CO-Tolerant Pt–BeO as a Novel Anode Electrocatalyst in Proton Exchange Membrane Fuel Cells
Abstract
:1. Introduction
2. Results and Discussion
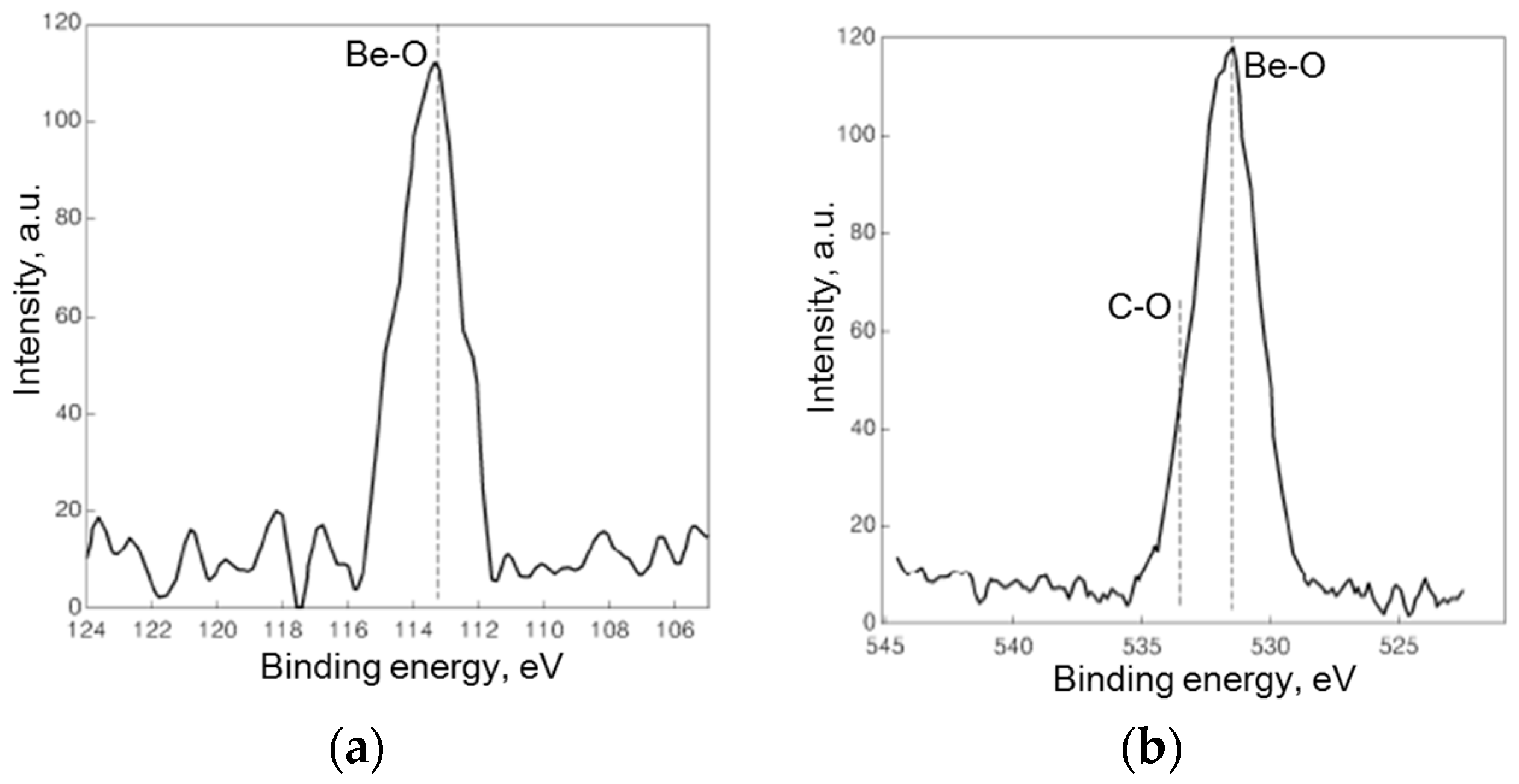
2.1. Physicochemical Properties of Carbon-Supported Pt–BeO
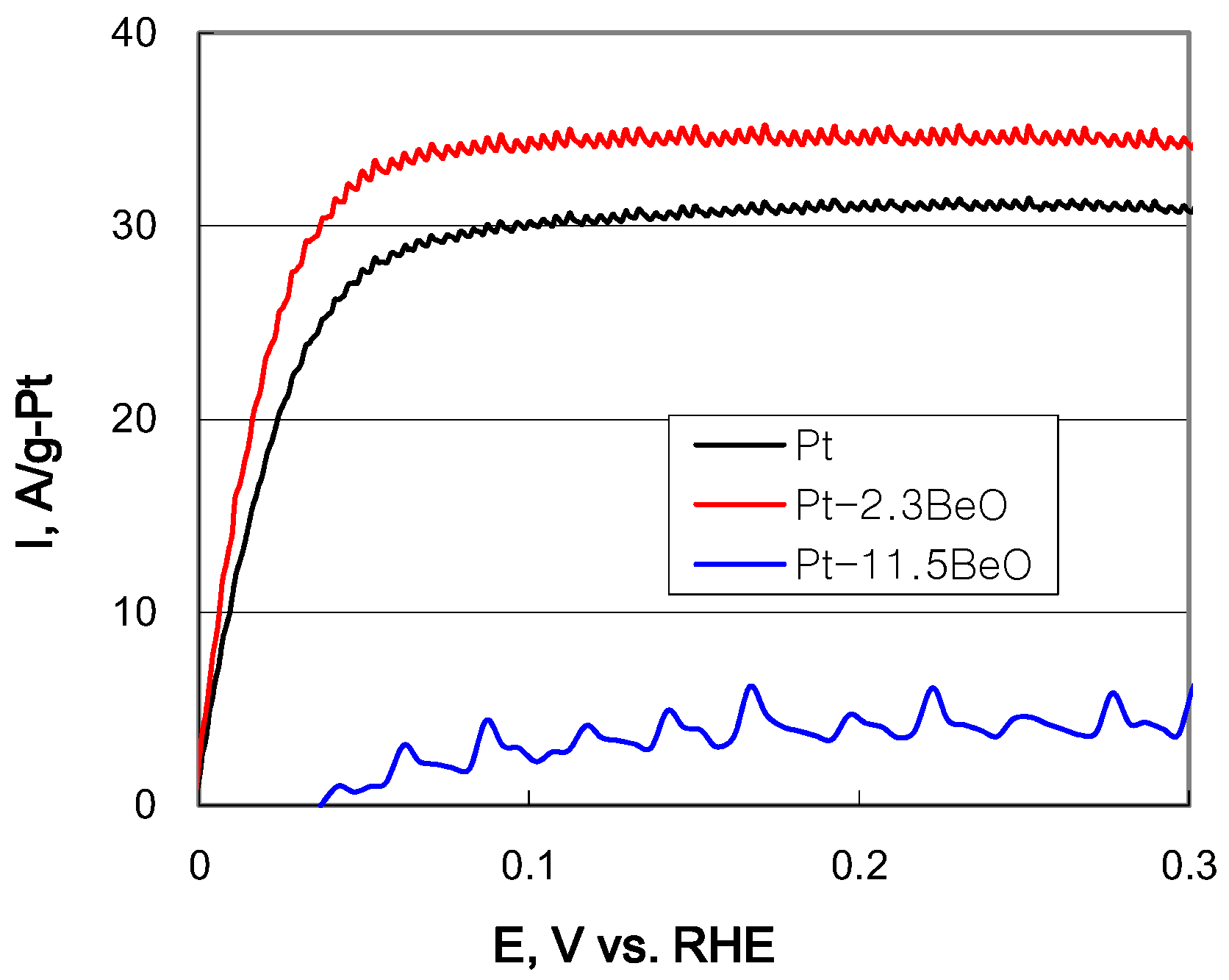
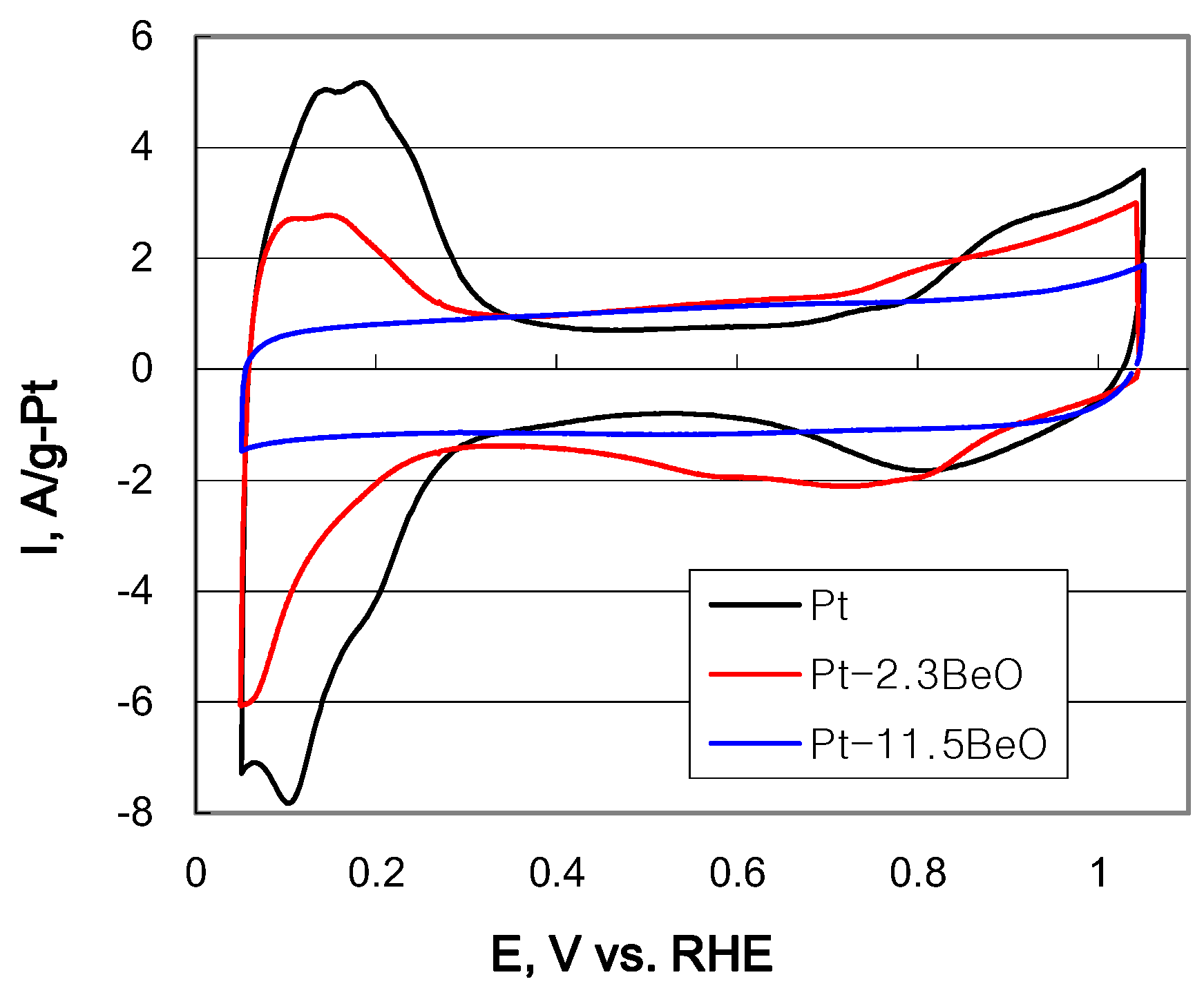
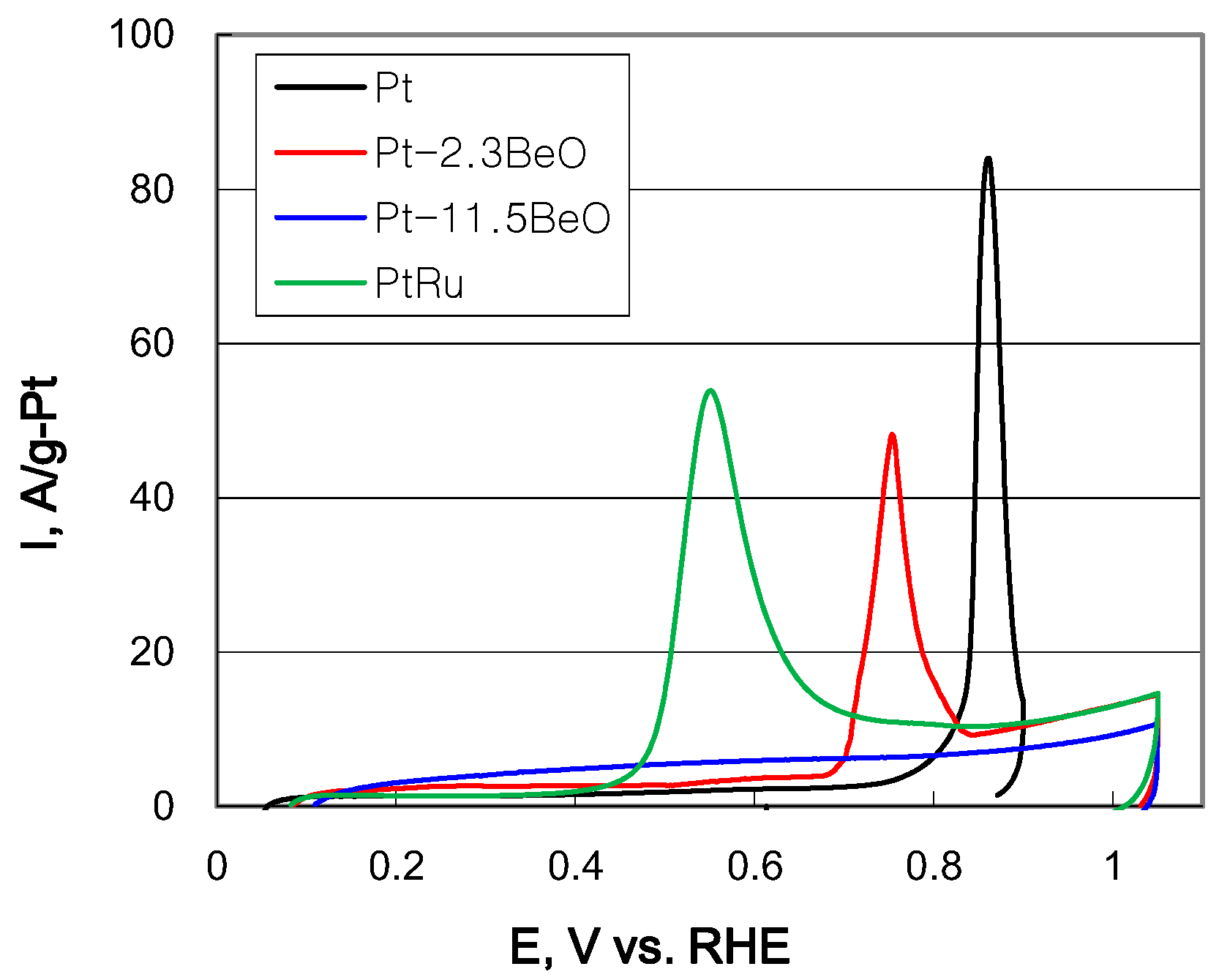
2.2. Electrochemical Properties of Carbon-Supported Pt–BeO
3. Materials and Methods
3.1. Preparation of Catalysts
3.2. Electrochemical and Physicochemical Characterization
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| PEMFC | Proton exchange membrane fuel cell |
| HOR | Hydrogen oxidation reduction |
| RDE | Rotating disk electrode |
| RHE | Reversible hydrogen electrode |
References
- Springer, T.E.; Rockward, T.; Zawodzinski, T.A.; Gottesfeld, S. Model for polymer electrolyte fuel cell operation on reformate feed effects of CO, H2 dilution, and high fuel utilization. J. Electrochem. Soc. 2001, 148, A11–A23. [Google Scholar] [CrossRef]
- Kwon, K.; Yoo, D.Y.; Park, J.O. Experimental factors that influence carbon monoxide tolerance of high-temperature proton-exchange membrane fuel cells. J. Power Sources 2008, 185, 202–206. [Google Scholar] [CrossRef]
