Oxygen Vacancy-Engineered Ni:Co3O4/Attapulgite Photothermal Catalyst from Recycled Spent Lithium-Ion Batteries for Efficient CO2 Reduction
Abstract
1. Introduction
2. Results and Discussion
2.1. XRD Analysis
2.2. TEM Analysis
2.3. UV–Vis Analysis
2.4. Thermal Imaging Analysis
2.5. XPS Analysis
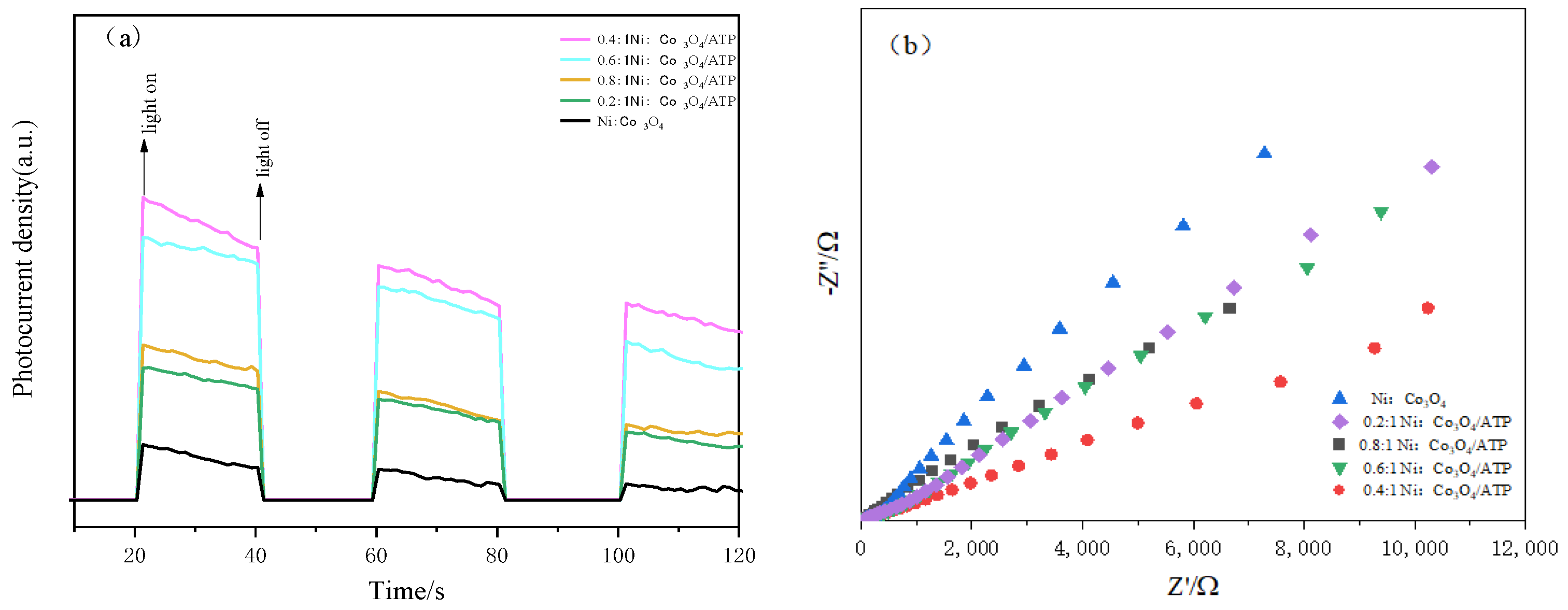
2.6. Electrochemical Analysis
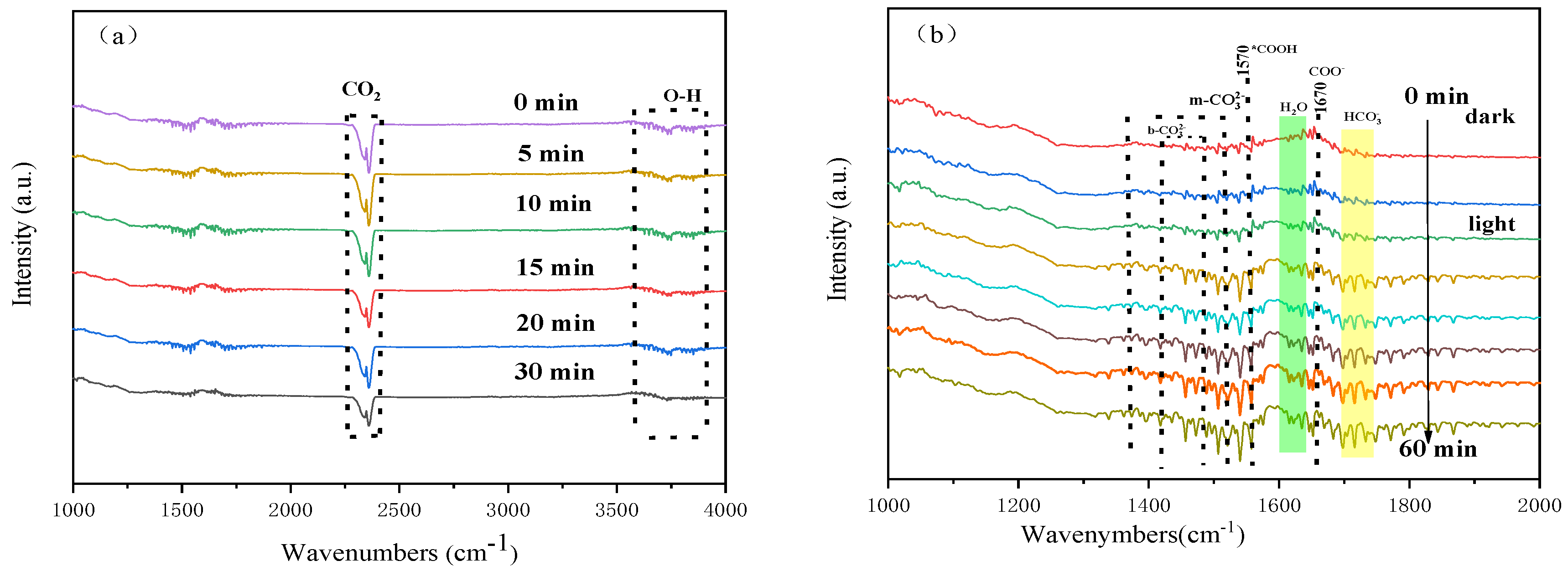
2.7. In Situ DRIFTS Analysis
2.8. EPR Analysis
2.9. Photothermal CO2 Reduction Performance
2.10. Mechanism of Photothermal Catalytic CO2 Reduction
3. Experimental Section
3.1. Materials
3.2. Recovery of Valuable Metals from Spent LIBs
3.3. Synthesis of Ni:Co3O4/ATP Composite
3.4. Material Characterization
3.5. Photocatalytic CO2 Reduction Experiment
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Zeng, X.; Chen, H.; Liu, Y.; Fang, W.; Huang, Z.; Zhao, L. Photo-thermal coupling-mediated enhancement in CO2 conversion: Key role of thermal effect and cobalt valence change-regulated electron-transfer orientation. J. Colloid. Interface Sci. 2025, 686, 733–742. [Google Scholar] [CrossRef]
- Li, M.; Zhang, S.; Li, L.; Han, J.; Zhu, X.; Ge, Q.; Wang, H. Construction of Highly Active and Selective Polydopamine Modified Hollow ZnO/Co3O4 p-n Heterojunction Catalyst for Photocatalytic CO2 Reduction. ACS Sustain. Chem. Eng. 2020, 8, 11465–11476. [Google Scholar] [CrossRef]
- Wang, Y.; Zheng, K.; Hu, X.; Zhou, W.; Wei, X.; Zhao, Y. Y2O3 promoted Co3O4 catalyst for catalytic decomposition of N2O. Mol. Catal. 2019, 470, 104–111. [Google Scholar] [CrossRef]
- Zheng, Y.-L.; Zhou, L.; Sun, X.-C.; Zhang, Q.; Yin, H.-J.; Du, P.; Yang, X.-F.; Zhang, Y.-W. Visible-Light Driven CO2 Reduction to CO by Co3O4 Supported on Tungsten Oxide. J. Phys. Chem. C 2022, 126, 3017–3028. [Google Scholar] [CrossRef]
