Fabrication and Catalytic Activity of Thermally Stable Gold Nanoparticles on Ultrastable Y (USY) Zeolites
Abstract
:1. Introduction
2. Results and Discussion
2.1. Loading of Au on Y-Type Zeolites
| Support | Si/Al2 | Loading/wt% | Support | Si/Al2 | Loading/wt% |
|---|---|---|---|---|---|
| NH4-ZSM-5 | 90 | 0.1 | H-USY | 7.7 | 0.1 |
| NH4-MOR | 15 | 0.1 | NH4-Y | 5.5 | 3.1 |
| NH4-beta | 20 | 1.8 | NH4-CaY | 7.7 | 3.3 |
| Na-Y | 5.5 | 0.1 | NH4-USY | 7.7 | 3.1 |
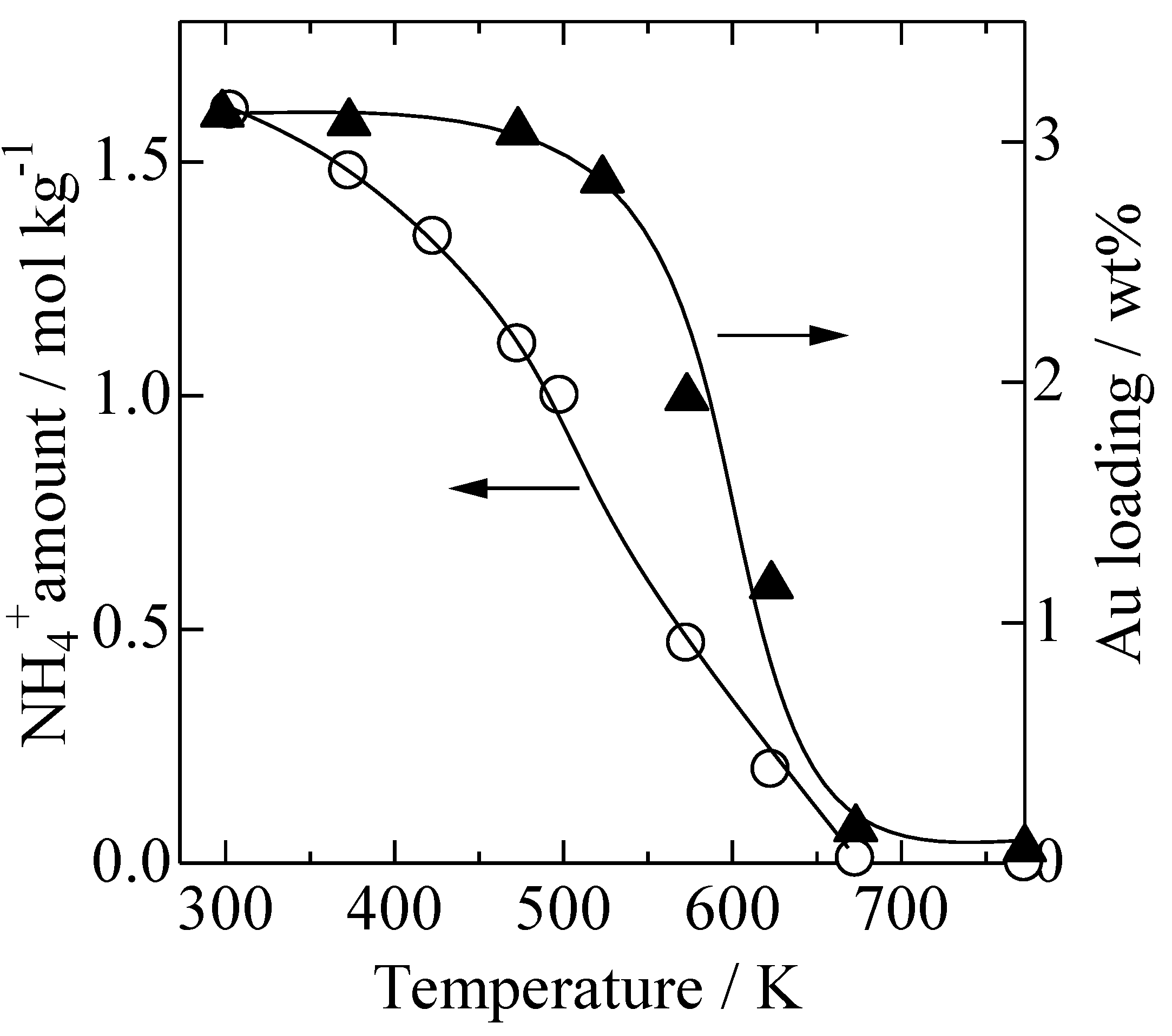
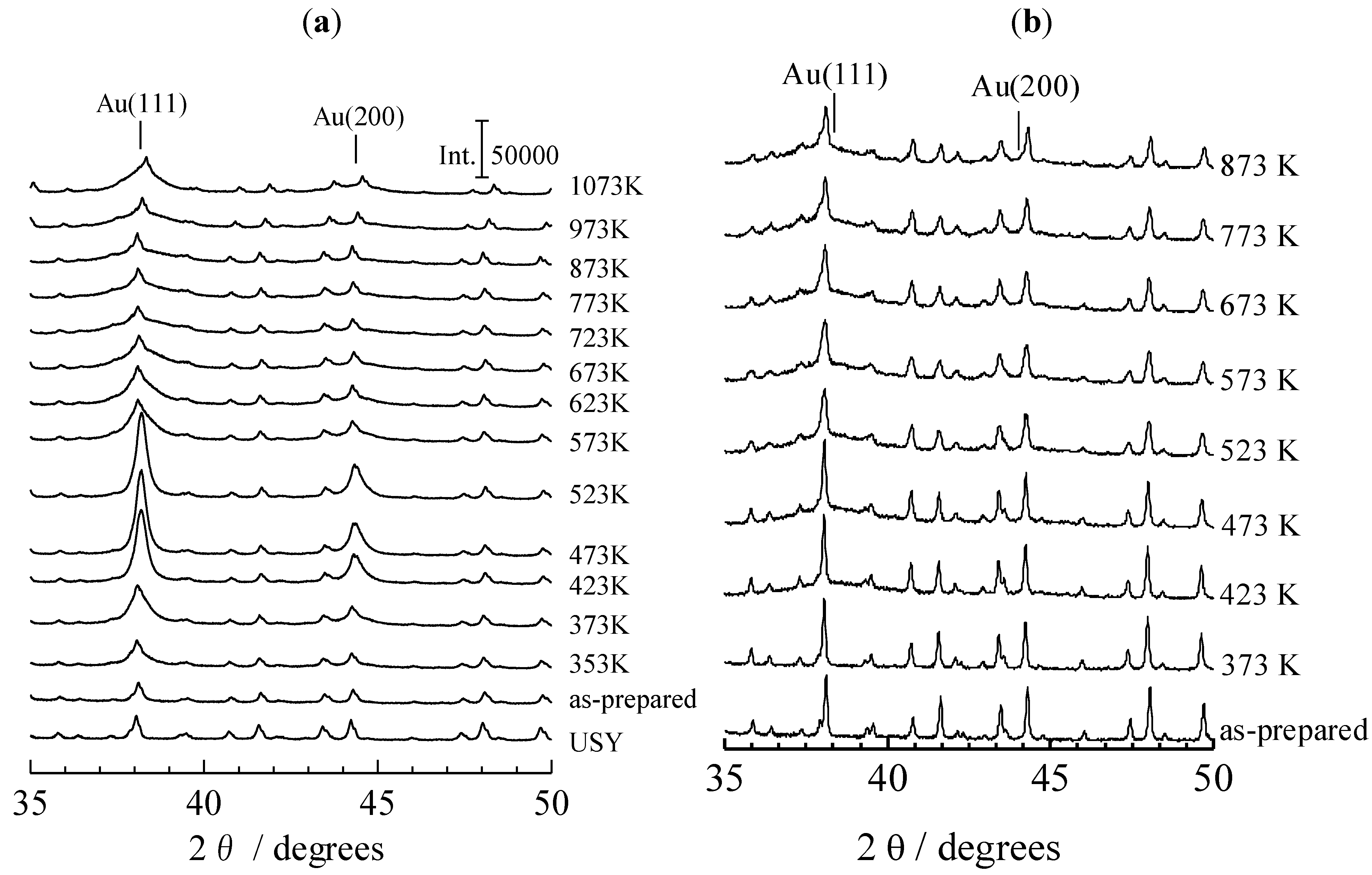
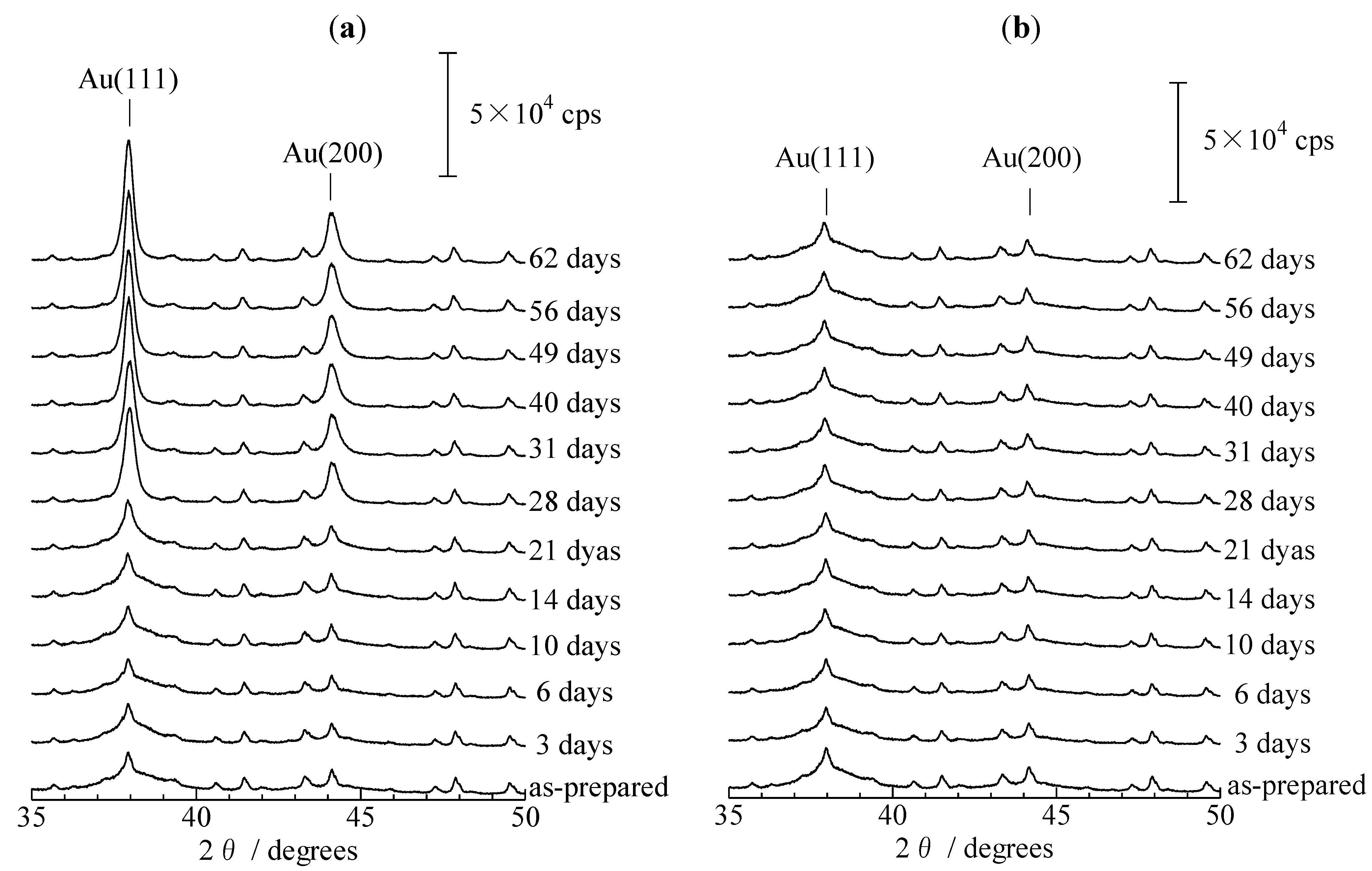
2.2. XRD Patterns Measured under Ex Situ and In Situ Conditions
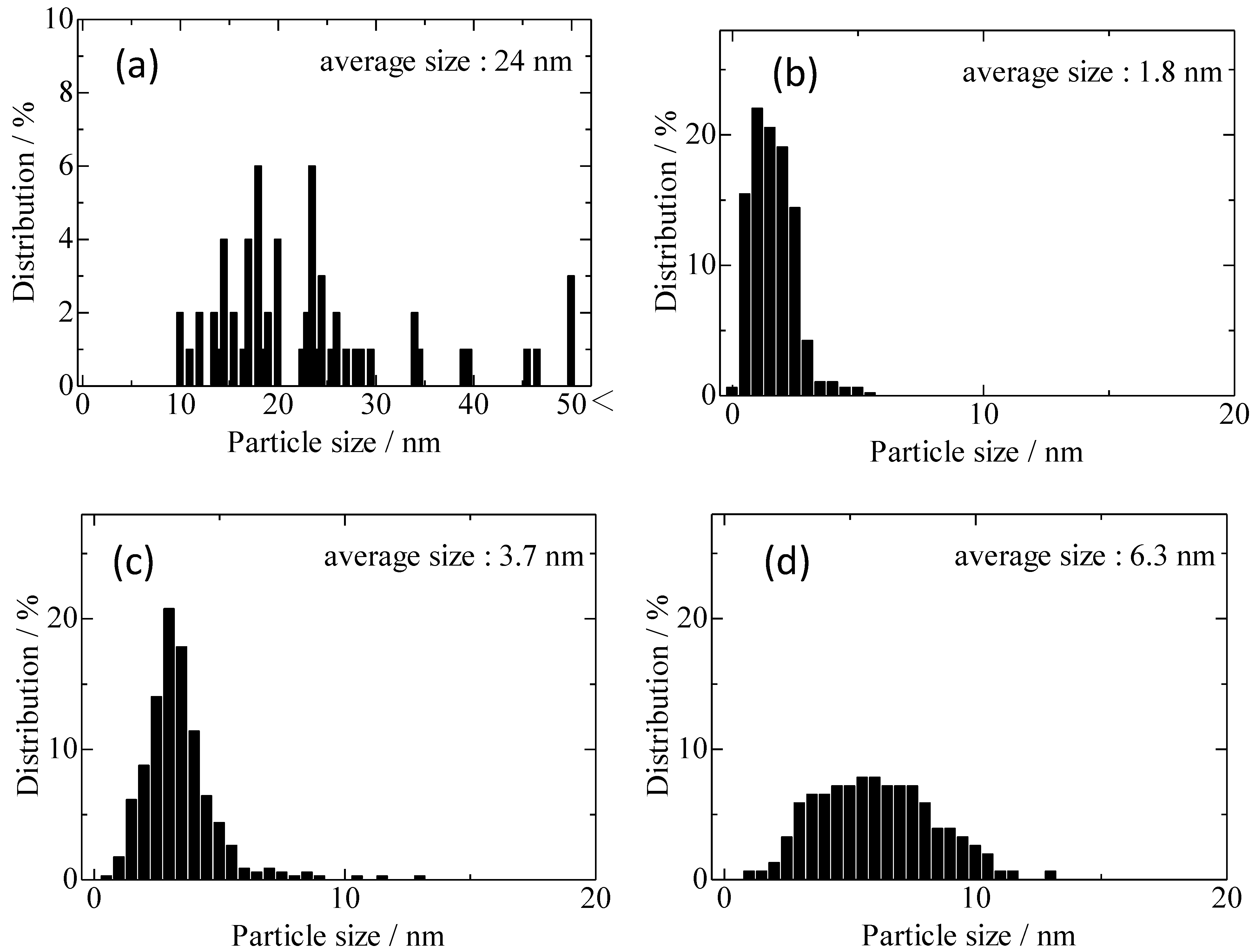
2.3. TEM Images of 3 wt%-Au/USY


2.4. IR Study of Adsorbed CO
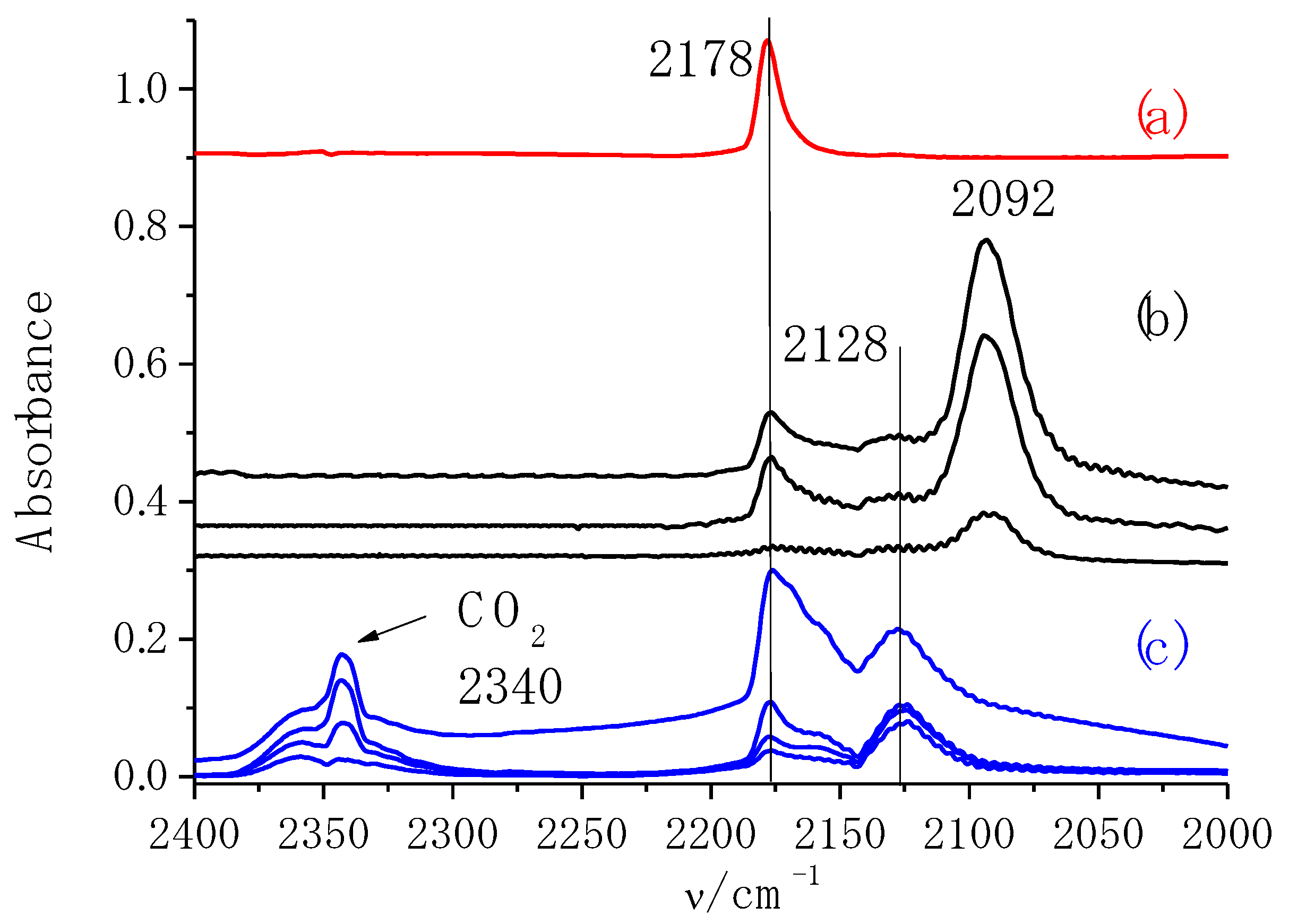

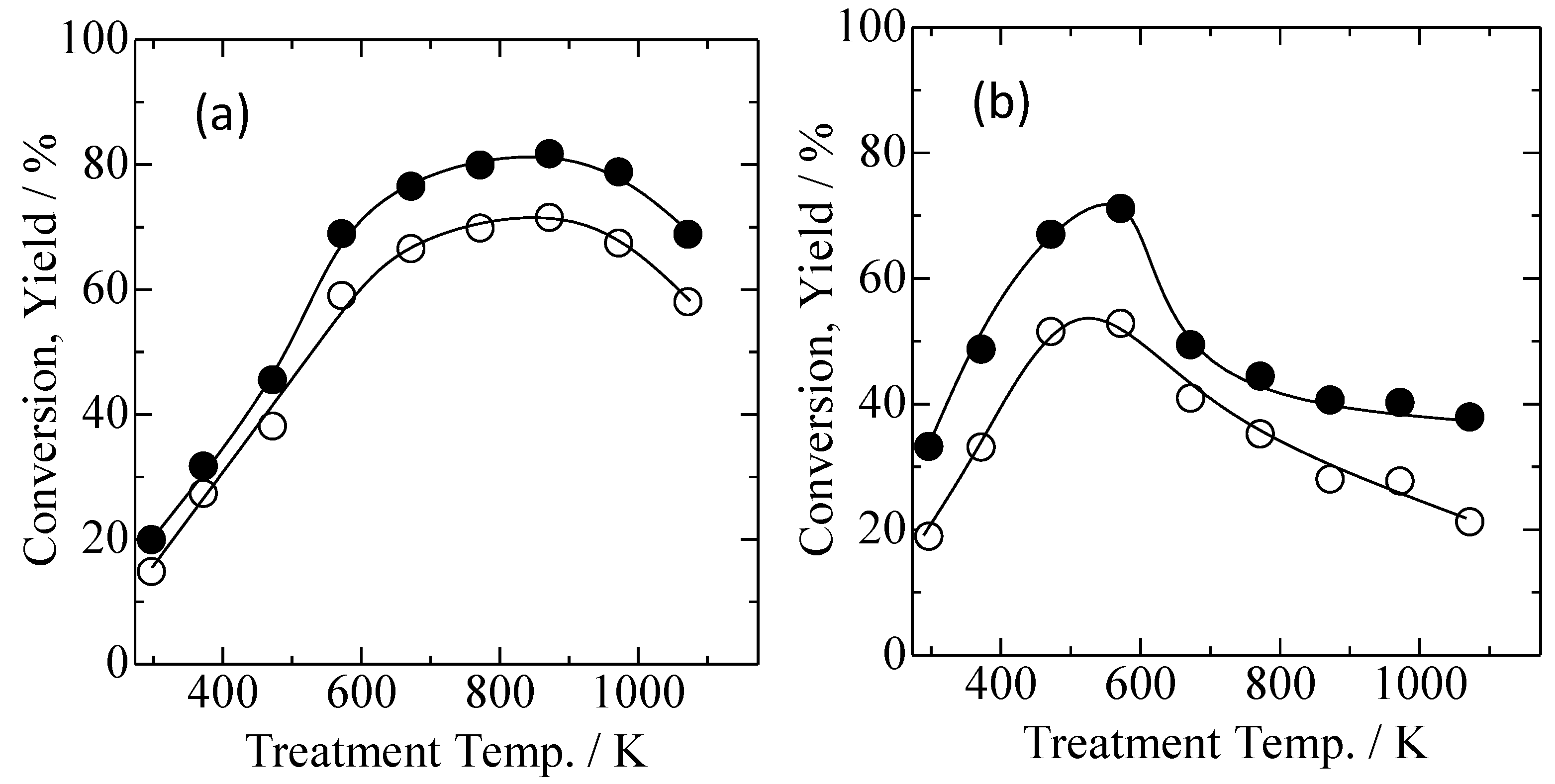
2.5. Catalytic Performance in Aerobic Oxidation of Benzyl Alcohol
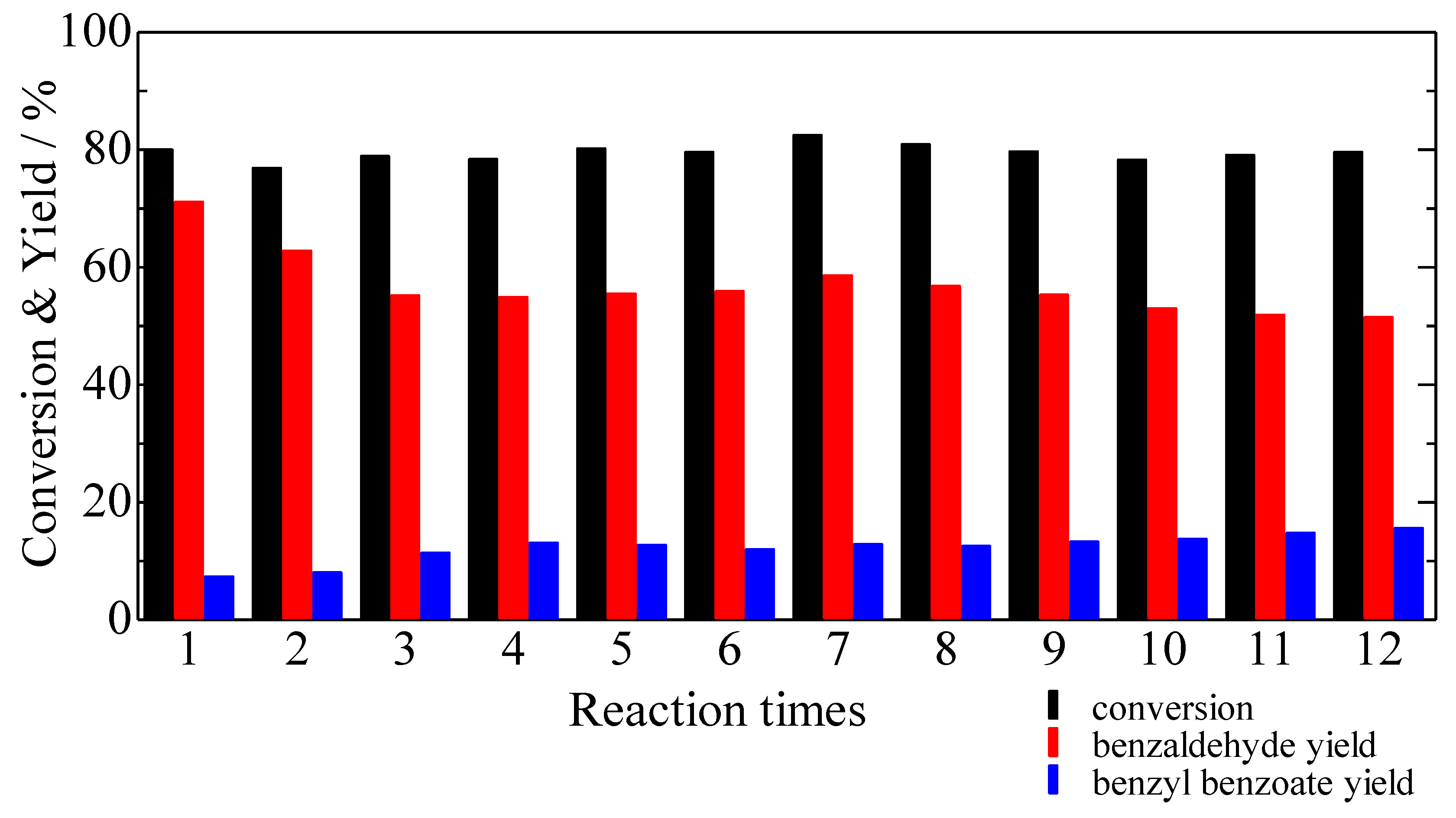
3. Experimental Section
