Effect of the Heterovalent Sc3+ and Nb5+ Doping on Photoelectrochemical Behavior of Anatase TiO2
Abstract
1. Introduction
2. Results and Discussions
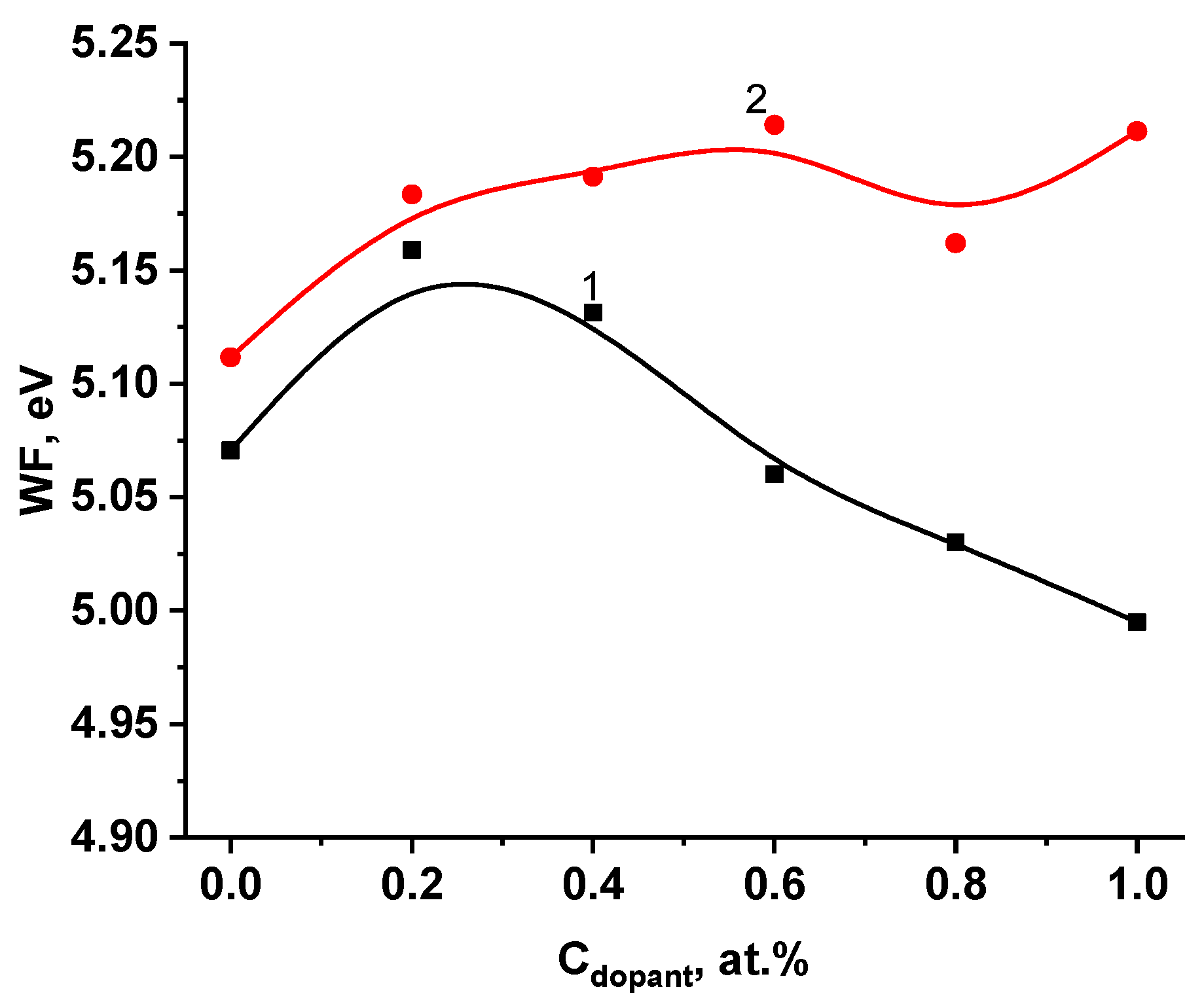
2.1. Physical–Chemical Characterization of the Doped Electrodes
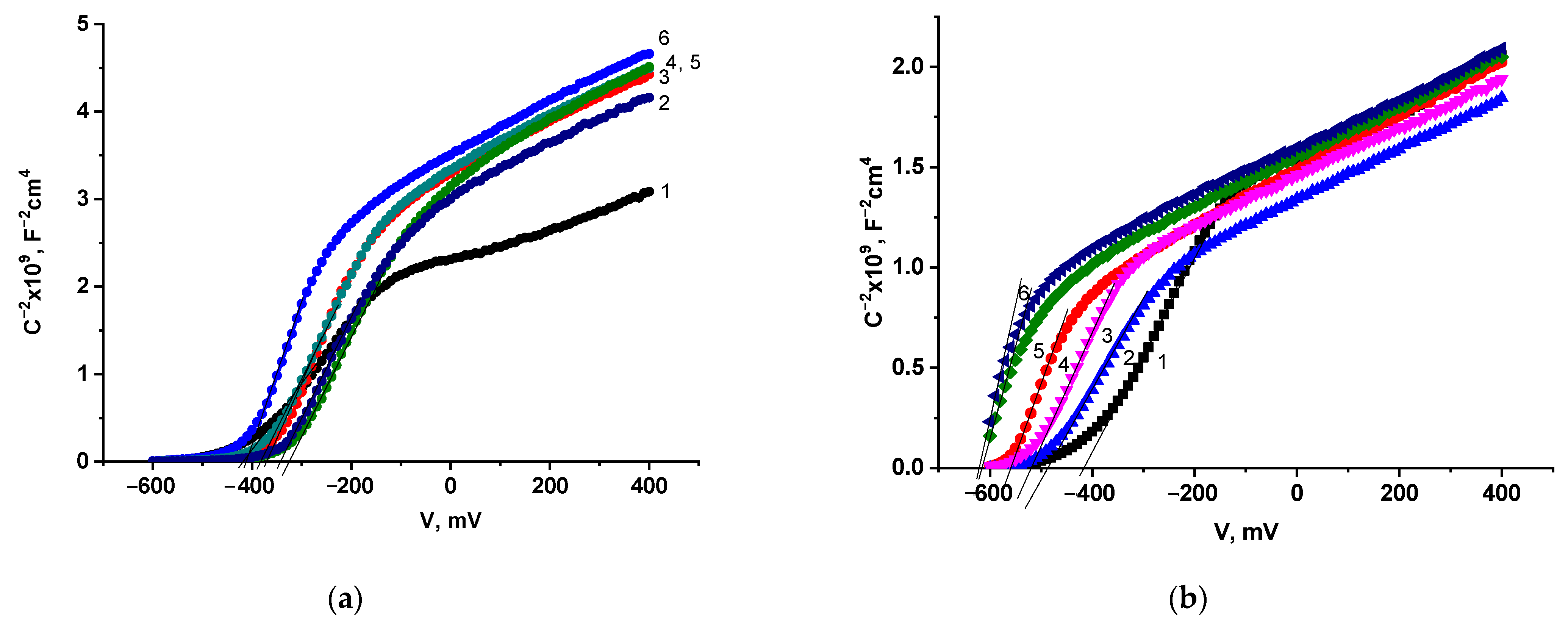
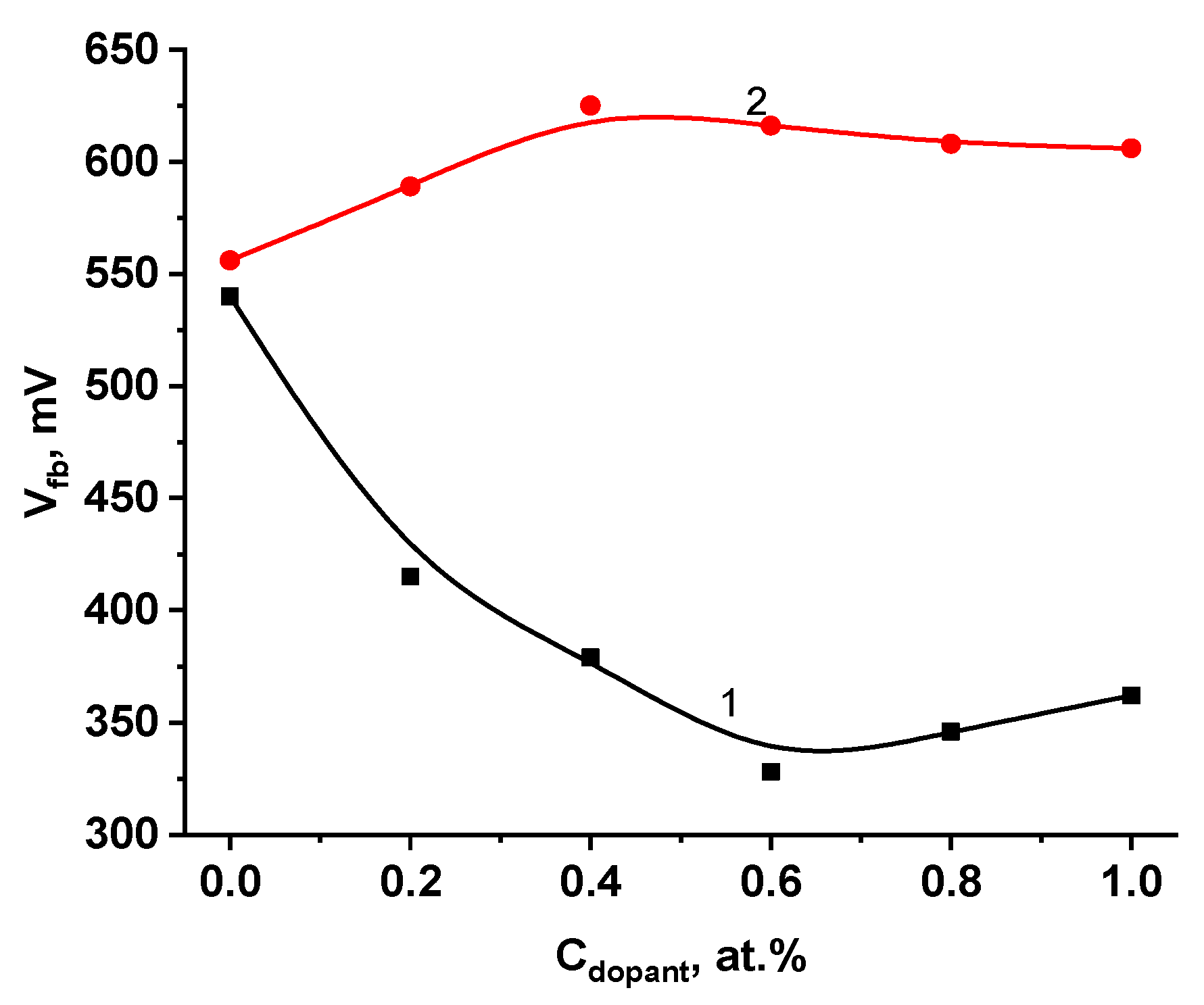
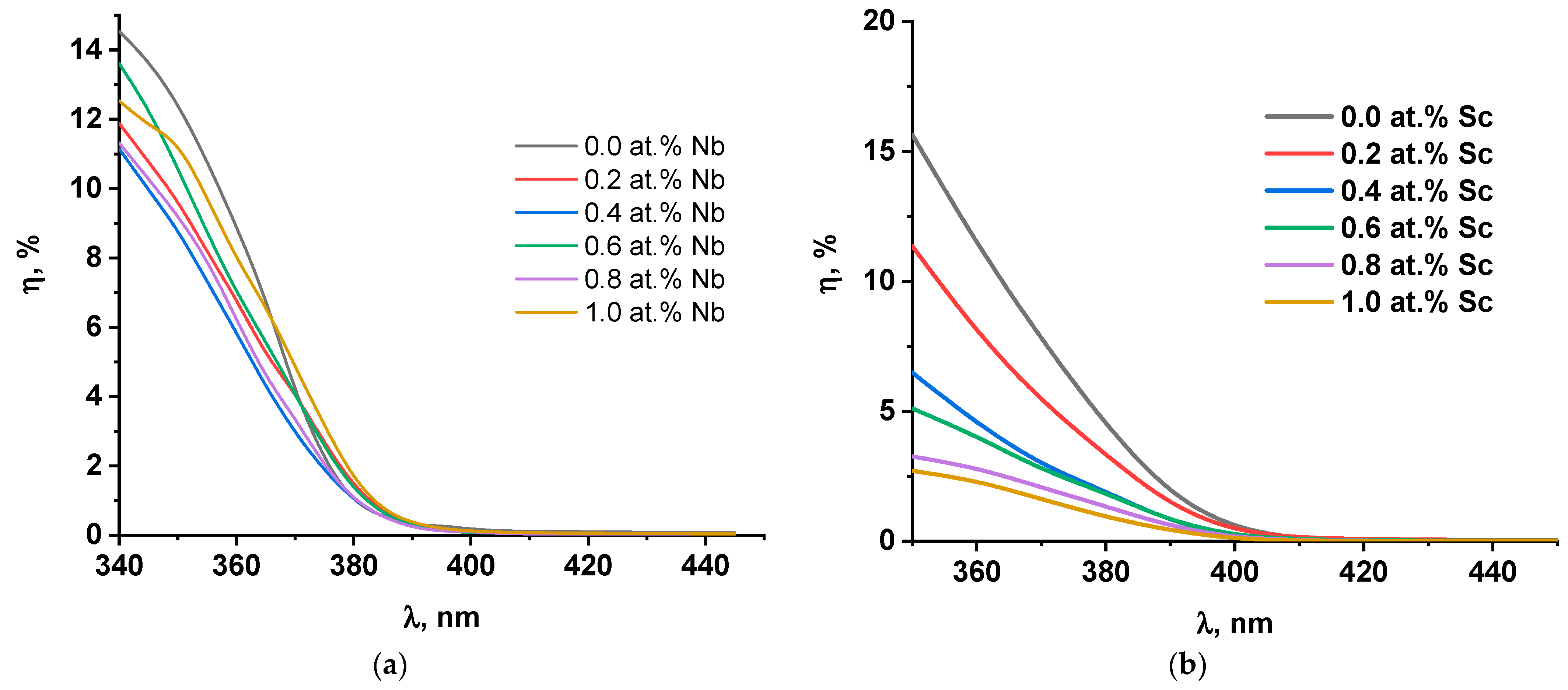
2.2. Photoelectrochemical Studies of the Doped Electrodes
3. Materials and Methods
3.1. Electrode Preparation
3.2. Sample Characterization
3.3. Photoelectrochemical Measurements
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Anpo, M. Applications of titanium oxide photocatalysts and unique second-generation TiO2 photocatalysts able to operate under visible light irradiation for the reduction of environmental toxins on a global scale. Stud. Surf. Sci. Catal. 2000, 130, 157–166. [Google Scholar] [CrossRef]
- Khaki, M.R.D.; Shafeeyan, M.S.; Raman, A.A.A.; Daud, W.M.A.W. Application of doped photocatalysts for organic pollutant degradation—A review. J. Environ. Manag. 2017, 198, 78–94. [Google Scholar] [CrossRef] [PubMed]
- Jeon, J.-P.; Kweon, D.H.; Jang, B.J.; Ju, M.J.; Baek, J.-B. Enhancing the photocatalytic activity of TiO2 catalysts. Adv. Sustain. Syst. 2020, 4, 2000197. [Google Scholar] [CrossRef]
- Schneider, J.; Matsuoka, M.; Takeuchi, M.; Zhang, J.; Horiuchi, Y.; Anpo, M.; Bahnemann, D.W. Understanding TiO2 photocatalysis: Mechanisms and materials. Chem. Rev. 2014, 114, 9919–9986. [Google Scholar] [CrossRef]
