Application of Spectroscopy Techniques for Monitoring (Bio)Catalytic Processes in Continuously Operated Microreactor Systems
Abstract
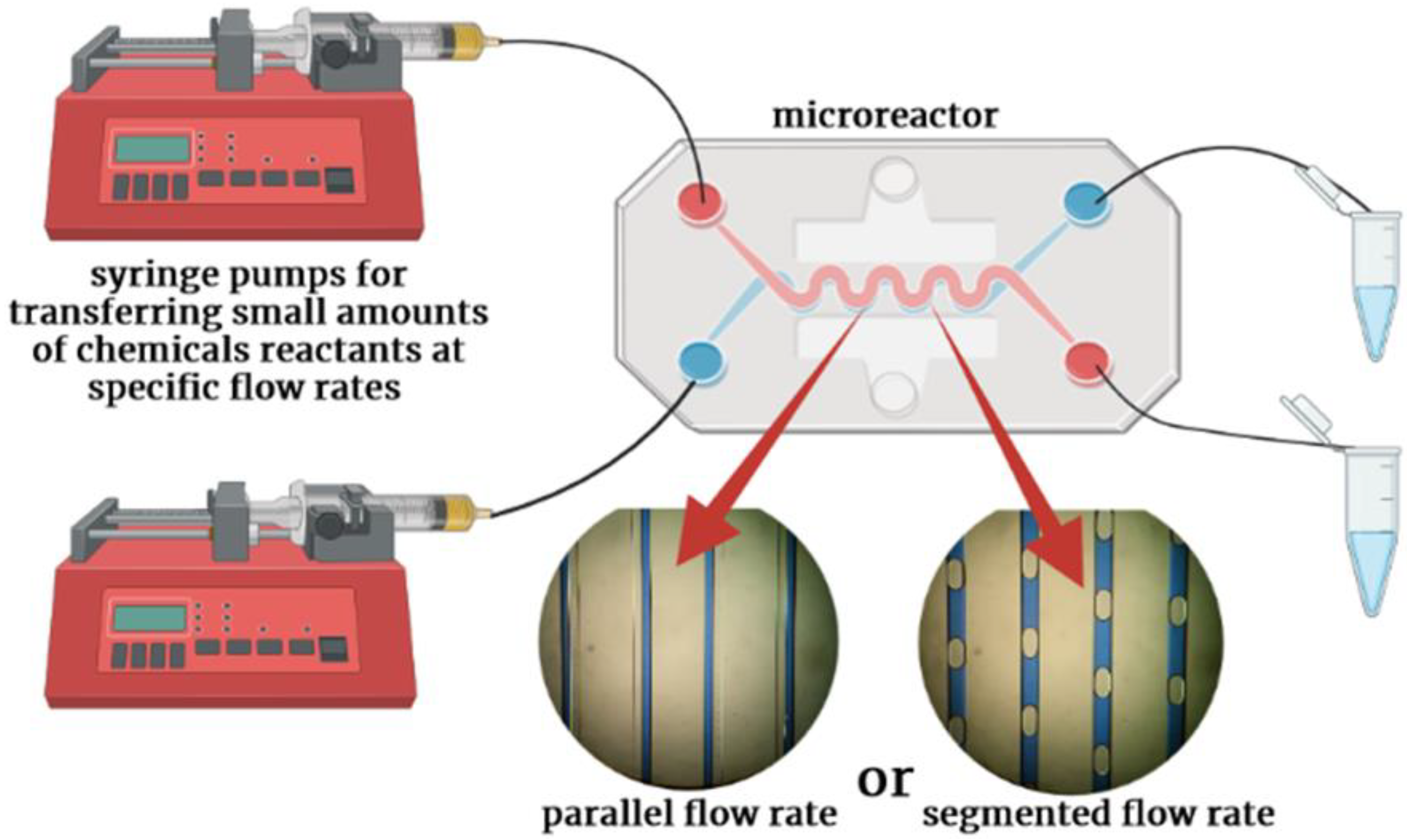
1. Flow Chemistry in Microreactors
2. Spectral Analysis Techniques and Chemometrics
2.1. Ultraviolet–Visible Spectroscopy
2.2. Infrared Spectroscopy
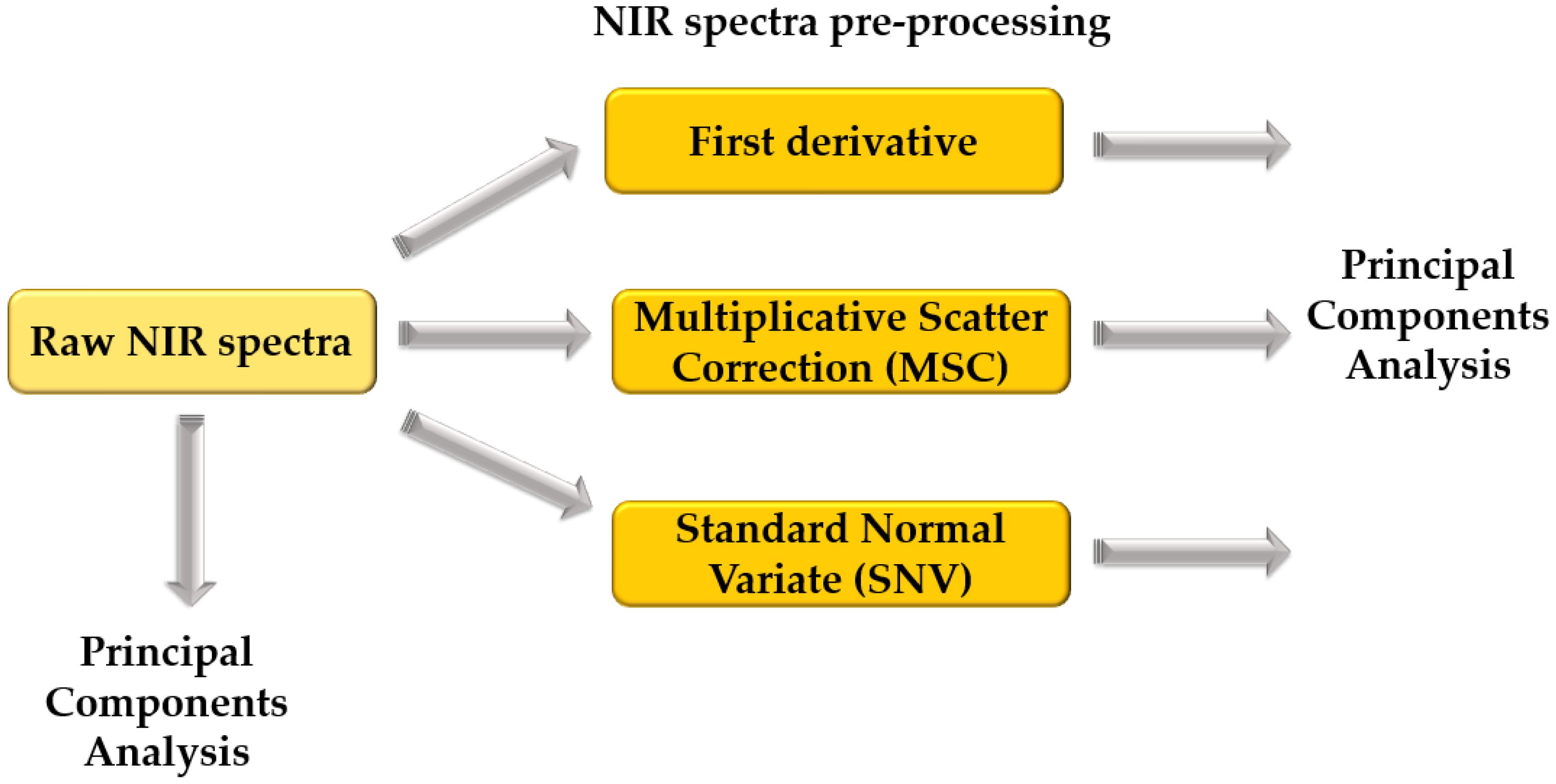
2.3. Near-Infrared Spectroscopy
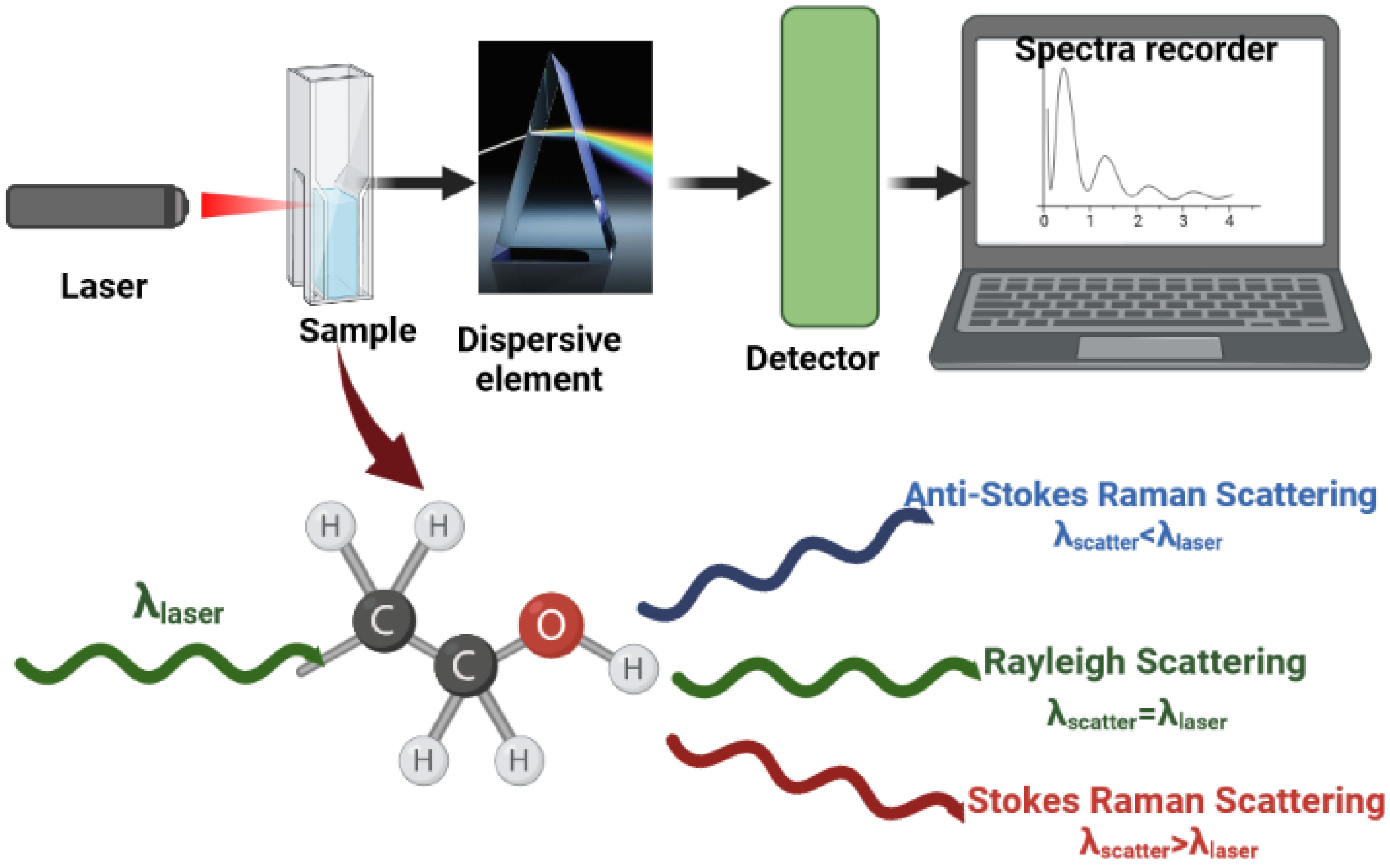
2.4. Raman Spectroscopy
2.5. Nuclear Magnetic Resonance Spectroscopy
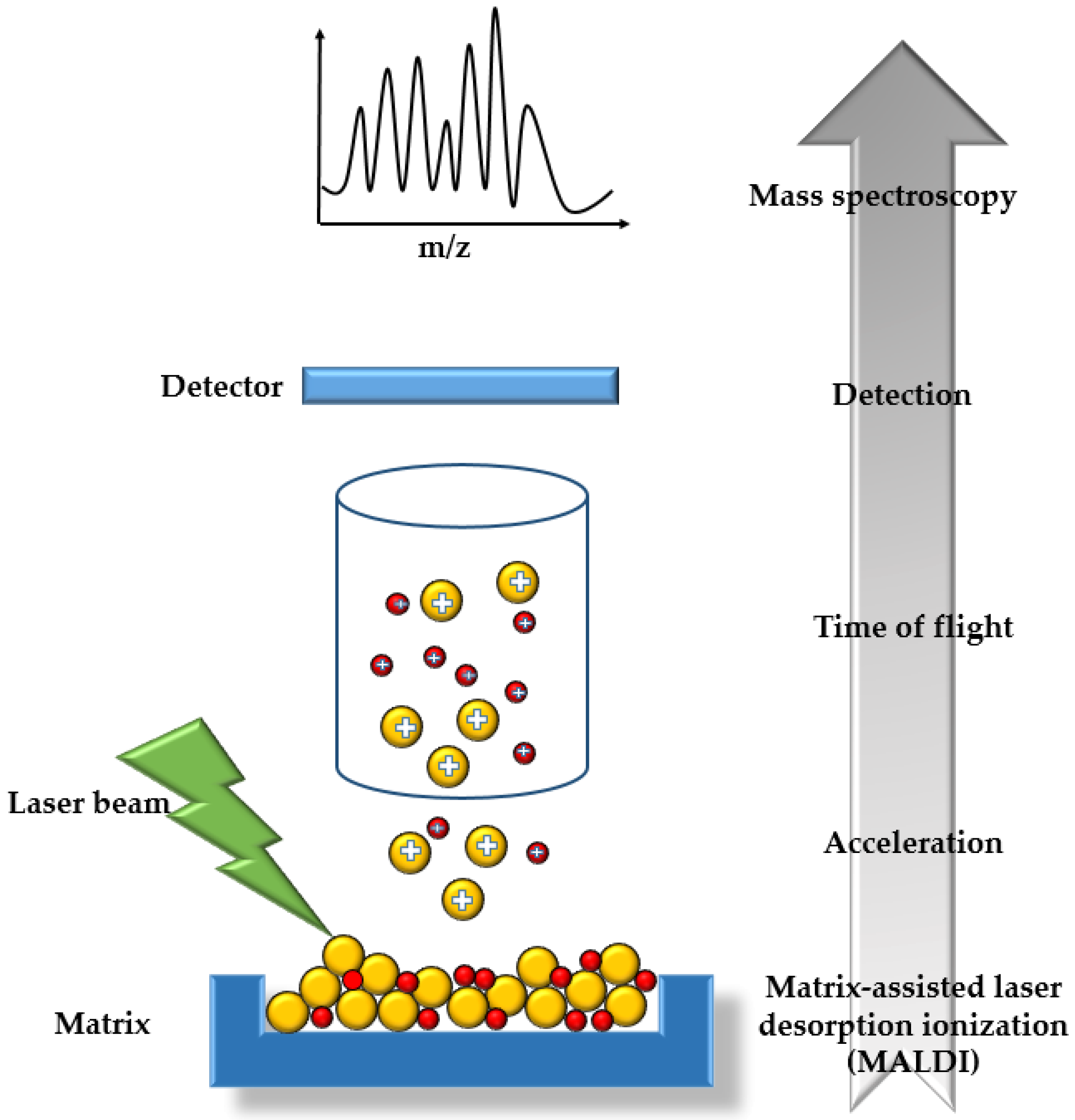
2.6. Matrix-Assisted Laser Desorption Ionization Time-of-Flight Mass Spectrometry
2.7. Electrospray Ionization Mass Spectrometry
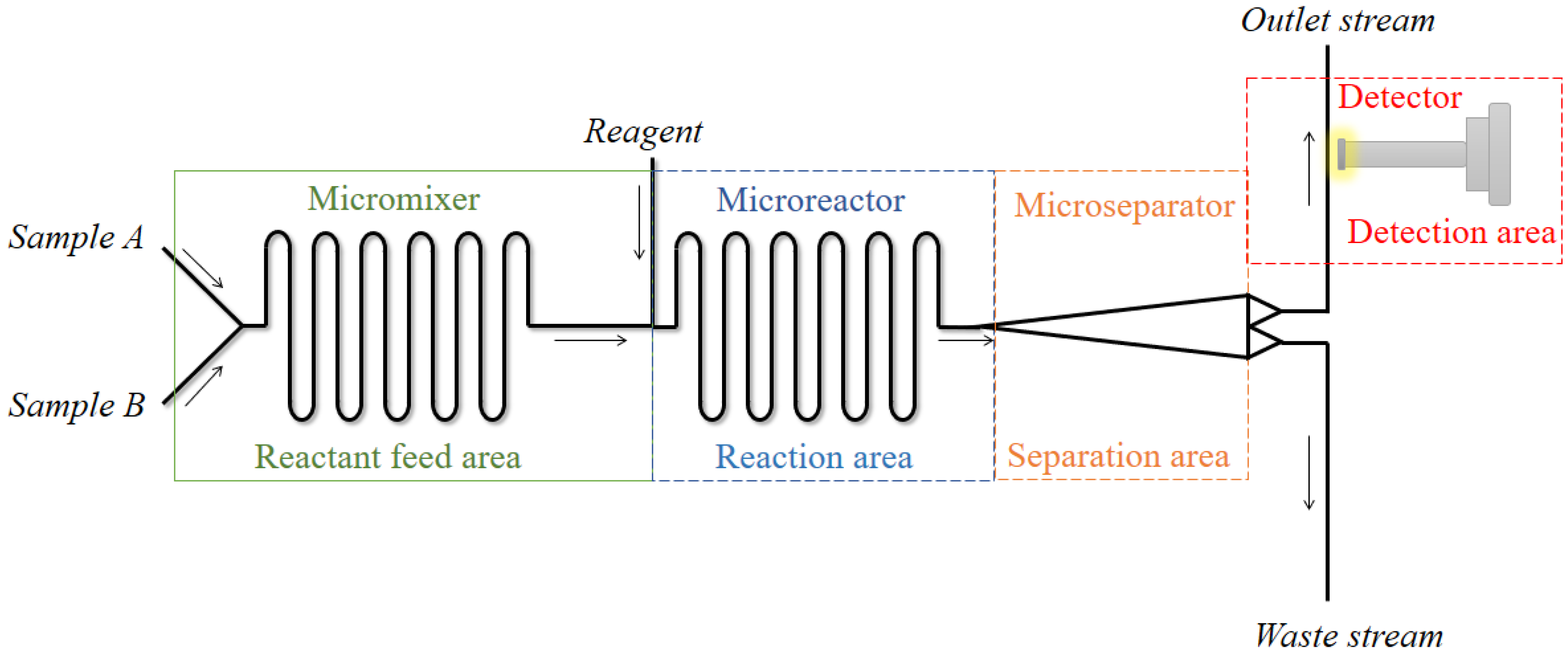
3. Microfluidic Devices Coupled with Spectroscopic Techniques
| Method | Microfluidic Device | Process | Reference |
|---|---|---|---|
| UV-Vis | Glass microfluidic chip with three sections, namely reaction zone, gas–liquid separator zone, and collocation and UV-Vis detection chamber | Gold nanoparticles (AuNP) synthesized using atmospheric-pressure helium plasma as the reducing agent followed by on-site mercury ion detection | [141] |
| 3D printed poly(lactic acid)/poly(methyl methacrylate) hybrid microfluidic device | Silver nanoparticles were synthesized using different concentrations of sodium borohydride while gold nanoparticles were synthesized varying the concentration of trisodium citrate | [142] | |
| Polydimethylsiloxane (PDMS) microfluidic dielectrophoretic droplet sorter | High-throughput label-free chemical identification and enzyme screening. The platform is used to measure ergothionase enzyme activity from monoclonal microcolonies isolated in droplets | [143] | |
| Polydimethylsiloxane microfluidic channel coupled with UV-Vis fibre-optic spectrometer and new synthesized colorimetric probe | Quantification of F-ions in flow streams | [144] | |
| Photonic lab-on-a-chip platform fabricated from polydimethylsiloxane | Detection and quantification of U(IV) concentrations in flow streams | [145] | |
| Paper microfluidic device. The device had a total of eight circular reaction zones, 10 mm diameter, printed with 0.7 mm line width in green wax, and six coloured squares as internal standard. This device would allow the measurement of eight separate standard concentrations of the drug on the same paper | Detection of decongestant phenylephrine hydrochloride (PHP) in solution | [146] | |
| Catalytic microreactor cell | In situ characterization of the activity of the silica-supported platinum (Pt) catalyst toward the dehydrogenation of 1-methyl-1,4-cyclohexadiene | [147] | |
| Hollow-core photonic crystal fibre (HC-PCF) microreactor | Analysis of the relationship between bimetallic nanoparticles and their activity on the hydrogenation of azobenzene | [148] | |
| Spiral-shaped tubular microreactors inserted inside a channel carved in a flat aluminium plate and wound in a spiral geometry | Kinetic parameters test of photochromic system AB involving 1,3,3-trimethylindolino-6′-nitrobenzopyrylospiran | [149] | |
| NIR | Micropillar array constructed on the surface of a poly(ethylene-vinyl acetate) copolymer | Ascorbic acid detection using this digital microfluidic platform | [150] |
| Microfluidic reactor consisting of: (1) a syringe pump, (2) a tubular microfluidic reactor constructed with polyetheretherketone (PEEK) or stainless steel (SS), and (3) a sample collector | GdF3:Eu theranostic scintillating nanoparticles synthesis | [151] | |
| Rectangular glass micro-capillaries in borosilicate glass | Distinguish water, ethanol, isopropanol, and ethylene glycol in flow regime | [152] | |
| Tubular microreactor | Quantification of the diffusion coefficient of aqueous solutions of sodium pentaborate | [153] | |
| Microfluidic chip prepared by sandwiching a Y-shaped cut-out silicone plate between two glass plates | Simultaneous measurement of the concentrations of acid (HCl and H2SO4), base (NaOH), and produced salt (NaCl and Na2SO4) during neutralization in a microfluidic channel | [154] | |