- Ren, J.-T.; Zheng, Y.-L.; Yuan, K.; Zhou, L.; Wu, K.; Zhang, Y.-W. Self-templated synthesis of Co3O4 hierarchical nanosheets from a metal–organic framework for efficient visible-light photocatalytic CO2 reduction. Nanoscale 2020, 12, 755–762. [Google Scholar] [CrossRef]
- Gu, Y.; Ding, J.; Tong, X.; Yao, H.; Yang, R.; Zhong, Q. Photothermal catalyzed hydrogenation of carbon dioxide over porous nanosheet Co3O4. J. CO2 Util. 2022, 61, 102003. [Google Scholar] [CrossRef]
- Leal, V.M.; Magnago, L.B.; dos Santos, G.F.; Ferreira, R.Q.; Ferreira, S.A.; Lelis, M.F.; Freitas, M.B. Efficient recycling of Co3O4 from spent lithium-ion batteries and its application as an electrochemical sensor and photocatalyst. Sustain. Mater. Technol. 2023, 37, e00688. [Google Scholar] [CrossRef]
- Hossain, R.; Sahajwalla, V. Microrecycled Co3O4 from waste lithium-ion battery: Synthesis, characterisation and implication in environmental application. J. Environ. Chem. Eng. 2022, 10, 107858. [Google Scholar] [CrossRef]
- Assefi, M.; Maroufi, S.; Yamauchi, Y.; Sahajwalla, V. Core–Shell Nanocatalysts of Co3O4 and NiO Shells from New (Discarded) Resources: Sustainable Recovery of Cobalt and Nickel from Spent Lithium-Ion Batteries, Ni–Cd Batteries, and LCD Panel. ACS Sustain. Chem. Eng. 2019, 7, 19005–19014. [Google Scholar] [CrossRef]
- Guan, D.; Shi, C.; Xu, H.; Gu, Y.; Zhong, J.; Sha, Y.; Hu, Z.; Ni, M.; Shao, Z. Simultaneously mastering operando strain and reconstruction effects via phase-segregation strategy for enhanced oxygen-evolving electrocatalysis. J. Energy Chem. 2023, 82, 572–580. [Google Scholar] [CrossRef]
- Zhang, H.; Shi, J.; Han, C.; Song, Z.; Xiao, Y.; Li, X. Ultrasound-induced construction of CuxCo3−xO4/attapulgite for catalytic degradation of toluene. Catalysts 2025, 15, 252. [Google Scholar] [CrossRef]
