Abstract
Microreactors have become an efficient tool for many enzymatic reactions because the laminar fluid flow within the microchannel enables precise process control, rapid mixing, and short residence time. This paper provides a systematic overview of the application of reaction kinetics and the mathematical modeling of enzymatic processes in microreactors. Rapid heat and mass transfer and a high surface-to-volume ratio are usually the reasons why reactions in microchannels proceed faster and with higher yields and productivity compared to conventional macroreactors. Since there are no radial diffusion limitations, microreactors are also an effective tool for determining the kinetic parameters of enzyme-catalyzed reactions. By eliminating the mass transfer effect on the reaction rate, the kinetics estimated in the microreactor are closer to the intrinsic kinetics of the reaction. In this review, the advantages and disadvantages of using microreactors are highlighted and the potential of their application is discussed. Advances in microreactors result in process intensification and more efficient biocatalytic processes in line with the advantages offered by the application of microreactors, such as (i) higher yields, (ii) a cleaner and improved product profile, (iii) scale-independent synthesis, (iv) increased safety, and (v) the constant quality of the output product through (vi) accelerated process development. Furthermore, microreactors are an excellent tool for kinetic studies under specified mass transfer conditions, enhancing the capabilities of other methods.
1. Enzyme Microreactors
1.1. Microreactors: Definitions and Some Basic Properties
According to the literature, a microreactor can be defined as a device made up of a series of interconnected microchannels (typically with an internal diameter less than 1 mm) in which small amounts of chemical can react [1,2,3]. The internal volume of microreactors can vary from less than 1 µL to several µL, while the internal volume of conventional macroreactors ranges from 100 mL to several liters [4,5,6]. Microchannels are basically straight tubes, but their structure can be extremely complex and includes, for example, micromixers. They are typically produced as circular or rectangular in their cross-sections. Microreactors are made from stable and inert materials such as glass, silicon, stainless steel, ceramics, or polymers. The material used in the construction of microreactors is determined by their use and/or the working parameters of the reaction that takes place. To prevent unwanted effects, materials must be useful and durable, have superior electrical and temperature resistance, and be chemically inert [7]. The surface properties of the reactor microchannels depend on the material construction and also on the method of fabrication. As is described and discussed in detail by Suryawanshi et al. [8], a variety of manufacturing processes can satisfy the general requirements for building an efficient and compact microreactor. The most commonly used manufacturing methods are: micromachining, wet etching, injection molding, laser ablation microforming, soft lithography, photolithography, hot embossing, nanoimprinting, electroforming, and micro-electro-discharge machining [8].
To perform chemical or biochemical reactions, microreactors are equipped with a tunable pumping system, reagent reservoirs, and system for the selective collection of reaction products. When working with continuously operated systems, pumps are a very important part of the equipment as they must ensure stable and accurate flow rates [9], which is why syringe pumps are mostly used (Figure 1).
Figure 1.
Simplified schematic presentation of microreactor system.
The small dimensions of microchannels ensure microreactor systems have numerous advantages over macro- and mezoreactors. Some of the most significant of these are as follows: (i) fast mixing and efficient mass transfer due to no concentration gradients and mixing based on molecular diffusion [10,11,12]; (ii) high surface-to-volume ratio in range from 10,000 to 50,000 m2/m3 that ensures efficient mass and energy transfer [13,14,15]; (iii) laminar flow characterized by a Reynolds number less than 100, which ensures the better control of reaction conditions and eliminates back mixing [16,17]; (iv) the usage of small substrate volumes what results in significant cost reduction [18,19]; (v) environmental friendly so-called “green technology”, due to the small amount of chemicals used and due to limited energy consumption [20,21,22]; (vi) safe reaction conditions due to the adaptability of small volumes and production materials under extreme reaction conditions such as high pressure and temperature [23]; (vii) the enhanced selectivity of the specific product synthesis [24,25]; (viii) rapid reaction rates due to high surface-to-volume ratios [26,27]; and (ix) numbering-up instead of complex scale-up allows the simple replacement of broken units without disturbing other units [28,29,30]. However, microreactor systems are still not perfect and have some disadvantages, such as: (i) clogging, due to the accumulation of solid materials on the microchannel surface reduces the flow rate and can cause the complete blocking of the channel [31,32]; (ii) the production price of microsystems depends on the used material, production technique, and additional used equipment [19]; and (iii) industrial applications are limited to pharmaceutical and fine chemical production due to low production volumes [33].
When enzymatic reactions in a microreactor are considered, an important factor is the microreactor itself and its shortcomings. According to Gojun et al. [34], the main disadvantages of their application can be: (i) clogging, (ii) production costs (the materials used, the production techniques, and the complexity of the reactor but also the use of additional equipment, such as pumps, sensors, etc.), (iii) handling due to the formation of a solid substance, (iv) the influence of the surface (microchannel wall) and chemical adsorption on that wall, and (v) the potential of the industrial application in terms of production volumes. The kinetics of chemical and biological reactions in microreactors may differ due to hydrodynamic and transport limitations, as the nature of the variation in key concentrations depends on the choice of values for these parameters [35,36]. However, the general advantages overcome the limitations, namely scale-independent synthesis, product profile improvement, accelerated process development, increased safety, consistent product quality, cleaner product profile, and higher yields [37].
As Fu et al. [38] stated, in the case of enzyme microreactors, it is extremely important to assess the viability and analyze the profitability and cost of the application [39]. The cost analysis must include the (i) the labor (cost and requirements), (ii) the process of chemicals, (iii) the equipment (cost, lifetime and utilization), (iv) the nanosprings (production and cost of non-functionalized one), (v) the sample dimensions, and (vi) the number of samples that can be treated in parallel [38,40]. In the process itself, the price of the biocatalyst is often critical, and its maximum cost depends on the process in which it is used, which directly affects the value of the final product. The current approach to chemical analysis focuses on minimizing chemicals, and precisely such an approach shows how it contributes to an additional cost increase in costs of about 5% [40]. In scientific research, enzyme immobilization is observed at the laboratory scale, where the catalyst/enzyme system is prepared on a small scale, and the cost itself is not specifically considered. However, in an industrial setting, the cost of catalyst preparation is as important as equipment and operating costs. All of the above makes it difficult to estimate operating costs. Any industrial process dealing with synthesis (such as catalyst preparation) involves equipment (installation) maintenance, equipment cleaning, product analysis, and particle characterization. All of this affects the price and duration of the process. The advantages of using enzyme kinetics in microreactors are as follows: (i) the amounts of enzyme and substrate used are extremely small; (ii) the enzyme stability is better; (iii) in addition to enzymes, other chemical species can be immobilized in the microchannel (and the enzyme involved in a specific reaction can be injected into the system); (iv) contact times are in the range of milliseconds to seconds; and (v) the use of resources, time, and labor is minimized [41,42]. Consequently, the application of microreactors offers a great advantage because very small amounts of immobilized enzymes are required and the methods are easily amenable to automated protocols, which shortens the analysis [41] and reduces the risk of the process contamination. In addition, they are considered to be more sustainable overall due to the possibility of reusing the catalyst several times [42].
As described by Verdnik et al. [43], production using microreactor technology has benefits not only in terms of selectivity, improved product efficiency, environmentally friendly processes, and simpler scale-up but also in terms of cost. The economic advantages are mainly due to the high yield and low amounts of by-products but also due to the plant safety, low energy consumption, and lower carbon footprint. However, switching from batch to continuous manufacturing generates costs, which is why an economic analysis must be part of the process development [44]. For example, Krtschil et al. [45] performed a cost analysis for an economical production of 4-cyanophenylboronic acid using microprocess technology. The analysis included investment costs (CAPEX) and operating costs (OPEX) of different production scenarios. Their results showed that the cost of microstructured reactors often represents less than 10% of the total plant-related expenditure and should therefore have a modest influence on choice of the innovative technology. Furthermore, the cost of increased production capacity was also analyzed. Total expenditures were decreased by 33% for the microchemical process with five-fold capacity and by another 25% for the process with ten-fold capacity. Results indicate that both process intensification and numbering-up are feasible methods to increase profitability. Benaskar et al. [46] performed a cost study for 14 process scenarios to analyze the cost impact of microprocessing and microwaves separately or in combination for two liquid-phase model reactions in fine chemicals synthesis, namely the Ullmann C–O cross-coupling reaction and aspirin synthesis. Their results showed that the OPEX in the Ullmann synthesis were material-based (reactant excess, pretreatment, and catalyst synthesis), while the OPEX in the aspirin synthesis seemed to be more downstream-based (workup, waste treatment). The CAPEX in the Ullmann synthesis was minimal relative to OPEX, but CAPEX for aspirin synthesis was about 40%. Furthermore, various scenarios resulted in a cost-effective and profitable processes. Based on that, integrated microwave heating and microflow processing resulted in a cost-effective system using a micropacked-bed reactor compared to a wall-coated microreactor, with a 20% profit increase. Similarly, Budžaki et al. [47] performed a cost analysis of enzymatic biodiesel production in a small-scale packed bed reactor and found that a large part of the scale-up production cost is the enzyme cost and an even larger part is the cost of the support material. Thus, it is challenging to select a support material that both ensure reaction stability and is cost effective. In addition, the reactor system must be sized to ensure sufficient production rates [48]. Furthermore, Yusuf et al. [49] demonstrated that the continuous production of biodiesel in fabricated microreactor systems would allow more fuel to be produced per unit of labor and reduce costs on a larger scale, while Tongtummachat et al. [50] stated that in the development of the continuously operated microfluidic process, possible substrate pretreatments and product separations and purifications must be considered in the cost analysis.
In this paper, information about the basic principles of working with enzymatic microreactors is described. The advantages and disadvantages of microreaction systems with dispersed enzymes and systems with immobilized enzymes are also composed. Furthermore, the challenges of performing kinetic studies and the mathematical modeling of the biotransformation process in continuously operated microsystems, as well as future perspectives in the presented field, have been discussed.
