.; Rossi, S.; Menale, C.; Godjevargova, T.; Ivanov, Y.; Bianco, M.; Mita, L.; Bencivenga, U.; Mita, D.; Diano, N. Biodegradation of bisphenols with immobilized laccase or tyrosinase on polyacrylonitrile beads. Biodegradation 2011, 22, 673–683. [Google Scholar] [CrossRef] [PubMed]
- Lloret, L.; Hollmann, F.; Eibes, G.; Feijoo, G.; Moreira, M.; Lema, J.M. Immobilisation of laccase on Eupergit supports and its application for the removal of endocrine disrupting chemicals in a packed-bed reactor. Biodegradation 2012, 23, 373–386. [Google Scholar] [CrossRef] [PubMed]
- Touahar, I.E.; Haroune, L.; Ba, S.; Bellenger, J.P.; Cabana, H. Characterization of combined cross-linked enzyme aggregates from laccase, versatile peroxidase and glucose oxidase, and their utilization for the elimination of pharmaceuticals. Sci. Total Environ. 2014, 481, 90–99. [Google Scholar] [CrossRef] [PubMed]
- Wong, K.S.; Huang, Q.; Au, C.H.; Wang, J.; Kwan, H.S. Biodegradation of dyes and polyaromatic hydrocarbons by two allelic forms of Lentinula edodes laccase expressed from Pichia pastoris. Bioresour. Technol. 2012, 104, 157–164. [Google Scholar] [CrossRef] [PubMed]
- Demarche, P.; Junghanns, C.; Nair, R.R.; Agathos, S.N. Harnessing the power of enzymes for environmental stewardship. Biotechnol. Adv. 2012, 30, 933–953. [Google Scholar] [CrossRef] [PubMed]
- Cardinal-Watkins, C.; Nicell, J.A. Enzyme-catalyzed oxidation of 17Î2-Estradiol using immobilized laccase from Trametes versicolor. Enzym. Res. 2011, 2011, 11. [Google Scholar] [CrossRef] [PubMed]
- Macellaro, G.; Pezzella, C.; Cicatiello, P.; Sannia, G.; Piscitelli, A. Fungal laccases degradation of endocrine disrupting compounds. BioMed Res. Int. 2014, 2014, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, L.N.; Hai, F.I.; Price, W.E.; Kang, J.; Leusch, F.D.; Roddick, F.; van de Merwe, J.P.; Magram, S.F.; Nghiem, L.D. Degradation of a broad spectrum of trace organic contaminants by an enzymatic membrane reactor: Complementary role of membrane retention and enzymatic degradation. Int. Biodeterior. Biodegrad. 2015, 99, 115–122. [Google Scholar] [CrossRef]
- Pham, T.N.; Samikannu, A.; Rautio, A.R.; Juhasz, K.L.; Konya, Z.; Wärnå, J.; Kordas, K.; Mikkola, J.P. Catalytic hydrogenation of d-xylose over Ru decorated carbon foam catalyst in a SpinChem® rotating bed reactor. Top. Catal. 2016, 59, 1165–1177. [Google Scholar] [CrossRef]
- Barig, S.; Alisch, R.; Nieland, S.; Wuttke, A.; Gräser, Y.; Huddar, M.; Schnitzlein, K.; Stahmann, K.P. Monoseptic growth of fungal lipase producers under minimized sterile conditions: Cultivation of Phialemonium curvatum in 350 L scale. Eng. Life Sci. 2011, 11, 387–394. [Google Scholar] [CrossRef]
- Noel, S.; Liberelle, B.; Robitaille, L.; de Crescenzo, G. Quantification of primary amine groups available for subsequent biofunctionalization of polymer surfaces. Bioconjug. Chem. 2011, 22, 1690–1699. [Google Scholar] [CrossRef] [PubMed]