| MicroNIR | Tubular microreactor | In-line monitoring of the dehydration reaction of D-Fructose into -hydroxymethylfurfural | [155] |
| FTIR | Plug-flow microreactor | NO oxidation reaction at high temperatures (T > 423 K) catalysed by three zeolite frameworks (high-silica chabazites, MFI, and zeolite beta) | [156] |
| Microreactor setup consisting of coiled 1/16-inch stainless steel capillaries | Estimation and modelling of kinetics of dataimine synthesis of benzaldehyde with benzylamine and deprotonation reaction with n-butyllithium | [157] | |
| Plate microreactor directly connected to a capillary microreactor | Exothermic deprotonation reaction of a CH-acidic compound in tetrahydrofuran THF | [158] | |
| Punched Y-shape microchannel of 3 cm by 5 mm in 100 μm thick LAMINAR® E9012 dry film photopolymer | Imaging of the heat and molar concentration fields of all the species included in exothermal chemical reaction (NaOH + HCl → NaCl + H2O) in a microfluidic reactor | [159] | |
| IR | Microfluidic device composed of sapphire substrates, thin polyethylene terephthalate (PET) film, a metal chassis, and a heater | Free radical polymerization of styrene in the presence of 2,2′—azobis (isobutyronitrile) (AIBN) as the initiator | [160] |
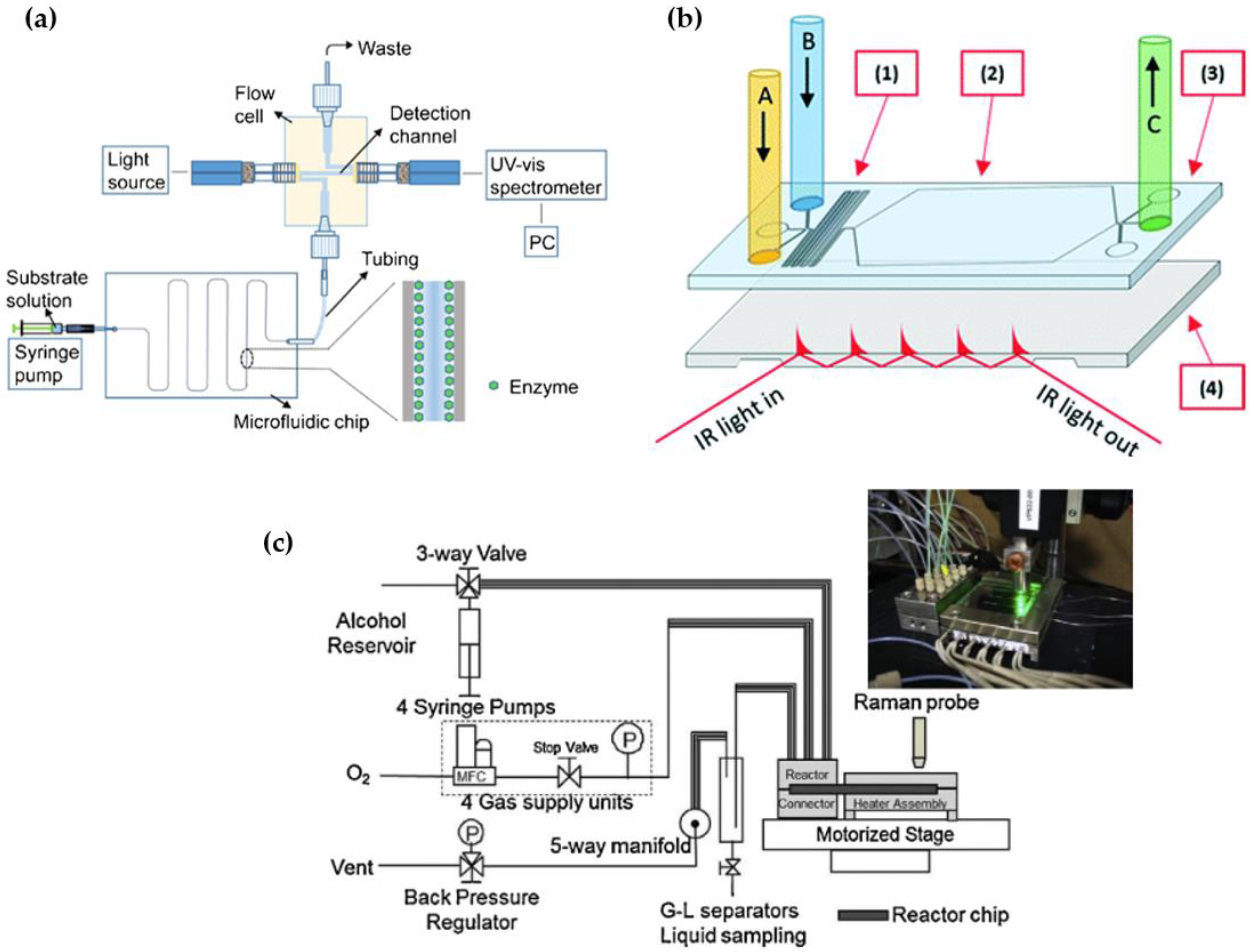
| Simultaneous application of UV-Vis and micro-Raman spectroscopies | Microfluidic chip fabricated from three fused silica plates that are thermally fused. The two outer plates form the chip channel depth | In-situ detection and quantification of both the Nd3+ (UV-Vis active) and HNO3 (Raman active) concentrations in the same sample | [161] |
| Photonic lab-on-a-chip platform | Actinide concentration monitoring along the plutonium uranium refining extraction | [162] | |
| Micro-Raman technology | Microfluidic device with microchannel width of 300 μm | Concentration measurements in organic and aqueous segments in microfluidic channel. The two-phase system was comprised of HNO3 as the aqueous phase and 30% (v/v) tributyl phosphate in n-dodecane as the organic phase, which simulated the plutonium uranium reduction extraction (PUREX) process | [163] |
| Microfluidic device with microchannel with following dimensions: width of 300 μm and depth of 250 μm | Quantification of nitric acid (HNO3) in solution | [164,165] | |
| Raman spectroscopy | The ceramic fixed-bed flow microreactor reactor | Mixed model molybdate catalysts that contain CoMoO4, Bi2Mo3O12, and Fe2Mo3O12 were investigated in the ammoxidation reaction of propene to acrylonitrile | [166] |
| Optofluidic hollow-core fibre microreactor | Monitoring of reactions involving photo-induced electron transfer processes | [167] | |
| Silicon-based microfluidic semi-flow device | Investigation of bulk-to-bulk (toluene–, diethyl ether–, and xylenes–water) interactions for liquid–liquid immiscible systems | [168] | |
| Surface-enhanced Raman scattering | Paper-based (Whatman qualitative filter paper) microfluidic device | Detection and quantification of the thiram residue | [169] |
| Glass plug-in optofluidic platform | Gold-catalysed reduction of para-nitrothiophenol by sodium borohydride | [170] | |
| Polydimethylsiloxane microfluidic devices fabricated using photo- and soft-lithography techniques. Chip included area for the nanoparticle synthesis and chamber for the Raman spectroscopy | A microfluidic device was used to encapsulate single prostate cancer cells and wheat germ agglutinin (WGA)-functionalized SERS nanoprobes in water-in-oil droplets that were subsequently locked into a storage droplet array for spectroscopic investigation | [171] | |
| Three-dimensional microfluidic chip. Microfluidic device was based on the integration of a nanoporous polycarbonate track-etched (PCTE) membrane that connects microchannels on two different levels with each other | Separation and concentration of target molecules present in a complex food sample simultaneously, which shows excellent potential in the rapid detection of a food contaminant | [172] | |