1.2. Microreactors with Dispersed Enzymes as Biocatalysts
Flow chemistry is a straightforward idea, where fluid containing a reactant or reactants is continuously pumped through a reactor to produce a stream of product [51]. According to Roberge et al. [52], 50% of reactions in fine chemical and pharmaceutical production could benefit from continuous production, while for 40% of them, microreactors would be the best reactor system for production. Taking into account the requirements of green chemistry, chemical catalytic processes are replaced with biocatalytic processes in which heavy organic solvents are replaced by green organic solvents and enzymes are used as catalysts. Enzymes are non-toxic biocatalysts, biodegradable, recyclable, naturally made from renewable resources, and known as green catalysts [53]. It can be concluded that the combination of microreactors as environmentally friendly reactor systems and bioanalytical processes can be considered as “green technology”. Biocatalysts can be either cell-free enzymes or whole cells. Free enzymes are considered more efficient than whole cells because there are no additional barriers between the substrates and the catalysts [51]. Nevertheless, enzymes, a strong catalyst utilized in biocatalysis, exhibit poor stability in solution over extended periods of time and a progressive reduction in activity during storage [54]. As described by De Santis et al. [55], by using free enzymes, the substrate and enzyme are fed through the reactor at the same time. The time-independent and location-dependent enzymatic reaction occurs inside the specific volume element, and the residence time is the most important element in achieving complete conversion. Depending on the selected physical properties of solvents for introducing both substrates and enzymes in a microreactor, two different flow patterns can be achieved, namely (i) parallel flow and (ii) segmented or slug flow [56]. The parallel flow is characterized by a layer of one phase flowing next to a layer of another phase, while in the slug flow, segments of one phase are dispersed in another phase in uniform and constant size and shape. To reduce the production cost and ensure the sustainability of the biotransformation process, a parallel flow pattern can be considered preferable due to the simple phase separation and possible reuse of the enzyme in the second cycle [57]. On the other hand, the segmented flow provides a larger interface and therefore can contribute to mass transfer [58]. When working with enzymes, a very important aspect is enzyme activity, which has to be preserved to achieve maximum production yield. Organic solvents used in biotransformations may reduce enzyme activity with long residence times. As mentioned earlier, in situ product separation has been presented as a means to enhance bioprocess performance and productivity and ionic liquids can be used as designer solvents to enhance simultaneous product separation. This approach has been described by Deng et al. [59] for the lipase-catalyzed hydrolysis of 4-nitrophenyl acetate using 1-butyl-3-methyl-imidazolium ionic liquids as the reactant solvent. The authors showed that continuous-flow biocatalytic reactions allow for high-level product purification and the three-time recycling of the enzyme. The examples of certain biotransformations with free enzymes performed in the microreactor are listed in Table 1. Based on the presented results, it can be concluded that the application of microreactor systems with dispersed enzymes as biocatalysts ensures a significantly higher production yield compared to traditional batch processing at short residence times. The presented results indicate that microreactor systems can be considered optimal reactor geometries for biotransformation with dispersed enzymes.

Table 1.
Application of microreactors for biotransformation’s with free enzymes.
1.3. Microreactors with Immobilized Biocatalysts
To ensure enzyme stability and, consequently, higher activity in the bioanalytical process, enzyme immobilization has been extensively used. As described by Hamidović et al. [69], nowadays, immobilized enzymes are preferred over free enzymes because immobilization allows enzyme recycling, making the process more cost-effective and ecologically beneficial. Furthermore, immobilization ensures long-term stability and temperature tolerance of the enzymes. The biocatalytic reaction rate is highly influenced by the quantity and activity of the immobilized enzyme in the microfluidic device. Enzyme immobilization is the binding of an enzyme to a supporting material, such as beads, gels, nanopores, magnetic nanoparticles, and/or polymer nanofibers [25]. Immobilization holds enzymes in situ all across the biochemical process, allowing them to be readily removed from the products and employed again for series of biochemical processes, allowing enzyme recycling. As described by Coloma et al. [54], here are three approaches for the preparation of immobilized enzyme microreactors, namely (i) the wall-coated type, where the enzyme is directly immobilized onto the inner wall surface of the microchannel; (ii) the monolithic type, where microchannels are filled with monolithic materials with interconnected meso-or microporous structures; and (iii) the packed-bed type, where enzymes are immobilized on polymeric or inorganic particles that are then packed into the microchannels of immobilized enzyme microreactors (Figure 2). The enzyme is directly immobilized on the inner wall surface of the microchannel in its wall-coated form. The accessible surface areas of the microfluidic walls are extremely limited, and the enzyme-loading capacity is restricted. Moreover, the substrate diffusion pathway is quite long under these circumstances, resulting in a poor biocatalytic conversion rate [70,71]. Therefore, researchers are working to improve the surface-to-volume ratio of microchannels, which will enhance the enzyme-loading capacity [72,73,74]. To enhance the surface-to-volume ratio, enzymes can be immobilized on polymeric or inorganic materials, which are then packed into microchannels [42,75,76]. Microchannels can also be filled with linked meso- or microporous structures [77,78,79]. Monolithic reactors contain monoliths composed of permeable catalytic media or catalytic materials arranged in the tubes of an inactive monolithic base [80]. In addition to the advantages in using immobilized enzymes, such as cost reduction in downstream processes, immediate reuse, and enzyme stabilization in the immobilization matrix [81,82], there are also disadvantages and/or challenges in working with immobilized enzymes. Immobilization requires more time, equipment, and resources, which makes it more expensive to implement [83,84]. Moreover, the removal of system contaminants is expensive because the entire system must be shut down [81,82,83,84].
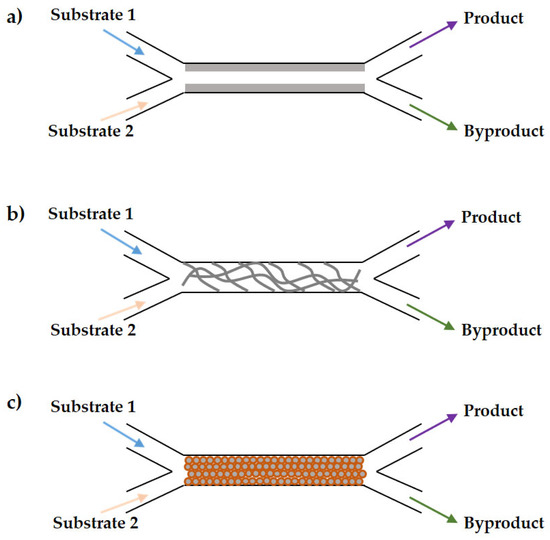
Figure 2.
Approaches for enzyme immobilization within microchannels (a) wall-coated types, (b) monolithic types, and (c) packed-bed type.
In recent years, several review papers have described and discussed current progress, advantages, problems, and challenges in working with microreactors with immobilized biocatalysts. For example, Zhu et al. [70] provided a thorough overview of the elements that influence the efficiency of continuous biocatalysis in microreactors with immobilized biocatalysts utilizing a top-down approach, while Bolivar and López-Gallego [71] presented the most important aspects when constructing immobilized enzyme reactors to ensure productive and stable flow conditions. They also investigate the efficiency and lifetime of immobilized enzymes incorporated into flow-reactors using protein loading, activity recovery, space–time yield, specific productivity, and turnover number. Furthermore, Thompson et al. [72] and Tamborini et al. [73] focused their reviews on the integration of enzyme immobilization and microfluidic devices for fine chemical production, with a particular emphasis on cutting-edge data. Romero-Fernández and Paradisi [74] presented most recent developments in continuous flow biocatalysis with an emphasis on novel enzyme immobilization and encapsulation technologies. De Santis et al. [55] focused on enzyme-catalyzed reactions, and Stojkovič and Žnidaršič-Plazl [75] described a method for immobilizing eukaryotic and prokaryotic cells onto the inner surfaces of microreactors made of various polymeric materials and glass. Some examples of microreactors for biotransformations with immobilized biocatalysts from recent years are listed in Table 2.

Table 2.
Application of microreactors for biotransformations with immobilized enzymes.
2. Kinetics of the Enzyme Reactions in Microreactors
The modeling of biotransformations as a technique for enzyme reaction engineering is fundamental for the design of enzyme-catalyzed reactions, both for large-scale production and microreactors, respectively [101]. Understanding enzyme kinetics enables the determination of optimal operating parameters and the identification of the most efficient mode of production system [41], as reaction kinetics is a key parameter for equipment design both in the laboratory and in industrial production. When it comes to determining the reaction kinetics of a chemical reaction, concentration, temperature, and pressure play an important role. Since most enzymatic reactions occur at mild reaction conditions, the concentration of substrate and products has the greatest influence. Therefore, to determine enzymatic kinetics, it is important to know the mechanism behind the interactions between enzyme and substrate and enzyme and product. To accurately determine the kinetics, it is important to accurately measure the concentration and reaction time. In addition, each reaction is also determined by mass transfer. According to Yan et al. [102] when the mass transfer is not favorable, the obtained rate can only reflect the characteristics of the mass transfer rather than the reaction itself.
When analyzing the kinetics of the enzyme-catalyzed reactions, it is necessary to consider the effects of the process variables, namely pH, temperature, substrate concentration, and enzyme concentration [1,18]. Each enzyme has an optimum pH and a change in pH outside this range will cause enzyme activity to slow down. Enzymes can be denatured by high pH [18,103,104]. Higher temperatures lead to more interactions between molecules, increasing the rate of a process. More collisions enhance the probability that the substrate will interact with the enzyme active site, increasing the rate of an enzyme-catalyzed process [105,106]. Higher substrate concentrations result in more molecular collisions and enzymes are more likely to come into contact with substrate molecules [41,92,107,108,109]. Therefore, the reaction reaches its maximum rate when the active sites are practically constantly filled. Moreover, if the enzyme concentration is low, the reaction does not continue as fast as it normally would [41,107,108]. The importance of molecules that reduce the enzyme activity must also be considered when analyzing enzyme kinetics. Many compounds can inhibit the activity of enzymes and lead to complete enzyme inactivation or a reduction in enzyme activity [105,110]. Reactive chemicals could form covalent bonds with important amino acids and prevent the enzyme from performing its reaction process, resulting in inactivation. Inactivation is usually irreversible [111]. Chemicals that bind to an enzyme and limit its ability to catalyze a reaction cause reversible inhibition [110,111,112]. The binding is non-covalent and reversible, and removing the inhibitor restores normal activity. Several strategies of reversible inhibition defined by the interaction between the inhibitor and the enzyme’s substrate are described in the literature [113,114,115,116]: (i) competitive inhibition occurs when an enzyme binds a substrate or an inhibitor but not both; (ii) non-competitive inhibition occurs if the inhibitor can bind to the enzyme whether the substrate is bound or not, implying that substrate binding has no effect on inhibition; (iii) non-competitive inhibition occurs if the inhibitor can only bind to the enzyme-substrate complex if the substrate is initially bound; and (iv) mixed inhibition is the combination of mechanisms that do not compete with any of the other processes.