- Gasteiger, H.A.; Markovic, N.; Ross, P.N.; Cairns, E.J. CO electrooxidation on well-characterized Pt-Ru alloys. J. Phys. Chem. 1994, 98, 617–625. [Google Scholar] [CrossRef]
- Schmidt, T.J.; Jusys, Z.; Gasteiger, H.A.; Behm, R.J.; Endruschat, U.; Boennemann, H. On the CO tolerance of novel colloidal PdAu/carbon electrocatalysts. J. Electroanal. Chem. 2001, 501, 132–140. [Google Scholar] [CrossRef]
- Papageorgopoulos, D.C.; Keijzer, M.; Veldhuis, J.B.J.; de Brujin, F.A. CO tolerance of Pd-rich platinum palladium carbon-supported electrocatalysts. J. Electrochem. Soc. 2002, 149, A1400–A1404. [Google Scholar] [CrossRef]
- Hung, W.-Z.; Chung, W.-H.; Tsai, D.-S.; Wilkinson, D.P.; Huang, Y.-S. CO tolerance and catalytic activity of Pt/Sn/SnO2 nanowires loaded on a carbon paper. Electrochim. Acta 2010, 55, 2116–2122. [Google Scholar] [CrossRef]
- Sims, C.M.; Ponce, A.A.; Gaskell, K.J.; Eichhorn, B.W. CO tolerance of Pt and PtSn intermetallic electrocatalysts on synthetically modified reduced graphene oxide supports. Dalton Trans. 2015, 44, 977–987. [Google Scholar] [CrossRef] [PubMed]
