Recent Advances in Graphene Based TiO2 Nanocomposites (GTiO2Ns) for Photocatalytic Degradation of Synthetic Dyes
Abstract
1. Introduction
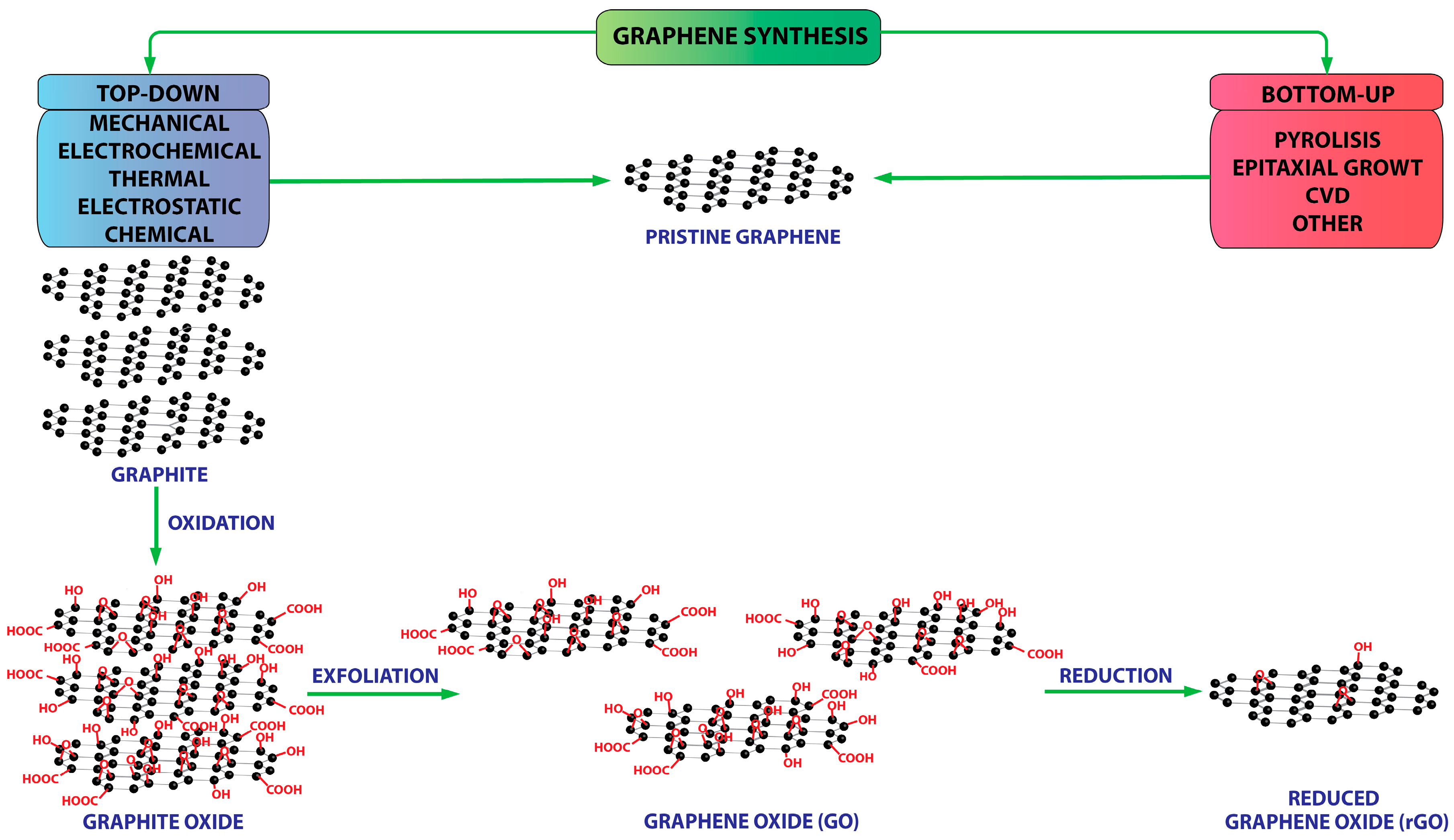
2. Graphene Materials: Concepts and Properties
3. The Photocatalytic Process: Fundamentals of Graphene-TiO2 Photocatalysts
- (1)
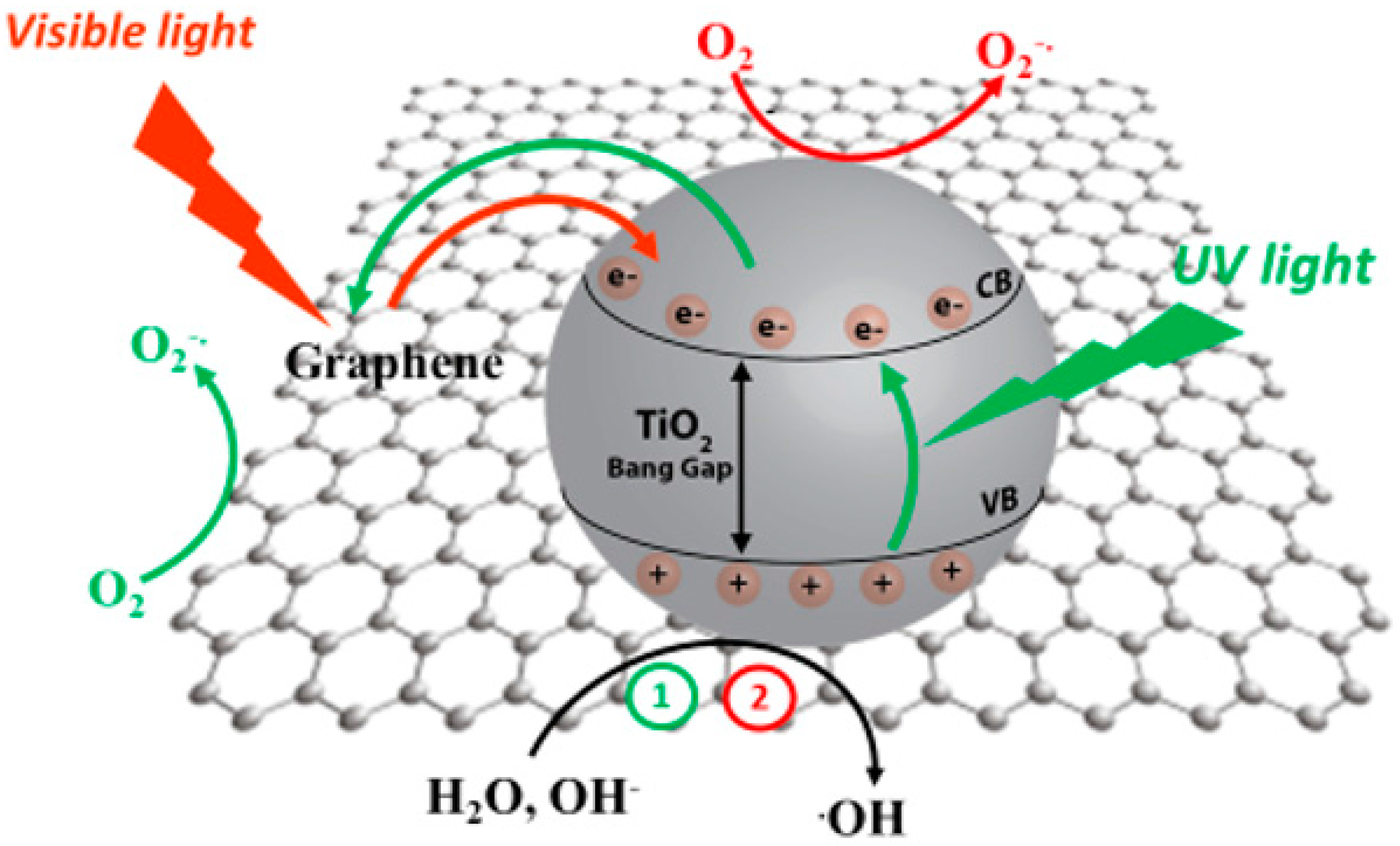
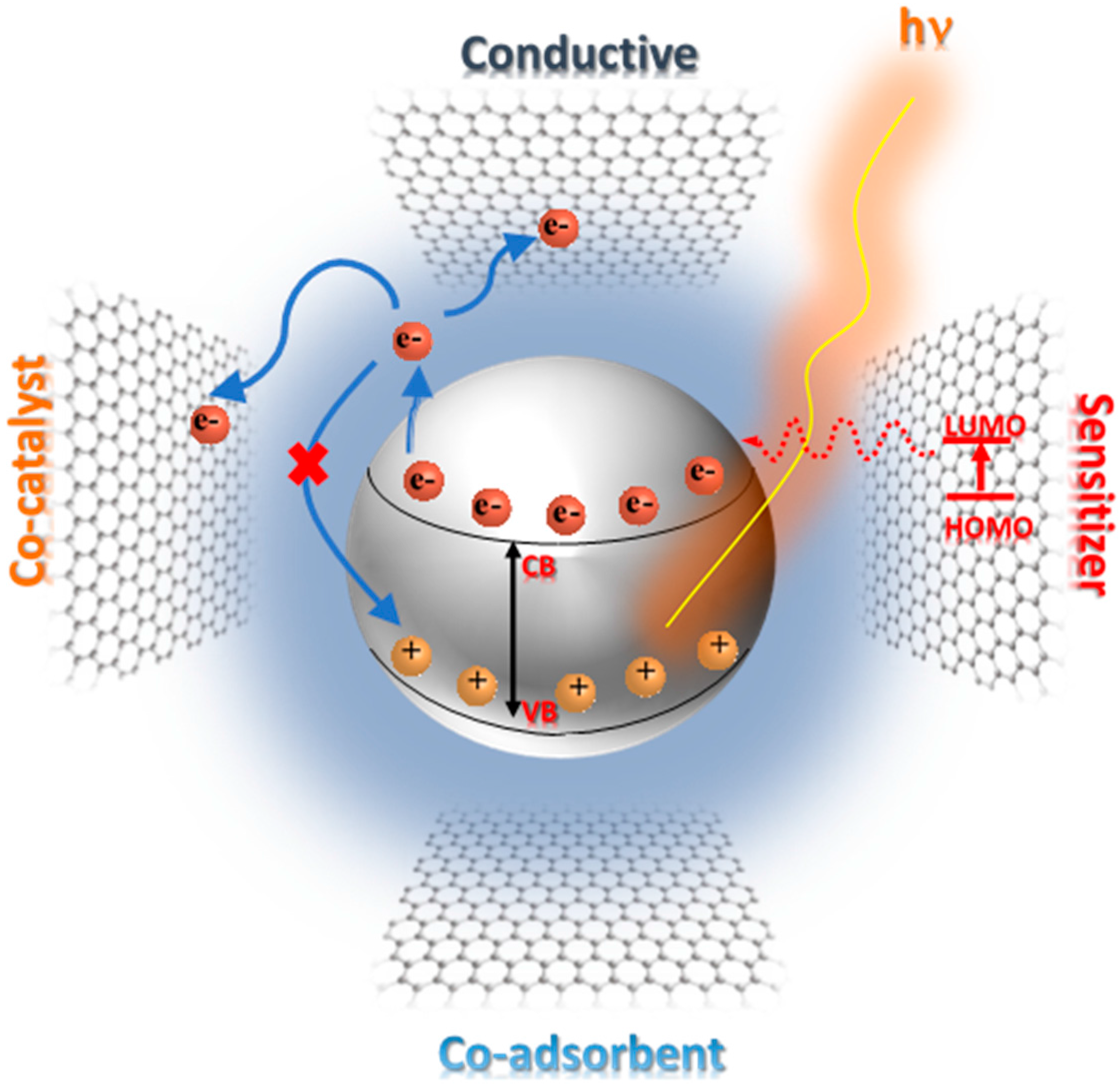
- UV light excitation of GTiO2Ns photogenerates the electron–hole pairs and the electrons are then injected into graphene due to its more positive Fermi level [144]. This process is favored by the position of the work function of graphene that is −4.42 eV, with respect the conduction band of TiO2 that is located at −4.20 eV [145]; from this consideration, the electron in the CB of TiO2 is injected to G. Graphene scavengesphotogenerated electrons by dissolved oxygen; facilitates the hole-electron separation, reducing the recombination of e-(CB) and the holes (VB); and, due to its high carrier mobility, accelerates the electron transport, thus enhancing the photocatalytic performance [142,146].