Conventionally, the kinetic studies of enzyme-catalyzed reactions are performed in a cuvette or a batch reactor. The two most common methods involve using the initial reaction rate method and the determination of the change in the concentration of substrate or product in time. When it comes to moderate, conventional reactions that are not too fast, when the reaction system is homogenous, this approach is preferable, since it is fast and easy to perform. However, when the reactions are fast and heterogeneous, the previously mentioned approach has limitations. The analysis of the kinetics of rapid reactions is frequently challenging to examine in a batch reactor due to the system’s poor mass transfer characteristics. Therefore, for such processes, the use of a microchannel reactor improves mass transfer rates and eliminates the mixing effect, which may ensure that the reaction works near to intrinsic kinetics [41,117]. Furthermore, with a constant substrate concentration, an inhibitor concentration gradient in a single microchannel, and a single starting inhibitor concentration, enzyme inhibition may be determined quickly [118]. Additionally, since the accurate measurement of concentration and concentration variation in reaction time are basic kinetic measurements, microreactors could be preferable tool. The residence time distribution in microchannel is very narrow, and sub-second and milli-second residence times can also be achieved [119], indicating that microreactors can be a strong tool for kinetic measurements [120]. According to Yen et al. [102], when it comes to mass transfer resistance in microreactor, two dimensionless parameters should be considered. The first one is the Hatta number, which represents the relative speed of the reaction rate to the mass transfer rate. The second is the Thiele modulus, often used to describe the effect of the internal mass transfer. In the microchannel, the Hatta number is less than 0.3, and the Thiele modulus is 6·10−4, indicating that mass transfer resistance and internal mass transfer can be ignored in a microreactor. As previously described by Tušek et al. [101], a thorough understanding of reaction kinetics is required for the reaction development and the collection of data on the catalyst under operational settings. Given that the settings in microreactors differ from those in traditional macroreactors (high mass transfer rate, short residence time, large volume-to-surface area ratio), the kinetic parameter values of enzyme processes must be established for microreactors as well. When it comes to the determination of enzyme kinetics, the use of microreactors has another advantage. Namely, most enzymes are expensive and hard to obtain. By applying microreactors, small volumes of reagents and enzymes are sufficient for the measurement.
When it comes to studying the enzyme kinetics in microfluidics, different strategies can be applied [121], such as multi-stream laminar flow parallel reaction chambers [122], droplet microfluidics [123], and microfluidic devices with continuous flow mixers [124,125,126]. As described by Sjostrom et al. [127], multi-stream laminar flow parallel reaction systems have two main drawbacks, slow mixing and Taylor dispersion, and therefore, droplet-based microfluidic systems are considered as efficient alternative technology. Droplet-based microfluidic systems technology can provide millisecond mixing and prevents dispersion [128,129]. Some examples available in the literature are given in Table 3.

Table 3.
Strategies for enzyme kinetics studying in microfluidic devices.
Some additional examples of kinetic studies using free and immobilized enzymes in microreactor systems can be found in the literature. Kundu et al. [106] investigated enzyme-catalyzed ring-opening polymerization of caprolactone to polycaprolactone in microreactors. The procedure was carried out with the help of a commercially available immobilized lipase preparation containing Candida antarctica lipase B. Their findings demonstrated that continuously operated microreactors are a more efficient polymerization system than traditional macro-batch reactors. They also showed that conversion is temperature- and time-dependent. The efficiency of the used reactor systems was also compared based on the kinetic study. Experimental data were fitted to first-order reaction kinetics, and they showed that microreactors have an apparent rate of reaction that is at least an order of magnitude faster than the reaction rate achieved in batch reactors. Kundu et al. [106] proposed two possible explanations for that phenomenon: (1) because microreactors have a much smaller volume than batch reactors, reactants are forced to come into contact with enzyme active sites; and (2) because the surface-to-volume ratio in microreactors is much larger than in batch reactors, more enzyme active sites are available to reactants at any given time. Similarly, Jurinjak Tušek et al. [66] demonstrated that the reaction rate of the laccase-catalyzed oxidation of catechol in a microreactor was approximately two times that of batch experiments. They estimated kinetic parameters for the catechol oxidation reaction from the independent experimental results collected in a continuously operated microreactor. The parameters were estimated using non-linear regression of double substrate Michaelis–Menten kinetics (maximal reaction rate, saturation constant for catechol and saturation constant for oxygen). Tušek et al. [101] used the same approach for the kinetic analysis of the alcohol dehydrogenase-catalyzed oxidation of hexanol, where double substrate Michaelis–Menten kinetic constants were also estimated based on experimental data from independent microreactor experiments. For the estimation of the kinetic parameters, the concentration of the first reactant was varied while the second was kept at a constant value and vice versa. The calculated parameter values show that the maximum initial rate (197.276 ± 79.334 U/mg) measured in the microreactor is much higher than the one seen in the cuvette (6.755 ± 0.089 U/mg). Because the microchannels lack radial diffusion constraints (small diameter compared to channel length), they are useful for determining the kinetic data of enzyme-catalyzed processes. Additionally, the enzyme alcohol dehydrogenase prefers hexanol and NAD+ as substrates (low Km values). Since this parameter is a property of the enzyme utilized, the Michaelis–Menten Km constants for hexanol are almost identical when measured in a cuvette and in a microreactor.
Because of the growing interest in biodiesel production, a kinetic analysis of lipase-catalyzed transesterification was performed. Gojun et al. [107] used edible sunflower oil and methanol as substrates, while lipase from Thermomyces lanuginosus (Lipolase L100) was used as a catalyst for biodiesel synthesis by transesterification. Experiments were performed in a polytetrafluoroethylene (PTFE) microreactor with three inlets and in glass microreactors with two and three inlets. The kinetic parameters were estimated from independent experiments performed in a microreactor. The influence of fatty acids and the methanol concentration on the initial reaction rate was investigated to estimate the kinetic parameters of the Michaelis–Menten kinetic model. Similarly, Abdulla Yusuf et al. [108] used sunflower oil, methanol, and lipase from Thermomyces lanuginous for biodiesel production. Both studies showed similar results for kinetic parameters, indicating the better performance of the reactor in comparison to conventional reactors. Previously described examples discussed biocatalytic processed with dispersed enzymes. When working with immobilized enzymes, a different approach has to be used. A Michaelis–Menten model can represent the kinetics of an immobilized enzyme in a batch reactor. The experimental data are fitted to a Lineweaver–Burk relationship, where r is reaction rate, Vmax is the reaction’s maximum velocity, Km is the kinetic constant, and S is the substrate concentration. The experimental results provided the values for Km and Vmax (Equation (1)).
The Lilly–Hornby model is an adaptation of the classic Michaelis–Menten kinetic model, which has been used to investigate the flow kinetics of enzymes immobilized in microreactor systems [96] (Equation (2)).
where f is the fraction of substrate converted to product during the reaction, Q is the flow rate of the substrate, [A0] is the initial substrate concentration, C is the reaction capacity of the microreactor, and Km(app) is the kinetic parameter [109]. That methodology was used by (i) Bellou et al. [96] for the description of the hydrolysis of p-nitrophenyl butyrate catalyzed by lipase from Candida antarctica covalently immobilized on tubular microfluidic reactor, by (ii) Carvalho and Fernandes [110] and Carvalho et al. [111] for the description of packed-bed microreactor loaded with immobilized invertaze for the hydrolysis of sucrose into inverted sugar syrup, by (iii) Gong et al. [109] for description of isoquercitine production by graphene-immobilized naringinase, by (iv) Abd Razak et al. [53] for description of glycerol dioelate by immobilized lipase form Candida antartica in a continuous flow packed-bed milli-reactor, and by (v) Abdul Halim et al. [112] for the description of 2-amino-1,3,4-butanetriol synthesis catalyzed by transketolaze and transaminase immobilized onto agarose beads and packed into tubes to enable multistep enzyme reactions. All researchers present similar results: Km(app) values decreased with increasing flow rates, whereas for higher flow rates, the Km(app) tends to stabilize at values close to those observed for the enzyme in its free form.
Although many kinetic measurements are typically performed on a single chip, microfluidic platforms have recently improved kinetic measurements significantly. Significant increases in throughput and minimized sample usage are the most important features of these systems. The superiority of the microfluidic platform, or “microfluidic mapper”, was demonstrated in the paper presented by Rho et al. [122]. The authors created a platform that allowed them to map the enzymatic kinetics of 36 parallel biochemical reactions catalyzed by horseradish peroxidase with 36 combinations of two reagents (Amplex Red and hydrogen peroxidase). The authors obtained a 3D kinetic plot containing all information about the kinetics of the target enzyme system using a single experiment.
Another successful example is the adaptation of an easy-to-use commercial microfluidic qPCR. In their paper, Rembeza and Engqvist, [113] adapted the mentioned system in order to determine the kinetic properties of three oxidases (lactate oxidase, glutamate oxidase, and glucose oxidase) in a resorufin-based fluorescence assay. When the results were compared to those obtained in the microfluidic chip and those reported in the literature, they were comparable, but the process was much faster when performed in plates.
A droplet based microfluidic platform was developed by Hess at al. [114]. The kinetics of four enzymes were studied: β-galactosidase, horseradish peroxidase, microperoxidase, and the engineered variants of haloalkane dehalogenases. When compared with conventional methods, their platform reduced assay volumes by six orders of magnitude and increased throughput to 9000 reactions/min.
With the premise “Go big or you’ll get lost”, Markin et al. [115] developed a high-throughput microfluidic platform, which they used to express and purify more than 1500 enzyme mutants in few hours and to perform their functional characterization in few days. For each mutant, they measured Michaelis–Menten kinetics for multiple substrates, inhibition constants, and effects on folding, in the end obtaining more than 5000 kinetic and thermodynamic constants from more than 670,000 total reactions.
On the other hand, despite the fact that microreactors improve the study of enzyme reactions, in the paper presented by Li et al. [116] the authors challenged the above mentioned strategies. They stated that, in the background of classical kinetic measurements in microreactors are complex experimental setup and procedures, equipment, and sophisticated microfabrication. All of this limits the application of these systems to a wide biological community. Additionally, they stress that all of the advantages of microreactors are outweighed by their complex operation and sample preparation outside the chip. In order to overcome the mentioned disadvantages, the authors report a one-step assay to measure enzyme kinetics using a 3D-printed microfluidic device, which eliminates the steps of preparing and handling multiple solutions, thereby simplifying the whole workflow significantly. In the system they proposed, the series of solutions with different substrate concentrations required by the Michaelis–Menten kinetics are all generated on-chip with minimal extra sample preparation steps outside the chip. In order to perform measurements, only flow rates need to be adjusted. The overall system was simplified and made more user friendly for non-microfabrication experts.
With the problems that are highlighted in the work of Li et al. [129] considering microreactor application in enzyme kinetic measurements, there are still some problems that should be solved in future work. Some of these are highlighted in the paper presented by Yan et al. [102], and although they refer to the determination of kinetics in chemical reactions, we can see some parallels with enzymatic kinetics. The first thing the authors mention is that most descriptions of the excellence of microreactors are still qualitative. So, more effort should be made to quantify the superior performance of microreactors. The authors’ second point is that most kinetic research is performed on a single type of microreactor. If we assume that microreactors with different properties (e.g., the presence of micromixers, different surface roughness, etc.), such as different mass transfer and mixing properties, this may lead to different kinetic laws. On the other hand, this could be an advantage, as a more specific and tailored microreactor can be made for each reaction to achieve the best performance. The third problem is that, when it comes to kinetic studies in microreactors, they are all still focused on conventional reactions with clear mechanisms and pathways. When it comes to the kinetics of complex reactions, the equilibrium hypothesis and steady-state hypothesis, which are common for batch measurements, can be neglected. With the inline analysis, more information about reaction transition states and intermediates can be obtained, meaning that reaction mechanisms can be reflected more precisely.