- Freitas, K.S.; Lopes, P.P.; Ticianelli, E.A. Electrocatalysis of the hydrogen oxidation in the presence of CO on RhO2/C-supported Pt nanoparticles. Electrochim. Acta 2010, 56, 418–426. [Google Scholar] [CrossRef]
- Takimoto, D.; Ohnishi, T.; Sugimoto, W. Suppression of CO adsorption on PtRu/C and Pt/C with RuO2 nanosheets. ECS Electrochem. Lett. 2015, 4, F35–F37. [Google Scholar] [CrossRef]
- Pereira, L.G.S.; dos Santos, F.R.; Pereira, M.E.; Paganin, V.A.; Ticianelli, E.A. CO tolerance effects of tungsten-based PEMFC anodes. Electrochim. Acta 2006, 51, 4061–4066. [Google Scholar] [CrossRef]
- Micoud, F.; Maillard, F.; Gourgaud, A.; Chatenet, M. Unique CO tolerance of Pt-WOx materials. Electrochem. Commun. 2009, 11, 651–654. [Google Scholar] [CrossRef]
- Cui, X.; Guo, L.; Cui, F.; He, Q.; Shi, J. Electrocatalytic activity and CO tolerance properties of mesostructured Pt/WO3 composite as an anode catalyst for PEMFCs. J. Phys. Chem. C 2009, 113, 4134–4138. [Google Scholar] [CrossRef]
