Bandgap-Tunable ZnxCd1−xS Solid Solutions for Effective Photocatalytic Degradation of Norfloxacin Under Visible Light and Natural Sunlight
Abstract
1. Introduction
2. Results
2.1. Structural Characterization
2.2. Surface Morphology Characterization
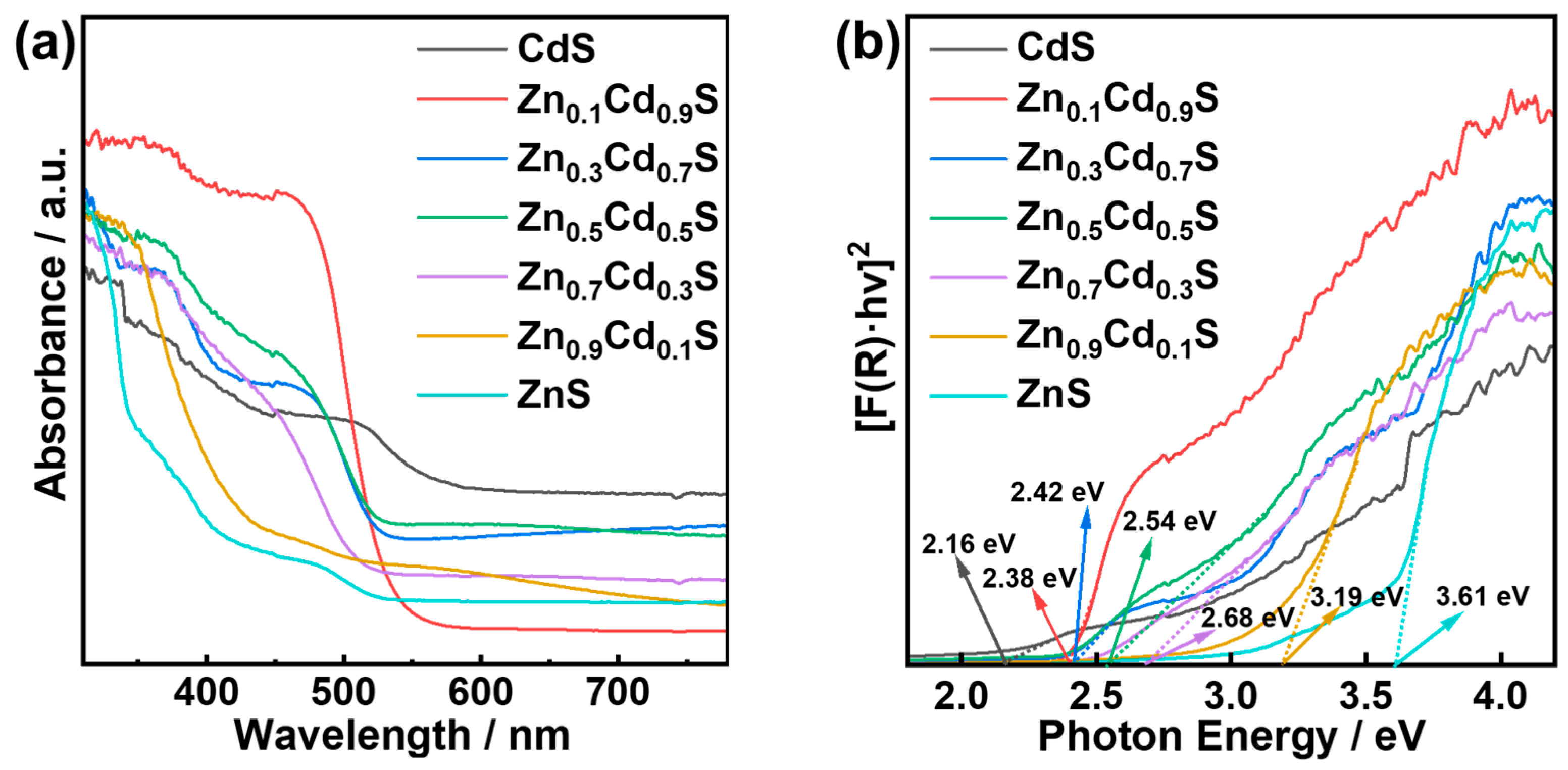
2.3. Optical Absorption
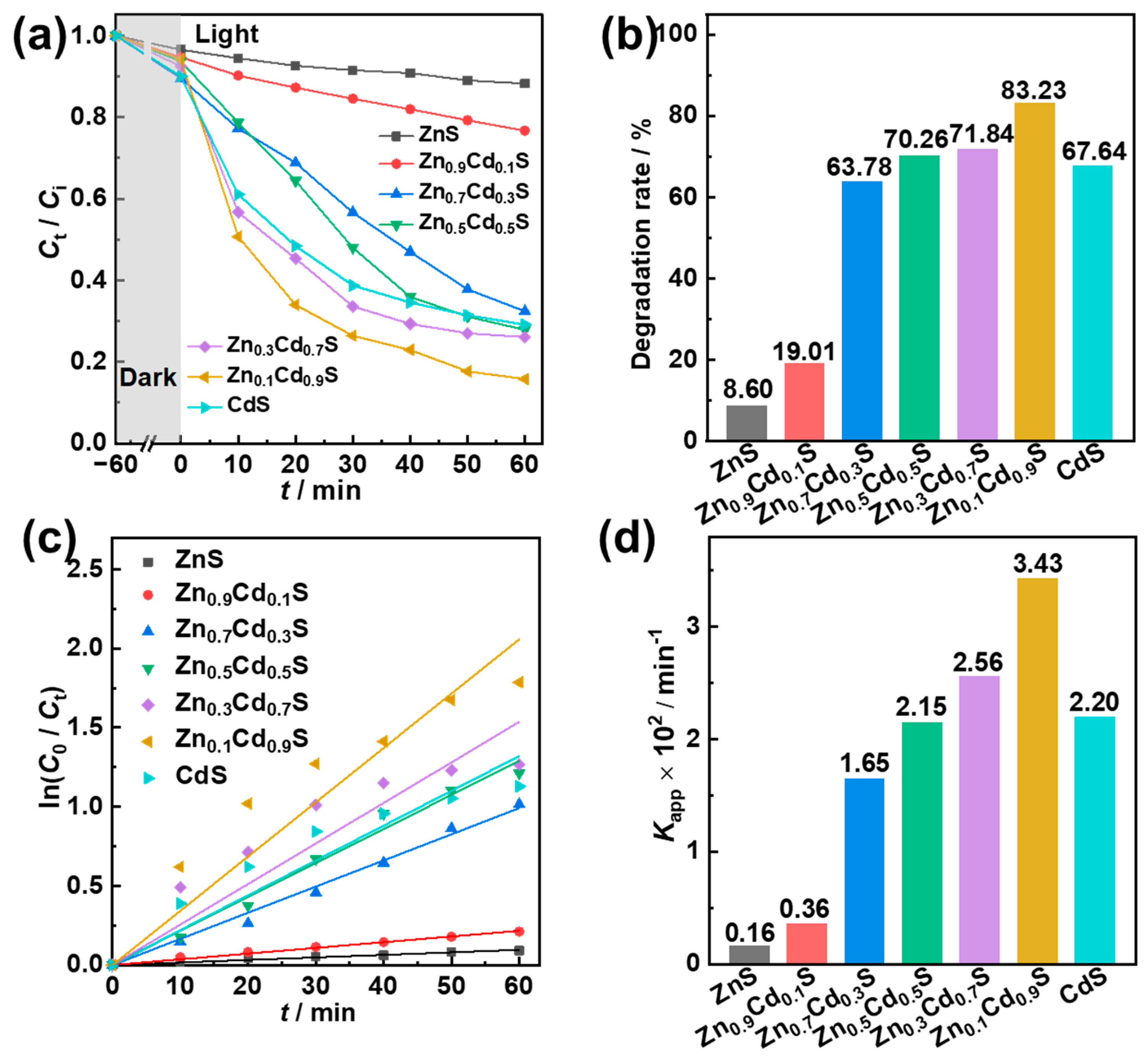
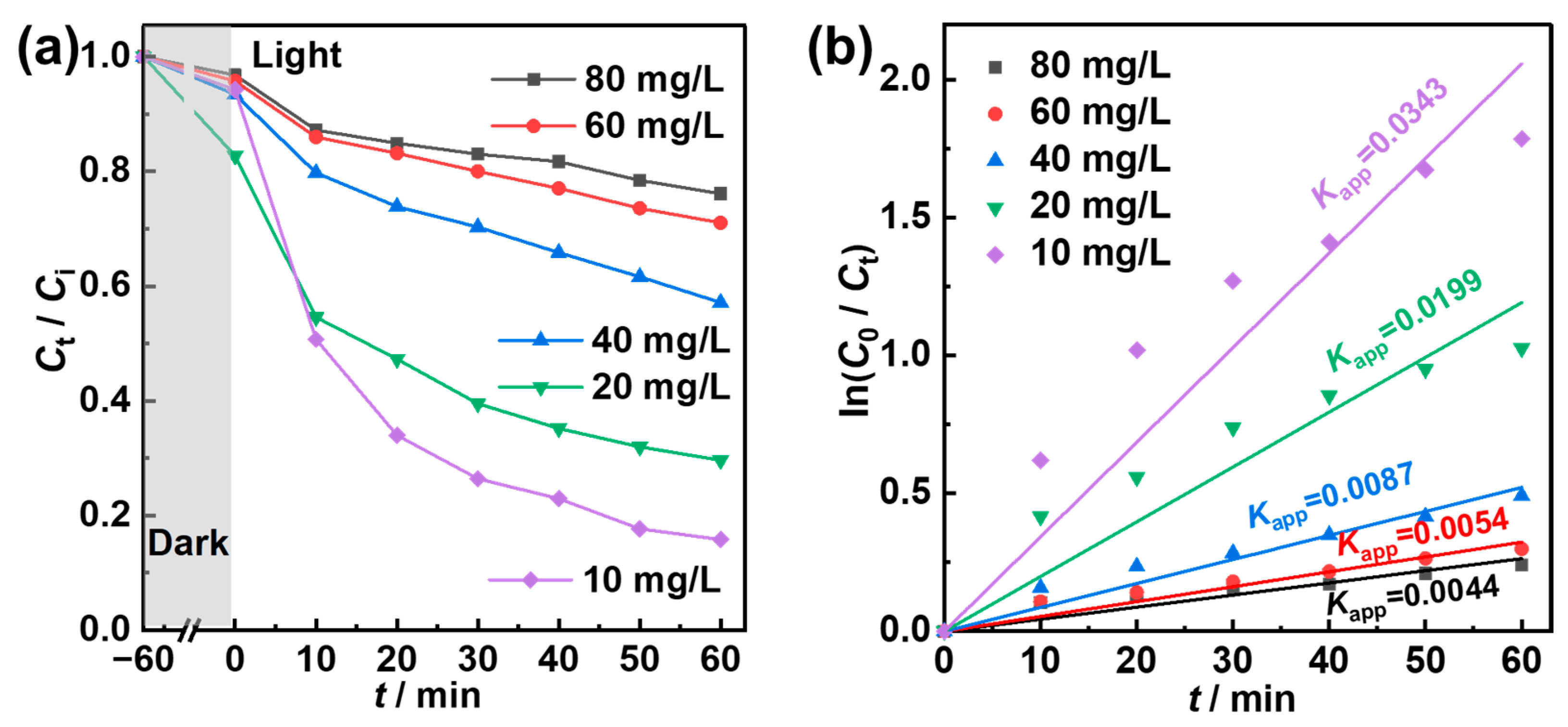
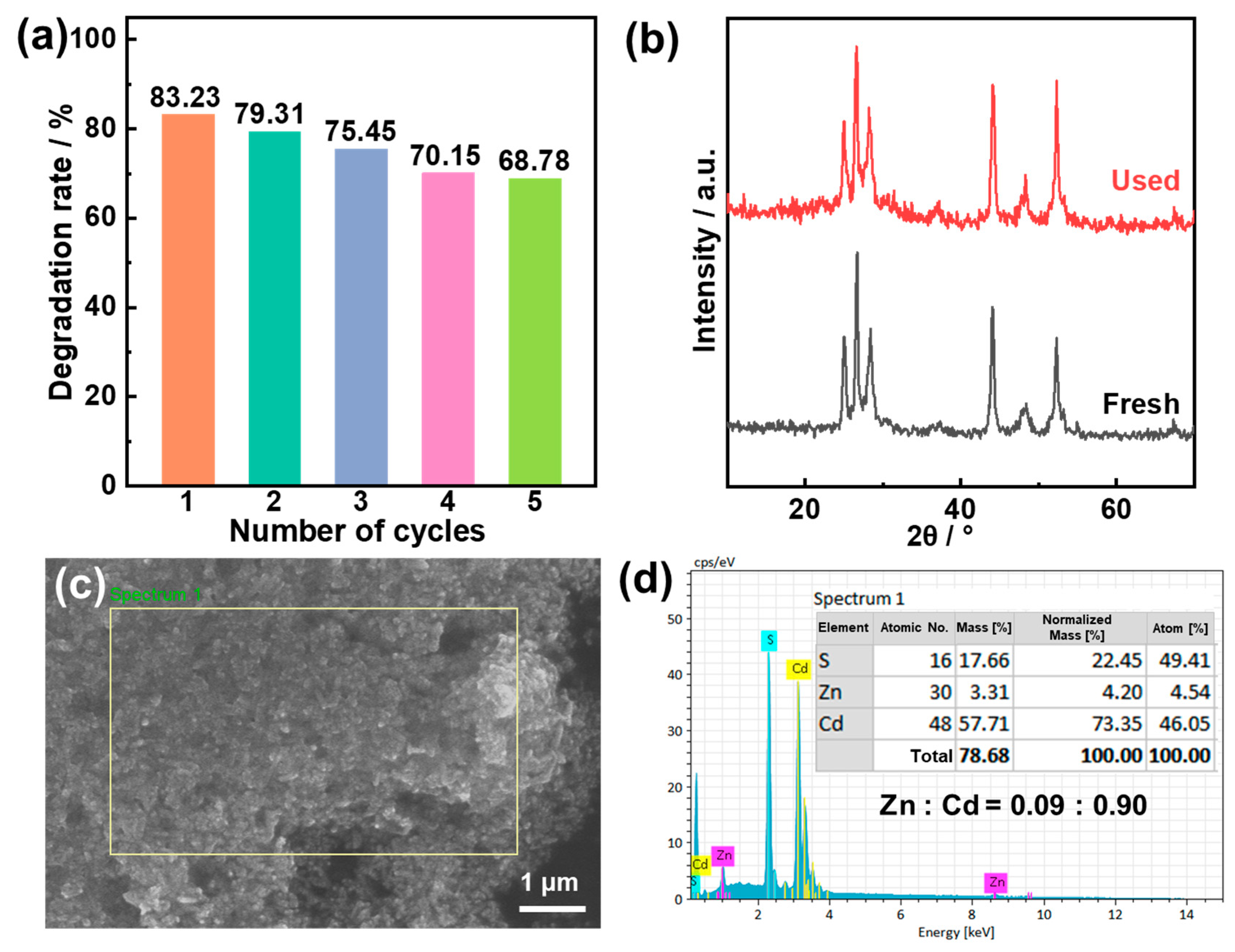
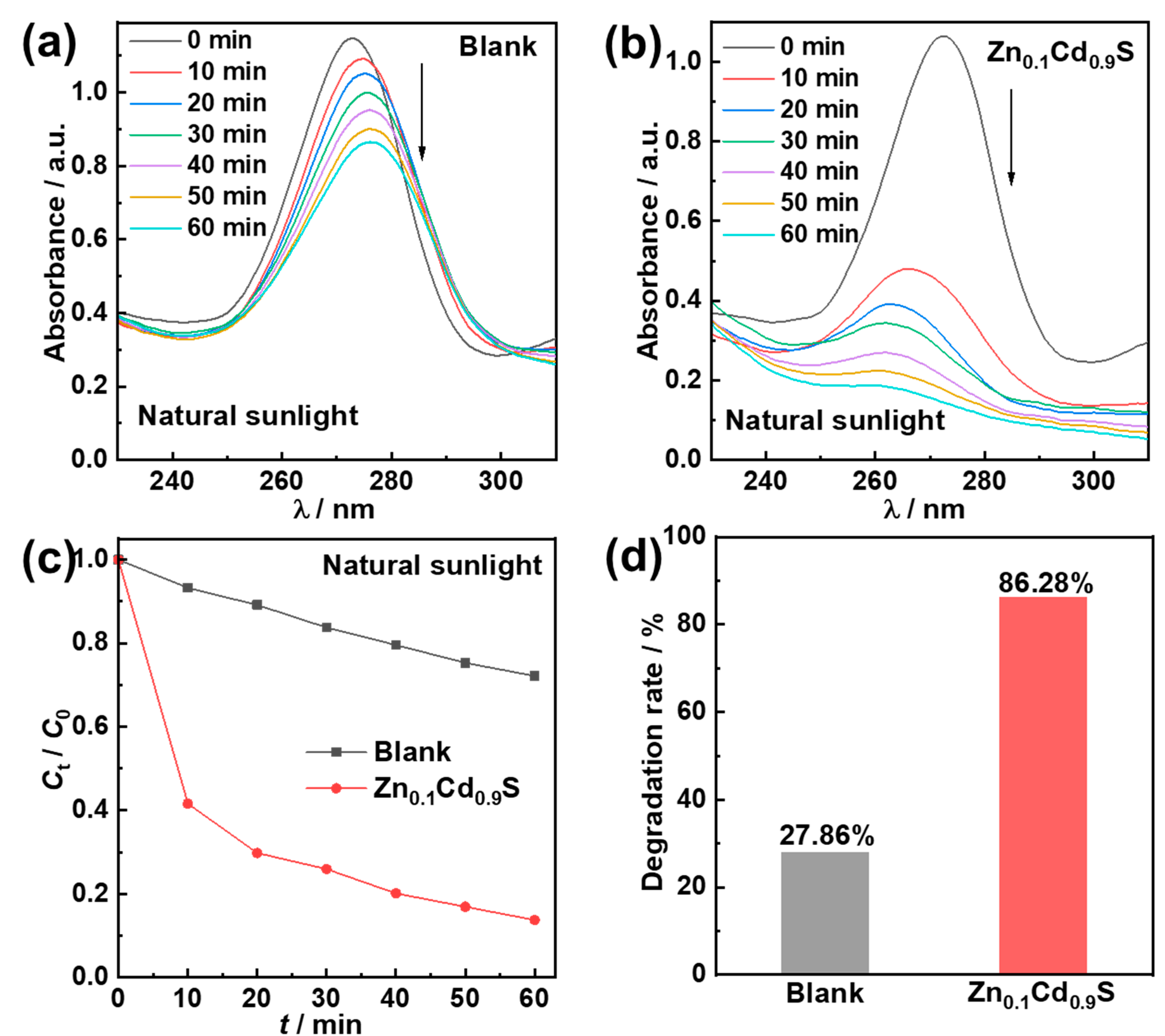
2.4. Photocatalytic Degradation Performance
3. Materials and Methods
3.1. Materials
3.2. Synthesis of ZnxCd1−xS (x = 0~1) Solid Solutions
3.3. Physicochemical Characterization of Obtained Samples
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Luo, Q.; Zhuang, W.; Sui, M. Combating Antibiotic Resistance in Persulfate-Based Advanced Oxidation Processes: Activation Methods and Energy Consumption. Environ. Res. 2025, 270, 120932. [Google Scholar] [CrossRef]
- Xu, L.; Zhang, H.; Xiong, P.; Zhu, Q.; Liao, C.; Jiang, G. Occurrence, fate, and risk assessment of typical tetracycline antibiotics in the aquatic environment: A review. Sci. Total Environ. 2021, 753, 141975. [Google Scholar] [CrossRef]
- Yang, X.; Chen, Z.; Zhao, W.; Liu, C.; Qian, X.; Zhang, M.; Wei, G.; Khan, E.; Hau Ng, Y.; Sik Ok, Y. Recent advances in photodegradation of antibiotic residues in water. Chem. Eng. J. 2021, 405, 126806. [Google Scholar] [CrossRef]
- Bhatt, S.; Chatterjee, S. Fluoroquinolone antibiotics: Occurrence, mode of action, resistance, environmental detection, and remediation—A comprehensive review. Environ. Pollut. 2022, 315, 120440. [Google Scholar] [CrossRef]