3. Modelling of Enzyme Reactions in Microreactors
To obtain detailed insight into the performance of microreactor systems with dispersed or immobilized enzymes, mathematical modelling is necessary. A mathematical model describing enzymatic processes depends on a kinetic model for different biochemical transformations [130]. As described by Sudar et al. [68] each component in the reaction media, as well as the biocatalyst stability in the reactor, has an influence on the reaction rate, which must be specified for a good mathematical description. Once a mathematical model and its results are established, simulations may be highly beneficial for optimizing and improving a process, as well as evaluating the performance of a biocatalyst. Tišma et al. [131] stated that the mathematical modelling of enzymatic microreactors, including flow distribution, transport phenomena, and enzyme reaction kinetics, would allow process description and optimization. They listed the steps of the development of the mathematical model for the description of the dispersed enzyme biotransformation in a microreactor, namely (i) setting the velocity profile, (ii) the description of the mass transfer phenomena, and (iii) description of the enzyme kinetics. Several examples of such an approach are available in the literature. Tišma et al. [131] developed and validated a 2D mathematical model for the description of laccase-catalyzed L-DOPA oxidation in a microreactor. Two aqueous phases were fed into microreactor under laminar flow conditions and parabolic velocity profile was assumed. Furthermore, the concentration profiles of both reaction substrates (L-DOPA and oxygen) and enzyme were described by convection in the flow direction and diffusion in two directions considering the square cross-section of the microchannel. The kinetics of the enzyme reaction were described by double substrate Michaelis–Menten kinetics, and kinetic parameter values were adopted from the batch experiments. The developed model described the experiment data with high accuracy for all inlet L-DOPA and oxygen concentrations and for all applied flow rates. The same approach was efficiently used for modelling the catechol oxidation catalyzed by laccase in a microreactor [66] with the exception that kinetic parameter values were estimated based on microreactor experiments. The rate of catechol oxidation was modelled with a double substrate Michaelis–Menten equation (Equation (3)).
where Vm is maximum reaction rate, is saturation constant of catechol, is saturation constant of oxygen, γlaccase is enzyme concentration, and c is concentration of substrates.
The mathematical model for steady-state conditions in a microreactor was composed of dimensionless partial differential equations for catechol, oxygen, and laccase and the corresponding boundary and initial conditions (Equations (4)–(6)):
Catechol:
Oxygen:
Laccase:
where ν represents linear velocity; ξ and ψ represent independent dimensionless variables ξ = x/W and ψ = y/W; and x and y are coordinated in the length (L) and microchannel width (2W). Dcatechol/w, Doxygen/w, and Dlaccase/w are the diffusion coefficients for catechol, oxygen, and laccase in water. Aside from solving the set of partial differential equations for description of the laccase-catalyzed catechol oxidation, Jurinjak Tušek et al. [66,68] proposed a simple numerical approximation. A microreactor was described as an ideal plug flow reactor in a steady state. Mass transfer between the two plug flow reactors is modelled with diffusion, which is described as the quotient of the concentration gradient between the two positions in a microreactor and the distance between the selected positions. Their results showed that numerical simplification efficiently described the experimental data for the highest inlet concentration of oxygen at long residence times. Aside from the aqueous two-phase systems of enzyme-catalyzed biotransformations in microreactor systems, efficient mathematical models have also been developed for the description of the enzyme-catalyzed process in systems involving two miscible solvents.
Žnidaršič-Plazl and Plazl [67] developed and validated a 3D model involving convection, diffusion, and enzyme reaction terms for n-hexane–water system with dissolved lipase B from Candida antarctica for conducting the reaction using isoamyl alcohol and acetic acid as reactants. Stojkovič et al. [132] proposed a 3D model for analysis of L-malic acid production from fumaric acid in a glass microreactor using surface-immobilized fumarase, while Gojun et al. [60,107] applied a 2D model to the biodiesel transesterification process in a microreactor. Gojun et al. [107] offered a comparison of multiple mathematical models used to describe biodiesel production in a lipase-catalyzed microreactor. The Bi-Bi ping-pong mechanism is the most widely reported mechanism in the literature for biodiesel synthesis. Nonetheless, due to the low order of approximation, the authors stated that it is not suitable for microprocess characterization. Among the three mathematical process models investigated (the 2D mathematical process model, the process model of steady-state two parallel plug flow reactors, and the process model of steady-state plug flow reactors), the 2D mathematical process model and the process model of steady-state two parallel plug flow reactors demonstrated good agreement between experimental results and model simulation. Moreover, the transesterification and hydrolysis reaction rates were modelled using double substrate Michaelis–Menten kinetics, Bi-Bi ping–pong kinetics, and Hill kinetics. The Hill model was presented as the appropriate kinetic model for lipase-catalyzed biodiesel generation based on the model selection criterion. In contrast to the described results, Razak et al. [133] proposed a plug flow profile for modelling the hydrolysis of p-nitrophenylpalmitate by lipase using kinetic parameter values estimated based on experimental data from a microreactor. All the researchers mentioned agree with the necessity of a detailed kinetic study of the enzyme-catalyzed reaction in microreactors.
An interesting approach was presented by Elbinger et al. [134], in which the spatial-temporal dynamics of metabolite concentrations during the conversion of sucrose to glucose-6-phosphate was analyzed. The model included a system of diffusion equations together with boundary and initial conditions. The results of the proposed model simulations were used to design the microreactor prototypes. These microreactors consisted of compartments divided by membranes containing specific transporters for substrate input and product output. Multienzyme complexes formed on nanobeads were employed to carry out metabolic processes inside the reactor compartments. Furthermore, Tměj et al. [135] proposed a mathematical model of the enzymatic-separating microreactor, including the mass balances of all reaction components in the liquid phase and in the solid phase, with the enthalpy balance and the equation describing the thermal enzyme inactivation. The microreactor used was constructed in the form of a thin channel filled with a gel containing an immobilized enzyme and an adsorbent, in which the enzyme reaction, molecular diffusion, electro-osmotic flux and adsorption took place. The numerical analysis of the mathematical model demonstrated the potential of the presented reactor design and operation mode and allowed the process optimization to take place without experimental studies. The mathematical model of a biosensor based on an array of enzyme microreactors immobilized on a single electrode was efficiently used for the analysis of the influence of a microreactor geometry on the biosensor response [136]. The model included diffusion equations with a non-linear component related to Michaelis–Menten enzymatic kinetics. Iliuta and Larachi [137] developed a model to describe nicotinamide synthesis in laminar flow microreactors, in which the enzyme nitrile hydratase was uniformly immobilized on the microchannel wall and/or on the surface of microparticles dispersed in the liquid phase. The model was proposed assuming the laminar flow, where mass transport in the radial direction occurs by molecular diffusion and mass transport in the axial direction occurs by both advection and diffusion. The diffusion of particles, depending on the Brownian motion, was described by the Stokes–Einstein equation. The results of the simulations were used to select the optimal microchannel geometry to achieve maximum nicotinamide synthesis. Furthermore, Krull and Peterat [138] modeled the aerobic cultivation of Saccharomyces cerevisiae in a continuous chemostat using a multiphase microreactor. The model was based on a steady-state equation describing the concentration changes of biomass, glucose, and ethanol with high accuracy. Schilke et al. [139] applied a mathematical modeling approach to analyze the hydrolysis of o-nitrophenyl-β-D-galactosylpyranoside with β-galactosidase from Aspergillus oryzae immobilized on the thiolated nanosprings by reversible disulfide linkages in a microreactor system. The two-dimensional mass transfer and reaction of the species was modeled by Fickian convection/diffusion equation, and the simulation results were used to improve the microreactor performance. Interestingly, Elagli et al. [140] used a stochastic algorithm based on Michaelis–Menten kinetics to describe hemoglobin hydrolysis by pepsin. The motivation for model development was the idea to modulate the kinetics of the reaction in order to obtain a new kinetic behavior, leading to altered peptides appearance kinetics.
Mathematical modeling is critical in the creation of microreactor [141]. Computational fluid dynamics is a modeling approach with strong visualization capabilities that can interact with a wide range of geometry and boundary conditions [137]. Computational fluid dynamics modeling is required to offer knowledge of the operating characteristics of microreactors and to assess the benefits and drawbacks of various flow profiles. Computational fluid dynamics modeling is useful not only for efficient design but also for interpreting experimental results. Different CDF commercial softwares are being used for microreactors modelling. A computational fluid dynamics modeling engineering approach to novel reformer designs can save time and money by avoiding costly iterative experimental design [12,142,143,144]. Venezia et al. [145] performed the computational fluid dynamics simulations of the hydrolysis reaction in a monolith channel coated by β-glucosidase adsorbed on a wrinkled silica nanoparticles. Their simulation results showed high agreement with the experimental results, demonstrating the validity of this model. Furthermore, simulations were carried out to estimate cellobiose conversion under different inlet conditions (i.e., mixture inlet velocity variation), without carrying out further experimental analyses, which is significantly more effective. Furthermore, Strniša et al. [106] used the mesoscopic lattice Boltzman method to analyze the resident time distribution within a micro-packed bed reactor composed in a two parallel-plate configuration with immobilized Candida antarctica lipase B used for the transesterification of vinyl butyrate and 1-butanol into butyl butyrate. Simulation results were in good agreement with experimental data and were used to compare the reaction efficiency of different micro-packed bed reactor geometries. Similarly, Peñaranda et al. [146] used CFD modelling to select the optimal geometry of magnetic microreactor used for phenol removal from wastewater. Laccasse used for the oxidation of phenol was covalently immobilized on amino-terminated, silanized magnetic nanoparticles. Multiphysics simulations revealed that particle mobility caused mixing patterns that were able to promote interactions between wastewater and the bionanocomposites.
4. Conclusions and Future Perspective
Microchannel devices have the benefits of decreased area and laminar flow compared to conventional reaction apparatuses. They can mimic a biological reaction apparatus, such as cellular surfaces and vascular systems. The further development of microreaction technologies will lead to improved process intensification and more efficient methods for biocatalytic processes. Particular research interest is focused on improving selective and reversible enzyme immobilization in microreactor systems [145], which would improve process performance. With the development of nanotechnology [25] and additive manufacturing or 3D printing [137], nanoscale support matrices are increasingly used in enzyme-based biotransformation processes [138,139,140]. Furthermore, with the development of analytical systems compatible with microfluidic devices, there is significant interest in the online measurements of the reaction occurring in microreactors. The real-time monitoring of chemical/biochemical reactions is an important tool in chemical/biochemical engineering. Online monitoring assists operators in process design and optimization and enables early intervention in case of unusual behavior [141,142]. Furthermore, microreactors are an excellent tool for kinetic studies under specific mass transport conditions, enhancing the capabilities of other methods and the future application of those systems will likely focus on the use of enzymes in multiphase systems to perform cascade reactions [143,144,145]. This is because, as noted by Zhang et al. [147] and Guo et al. [148], the continuous flow cascade of multistep heterogeneous catalysis is an important approach that has the potential to revolutionize stepwise catalytic synthesis, but it remains difficult to implement in practice.