- Wolkenstein, T. The Electronic Theory of Catalysis on Semiconductors, 1st ed.; Pergamon Press: Oxford, UK, 1963; 169p. [Google Scholar]
- Wolkenstein, T. the electronic theory of photocatalytic reactions on semiconductors. Adv. Catal. 1973, 23, 157–208. [Google Scholar] [CrossRef]
- Hauffe, K.; Stechemesser, R. Zur Randschicht-Theorie der Adsorption und Katalyse an Halbleiter-Katalysatoren. In Electronic Phenomena in Chemisorption and Catalysis on Semiconductors. Symposium on Electronic Phenomena in Chemisorption and Catalysis on Semiconductors Held in Moscow, July 2–4, 1968, 1st ed.; Hauffe, K., Wolkenstein, T., Eds.; Walter De Gruyter & Co.: Berlin, Germany, 1969; Chapter 1; pp. 1–27. [Google Scholar] [CrossRef]
- Etacheri, V.; Di Valentin, C.; Schneider, J.; Bahnemann, D.; Pillai, S.C. Visible-light activation of TiO2 photocatalysts: Advances in theory and experiments. J. Photochem. Photobiol. 2015, 25, 1–29. [Google Scholar] [CrossRef]
- Kang, X.; Liu, S.; Dai, Z.; He, Y.; Song, X.; Tan, Z. Titanium dioxide: From engineering to applications. Catalysts 2019, 9, 191. [Google Scholar] [CrossRef]
- Fujishima, A.; Zhang, X.; Tryk, D.A. TiO2 photocatalysis and related surface phenomena. Surf. Sci. Rep. 2008, 63, 515–582. [Google Scholar] [CrossRef]
- Pan, J.W.; Li, C.; Zhao, Y.F.; Liu, R.X.; Gong, Y.Y.; Niu, L.Y.; Liu, X.J.; Chi, B.Q. Electronic properties of TiO2 doped with Sc, Y, La, Zr, Hf, V, Nb and Ta. Chem. Phys. Lett. 2015, 628, 43–48. [Google Scholar] [CrossRef]