- Yang, F.; Lu, Y.; Liu, M.; Yang, S.; Tu, W.; Zhang, W.; Zhu, C.; Guo, Z.; Yuan, A. Carbon-framework-encapsulated CoMn2O4 spinel derived from electrospun nanofiber coupling via the photothermal approach reinforces PMS activation to eliminate 2, 4-dichlorophenol. Mater. Chem. Front. 2022, 6, 2810–2825. [Google Scholar] [CrossRef]
- Qin, S.; Ge, C.; Kong, X.; Fu, M.; Zhuang, Z.; Li, X. Photothermal catalytic reduction of CO2 by cobalt silicate heterojunction constructed from clay minerals. Catalysts 2023, 13, 32. [Google Scholar] [CrossRef]
- Ke, Y.; Liang, Q.; Zhao, S.; Zhang, Z.; Li, X.; Li, Z. In situ self-assembled ZIF-67/MIL-125-derived Co3O4/TiO2 p–n heterojunctions for enhanced photocatalytic CO2 reduction. Inorg. Chem. 2022, 61, 2652–2661. [Google Scholar] [CrossRef]
- Li, X.; Wang, X.; Nan, Y.; Sun, Y.; Xu, H.; Chi, L.; Huang, Y.; Duan, J.; Hou, B. Effect of Co3O4/TiO2 heterojunction photoanode with enhanced photocathodic protection on 304 stainless steel under visible light. Colloids Surf. A Physicochem. Eng. Asp. 2023, 664, 131150. [Google Scholar] [CrossRef]
- Zhang, H.; Guan, D.; Gu, Y.; Xu, H.; Wang, C.; Shao, Z.; Guo, Y. Tuning synergy between nickel and iron in Ruddlesden–Popper perovskites through controllable crystal dimensionalities towards enhanced oxygen-evolving activity and stability. Carbon. Energy 2024, 6, e465. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, X.; Yan, Z.; Xu, C.; Zhang, W.; Ban, H.; Li, C. Activation reconstructing CuZnO/SiO2 catalyst for CO2 hydrogenation. J. Catal. 2022, 412, 10–20. [Google Scholar] [CrossRef]
- Wang, C.; Jiao, L.; Ji, P. Interfacial synthesis of Co-based ultrathin nanosheets with NaF crystal as template and inducer: Fluorine doped Co(OH)2, porous fluorine doped Co3O4 and porous Co3O4. Appl. Surf. Sci. 2022, 589, 152923. [Google Scholar] [CrossRef]
- Zuo, S.; Qin, S.; Xue, B.; Xu, R.; Shi, H.; Lu, X.; Yao, C.; Gui, H.; Li, X. Development of Plasmonic Attapulgite/Co(Ti)Ox Nanocomposite Using Spent Batteries toward Photothermal Reduction of CO2. Molecules 2024, 29, 2865. [Google Scholar] [CrossRef]
