Synergistic Enhancement in Catalytic Performance of Superparamagnetic Fe3O4@Bacilus subtilis as Recyclable Fenton-Like Catalyst
Abstract
1. Introduction
2. Results and Discussion
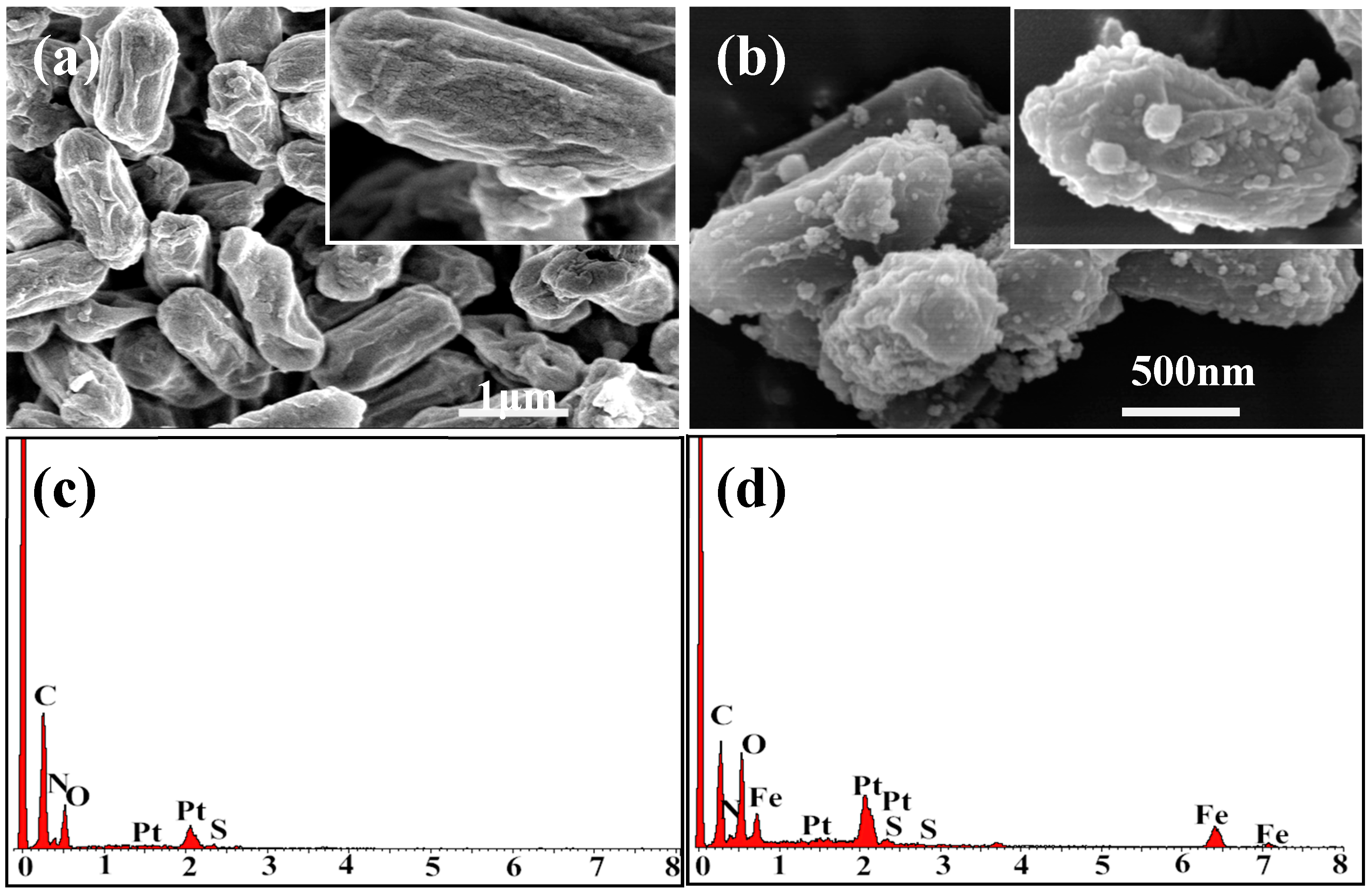
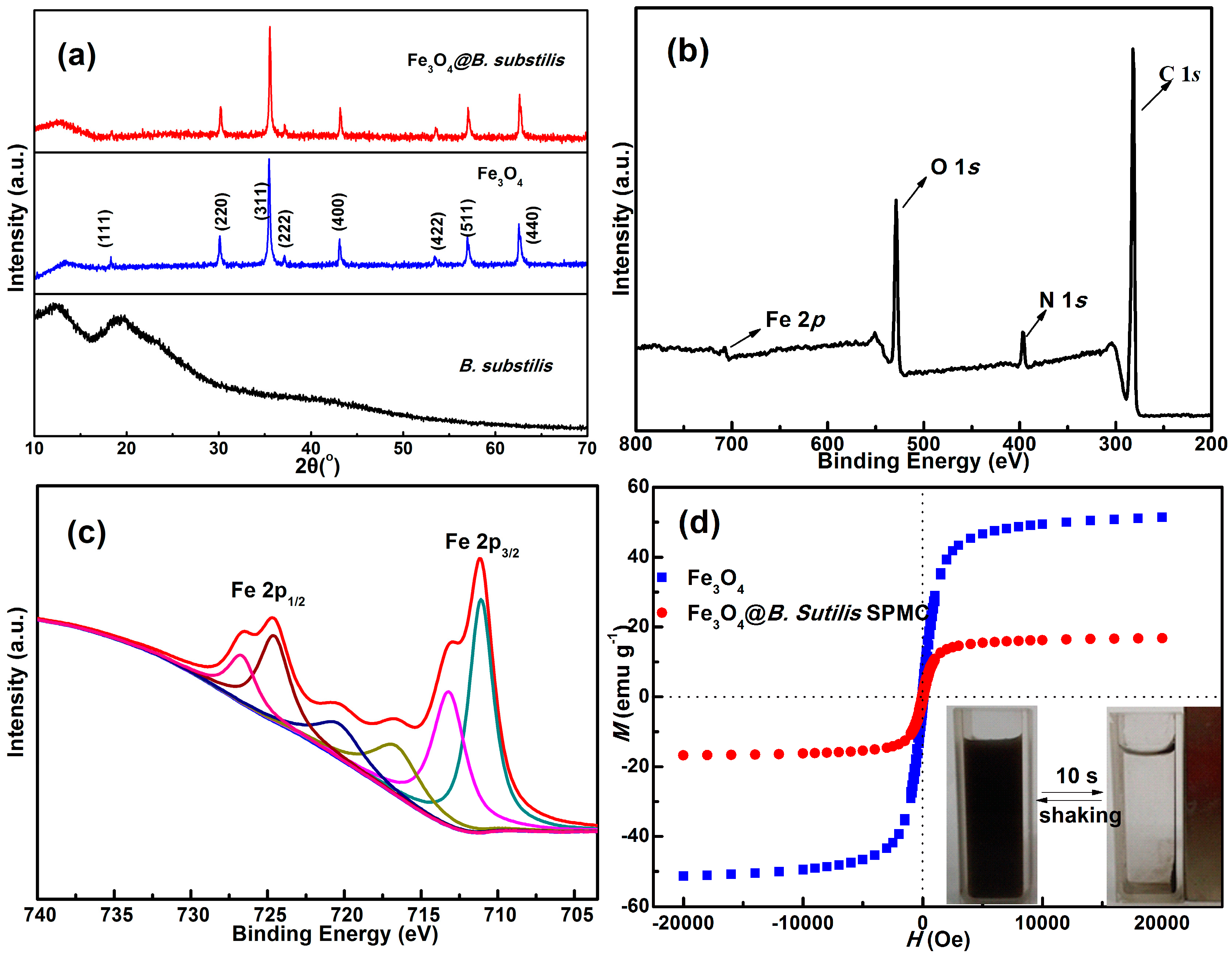
2.1. Characterization
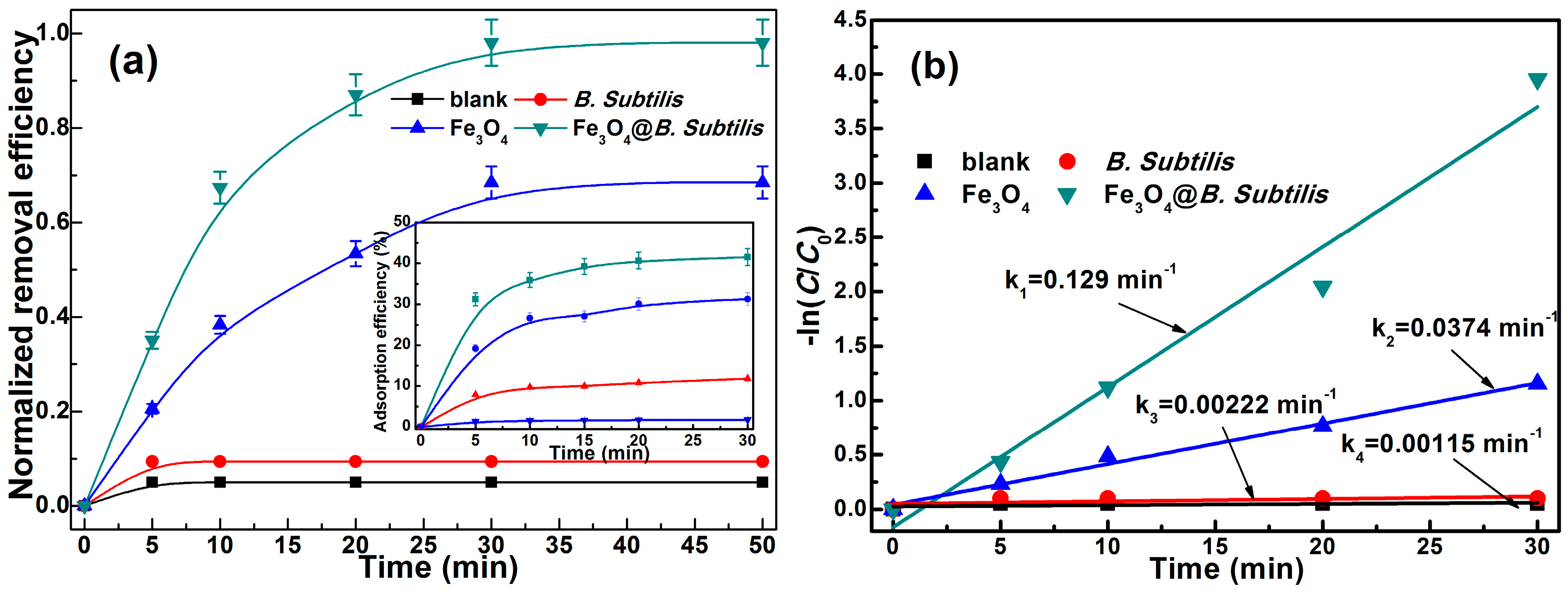
2.2. Fenton-Like Catalytic Degradation of Doxycycline
2.3. Effect of H2O2 Dosage on Degradation of DC
2.4. Iron Ion Leaching
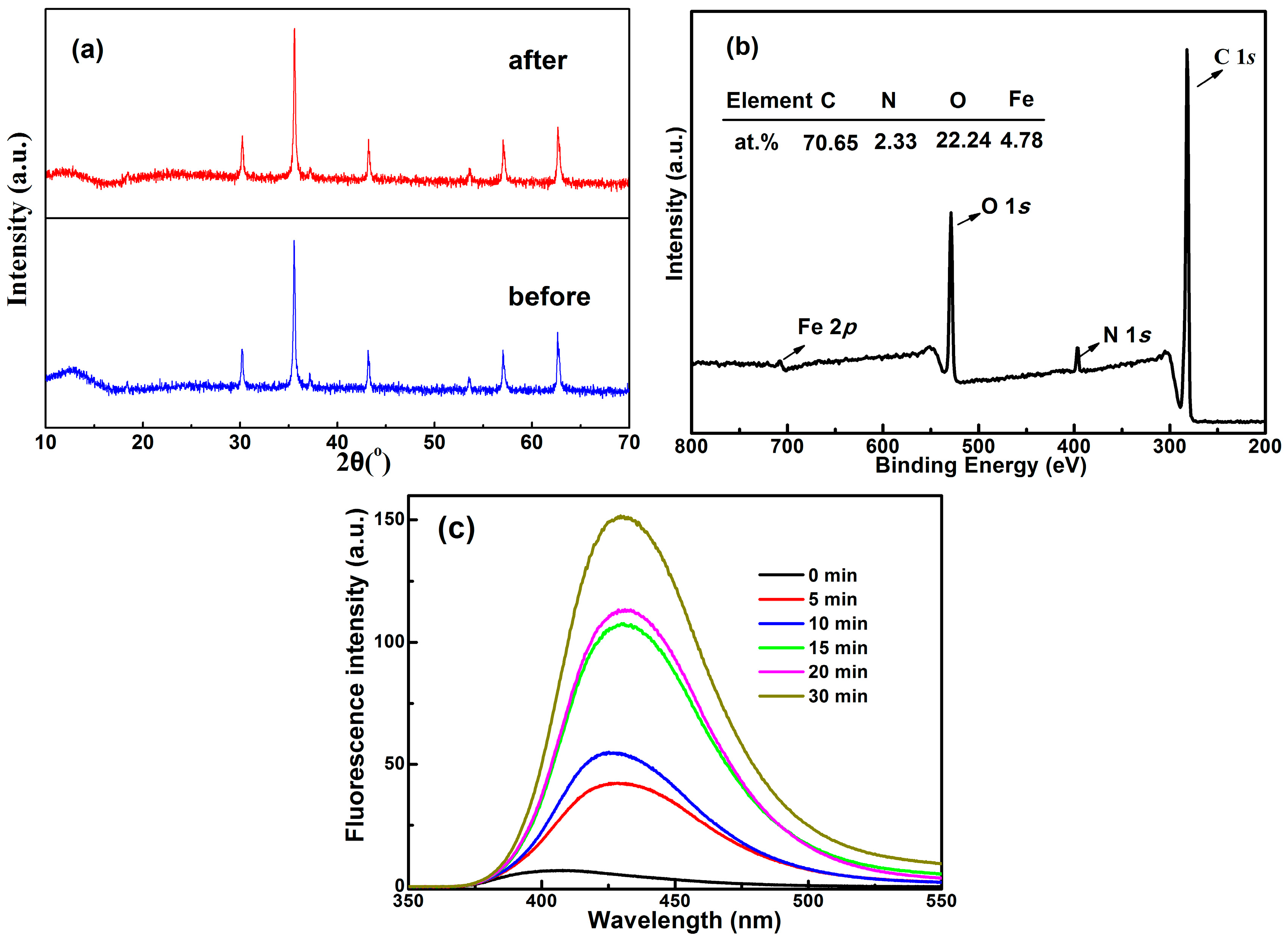
2.5. Stability and Reusability
2.6. Degradation Mechanism
3. Experimental
3.1. Materials
3.2. Synthesis of Fe3O4 MNPs
3.3. Preparation of Fe3O4@B. subtilis Superparamagnetic Composite (SPMC)
3.4. Characterizations
3.5. Catalytic Tests
3.6. Detection of Radical Species
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- He, C.; Wu, S.; Zhao, N.; Shi, C.; Liu, E.; Li, J. Carbon-encapsulated Fe3O4 nanoparticles as a high-rate lithium ion battery anode material. ACS Nano 2013, 7, 4459–4469. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Chen, W.; Huang, J.; Zhou, Y.; Zhu, Y.; Li, C. Rapid degradation of methylene blue in a novel heterogeneous Fe3O4@rGO@TiO2-catalyzed photo-Fenton system. Sci. Rep. 2015, 5, 10632. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Pauletti, G.M.; Wang, J.; Zhang, J.; Ewing, R.C.; Wang, Y.; Shi, D. Dual surface-functionalized janus nanocomposites of polystyrene/Fe3O4@SiO2 for simultaneous tumor cell targeting and stimulus-induced drug release. Adv. Mater. 2013, 25, 3485–3489. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Wang, J. Magnetic nanoscaled Fe3O4/CeO2 composite as an efficient Fenton-like heterogeneous catalyst for degradation of 4-chlorophenol. Environ. Sci. Technol. 2012, 46, 10145–10153. [Google Scholar] [CrossRef] [PubMed]