- De Angelis, F.; Di Valentin, C.; Fantacci, S.; Vittadini, A.; Selloni, A. Theoretical studies on anatase and less common TiO2 phases: Bulk, surfaces, and nanomaterials. Chem. Rev. 2014, 114, 9708–9753. [Google Scholar] [CrossRef]
- Meher, S.R. Transition metal oxide-based materials for visible-light-photocatalysis. In Nanostructured Materials for Visible Light Photocatalysis, 1st ed.; Nayak, A.K., Sahu, N.K., Eds.; Elsevier Inc.: Amsterdam, The Netherlands, 2022. [Google Scholar] [CrossRef]
- Bloh, J.Z.; Dillert, R.; Bahnemann, D.W. Designing optimal metal-doped photocatalysts: Correlation between photocatalytic activity, doping ratio, and particle size. J. Phys. Chem. C 2012, 116, 25558–25562. [Google Scholar] [CrossRef]
- Stashans, A.; Bermeo, S. Al-bound hole polarons in TiO2. Chem. Phys. 2009, 363, 100–103. [Google Scholar] [CrossRef]
- Deak, P.; Aradi, B.; Frauenheim, T. Polaronic effects in TiO2 calculated by the HSE06 hybrid functional: Dopant passivation by carrier self-trapping. Phys. Rev. B 2011, 83, 155207. [Google Scholar] [CrossRef]
- Di Valentin, C.; Pacchioni, G.; Selloni, A. Reduced and n-type doped TiO2: Nature of Ti3+ species. J. Phys. Chem. C 2009, 113, 20543–20552. [Google Scholar] [CrossRef]
- Lee, H.Y.; Robertson, J. Doping and compensation in Nb-doped anatase and rutile TiO2. J. Appl. Phys. 2013, 113, 213706. [Google Scholar] [CrossRef]
- Vequizo, J.J.M.; Matsunaga, H.; Ishiku, T.; Kamimura, S.; Ohno, T.; Yamakata, A. Trapping-induced enhancement of photocatalytic activity on brookite TiO2 powders: Comparison with anatase and rutile TiO2 powders. ACS Catal. 2017, 7, 2644–2651. [Google Scholar] [CrossRef]