3.1. Sample Preparation
3.2. TEM and XRD Data Collection and Analysis
3.3. IR Measurements and Analysis
3.4.Catalytic Reactions
4. Conclusions
Acknowledgments
References
- Haruta, M. Size- and support-dependency in the catalysis of gold. Catal. Today 1997, 36, 153–166. [Google Scholar]
- Daniel, M.C.; Astruc, D. Gold nanoparticles Assembly, supramolecular chemistry, quantum-size-related properties, and applications toward biology, catalysis, and nanotechnology. Chem. Rev. 2004, 104, 293–346. [Google Scholar] [CrossRef]
- Tsukuda, T.; Tsunoyama, H.; Sakurai, H. Aerobic Oxidations Catalyzed by Colloidal Nanogold. Chem. Asian J. 2011, 6, 736–748. [Google Scholar] [CrossRef]
- Haruta, M.; Date, M. Advances in the catalysis of Au nanoparticles. Appl. Catal. A 2001, 222, 427–437. [Google Scholar]
- Sardar, R.; Funston, A.M.; Mulvaney, P.; Murray, R.W. Gold Nanoparticles: Past, Present, and Future. Langmuir 2009, 25, 13840–13851. [Google Scholar] [CrossRef]
- Corma, A.; Garcia, H. Supported gold nanoparticles as catalysts for organic reactions. Chem. Soc. Rev. 2008, 37, 2096–2126. [Google Scholar] [CrossRef]
- Veith, G.M.; Lupini, A.R.; Rashkeev, S.; Pennycook, S.J.; Mullins, D.R.; Schwartz, V.; Bridges, C.A.; Dudney, N.J. Thermal stability and catalytic activity of gold nanoparticles supported on silica. J. Catal. 2009, 262, 92–101. [Google Scholar] [CrossRef]
- Okumura, M.; Nakamura, S.; Tsubota, S.; Nakamura, T.; Azuma, M.; Haruta, M. Chemical vapor deposition of gold on Al2O3, SiO2, and TiO2 for the oxidation of CO and of H2. Catal. Lett. 1998, 51, 53–58. [Google Scholar]
- Zanella, R.; Giorgio, S.; Henry, C.R.; Louis, C. Alternative methods for the preparation of gold nanoparticles supported on TiO2. J. Phys. Chem. B 2002, 106, 7634–7642. [Google Scholar] [CrossRef]
- Zanella, R.; Giorgio, S.; Shin, C.H.; Henry, C.R.; Louis, C. Characterization and reactivity in CO oxidation of gold nanoparticles supported on TiO2 prepared by deposition-precipitation with NaOH and urea. J. Catal. 2004, 222, 357–367. [Google Scholar] [CrossRef]