- Bourbonnais, R.; Paice, M.G.; Reid, I.D.; Lanthier, P.; Yaguchi, M. Lignin oxidation by laccase isozymes from Trametes versicolor and role of the mediator 2,2′-azinobis(3-ethylbenzthiazoline-6-sulfonate) in kraft lignin depolymerization. Appl. Environ. Microbiol. 1995, 61, 1876–1880. [Google Scholar] [PubMed]
- Leirião, P.; Fonseca, L.; Taipa, M.; Cabral, J.; Mateus, M. Horseradish peroxidase immobilized through its carboxylic groups onto a polyacrylonitrile membrane. Appl. Biochem. Biotechnol. 2003, 110, 1–10. [Google Scholar] [CrossRef]
- Chen, Y.; Zhang, Y. Fluorescent quantification of amino groups on silica nanoparticle surfaces. Anal. Bioanal. Chem. 2011, 399, 2503–2509. [Google Scholar] [CrossRef] [PubMed]
- Ramanathan, T.; Fisher, F.T.; Ruoff, R.S.; Brinson, L.C. Amino-functionalized carbon nanotubes for binding to polymers and biological systems. Chem. Mater. 2005, 17, 1290–1295. [Google Scholar] [CrossRef]
- Norde, W.; Anusiem, A.C. Adsorption, desorption and re-adsorption of proteins on solid surfaces. Colloids Surf. 1992, 66, 73–80. [Google Scholar] [CrossRef]
- Durán, N.; Esposito, E. Potential applications of oxidative enzymes and phenoloxidase-like compounds in wastewater and soil treatment: A review. Appl. Catal. B Environ. 2000, 28, 83–99. [Google Scholar] [CrossRef]
- Becker, D.; Rodriguez-Mozaz, S.; Insa, S.; Schoevaart, R.; Barceló, D.; de Cazes, M.; Belleville, M.P.; Sanchez-Marcano, J.; Misovic, A.; Oehlmann, J.; et al. Removal of endocrine disrupting chemicals in wastewater by enzymatic treatment with fungal laccases. Org. Process Res. Dev. 2017, 21, 480–491. [Google Scholar] [CrossRef]
- Daughton, C.G.; Ternes, T.A. Pharmaceuticals and personal care products in the environment: Agents of subtle change? Environ. Health Perspect. 1999, 107, 907–938. [Google Scholar] [CrossRef] [PubMed]
- Basile, T.; Petrella, A.; Petrella, M.; Boghetich, G.; Petruzzelli, V.; Colasuonno, S.; Petruzzelli, D. Review of endocrine-disrupting-compound removal technologies in water and wastewater treatment plants: An EU perspective. Ind. Eng. Chem. Res. 2011, 50, 8389–8401. [Google Scholar] [CrossRef]
- Rodriguez, S.; Santos, A.; Romero, A. Effectiveness of AOP’s on abatement of emerging pollutants and their oxidation intermediates: Nicotine removal with Fenton’s Reagent. Desalination 2011, 280, 108–113. [Google Scholar] [CrossRef]
- Savchuk, N.; Krizova, P. Membrane and AOP processes—Their application and comparison in treatment of wastewater with high organics content. Desalin. Water Treat. 2015, 56, 3247–3251. [Google Scholar] [CrossRef]
- Pompe, T.; Salchert, K.; Alberti, K.; Zandstra, P.; Werner, C. Immobilization of growth factors on solid supports for the modulation of stem cell fate. Nat. Protoc. 2010, 5, 1042–1050. [Google Scholar] [CrossRef] [PubMed]
- Sano, S.; Kato, K.; Ikada, Y. Introduction of functional groups onto the surface of polyethylene for protein immobilization. Biomaterials 1993, 14, 817–822. [Google Scholar] [CrossRef]