- Kaleji, B.K.; Sarraf-Mamoory, R.; Fujishima, A. Influence of Nb dopant on the structural and optical properties of nanocrystalline TiO2 thin films. Mater. Chem. Phys. 2012, 132, 210–215. [Google Scholar] [CrossRef]
- Osorio-Guillén, J.; Lany, S.; Zunger, A. Atomic control of conductivity versus ferromagnetism in wide-gap Oxides via selective doping: V, Nb, Ta in anatase TiO2. Phys. Rev. Lett. 2008, 100, 036601. [Google Scholar] [CrossRef]
- Furubayashi, Y.; Hitosugi, T.; Yamamoto, Y.; Inaba, K.; Kinoda, G.; Hirose, Y.; Shimada, T.; Hasegawa, T. A transparent metal: Nb-doped anatase TiO2. Appl. Phys. Lett. 2005, 86, 252101. [Google Scholar] [CrossRef]
- Singh, S.; Sharma, V.; Sachdev, K. Investigation of effect of doping concentration in Nb-doped TiO2 thin films for TCO applications. J. Mater. Sci. 2017, 52, 11580–11591. [Google Scholar] [CrossRef]
- Lü, X.; Mou, X.; Wu, J.; Zhang, D.; Zhang, L.; Huang, F.; Xu, F.; Huang, S. Improved-performance dye-sensitized solar cells using Nb-doped TiO2 electrodes: Efficient electron injection and transfer. Adv. Funct. Mater. 2010, 20, 509–515. [Google Scholar] [CrossRef]
- Wu, M.-C.; Lin, T.-H.; Chih, J.-S.; Hsiao, K.-C.; Wu, P.-Y. Niobium doping induced morphological changes and enhanced photocatalytic performance of anatase TiO2. Jpn. J. Appl. Phys. 2017, 56, 04CP07. [Google Scholar] [CrossRef]
- Ratova, M.; Kelly, P.J.; West, G.T.; Iordanova, I. Enhanced properties of magnetron sputtered photocatalytic coatings via transition metal doping. Surf. Coat. Technol. 2013, 228, S544–S549. [Google Scholar] [CrossRef]