- Shurbaji, S.; Huong, P.T.; Altahtamouni, T.M. Review on the Visible Light Photocatalysis for the Decomposition of Ciprofloxacin, Norfloxacin, Tetracyclines, and Sulfonamides Antibiotics in Wastewater. Catalysts 2021, 11, 437. [Google Scholar] [CrossRef]
- Ahtasham Iqbal, M.; Akram, S.; Khalid, S.; Lal, B.; Hassan, S.U.; Ashraf, R.; Kezembayeva, G.; Mushtaq, M.; Chinibayeva, N.; Hosseini-Bandegharaei, A. Advanced photocatalysis as a viable and sustainable wastewater treatment process: A comprehensive review. Environ. Res. 2024, 253, 118947. [Google Scholar] [CrossRef]
- Pouramini, Z.; Mousavi, S.M.; Babapoor, A.; Hashemi, S.A.; Lai, C.W.; Mazaheri, Y.; Chiang, W.-H. Effect of Metal Atom in Zeolitic Imidazolate Frameworks (ZIF-8 & 67) for Removal of Dyes and Antibiotics from Wastewater: A Review. Catalysts 2023, 13, 155. [Google Scholar] [CrossRef]
- Thakur, V.; Singh, S.; Kumar, P.; Rawat, S.; Chandra Srivastava, V.; Lo, S.-L.; Štangar, U.L. Photocatalytic behaviors of bismuth-based mixed oxides: Types, fabrication techniques and mineralization mechanism of antibiotics. Chem. Eng. J. 2023, 475, 146100. [Google Scholar] [CrossRef]
- Rathi, A.; Barman, S.; Basu, S.; Arya, R.K. Post-fabrication structural changes and enhanced photodegradation activity of semiconductors@zeolite composites towards noxious contaminants. Chemosphere 2022, 288, 132609. [Google Scholar] [CrossRef] [PubMed]