- Wei, Y.; Yin, G.; Ma, C.; Huang, Z.; Chen, X.; Liao, X.; Yao, Y.; Yin, H. Synthesis and cellular compatibility of biomineralized Fe3O4 nanoparticles in tumor cells targeting peptides. Colloids Surf. B Biointerfaces 2013, 107, 180–188. [Google Scholar] [CrossRef] [PubMed]
- Zubir, N.A.; Yacou, C.; Motuzas, J.; Zhang, X.; da Costa, J.C.D. Structural and functional investigation of graphene oxide–Fe3O4 nanocomposites for the heterogeneous Fenton-like reaction. Sci. Rep. 2014, 4, 4594. [Google Scholar] [CrossRef] [PubMed]
- Kuznetsova, E.; Savinov, E.; Vostrikova, L.; Parmon, V. Heterogeneous catalysis in the Fenton-type system FeZSM-5/H2O2. Appl. Catal. B 2004, 51, 165–170. [Google Scholar] [CrossRef]
- Aravindhan, R.; Fathima, N.N.; Rao, J.R.; Nair, B.U. Wet oxidation of acid brown dye by hydrogen peroxide using heterogeneous catalyst Mn-salen-Y zeolite: A potential catalyst. J. Hazard. Mater. 2006, 138, 152–159. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Zhang, X.; Ma, Y.; Huang, Y.; Wang, Y.; Chen, Y. Superparamagnetic graphene oxide–Fe3O4 nanoparticles hybrid for controlled targeted drug carriers. J. Mater. Chem. 2009, 19, 2710–2714. [Google Scholar] [CrossRef]
- Yu, L.; Yang, X.; Ye, Y.; Wang, D. Efficient removal of atrazine in water with a Fe3O4/MWCNTs nanocomposite as a heterogeneous Fenton-like catalyst. RSC Adv. 2015, 5, 46059–46066. [Google Scholar] [CrossRef]