- (2)
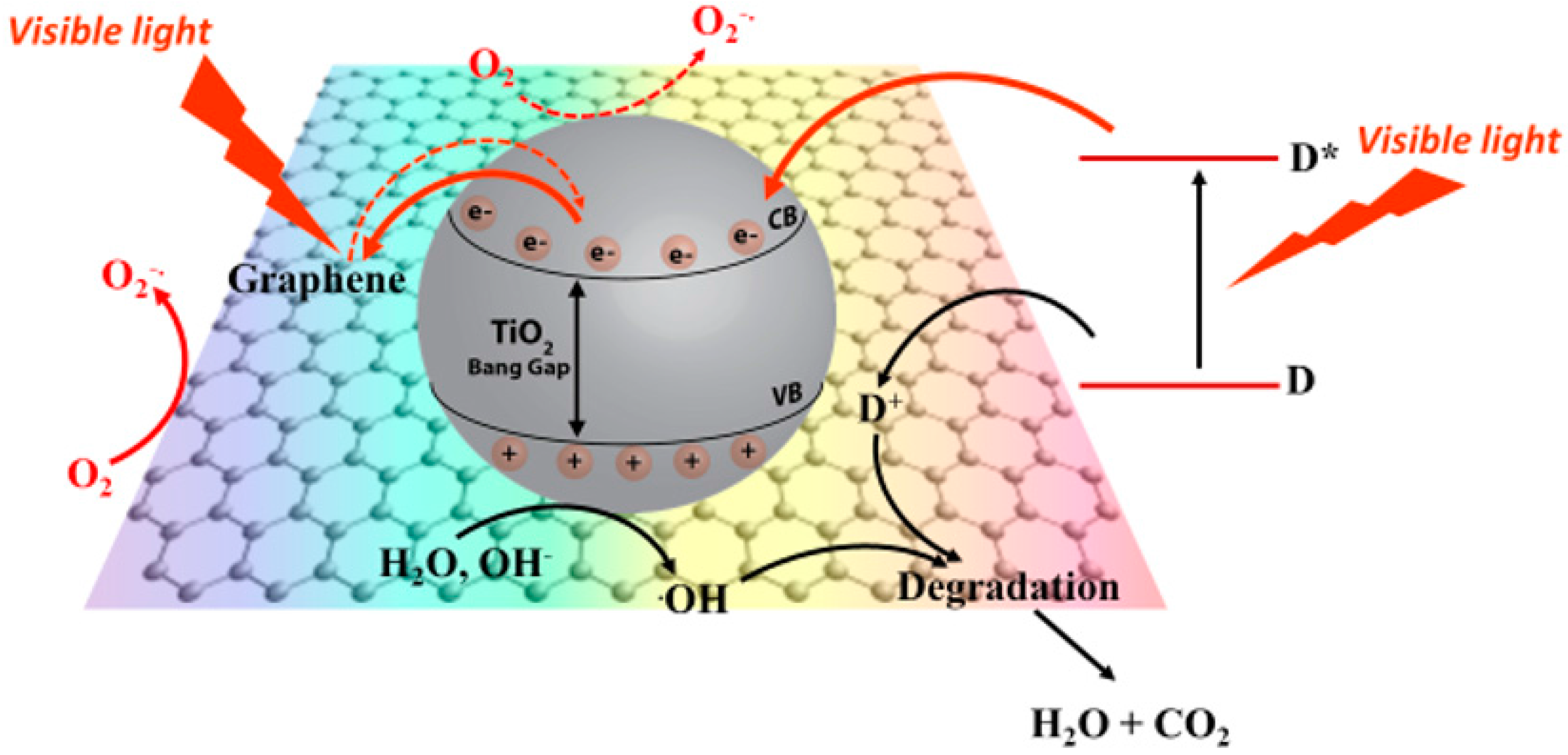
- When the operational mechanism takes place via visible light, the electron transfer of thephotogenerated electron is promoted from the G photoexcited state and then delocalized to the TiO2 surface. M.T Silva et al. indicated, by rGO photoluminescence study, that the photogenerated electrons under Vis or NIR laser can be transferred to the surface of TiO2 with a consequent quenching of photoluminescence; also in this case, charge recombination is inhibited with a consequent increasing of photocatalytic activity under visible light [147]. It is important to know that the presence of G in GTiO2Ns photocatalyst produces a red shift in the absorption, reducing its band gap and thus extending the photoresponse to a longer wavelength [148]. The explanation of visible light activation in the GTiO2Ns composites is not clear, but it is possible to attribute this phenomena to the sensitization of TiO2 due to the presence of graphene [147,149,150]. In this case, in the visible light excitation of GTiO2Ns, graphene absorbs the light, and the photoexcited electrons in high energy G states are delocalized into the CB of the TiO2 surface with the dissipation of excess energy due to electron vibrational interaction [151]; successively electrons react with oxygen, resulting in the formation of superoxide radicals. In Figure 3, the activation mechanisms of GTiO2Ns under UV and visible light are reported.
4. Preparation Methods of GTiO2Ns
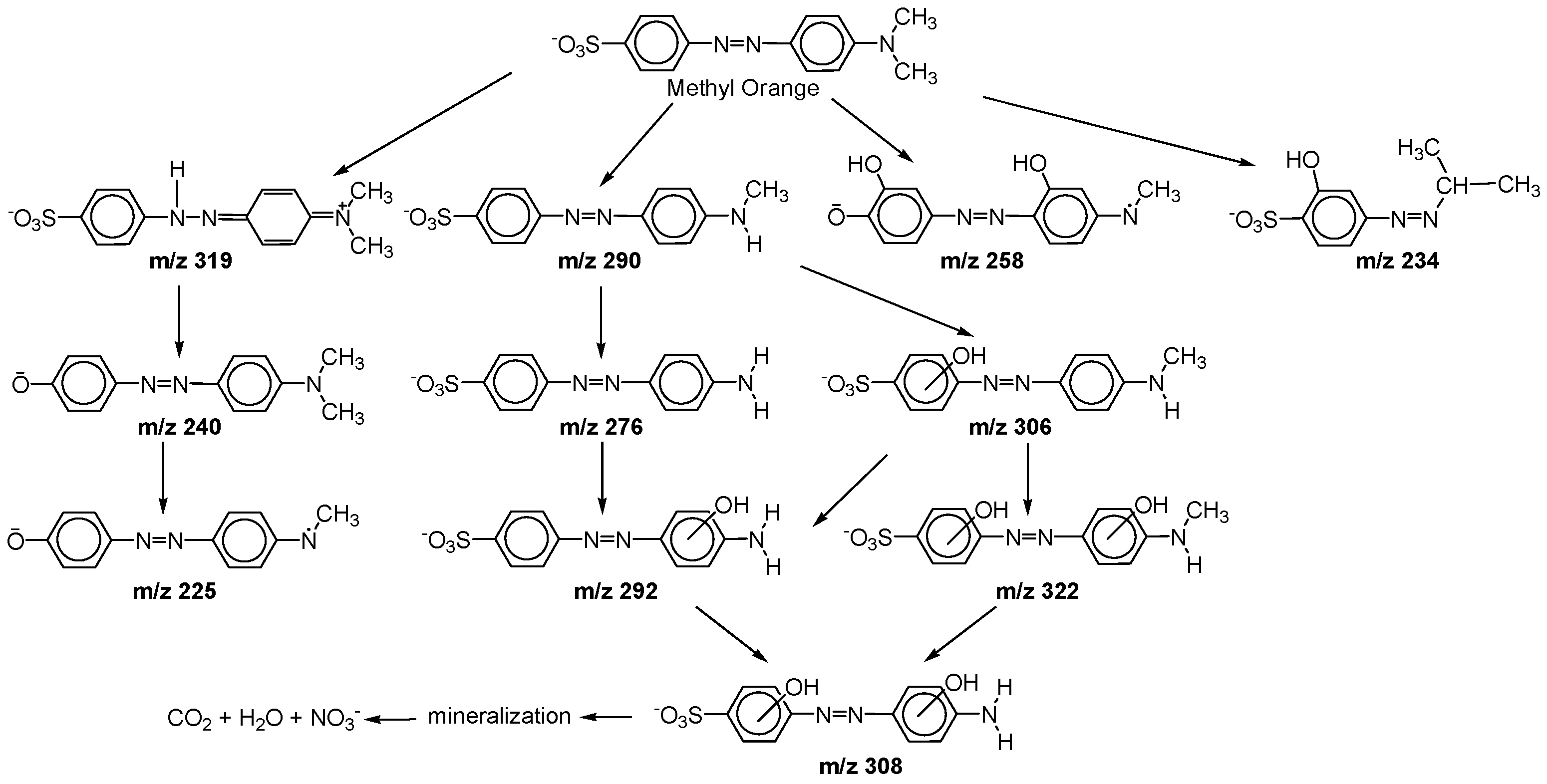
5. Photocatalytic Degradation of Dyes with GTiO2Ns
6. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Peirce, J.J.; Weiner, R.; Vesilind, P.A. Environmental Pollution and Control, 4th ed.; Elsevier: Oxford, UK, 1998; ISBN 978-0-7506-9899-3. [Google Scholar]
- Andreozzi, R.; Caprio, V.; Insola, A.; Marotta, R. Advanced oxidation processes (AOP) for water purification and recovery. Catal. Today 1999, 53, 51–59. [Google Scholar] [CrossRef]
- Chaukura, N.; Edna, C.; Murimba, E.C.; Gwenzi, W. Sorptive removal of methylene blue from simulated wastewater using biochars derived from pulp and paper sludge. Environ. Technol. Innov. 2017, 8, 132–140. [Google Scholar] [CrossRef]
- Solís, M.; Solís, A.; Inés Pérez, H.; Manjarrez, N.; Flores, M. Microbial decolouration of azo dyes: A review. Process. Biochem. 2012, 47, 1723–1748. [Google Scholar] [CrossRef]
- Zhang, Z.; Yang, R.Y.; Gao, Y.S.; Zhao, Y.F.; Wang, J.Y.; Huang, L.; Guo, J.; Zhou, T.T.; Lu, P.; Guo, Z.H.; et al. Novel Na2Mo4O13/α-MoO3 hybrid material as highly efficient CWAO catalyst for dye degradation at ambient conditions. Sci. Rep. 2014, 4, 6797–6809. [Google Scholar] [CrossRef] [PubMed]
- Santhosh, C.; Velmurugan, V.; Jacob, G.; Jeong, S.K.; Grace, A.N.; Bhatnagar, A. Role of nanomaterials in water treatment applications: A review. Chem. Eng. J. 2016, 306, 1116–1137. [Google Scholar] [CrossRef]
- Padikkaparambil, S.; Narayanan, B.; Yaakob, Z.; Viswanathan, S.; Tasirin, S.M. Au/TiO2 reusable photocatalysts for dye degradation. Int. J. Photoenergy 2013, 2013. [Google Scholar] [CrossRef]
- Cho, K.M.; Kim, K.H.; Choi, H.O.; Jung, H.T. A highly photoactive, visible-light-driven graphene/2D mesoporous TiO2 photocatalyst. Green Chem. 2015, 17, 3972–3978. [Google Scholar] [CrossRef]
- Ibhadon, A.O.; Fitzpatrick, P. Heterogeneous photocatalysis: Recent advances and applications. Catalysts 2013, 3, 189–218. [Google Scholar] [CrossRef]
- Wei, W.; Liu, D.; Wei, Z.; Zhu, Y. Short-range π-π stacking assembly on P25 TiO2 nanoparticle for enhanced visible-light photocatalysis. ACS Catal. 2017, 7, 652–663. [Google Scholar] [CrossRef]
- Jiang, W.J.; Liu, Y.F.; Wang, J.; Zhang, M.; Luo, W.J.; Zhu, Y.F. Separation-free polyaniline/TiO2 3D hydrogel with high photocatalytic activity. Adv. Mater. Interfaces 2016, 3, 9. [Google Scholar]
- Tatsuma, T.; Saitoh, S.; Ohko, Y.; Fujishima, A. TiO2-WO3 photoelectrochemical anticorrosion system with an energy storage ability. Chem. Mater. 2001, 13, 2838–2842. [Google Scholar] [CrossRef]
- Sajan, C.P.; Wageh, S.; Al-Ghamdi, A.A.; Yu, J.G.; Cao, S.W. TiO2 nanosheets with exposed {001} facets for photocatalytic applications. Nano Res. 2016, 9, 3–27. [Google Scholar] [CrossRef]
- Chen, X.; Mao, S.S. Titanium dioxide nanomaterials: Synthesis, properties, modifications, and applications. Chem. Rev. 2007, 107, 2891–2959. [Google Scholar] [CrossRef] [PubMed]
- Tong, H.; Ouyang, S.X.; Bi, Y.P.; Umezawa, N.; Oshikiri, M.; Ye, J.H. Nano-photocatalytic materials: Possibilities and challenges. Adv. Mater. 2012, 24, 229–251. [Google Scholar] [CrossRef] [PubMed]
- Anpo, M.; Tackeuchi, M. The design and development of highly reactive titanium oxide photocatalysts operating under visible light irradiation. J. Catal. 2003, 216, 505–516. [Google Scholar] [CrossRef]
- Al-Harbi, L.M.; El-Mossalamy, E.H.; Arafa, H.M.; Al-Owais, A.; Shah, M.A. TiO2 Nanoparticles with Tetra-pad Shape Prepared by an Economical and Safe Route at very Low Temperature. Mod. Appl. Sci. 2011, 5, 130–135. [Google Scholar] [CrossRef]
- Giovannetti, R.; D’Amato, C.A.; Zannotti, M.; Rommozzi, E.; Gunnella, R.; Minicucci, M.; Di Cicco, A. Visible light photoactivity of polypropylene coated nano-TiO2 for dyes degradation in water. Sci. Rep. 2015, 2. [Google Scholar] [CrossRef] [PubMed]
- Augugliaro, V.; Bellardita, M.; Loddo, V.; Palmisano, G.; Palmisano, L.; Yurdakal, S. Overview on oxidation mechanisms of organic compounds by TiO2 in heterogeneous photocatalysis. J. Photochem. Photobiol. C Photochem. Rev. 2012, 13, 224–245. [Google Scholar] [CrossRef]
- Sud, D.; Kaur, P. Heterogeneous photocatalytic degradation of selected organophosphate pesticides: A review. Crit. Rev. Environ. Sci. Technol. 2012, 42, 2365–2407. [Google Scholar] [CrossRef]
- Wang, J.L.; Xu, L.J. Advanced oxidation processes for wastewater treatment: Formation of hydroxyl radical and application. Crit. Rev. Environ. Sci. Technol. 2012, 42, 251–325. [Google Scholar] [CrossRef]
- Hashimoto, K.; Irie, H.; Fujishima, A. TiO2 photocatalysis: A historical overview and future prospects. Jpn. J. Appl. Phys. 2005, 12, 8269–8285. [Google Scholar] [CrossRef]
- Sheikh, M.U.D.; Naikoo, G.A.; Thomas, M.; Bano, M.; Khan, F. Solar-assisted photocatalytic reduction of methyl orange azo dye over porous TiO2 nanostructures. New J. Chem. 2016, 40, 5483–5494. [Google Scholar] [CrossRef]
- Zhang, N.; Zhang, Y.; Xu, Y.-J. Recent progress on graphene-based photocatalysts: Current status and future perspectives. Nanoscale 2012, 4, 5792–5813. [Google Scholar] [CrossRef] [PubMed]
- Malato, S.; Fernández-Ibáñez, P.; Maldonado, M.I.; Blanco, J.; Gernjak, W. Decontamination and disinfection of water by solar photocatalysis: Recent overview and trends. Catal. Today 2009, 147, 1–59. [Google Scholar] [CrossRef]
- Wang, W.K.; Chen, J.J.; Gao, M.; Huang, Y.X.; Zhang, X.; Yu, H.Q. Photocatalytic degradation of atrazine by boron-doped TiO2 with a tunable rutile/anatase ratio. Appl. Catal. B Environ. 2016, 195, 69–76. [Google Scholar] [CrossRef]
- Hao, R.R.; Wang, G.H.; Tang, H.; Sun, L.L.; Xu, C.; Han, D.Y. Template-free preparation of macro/mesoporous g-C3N4/TiO2 heterojunction photocatalysts with enhanced visible light photocatalytic activity. Appl. Catal. B Environ. 2016, 187, 47–58. [Google Scholar] [CrossRef]
- Gua, Y.; Xinga, M.; Zhang, J. Synthesis and photocatalytic activity of graphene based doped TiO2 nanocomposites. Appl. Surf. Sci. 2014, 319, 8–15. [Google Scholar] [CrossRef]
- Zaleska, A. Doped-TiO2: A review. Recent Pat. Eng. 2008, 2, 157–164. [Google Scholar] [CrossRef]
- Yang, H.G.; Sun, C.H.; Qiao, S.Z.; Zou, J.; Liu, G.; Smith, S.C.; Cheng, H.M.; Lu, G.Q. Anatase TiO2 single crystals with a large percentage of reactive facets. Nature 2008, 453, 638–641. [Google Scholar] [CrossRef] [PubMed]
- Adán, C.; Bahamonde, A.; Fernández-García, M.; Martínez-Arias, A. Structure and activity of nanosized iron-doped anatase TiO2 catalysts for phenol photocatalytic degradation. Appl. Catal. B Environ. 2007, 72, 11–17. [Google Scholar] [CrossRef]
- Cai, J.; Wu, X.; Li, S.; Zheng, F. Controllable location of Au nanoparticles as cocatalyst onto TiO2@CeO2 nanocomposite hollow spheres for enhancing photocatalytic activity. Appl. Catal. B Environ. 2017, 201, 12–21. [Google Scholar] [CrossRef]
- Cheng, L.; Qiu, S.; Chen, J.; Shao, J.; Cao, S. A practical pathway for the preparation of Fe2O3 decorated TiO2 photocatalyst with enhanced visible-light photoactivity. Mater. Chem. Phys. 2017, 190, 53–61. [Google Scholar] [CrossRef]
- Xie, Y.; Meng, Y.; Wu, M. Visible-light-driven self-cleaning SERS substrate of silver nanoparticles and graphene oxide decorated nitrogen-doped Titania nanotube array. Surf. Interface Anal. 2016, 48, 334–340. [Google Scholar] [CrossRef]
- Xing, Z.; Zong, X.; Zhu, Y.; Chen, Z.; Bai, Y.; Wang, L. A nanohybrid of CdTe@CdS nanocrystals and Titania nanosheets with p-n nanojunctions for improved visible light-driven hydrogen production. Catal. Today 2016, 264, 229–235. [Google Scholar] [CrossRef]
- Hamzezadeh-Nakhjavani, S.; Tavakoli, O.; Akhlaghi, S.P.; Salehi, Z.; Esmailnejad-Ahranjani, P.; Arpanaei, A. Efficient photocatalytic degradation of organic pollutants by magnetically recoverable nitrogen-doped TiO2 nanocomposite photocatalysts under visible light irradiation. Environ. Sci. Pollut. Res. 2015, 22, 18859–18873. [Google Scholar] [CrossRef] [PubMed]
- Qi, K.; Selvaraj, R.; Al Fahdi, T.; Al-Kindy, S.; Kim, Y.; Wang, G.-C.; Tai, C.-W.; Sillanpää, M. Enhanced photocatalytic activity of anatase-TiO2 nanoparticles by fullerene modification: A theoretical and experimental study. Appl. Surf. Sci. 2016, 387, 750–758. [Google Scholar] [CrossRef]
- Qin, X.; He, F.; Chen, L.; Meng, Y.; Liu, J.; Zhao, N.; Huang, Y. Oxygen-vacancy modified TiO2 nanoparticles as enhanced visible-light driven photocatalysts by wrapping and chemically bonding with graphite-like carbon. RSC Adv. 2016, 6, 10887–10894. [Google Scholar] [CrossRef]