| Polydimethylsiloxane spiral shape microfluidic device | In-situ patterning of silver nanoparticles on a silicon substrate | [173] | |
| 2D periodic metal (Cu-Ag) nanostructures inside 3D glass microfluidic channels using all-femtosecond-laser-processing | Efficient detection of Cd at levels as low as 10 ppb | [68] |
3.1. UV-Vis Spectroscopy Monitoring of (Bio)Catalytic Processes in Continuously Operated Microreactor Systems
3.2. IR Spectroscopy Monitoring of (Bio)Catalytic Processes in Continuously Operated Microreactor Systems
3.3. Raman Spectroscopy Monitoring of (Bio)Catalytic Processes in Continuously Operated Microreactor Systems
3.4. MALDI-TOF MS for (Bio)Catalytic Processes in Continuously Operated Microreactor Systems
3.5. ESI-MS for (Bio)Catalytic Processes in Continuously Operated Microreactor Systems
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Rogers, L.; Jensen, K.F. Continuous Manufacturing—The Green Chemistry Promise? Green Chem. 2019, 21, 3481–3498. [Google Scholar] [CrossRef]
- Porta, R.; Benaglia, M.; Puglisi, A. Flow Chemistry: Recent Developments in the Synthesis of Pharmaceutical Products. Org. Process Res. Dev. 2016, 20, 2–25. [Google Scholar] [CrossRef]
- Hartman, R.L. Flow Chemistry Remains an Opportunity for Chemists and Chemical Engineers. Curr. Opin. Chem. Eng. 2020, 29, 42–50. [Google Scholar] [CrossRef]
- Heretsch, P. Modern Flow Chemistry-Prospect and Advantage. Beilstein J. Org. Chem 2023, 19, 33–35. [Google Scholar] [CrossRef]
- Van Herck, J.; Junkers, T. Rapid Kinetic Screening via Transient Timesweep Experiments in Continuous Flow Reactors. Chem. Methods 2022, 2, e202100090. [Google Scholar] [CrossRef]
- Mohamed, M.G.A.; Kheiri, S.; Islam, S.; Kumar, H.; Yang, A.; Kim, K. An Integrated Microfluidic Flow-Focusing Platform for on-Chip Fabrication and Filtration of Cell-Laden Microgels. Lab Chip 2019, 19, 1621–1632. [Google Scholar] [CrossRef]
- Bogdan, A.R.; Dombrowski, A.W. Emerging Trends in Flow Chemistry and Applications to the Pharmaceutical Industry. J. Med. Chem. 2019, 62, 6422–6468. [Google Scholar] [CrossRef]
- Britton, J.; Majumdar, S.; Weiss, G.A. Continuous Flow Biocatalysis. Chem. Soc. Rev. 2018, 47, 5891–5918. [Google Scholar] [CrossRef]
- Holtze, C.; Boehling, R. Batch or Flow Chemistry?—A Current Industrial Opinion on Process Selection. Curr. Opin. Chem. Eng. 2022, 36, 100798. [Google Scholar] [CrossRef]
- Wan, L.; Kong, G.; Liu, M.; Jiang, M.; Cheng, D.; Chen, F. Flow Chemistry in the Multi-Step Synthesis of Natural Products. Green Synth. Catal. 2022, 3, 243–258. [Google Scholar] [CrossRef]
- Bojang, A.A.; Wu, H.S. Design, Fundamental Principles of Fabrication and Applications of Microreactors. Processes 2020, 8, 891. [Google Scholar] [CrossRef]
- Suryawanshi, P.L.; Gumfekar, S.P.; Bhanvase, B.A.; Sonawane, S.H.; Pimplapure, M.S. A Review on Microreactors: Reactor Fabrication, Design, and Cutting-Edge Applications. Chem. Eng. Sci. 2018, 189, 431–448. [Google Scholar] [CrossRef]
- Rogolino, A.; Savio, G. Trends in Additively Manufactured Microfluidics, Microreactors and Catalytic Materials. Mater. Adv. 2021, 2, 845–855. [Google Scholar] [CrossRef]
- Abdulla Yusuf, H.; Hossain, S.M.Z.; Aloraibi, S.; Alzaabi, N.J.; Alfayhani, M.A.; Almedfaie, H.J. Fabrication of Novel Microreactors In-House and Their Performance Analysis via Continuous Production of Biodiesel. Chem. Eng. Process. Process Intensif. 2022, 172, 108792. [Google Scholar] [CrossRef]
- Vrsaljko, D.; Ćevid, I.; Car, F.; Rahelić, T. Production of Microreactor Systems by Additive Manufacturing Technology. Eng. Power Bull. Croat. Acad. Eng. 2019, 14, 29–34. [Google Scholar]
- Domínguez, M.I.; Centeno, M.A.; Martínez, M.; Bobadilla, L.F.; Laguna, Ó.H.; Odriozola, J.A. Current Scenario and Prospects in Manufacture Strategies for Glass, Quartz, Polymers and Metallic Microreactors: A Comprehensive Review. Chem. Eng. Res. Des. 2021, 1, 13–35. [Google Scholar] [CrossRef]
