Naproxen Degradation Using NiO Synthesized via Ultrasonic Spray Pyrolysis on Ni–Fe Foam by Ozone
Abstract
1. Introduction
2. Results and Discussion
2.1. Evaluation of the Catalytic Activity of NiFeF and NiO/NiFeF in NPX Ozonation
2.2. Crystalline Structure of NiFeF and NiO/NiFeF
2.3. SEM Analysis
2.4. XPS Analysis
2.5. Catalytic Reaction Pathways
2.6. Identification of ROS Formed in Catalytic Ozonation
2.7. Catalyst Stability Study
3. Experimental Section
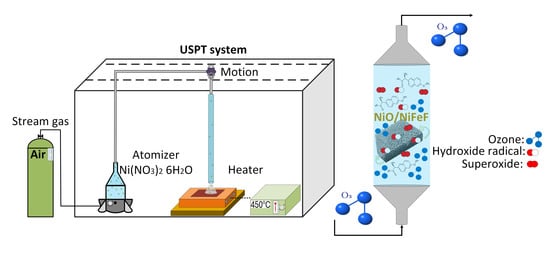
3.1. Preparation of the Catalysts
3.2. Catalyst Characterization
3.3. Ozonation Procedure
3.4. Analytical Methods
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Barrios-Estrada, C.; de Jesús Rostro-Alanis, M.; Muñoz-Gutiérrez, B.D.; Iqbal, H.M.N.; Kannan, S.; Parra-Saldívar, R. Emergent contaminants: Endocrine disruptors and their laccase-assisted degradation—A review. Sci. Total Environ. 2018, 612, 1516–1531. [Google Scholar] [CrossRef] [PubMed]
- Wada, O.Z.; Olawade, D.B. Recent occurrence of pharmaceuticals in freshwater, emerging treatment technologies, and future considerations: A review. Chemosphere 2025, 374, 144153. [Google Scholar] [CrossRef]
- Komorowska-Kaufman, M.; Zembrzuska, J. Application of oxidation processes in wastewater quaternary treatment for organic compounds, antibiotics and nonsteroidal anti-inflammatory drugs removal and disinfection. Desalination Water Treat. 2025, 321, 101059. [Google Scholar] [CrossRef]
- Wojcieszyńska, D.; Guzik, U. Naproxen in the environment: Its occurrence, toxicity to nontarget organisms and biodegradation. Appl. Microbiol. Biotechnol. 2020, 104, 1849–1857. [Google Scholar] [CrossRef]
- Bindu, S.; Mazumder, S.; Bandyopadhyay, U. Non-steroidal anti-inflammatory drugs (NSAIDs) and organ damage: A current perspective. Biochem. Pharmacol. 2020, 180, 114147. [Google Scholar] [CrossRef]
- Moreno Ríos, A.L.; Gutierrez-Suarez, K.; Carmona, Z.; Ramos, C.G.; Silva Oliveira, L.F. Pharmaceuticals as emerging pollutants: Case naproxen an overview. Chemosphere 2022, 291, 132822. [Google Scholar] [CrossRef]
- Li, Z.; Liu, G.; Su, Q.; Lv, C.; Jin, X.; Wen, X. UV-Induced Photodegradation of Naproxen Using a Nano γ-FeOOH Composite: Degradation Kinetics and Photocatalytic Mechanism. Front. Chem. 2019, 7, 847. [Google Scholar] [CrossRef] [PubMed]
- Ray, S.K.; Dhakal, D.; Lee, S.W. Rapid degradation of naproxen by AgBr-α-NiMoO4 composite photocatalyst in visible light: Mechanism and pathways. Chem. Eng. J. 2018, 347, 836–848. [Google Scholar] [CrossRef]
- Arany, E.; Szabó, R.K.; Apáti, L.; Alapi, T.; Ilisz, I.; Mazellier, P.; Dombi, A.; Gajda-Schrantz, K. Degradation of naproxen by UV, VUV photolysis and their combination. J. Hazard. Mater. 2013, 262, 151–157. [Google Scholar] [CrossRef]