- Rahbar, M.; Behpour, M. Multi-walled carbon nanotubes/TiO2 thin layer for photocatalytic degradation of organic pollutant under visible light irradiation. J. Mater. Sci. Mater. Electron. 2016, 27, 8348–8355. [Google Scholar] [CrossRef]
- Lin, L.; Wang, H.; Xu, P. Immobilized TiO2-reduced graphene oxide nanocomposites on optical fibers as high performance photocatalysts for degradation of pharmaceuticals. Chem. Eng. J. 2017, 310, 389–398. [Google Scholar] [CrossRef]
- Stoller, M.D.; Park, S.; Zhu, Y.; An, J.; Ruoff, R.S. Graphene-based ultracapacitors. Nano Lett. 2008, 8, 3498–3502. [Google Scholar] [CrossRef] [PubMed]
- Novoselov, K.S.; Geim, A.K.; Morozov, S.V.; Jiang, D.; Zhang, Y.; Dubonos, S.V.; Grigorieva, I.V.; Firsov, A.A. Electric field effect in atomically thin carbon films. Science 2004, 306, 666–669. [Google Scholar] [CrossRef] [PubMed]
- Balandin, A.A.; Ghosh, S.; Bao, W.; Calizo, I.; Teweldebrhan, D.; Miao, F.; Lau, C.N. Superior thermal conductivity of single-layer graphene. Nano Lett. 2008, 8, 902–907. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Y.; Zhou, S.; Hu, P.; Zhao, G.; Li, Y.; Zhang, X.; Han, W. Enhanced mechanical, thermal, and electric properties of graphene aerogels via supercritical ethanol drying and high-temperature thermal reduction. Sci. Rep. 2017, 7. [Google Scholar] [CrossRef] [PubMed]
- Sun, W.; Du, A.; Gao, G.; Shen, J.; Wu, G. Graphene-templated carbon aerogels combining with ultra-high electrical conductivity and ultra-low thermal conductivity. Microporous Mesoporous Mater. 2017, 253, 71–79. [Google Scholar] [CrossRef]
- Tran, V.-T.; Saint-Martin, J.; Dollfus, P.; Volz, S. Optimizing the thermoelectric performance of graphene nano-ribbons without degrading the electronic properties. Sci. Rep. 2017, 7. [Google Scholar] [CrossRef] [PubMed]
- Wei, A.; Li, Y.; Li, Y.; Ye, H. Thermal characteristics of graphene nanosheet with graphane domains of varying morphologies. Comput. Mater. Sci. 2017, 138, 192–198. [Google Scholar] [CrossRef]
- Geim, A.K.; Novoselov, K.S. The rise of graphene. Nat. Mater. 2007, 6, 183–191. [Google Scholar] [CrossRef] [PubMed]
- Leary, R.; Westwood, A. Carbonaceous nanomaterials for the enhancement of TiO2 photocatalysis. Carbon 2011, 49, 741–772. [Google Scholar] [CrossRef]
- Hamandia, M.; Berhault, G.; Guillardb, C.; Kochkara, H. Influence of reduced graphene oxide on the synergism between rutileand anatase TiO2 particles in photocatalytic degradation of formic acid. Mol. Catal. 2017, 432, 125–130. [Google Scholar] [CrossRef]
- Morales-Torres, S.; Pastrana-Martínez, L.M.; Figueiredo, J.L.; Faria, J.J.; Silva, A.M.T. Design of graphene-based TiO2 photocatalysts—A review. Environ. Sci. Pollut. Res. 2012, 19, 3676–3687. [Google Scholar] [CrossRef] [PubMed]
- Adamu, H.; Dubey, P.; Anderson, J.A. Probing the role of thermally reduced graphene oxide in enhancing performance of TiO2 in photocatalytic phenol removal from aqueous environments. Chem. Eng. J. 2016, 284, 380–388. [Google Scholar] [CrossRef]
- Giovannetti, R.; Rommozzi, E.; Zannotti, M.; D’Amato, C.A.; Ferraro, S.; Cespi, M.; Bonacucina, G.; Minicucci, M.; Di Cicco, A. Exfoliation of graphite into graphene in aqueous solution: An application as graphene/TiO2 nanocomposite to improve visible light photocatalytic activity. RSC Adv. 2016, 6, 93048–93055. [Google Scholar] [CrossRef]
- Truppi, A.; Petronella, F.; Placido, T.; Striccoli, M.; Agostiano, A.; Curri, M.L.; Comparelli, R. Visible-light-active TiO2-based hybrid nanocatalysts for environmental applications. Catalysts 2017, 7, 100. [Google Scholar] [CrossRef]
- Xiang, Q.; Yu, J.; Jaroniec, M. Graphene-based semiconductor photocatalysts. Chem. Soc. Rev. 2012, 41, 782–796. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Tang, Z.R.; Fu, X.Y.; Xu, J. TiO2-graphene nanocomposites for gas-phase photocatalytic degradation of volatile aromatic pollutant: Is TiO2-graphene truly different from other TiO2-carbon composite materials? ACS Nano 2010, 4, 7303–7314. [Google Scholar] [CrossRef] [PubMed]
- Surender Kumar, S. Complex Magnetic Nanostructures: Synthesis, Assembly and Applications; Springer: Cham, Switzerland, 2017. [Google Scholar]
- Syväjärvi, M.; Tiwari, A. Graphene Materials: Fundamentals and Emerging Applications; Wiley: Hoboken, NJ, USA, 2015. [Google Scholar]
- Singh, V.; Joung, D.; Zhai, L.; Das, S.; Khondaker, S.I.; Seal, S. Graphene based materials: Past, present and future. Progress Mater. Sci. 2011, 56, 1178–1271. [Google Scholar] [CrossRef]
- Cong, H.-P.; Chena, J.-F.; Yu, S.-H. Graphene-based macroscopic assemblies and architectures: An emerging material system. Chem. Soc. Rev. 2014, 43, 7295–7325. [Google Scholar] [CrossRef] [PubMed]
- Mahmood, N.; Zhang, C.; Yin, H.; Hou, Y. Graphene-based nanocomposites for energy storage and conversion in lithium batteries, supercapacitors and fuel cells. J. Mater. Chem. A 2014, 2, 15–32. [Google Scholar] [CrossRef]
- Li, X.; Yu, J.; Wageh, S.; Al-Ghamdi, A.A.; Xie, J. Graphene in photocatalysis: A review. Small 2016, 12, 6640–6696. [Google Scholar] [CrossRef] [PubMed]
- Xu, C.; Zhu, J.; Yuan, R.; Fu, X. More effective use of graphene in photocatalysis by conformal attachment of small sheets to TiO2 spheres. Carbon 2016, 96, 394–402. [Google Scholar] [CrossRef]
- Zhu, J.; Yang, D.; Yin, Z.; Yan, Q.; Zhang, H. Graphene and graphene-based materials for energy storage applications. Small 2014, 10, 3480–3498. [Google Scholar] [CrossRef] [PubMed]
- Piao, Y. Preparation of porous graphene-based nanomaterials for electrochemical energy storage devices. In Nano Devices and Circuit Techniques for Low-Energy Applications and Energy Harvesting; Kyung, C.M., Ed.; KAIST Research Series; Springer: Dordrecht, The Netherland, 2016. [Google Scholar]
- Li, N.; Liu, G.; Zhen, C.; Li, F.; Zhang, L.; Cheng, H.M. Battery performance and photocatalytic activity of mesoporous anatase TiO2 nanospheres/graphene composites by template-free self-assembly. Adv. Funct. Mater. 2011, 21, 1717–1722. [Google Scholar] [CrossRef]
- Maroni, F.; Raccichini, R.; Birrozzi, A.; Carbonari, G.; Tossici, R.; Croce, F.; Marassi, R.; Nobili, F. Graphene/silicon nanocomposite anode with enhanced electrochemical stability for lithium-ion battery applications. J. Power Sources 2014, 269, 873–882. [Google Scholar] [CrossRef]
- Kaplas, T.; Kuzhir, P. Ultra-thin Graphitic Film: Synthesis and Physical Properties. Nanoscale Res. Lett. 2016, 11, 54. [Google Scholar] [CrossRef] [PubMed]
- Allagui, A.; Abdelkareem, M.A.; Alawadhi, H.; Elwakil, A.S. Reduced graphene oxide thin film on conductive substrates by bipolar electrochemistry. Sci. Rep. 2016, 6. [Google Scholar] [CrossRef] [PubMed]
- Bhuyan, M.S.A.; Uddin, M.N.; Islam, M.M.; Bipasha, F.A.; Hossain, S.S. Synthesis of graphene. Int. Nano Lett. 2016, 6, 65–83. [Google Scholar] [CrossRef]
- Tetlow, H.; de Boer, J.P.; Ford, I.J.; Vvedenskyb, D.D.; Coraux, J.; Kantorovich, L. Growth of epitaxial graphene: Theory and experiment. Phys. Rep. 2014, 542, 195–295. [Google Scholar] [CrossRef]
- Cui, Y.; Zhang, H.; Chen, W.; Yang, Z.; Cai, Q. Structural evolution of flower defects and effects on the electronic structures of epitaxial graphene. J. Phys. Chem. C 2017, 121, 15282–15287. [Google Scholar] [CrossRef]
- Galves, L.A.; Wofford, J.M.; Soares, G.V.; Jahn, U.; Pfüller, C.; Riechert, H.; Lopes, J.M.J. The effect of the SiC(0001) surface morphology on the growth of epitaxial mono-layer graphene nanoribbons. Carbon 2017, 115, 162–168. [Google Scholar] [CrossRef]
- Ramnani, P.; Neupane, M.R.; Ge, S.; Balandin, A.A.; Lake, R.K.; Mulchandani, A. Raman spectra of twisted CVD bilayer graphene. Carbon 2017, 123, 302–306. [Google Scholar] [CrossRef]
- Chena, X.; Zhangb, L.; Chena, S. Large area CVD growth of graphene. Synth. Met. 2015, 210, 95–108. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhang, L.; Zhou, C. Review of chemical vapor deposition of graphene and related applications. Acc. Chem. Res. 2013, 46, 2329–2339. [Google Scholar] [CrossRef] [PubMed]
- Lukosius, M.; Dabrowski, J.; Kitzmann, J.; Fursenko, O.; Akhtar, F.; Lisker, M.; Lippert, G.; Schulze, S.; Yamamoto, Y.; Schubert, M.A.; et al. Metal-free CVD graphene synthesis on 200 mm Ge/Si(001) substrates. ACS Appl. Mater. Interfaces 2016, 8, 33786–33793. [Google Scholar] [CrossRef] [PubMed]
- Robert, M.; Jacobberger, R.M.; Machhi, R.; Wroblewski, J.; Ben Taylor, B.; Anne Lynn Gillian-Daniel, A.L.; Arnold, M.S. Simple graphene synthesis via chemical vapor deposition. J. Chem. Educ. 2015, 92, 1903–1907. [Google Scholar]
- Kim, K.S.; Zhao, Y.; Jang, H.; Lee, S.Y.; Kim, J.M.; Kim, K.S.; Ahn, J.H.; Kim, P.; Choi, J.Y.; Hong, B.H. Large-scale pattern growth of graphene films for stretchable transparent electrodes. Nature 2009, 457, 706–710. [Google Scholar] [CrossRef] [PubMed]
- Emtsev, K.V.; Bostwick, A.; Horn, K.; Jobst, J.; Kellogg, G.L.; Ley, L.; McChesney, J.L.; Ohta, T.; Reshanov, S.A.; Rohrl, J.; et al. Towards wafer-size graphene layers by atmospheric pressure graphitization of silicon carbide. Nat. Mater. 2009, 8, 203–207. [Google Scholar] [CrossRef] [PubMed]
- Juang, Z.Y.; Wu, C.Y.; Lu, A.Y.; Su, C.Y.; Leou, K.C.; Chen, F.R.; Tsai, C.H. Graphene synthesis by chemical vapor deposition and transfer by a roll-to-roll process. Carbon 2010, 48, 3169–3174. [Google Scholar] [CrossRef]
- Wei, D.C.; Liu, Y.Q.; Wang, Y.; Zhang, H.L.; Huang, L.P.; Yu, G. Synthesis of N-doped graphene by chemical vapor deposition and its electrical properties. Nano Lett. 2009, 9, 1752–1758. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Zhang, X.; Sun, X.; Ma, Y. Shape-controlled synthesis of nanocarbons through direct conversion of carbon dioxide. Sci. Rep. 2013, 3. [Google Scholar] [CrossRef] [PubMed]