- Tsubota, S.; Cunningham, D.; Bando, Y.; Haruta, M. CO Oxidation over Gold Supported on TiO2. Stud. Surf. Sci. Catal. 1993, 77, 325–328. [Google Scholar] [CrossRef]
- Li, G.; EdwardS, J.; Carley, A.F.; Hutchings, G.J. Direct synthesis of hydrogen peroxide from H2 and O2 using zeolite-supported Au catalysts. Catal. Today 2006, 114, 369–371. [Google Scholar] [CrossRef]
- Guillemot, D.; Borovkov, V.Y.; Kazansky, V.B.; PolissetThfoin, M.; Fraissard, J. Surface characterization of Au/HY by Xe-129 NMR and diffuse reflectance IR spectroscopy of adsorbed CO. Formation of electron-deficient gold particles inside HY cavities. J. Chem. Soc.-Faraday Trans. 1997, 93, 3587–3591. [Google Scholar] [CrossRef]
- Hojholt, K.T.; Laursen, A.B.; Kegnaes, S.; Christensen, C.H. Size-Selective Oxidation of Aldehydes with Zeolite Encapsulated Gold Nanoparticles. Top. Catal. 2011, 54, 1026–1033. [Google Scholar] [CrossRef]
- Lin, C.H.; Lin, S.D.; Lee, J.F. Chlorine residue in the Au/gamma-Al2O3 prepared by AuCl3 impregnation—An EXAFS analysis. Catal. Lett. 2003, 89, 235–242. [Google Scholar]
- Okumura, K.; Tomiyama, T.; Morishita, N.; Sanada, T.; Kamiguchi, K.; Katada, N.; Niwa, M. Evolution of strong acidity and high-alkane-cracking activity in ammonium-treated USY zeolites. Appl. Catal. A 2011, 405, 8–17. [Google Scholar] [CrossRef]
- Rodriguez-Reyes, J.C.F.; Friend, C.M.; Madix, R.J. Origin of the selectivity in the gold-mediated oxidation of benzyl alcohol. Surf. Sci. 2012, 606, 1129–1134. [Google Scholar] [CrossRef]
- Zhou, L.P.; Yu, W.J.; Wu, L.; Liu, Z.; Chen, H.J.; Yang, X.M.; Su, Y.L.; Xu, J. Nanocrystalline gold supported on NaY as catalyst for the direct oxidation of primary alcohol to carboxylic acid with molecular oxygen in water. Appl. Catal. A 2013, 451, 137–143. [Google Scholar] [CrossRef]