- Isac, L.; Enesca, A. Recent Developments in ZnS-Based Nanostructures Photocatalysts for Wastewater Treatment. Int. J. Mol. Sci. 2022, 23, 15668. [Google Scholar] [CrossRef] [PubMed]
- Jie, L.; Gao, X.; Cao, X.; Wu, S.; Long, X.; Ma, Q.; Su, J. A review of CdS photocatalytic nanomaterials: Morphology, synthesis methods, and applications. Mater. Sci. Semicond. Process. 2024, 176, 108288. [Google Scholar] [CrossRef]
- Le, Y.; Wang, H. Remediation of wastewater by using CdS-based biohybrids: Challenges and enhancement strategies. Bioresour. Technol. 2025, 426, 132379. [Google Scholar] [CrossRef]
- Khan, M.M.; Abdulwahab, K.O. Metals- and non-metals-doped ZnS for various photocatalytic applications. Mater. Sci. Semicond. Process. 2024, 181, 108634. [Google Scholar] [CrossRef]
- Zhang, H.; Wang, Z.; Zhang, J.; Dai, K. Metal-sulfide-based heterojunction photocatalysts: Principles, impact, applications, and in-situ characterization. Chin. J. Catal. 2023, 49, 42–67. [Google Scholar] [CrossRef]
- Huang, D.; Wen, M.; Zhou, C.; Li, Z.; Cheng, M.; Chen, S.; Xue, W.; Lei, L.; Yang, Y.; Xiong, W.; et al. ZnxCd1−xS based materials for photocatalytic hydrogen evolution, pollutants degradation and carbon dioxide reduction. Appl. Catal. B Environ. 2020, 267, 118651. [Google Scholar] [CrossRef]
- Liu, Y.; Zheng, X.; Yang, Y.; Song, Y.; Yang, Y.; Li, J.; Shim, C.M.; Shen, Y.; Tian, X. Recent Advances in the Hydrogen Evolution Reaction of ZnxCd1−xS-Based Photocatalysts. Sol. RRL 2022, 6, 2101061. [Google Scholar] [CrossRef]