- Hajian, M.; Zareie, M.; Hashemiana, D.; Bahrami, M. Synthesis of graphene through direct decomposition of CO2. RSC Adv. 2016, 6, 73331–73335. [Google Scholar] [CrossRef]
- Qin, B.; Zhang, T.; Chen, H.; Ma, Y. The growth mechanism of few-layer graphene in the arc discharge process. Carbon 2016, 102, 494–498. [Google Scholar] [CrossRef]
- Poorali, M.S.; Bagheri-Mohagheghi, M.M. Synthesis and physical properties of multi-layered graphene sheets by Arc-discharge method with TiO2 and ZnO catalytic. J. Mater. Sci. Mater. Electron. 2017, 28, 6186–6193. [Google Scholar] [CrossRef]
- Xiao, B.; Li, X.; Li, X.; Wang, B.; Langford, C.; Li, R.; Sun, X. Graphene Nanoribbons Derived from the Unzipping of Carbon Nanotubes: Controlled Synthesis and Superior Lithium Storage. J. Phys. Chem. C 2014, 118, 881–890. [Google Scholar] [CrossRef]
- Yang, X.; Dou, X.; Rouhanipour, A.; Zhi, L.; Räder, H.J.; Mülle, K. Two-dimensional graphene nanoribbons. J. Am. Chem. Soc. 2008, 130, 4216–4217. [Google Scholar] [CrossRef] [PubMed]
- Kharisov, B.I.; Kharissova, O.V. Synthesis of graphene by pyrolysis of organic matter. In Graphene Science Handbook: Fabrication Methods; Aliofkhazraei, M., Ali, N., Milne, W.I., Ozkan, C.S., Mitura, S., Gervasoni, J.L., Eds.; CRC Press: Boca Raton, FL, USA, 2016; pp. 345–360. [Google Scholar]
- Zou, B.; Wang, X.X.; Huang, X.X.; Wang, J.N. Continuous synthesis of graphene sheets by spray pyrolysis and their use as catalysts for fuel cells. Chem. Commun. 2015, 51, 741–744. [Google Scholar] [CrossRef] [PubMed]
- Jabari-Seresht, R.; Jahanshahi, M.; Rashidi, A.; Ghoreyshi, A.A. Fabrication and evaluation of nonporous graphene by a unique spray pyrolysis method. Chem. Eng. Technol. 2013, 36, 1550–1558. [Google Scholar] [CrossRef]
- Khan, M.; Tahir, M.N.; Adil, S.F.; Khan, H.U.; Siddiqui, M.R.H.; Al-warthan, A.A.; Tremel, W. Graphene based metal and metal oxide nanocomposites: Synthesis, properties and their applications. J. Mater. Chem. A 2015, 3, 18753–18808. [Google Scholar] [CrossRef]
- Kim, H.; Abdala, A.A.; Macosko, C.W. Graphene/polymer nanocomposites. Macromolecules 2010, 43, 6515–6530. [Google Scholar] [CrossRef]
- Yi, M.; Shen, Z. A review on mechanical exfoliation for the scalable production of graphene. J. Mater. Chem. A 2015, 3, 11700–11715. [Google Scholar] [CrossRef]
- Hernandez, Y.; Nicolosi, V.; Lotya, M.; Blighe, F.M.; Sun, Z.Y.; De, S.; McGovern, I.T.; Holland, B.; Byrne, M.; Gunko, Y.K.; et al. High-yield production of graphene by liquid-phase exfoliation of graphite. Nat. Nanotechnol. 2008, 3, 563–568. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Zhang, G.; Bai, X.; Sun, X.; Wang, X.; Wang, E.; Dai, H. Highly conducting graphene sheets and Langmuir-Blodgett films. Nat. Nanotechnol. 2008, 3, 538–542. [Google Scholar] [CrossRef] [PubMed]
- Coscia, U.; Palomba, M.; Ambrosone, G.; Barucca, G.; Cabibbo, M.; Mengucci, P.; de Asmundis, R.; Carotenuto, G. A new micromechanical approach for the preparation of graphene nanoplatelets deposited on polyethylene. Nanotechnology 2017, 28. [Google Scholar] [CrossRef] [PubMed]
- Zhong, Y.L.; Swager, T.M. Enhanced electrochemical expansion of graphite for in situ electrochemical functionalization. J. Am. Chem. Soc. 2012, 134, 17896–17899. [Google Scholar] [CrossRef] [PubMed]
- Leroux, Y.R.; Jean-François Bergamini, J.-F.; Ababou, S.; Le Breton, J.-C.; Hapiot, P. Synthesis of functionalized few-layer graphene through fast electrochemical expansion of graphite. J. Electroanal. Chem. 2015, 753, 42–46. [Google Scholar] [CrossRef]
- Hossaina, S.T.; Wanga, R. Electrochemical exfoliation of graphite: Effect of temperature and hydrogen peroxide addition. Electrochim. Acta 2016, 216, 253–260. [Google Scholar] [CrossRef]
- McAllister, M.J.; Li, J.L.; Adamson, D.H.; Schniepp, H.C.; Abdala, A.A.; Liu, J.; Herrera-Alonso, M.; Milius, D.L.; Car, R.; Prudhomme, R.K.; et al. Single sheet functionalized graphene by oxidation and thermal expansion of graphite. Chem. Mater. 2007, 19, 4396–4404. [Google Scholar] [CrossRef]
- Beidaghi, M.; Wang, Z.; Gu, L.; Wang, C. Electrostatic spray deposition of graphene nanoplatelets for high-power thin-film supercapacitor electrodes. J. Solid State Electrochem. 2012, 16, 3341–3348. [Google Scholar] [CrossRef]
- Allen, M.J.; Tung, V.C.; Kaner, R.B. Honeycomb carbon: A review of graphene. Chem. Rev. 2010, 110, 132–145. [Google Scholar] [CrossRef] [PubMed]
- Viculis, L.M.; Mack, J.J.; Kaner, R.B. A chemical route to carbon nanoscrolls. Science 2003, 299. [Google Scholar] [CrossRef] [PubMed]
- Park, S.; Ruoff, R.S. Chemical methods for the production of graphenes. Nat. Nanotechnol. 2009, 4, 217–224. [Google Scholar] [CrossRef] [PubMed]
- Jiao, L.Y.; Wang, X.R.; Diankov, G.; Wang, H.L.; Dai, H.J. Facile synthesis of high-quality graphene nanoribbons. Nat. Nanotechnol. 2010, 5, 321–325. [Google Scholar] [CrossRef] [PubMed]
- Kosynkin, D.V.; Higginbotham, A.L.; Sinitskii, A.; Lomeda, J.R.; Dimiev, A.; Price, B.K.; Tour, J.M. Longitudinal unzipping of carbon nanotubes to form graphene nanoribbons. Nature 2009, 458, 872–876. [Google Scholar] [CrossRef] [PubMed]
- Jiao, L.Y.; Zhang, L.; Wang, X.R.; Diankov, G.; Dai, H.J. Narrow graphene nanoribbons from carbon nanotubes. Nature 2009, 458, 877–880. [Google Scholar] [CrossRef] [PubMed]
- Lin, T.; Liu, Z.; Zhou, M.; Bi, H.; Zhang, K.; Huang, F.; Wan, D.; Zhong, Y. Rapid microwave synthesis of graphene directly on h-BN with excellent heat dissipation performance. ACS Appl. Mater. Interfaces 2014, 6, 3088–3092. [Google Scholar] [CrossRef] [PubMed]
- Xin, G.Q.; Hwang, W.; Kim, N.; Cho, S.M.; Chae, H. A graphene sheet exfoliated with microwave irradiation and interlinked by carbon nanotubes for high-performance transparent flexible electrodes. Nanotechnology 2010, 21. [Google Scholar] [CrossRef] [PubMed]
- Shen, Y.; Jing, T.; Ren, W.; Zhang, J.; Jiang, Z.-G.; Yu, Z.-Z.; Dasari, A. Chemical and thermal reduction of graphene oxide and its electrically conductive polylactic acid nanocomposites. Compos. Sci. Technol. 2012, 72, 1430–1435. [Google Scholar] [CrossRef]
- Cote, L.J.; Kim, F.; Huang, J. Langmuir-Blodgett assembly of graphite oxide single layers. J. Am. Chem. Soc. 2009, 131, 1043–1049. [Google Scholar] [CrossRef] [PubMed]
- Loh, K.P.; Bao, Q.L.; Ang, P.K.; Yang, J.X. The chemistry of graphene. J. Mater. Chem. 2010, 20, 2277–2289. [Google Scholar] [CrossRef]
- Moon, I.K.; Lee, J. Highly qualified reduced graphene oxides: The best chemical reduction. Chem. Commun. 2011, 47, 9681–9683. [Google Scholar] [CrossRef] [PubMed]
- Chua, C.K.; Pumera, M. Chemical reduction of graphene oxide: A synthetic chemistry viewpoint. Chem. Soc. Rev. 2014, 43, 291–312. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Hernandez, Y.; Feng, X.; Mullen, K. From nanographene and graphene nanoribbons to graphene sheets: Chemical synthesis. Angew. Chem. Int. Ed. Engl. 2012, 51, 7640–7654. [Google Scholar] [CrossRef] [PubMed]
- Barg, S.; Perez, F.M.; Ni, N.; Pereira, P.V.; Maher, R.C.; Garcia-Tunon, E.; Eslava, S.; Agnoli, S.; Mattevi, C.; Saiz, E. Mesoscale assembly of chemically modified graphene into complex cellular networks. Nat. Commun. 2014, 5, 4328. [Google Scholar] [CrossRef] [PubMed]
- Ruoff, R. Graphene: Calling all chemists. Nat. Nano 2008, 3, 10–11. [Google Scholar] [CrossRef] [PubMed]
- Paredes, J.I.; Villar-Rodil, S.; Martinez-Alonso, A.; Tascon, J.M.D. Graphene oxide dispersions in organic solvents. Langmuir 2008, 24, 10560–10564. [Google Scholar] [CrossRef] [PubMed]
- Bianco, A.; Cheng, H.-M.; Enoki, T.; Gogotsi, Y.; Hurt, R.H.; Koratkar, N.; Kyotani, T.; Monthioux, M.; Park, C.R.; Tascon, J.M.D.; et al. All in the graphene family—A recommended nomenclature for two dimensional carbon materials. Carbon 2013, 65, 1–6. [Google Scholar] [CrossRef]
- Poh, H.L.; Sanek, F.; Ambrosi, A.; Zhao, G.; Sofer, Z.; Pumera, M. Graphenes prepared by Staudenmaier, Hofmann and Hummer’s methods with consequent thermal exfoliation exhibit very different electrochemical properties. Nanoscale 2012, 4, 3515–3522. [Google Scholar] [CrossRef] [PubMed]
- Compton, O.C.; Nguyen, S.T. Graphene oxide, highly reduced graphene oxide, and graphene: Versatile building blocks for carbon-based materials. Small 2010, 6, 711–723. [Google Scholar] [CrossRef] [PubMed]
- Dreyer, D.R.; Park, S.; Bielawski, C.W.; Ruoff, R.S. The chemistry of graphene oxide. Chem. Soc. Rev. 2010, 39, 228–240. [Google Scholar] [CrossRef] [PubMed]
- Brodie, B.C. On the atomic weight of graphite. Philos. Trans. R. Soc. Lond. 1859, 149, 249–259. [Google Scholar] [CrossRef]
- Staudenmaier, L. Verfahren zur darstellung der graphitsaure. Ber. Dtsch. Chem. Ges. 1898, 31, 1481–1487. [Google Scholar] [CrossRef]
- Hummers, W.S., Jr.; Offeman, R.E. Preparation of graphitic oxide. J. Am. Chem. Soc. 1958, 80, 1339. [Google Scholar] [CrossRef]
- Schniepp, H.C.; Li, J.L.; McAllister, M.J.; Sai, H.; Herrera-Alonso, M.; Adamson, D.H.; Prud’homme, R.K.; Car, R.; Saville, D.A.; Aksay, I.A. Functionalized single graphene sheets derived from splitting graphite oxide. J. Phys. Chem. B 2006, 110, 8535–8539. [Google Scholar] [CrossRef] [PubMed]
- Gao, X.F.; Jang, J.; Nagase, S. Hydrazine and thermal reduction of graphene oxide: Reaction mechanisms, product structures, and reaction design. J. Phys. Chem. C 2010, 114, 832–842. [Google Scholar] [CrossRef]
- Ramesha, G.K.; Sampath, S. Electrochemical reduction of oriented graphene oxide films: An in situ Raman spectroelectrochemical study. J. Phys. Chem. C 2009, 113, 7985–7989. [Google Scholar] [CrossRef]
- Abdelsayed, V.; Moussa, S.; Hassan, H.M.; Aluri, H.S.; Collinson, M.M.; El-Shall, M.S. Photothermal deoxygenation of graphite oxide with laser excitation in solution and graphene-aided increase in water temperature. J. Phys. Chem. Lett. 2010, 1, 2804–2809. [Google Scholar] [CrossRef]