- Liu, Y.; Tang, Y.; Wu, Y.; Feng, L.; Zhang, L. Degradation of naproxen in chlorination and UV/chlorine processes: Kinetics and degradation products. Environ. Sci. Pollut. Res. 2019, 26, 34301–34310. [Google Scholar] [CrossRef]
- Karaca, M.; Kıranşan, M.; Karaca, S.; Khataee, A.; Karimi, A. Sonocatalytic removal of naproxen by synthesized zinc oxide nanoparticles on montmorillonite. Ultrason. Sonochem. 2016, 31, 250–256. [Google Scholar] [CrossRef]
- Dulova, N.; Kattel, E.; Trapido, M. Degradation of naproxen by ferrous ion-activated hydrogen peroxide, persulfate and combined hydrogen peroxide/persulfate processes: The effect of citric acid addition. Chem. Eng. J. 2017, 318, 254–263. [Google Scholar] [CrossRef]
- Aguilar, C.M.; Vazquez-Arenas, J.; Castillo-Araiza, O.O.; Rodríguez, J.L.; Chairez, I.; Salinas, E.; Poznyak, T. Improving ozonation to remove carbamazepine through ozone-assisted catalysis using different NiO concentrations. Environ. Sci. Pollut. Res. 2020, 27, 22184–22194. [Google Scholar] [CrossRef]
- Biard, P.-F.; Werghi, B.; Soutrel, I.; Orhand, R.; Couvert, A.; Denicourt-Nowicki, A.; Roucoux, A. Efficient catalytic ozonation by ruthenium nanoparticles supported on SiO2 or TiO2: Towards the use of a non-woven fiber paper as original support. Chem. Eng. J. 2016, 289, 374–381. [Google Scholar] [CrossRef]
- Yu, G.; Wang, Y.; Cao, H.; Zhao, H.; Xie, Y. Reactive Oxygen Species and Catalytic Active Sites in Heterogeneous Catalytic Ozonation for Water Purification. Environ. Sci. Technol. 2020, 54, 5931–5946. [Google Scholar] [CrossRef]
- Jin, X.; Wu, C.; Fu, L.; Tian, X.; Wang, P.; Zhou, Y.; Zuo, J. Development, dilemma and potential strategies for the application of nanocatalysts in wastewater catalytic ozonation: A review. J. Environ. Sci. 2023, 124, 330–349. [Google Scholar] [CrossRef]
- Aguilar-Melo, C.M.; Rodríguez, J.L.; Chairez, I.; Salgado, I.; Andraca Adame, J.A.; Galaviz-Pérez, J.A.; Vazquez-Arenas, J.; Poznyak, T. Enhanced Naproxen Elimination in Water by Catalytic Ozonation Based on NiO Films. Catalysts 2020, 10, 884. [Google Scholar] [CrossRef]
- Huang, Y.; Luo, M.; Xu, Z.; Zhang, D.; Li, L. Catalytic ozonation of organic contaminants in petrochemical wastewater with iron-nickel foam as catalyst. Sep. Purif. Technol. 2019, 211, 269–278. [Google Scholar] [CrossRef]
- Feng, C.; Diao, P. Nickel foam supported NiFe2O4-NiO hybrid: A novel 3D porous catalyst for efficient heterogeneous catalytic ozonation of azo dye and nitrobenzene. Appl. Surf. Sci. 2021, 541, 148683. [Google Scholar] [CrossRef]