- Leiu, Y.X.; Ling, G.Z.S.; Mohamed, A.R.; Wang, S.; Ong, W.-J. Atomic-level tailoring ZnxCd1−xS photocatalysts: A paradigm for bridging structure-performance relationship toward solar chemical production. Mater. Today Energy 2023, 34, 101281. [Google Scholar] [CrossRef]
- Wang, Z.; Lu, D.; Kondamareddy, K.K.; He, Y.; Gu, W.; Li, J.; Fan, H.; Wang, H.; Ho, W. Recent Advances and Insights in Designing ZnxCd1–xS-Based Photocatalysts for Hydrogen Production and Synergistic Selective Oxidation to Value-Added Chemical Production. ACS Appl. Mater. Interfaces 2024, 16, 48895–48926. [Google Scholar] [CrossRef]
- Liu, R.; Guo, J.; Gan, W.; Chen, R.; Ding, S.; Zhao, Z.; Li, J.; Zhang, M.; Sun, Z. Construction of ZnCdS/TiO2 Z-Scheme heterostructure with excellent photocatalytic performance for degradation of norfloxacin. Appl. Surf. Sci. 2024, 650, 159229. [Google Scholar] [CrossRef]
- Lu, G.; Li, W.; Li, Z.; Gu, G.; Han, Q.; Liang, J.; Chen, Z. Enhanced Degradation of Norfloxacin Under Visible Light by S-Scheme Fe2O3/g–C3N4 Heterojunctions. Molecules 2024, 29, 5212. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.; Chen, Q.; Wu, S. Photocatalytic degradation of norfloxacin antibiotics on ZnxCd(1-x)S/g-C3N4 composites in water. Environ. Sci. Pollut. Res. 2024, 31, 16473–16484. [Google Scholar] [CrossRef]
- Xing, C.; Zhang, Y.; Yan, W.; Guo, L. Band structure-controlled solid solution of Cd1-xZnxS photocatalyst for hydrogen production by water splitting. Int. J. Hydrogen Energy 2006, 31, 2018–2024. [Google Scholar] [CrossRef]