- Ng, Y.H.; Lightcap, I.V.; Goodwin, K.; Matsumura, M.; Kamat, P.V. To what extent do graphene scaffolds improve the photovoltaic and photocatalytic response of TiO2 nanostructured films? J. Phys. Chem. Lett. 2010, 1, 2222–2227. [Google Scholar] [CrossRef]
- Golsheikh, A.M.; Lim, H.N.; Zakaria, R.; Huang, N.M. Sonochemical synthesis of reduced graphene oxide uniformly decorated with hierarchical ZnS nanospheres and its enhanced photocatalytic activities. RSC Adv. 2015, 5, 12726–12735. [Google Scholar] [CrossRef]
- Vinodgopal, K.; Neppolian, B.; Lightcap, I.V.; Grieser, F.; Ashokkumar, M.; Kamat, P.V. Sonolytic design of graphene-Au nanocomposites. Simultaneous and sequential reduction of graphene oxide and Au(III). J. Phys. Chem. Lett. 2010, 1, 1987–1993. [Google Scholar] [CrossRef]
- Voiry, D.; Yang, J.; Kupferberg, J.; Fullon, R.; Lee, C.; Jeong, H.Y.; Shin, H.S.; Chhowalla, M. High-quality graphene via microwave reduction of solution-exfoliated graphene oxide. Science 2016, 353, 1413–1416. [Google Scholar] [CrossRef] [PubMed]
- Mills, A.; Le Hunte, S. An overview of semiconductor photocatalysis. J. Photochem. Photobiol. A 1997, 108, 1–35. [Google Scholar] [CrossRef]
- Mahlambi, M.M.; Ngila, C.J.; Mamba, B.B. Recent developments in environmental photocatalytic degradation of organic pollutants: The case of Titanium dioxide nanoparticles—A review. J. Nanomater. 2015, 2015. [Google Scholar] [CrossRef]
- Nosaka, Y.; Nosaka, A. Understanding hydroxyl radical (•OH) generation processes in photocatalysis. ACS Energy Lett. 2016, 1, 356–359. [Google Scholar] [CrossRef]
- Zhang, Y.; Tan, Y.; Stormer, H.L.; Kim, P. Experimental observation of the quantum Hall effect and Berry’s phase in graphene. Nature 2005, 438, 201–204. [Google Scholar] [CrossRef] [PubMed]
- Teh, C.M.; Mohamed, A.R. Roles of titanium dioxide and ion-doped titanium dioxide on photocatalytic degradation of organic pollutants (phenolic compounds and dyes) in aqueous solutions: A review. J. Alloys Compd. 2011, 509, 1648–1660. [Google Scholar] [CrossRef]
- Hager, S.; Bauer, R. Heterogeneous photocatalytic oxidation of organics for air purification by near UV irradiated titanium dioxide. Chemosphere 1999, 38, 1549–1559. [Google Scholar] [CrossRef]
- Faraldos, M.; Bahamonde, A. Environmental applications of titania-graphene photocatalysts. Catal. Today 2017, 285, 13–28. [Google Scholar] [CrossRef]
- Upadhyay, R.K.; Soin, N.; Ro, S.S. Role of graphene/metal oxide composites as photocatalysts, adsorbents and disinfectants in water treatment: A review. RSC Adv. 2014, 4, 3823–3851. [Google Scholar] [CrossRef]
- Fujishima, A.; Rao, T.N.; Tryk, D.A. Titanium dioxide photocatalysis. J. Photochem. Photobiol. C Photochem. Rev. 2000, 1, 1–21. [Google Scholar] [CrossRef]
- Hu, G.; Tang, B. Photocatalytic mechanism of graphene/titanate nanotubes photocatalyst under visible-light irradiation. Mater. Chem. Phys. 2013, 138, 608–614. [Google Scholar] [CrossRef]
- Cruz, M.; Gomez, C.; Duran-Valle, C.J.; Pastrana-Martínez, L.M.; Faria, J.L.; Silva, A.M.T.; Faraldosa, M.; Bahamondea, A. Bare TiO2 and graphene oxide TiO2 photocatalysts on the degradation of selected pesticides and influence of the water matrix. Appl. Surf. Sci. 2017, 416, 1013–1021. [Google Scholar] [CrossRef]
- Wang, C.; Cao, M.; Wang, P.; Ao, Y.; Hou, J.; Qian, J. Preparation of graphene-TiO2 composites with enhanced photocatalytic activity for the removal of dye and Cr (VI). Appl. Catal. A Gen. 2014, 473, 83–89. [Google Scholar] [CrossRef]
- Pastrana-Martínez, L.M.; Morales-Torres, S.; Likodimos, V.; José, L.; Figueiredo, J.L.; Faria, J.L.; Falaras, P.; Silva, A.M.T. Advanced nanostructured photocatalysts based on reduced graphene oxide-TiO2 composites for degradation of diphenhydramine pharmaceutical and methyl orange dye. Appl. Catal. B 2012, 123, 241–256. [Google Scholar] [CrossRef]
- Liu, S.; Sun, H.; Liu, S.; Wang, S. Graphene facilitated visible light photodegradation of methylene blue over titanium dioxide photocatalysts. Chem. Eng. J. 2013, 214, 298–303. [Google Scholar] [CrossRef]
- Zhang, L.-W.; Fu, H.-B.; Zhu, Y.-F. Efficient TiO2 photocatalysts from surface hybridization of TiO2 particles with graphite-like carbon. Adv. Funct. Mater. 2008, 18, 2180–2189. [Google Scholar] [CrossRef]
- Ren, L.; Qi, X.; Liu, Y.; Huang, Z.; Wei, X.; Li, J.; Yang, L.; Zhong, J. Upconversion-P25–graphene composite as an advanced sunlight driven photocatalytic hybrid material. J. Mater. Chem. 2012, 22, 11765–11771. [Google Scholar] [CrossRef]
- Long, R.; English, N.J.; Prezhdo, O.V. Photo-induced charge separation across the graphene-TiO2 interface is faster than energy losses: A time-domain ab initio analysis. J. Am. Chem. Soc. 2012, 134, 14238–14248. [Google Scholar] [CrossRef] [PubMed]
- Rehman, S.; Ullah, R.; Butt, A.M.; Gohar, N.D. Strategies of making TiO2 and ZnO visible light active. J. Hazard. Mater. 2009, 170, 560–569. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.; Wu, X.-L.; Wang, Z.-M.; Aoki, H.; Kutsuna, S.; Keiko Jimura, K.; Hayashi, S. Anchoring titanium dioxide on carbon spheres for high-performancevisible light photocatalysis. Appl. Catal. B Environ. 2017, 207, 255–266. [Google Scholar] [CrossRef]
- Warkhade, S.K.; Gaikwad, G.S.; Zodape, S.P.; Pratap, U.; Maldhure, A.V.; Wankhade, A.V. Low temperature synthesis of pure anatase carbon doped titanium dioxide: An efficient visible light active photocatalyst. Mater. Sci. Semicond. Process. 2017, 63, 18–24. [Google Scholar] [CrossRef]
- Jiang, W.J.; Zhu, Y.F.; Zhu, G.X.; Zhang, Z.J.; Chen, X.J.; Yao, W.Q. Three-dimensional photocatalysts with a network structure. J. Mater. Chem. A 2017, 5, 5661–5679. [Google Scholar] [CrossRef]
- Williams, G.; Seger, B.; Kamat, P.V. TiO2–graphene nanocomposites. UV-assisted photocatalytic reduction of graphene oxide. ACS Nano 2008, 2, 1487–1491. [Google Scholar] [CrossRef] [PubMed]
- Pan, X.; Yang, M.-Q.; Tang, Z.R.; Xu, Y.-J. Noncovalently functionalized graphene-directed synthesis of ultralarge graphene-based TiO2 nanosheet composites: Tunable morphology and photocatalytic applications. J. Phys. Chem. C 2014, 118, 27325–27335. [Google Scholar] [CrossRef]
- Kamegawa, T.; Yamahana, D.; Yamashita, H. Graphene coating of TiO2 nanoparticles loaded on mesoporous silica for enhancement of photocatalytic activity. J. Phys. Chem. C 2010, 114, 15049–15053. [Google Scholar] [CrossRef]
- Bekyarova, E.; Itkis, M.E.; Ramesh, P.; Berger, C.; Sprinkle, M.; de Heer, W.A.; Haddon, R.C. Chemical modification of epitaxial graphene: Spontaneous grafting of aryl groups. J. Am. Chem. Soc. 2009, 131, 1336–1337. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Bao, N.; Wang, X.; Hu, X.; Miao, X.; Chaker, M.; Ma, D. Advanced fabrication of chemically bonded graphene/TiO2 continuous fibers with enhanced broadband photocatalytic properties and involved mechanisms exploration. Sci. Rep. 2016, 6. [Google Scholar] [CrossRef] [PubMed]
- Yao, Y.; Jie, G.; Bao, F.; Jiang, S.; Zhang, X.; Ma, R. Covalent functionalization of graphene with polythiophene through a Suzuki coupling reaction. RSC Adv. 2015, 5, 42754–42761. [Google Scholar] [CrossRef]
- He, F.; Fan, J.; Ma, D.; Zhang, L.; Leung, C.; Chan, H.L. The attachment of Fe3O4 nanoparticles to graphene oxide by covalent bonding. Carbon 2010, 48, 3139–3144. [Google Scholar] [CrossRef]
- Morawskia, A.W.; Kusiak-Nejmana, E.; Wanaga, A.; Kapica-Kozara, J.; Wróbela, R.J.; Ohtanib, B.; Aksienionekc, M.; Lipinska, L. Photocatalytic degradation of acetic acid in the presence of visiblelight-active TiO2-reduced graphene oxide photocatalysts. Catal. Today 2017, 280, 108–113. [Google Scholar] [CrossRef]
- Liu, Y.; Zhang, D.; Shang, Y.; Zang, W.; Li, M. Construction of multifunctional films based on graphene-TiO2 composite materials for strain sensing and photodegradation. RSC Adv. 2015, 5, 104785–104791. [Google Scholar] [CrossRef]
- Raliya, R.; Avery, C.; Chakrabarti, S.; Biswas, P. Photocatalytic degradation of methyl orange dye by pristine titanium dioxide, zinc oxide, and graphene oxide nanostructures and their composites under visible light irradiation. Appl. Nanosci. 2017, 7, 253–259. [Google Scholar] [CrossRef]
- Kuila, T.; Bose, S.; Mishra, A.K.; Kharna, P.; Kim, N.H.; Lee, J.H. Recent advances in graphene based polymer composites. Progress Mater. Sci. 2012, 57, 1061–1105. [Google Scholar] [CrossRef]
- Huang, X.; Qi, X.; Freddy Boeyab, F.; Zhang, H. Graphene-based composites. Chem. Soc. Rev. 2011, 41, 666–686. [Google Scholar] [CrossRef] [PubMed]
- Bai, H.; Li, C.; Shi, G. Functional composite materials based on chemically converted graphene. Adv. Mater. 2011, 23, 1089–1115. [Google Scholar] [CrossRef] [PubMed]
- Sreeprasad, T.S.; Berry, V. How do the electrical properties of graphene change with its functionalization? Small 2013, 9, 341–350. [Google Scholar] [CrossRef] [PubMed]
- Pan, X.; Zhao, Y.; Liu, S.; Korzeniewski, C.L.; Wang, S.; Fan, Z. Comparing graphene-TiO2 nanowire and graphene-TiO2 nanoparticle composite photocatalysts. ACS Appl. Mater. Interfaces 2012, 4, 3944–3950. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.S.; You, K.H.; Park, C.B. Highly photoactive, low bandgap TiO2 nanoparticles wrapped by graphene. Adv. Mater. 2012, 24, 1084–1088. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Salihi, E.C.; Šiller, L. Green reduction of graphene oxide using alanine. Mater. Sci. Eng. C 2017, 72, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Pei, S.; Cheng, H.-M. The reduction of graphene oxide. Carbon 2012, 50, 3210–3228. [Google Scholar] [CrossRef]
- Bell, N.J.; Ng, Y.H.; Du, A.; Coster, H.; Smith, S.C.; Amal, R. Understanding the enhancement in photoelectrochemical properties of photocatalytically prepared TiO2- reduced graphene oxide composite. J. Phys. Chem. C 2011, 115, 6004–6009. [Google Scholar] [CrossRef]