- Khalik, W.F.; Ho, L.N.; Ong, S.A.; Wong, Y.S.; Yusoff, N.A.; Lee, S.L. Revealing the influences of functional groups in azo dyes on the degradation efficiency and power output in solar photocatalytic fuel cell. J. Environ. Health Sci. Eng. 2020, 18, 769–777. [Google Scholar] [CrossRef]
- Li, J.; Ban, X.; Sun, A.; Lai, H.; Li, H.; Yang, Z.; Pan, P.; He, J.; Zhang, R. A highly sensitive non-enzymatic glucose sensor based on coral-like Cu/nickel foam bimetallic structure. Mater. Today Commun. 2025, 46, 112791. [Google Scholar] [CrossRef]
- Zhu, L.; Zhou, S.; Cheng, H.; Ma, J.; Imanova, G.; Komarneni, S. Cu2S/Ni3S2 nanosheets combined with nickel foam substrate for efficient catalytic ozonation of p-nitrophenol in wastewater. J. Environ. Chem. Eng. 2024, 12, 113591. [Google Scholar] [CrossRef]
- Zhang, R.; Ran, T.; Cao, Y.; Zhang, Q.; Dong, F.; Yang, G.; Zhou, Y. Surface Hydrogen Atoms Promote Oxygen Activation for Solar Light-Driven NO Oxidization over Monolithic α-Ni(OH)2/Ni Foam. Environ. Sci. Technol. 2020, 54, 16221–16230. [Google Scholar] [CrossRef]
- Feng, C.; Qiu, S.; Diao, P. Copper Foam-Supported CuxO@Fe2O3 Core–Shell Nanotubes: An Efficient Ozonation Catalyst for Degradation of Organic Pollutants. ACS EST Water 2023, 3, 465–474. [Google Scholar] [CrossRef]
- Zhou, S.; Zhu, F.; Cheng, H.; Komarneni, S.; Ma, J. In-situ growth of Ni3S2@Mo2S3 catalyst on Mo-Ni foam for degradation of p-nitrophenol with a good synergetic effect by using ozone. J. Environ. Chem. Eng. 2023, 11, 111477. [Google Scholar] [CrossRef]
- Zhan, S.; Hu, X.; Lou, Z.; Zhu, J.; Xiong, Y.; Tian, S. In-situ growth of defect-enriched NiO film on nickel foam (NF@NiO) monolithic catalysts for ozonation of gaseous toluene. J. Alloys Compd. 2022, 893, 162160. [Google Scholar] [CrossRef]
- Wang, H.; Peng, L.; Li, G.; Liu, H.; Liang, Z.; Zhao, H.; An, T. Enhanced catalytic ozonation inactivation of bioaerosols by MnO2/Ni foam with abundant oxygen vacancies and O3 at atmospheric concentration. Appl. Catal. B Environ. Energy 2024, 344, 123675. [Google Scholar] [CrossRef]
- Zhou, S.; Cheng, H.; Komarneni, S.; Ma, J. Enhanced heterogeneous catalytic ozonation to degrade p-nitrophenol by Co-doped Ni3S2/NF nanosheets. Colloids Surf. A Physicochem. Eng. Asp. 2024, 689, 133717. [Google Scholar] [CrossRef]
- Yao, W.; Yang, T.; Liu, D.; Liu, F.; Zhang, L.; Cheng, C.; Hu, J.; Huang, H. Preparation of LMO@FC catalysts and degradation of tetracycline by catalytic ozonation. J. Alloys Compd. 2024, 1004, 175848. [Google Scholar] [CrossRef]
- Tian, S.; Zhan, S.; Lou, Z.; Zhu, J.; Feng, J.; Xiong, Y. Electrodeposition synthesis of 3D-NiO1−δ flowers grown on Ni foam monolithic catalysts for efficient catalytic ozonation of VOCs. J. Catal. 2021, 398, 1–13. [Google Scholar] [CrossRef]