Author Contributions
Conceptualization, A.J.T. and A.Š.; methodology, D.V.; writing—original draft preparation, T.S.C. and T.J.; writing—review and editing, B.Z., A.Š. and A.J.T.; visualization, M.B.; supervision, B.Z. and J.G.K. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Laurenti, E.; Vianna, A.D.S., Jr. Enzymatic microreactors in biocatalysis: History, features, and future perspectives. Biocatalysis 2016, 1, 148–165. [Google Scholar] [CrossRef]
- Šalić, A.; Tušek, A.; Zelić, B. Application of microreactors in medicine and biomedicine. J. Appl. Biomed. 2012, 10, 137–153. [Google Scholar] [CrossRef]
- Zhang, X.; Wiles, C.; Painter, S.L.; Watts, P.; Haswell, S.J. Microreactors as tools for chemical research. Chem. Today 2006, 24, 377–386. [Google Scholar]
- Jähnisch, K.; Hessel, V.; Löwe, H.; Baerns, M. Chemistry in microstructured reactors. Angew. Chem. Int. Ed. 2004, 43, 406–446. [Google Scholar] [CrossRef]
- Illg, T.; Löb, P.; Hessel, V. Flow chemistry using milli- and microstructured reactors-From conventional to novel process windows. Bioorg. Med. Chem. 2010, 18, 3707–3719. [Google Scholar] [CrossRef]
- Kitson, P.J.; Rosnes, M.H.; Sans, V.; Dragone, V.; Cronin, L. Configurable 3D-Printed millifluidic and microfluidic “lab on a chip” reactionware devices. Lab. Chip. 2012, 12, 3267–3271. [Google Scholar] [CrossRef]
- Domínguez, M.I.; Centeno, M.A.; Martínez, M.; Bobadilla, L.F.; Laguna, Ó.H.; Odriozola, J.A. Current scenario and prospects in manufacture strategies for glass, quartz, polymers and metallic microreactors: A comprehensive review. Chem. Eng. Res. Des. 2021, 1, 13–35. [Google Scholar] [CrossRef]
- Suryawanshi, P.L.; Gumfekar, S.P.; Bhanvase, B.A.; Sonawane, S.H.; Pimplapure, M.S. A review on microreactors: Reactor fabrication, design, and cutting-edge applications. Chem. Eng. Sci. 2018, 189, 431–448. [Google Scholar] [CrossRef]
- Tonomura, O.; Okamoto, K.; Taniguchi, S.; Hasebe, S. Design of microreactor systems with minimization of flow pulsation. Comput. Aided Chem. Eng. 2019, 46, 1795–1800. [Google Scholar]
- Mielke, E.; Plouffe, P.; Mongeon, S.S.; Aellig, C.; Filliger, S.; Macchi, A.; Roberge, D.M. Micro-reactor mixing unit interspacing for fast liquid-liquid reactions leading to a generalized scale-up methodology. Chem. Eng. J. 2018, 352, 682–694. [Google Scholar] [CrossRef]
- Mielke, E.; Roberge, D.M.; MaCchi, A. Microreactor mixing-unit design for fast liquid-liquid reactions. J. Flow Chem. 2016, 6, 279–287. [Google Scholar] [CrossRef]
- Liu, H.; Wang, C.; Wang, R.; Yang, X. Design, heat transfer, and visualization of the milli-reactor by CFD and ANN. Processes 2022, 10, 2329. [Google Scholar] [CrossRef]
- Kwapiszewski, R.; Ziolkowska, K.; Zukowski, K.; Chudy, M.; Dybko, A.; Brzozka, Z. Effect of a high surface-to-volume ratio on fluorescence-based assays. Anal. Bioanal. Chem. 2012, 403, 151–155. [Google Scholar] [CrossRef]
- Namvar, A.; Blanch, A.J.; Dixon, M.W.; Carmo, O.M.S.; Liu, B.; Tiash, S.; Looker, O.; Andrew, D.; Chan, L.J.; Tham, W.H.; et al. Surface area-to-volume ratio, not cellular viscoelasticity, is the major determinant of red blood cell traversal through small channels. Cell. Microbiol. 2021, 23, e13270. [Google Scholar] [CrossRef]
- Xue, X.; Patel, M.K.; Kersaudy-Kerhoas, M.; Desmulliez, M.P.Y.; Bailey, C.; Topham, D. Analysis of fluid separation in microfluidic T-channels. Appl. Math. Model. 2012, 36, 743–755. [Google Scholar] [CrossRef]
- Fang, W.F.; Yang, J.T. High performance Microreactor for Rapid Fluid Mixing and Redox Reaction of Ascorbic Acid. In Proceedings of the 2008 Symposium on Design, Test, Integration and Packaging of MEMS/MOEMS, Nice, France, 9–11 April 2008. [Google Scholar]
- Moreau, M.; Di Miceli Raimondi, N.; Le Sauze, N.; Gourdon, C.; Cabassud, M.; Cabas-sud, M.; Miceli Raimondi, D.; Sauze, L. A new numerical method for axial dispersion characterization in microreactors A new numerical method for axial dispersion characterization in microreactors Open Archive TOULOUSE Archive Ouverte (OATAO) A new numerical method for axial dispersion characterization in microreactors. Chem. Eng. Sci. 2017, 168, 178–188. [Google Scholar]
- Heinzler, R.; Hübner, J.; Fischöder, T.; Elling, L.; Franzreb, M. A compartmented flow microreactor system for automated optimization of bioprocesses applying immobilized enzymes. Front. Bioeng. Biotechnol. 2018, 6, 189. [Google Scholar] [CrossRef]
- Bojang, A.A.; Wu, H.S. Design, Fundamental Principles of Fabrication and Applications of Microreactors. Process 2020, 8, 891. [Google Scholar] [CrossRef]
- He, Y.; Guo, S.; Chen, K.; Li, S.; Zhang, L.; Yin, S. Sustainable green production: A review of recent development on rare earths extraction and separation using microreactors. ACS Sustain. Chem. Eng. 2019, 7, 17616–17626. [Google Scholar] [CrossRef]
- Yue, J. Green process intensification using microreactor technology for the synthesis of biobased chemicals and fuels. Chem. Eng. Process. Process Intensif. 2022, 177, 109002. [Google Scholar] [CrossRef]
- Fanelli, F.; Parisi, G.; Degennaro, L.; Luisi, R. Contribution of microreactor technology and flow chemistry to the development of green and sustainable synthesis. Beilstein J. Org. Chem. 2017, 13, 520–542. [Google Scholar] [CrossRef] [PubMed]
- Pohar, A.; Plazl, I. Process Intensification through Microreactor Application. Chem. Biochem. Eng. Q 2009, 23, 537–544. [Google Scholar]
- Han, C.; Xu, H.; Waclawik, E.R.; Li, X.H.; Xu, J. A bioinspired microreactor with interfacial regulation for maximizing selectivity in a catalytic reaction. Chem. Commun. 2020, 56, 8059–8062. [Google Scholar] [CrossRef] [PubMed]
- Imarah, A.O.; Silva, F.M.W.G.; Tuba, L.; Malta-Lakó, Á.; Szemes, J.; Sánta-Bell, E.; Poppe, L. A Convenient U-shape microreactor for continuous flow biocatalysis with enzyme-coated magnetic nanoparticles-lipase-catalyzed enantiomer selective acylation of 4-(Morpholin-4-yl)butan-2-ol. Catalysts 2022, 12, 1065. [Google Scholar] [CrossRef]
- Wang, X.; Zhang, T.; Lv, L.; Tang, W.; Gupta, R.K.; Tang, S. Reaction performance and flow behavior of isobutane/1-butene and H2SO4 in the microreactor configured with the micro-mixer. Ind. Eng. Chem. Res. 2022, 61, 9122–9135. [Google Scholar] [CrossRef]
- Aroh, K.C.; Jensen, K.F. Efficient kinetic experiments in continuous flow microreactors. React. Chem. Eng. 2018, 3, 94–101. [Google Scholar] [CrossRef]
- Dong, Z.; Wen, Z.; Zhao, F.; Kuhn, S.; Noël, T. Scale-up of micro- and milli-reactors: An overview of strategies, design principles and applications. Chem. Eng. Sci. X 2021, 10, 100097. [Google Scholar] [CrossRef]
- Qiu, M.; Zha, L.; Song, Y.; Xiang, L.; Su, Y. Numbering-up of capillary microreactors for homogeneous processes and its application in free radical polymerization. React. Chem. Eng. 2019, 4, 351–361. [Google Scholar] [CrossRef]
- Ahn, G.N.; Yu, T.; Lee, H.J.; Gyak, K.W.; Kang, J.H.; You, D.; Kim, D.P. A numbering-up metal microreactor for the high-throughput production of a commercial drug by copper catalysis. Lab. Chip. 2019, 19, 3535–3542. [Google Scholar] [CrossRef]
- Zong, J.; Yue, J. Continuous solid particle flow in microreactors for efficient chemical conversion. Ind. Eng. Chem. Res. 2022, 61, 6269–6291. [Google Scholar] [CrossRef]
- Sicignano, L.; Tomaiuolo, G.; Perazzo, A.; Nolan, S.P.; Maffettone, P.L.; Guido, S. The effect of shear flow on microreactor clogging. Chem. Eng. J. 2018, 341, 639–647. [Google Scholar] [CrossRef]
- Vrsaljko, D.; Ćevid, I.; Car, F.; Rahelić, T. Production of microreactor systems by additive manufacturing technology. Eng. Power: Bull. Croat. Acad. Eng. 2019, 14, 29–32. [Google Scholar]
- Gojun, M.; Šalić, A.; Tušek, A.J.; Valinger, D.; Tišma, M.; Zelić, B.; Croatia, Z.; Strossmayer, J.J. The Smaller, The Better—Microtechnology for a Macroresults. Eng. Power Bull. Croat. Acad. Eng. 2019, 14, 2–7. [Google Scholar]
- Patnaik, P.R. Parametric constraints on the dynamic behavior of immobilized enzyme kinetics in a microreactor. J. Biochem. Technol. 2011, 3, 270–273. [Google Scholar]
- Miložič, N.; Lubej, M.; Lakner, M.; Žnidaršič-Plazl, P.; Plazl, I. Theoretical and experimental study of enzyme kinetics in a microreactor system with surface-immobilized biocatalyst. Chem. Eng. J. 2017, 313, 374–381. [Google Scholar] [CrossRef]
- Mason, B.P.; Price, K.E.; Steinbacher, J.L.; Bogdan, A.R.; McQuade, T.D. Greener approaches to organic synthesis using microreactor technology. Chem. Rev. 2007, 107, 2300–2318. [Google Scholar] [CrossRef]
- Fu, H.; Dencic, I.; Tibhe, J.; Sanchez Pedraza, C.A.; Wang, Q.; Noel, T.; Meuldijk, J.; de Croon, M.; Hessel, V.; Weizenmann, N.; et al. Threonine aldolase immobilization on different supports for engineering of productive, cost-efficient enzymatic microreactors. Chem. Eng. J. 2012, 207–208, 564–576. [Google Scholar] [CrossRef]
- Flickinger, M.C.; Drew, S.W.; Murray, T.J.; Mehlman, M.J.; Gaden, E.; Blanch, W.; Chisti, Y.; Demain, A.; Dunnill, P.; Estell, D.; et al. Encyclopedia of Bioprocess Technology: Fermentation, Biocatalysis and Bioseparation; Wiley-Interscience: Hoboken, NJ, USA, 1999. [Google Scholar]
- Tufvesson, P.; Lima-Ramos, J.; Nordblad, M.; Woodley, J.M. Guidelines and cost analysis for catalyst production in biocatalytic processes. Org. Process Res. Dev. 2011, 15, 266–274. [Google Scholar] [CrossRef]
- Urban, P.L.; Goodall, D.M.; Bruce, N.C. Enzymatic microreactors in chemical analysis and kinetic studies. Biotechnol. Adv. 2006, 24, 42–57. [Google Scholar] [CrossRef]
- Brás, E.J.S.; Domingues, C.; Chu, V.; Fernandes, P.; Conde, P. Microfluidic bioreactors for enzymatic synthesis in packed-bed reactors-Multi-step reactions and upscaling. J. Biotechnol. 2020, 323, 24–32. [Google Scholar] [CrossRef]
- Verdnik, A.; Pintarič, Z.N.; Kravanja, Z. Process Intensification with Microprocess Engineering. Chem. Eng. Trans. 2022, 94, 589–594. [Google Scholar]
- Abou-Jaoude, A.; Arafat, Y.; Foss, A.W.; Dixon, B.W. An Economics-By-Design Approach Applied to a Heat Pipe Microreactor Concept; Idaho National Laboratory Nuclear Science & Technology: Idaho Falls, ID, USA, 2021.