- Xu, H.-Y.; Shi, T.-N.; Zhao, H.; Jin, L.-G.; Wang, F.-C.; Wang, C.-Y.; Qi, S.-Y. Heterogeneous Fenton-like discoloration of methyl orange using Fe3O4/MWCNTs as catalyst: Process optimization by response surface methodology. Front. Mater. Sci. 2016, 10, 45–55. [Google Scholar] [CrossRef]
- Li, X.; Huang, X.; Liu, D.; Wang, X.; Song, S.; Zhou, L.; Zhang, H. Synthesis of 3D hierarchical Fe3O4/graphene composites with high lithium storage capacity and for controlled drug delivery. J. Phys. Chem. C 2011, 115, 21567–21573. [Google Scholar] [CrossRef]
- Yan, C.; Feng, D.; Jiang, Y.; An, X.; Ye, L.; Guan, W.; Bai, B. Bio-template Route for the Facile Fabrication of TiO2@ Bacillus subtilis Composite Particles and Their Application for the Degradation of Rhodamine B. Catal. Lett. 2015, 145, 1301–1306. [Google Scholar] [CrossRef]
- Filip, Z.; Herrmann, S.; Kubat, J. FT-IR spectroscopic characteristics of differently cultivated Bacillus subtilis. Microbiol. Res. 2004, 159, 257–262. [Google Scholar] [CrossRef] [PubMed]
- Zheng, P.; Bai, B.; Guan, W.; Wang, H.; Suo, Y. Degradation of tetracycline hydrochloride by heterogeneous Fenton-like reaction using Fe@ Bacillus subtilis. RSC Adv. 2016, 6, 4101–4107. [Google Scholar] [CrossRef]
- Ayla, A.; Çavuş, A.; Bulut, Y.; Baysal, Z.; Aytekin, Ç. Removal of methylene blue from aqueous solutions onto Bacillus subtilis: Determination of kinetic and equilibrium parameters. Desalination Water Treat. 2013, 51, 7596–7603. [Google Scholar] [CrossRef]