- Shen, T.; Wang, P.; Shi, F.; Xu, P.; Zhang, G. Metal foam-based functional materials application in advanced oxidation and reduction processes for water remediation: Design, Mechanisms, and Prospects. Chem. Eng. J. 2024, 500, 156825. [Google Scholar] [CrossRef]
- Rodríguez, J.A.-O.; Valenzuela, M.A. Ni-based catalysts used in heterogeneous catalytic ozonation for organic pollutant degradation: A minireview. Environ. Sci. Pollut. Res. 2022, 29, 84056–84075. [Google Scholar] [CrossRef]
- Gubóová, A.; Oriňaková, R.; Strečková, M.; Paračková, M.; Petruš, O.; Plešingerová, B.; Mičušík, M. Iron-nickel metal foams modified by phosphides as robust catalysts for a hydrogen evolution reaction. Mater. Today Chem. 2023, 34, 101778. [Google Scholar] [CrossRef]
- Krishnamurthy, P.; Maiyalagan, T.; Panomsuwan, G.; Jiang, Z.; Rahaman, M. Iron-Doped Nickel Hydroxide Nanosheets as Efficient Electrocatalysts in Electrochemical Water Splitting. Catalysts 2023, 13, 1095. [Google Scholar] [CrossRef]
- Solís, C.; Toldra-Reig, F.; Balaguer, M.; Somacescu, S.; Garcia-Fayos, J.; Palafox, E.; Serra, J.M. Mixed Ionic–Electronic Conduction in NiFe2O4–Ce0.8Gd0.2O2−δ Nanocomposite Thin Films for Oxygen Separation. ChemSusChem 2018, 11, 2638–2836. [Google Scholar] [CrossRef]
- Biesinger, M.C.; Payne, B.P.; Grosvenor, A.P.; Lau, L.W.; Gerson, A.R.; Smart, R.S.C. Resolving surface chemical states in XPS analysis of first row transition metals, oxides and hydroxides: Cr, Mn, Fe, Co and Ni. Appl. Surf. Sci. 2011, 257, 2717–2730. [Google Scholar] [CrossRef]
- Li, G.; Li, R.; Zhou, W. A Wire-Shaped Supercapacitor in Micrometer Size Based on Fe3O4 Nanosheet Arrays on Fe Wire. Nano-Micro Lett. 2017, 9, 46. [Google Scholar] [CrossRef]
- Mullet, M.; Khare, V.; Ruby, C. XPS study of Fe(II)—Fe(III) (oxy)hydroxycarbonate green rust compounds. Surf. Interface Anal. 2008, 40, 125–892. [Google Scholar] [CrossRef]
- Ding, T.; Lin, K.; Yang, B.; Yang, M.; Li, J.; Li, W.; Gan, J. Biodegradation of naproxen by freshwater algae Cymbella sp. and Scenedesmus quadricauda and the comparative toxicity. Bioresour. Technol. 2017, 238, 164–173. [Google Scholar] [CrossRef] [PubMed]
- Pourakbar, M.; Ghanbari, F.; Khavar, A.H.C.; Khashij, M.; Mehralian, M.; Behnami, A.; Satari, M.; Mahdaviapour, M.; Oghazyan, A.; Aghayani, E. Comparative study of naproxen degradation via integrated UV/O3/PMS process: Degradation products, reaction pathways, and toxicity assessment. Korean J. Chem. Eng. 2022, 39, 2725–2735. [Google Scholar] [CrossRef]
- Hayyan, M.; Hashim, M.A.; AlNashef, I.M. Superoxide Ion: Generation and Chemical Implications. Chem. Rev. 2016, 116, 3029–3085. [Google Scholar] [CrossRef]
- Guo, Y.; Zhang, Y.; Yu, G.; Wang, Y. Revisiting the role of reactive oxygen species for pollutant abatement during catalytic ozonation: The probe approach versus the scavenger approach. Appl. Catal. B Environ. 2021, 280, 119418. [Google Scholar] [CrossRef]