- Muhamad, E.N.; Takeguchi, T.; Wang, F.; Wang, G.; Yamanaka, T.; Ueda, W. A comparative study of variously prepared carbon-supported Pt/MoOx anode catalysts for a polymer electrolyte fuel cell. J. Electrochem. Soc. 2009, 156, B1361–B1368. [Google Scholar] [CrossRef]
- Elezovic, N.R.; Gajic-Krstajic, L.M.; Vracar, L.M.; Krstajic, N.V. Effect of chemisorbed CO on MoOx-Pt/C electrode on the kinetics of hydrogen oxidation reaction. Int. J. Hydrogen Energy 2010, 35, 12878–12887. [Google Scholar] [CrossRef]
- Lin, R.; Cao, C.; Zhang, H.; Huang, H.; Ma, J. Electro-catalytic activity of enhanced CO-tolerant cerium-promoted Pt/C catalyst for PEM fuel cell anode. Int. J. Hydrogen Energy 2012, 37, 4648–4656. [Google Scholar] [CrossRef]
- Kolla, P.; Smirnova, A. Methanol oxidation on hybrid catalysts: PtRu/C nanostructures promoted with cerium and titanium oxides. Int. J. Hydrogen Energy 2013, 38, 15152–15159. [Google Scholar] [CrossRef]
- Lim, K.R.; Kim, C.E.; Yun, Y.S.; Kim, W.T.; Soon, A.; Kim, D.H. Remarkably stable amorphous metal oxide grown on Zr-Cu-Be metallic glass. Sci. Rep. 2015, 5, 18196. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, T.J.; Gasteiger, H.A.; Behm, R.J. Rotating disk electrode measurements on the CO tolerance of a high-surface area Pt/Vulcan carbon fuel cell catalyst. J. Electrochem. Soc. 1999, 146, 1296–1304. [Google Scholar] [CrossRef]
- Hwang, D.-Y.; Mebel, A.M. Conversion of CO to formaldehyde catalyzed by BeO: A theoretical study. J. Phys. Chem. A 2001, 105, 10433–10438. [Google Scholar] [CrossRef]
- Stoffelsma, C.; Rodriguez, P.; Garcia, G.; Garcia-Araez, N.; Strmcnik, D.; Markovic, N.M.; Koper, M.T.M. Promotion of the oxidation of carbon monoxide at stepped platinum single-crystal electrodes in alkaline media by lithium and beryllium cations. J. Am. Chem. Soc. 2010, 132, 16127–16133. [Google Scholar] [CrossRef] [PubMed]
- Sa, Y.J.; Kwon, K.; Cheon, J.Y.; Kleitz, F.; Joo, S.H. Ordered mesoporous Co3O4 spinels as stable, bifunctional, noble metal-free oxygen electrocatalysts. J. Mater. Chem. A 2013, 1, 9992–10001. [Google Scholar] [CrossRef]
- Kwon, K.; Jin, S.; Lee, K.H.; You, D.J.; Pak, C. Performance enhancement of Pd-based hydrogen oxidation catalysts using tungsten oxide. Catal. Today 2014, 235, 175–178. [Google Scholar] [CrossRef]

| Catalyst | BET SSA (m2/g) | ESA (m2/g) 1 |