- Zhang, Y.; Pan, C. TiO2/graphene composite from thermal reaction of graphene oxide and its photocatalytic activity in visible light. J. Mater. Sci. 2011, 46, 2622–2626. [Google Scholar] [CrossRef]
- Zhang, X.-Y.; Li, H.-P.; Cui, X.-L.; Linb, Y. Graphene/TiO2 nanocomposites: Synthesis, characterization and application in hydrogen evolution from water photocatalytic splitting. J. Mater. Chem. 2010, 20, 2801–2806. [Google Scholar] [CrossRef]
- Zabihi, F.; Ahmadian-Yazdi, M.-R.; Eslamian, M. Photocatalytic graphene-TiO2 thin films fabricated by low-temperature ultrasonic vibration-assisted spin and spray coating in a sol-gel process. Catalysts 2017, 7, 136. [Google Scholar] [CrossRef]
- Gopalakrishnan, A.; Binitha, N.N.; Yaakob, Z. Excellent photocatalytic activity of titania-graphene nanocomposites prepared by a facile route. J. Sol-Gel Sci. Technol. 2016, 80, 189–200. [Google Scholar] [CrossRef]
- Shao, L.; Quan, S.; Liu, Y.; Guo, Z.; Wang, Z. A novel “gel-sol” strategy to synthesize TiO2 nanorod combining reduced graphene oxide composites. Mater. Lett. 2013, 107, 307–310. [Google Scholar] [CrossRef]
- Long, M.; Qin, Y.; Chen, C.; Guo, X.; Tan, B.; Cai, W. Origin of visible light photoactivity of reduced graphene oxide/TiO2 by in situ hydrothermal growth of undergrown TiO2 with graphene oxide. J. Phys. Chem. C 2013, 117, 16734–16741. [Google Scholar] [CrossRef]
- Zhang, H.; Lv, X.; Li, Y.; Wang, Y.; Li, J. P25-graphene composite as a high performance photocatalyst. ACS Nano 2010, 4, 380–386. [Google Scholar] [CrossRef] [PubMed]
- Hu, G.; Yang, J.; Zhao, D.; Chen, Y.; Cao, Y. Research on photocatalytic properties of TiO2-graphene composites with different morphologies. J. Mater. Eng. Perform. 2017, 26, 3263–3270. [Google Scholar] [CrossRef]
- Hamandi, M.; Berhault, G.; Guillard, C.; Kochkar, H. Reduced graphene oxide/TiO2 nanotube composites for formic acid photodegradation. Appl. Catal. B Environ. 2017, 209, 203–213. [Google Scholar] [CrossRef]
- Lv, S.; Wan, J.; Shen, Y.; Hu, Z. Preparation of superlong TiO2 nanotubes and reduced graphene oxide composite photocatalysts with enhanced photocatalytic performance under visible light irradiation. J. Mater. Sci. Mater. Electron. 2017, 28, 14769–14776. [Google Scholar] [CrossRef]
- Kusiak-Nejmana, E.; Wanaga, A.; Kowalczyka, L.; Kapica-Kozara, J.; Colbeau-Justinb, C.; Mendez Medrano, M.G.; Morawsk, A.W. Graphene oxide-TiO2 and reduced graphene oxide-TiO2 nanocomposites: Insight in charge-carrier lifetime measurements. Catal. Today 2017, 287, 189–195. [Google Scholar] [CrossRef]
- Li, T.; Wang, T.; Qu, G.; Qu, G.; Liang, D.; Hu, S. Synthesis and photocatalytic performance of reduced graphene oxide-TiO2 nanocomposites for orange II degradation under UV light irradiation. Environ. Sci. Pollut. Res. 2017, 24, 12416–12425. [Google Scholar] [CrossRef] [PubMed]
- Yadav, H.M.; Kim, J.-S. Solvothermal synthesis of anatase TiO2-graphene oxide nanocomposites and their photocatalytic performance. J. Alloys Compd. 2016, 688, 123–129. [Google Scholar] [CrossRef]
- Hu, J.J.; Lia, H.; Muhammada, S.; Wua, Q.; Zhaoa, Y.; Jiaoa, Q. Surfactant-assisted hydrothermal synthesis of TiO2/reduced graphene oxide nanocomposites and their photocatalytic performances. J. Solid State Chem. 2017, 253, 113–120. [Google Scholar] [CrossRef]
- Ge, M.-Z.; Li, S.-H.; Huang, J.-Y.; Zhang, K.-Q.; Al-Deyabc, S.S.; Lai, Y.-K. TiO2 nanotube arrays loaded with reduced graphene oxide films: Facile hybridization and promising photocatalytic application. J. Mater. Chem. A 2015, 3, 3491–3499. [Google Scholar] [CrossRef]
- De Oliveira, A.G.; Nascimento, J.P.; de Fátima Gorgulho, H.; Martelli, P.B.; Furtado, C.A.; Figueiredo, J.L. Electrochemical synthesis of TiO2/Graphene oxide composite films for photocatalytic applications. J. Alloys Compd. 2016, 654, 514–522. [Google Scholar] [CrossRef]
- Shanmugam, M.; Alsalme, A.; Alghamdi, A.; Jayavel, R. In-situ microwave synthesis of graphene-TiO2 nanocomposites with enhanced photocatalytic properties for the degradation of organic pollutants. J. Photochem. Photobiol. B Biol. 2016, 163, 216–223. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Gao, H.; Chen, M.; Xu, X.; Wang, X.; Pan, C.; Gao, J. Microwave-assisted synthesis of reduced graphene oxide/titania nanocomposites as an adsorbent for methylene blue adsorption. J. Appl. Surf. Sci. 2016, 360, 840–848. [Google Scholar] [CrossRef]
- Yang, Y.; Liu, E.; Fan, J.; Hu, X.; Hou, W.; Wu, F.; Ma, Y. Green and facile microwave-assisted synthesis of TiO2/graphene nanocomposite and their photocatalytic activity for methylene blue degradation. Russ. J. Phys. Chem. A 2014, 88, 478–483. [Google Scholar] [CrossRef]
- Pu, S.; Zhu, R.; Ma, H.; Deng, D.; Pei, X.; Qi, F.; Chu, W. Facile in-situ design strategy to disperse TiO2 nanoparticles on graphene for the enhanced photocatalytic degradation of rhodamine 6G. Appl. Catal. B Environ. 2017, 218, 208–219. [Google Scholar] [CrossRef]
- Pokhrel, D.; Viraraghavan, T. Treatment of pulp and paper mill wastewater—A review. Sci. Total Environ. 2004, 333, 37–58. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Zhang, B.; Zhang, Y.; He, Y.; Huang, L.; Tan, S.; Cai, X. Simultaneous removal of cationic and anionic dyes from environmental water using montmorillonite-pillared graphene oxide. J. Chem. Eng. Data 2015, 60, 1270–1278. [Google Scholar] [CrossRef]
- Bhullar, N.; Sharma, S.; Sud, D. Studies of adsorption for dye effluent and decolonization of methylene blue and methyl blue based on the surface of biomass adsorbent. Pollut. Res. 2012, 31, 681–686. [Google Scholar]
- Hameed, B.H.; Din, A.T.M.; Ahmad, A.L. Adsorption of methylene blue onto bamboo-based activated carbon: Kinetics and equilibrium studies. J. Hazard. Mater. 2007, 141, 819–825. [Google Scholar] [CrossRef] [PubMed]
- Mohamed, H.H. Biphasic TiO microspheres/reduced graphene oxide for effective simultaneous photocatalytic reduction and oxidation processes. Appl. Catal. A Gen. 2017, 541, 25–34. [Google Scholar] [CrossRef]
- Zhang, N.; Li, B.; Li, S.; Yang, S. Graphene-supported mesoporous Titania nanosheets for efficient photodegradation. J. Colloid Interface Sci. 2017, 505, 711–718. [Google Scholar] [CrossRef] [PubMed]
- Najafi, M.; Kermanpur, A.; Rahimipour, M.R.; Najafizadeh, A. Effect of TiO2 morphology on structure of TiO2-graphene oxide nanocomposite synthesized via a one-step hydrothermal method. J. Alloys Compd. 2017, 722, 272–277. [Google Scholar] [CrossRef]
- Divya, K.S.; Madhu, A.K.; Umadevi, T.U.; Suprabha, T.; Radhakrishnan Nair, P.; Suresh, M. Improving the photocatalytic performance of TiO2 via hybridizing with graphene. J. Semiconduct. 2017, 38. [Google Scholar] [CrossRef]
- Sohail, M.; Xue, H.; Jiao, Q.; Li, H.; Khan, K.; Wang, S. Synthesis of well-dispersed TiO2@reduced graphene oxide (rGO) nanocomposites and their photocatalytic properties. Mater. Res. Bull. 2017, 90, 125–130. [Google Scholar] [CrossRef]
- Tseng, I.-H.; Sung, Y.-M.; Chang, P.-Y.; Lin, S.-W. Photocatalytic performance of Titania nanosheets templated by graphene oxide. J. Photochem. Photobiol. A Chem. 2017, 339, 1–11. [Google Scholar] [CrossRef]
- Minella, M.; Sordello, F.; Minero, C. Photocatalytic process in TiO2/graphene hybrid materials. Evidence of charge separation by electron transfer from reduced graphene oxide to TiO2. Catal. Today 2017, 281, 29–37. [Google Scholar] [CrossRef]
- Raja, V.; Shiamala, L. Biphasic TiO2 nanoparticles decorated graphene nanosheets for visible light driven photocatalytic degradation of organic dyes. Appl. Surf. Sci. 2017, 31. [Google Scholar] [CrossRef]
- Verma, R.; Samdarshi, S.K.; Sagar, K. Nanostructured bi-phasic TiO2 nanoparticles grown on reduced graphene oxide with high visible light photocatalytic detoxification. Mater. Chem. Phys. 2017, 186, 202–211. [Google Scholar] [CrossRef]
- Atchudan, R.; Jebakumar Immanuel Edison, T.N.; Perumal, S.; Karthikeyan, D.; Lee, Y.R. Effective photocatalytic degradation of anthropogenic dyes using graphene oxide grafting titanium dioxide nanoparticles under UV-light irradiation. J. Photochem. Photobiol. A Chem. 2017, 333, 92–104. [Google Scholar] [CrossRef]
- Darvishi, M.; Seyed-Yazdi, J. Characterization and comparison of photocatalytic activities of prepared TiO2/graphene nanocomposites using titanium butoxide and TiO2 via microwave irradiation method. Mater. Res. Express 2016, 3. [Google Scholar] [CrossRef]
- Rezaei, M.; Salem, S. Photocatalytic activity enhancement of anatase-graphene nanocomposite for methylene removal: Degradation and kinetics. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2016, 167, 41–49. [Google Scholar] [CrossRef] [PubMed]
- Rezaei, M.; Salem, S. Optimal TiO2-graphene oxide nanocomposite for photocatalytic activity under sunlight condition: Synthesis, characterization, and kinetics. Int. J. Chem. Kinet. 2016, 48, 573–583. [Google Scholar] [CrossRef]
- Charoensuk, J.; Charupongtawitch, R.; Usakulwattana, A. Kinetics of photocatalytic degradation of methylene blue by TiO2-graphene nanocomposites. J. Nanosci. Nanotechnol. 2016, 16, 296–302. [Google Scholar]
- Wang, R.; Yang, R.; Wang, B. Efficient degradation of methylene blue by the nano TiO2-functionalized graphene oxide nanocomposite photocatalyst for wastewater treatment. Water Air Soil Pollut. 2016, 227. [Google Scholar] [CrossRef]
- Liu, G.; Wang, R.; Liu, H.; Han, K.; Cui, H. Highly dispersive nano-TiO2 in situ growing on functional graphene with high photocatalytic activity. J. Nanopart. Res. 2016, 18, 1–8. [Google Scholar] [CrossRef]
- Baldissarelli, V.Z.; De Souza, T.; Andrade, L.; Oliveira, L.F.C.D.; José, H.J. Preparation and photocatalytic activity of TiO2-exfoliated graphite oxide composite using an ecofriendly graphite oxidation method. Appl. Surf. Sci. 2015, 359, 868–874. [Google Scholar] [CrossRef]