- Liu, J.; Che, R.; Chen, H.; Zhang, F.; Xia, F.; Wu, Q.; Wang, M. Microwave absorption enhancement of multifunctional composite microspheres with spinel Fe3O4 cores and anatase TiO2 shells. Small 2012, 8, 1214–1221. [Google Scholar] [CrossRef] [PubMed]
- Kaboudin, B.; Mostafalu, R.; Yokomatsu, T. Fe3O4 nanoparticle-supported Cu (II)-β-cyclodextrin complex as a magnetically recoverable and reusable catalyst for the synthesis of symmetrical biaryls and 1, 2, 3-triazoles from aryl boronic acids. Green Chem. 2013, 15, 2266–2274. [Google Scholar] [CrossRef]
- Mahdavi, M.; Namvar, F.; Ahmad, M.B.; Mohamad, R. Green biosynthesis and characterization of magnetic iron oxide (Fe3O4) nanoparticles using seaweed (Sargassum muticum) aqueous extract. Molecules 2013, 18, 5954–5964. [Google Scholar] [CrossRef] [PubMed]
- Hassan, M.S.; Amna, T.; Yang, O.-B.; Kim, H.-C.; Khil, M.-S. TiO2 nanofibers doped with rare earth elements and their photocatalytic activity. Ceram. Int. 2012, 38, 5925–5930. [Google Scholar] [CrossRef]
- Deng, Y.; Qi, D.; Deng, C.; Zhang, X.; Zhao, D. Superparamagnetic high-magnetization microspheres with an Fe3O4@ SiO2 core and perpendicularly aligned mesoporous SiO2 shell for removal of microcystins. J. Am. Chem. Soc. 2008, 130, 28–29. [Google Scholar] [CrossRef] [PubMed]
- Tian, S.; Zhang, J.; Chen, J.; Kong, L.; Lu, J.; Ding, F.; Xiong, Y. Fe2(MoO4)3 as an effective photo-Fenton-like catalyst for the degradation of anionic and cationic dyes in a wide pH range. Ind. Eng. Chem. Res. 2013, 52, 13333–13341. [Google Scholar] [CrossRef]