- Michalow, K.A.; Flak, D.; Heel, A.; Parlinska-Wojtan, M.; Rekas, M.; Graule, T. Effect of Nb doping on structural, optical and photocatalytic properties of flame-made TiO2 nanopowder. Environ. Sci. Pollut. Res. 2012, 19, 3696–3708. [Google Scholar] [CrossRef]
- Nogueira, M.V.; Lustosa, G.M.M.M.; Kobayakawa, Y.; Kogler, W.; Ruiz, M.; Monteiro Filho, E.S.; Zaghete, M.A.; Perazolli, L.A. Nb-doped TiO2 photocatalysts used to reduction of CO2 to methanol. Adv. Mater. Sci. Eng. 2018, 2018, 7326240. [Google Scholar] [CrossRef]
- Qian, X.; Yang, W.; Gao, S.; Xiao, J.; Basu, S.; Yoshimura, A.; Shi, Y.; Meunier, V.; Li, Q. Highly selective, defect-induced photocatalytic CO2 reduction to acetaldehyde by the Nb-doped TiO2 nanotube array under simulated solar illumination. ACS Appl. Mater. Interfaces 2020, 12, 55982–55993. [Google Scholar] [CrossRef] [PubMed]
- Emeline, A.V.; Furubayashi, Y.; Zhang, X.; Jin, M.; Murakami, T.; Fujishima, A. Photoelectrochemical behavior of Nb-doped TiO2 electrodes. J. Phys. Chem. B 2005, 109, 24441–24444. [Google Scholar] [CrossRef] [PubMed]
- Mei, B.; Byford, H.; Bledowski, M.; Wang, L.; Strunk, J.; Muhler, M.; Beranek, R. Beneficial effect of Nb doping on the photoelectrochemical properties of TiO2 and TiO2-polyheptazine hybrids. Sol. Energy Mater. Sol. Cells 2013, 117, 48–53. [Google Scholar] [CrossRef]
- Li, D.; Li, R.; Qin, X.; Yan, W. First-principle studies of the magnetism and optical properties of (Sc, Y)-codoped anatase TiO2 (101) surface. Phys. Lett. A 2019, 383, 2679–2684. [Google Scholar] [CrossRef]