- Gu, L.; Zhang, H.; Jiao, Z.; Li, M.; Wu, M.; Lei, Y. Glucosamine-induced growth of highly distributed TiO2 nanoparticles on graphene nanosheets as high-performance photocatalysts. RSC Adv. 2016, 6, 67039–67048. [Google Scholar] [CrossRef]
- Fan, J.; Liu, E.; Hu, X.; Ma, Y.; Fan, X.; Li, Y.; Tang, C. Facile hydrothermal synthesis of TiO2 nanospindles-reduced graphene oxide composite with a enhanced photocatalytic activity. J. Alloys Compd. 2015, 623, 298–303. [Google Scholar]
- Sun, M.; Li, W.; Sun, S.; He, J.; Zhang, Q.; Shi, Y. One-step in situ synthesis of graphene-TiO2 nanorod hybrid composites with enhanced photocatalytic activity. Mater. Res. Bull. 2015, 61, 280–286. [Google Scholar] [CrossRef]
- Rong, X.; Qiu, F.; Zhang, C.; Fu, L.; Wang, Y.; Yang, D. Preparation, characterization and photocatalytic application of TiO2-graphene photocatalyst under visible light irradiation. Ceram. Int. 2015, 41, 2502–2511. [Google Scholar] [CrossRef]
- Gupta, B.K.; Kedawat, G.; Agrawal, Y.; Kumar, P.; Dwivedi, J. A novel strategy to enhance ultraviolet light driven photocatalysis from graphene quantum dots infilled TiO2 nanotube arrays. RSC Adv. 2015, 5, 10623–10631. [Google Scholar] [CrossRef]
- Sha, J.; Zhao, N.; Liu, E.; Shi, C.; He, C.; Li, J. In situ synthesis of ultrathin 2-D TiO2 with high energy facets on graphene oxide for enhancing photocatalytic activity. Carbon 2014, 68, 352–359. [Google Scholar] [CrossRef]
- Yang, Y.; Xu, L.; Wang, H.; Wang, W.; Zhang, L. TiO2/graphene porous composite and its photocatalytic degradation of methylene blue. Mater. Des. 2016, 108, 632–639. [Google Scholar] [CrossRef]
- Suave, J.; Amorim, S.M.; Moreira, R.F.P.M. TiO2-graphene nanocomposite supported on floating autoclaved cellular concrete for photocatalytic removal of organic compounds. J. Environ. Chem. Eng. 2017, 5, 3215–3223. [Google Scholar] [CrossRef]
- Ai, L.; Zhang, C.; Meng, L. Adsorption of methyl orange from aqueous solution on hydrothermal synthesized Mg–Al layered double hydroxide. J. Chem. Eng. Data 2011, 56, 4217–4225. [Google Scholar] [CrossRef]
- Lavanya, T.; Dutta, M.; Ramaprabhu, S.; Satheesh, K. Superior photocatalytic performance of graphene wrapped anatase/rutile mixed phase TiO2 nanofibers synthesized by a simple and facile route. J. Environ. Chem. Eng. 2017, 5, 494–503. [Google Scholar] [CrossRef]
- Hou, X.; Sun, T.; Zhao, X. Calcination of reduced graphene oxide decorated TiO2 composites for recovery and reuse in photocatalytic applications. Ceram. Int. 2017, 43, 1150–1159. [Google Scholar]
- Zhao, F.; Dong, B.; Gao, R.; Su, G.; Liu, W.; Shi, L.; Xia, C. A three-dimensional graphene-TiO2 nanotube nanocomposite with exceptional photocatalytic activity for dye degradation. Appl. Surf. Sci. 2015, 351, 303–308. [Google Scholar] [CrossRef]
- Lu, Z.; Chen, G.; Hao, W.; Sun, G.; Li, Z. Mechanism of UV-assisted TiO2/reduced graphene oxide composites with variable photodegradation of methyl orange. RSC Adv. 2015, 5, 72916–72922. [Google Scholar] [CrossRef]
- Xia, H.Y.; He, G.Q.; Min, Y.L.; Liu, T. Role of the crystallite phase of TiO2 in graphene/TiO2 photocatalysis. J. Mater. Sci. Mater. Electron. 2015, 26, 3357–3363. [Google Scholar] [CrossRef]
- Jiang, Y.; Wang, W.-N.; Biswas, P.; Fortner, J.D. Facile aerosol synthesis and characterization of ternary crumpled graphene-TiO2-magnetite nanocomposites for advanced watertreatment. ACS Appl. Mater. Interfaces 2014, 6, 11766–11774. [Google Scholar] [CrossRef] [PubMed]
- Han, W.; Ren, L.; Zhang, Z.; Qi, X.; Liu, Y.; Huang, Z.; Zhong, J. Graphene-supported flocculent-like TiO2 nanostructures for enhanced photoelectrochemical activity and photodegradation performance. J. Ceram. Int. 2015, 41, 7471–7477. [Google Scholar] [CrossRef]
- Lavanya, T.; Satheesh, K.; Dutta, M.; Victor Jaya, N. Superior photocatalytic performance of reduced graphene oxide wrapped electrospun anatase mesoporous TiO2 nanofibers. J. Alloys Compd. 2014, 615, 643–650. [Google Scholar] [CrossRef]
- Wang, Y.; Li, Z.; He, Y.; Li, F.; Liu, X. Low-temperature solvothermal synthesis of graphene-TiO2 nanocomposite and its photocatalytic activity for dye degradation. Mater. Lett. 2014, 134, 115–118. [Google Scholar] [CrossRef]
- Athanasekou, C.P.; Morales-Torres, S.; Likodimos, V.; Romanos, G.E.; Pastrana-Martinez, L.M.; Falaras, P.; Faria, J.L.; Figueiredo, J.L. Prototype composite membranes of partially reduced graphene oxide/TiO2 for photocatalytic ultrafiltration water treatment under visible light. Appl. Catal. B Environ. 2014, 158–159, 361–372. [Google Scholar] [CrossRef]
- Liu, Y. Hydrothermal synthesis of TiO2-RGO composites and their improved photocatalytic activity in visible light. RSC Adv. 2014, 4, 36040–36045. [Google Scholar] [CrossRef]
- Shakir, K.; Elkafrawy, A.F.; Ghoneimy, H.F.; Elrab Beheir, S.G.; Refaat, M. Removal of rhodamine B (a basic dye) and thoron (an acidic dye) from dilute aqueous solutions and wastewater simulants by ion flotation. Water Res. 2010, 44, 1449–1461. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.; Zhou, M.; Zhang, Z.; Tang, T.; Wang, T. Preparation of TiO2 nanotubes/reduced graphene oxide binary nanocomposites enhanced photocatalytic properties. J. Mater. Sci. Mater. Electron. 2017, 28, 9416–9422. [Google Scholar] [CrossRef]
- Chen, Y.; Dong, X.; Cao, Y.; Xiang, J. Enhanced photocatalytic activities of low-bandgap TiO2-reduced graphene oxide nanocomposites. J. Nanopart. Res. 2017, 19. [Google Scholar] [CrossRef]
- Wang, W.; Huang, X.; Lai, M. RGO/TiO2 nanosheets immobilized on magnetically actuated artificial cilia film: A new mode for efficient photocatalytic reaction. RSC Adv. 2017, 7, 10517–10523. [Google Scholar] [CrossRef]
- Biris, A.R.; Toloman, D.; Popa, A.; Lazar, M.D.; Kannarpady, G.K.; Saini, V.; Watanabe, F.; Chhetri, B.P.; Ghosh, A. Synthesis of tunable core-shell nanostructures based on TiO2-graphene architectures and their application in the photodegradation of rhodamine dyes. Phys. E Low Dimens. Syst. Nanostruct. 2016, 81, 326–333. [Google Scholar] [CrossRef]
- Kim, T.-W.; Park, M.; Kim, H.Y.; Park, S.-J. Preparation of flower-like TiO2 sphere/reduced graphene oxide composites for photocatalytic degradation of organic pollutants. J. Solid State Chem. 2016, 239, 91–98. [Google Scholar] [CrossRef]
- Liu, H.; Wang, Y.; Shi, L.; Xu, R.; Huang, L.; Tan, S. Utilization of reduced graphene oxide for the enhancement of photocatalytic property of TiO2 nanotube. Desalination Water Treat. 2016, 57, 13263–13272. [Google Scholar] [CrossRef]
- Sedghi, R.; Heidari, F. A novel & effective visible light-driven TiO2/magnetic porous graphene oxide nanocomposite for the degradation of dye pollutants. RSC Adv. 2016, 6, 49459–49468. [Google Scholar]
- Zhang, J.-J.; Wu, Y.-H.; Mei, J.-Y.; Zheng, G.-P.; Yan, T.-T.; Zheng, X.-C.; Liu, P.; Guan, X.-X. Synergetic adsorption and photocatalytic degradation of pollutants over 3D TiO2-graphene aerogel composites synthesized: Via a facile one-pot route. Photochem. Photobiol. Sci. 2016, 15, 1012–1019. [Google Scholar] [CrossRef] [PubMed]
- He, R.; He, W. Ultrasonic assisted synthesis of TiO2-reduced graphene oxide nanocomposites with superior photovoltaic and photocatalytic activities. Ceram. Int. 2016, 42, 5766–5771. [Google Scholar] [CrossRef]
- Fang, R.; Liang, Y.; Ge, X.; Du, M.; Li, S.; Li, T. Preparation and photocatalytic degradation activity of TiO2/rGO/polymer composites. Colloid Polym. Sci. 2015, 293, 1151–1157. [Google Scholar] [CrossRef]
- Liang, D.; Cui, C.; Hub, H.; Wang, Y.; Xu, S.; Ying, B.; Li, P.; Lu, B. One-step hydrothermal synthesis of anatase TiO2/reduced graphene oxide nanocomposites with enhanced photocatalytic activity. J. Alloys Compd. 2014, 582, 236–240. [Google Scholar] [CrossRef]
- Li, Q.; Bian, J.; Zhang, L.; Zhang, R.; Wang, G. Synthesis of carbon materials-TiO2 hybrid nanostructures and their visible-light photo-catalytic activity. Chem Plus Chem 2014, 79, 454–461. [Google Scholar]
- Rajoriya, S.; Bargole, S.; Saharan, V.K. Degradation of a cationic dye (Rhodamine 6G) using hydrodynamic cavitation coupled with other oxidative agents: Reaction mechanism and pathway. Ultrason. Sonochem. 2017, 34, 183–194. [Google Scholar] [CrossRef] [PubMed]
- Dai, B.; Tao, H.; Lin, Y.-J.; Chang, C.-T. Study of various nanostructures Titania with graphene composites: The preparation and photocatalytic activities. Nano 2016, 11. [Google Scholar] [CrossRef]
- Liang, X.; Tao, H.; Zhang, Q. High performance photocatalytic degradation by graphene/titanium nanotubes under near visible light with low energy irradiation. J. Nanosci. Nanotechnol. 2015, 15, 4887–4894. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Liang, X.; Chen, B.-Y.; Chang, C. Deciphering effects of functional groups and electron density on azodyes degradation by graphene loaded TiO2. Appl. Surf. Sci. 2015, 357, 1064–1071. [Google Scholar] [CrossRef]
- Posa, V.R.; Annavaram, V.; Somala, A.R. Fabrication of graphene-TiO2 nanocomposite with improved photocatalytic degradation for acid orange 7 dye under solar light irradiation. Bull. Mater. Sci. 2016, 39, 759–767. [Google Scholar] [CrossRef]
- Gao, P.; Li, A.; Sun, D.D.; Ng, W.J. Effects of various TiO2 nanostructures and graphene oxide on photocatalytic activity of TiO2. J. Hazard. Mater. 2014, 279, 96–104. [Google Scholar] [CrossRef] [PubMed]
- Muthirulan, P.; Devi, C.N.; Sundaram, M.M. TiO2 wrapped graphene as a high performance photocatalyst for acid orange 7 dye degradation under solar/UV light irradiations. Ceram. Int. 2014, 40, 5945–5957. [Google Scholar] [CrossRef]

| Results | Highlight | Refs. | |
|---|---|---|---|
| Epitaxial growth |
| Experimental and theoretical aspect | [71] |
| Enhancements in the electronic properties of G-based devices | [72] | |
| Influence of the substrate step height on the energy barrier | [73] | |