- Li, X.; Wang, S.; Liu, Y.; Yang, D.; Yuan, F.; Gao, J. The bandgap tunable Zn1−xCdxS solid solutions with enhanced photocatalytic property in water environmental treatment. J. Aust. Ceram. Soc. 2023, 59, 1197–1204. [Google Scholar] [CrossRef]
- Xue, F.; Fu, W.; Liu, M.; Wang, X.; Wang, B.; Guo, L. Insight into Cd0.9Zn0.1S solid-solution nanotetrapods: Growth mechanism and their application for photocatalytic hydrogen production. Int. J. Hydrogen Energy 2016, 41, 20455–20464. [Google Scholar] [CrossRef]
- Jubeer, E.M.; Manthrammel, M.A.; Subha, P.A.; Shkir, M.; Biju, K.P.; AlFaify, S.A. Defect engineering for enhanced optical and photocatalytic properties of ZnS nanoparticles synthesized by hydrothermal method. Sci. Rep. 2023, 13, 16820. [Google Scholar] [CrossRef]
- Wei, Z.; Ji, Y.; Bielan, Z.; Yue, X.; Xu, Y.; Sun, J.; Chen, S.; Yi, G.; Chang, Y.; Kowalska, E. Platinum-Modified Rod-like Titania Mesocrystals with Enhanced Photocatalytic Activity. Catalysts 2024, 14, 283. [Google Scholar] [CrossRef]
- Wu, T.; Ren, X.; Zhao, X. Construction of Lamellar CoFe-LDHs@MoS2 to Promote Permonosulfate Properties Leading to Effective Photocatalytic Degradation of Norfloxacin. Catalysts 2024, 14, 860. [Google Scholar] [CrossRef]
- Gao, Y.; Cao, T.; Du, J.; Qi, X.; Yan, H.; Xu, X. The Bi-Modified (BiO)2CO3/TiO2 Heterojunction Enhances the Photocatalytic Degradation of Antibiotics. Catalysts 2025, 15, 56. [Google Scholar] [CrossRef]
- Zheng, C.; Li, T.; Wang, Y.; Wu, Y.; Wu, J.; Yu, F.; Lu, C.; Xie, Y. Epitaxially induced carrier space vector separation over Zn0.1Cd0.9S/Co9S8 Schottky junctions. Sep. Purif. Technol. 2025, 364, 132514. [Google Scholar] [CrossRef]