- Lv, H.; Yang, Z.; Zhang, J.; Qian, G.; Duan, X.; Shu, Z.; Zhou, X. Liquid Flow and Mass Transfer Behaviors in a Butterfly-Shaped Microreactor. Micromachines 2021, 12, 883. [Google Scholar] [CrossRef]
- Dong, Z.; Wen, Z.; Zhao, F.; Kuhn, S.; Noël, T. Scale-up of Micro- and Milli-Reactors: An Overview of Strategies, Design Principles and Applications. Chem. Eng. Sci. X 2021, 10, 100097. [Google Scholar] [CrossRef]
- Lozeman, J.J.A.; Elsbecker, T.; Bohnenn, S.; De Boer, H.L.; Krakers, M.; Mul, G.; Van Den Berg, A.; Odijk, M. Modular Microreactor with Integrated Reflection Element for Online Reaction Monitoring Using Infrared Spectroscopy. Lab Chip 2020, 20, 4166–4174. [Google Scholar] [CrossRef]
- Culbertson, C.T.; Mickleburgh, T.G.; Stewart-James, S.A.; Sellens, K.A.; Pressnall, M. Micro Total Analysis Systems: Fundamental Advances and Biological Applications. Anal. Chem. 2014, 86, 95. [Google Scholar] [CrossRef]
- Sridhar, A.; Kapoor, A.; Kumar, P.S.; Ponnuchamy, M.; Sivasamy, B.; Vo, D.V.N. Lab-on-a-Chip Technologies for Food Safety, Processing, and Packaging Applications: A Review. Environ. Chem. Lett. 2021, 20, 901–927. [Google Scholar] [CrossRef]
- Narayanamurthy, V.; Jeroish, Z.E.; Bhuvaneshwari, K.S.; Bayat, P.; Premkumar, R.; Samsuri, F.; Yusoff, M.M. Advances in Passively Driven Microfluidics and Lab-on-Chip Devices: A Comprehensive Literature Review and Patent Analysis. RSC Adv. 2020, 10, 11652–11680. [Google Scholar] [CrossRef]
- Patabadige, D.E.W.; Jia, S.; Sibbitts, J.; Sadeghi, J.; Sellens, K.; Culbertson, C.T. Micro Total Analysis Systems: Fundamental Advances and Applications. Anal. Chem. 2016, 88, 320–338. [Google Scholar] [CrossRef] [PubMed]
- Lee, N.Y. Recent Progress in Lab-on-a-Chip Technology and Its Potential Application to Clinical Diagnoses. Int. Neurourol. J. 2013, 17, 2. [Google Scholar] [CrossRef] [PubMed]
- Ghasemi, A.; Amiri, H.; Zare, H.; Masroor, M.; Hasanzadeh, A.; Beyzavi, A.; Aref, A.R.; Karimi, M.; Hamblin, M.R. Carbon Nanotubes in Microfluidic Lab-on-a-Chip Technology: Current Trends and Future Perspectives. Microfluid. Nanofluidics 2017, 21. [Google Scholar] [CrossRef] [PubMed]
- Gorjikhah, F.; Davaran, S.; Salehi, R.; Bakhtiari, M.; Hasanzadeh, A.; Panahi, Y.; Emamverdy, M.; Akbarzadeh, A. Improving “Lab-on-a-Chip” Techniques Using Biomedical Nanotechnology: A Review. Artif. Cells Nanomed. Biotechnol. 2016, 44, 1609–1614. [Google Scholar] [CrossRef]
- Uson, L.; Arruebo, M.; Sebastian, V.; Santamaria, J. Single Phase Microreactor for the Continuous, High-Temperature Synthesis of <4 nm Superparamagnetic Iron Oxide Nanoparticles. Chem. Eng. J. 2018, 340, 66–72. [Google Scholar] [CrossRef]
- Dobhal, A.; Kulkarni, A.; Dandekar, P.; Jain, R. A Microreactor-Based Continuous Process for Controlled Synthesis of Poly-Methyl-Methacrylate-Methacrylic Acid (PMMA) Nanoparticles. J. Mater. Chem. B 2017, 5, 3404–3417. [Google Scholar] [CrossRef]
- Dasankoppa, F.S.; Sholapur, H.N.; Byahatti, A.; Abbas, Z.; Subrata, K. Application of Response Surface Optimization Methodology in Designing Ordispersible Tablets of Antdiabetic Drug. J. Young Pharm. 2020, 12, 173–177. [Google Scholar] [CrossRef]
- Imarah, A.O.; Silva, F.M.W.G.; Tuba, L.; Malta-Lakó, Á.; Szemes, J.; Sánta-Bell, E.; Poppe, L. A Convenient U-Shape Microreactor for Continuous Flow Biocatalysis with Enzyme-Coated Magnetic Nanoparticles-Lipase-Catalyzed Enantiomer Selective Acylation of 4-(Morpholin-4-Yl)Butan-2-Ol. Catalysts 2022, 12, 1065. [Google Scholar] [CrossRef]
- Bellou, M.G.; Gkantzou, E.; Skonta, A.; Moschovas, D.; Spyrou, K.; Avgeropoulos, A.; Gournis, D.; Stamatis, H. Development of 3D Printed Enzymatic Microreactors for Lipase-Catalyzed Reactions in Deep Eutectic Solvent-Based Media. Micromachines 2022, 13, 1954. [Google Scholar] [CrossRef] [PubMed]
- Nagy, C.; Szabo, R.; Gaspar, A. Microfluidic Immobilized Enzymatic Reactors for Proteomic Analyses—Recent Developments and Trends (2017–2021). Micromachines 2022, 13, 311. [Google Scholar] [CrossRef] [PubMed]
- Gong, A.; Zhu, C.T.; Xu, Y.; Wang, F.Q.; Tsabing, D.K.; Wu, F.A.; Wang, J. Moving and Unsinkable Graphene Sheets Immobilized Enzyme for Microfluidic Biocatalysis. Sci. Rep. 2017, 7, 4309. [Google Scholar] [CrossRef] [PubMed]