- Hennig, A.; Borcherding, H.; Jaeger, C.; Hatami, S.; Würth, C.; Hoffmann, A.; Hoffmann, K.; Thiele, T.; Schedler, U.; Resch-Genger, U. Scope and limitations of surface functional group quantification methods: Exploratory study with poly(acrylic acid)-grafted micro- and nanoparticles. J. Am. Chem. Soc. 2012, 134, 8268–8276. [Google Scholar] [CrossRef] [PubMed]

| Pore diameter | 30–240 μm |
| Fillet width | 4–7 μm |
| BET surface | 5.91 m2·g−1 |
| Adsorptive | Covalent | ||||
|---|---|---|---|---|---|
| Quantity on BT (μg·mg−1) | Activity (U·mg−1enzyme) | Quantity on BT (μg·mg−1) | Activity (U·mg−1enzyme) | Activity of Free Enzyme (U·mg−1enzyme) | |
| Lipase | 5.8 ± 1.2 | 0.3 ± 0.1 | 4.3 ± 0.4 | 0.5 ± 0.1 | 2.2 ± 0.1 |
| Laccase | 5.8 ± 1.6 | 4.5 ± 0.8 | 4.1 ± 1.2 | 4.2 ± 0.7 | 18.5 ± 3.6 |
| Surface Coverage | Lipase | Laccase | |
|---|---|---|---|
| Theoretical (ng·cm−2) | min | 25 | 114 |
| max | 44 | 125 | |
| After adsorptive immobilization (ng·cm−2) | without SDS treatment | 98 ± 20 | 98 ± 37 |
| after SDS treatment | 17 ± 15 | 63 ± 19 | |
| After covalent immobilization (ng·cm−2) | without SDS treatment | 113 ± 14 | 126 ± 36 |
| after SDS treatment | 73 ± 7 | 69 ± 20 |
| Enzyme Concentration in Solution (mg·mL−1) | Immobilization Time (h) | Composition of Immobilization Solution | Wash Procedure | |
|---|---|---|---|---|
| Lipase | 3.0 | 0.5 (adsorptive) 2.0 (covalent) | 1% Propylene glycol, 1.6% PEG400, 0.01% CaCl2, pH = 6.1 | Five-times dipping into 30 mL ultrapure water, three wash cycles |
| Laccase | 0.5 | 4.0 | 0.2 M sodium citrate buffer, pH = 4.5 | Incubation in 5 mL 0.2 M sodium citrate buffer (pH = 4.5) on orbital shaker at 110 rpm for 15 min, three wash cycles |
| Precursor Ion | Product Ion | Frag-Mentor (V) | Collision Energy (V) | CAV (V) | |
|---|---|---|---|---|---|
| BPA-16d | 241 | 223 | 140 | 17 | 4 |
| 241 | 142 | 140 | 23 | 3 | |
| BPA | 227 | 212 | 140 | 17 | 4 |
| 227 | 195 | 140 | 42 | 5 | |
| 227 | 133 | 140 | 23 | 3 | |
| 227 | 117 | 140 | 57 | 4 |
| Parameter | Value |
|---|---|
| Temperature | 35 °C |
| Flow | 0.4 mL·min−1 |
| Injection volume | 15 μL |
| Source | 300 °C |
| Gas flow (N2) | 11 L·min−1 |
| Capillary | 4000 V |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Allertz, P.J.; Berger, S.; Sellenk, G.; Dittmer, C.; Dietze, M.; Stahmann, K.-P.; Salchert, K. Approaching Immobilization of Enzymes onto Open Porous Basotect®. Catalysts 2017, 7, 359. https://doi.org/10.3390/catal7120359
Allertz PJ, Berger S, Sellenk G, Dittmer C, Dietze M, Stahmann K-P, Salchert K. Approaching Immobilization of Enzymes onto Open Porous Basotect®. Catalysts. 2017; 7(12):359. https://doi.org/10.3390/catal7120359
Chicago/Turabian StyleAllertz, Peter J., Steffen Berger, Grit Sellenk, Christin Dittmer, Marco Dietze, Klaus-Peter Stahmann, and Katrin Salchert. 2017. "Approaching Immobilization of Enzymes onto Open Porous Basotect®" Catalysts 7, no. 12: 359. https://doi.org/10.3390/catal7120359
APA StyleAllertz, P. J., Berger, S., Sellenk, G., Dittmer, C., Dietze, M., Stahmann, K.-P., & Salchert, K. (2017). Approaching Immobilization of Enzymes onto Open Porous Basotect®. Catalysts, 7(12), 359. https://doi.org/10.3390/catal7120359