- Mathon, B.; Coquery, M.; Liu, Z.; Penru, Y.; Guillon, A.; Esperanza, M.; Miège, C.; Choubert, J.M. Ozonation of 47 organic micropollutants in secondary treated municipal effluents: Direct and indirect kinetic reaction rates and modelling. Chemosphere 2021, 262, 127969. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.; Kovalova, L.; McArdell, C.S.; von Gunten, U. Prediction of micropollutant elimination during ozonation of a hospital wastewater effluent. Water Res. 2014, 64, 134–148. [Google Scholar] [CrossRef]
- Wang, Y.; Xie, Y.; Sun, H.; Xiao, J.; Cao, H.; Wang, S. Efficient Catalytic Ozonation over Reduced Graphene Oxide for p-Hydroxylbenzoic Acid (PHBA) Destruction: Active Site and Mechanism. ACS Appl. Mater. Interfaces 2016, 8, 9710–9720. [Google Scholar] [CrossRef]
- Jary, W.G.; Ganglberger, T.; Pöchlauer, P.; Falk, H. Generation of Singlet Oxygen from Ozone Catalysed by Phosphinoferrocenes. Monatshefte Für Chem./Chem. Mon. 2005, 136, 537–541. [Google Scholar] [CrossRef]
- Feng, X.; Sun, D. Degradation characteristics of refractory organic matter in naproxen pharmaceutical secondary effluent using vacuum ultraviolet–ozone treatment. J. Hazard. Mater. 2023, 459, 132056. [Google Scholar] [CrossRef]
- Huaccallo-Aguilar, Y.; Álvarez-Torrellas, S.; Gil, M.V.; Larriba, M.; García, J. Insights of emerging contaminants removal in real water matrices by CWPO using a magnetic catalyst. J. Environ. Chem. Eng. 2021, 9, 106321. [Google Scholar] [CrossRef]
- Rasool, R.T.; Ashraf, G.A.; Fadhali, M.M.; Al-Sulaimi, S.; Ghernaout, D.; El Jery, A.; Aldrdery, M.; Elkhaleefa, A.; Hassan, N.; Ajmal, Z.; et al. Peroxymonosulfate-based photodegradation of naproxen by stimulating (Mo, V, and Zr)-carbide nanoparticles. J. Water Process Eng. 2023, 54, 104027. [Google Scholar] [CrossRef]
- He, L.; Zhi, J.; Li, H.; Jia, Y.; Gao, Q.; Wang, J.; Xu, Y.; Li, X. Peroxymonosulfate activation by magnetic NiCo layered double hydroxides for naproxen degradation. Colloids Surf. A Physicochem. Eng. Asp. 2022, 642, 128696. [Google Scholar] [CrossRef]
- Li, L.; Wang, Y.; Gao, Y.; Huang, Y.; Liang, Y. Theoretical evidence of enhanced interaction between H2O and O3 leads to improved performance of catalytic ozonation with copper doping α-FeOOH. Surf. Interfaces 2024, 44, 103772. [Google Scholar] [CrossRef]
- Sheydaei, M.; Haseli, A.; Ayoubi-Feiz, B.; Vatanpour, V. MoS2/N-TiO2/Ti mesh plate for visible-light photocatalytic ozonation of naproxen and industrial wastewater: Comparative studies and artificial neural network modeling. Environ. Sci. Pollut. Res. 2022, 29, 22454–22468. [Google Scholar] [CrossRef] [PubMed]