- Benković, M.; Valinger, D.; Jurina, T.; Gajdoš Kljusurić, J.; Jurinjak Tušek, A. Biocatalysis as a Green Approach for Synthesis of Iron Nanoparticles—Batch and Microflow Process Comparison. Catalysts 2023, 13, 112. [Google Scholar] [CrossRef]
- Rabiee, N.; Ahmadi, S.; Fatahi, Y.; Rabiee, M.; Bagherzadeh, M.; Dinarvand, R.; Bagheri, B.; Zarrintaj, P.; Saeb, M.R.; Webster, T.J. Nanotechnology-Assisted Microfluidic Systems: From Bench to Bedside. Nanomedicine 2021, 16, 237–258. [Google Scholar] [CrossRef]
- Khizar, S.; Ben Halima, H.; Ahmad, N.M.; Zine, N.; Errachid, A.; Elaissari, A. Magnetic Nanoparticles in Microfluidic and Sensing: From Transport to Detection. Electrophoresis 2020, 41, 1206–1224. [Google Scholar] [CrossRef]
- Khan, N.I.; Song, E. Lab-on-a-Chip Systems for Aptamer-Based Biosensing. Micromachines 2020, 11, 220. [Google Scholar] [CrossRef]
- Xiang, Y.; Hu, C.; Wu, G.; Xu, S.; Li, Y. Nanomaterial-Based Microfluidic Systems for Cancer Biomarker Detection: Recent Applications and Future Perspectives. TrAC Trends Anal. Chem. 2023, 158, 116835. [Google Scholar] [CrossRef]
- Medina-Sánchez, M.; Miserere, S.; Merkoçi, A. Nanomaterials and Lab-on-a-Chip Technologies. Lab Chip 2012, 12, 1932–1943. [Google Scholar] [CrossRef]
- Monošík, R.; Angnes, L. Utilisation of Micro- and Nanoscaled Materials in Microfluidic Analytical Devices. Microchem. J. 2015, 119, 159–168. [Google Scholar] [CrossRef]
- Ali, M.A.; Solanki, P.R.; Srivastava, S.; Singh, S.; Agrawal, V.V.; John, R.; Malhotra, B.D. Protein Functionalized Carbon Nanotubes-Based Smart Lab-on-a-Chip. ACS Appl. Mater. Interfaces 2015, 7, 5837–5846. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Huang, Y.; Chen, C.; Xiao, A.; Guo, T.; Guan, B.O. Improved Detection Sensitivity of γ-Aminobutyric Acid Based on Graphene Oxide Interface on an Optical Microfiber. Phys. Chem. Chem. Phys. 2018, 20, 14117–14123. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.P.; Chen, P.; Dai, J.W.; Liu, D.; Li, J.Y.; Xu, Y.P.; Chu, X.L. Recent Advances of Chemometric Calibration Methods in Modern Spectroscopy: Algorithms, Strategy, and Related Issues. TrAC Trends Anal. Chem. 2022, 153, 116648. [Google Scholar] [CrossRef]
- Ríos-Reina, R.; Azcarate, S.M. How Chemometrics Revives the UV-Vis Spectroscopy Applications as an Analytical Sensor for Spectralprint (Nontargeted) Analysis. Chemosensors 2022, 11, 8. [Google Scholar] [CrossRef]
- Ríos-Reina, R.; Camiña, J.M.; Callejón, R.M.; Azcarate, S.M. Spectralprint Techniques for Wine and Vinegar Characterization, Authentication and Quality Control: Advances and Projections. TrAC Trends Anal. Chem. 2021, 134, 116121. [Google Scholar] [CrossRef]
- Hasbi, N.H.; Bade, A.; Chee, F.P. Pattern Recognition for Ultraviolet and Fourier Transform Data: A Walkthrough of Techniques and Direction. J. Phys. Conf. Ser. 2022, 2314, 012012. [Google Scholar] [CrossRef]
- Ballabio, D.; Consonni, V. Classification Tools in Chemistry. Part 1: Linear Models. PLS-DA. Anal. Methods 2013, 5, 3790–3798. [Google Scholar] [CrossRef]
- Indahl, U.G. The Geometry of PLS1 Explained Properly: 10 Key Notes on Mathematical Properties of and Some Alternative Algorithmic Approaches to PLS1 Modelling. J. Chemom. 2014, 28, 168–180. [Google Scholar] [CrossRef]
- Jurinjak Tušek, A.; Jurina, T.; Čulo, I.; Valinger, D.; Gajdoš Kljusurić, J.; Benković, M. Application of NIRs Coupled with PLS and ANN Modelling to Predict Average Droplet Size in Oil-in-Water Emulsions Prepared with Different Microfluidic Devices. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2022, 270, 120860. [Google Scholar] [CrossRef]
- Santos, G.R.; Paulino, G.S.P.; Borges, G.P.I.; Santiago, A.F.; da Silvac, G.A. Avanços Analíticos baseados em Modelos de Calibração de Primeira ordem e Espectroscopia UV-VIS para Avaliação da Qualidade da Água: UMA Revisão—Parte 1. Quim. Nova 2022, 45, 314–323. [Google Scholar] [CrossRef]