| Chemical vapor deposition (CVD) |
| Raman spectra measured as a function of the rotation angle | [74] |
| Review: challenges and future perspective | [75] | |
| Representative applications | [76] | |
| Absence of metallic contaminations | [77] | |
| Educational experiments: economical, safe, and simple technique in 30–45 min | [78] | |
| Two methods to shape and transfer films to specific substrates | [79] | |
| Ex-situ graphitization in argon atmosphere | [80] | |
| Efficient roll-to-roll process | [81] | |
| N-type behavior useful to modulate G electrical properties | [82] | |
| Electrochemical reduction of CO and CO2 |
| Direct reaction of CO2 with Mg metal | [83] |
| Room-temperature synthesis method on copper foil from different carbon sources using external charges | [84] | |
| Arc discharge |
| The reactivity of buffer gases (helium, oxygen-helium, and hydrogen-helium) is the key factor | [85] |
| Different mechanisms in the presence and absence of TiO2 and ZnO catalysts | [86] | |
| Unzipping carbon-nanotubes (CNT) |
| Radial and shear loading unzipping modes with cryomill method at 150 K | [70] |
| Linear longitudinal opening of the Multi Wall CNT (MWCNT) | [87] | |
| Organic synthesis |
| Highly ordered monolayers (2D crystal) of larger G ribbons | [88] |
| Pyrolysis |
| Pyrolysis of organic matter | [89] |
| Spray pyrolysis of iron carbonyl and pyridine | [90] | |
| Spray pyrolysis at different temperatures, graphene oxide-based precursor, nitrogen carrier gas | [91] |
| Results | Highlight | Refs. | |
|---|---|---|---|
| Mechanical |
| Exfoliation of small mesas of highly oriented pyrolytic graphite | [42] |
| Review: general description | [94] | |
| Dispersion and exfoliation of graphite in N-methyl-pyrrolidone | [95] | |
| Exfoliation–reintercalation–expansion of graphite | [96] | |
| Thin films on a low-density polyethylene substrate | [97] | |
| Electrochemical expansion |
| Hyperexpanded graphite by electrolysis in a Li+ containing electrolyte and in situ electrochemical diazonium functionalization | [98] |
| Cathodic graphite expansion in dimethylformamide (DMF) and functionalization by reducing aryl diazonium salts in organic solution | [99] | |
| Exfoliation temperature increase from 25 to 95 °C reuslt in decrease of defects and increase of thermal stability (with H2O2 addition) | [100] | |
| Chemical synthesis |
| Review: general description | [103] |
| Low-temperature, catalyst-free graphite intercalation with alkali metals | [104] | |
| From colloidal suspensions | [105] | |
| Unzipping nanotube |
| Mechanical sonication and gas-phase oxidation in organic solvent of multiwalled carbon nanotubes | [106] |
| Lengthwise cutting of MWCNTs by a solution-based oxidative process | [107] | |
| Plasma etching of CNT partly embedded in a polymer film | [108] | |
| Microwave synthesis |
| Microwave G growth on polymethyl methacrylate (PMMA)-coated h-BN flakes | [109] [110] |
| Chemical and/or thermal graphene oxide (GO) reduction |
| Glucose and polhyvinilpyrrolidone (PVP) reduction of GO mixed with PLA | [111] |
| Reduction |
| Two different reducing mixed reagents: HI/NH and NH/HI. Review: 50 types of reducing agents | [114] [115] |
| Other |
| Review: different sizes and chemical compositions. Controlled segregation of G chemically modified on liquid interfaces | [116] [117] |
| Ex-Situ Hybridization | ||||
| Photocatalyst | Synthetic Route | Starting Graphite | Starting Semiconductor | Refs. |
| rGO/TiO2 | Mechanical mixing and ultrasonication | rGO | TiO2NPs in 1-butyl alcohol | [163] |
| G/TiO2 | Mechanical mixing and ultrasonication | G in PVP/water | TiO2NPs anatase in water | [164] |
| GO/TiO2 | Mechanical mixing | GO | Titanium isopropoxide | [165] |
| In-Situ Crystallization | ||||
| rGO/TiO2 | M with GO and PR reduction | GO | TiO2NPs | [174] |
| M and GO and ST reduction | GO | TiO2NPs | [175] | |
| G/TiO2 | ST | GO | Tetrabutyl titanate | [145] |
| G/TiO2 | SG | rGO | Titanium isopropoxide | [146] |
| G/TiO2 | HD | GO | P25 | [147] |
| rGO/TiO2 | SG | GO | Tetrabutyl titanate | [176] |
| G/TiO2 | SG | G dispersion | Titanium isopropoxide | [177] |
| rGO/TiO2 | Solvothermal SG | GO sheets | TiO2 sol | [178] |
| rGO/TiO2 | SG | GO | Titanium isopropoxide | [179] |
| G/TiO2 | HD | GO suspension | TiO2 sol | [180] |
| G/TiO2 | one-step HD | GO water/ethanol | TiO2 | [181] |
| G/TiO2 | HD | GO | Different Ti sources | [182] |
| G/TiO2 | Wet impregnation and thermal reduction (H2) | GO | TiO2 nanotubes from HD of TiO2 P25 | [183] |
| rGO superlong TiO2 | HD and heating reflux | GO | Super long TiO2 | [184] |
| GO and rGO/TiO2 (nanocristals) | Elevated pressure HD | GO and rGO | TiO2 | [185] |
| rGO/TiO2 sandwich-like structure | HD/ST | rGO | Butyl titanate | [186] |
| nanocrystalline anatase TiO2-GO | SG and GO decoration by ST | GO | Anatase TiO2NPs by SG | [187] |
| TiO2/rGO-X nanocomposites | One-step surfactant (X)-assisted HD | GO | TiO2 (P25) | [188] |
| rGO films on TiO2 nanotubes | Two-step ED and carbonation techniques | rGO | Two-step anodized TiO2 nanotubes from Ti foils | [189] |
| Ti plate deposited TiO2 and GO film | ED | GO from nanographite | Ti plate and K2TiF6 aqueous solution | [190] |
| G/TiO2 | MW | GO | TiCl4 | [191] |
| rGO/TiO2 | MW | GO | Ti powder | [192] |
| rGO/TiO2 | MW | GO | TiO2NPs | [193] |
| rGO/TiO2 | PR | GO | TiO2NPs | [194] |
| rGO/TiO2 | PR | GO | Colloidal TiO2 | [156] |
| Photoactive Nanomaterials | Dye Conc. (mg/L) | Catalyst Quantity (g/L) | Light Source | Irradiation Time (min) | Degradation (%) | Refs. |
|---|---|---|---|---|---|---|
| G/TiO2 | 9.60 | 0.33 | Visible | 180 | 90 | [8] |
| P25-G | 8.64 | 0.6 | UV | 60 | 85 | [199] |
| P25-G | 8.64 | 0.6 | Visible | 60 | 65 | [199] |
| G/TiO2 | 10 | 0.6 | UV | 120 | 100 | [200] |
| G/TiO2 | 10 | 0.2 | UV | 40 | 85 | [202] |
| TiO2@rGO | -- | 0.1 | UV | 120 | 92 | [203] |
| GO/TiO2 | 15 | 0.2 | UV | 350 | 92 | [204] |
| TiO2-G | 0.13 | 0.5 | UV-Vis | 450 | 100 | [205] |
| rGO/TiO2 | 320 | 0.5 | Visible | 90 | 95 | [207] |
| TiO2/GO | -- | 0.2 | UV | 25 | 100 | [208] |
| TiO2/G | 0.13 | 0.4 | UV | 60 | 96 | [209] |
| TiO2/G | 3 | 0.48 | Visible | 90 | 100 | [210] |
| G/TiO2 | 3.2 | 0.2 | UV | 180 | 97 | [191] |
| G/TiO2 | 3.2 | 0.2 | Visible | 240 | 96 | [191] |
| TiO2/GO | 3 | 0.48 | Visible | 60 | 94 | [211] |
| TiO2/GO | 5 | 0.1 | UV | 40 | 93 | [213] |
| TiO2/GO | 5 | 0.1 | Visible | 40 | 70 | [213] |
| TiO2-Graphite Oxide | 10 | 0.5 | UV | 60 | 100 | [215] |
| TiO2/G | 10 | 0.2 | UV | 20 | 97 | [216] |
| TiO2/rGO | 10 | 0.17 | UV | 60 | 100 | [217] |
| G/TiO2 | 5 | 0.5 | Visible | 100 | 70 | [218] |
| TiO2/G | 10 | 0.8 | Visible | 100 | 98.8 | [219] |
| Graphene quantum dots/TiO2 | 6.4 | -- | UV | 180 | 100 | [220] |
| TiO2/GO | 10 | 0.1 | UV | 180 | 100 | [221] |
| TiO2/G | 10 | 0.01 | Visible | 150 | 100 | [222] |
| TiO2/G | 10 | 0.5 | UV | 90 | 100 | [223] |
| Graphene quantum dots/TiO2 | 6.4 | -- | UV | 180 | 100 | [220] |
| Photoactive Nanomaterials | Dye Conc. (mg/L) | Catalyst Quantity (g/L) | Light Source | Irradiation Time (min) | Degradation (%) | Refs. |
|---|---|---|---|---|---|---|
| rGO/TiO2 (mix anatase/rutile nanofibers) | 10 | 0.4 | UV | 120 | 97 | [225] |
| G-pasted TiO2 spheres | 12 | 0.5 | UV | 75 | 95 | [63] |
| GO/TiO2 | 13 | 1 | UV | 60 | 88 | [226] |
| 1 | Vis | 60 | 80 | |||
| rGO/TiO2 | 1 | UV | 60 | 70 | ||
| 1 | Vis | 60 | 99 | |||
| rGO/TiO2 anotube | 20 | 0.25 | UV | 210 | 100 | [227] |
| rGO/TiO2 | 10 | 0.5 | UV | 75 | 70 | [228] |
| rGO/TiO2 mix anatasio/rutilio | 6.55 | 0.6 | UV | 100 | 100 | [229] |
| Vis | 100 | 50 | ||||
| G/TiO2/Magnetite | 9.6 | 0.16 | UV | 90 | 99 | [230] |
| Flocculent likeTiO2/G | 20 | 0.8 | UV | 60 | 70 | [231] |
| rGO/TiO2 nanofibers | 15 | 0.4 | UV | 120 | 100 | [232] |
| G/TiO2 | 10 | 0.6 | UV | 60 | 80 | [233] |
| TiO2/rGO | 10 | 0.5 | Vis | 240 | 90 | [235] |
| Photoactive Nanomaterials | Dye Conc. (mg/L) | Catalyst Quantity (g/L) | Light Source | Irradiation Time (min) | Degradation (%) | Refs. |
|---|---|---|---|---|---|---|
| TiO2/rGO | 30 | 0.2 | Vis | 40 | 100 | [238] |
| rGO/TiO2 nanosheets onmagnetically cilia film rGO/TiO2-Au | 20 | 0.2 | UV | 180 | 83 100 | [239] |
| Core-shell TiO2/G | 4.79 | 0.6 | UV | 270 | 100 | [240] |
| Flower-like TiO2 sphere /rGO | 15 | 0.4 | Simulated solar | 120 | 100 | [241] |
| TiO2 nanotubes/rGO | 10 | 0.5 | UV | 20 | 100 | [242] |
| TiO2/magnetic porous GO | 10 | 0. 1 | Vis | 20 | 100 | [243] |
| 3D TiO2/G aerogel | 20 | 0.2 | Visible light | 180 | 99 | [244] |
| TiO2/rGO | 4.79 | 0.4 | Visible light | 180 | 100 | [245] |
| TiO2/rGO/polymer | 8 | 0.2 | Visible light | 30 | 96 | [246] |
| TiO2/rGO | 20 | 0.5 | UV | 30 | 100 | [247] |
| GTiO2 | 5 | 0.5 | Visible light | 60 | 80 | [248] |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Giovannetti, R.; Rommozzi, E.; Zannotti, M.; D’Amato, C.A. Recent Advances in Graphene Based TiO2 Nanocomposites (GTiO2Ns) for Photocatalytic Degradation of Synthetic Dyes. Catalysts 2017, 7, 305. https://doi.org/10.3390/catal7100305
Giovannetti R, Rommozzi E, Zannotti M, D’Amato CA. Recent Advances in Graphene Based TiO2 Nanocomposites (GTiO2Ns) for Photocatalytic Degradation of Synthetic Dyes. Catalysts. 2017; 7(10):305. https://doi.org/10.3390/catal7100305
Chicago/Turabian StyleGiovannetti, Rita, Elena Rommozzi, Marco Zannotti, and Chiara Anna D’Amato. 2017. "Recent Advances in Graphene Based TiO2 Nanocomposites (GTiO2Ns) for Photocatalytic Degradation of Synthetic Dyes" Catalysts 7, no. 10: 305. https://doi.org/10.3390/catal7100305
APA StyleGiovannetti, R., Rommozzi, E., Zannotti, M., & D’Amato, C. A. (2017). Recent Advances in Graphene Based TiO2 Nanocomposites (GTiO2Ns) for Photocatalytic Degradation of Synthetic Dyes. Catalysts, 7(10), 305. https://doi.org/10.3390/catal7100305