- Okumura, K.; Murakami, C.; Oyama, T.; Sanada, T.; Isoda, A.; Katada, N. Formation of nanometer-sized Au particles on USY zeolites under hydrogen atmosphere. Gold Bull. 2012, 45, 83–90. [Google Scholar] [CrossRef]
- Sadowska, K.; Gora-Marek, K.; Datka, J. Hierarchic zeolites studied by IR spectroscopy: Acid properties of zeolite ZSM-5 desilicated with NaOH and NaOH/tetrabutylamine hydroxide. Vib. Spectrosc. 2012, 63, 418–425. [Google Scholar] [CrossRef]
- Grunwaldt, J.-D.; Maciejewski, M.; Becker, O.S.; Fabrizioli, P.; Baiker, A. Comparative Study of Au/TiO2 and Au/ZrO2 Catalysts for Low-Temperature CO Oxidation. J. Catal. 1999, 186, 458–469. [Google Scholar] [CrossRef]
- Venkov, T.; Fajerwerg, K.; Delannoy, L.; Klimev, H.; Hadjiivanov, K.; Louis, C. Effect of the activation temperature on the state of gold supported on titania: An FT-IR spectroscopic study. Appl. Catal. A 2005, 301, 106–114. [Google Scholar]
- Lee, J.Y.; Schwank, J. Infrared Spectroscopic Study of NO Reduction by H2 on Supported Gold Catalysts. J. Catal. 1986, 102, 207–215. [Google Scholar] [CrossRef]
- Debeila, M.A.; Coville, N.J.; Scurrell, M.S.; Hearne, G.R. DRIFTS studies of the interaction of nitric oxide and carbon monoxide on Au-TiO2. Catal. Today 2002, 72, 79–87. [Google Scholar] [CrossRef]
- Zahmakiran, M.; Ozkar, S. The preparation and characterization of gold(0) nanoclusters stabilized by zeolite framework: Highly active, selective and reusable catalyst in aerobic oxidation of benzyl alcohol. Mater. Chem. Phys. 2010, 121, 359–363. [Google Scholar] [CrossRef]
- Noda, T.; Suzuki, K.; Katada, N.; Niwa, M. Combined study of IRMS-TPD measurement and DFT calculation on Bronsted acidity and catalytic cracking activity of cation-exchanged Y zeolites. J. Catal. 2008, 259, 203–210. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Sanada, T.; Murakami, C.; Góra-Marek, K.; Iida, K.; Katada, N.; Okumura, K. Fabrication and Catalytic Activity of Thermally Stable Gold Nanoparticles on Ultrastable Y (USY) Zeolites. Catalysts 2013, 3, 599-613. https://doi.org/10.3390/catal3030599
Sanada T, Murakami C, Góra-Marek K, Iida K, Katada N, Okumura K. Fabrication and Catalytic Activity of Thermally Stable Gold Nanoparticles on Ultrastable Y (USY) Zeolites. Catalysts. 2013; 3(3):599-613. https://doi.org/10.3390/catal3030599
Chicago/Turabian StyleSanada, Takashi, Chika Murakami, Kinga Góra-Marek, Keiko Iida, Naonobu Katada, and Kazu Okumura. 2013. "Fabrication and Catalytic Activity of Thermally Stable Gold Nanoparticles on Ultrastable Y (USY) Zeolites" Catalysts 3, no. 3: 599-613. https://doi.org/10.3390/catal3030599
APA StyleSanada, T., Murakami, C., Góra-Marek, K., Iida, K., Katada, N., & Okumura, K. (2013). Fabrication and Catalytic Activity of Thermally Stable Gold Nanoparticles on Ultrastable Y (USY) Zeolites. Catalysts, 3(3), 599-613. https://doi.org/10.3390/catal3030599