- Lavine, B. A User-Friendly Guide to Multivariate Calibration and Classification, Tomas Naes, Tomas Isakson, Tom Fearn and Tony Davies, NIR Publications, Chichester, 2002, ISBN 0-9528666-2-5, £45.00. J. Chemom. 2003, 17, 571–572. [Google Scholar] [CrossRef]
- Becerra, E.; Danchana, K.; Cerdà, V. WinMLR, a Software Program for the Simultaneous Determination of Several Components in Mixtures Using Multilinear Regression Analysis. Talanta 2020, 213, 120830. [Google Scholar] [CrossRef] [PubMed]
- Mevik, B.H.; Wehrens, R. The Pls Package: Principal Component and Partial Least Squares Regression in R. J. Stat. Softw. 2007, 18, 1–23. [Google Scholar] [CrossRef]
- Mevik, B.H.; Segtnan, V.H.; Næs, T. Ensemble Methods and Partial Least Squares Regression. J. Chemom. 2004, 18, 498–507. [Google Scholar] [CrossRef]
- Balabin, R.M.; Safieva, R.Z.; Lomakina, E.I. Comparison of Linear and Nonlinear Calibration Models Based on near Infrared (NIR) Spectroscopy Data for Gasoline Properties Prediction. Chemom. Intell. Lab. Syst. 2007, 88, 183–188. [Google Scholar] [CrossRef]
- Balabin, R.M.; Safieva, R.Z.; Lomakina, E.I. Wavelet Neural Network (WNN) Approach for Calibration Model Building Based on Gasoline near Infrared (NIR) Spectra. Chemom. Intell. Lab. Syst. 2008, 93, 58–62. [Google Scholar] [CrossRef]
- Valinger, D.; Kušen, M.; Tušek, A.J.; Panić, M.; Jurina, T.; Benković, M.; Radojčić Redovniković, I.; Gajdoš Kljusurić, J. Development of Near Infrared Spectroscopy Models for Quantitative Prediction of the Content of Bioactive Compounds in Olive Leaves. Dev. Near Infrared Spectrosc. Model. Chem. Biochem. Eng. Q 2018, 32, 535–543. [Google Scholar] [CrossRef]
- Kundu, P.; Paul, V.; Kumar, V.; Mishra, I.M. Formulation Development, Modeling and Optimization of Emulsification Process Using Evolving RSM Coupled Hybrid ANN-GA Framework. Chem. Eng. Res. Des. 2015, 104, 773–790. [Google Scholar] [CrossRef]
- Verma, G.; Mishra, M. Development and Optimization of UV-VIS Spectroscopy—A Review. World J. Pharm. Res. 2018, 7. [Google Scholar] [CrossRef]
- Mäntele, W.; Deniz, E. UV–VIS Absorption Spectroscopy: Lambert-Beer Reloaded. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2017, 173, 965–968. [Google Scholar] [CrossRef]
- Soo, M.H.; Samad, N.A.; Zaidel, A.; Jusoh, M.; Muhamad, Y.M.; Hashim, I.I. Extraction of Plant Based Protein from Moringa Oleifera Leaves Using Alkaline Extraction and Isoelectric Precipitation Method. Chem. Eng. Trans. 2021, 89, 253–258. [Google Scholar] [CrossRef]
- Cerdá-Bernad, D.; Baixinho, J.P.; Fernández, N.; Frutos, M.J. Evaluation of Microwave-Assisted Extraction as a Potential Green Technology for the Isolation of Bioactive Compounds from Saffron (Crocus Sativus L.) Floral By-Products. Foods 2022, 11, 2335. [Google Scholar] [CrossRef] [PubMed]
- Farag, M.A.; Sheashea, M.; Zhao, C.; Maamoun, A.A. UV Fingerprinting Approaches for Quality Control Analyses of Food and Functional Food Coupled to Chemometrics: A Comprehensive Analysis of Novel Trends and Applications. Foods 2022, 11, 2867. [Google Scholar] [CrossRef]
- Cerdà, V.; Phansi, P.; Ferreira, S. From Mono- to Multicomponent Methods in UV-VIS Spectrophotometric and Fluorimetric Quantitative Analysis—A Review. TrAC Trends Anal. Chem. 2022, 157, 116772. [Google Scholar] [CrossRef]
- Liauw, M.A.; Baylor, L.C.; O’Rourke, P.E. UV-Visible Spectroscopy for On-Line Analysis. In Process Analytical Technology: Spectroscopic Tools and Implementation Strategies for the Chememical and Pharmaceutical Industries, 2nd ed.; John Wiley & Sons: Hoboken, NJ, USA, 2010; pp. 81–106. [Google Scholar] [CrossRef]
- Claßen, J.; Aupert, F.; Reardon, K.F.; Solle, D.; Scheper, T. Spectroscopic Sensors for In-Line Bioprocess Monitoring in Research and Pharmaceutical Industrial Application. Anal. Bioanal. Chem. 2017, 409, 651–666. [Google Scholar] [CrossRef]
- Roberts, J.; Power, A.; Chapman, J.; Chandra, S.; Cozzolino, D. The Use of UV-Vis Spectroscopy in Bioprocess and Fermentation Monitoring. Fermentation 2018, 4, 18. [Google Scholar] [CrossRef]