- Zheng, C.; Wang, Y.; Wu, Y.; Wu, J.; Lu, J.; Xie, Y. Epitaxial induction driven charge vector Steering in Zn0.1Cd0.9S/Cu2WS4 S-scheme heterojunctions for superior photocatalytic hydrogen evolution. J. Colloid Interface Sci. 2025, 696, 137890. [Google Scholar] [CrossRef] [PubMed]
- Patel, J.; Singh, A.K.; Carabineiro, S.A.C. Assessing the Photocatalytic Degradation of Fluoroquinolone Norfloxacin by Mn:ZnS Quantum Dots: Kinetic Study, Degradation Pathway and Influencing Factors. Nanomaterials 2020, 10, 964. [Google Scholar] [CrossRef]
- Wu, Z.; Wang, X.-L.; Wang, X.; Xu, X.; Li, D.-S.; Wu, T. 0D/2D heterostructure constructed by ultra-small chalcogenide-cluster aggregated quaternary sulfides and g-C3N4 for enhanced photocatalytic H2 evolution. Chem. Eng. J. 2021, 426, 131216. [Google Scholar] [CrossRef]
- Li, J.; Liu, C.; Wang, X.; Ding, Y.; Wu, Z.; Sun, P.; Tang, J.; Zhang, J.; Li, D.-S.; Chen, N.; et al. Stable 3D neutral gallium thioantimonate frameworks decorated with transition metal complexes for a tunable photocatalytic hydrogen evolution. Dalton Trans. 2022, 51, 978–985. [Google Scholar] [CrossRef]
- Kar, S.; Ibrahim, S.; Pal, T.; Ghosh, S. Enhance Solar-Light-Driven Photocatalytic Degradation of Norfloxacin Aqueous Solution by RGO-Based CdxZn1−xS Alloy Composite with Band-Gap Tuneability. ChemistrySelect 2020, 5, 54–60. [Google Scholar] [CrossRef]
- Wan, S.; Wang, W.; Cheng, B.; Luo, G.; Shen, Q.; Yu, J.; Zhang, J.; Cao, S.; Zhang, L. A superlattice interface and S-scheme heterojunction for ultrafast charge separation and transfer in photocatalytic H2 evolution. Nat. Commun. 2024, 15, 9612. [Google Scholar] [CrossRef]