|---|---|---|
| Pt/C | 180 | 91 |
| Pt-2.3BeO/C | 79 | 39 |
| Pt-11.5BeO/C | 74 | 0 |
| PtRu/C 2 | 215 | - |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kwon, K.; Jung, Y.; Ku, H.; Lee, K.H.; Kim, S.; Sohn, J.; Pak, C. CO-Tolerant Pt–BeO as a Novel Anode Electrocatalyst in Proton Exchange Membrane Fuel Cells. Catalysts 2016, 6, 68. https://doi.org/10.3390/catal6050068
Kwon K, Jung Y, Ku H, Lee KH, Kim S, Sohn J, Pak C. CO-Tolerant Pt–BeO as a Novel Anode Electrocatalyst in Proton Exchange Membrane Fuel Cells. Catalysts. 2016; 6(5):68. https://doi.org/10.3390/catal6050068
Chicago/Turabian StyleKwon, Kyungjung, Yeojin Jung, Heesuk Ku, Kang Hee Lee, Sookyung Kim, Jeongsoo Sohn, and Chanho Pak. 2016. "CO-Tolerant Pt–BeO as a Novel Anode Electrocatalyst in Proton Exchange Membrane Fuel Cells" Catalysts 6, no. 5: 68. https://doi.org/10.3390/catal6050068
APA StyleKwon, K., Jung, Y., Ku, H., Lee, K. H., Kim, S., Sohn, J., & Pak, C. (2016). CO-Tolerant Pt–BeO as a Novel Anode Electrocatalyst in Proton Exchange Membrane Fuel Cells. Catalysts, 6(5), 68. https://doi.org/10.3390/catal6050068