- Bai, S.; Serien, D.; Hu, A.; Sugioka, K. 3D Microfluidic Surface-Enhanced Raman Spectroscopy (SERS) Chips Fabricated by All-Femtosecond-Laser-Processing for Real-Time Sensing of Toxic Substances. Adv. Funct. Mater. 2018, 28, 1706262. [Google Scholar] [CrossRef]
- Yue, J.; Schouten, J.C.; Alexander Nijhuis, T. Integration of Microreactors with Spectroscopic Detection for Online Reaction Monitoring and Catalyst Characterization. Ind. Eng. Chem. Res. 2012, 51, 14583–14609. [Google Scholar] [CrossRef]
- Jurinovich, S.; Domenici, V. Digital Tool for the Analysis of UV-Vis Spectra of Olive Oils and Educational Activities with High School and Undergraduate Students. J. Chem. Educ. 2022, 99, 787–798. [Google Scholar] [CrossRef]
- Hong, T.; Yin, J.Y.; Nie, S.P.; Xie, M.Y. Applications of Infrared Spectroscopy in Polysaccharide Structural Analysis: Progress, Challenge and Perspective. Food Chem. X 2021, 12, 100168. [Google Scholar] [CrossRef]
- Joshi, R.; Sathasivam, R.; Park, S.U.; Lee, H.; Kim, M.S.; Baek, I.; Cho, B.K. Application of Fourier Transform Infrared Spectroscopy and Multivariate Analysis Methods for the Non-Destructive Evaluation of Phenolics Compounds in Moringa Powder. Agriculture 2022, 12, 10. [Google Scholar] [CrossRef]
- Günzler, H.; Gremlich, H.-U. IR Spectroscopy: An Introduction; Wiley-VCH: Weinheim, Germany, 2002; p. 361. [Google Scholar]
- Türker-Kaya, S.; Huck, C.W. A Review of Mid-Infrared and Near-Infrared Imaging: Principles, Concepts and Applications in Plant Tissue Analysis. Molecules 2017, 22, 168. [Google Scholar] [CrossRef] [PubMed]
- Toyama, T.; Fujioka, J.; Watanabe, K.; Yoshida, A.; Sakuma, T.; Inaba, K.; Imai, T.; Nakajima, T.; Tsukiyama, K.; Hamada, N.; et al. Investigation of Bactericidal Effect of a Mid-Infrared Free Electron Laser on Escherichia Coli. Sci. Rep. 2022, 12, 18111. [Google Scholar] [CrossRef]
- Magnussen, E.A.; Zimmermann, B.; Blazhko, U.; Dzurendova, S.; Dupuy–Galet, B.; Byrtusova, D.; Muthreich, F.; Tafintseva, V.; Liland, K.H.; Tøndel, K.; et al. Deep Learning-Enabled Inference of 3D Molecular Absorption Distribution of Biological Cells from IR Spectra. Commun. Chem. 2022, 5, 175. [Google Scholar] [CrossRef] [PubMed]
- Stiegler, J.M.; Abate, Y.; Cvitkovic, A.; Romanyuk, Y.E.; Huber, A.J.; Leone, S.R.; Hillenbrand, R. Nanoscale Infrared Absorption Spectroscopy of Individual Nanoparticles Enabled by Scattering-Type near-Field Microscopy. ACS Nano 2011, 5, 6494–6499. [Google Scholar] [CrossRef]
- Khan, S.A.; Khan, S.B.; Khan, L.U.; Farooq, A.; Akhtar, K.; Asiri, A.M. Fourier Transform Infrared Spectroscopy: Fundamentals and Application in Functional Groups and Nanomaterials Characterization. In Handbook of Materials Characterization; Springer: Cham, Switzerland, 2018; pp. 317–344. [Google Scholar] [CrossRef]
- Palou, A.; Miró, A.; Blanco, M.; Larraz, R.; Gómez, J.F.; Martínez, T.; González, J.M.; Alcalà, M. Calibration Sets Selection Strategy for the Construction of Robust PLS Models for Prediction of Biodiesel/Diesel Blends Physico-Chemical Properties Using NIR Spectroscopy. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2017, 180, 119–126. [Google Scholar] [CrossRef]
- de Carvalho Rocha, W.F.; Sheen, D.A. Determination of Physicochemical Properties of Petroleum Derivatives and Biodiesel Using GC/MS and Chemometric Methods with Uncertainty Estimation. Fuel 2019, 243, 413–422. [Google Scholar] [CrossRef]
- López-Fernández, J.; Moya, D.; Dolors Benaiges, M.; Valero, F.; Alcalà, M. Near Infrared Spectroscopy: A Useful Technique for Inline Monitoring of the Enzyme Catalyzed Biosynthesis of Third-Generation Biodiesel from Waste Cooking Oil. Fuel 2022, 319, 123794. [Google Scholar] [CrossRef]
- Reich, G. Near-Infrared Spectroscopy and Imaging: Basic Principles and Pharmaceutical Applications. Adv. Drug Deliv. Rev. 2005, 57, 1109–1143. [Google Scholar] [CrossRef]
- Menezes, J.C.; Ferreira, A.P.; Rodrigues, L.O.; Brás, L.P.; Alves, T.P. Chemometrics Role within the PAT Context: Examples from Primary Pharmaceutical Manufacturing. Compr. Chemom. 2009, 4, 313–355. [Google Scholar] [CrossRef]