- Cao, G.; Xing, H.; Gui, H.; Yao, C.; Chen, Y.; Chen, Y.; Li, X. Plasmonic quantum dots modulated nano-mineral toward photothermal reduction of CO2 coupled with biomass conversion. Nano Res. 2024, 17, 5061–5072. [Google Scholar] [CrossRef]
- Navarro-Jaén, S.; Virginie, M.; Bonin, J.; Robert, M.; Wojcieszak, R.; Khodakov, A.Y. Highlights and challenges in the selective reduction of carbon dioxide to methanol. Nat. Rev. Chem. 2021, 5, 564–579. [Google Scholar] [CrossRef] [PubMed]
- Dong, X.; Jing, X.; Mu, Y.; Yu, Y.; Miao, C.; Meng, C.; Huang, C.; Zhang, Y. Rational design of double-sandwich-like C@Co, CoO/Co2SiO4/rGO architectures boost electrochemical performances of Co2SiO4 for energy storage devices. Chem. Eng. J. 2022, 431, 133277. [Google Scholar] [CrossRef]










Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shi, J.; Xiao, Y.; Yu, M.; Li, X. Oxygen Vacancy-Engineered Ni:Co3O4/Attapulgite Photothermal Catalyst from Recycled Spent Lithium-Ion Batteries for Efficient CO2 Reduction. Catalysts 2025, 15, 732. https://doi.org/10.3390/catal15080732
Shi J, Xiao Y, Yu M, Li X. Oxygen Vacancy-Engineered Ni:Co3O4/Attapulgite Photothermal Catalyst from Recycled Spent Lithium-Ion Batteries for Efficient CO2 Reduction. Catalysts. 2025; 15(8):732. https://doi.org/10.3390/catal15080732
Chicago/Turabian StyleShi, Jian, Yao Xiao, Menghan Yu, and Xiazhang Li. 2025. "Oxygen Vacancy-Engineered Ni:Co3O4/Attapulgite Photothermal Catalyst from Recycled Spent Lithium-Ion Batteries for Efficient CO2 Reduction" Catalysts 15, no. 8: 732. https://doi.org/10.3390/catal15080732
APA StyleShi, J., Xiao, Y., Yu, M., & Li, X. (2025). Oxygen Vacancy-Engineered Ni:Co3O4/Attapulgite Photothermal Catalyst from Recycled Spent Lithium-Ion Batteries for Efficient CO2 Reduction. Catalysts, 15(8), 732. https://doi.org/10.3390/catal15080732