- Krtschil, U.; Hessel, V.; Kralisch, D.; Kreisel, G.; Küpper, M.; Schenk, R. Cost Analysis of a Commercial Manufacturing Process of a Fine Chemical Compound Using Micro Process Engineering. Chimia 2006, 60, 611–617. [Google Scholar] [CrossRef]
- Benaskar, F.; Ben-Abdelmoumen, A.; Patil, N.; Rebrov, E.; Meuldijk, J.; Hulshof, L.; Hessel, V.; Krtschil, U.; Schouten, J. Cost analysis for a continuously operated fine chemicals production plant at 10 Kg/day using a combination of microprocessing and microwave heating. J. Flow Chem. 2011, 1, 74–89. [Google Scholar] [CrossRef]
- Budžaki, S.; Miljić, G.; Sundaram, S.; Tišma, M.; Hessel, V. Cost analysis of enzymatic biodiesel production in small-scaled packed-bed reactors. Appl. Energy 2018, 210, 268–278. [Google Scholar] [CrossRef]
- Welter, R.; Silva, J., Jr.; Souza, M.d.; Lopes, M.; Taranto, O.; Santana, H. Are Microreactors the Future of Biodiesel Synthesis? Preprints. Org. 2022, 2022020257. [Google Scholar] [CrossRef]
- Abdulla Yusuf, H.; Hossain, S.M.Z.; Aloraibi, S.; Alzaabi, N.J.; Alfayhani, M.A.; Almedfaie, H.J. Fabrication of novel microreactors in-house and their performance analysis via continuous production of biodiesel. Chem. Eng. Process. Process Intensif. 2022, 172, 108792. [Google Scholar] [CrossRef]
- Tongtummachat, T.; Jaree, A.; Akkarawatkhoosith, N. Continuous hydrothermal furfural production from xylose in a microreactor with dual-acid catalysts. RSC Adv. 2022, 12, 23366–23378. [Google Scholar] [CrossRef]
- Britton, J.; Majumdar, S.; Weiss, G.A. Continuous Flow Biocatalysis. Chem. Soc. Rev. 2018, 47, 5891. [Google Scholar] [CrossRef]
- Roberge, D.M.; Ducry, L.; Bieler, N.; Cretton, P.; Zimmermann, B. Microreactor Technology: A Revolution for the Fine Chemical and Pharmaceutical Industries? Chem. Eng. Technol. 2005, 28, 318–323. [Google Scholar] [CrossRef]
- Abd Razak, N.N.; Cognet, P.; Pérès, Y.; Gew, L.T.; Aroua, M.K. Kinetics and hydrodynamics of Candida antartica lipase-catalyzed synthesis of glycerol dioleate (GDO) in a continuous flow packed-bed millireactor. J. Clean. Prod. 2022, 373, 133816. [Google Scholar] [CrossRef]
- Coloma, J.; Guiavarc’h, Y.; Hagedoorn, P.-L.; Hanefeld, U. Immobilisation and flow chemistry: Tools for implementing biocatalysis. Chem. Commun. 2021, 57, 11416–11428. [Google Scholar] [CrossRef]
- De Santis, P.; Meyer, L.E.; Kara, S. The rise of continuous flow biocatalysis—Fundamentals, very recent developments and future perspectives. React. Chem. Eng. 2020, 5, 2155–2184. [Google Scholar] [CrossRef]
- Žnidaršič-Plazl, P. Let the biocatalyst flow. Acta Chim. Slov. 2021, 68, 1–16. [Google Scholar] [CrossRef]
- Šalić, A.; Ivanković, M.; Ferk, E.; Zelić, B. ADH based NAD+ regeneration in a microreactor. J. Chem. Technol. Biotechnol. 2013, 88, 1721–1729. [Google Scholar] [CrossRef]
- Guan, T.; Gao, Y.; Pan, M.; Wu, Y.; Zhang, S.; Xu, L.; Zhu, L.; Yun, J. Slug flow hydrodynamics of immiscible fluids within a rectangular microchannel towards size-controllable fabrication of dextran-based cryogel beads. Chem. Eng. J. 2019, 369, 116–123. [Google Scholar] [CrossRef]
- Deng, Q.; Tran, N.N.; Razi Asrami, M.; Schober, L.; Gröger, H.; Hessel, V. Ionic Liquid/Water Continuous-Flow System with Compartmentalized Spaces for Automatic Product Purification of Biotransformation with Enzyme Recycling. Ind. Eng. Chem. Res. 2020, 59, 21001–21011. [Google Scholar] [CrossRef]
- Gojun, M.; Ljubić, A.; Bačić, M.; Jurinjak Tušek, A.; Šalić, A.; Zelić, B. Model-to-model: Comparison of mathematical process models of lipase catalysed biodiesel production in a microreactor. Comput. Chem. Eng. 2021, 145, 107200. [Google Scholar] [CrossRef]
- Gojun, M.; Šalić, A.; Zelić, B. Integrated microsystems for lipase-catalyzed biodiesel production and glycerol removal by extraction or ultrafiltration. Renew. Energy 2021, 180, 213–221. [Google Scholar] [CrossRef]
- Šalić, A.; Tušek, A.J.; Sander, A.; Zelić, B. Lipase catalysed biodiesel synthesis with integrated glycerol separation in continuously operated microchips connected in series. New Biotechnol. 2018, 47, 80–88. [Google Scholar] [CrossRef]
- Valinger, D.; Vrsalović Presečki, A.; Kurtanjek, Ž.; Pohl, M.; Findrik Blažević, Z.; Vasić-Rački, D. Continuous enzymatic carboligation of benzaldehyde and acetaldehyde in an enzyme ultrafiltration membrane reactor and laminar flow microreactors. J. Mol. Catal. B Enzym. 2014, 102, 132–137. [Google Scholar] [CrossRef]
- Šalić, A.; Zelić, B. ADH-catalysed hexanol oxidation with fully integrated NADH regeneration performed in microreactors connected in series. RSC Adv. 2014, 4, 41714–41721. [Google Scholar] [CrossRef]
- Šalić, A.; Tušek, A.; Kurtanjek, Ž.; Zelić, B. Biotransformation in a microreactor: New method for production of hexanal. Biotechnol. Bioprocess Eng. 2011, 16, 495–504. [Google Scholar] [CrossRef]
- Tušek, A.J.; Tišma, M.; Bregović, V.; Ptičar, A.; Kurtanjek, Ž.; Zelić, B. Enhancement of phenolic compounds oxidation using laccase from Trametes versicolor in a microreactor. Biotechnol. Bioprocess Eng. 2013, 18, 686–696. [Google Scholar] [CrossRef]
- Žnidaršič-Plazl, P.; Plazl, I. Modelling and experimental studies on lipase-catalyzed isoamyl acetate synthesis in a microreactor. Process Biochem. 2009, 44, 1115–1121. [Google Scholar] [CrossRef]
- Sudar, M.; Findrik, Z.; Szekrenyi, A.; Clapés, P.; Vasić-Rački, Đ. Reactor and microreactor performance and kinetics of the aldol addition of dihydroxyacetone to benzyloxycarbonyl-N-3-aminopropanal catalyzed by D-fructose-6-phosphate aldolase variant A129G. Chem. Eng. Commun. 2019, 206, 927–939. [Google Scholar] [CrossRef]
- Hamidović, M.; Ender, F.; Springer, A. A novel enzymatic microreactor: Towards transforming the pharmaceutical industry. In Proceedings of the International Conference on Medical and Biological Engineering, Banja Luka, Bosnia and Herzegovina, 16–18 May 2019; Volume 73, pp. 303–308. [Google Scholar]
- Zhu, Y.; Chen, Q.; Shao, L.; Jia, Y.; Zhang, X. Microfluidic immobilized enzyme reactors for continuous biocatalysis. React. Chem. Eng. 2020, 5, 9–32. [Google Scholar] [CrossRef]
- Bolivar, J.M.; López-Gallego, F. Characterization and evaluation of immobilized enzymes for applications in flow reactors. Curr. Opin. Green Sustain. Chem. 2020, 25, 100349. [Google Scholar] [CrossRef]
- Thompson, M.P.; Peñafiel, I.; Cosgrove, S.C.; Turner, N.J. Biocatalysis using immobilized enzymes in continuous flow for the synthesis of fine chemicals. Org. Process Res. Dev. 2019, 23, 9–18. [Google Scholar] [CrossRef]
- Tamborini, L.; Fernandes, P.; Paradisi, F.; Molinari, F. Flow bioreactors as complementary tools for biocatalytic process intensification. Trends Biotechnol. 2018, 36, 73–88. [Google Scholar] [CrossRef]
- Romero-Fernández, M.; Paradisi, F. Protein immobilization technology for flow biocatalysis. Curr. Opin. Chem. Biol. 2020, 55, 1–8. [Google Scholar] [CrossRef]
- Stojkovič, G.; Žnidaršič-Plazl, P. Covalent immobilization of microbial cells on microchannel surfaces. Methods Mol. Biol. 2020, 2100, 417–426. [Google Scholar]
- Meller, K.; Pomastowski, P.; Grzywiński, D.; Szumski, M.; Buszewski, B. Preparation and evaluation of dual-enzyme microreactor with co-immobilized trypsin and chymotrypsin. J. Chromatogr. A 2016, 1440, 45–54. [Google Scholar] [CrossRef]
- Miložič, N.; Stojkovič, G.; Vogel, A.; Bouwes, D.; Žnidaršič-Plazl, P. Development of microreactors with surface-immobilized biocatalysts for continuous transamination. New Biotechnol. 2018, 47, 18–24. [Google Scholar] [CrossRef]
- Lin, K.; Xia, A.; Huang, Y.; Zhu, X.; Cai, K.; Wei, Z.; Liao, Q. Efficient production of sugar via continuous enzymatic hydrolysis in a microreactor loaded with cellulase. Chem. Eng. J. 2022, 445, 136633. [Google Scholar] [CrossRef]
- Xia, A.; Lin, K.; Zhu, T.; Huang, Y.; Zhu, X.; Zhu, X.; Cai, K.; Wei, Z.; Liao, Q. Improving the saccharification efficiency of lignocellulosic biomass using a bio-inspired two-stage microreactor system loaded with complex enzymes. Green Chem. 2022, 24, 9519–9529. [Google Scholar] [CrossRef]