- Wei, Z.; Mei, W.; Xiyu, S.; Yachao, W.; Zhenyong, L. Electronic and optical properties of the doped TiO2 system. J. Semicond. 2010, 31, 072001. [Google Scholar] [CrossRef]
- Hirano, M.; Date, K. Scandium-doped anatase (TiO2) nanoparticles directly formed by hydrothermal crystallization. J. Am. Ceram. Soc. 2005, 88, 2604–2607. [Google Scholar] [CrossRef]
- Cavalheiro, A.A.; Bruno, J.C.; Saeki, M.J.; Valente, J.P.S.; Florentino, A.O. Effect of scandium on the structural and photocatalytic properties of titanium dioxide thin films. J. Mater. Sci. 2008, 43, 602–608. [Google Scholar] [CrossRef]
- Latini, A.; Cavallo, C.; Aldibaja, F.K.; Gozzi, D.; Carta, D.; Corrias, A.; Lazzarini, L.; Salviati, G. Efficiency improvement of DSSC photoanode by scandium doping of mesoporous titania beads. J. Phys. Chem. C 2013, 117, 25276–25289. [Google Scholar] [CrossRef]
- Nasir, M.; Lei, J.; Iqbal, W.; Zhang, J. Study of synergistic effect of Sc and C co-doping on the enhancement of visible light photo-catalytic activity of TiO2. Appl. Surf. Sci. 2016, 364, 446–454. [Google Scholar] [CrossRef]
- da Silva, D.W.; Manfroi, D.C.; Teixeira, G.F.; Perazolli, L.A.; Zaghete, M.A.; Cavalheiro, A.A. Photocatalytic decomposition of Rhodamine-B using scandium and silver-modified TiO2 powders. Adv. Mater. Res. 2014, 975, 213–218. [Google Scholar] [CrossRef]
- Berglund, S.P.; Hoang, S.; Minter, R.L.; Fullon, R.R.; Mullins, C.B. Investigation of 35 elements as single metal oxides, mixed metal oxides, or dopants for titanium dioxide for dye-sensitized solar cells. J. Phys. Chem. C 2013, 117, 25248–25258. [Google Scholar] [CrossRef]