| No. | Condensed Formula | Name | Structure | MM g/mol | O3 | NiFeF | NiO/NiFeF |
|---|---|---|---|---|---|---|---|
| 1. | C14H14O3 | Naproxen |  | 230.26 | X | X | X |
| 2. | C14H18O4 | (2S)-2-(3-(hydroxymethyl)-4-[(1E)-2-methoxyprop-1-en-1-yl]phenyl)propanoic acid |  | 250.29 | X | X | X |
| 3. | C13H12O5 | (2R)-2-(5,6-dihydroxynaphthalen-2-yl)-3-hydroxypropanoic acid |  | 248.23 | X | ||
| 4. | C15H16O3 | 2-(5,6-dimethylnaphthalen-2-yl)-2-hydroxypropanoic acid |  | 244.29 | X | ||
| 5. | C14H16O4 | (2S)-2-(8-hydroxy-6-methoxynaphthalen-2-yl)propane-1,1-diol |  | 248.09 | X | ||
| 6. | C13H18O4 | (2S)-2-[3-(hydroxymethyl)-4-(2-methoxyethyl)phenyl]propanoic acid |  | 238.28 | X | X | |
| 7. | C10H10O6 | 5-[(1S)-1-carboxy-2-hydroxyethyl]-2-hydroxybenzoic acid |  | 226.18 | X | ||
| 8. | C13H18O3 | (2S)-2-[4-(2-methoxyethyl)-3-methylphenyl]propanoic acid |  | 222.15 | X | X | X |
| 9. | C13H12O3 | (2S)-2-(6-hydroxynaphthalen-2-yl)propanoic acid |  | 216.24 | X | ||
| 10. | C13H16O2 | (2S)-2-[4-ethyl-3-(1-hydroxyethen-1-yl)phenyl]propanal |  | 204.08 | X | X | |
| 11. | C9H10O5 | 2-(3,4-dihydroxyphenyl)-2-hydroxypropanoic acid |  | 198.17 | X | ||
| 12. | C10H14O4 | 2-[3-hydroxy-4-(hydroxymethyl)phenyl]propane-1,1-diol |  | 198.04 | X | ||
| 13. | C11H16O3 | 2-(4-ethyl-3-hydroxyphenyl)propane-1,1-diol |  | 196.25 | X | ||
| 14. | C11H14O3 | (2S)-2-[4-(2-hydroxyethyl)phenyl]propanoic acid |  | 194.2 | X | X | X |
| 15. | C9H12O4 | 4-[(2S)-1,1-dihydroxypropan-2-yl]benzene-1,2-diol |  | 184.15 | X | ||
| 16. | C9H10O4 | (2S)-2-(3,4-dihydroxyphenyl)propanoic acid |  | 182.17 | X | ||
| 17. | C10H12O3 | (2S)-2-(3-hydroxy-4-methylphenyl)propanoic acid |  | 180.16 | X | X | X |
| 18. | C10H8O3 | naphthalene-2,3,6-triol |  | 176.17 | X | X | |
| 19. | C12H12O | 6-ethylnaphthalen-2-ol |  | 172.23 | X | ||
| 20. | C9H10O3 | (2S)-2-(3,4-dihydroxyphenyl)propanal |  | 166.07 | X | ||
| 21. | C11H10O | 6-methylnaphthalen-2-ol |  | 158.2 | X | X | |
| 22. | C7H6O4 | 3,4-dihydroxybenzoic acid |  | 154.12 | X | X | |
| 23. | C9H10O2 | 4-(prop-1-en-2-yl)benzene-1,2-diol |  | 150.99 | X | ||
| 24. | C11H10 | 2-methylnaphthalene |  | 142.20 | X | X | X |
| 25. | C8H10O2 | 4-ethylbenzene-1,2-diol |  | 138.10 | X | X | |
| 26. | C4H6O5 | (2S)-2-hydroxybutanedioic acid |  | 134.09 | X | ||
| 27. | C10H8 | naphthalene |  | 128.17 | X | ||
| 28. | C7H8O2 | 2-methylbenzene-1,4-diol |  | 124.14 | X | x | x |
| 29. | C7H6O2 | benzoic acid |  | 122.12 | X | ||
| 30. | C9H12 | 1-ethyl-4-methylbenzene |  | 120.19 | X | X | X |
| 31. | C6H6O2 | benzene-1,4-diol |  | 110.11 | X | X | X |
| 32. | C8H8 | ethenylbenzene |  | 104.15 | X | ||
| 33. | C5H10O2 | (3E)-pent-3-ene-1,4-diol |  | 102.13 | X | ||