- Valikhani, D.; Bolivar, J.M.; Viefhues, M.; McIlroy, D.N.; Vrouwe, E.X.; Nidetzky, B. A Spring in Performance: Silica Nanosprings Boost Enzyme Immobilization in Microfluidic Channels. ACS Appl. Mater. Interfaces 2017, 9, 34641–34649. [Google Scholar] [CrossRef]
- Obst, F.; Mertz, M.; Mehner, P.J.; Beck, A.; Castiglione, K.; Richter, A.; Voit, B.; Appelhans, D. Enzymatic synthesis of sialic acids in microfluidics to overcome cross-inhibitions and substrate supply limitations. ACS Appl. Mater. Interfaces 2021, 13, 49433–49444. [Google Scholar] [CrossRef]
- Liu, X.; Zhu, X.; Camara, M.A.; Qu, Q.; Shan, Y.; Yang, L. Surface modification with highly-homogeneous porous silica layer for enzyme immobilization in capillary enzyme microreactors. Talanta 2019, 197, 539–547. [Google Scholar] [CrossRef]
- Zhong, C.; Yang, B.; Huang, W.; Huang, H.; Zhang, S.; Yan, X.; Lu, Q.; Chen, Z.; Lin, Z. Self-assembly synthes is of trypsin-immobilized monolithic microreactor for fast and efficient proteolysis. J. Chromatogr. A 2021, 1635, 461742. [Google Scholar] [CrossRef]
- Peschke, T.; Bitterwolf, P.; Rabe, K.S.; Niemeyer, C.M. Self-immobilizing oxidoreductases for flow biocatalysis in miniaturized packed-bed reactors. Chem. Eng. Technol. 2019, 42, 2009–2017. [Google Scholar] [CrossRef]
- Svetozarević, M.; Šekuljica, N.; Onjia, A.; Barać, N.; Mihajlović, M.; Knežević-Jugović, Z.; Mijin, D. Biodegradation of synthetic dyes by free and cross-linked peroxidase in microfluidic reactor. Environ. Technol. Innov. 2022, 26, 102373. [Google Scholar] [CrossRef]
- Li, M.; Shen, H.; Zhou, Z.; He, W.; Su, P.; Song, J.; Yang, Y. Controllable and high-performance immobilized enzyme reactor: DNA-directed immobilization of multienzyme in polyamidoamine dendrimer-functionalized capillaries. Electrophoresis 2020, 41, 335–344. [Google Scholar] [CrossRef] [PubMed]
- Kecskemeti, A.; Gaspar, A. Preparation and characterization of a packed bead immobilized trypsin reactor integrated into a PDMS microfluidic chip for rapid protein digestion. Talanta 2017, 166, 275–283. [Google Scholar] [CrossRef] [PubMed]
- Ku, K.; Frey, C.; Arad, M.; Ghafourifar, G. Development of novel enzyme immobilization methods employing formaldehyde or triethoxysilylbutyraldehyde to fabricate immobilized enzyme microreactors for peptide mapping. Anal. Methods 2022, 14, 4053–4063. [Google Scholar] [CrossRef] [PubMed]
- Nagy, C.; Kecskemeti, A.; Gaspar, A. Fabrication of immobilized enzyme reactors with pillar arrays into polydimethylsiloxane microchip. Anal. Chim. Acta 2020, 1108, 70–78. [Google Scholar] [CrossRef]
- Menegatti, T.; Žnidaršič-Plazl, P. Copolymeric hydrogel-based immobilization of yeast cells for continuous biotransformation of fumaric acid in a microreactor. Micromachines 2019, 10, 867. [Google Scholar] [CrossRef]
- Menegatti, T.; Žnidaršič-Plazl, P. Hydrogel-based enzyme and cofactor co-immobilization for efficient continuous transamination in a microbioreactor. Front. Bioeng. Biotechnol. 2021, 9, 752064. [Google Scholar] [CrossRef]
- Strniša, F.; Bajić, M.; Panjan, P.; Plazl, I.; Sesay, A.M.; Žnidaršič-Plazl, P. Characterization of an enzymatic packed-bed microreactor: Experiments and modeling. Chem. Eng. J. 2018, 350, 541–550. [Google Scholar] [CrossRef]
- Bajić, M.; Plazl, I.; Stloukal, R.; Žnidaršič-Plazl, P. Development of a miniaturized packed bed reactor with ω-transaminase immobilized in LentiKats®. Process Biochem. 2017, 52, 63–72. [Google Scholar] [CrossRef]
- Molnár, Z.; Farkas, E.; Lakó, Á.; Erdélyi, B.; Kroutil, W.; Vértessy, B.G.; Paizs, C.; Poppe, L. Immobilized Whole-Cell Transaminase Biocatalysts for Continuous-Flow Kinetic Resolution of Amines. Catalysts 2019, 9, 438. [Google Scholar] [CrossRef]
- Bi, Y.; Zhou, H.; Jia, H.; Wei, P. A flow-through enzymatic microreactor immobilizing lipase based on layer-by-layer method for biosynthetic process: Catalyzing the transesterification of soybean oil for fatty acid methyl ester production. Process Biochem. 2017, 54, 73–80. [Google Scholar] [CrossRef]
- Bellou, M.G.; Gkantzou, E.; Skonta, A.; Moschovas, D.; Spyrou, K.; Avgeropoulos, A.; Gournis, D.; Stamatis, H. Development of 3D Printed Enzymatic Microreactors for Lipase-Catalyzed Reactions in Deep Eutectic Solvent-Based Media. Micromachines 2022, 13, 1954. [Google Scholar] [CrossRef]
- Tušek, A.J.; Šalić, A.; Zelić, B. Catechol removal from aqueous media using laccase immobilized in different macro- and microreactor systems. Appl. Biochem. Biotechnol. 2017, 182, 1575–1590. [Google Scholar] [CrossRef]
- Šalić, A.; Pindrić, K.; Podrepšek, G.H.; Novosel, N.; Leitgeb, M.; Zelić, B. NADH oxidation in a microreactor with an oscillating magnetic field. J. Flow Chem. 2016, 6, 27–32. [Google Scholar] [CrossRef]
- Wang, J.; Gu, S.S.; Cui, H.S.; Yang, L.Q.; Wu, X.Y. Rapid synthesis of propyl caffeate in ionic liquid using a packed bed enzyme microreactor under continuous-flow conditions. Bioresour. Technol. 2013, 149, 367–374. [Google Scholar] [CrossRef]
- Wei, C.; Zhou, Y.; Zhuang, W.; Li, G.; Jiang, M.; Zhang, H. Improving the performance of immobilized β-glucosidase using a microreactor. J. Biosci. Bioeng. 2018, 125, 377–384. [Google Scholar] [CrossRef]
- Tušek, A.; Šalić, A.; Kurtanjek, Ž.; Zelić, B. Modeling and kinetic parameter estimation of alcohol dehydrogenase-catalyzed hexanol oxidation in a microreactor. Eng. Life Sci. 2012, 12, 49–56. [Google Scholar] [CrossRef]
- Yan, Z.; Tian, J.; Du, C.; Deng, J.; Luo, G. Reaction kinetics determination based on microfluidic technology. Chinese J. Chem. Eng. 2022, 41, 49–72. [Google Scholar] [CrossRef]
- Hassan, S.U.; Nightingale, A.M.; Niu, X. Continuous measurement of enzymatic kinetics in droplet flow for point-of-care monitoring. Analyst 2016, 141, 3266–3273. [Google Scholar] [CrossRef]
- Song, H.; Ismagilov, R.F. Millisecond kinetics on a microfluidic chip using nanoliters of reagents. J. Am. Chem. Soc. 2003, 125, 14613–14619. [Google Scholar] [CrossRef]
- Jankowski, P.; Samborski, A.; Ostaszewski, R.; Garstecki, P. Evaluation of droplet-based microfluidic platforms as a convenient tool for lipases and esterases assays. Prep. Biochem. Biotechnol. 2019, 49, 727–734. [Google Scholar] [CrossRef] [PubMed]
- Kundu, S.; Bhangale, A.S.; Wallace, W.E.; Flynn, K.M.; Guttman, C.M.; Gross, R.A.; Beers, K.L. Continuous flow enzyme-catalyzed polymerization in a microreactor. J. Am. Chem. Soc. 2011, 133, 6006–6011. [Google Scholar] [CrossRef] [PubMed]
- Gojun, M.; Pustahija, L.; Tušek, A.J.; Šalić, A.; Valinger, D.; Zelić, B. Kinetic parameter estimation and mathematical modelling of lipase catalysed biodiesel synthesis in a microreactor. Micromachines 2019, 10, 759. [Google Scholar] [CrossRef] [PubMed]
- Abdulla Yusuf, H.; Elkanzi, E.M.; Hossain, S.M.Z.; Alsaei, A.M.; Alhindy, A.H.; Ebrahem, E. Design and performance assessment of an in-house fabricated microreactor for enzyme-catalysed biodiesel synthesis. Arab. J. Basic Appl. Sci. 2020, 27, 239–247. [Google Scholar] [CrossRef]
- Gong, A.; Zhu, C.T.; Xu, Y.; Wang, F.Q.; Tsabing, D.K.; Wu, F.A.; Wang, J. Moving and unsinkable graphene sheets immobilized enzyme for microfluidic biocatalysis. Sci. Rep. 2017, 7, 4309. [Google Scholar] [CrossRef]
- Carvalho, F.; Fernandes, P. Packed bed enzyme microreactor: Application in sucrose hydrolysis as proof-of-concept. Biochem. Eng. J. 2015, 104, 74–81. [Google Scholar] [CrossRef]
- Carvalho, F.; Marques, M.P.C.; Fernandes, P. Sucrose hydrolysis in a bespoke capillary wall-coated microreactor. Catalysts 2017, 7, 42. [Google Scholar] [CrossRef]
- Abdul Halim, A.; Szita, N.; Baganz, F. Characterization and multi-step transketolase-ω-transaminase bioconversions in an immobilized enzyme microreactor (IEMR) with packed tube. J. Biotechnol. 2013, 168, 567–575. [Google Scholar] [CrossRef]
- Rembeza, E.; Engqvist, M.K.M. Adaptation of a Microfluidic qPCR System for Enzyme Kinetic Studies. ACS Omega 2021, 6, 1985–1990. [Google Scholar]