- Murashkina, A.A.; Murzin, P.D.; Rudakova, A.V.; Ryabchuk, V.K.; Emeline, A.V.; Bahnemann, D.W. Influence of the dopant concentration on the photocatalytic activity: Al-doped TiO2. J. Phys. Chem. C 2015, 119, 24695–24703. [Google Scholar] [CrossRef]
- Murashkina, A.A.; Rudakova, A.V.; Ryabchuk, V.K.; Nikitin, K.V.; Mikhailov, R.V.; Emeline, A.V.; Bahnemann, D.W. Influence of the dopant concentration on the photoelectrochemical behavior of Al-doped TiO2. J. Phys. Chem. C 2018, 122, 7975–7981. [Google Scholar] [CrossRef]
- Murzin, P.D.; Murashkina, A.A.; Emeline, A.V.; Bahnemann, D.W. Effect of Sc3+/V5+ co-doping on photocatalytic activity of TiO2. Top. Catal. 2021, 64, 817–823. [Google Scholar] [CrossRef]
- Murzin, P.D.; Rudakova, A.V.; Emeline, A.V.; Bahnemann, D.W. Effect of the heterovalent doping of TiO2 with Sc3+ and Nb5+ on the defect distribution and photocatalytic activity. Catalysts 2022, 12, 484. [Google Scholar] [CrossRef]
- Shaitanov, L.; Murashkina, A.; Rudakova, A.; Ryabchuk, V.; Emeline, A.; Artemev, Y.; Kataeva, G.; Serpone, N. UV-induced formation of color centers in dispersed TiO2 particles: Effect of thermal treatment, metal (Al) doping, and adsorption of molecules. J. Photochem. Photobiol. A 2018, 354, 33–46. [Google Scholar] [CrossRef]
- Shannon, R.D. Revised effective ionic radii and systematic studies of interatomic distances in halides and chalcogenides. Acta Cryst. 1976, 32, 751–767. [Google Scholar] [CrossRef]




| x-Sc-TiO2 | Sc Content, at.% | x-Nb-TiO2 | Nb Content, at.% |