- Bigda, R.J. Consider Fentons chemistry for wastewater treatment. Chem. Eng. Prog. 1995, 91, 62–66. [Google Scholar]
- Coelho, J.V.; Guedes, M.S.; Prado, R.G.; Tronto, J.; Ardisson, J.D.; Pereira, M.C.; Oliveira, L.C. Effect of iron precursor on the Fenton-like activity of Fe2O3/mesoporous silica catalysts prepared under mild conditions. Appl. Catal. B 2014, 144, 792–799. [Google Scholar] [CrossRef]
- Li, X.; Liu, J.; Rykov, A.I.; Han, H.; Jin, C.; Liu, X.; Wang, J. Excellent photo-Fenton catalysts of Fe–Co Prussian blue analogues and their reaction mechanism study. Appl. Catal. B 2015, 179, 196–205. [Google Scholar] [CrossRef]
- Cleveland, V.; Bingham, J.-P.; Kan, E. Heterogeneous Fenton degradation of bisphenol A by carbon nanotube-supported Fe3O4. Sep. Purif. Technol. 2014, 133, 388–395. [Google Scholar] [CrossRef]
- Luo, W.; Zhu, L.; Wang, N.; Tang, H.; Cao, M.; She, Y. Efficient removal of organic pollutants with magnetic nanoscaled BiFeO3 as a reusable heterogeneous Fenton-like catalyst. Environ. Sci. Technol. 2010, 44, 1786–1791. [Google Scholar] [CrossRef] [PubMed]
- Hu, X.; Liu, B.; Deng, Y.; Chen, H.; Luo, S.; Sun, C.; Yang, P.; Yang, S. Adsorption and heterogeneous Fenton degradation of 17α-methyltestosterone on nano Fe3O4/MWCNTs in aqueous solution. Appl. Catal. B 2011, 107, 274–283. [Google Scholar] [CrossRef]