- Hess, D.; Dockalova, V.; Kokkonen, P.; Bednar, D.; Damborsky, J.; deMello, A.; Prokop, Z.; Stavrakis, S. Exploring mechanism of enzyme catalysis by on-chip transient kinetics coupled with global data analysis and molecular modeling. Chem 2021, 7, 1066–1079. [Google Scholar] [CrossRef]
- Markin, C.J.; Mokhtari, D.A.; Sunden, F.; Appel, M.J.; Akiva, E.; Longwell, S.A.; Sabatti, C.; Herschlag, D.; Fordyce, P.M. Revealing enzyme functional architecture via high-throughput microfluidic enzyme kinetics. Science 2021, 373, eabf8761. [Google Scholar] [CrossRef]
- Li, X.; He, Z.; Li, C.; Li, P. One-step enzyme kinetics measurement in 3D printed microfluidics devices based on a high-performance single vibrating sharp-tip mixer. Anal. Chim. Acta 2021, 1172, 338677. [Google Scholar] [CrossRef]
- Tadepalli, S.; Qian, D.; Lawal, A. Comparison of performance of microreactor and semi-batch reactor for catalytic hydrogenation of o-nitroanisole. Catal. Today 2007, 125, 64–73. [Google Scholar] [CrossRef]
- Wohlgemuth, R.; Plazl, I.; Žnidaršič-Plazl, P.; Gernaey, K.V.; Woodley, J.M. Microscale technology and biocatalytic processes: Opportunities and challenges for synthesis. Trends Biotechnol. 2015, 33, 302–314. [Google Scholar] [CrossRef]
- Han, Z.; Li, W.; Huang, Y.; Zheng, B. Measuring rapid enzymatic kinetics by electrochemical method in droplet-based microfluidic devices with pneumatic valves. Anal. Chem. 2009, 81, 5840–5845. [Google Scholar] [CrossRef]
- Keles, H.; Susanne, F.; Livingstone, H.; Hunter, S.; Wade, C.; Bourdon, R.; Rutter, A. Development of a robust and reusable microreactor employing laser based Mid-IR chemical imaging for the automated quantification of reaction kinetics. Org. Process Res. Dev. 2017, 21, 1761–1768. [Google Scholar] [CrossRef]
- Hess, D.; Yang, T.; Stavrakis, S. Droplet-based optofluidic systems for measuring enzyme kinetics. Anal. Bioanal. Chem. 2020, 412, 3265–3283. [Google Scholar] [CrossRef]
- Rho, H.S.; Hanke, A.T.; Ottens, M.; Gardeniers, H. Mapping of Enzyme Kinetics on a Microfluidic Device. PLoS ONE 2016, 11, e0153437. [Google Scholar] [CrossRef]
- Bui, M.P.N.; Li, C.A.; Han, K.N.; Choo, J.; Lee, E.K.; Seong, G.H. Enzyme kinetic measurements using a droplet-based microfluidic system with a concentration gradient. Anal. Chem. 2011, 83, 1603–1608. [Google Scholar] [CrossRef]
- Buchegger, W.; Haller, A.; van den Driesche, S.; Kraft, M.; Lendl, B.; Vellekoop, M. Studying enzymatic bioreactions in a millisecond microfluidic flow mixer. Biomicrofluidics 2012, 6, 012803. [Google Scholar] [CrossRef]
- Jang, H.; Pawate, A.S.; Bhargava, R.; Kenis, P.J.A. Polymeric microfluidic continuous flow mixer combined with hyperspectral FT-IR imaging for studying rapid biomolecular events. Lab Chip 2019, 19, 2598–2609. [Google Scholar] [CrossRef] [PubMed]
- Kise, D.P.; Reddish, M.J.; Brian Dyer, R. Sandwich-format 3D printed microfluidic mixers: A flexible platform for multi-probe analysis. J. Micromech. Microeng. 2015, 25, 124002. [Google Scholar] [CrossRef] [PubMed]
- Sjostrom, S.L.; Joensson, H.N.; Svahn, H.A. Multiplex analysis of enzyme kinetics and inhibition by droplet microfluidics using picoinjectors. Lab Chip 2013, 13, 1754–1761. [Google Scholar] [CrossRef] [PubMed]
- Badenhorst, C.P.S.; Bornscheuer, U.T. Droplet microfluidics: From simple activity screening to sophisticated kinetics. Chem 2021, 7, 835–838. [Google Scholar] [CrossRef]
- Song, H.; Chen, D.L.; Ismagilov, R.F. Reactions in droplets in microfluidic channels. Angew. Chem. Int. Ed. Engl. 2006, 45, 7336–7356. [Google Scholar] [CrossRef]
- Mai, V.Q.; Nhan, T.A.; Hammouch, Z. A mathematical model of enzymatic non-competitive inhibition by product and its applications. Phys. Scr. 2021, 96, 124062. [Google Scholar] [CrossRef]
- Tišma, M.; Zelić, B.; Vasić-Rački, D.; Žnidaršič-Plazl, P.; Plazl, I. Modelling of laccase-catalyzed l-DOPA oxidation in a microreactor. Chem. Eng. J. 2009, 149, 383–388. [Google Scholar] [CrossRef]
- Stojkovič, G.; Plazl, I.; Žnidaršič-Plazl, P. L-Malic acid production within a microreactor with surface immobilised fumarase. Microfluid. Nanofluidics 2011, 10, 627–635. [Google Scholar] [CrossRef]
- Razak, N.N.A.; Firmansyah, S.; Annuar, M.S.M. Effects of Microfluidization on kinetic parameter values of lipase hydrolysis reaction. Biocatal. Agric. Biotechnol. 2020, 27, 101660. [Google Scholar] [CrossRef]
- Elbinger, T.; Gahn, M.; Neuss-Radu, M.; Hante, F.M.; Voll, L.M.; Leugering, G.; Knabner, P. Model-based design of biochemical microreactors. Front. Bioeng. Biotechnol. 2016, 4, 13. [Google Scholar] [CrossRef]
- Tmĕj, F.; Limbergová, Z.; Hasal, P. Modelling and optimisation of enzymatic separating micro-reactor. Bioprocess Biosyst. Eng. 2005, 28, 123–130. [Google Scholar] [CrossRef]
- Baronas, R.; Ivanauskas, F.; Kulys, J. Mathematical modeling of biosensors based on an array of enzyme microreactors. Sensors 2006, 6, 453–465. [Google Scholar] [CrossRef]
- Iliuta, I.; Larachi, F. Enhanced Enzymatic synthesis of nicotinamide in laminar flow intensified microreactors: Models and simulations. Ind. Eng. Chem. Res. 2021, 60, 12210–12219. [Google Scholar] [CrossRef]
- Krull, R.; Peterat, G. Analysis of reaction kinetics during chemostat Cultivation of Saccharomyces cerevisiae using a multiphase microreactor. Biochem. Eng. J. 2016, 105, 220–229. [Google Scholar] [CrossRef]
- Schilke, K.F.; Wilson, K.L.; Cantrell, T.; Corti, G.; McIlroy, D.N.; Kelly, C. A novel enzymatic microreactor with Aspergillus oryzae β-galactosidase immobilized on silicon dioxide nanosprings. Biotechnol. Prog. 2010, 26, 1597–1605. [Google Scholar] [CrossRef]
- Elagli, A.; Laurette, S.; Treizebre, A.; Bocquet, B.; Froidevaux, R. Diffusion based kinetic selectivity modulation of enzymatic proteolysis in a microfluidic reactor: Experimental analysis and stochastic modeling. RSC Adv. 2014, 4, 3873–3882. [Google Scholar] [CrossRef]
- Chen, J.; Gao, X.; Yan, L.; Xu, D. Retracted Article: Computational fluid dynamics modeling of the millisecond methane steam reforming in microchannel reactors for hydrogen production. RSC Adv. 2018, 8, 25183–25200. [Google Scholar] [CrossRef]
- Zhai, X.; Ding, S.; Chang, Y.; Jin, Y.; Cheng, Y. CFD Simulation with detailed chemistry of steam reforming of methane for hydrogen production in an integrated micro-reactor. Int. J. Hydrogen Energy 2010, 35, 5383–5392. [Google Scholar] [CrossRef]
- Van Hoecke, L.; Boeye, D.; Gonzalez-Quiroga, A.; Patience, G.S.; Perreault, P. Experimental methods in chemical engineering: Computational Fluid Dynamics/Finite Volume Method—CFD/FVM. Can. J. Chem. Eng. 2023, 101, 545–561. [Google Scholar] [CrossRef]
- An, H.; Li, A.; Sasmito, A.P.; Kurnia, J.C.; Jangam, S.V.; Mujumdar, A.S. Computational Fluid Dynamics (CFD) analysis of micro-reactor performance: Effect of various configurations. Chem. Eng. Sci. 2012, 75, 85–95. [Google Scholar] [CrossRef]
- Venezia, V.; Califano, V.; Pota, G.; Costantini, A.; Landi, G.; Di Benedetto, A. CFD Simulations of Microreactors for the Hydrolysis of Cellobiose to Glucose by β-Glucosidase Enzyme. Micromachines 2020, 11, 790. [Google Scholar] [CrossRef] [PubMed]
- Peñaranda, P.A.; Noguera, M.J.; Florez, S.L.; Husserl, J.; Ornelas-Soto, N.; Cruz, J.C.; Osma, J.F. Treatment of wastewater, phenols and dyes using novel magnetic torus microreactors and laccase immobilized on magnetite nanoparticles. Nanomaterials 2022, 12, 1688. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Ettelaie, R.; Dong, L.; Li, X.; Li, T.; Zhang, X.; Binks, B.P.; Yang, H. Pickering emulsion droplet-based biomimetic microreactors for continuous flow cascade reactions. Nat. Commun. 2022, 13, 475. [Google Scholar] [CrossRef] [PubMed]
- Guo, X.; Xue, N.; Zhang, M.; Ettelaie, R.; Yang, H. A Supraparticle-based biomimetic cascade catalyst for continuous flow reaction. Nat. Commun. 2022, 13, 5935. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).