- Rana, S.; Kumar, A.; Wang, T.; Dhiman, P.; Sharma, G.; Shi, H. Incorporating C3N5 and NiCo2S4 to Form a Novel Z-Scheme Heterojunction for Superior Photocatalytic Degradation of Norfloxacin. Chemistry 2024, 6, 962–980. [Google Scholar] [CrossRef]
- Lee, E.; Jagan, G.; Choi, J.U.; Cha, B.; Yoon, Y.; Saravanakumar, K.; Park, C.M. Peroxymonosulfate-activated photocatalytic degradation of norfloxacin via a dual Z-scheme g-C3N5/BiVO4/CoFe − LDH heterojunction: Operation and mechanistic insights. Chem. Eng. J. 2024, 494, 152961. [Google Scholar] [CrossRef]
- Li, Y.; Wang, W.; Chen, L.; Ma, H.; Lu, X.; Ma, H.; Liu, Z. Visible-Light-Driven Z-Type Pg-C3N4/Nitrogen Doped Biochar/BiVO4 Photo-Catalysts for the Degradation of Norfloxacin. Materials 2024, 17, 1634. [Google Scholar] [CrossRef]
- Fu, S.; Huang, Z.; Wang, Y.; Zheng, B.; Yuan, W.; Li, L.; Deng, P.; Zhu, H.; Zhang, H.; Liu, B. Fabrication of a Novel Z-Scheme AgBiO3/BiOCl Heterojunction with Excellent Photocatalytic Performance towards Organic Pollutant. Materials 2024, 17, 4615. [Google Scholar] [CrossRef]
- Sasikumar, K.; Rajamanikandan, R.; Ju, H. Construction of Z-Scheme ZIF67/NiMoO4 Heterojunction for Enhanced Photocatalytic Degradation of Antibiotic Pollutants. Materials 2024, 17, 6225. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Song, D.; Liao, L.; Wang, B.; Li, Z.; Li, M.; Zhou, W. Bi/Mn-Doped BiOCl Nanosheets Self-Assembled Microspheres toward Optimized Photocatalytic Performance. Nanomaterials 2023, 13, 2408. [Google Scholar] [CrossRef] [PubMed]