- Kwan, W.P.; Voelker, B.M. Rates of hydroxyl radical generation and organic compound oxidation in mineral-catalyzed Fenton-like systems. Environ. Sci. Technol. 2003, 37, 1150–1158. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.-J.; Xu, X.-M.; Xu, J.; Han, Y.-F. Iron oxychloride (FeOCl): An efficient Fenton-like catalyst for producing hydroxyl radicals in degradation of organic contaminants. J. Am. Chem. Soc. 2013, 135, 16058–16061. [Google Scholar] [CrossRef] [PubMed]
- Song, R.; Bai, B.; Puma, G.L.; Wang, H.; Suo, Y. Biosorption of azo dyes by raspberry-like Fe3O4@ yeast magnetic microspheres and their efficient regeneration using heterogeneous Fenton-like catalytic processes over an up-flow packed reactor. React. Kinet. Mech. Cat. 2015, 115, 547–562. [Google Scholar] [CrossRef]


© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zheng, P.; Pan, Z.; Zhang, J. Synergistic Enhancement in Catalytic Performance of Superparamagnetic Fe3O4@Bacilus subtilis as Recyclable Fenton-Like Catalyst. Catalysts 2017, 7, 349. https://doi.org/10.3390/catal7110349
Zheng P, Pan Z, Zhang J. Synergistic Enhancement in Catalytic Performance of Superparamagnetic Fe3O4@Bacilus subtilis as Recyclable Fenton-Like Catalyst. Catalysts. 2017; 7(11):349. https://doi.org/10.3390/catal7110349
Chicago/Turabian StyleZheng, Pei, Zhe Pan, and Jun Zhang. 2017. "Synergistic Enhancement in Catalytic Performance of Superparamagnetic Fe3O4@Bacilus subtilis as Recyclable Fenton-Like Catalyst" Catalysts 7, no. 11: 349. https://doi.org/10.3390/catal7110349
APA StyleZheng, P., Pan, Z., & Zhang, J. (2017). Synergistic Enhancement in Catalytic Performance of Superparamagnetic Fe3O4@Bacilus subtilis as Recyclable Fenton-Like Catalyst. Catalysts, 7(11), 349. https://doi.org/10.3390/catal7110349