|---|---|---|---|
| 0-Sc-TiO2 | 0.00 | 0-Nb-TiO2 | 0.00 |
| 0.2-Sc-TiO2 | 0.25 | 0.2-Nb-TiO2 | 0.22 |
| 0.4-Sc-TiO2 | 0.45 | 0.4-Nb-TiO2 | 0.38 |
| 0.6-Sc-TiO2 | 0.60 | 0.6-Nb-TiO2 | 0.64 |
| 0.8-Sc-TiO2 | 0.82 | 0.8-Nb-TiO2 | 0.81 |
| 1.0-Sc-TiO2 | 1.07 | 1.0-Nb-TiO2 | 1.08 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Siliavka, E.S.; Rudakova, A.V.; Bakiev, T.V.; Murashkina, A.A.; Murzin, P.D.; Kataeva, G.V.; Emeline, A.V.; Bahnemann, D.W. Effect of the Heterovalent Sc3+ and Nb5+ Doping on Photoelectrochemical Behavior of Anatase TiO2. Catalysts 2024, 14, 76. https://doi.org/10.3390/catal14010076
Siliavka ES, Rudakova AV, Bakiev TV, Murashkina AA, Murzin PD, Kataeva GV, Emeline AV, Bahnemann DW. Effect of the Heterovalent Sc3+ and Nb5+ Doping on Photoelectrochemical Behavior of Anatase TiO2. Catalysts. 2024; 14(1):76. https://doi.org/10.3390/catal14010076
Chicago/Turabian StyleSiliavka, Elena S., Aida V. Rudakova, Tair V. Bakiev, Anna A. Murashkina, Petr D. Murzin, Galina V. Kataeva, Alexei V. Emeline, and Detlef W. Bahnemann. 2024. "Effect of the Heterovalent Sc3+ and Nb5+ Doping on Photoelectrochemical Behavior of Anatase TiO2" Catalysts 14, no. 1: 76. https://doi.org/10.3390/catal14010076
APA StyleSiliavka, E. S., Rudakova, A. V., Bakiev, T. V., Murashkina, A. A., Murzin, P. D., Kataeva, G. V., Emeline, A. V., & Bahnemann, D. W. (2024). Effect of the Heterovalent Sc3+ and Nb5+ Doping on Photoelectrochemical Behavior of Anatase TiO2. Catalysts, 14(1), 76. https://doi.org/10.3390/catal14010076