- Song, D.; Li, M.; Liao, L.; Guo, L.; Liu, H.; Wang, B.; Li, Z. High-Crystallinity BiOCl Nanosheets as Efficient Photocatalysts for Norfloxacin Antibiotic Degradation. Nanomaterials 2023, 13, 1841. [Google Scholar] [CrossRef] [PubMed]




Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, X.; Zhang, X.; Qu, Y.; Liu, T.; Luo, J.; Long, T.; Wu, L.; Tian, C.; Hu, Y. Bandgap-Tunable ZnxCd1−xS Solid Solutions for Effective Photocatalytic Degradation of Norfloxacin Under Visible Light and Natural Sunlight. Catalysts 2025, 15, 819. https://doi.org/10.3390/catal15090819
Wang X, Zhang X, Qu Y, Liu T, Luo J, Long T, Wu L, Tian C, Hu Y. Bandgap-Tunable ZnxCd1−xS Solid Solutions for Effective Photocatalytic Degradation of Norfloxacin Under Visible Light and Natural Sunlight. Catalysts. 2025; 15(9):819. https://doi.org/10.3390/catal15090819
Chicago/Turabian StyleWang, Xiang, Xidan Zhang, Yifei Qu, Tian Liu, Juejing Luo, Ting Long, Liang Wu, Chong Tian, and Yu Hu. 2025. "Bandgap-Tunable ZnxCd1−xS Solid Solutions for Effective Photocatalytic Degradation of Norfloxacin Under Visible Light and Natural Sunlight" Catalysts 15, no. 9: 819. https://doi.org/10.3390/catal15090819
APA StyleWang, X., Zhang, X., Qu, Y., Liu, T., Luo, J., Long, T., Wu, L., Tian, C., & Hu, Y. (2025). Bandgap-Tunable ZnxCd1−xS Solid Solutions for Effective Photocatalytic Degradation of Norfloxacin Under Visible Light and Natural Sunlight. Catalysts, 15(9), 819. https://doi.org/10.3390/catal15090819