| 34. | C5H8O2 | 2-methylidenebutanoic acid |  | 100.12 | X | X | |
| 35. | C2H2O4 | Oxalic acid |  | 90.03 | X | X | X |
| 36. | C3H4O3 | 2-oxopropanoic acid |  | 88.05 | X | X | |
| 37. | C5H10O | (2E)-pent-2-en-2-ol |  | 86.13 | X | ||
| 38. | C4H6O2 | 2-methylprop-2-enoic acid |  | 86.09 | X | X | |
| 39. | C6H6 | benzene |  | 78.11 | X | ||
| 40. | C2H4O3 | hydroxyacetic acid |  | 76.05 | X |
| Treatment | Catalyst | Mass (g) | NPX Concentration (mg⋅L−1) | pH | Ozone (mg⋅L−1) | Time (min) | TOC Removal, % | Ref | |
|---|---|---|---|---|---|---|---|---|---|
| AO | HCO | ||||||||
| HCO | NiO/NiFeF | 0.35 | 20 | 5.7 | 11 | 120 | 12 | 73 | This study |
| Vacuum ultraviolet ozone | - | - | 14.26–18.47 | 7.68–9.12 | 75 | 90 | 28.54 | 54.81 | [47] |
| Catalytic wet peroxide oxidation | Fe3O4/MWCNTs | 0.4 | 10 | 5.0 | - | 270 | - | 66 | [48] |
| Peroxymonosulfate-based photodegradation | (Mo, V, and Zr)-carbide nanoparticles | 0.75 | 10 | 3–7 | - | 60 | - | - | [49] |
| Peroxymonosulfate degradation | Fe3O4 @NiCo | 0.2 | 10 | - | 20 | 49.4 | [50] | ||
| Catalytic ozonation | Cu doped α-FeOOH | 0.3 | 20 | 7 | 7.4 | 25 | 34.7 | 40.8 | [51] |
| Visible-light photocatalytic ozonation | MoS2/N-TiO2/Ti (6 pieces) | 0.1 g N-TiO2, 0.1 g MoS2, 0.07 g Mg(NO3) | 3–12 | 4 | 3 L/min | 90 | - | - | [52] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Morejón Aguila, G.L.; Rodríguez Santillán, J.L.; Poznyak, T.; Cruz Narváez, Y.; Mendoza León, H.F.; Lartundo Rojas, L.; Ramos Torres, C.J.; Castro Arellano, J.J. Naproxen Degradation Using NiO Synthesized via Ultrasonic Spray Pyrolysis on Ni–Fe Foam by Ozone. Catalysts 2025, 15, 993. https://doi.org/10.3390/catal15100993
Morejón Aguila GL, Rodríguez Santillán JL, Poznyak T, Cruz Narváez Y, Mendoza León HF, Lartundo Rojas L, Ramos Torres CJ, Castro Arellano JJ. Naproxen Degradation Using NiO Synthesized via Ultrasonic Spray Pyrolysis on Ni–Fe Foam by Ozone. Catalysts. 2025; 15(10):993. https://doi.org/10.3390/catal15100993
Chicago/Turabian StyleMorejón Aguila, George Luis, Julia Liliana Rodríguez Santillán, Tatyana Poznyak, Yair Cruz Narváez, Héctor F. Mendoza León, Luis Lartundo Rojas, Claudia Jazmín Ramos Torres, and José J. Castro Arellano. 2025. "Naproxen Degradation Using NiO Synthesized via Ultrasonic Spray Pyrolysis on Ni–Fe Foam by Ozone" Catalysts 15, no. 10: 993. https://doi.org/10.3390/catal15100993
APA StyleMorejón Aguila, G. L., Rodríguez Santillán, J. L., Poznyak, T., Cruz Narváez, Y., Mendoza León, H. F., Lartundo Rojas, L., Ramos Torres, C. J., & Castro Arellano, J. J. (2025). Naproxen Degradation Using NiO Synthesized via Ultrasonic Spray Pyrolysis on Ni–Fe Foam by Ozone. Catalysts, 15(10), 993. https://doi.org/10.3390/catal15100993