- Ozaki, Y.; Huck, C.W.; Beć, K.B. Near-IR Spectroscopy and Its Applications. Mol. Laser Spectrosc. Adv. Appl. 2018, 11–38. [Google Scholar] [CrossRef]
- Siesler, A.H.W.; Ozaki, Y.; Kawata, S.; Heise, H.M. Near-Infrared Spectroscopy. Principles, Instruments, Applications. H. W. Siesler, Y. Ozaki, S. Kawata and H. M. Heise (Eds), Wiley-VCH, Weinheim, 2002, ISBN 3-527-30149-6, 348 Pp, £ 70.00. J. Chemom. 2002, 16, 636–638. [Google Scholar] [CrossRef]
- Blanco, M.; Romero, M.A. Near-Infrared Libraries in the Pharmaceutical Industry: A Solution for Identity Confirmation. Analyst 2001, 126, 2212–2217. [Google Scholar] [CrossRef] [PubMed]
- Beć, K.B.; Grabska, J.; Huck, C.W. Principles and Applications of Miniaturized Near-Infrared (NIR) Spectrometers. Chem. Eur. J. 2021, 27, 1514–1532. [Google Scholar] [CrossRef]
- Workman, J., Jr.; Weyer, L. Practical Guide and Spectral Atlas for Interpretive Near-Infrared Spectroscopy. In Practical Guide and Spectral Atlas for Interpretive Near-Infrared Spectroscscopy, 2nd ed.; CRC Press: Boca Raton, FL, USA, 2012; 326 p. [Google Scholar] [CrossRef]
- Jue, T.; Masuda, K. Application of near Infrared Spectroscopy in Biomedicine. In Application of Near Infrared Spectroscopy in Biomedicine; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2013; pp. 1–151. [Google Scholar] [CrossRef]
- Czarnecki, M.A.; Morisawa, Y.; Futami, Y.; Ozaki, Y. Advances in Molecular Structure and Interaction Studies Using Near-Infrared Spectroscopy. Chem. Rev. 2015, 115, 9707–9744. [Google Scholar] [CrossRef] [PubMed]
- Pinzi, S.; Alonso, F.; García Olmo, J.; Dorado, M.P. Near Infrared Reflectance Spectroscopy and Multivariate Analysis to Monitor Reaction Products during Biodiesel Production. Fuel 2012, 92, 354–359. [Google Scholar] [CrossRef]
- Blanco, M.; Alcalá, M.; González, J.M.; Torras, E. A Process Analytical Technology Approach Based on near Infrared Spectroscopy: Tablet Hardness, Content Uniformity, and Dissolution Test Measurements of Intact Tablets. J. Pharm. Sci. 2006, 95, 2137–2144. [Google Scholar] [CrossRef]
- Kumaravelu, C.; Gopal, A. A Review on the Applications of Near-Infrared Spectrometer and Chemometrics for the Agro-Food Processing Industries. In Proceedings of the 2015 IEEE Technological Innovation in ICT for Agriculture and Rural Development (TIAR), Chennai, India, 10–12 July 2015; pp. 8–12. [Google Scholar] [CrossRef]
- Alcalà, M.; Blanco, M.; Menezes, J.C.; Felizardo, P.M.; Garrido, A.; Pérez, D.; Zamora, E.; Pasquini, C.; Romañach, R.J. Near-Infrared Spectroscopy in Laboratory and Process Analysis. In Encyclopedia of Analytical Chemistry: Applications, Theory and Instrumentation; John Wiley & Sons: Hoboken, NJ, USA, 2012. [Google Scholar] [CrossRef]
- Rantanen, J. Process Analytical Applications of Raman Spectroscopy. J. Pharm. Pharmacol. 2010, 59, 171–177. [Google Scholar] [CrossRef]
- Kong, K.; Kendall, C.; Stone, N.; Notingher, I. Raman Spectroscopy for Medical Diagnostics—From in-Vitro Biofluid Assays to in-Vivo Cancer Detection. Adv. Drug Deliv. Rev. 2015, 89, 121–134. [Google Scholar] [CrossRef]
- Yang, D.; Ying, Y. Applications of Raman Spectroscopy in Agricultural Products and Food Analysis: A Review. Appl. Spectrosc. Rev. 2011, 46, 539–560. [Google Scholar] [CrossRef]
- Poornima Parvathi, V.; Parimaladevi, R.; Vasant, S.; Umadevi, M. Graphene Boosted Silver Nanoparticles as Surface Enhanced Raman Spectroscopic Sensors and Photocatalysts for Removal of Standard and Industrial Dye Contaminants. Sens. Actuators B Chem. 2019, 281, 679–688. [Google Scholar] [CrossRef]
- Shipp, D.W.; Sinjab, F.; Notingher, I. Raman Spectroscopy: Techniques and Applications in the Life Sciences. Adv. Opt. Photonics 2017, 9, 315–428. [Google Scholar] [CrossRef]
- Orlando, A.; Franceschini, F.; Muscas, C.; Pidkova, S.; Bartoli, M.; Rovere, M.; Tagliaferro, A. A Comprehensive Review on Raman Spectroscopy Applications. Chemosensors 2021, 9, 262. [Google Scholar] [CrossRef]
- Ida, N. Electromagnetic Waves and Propagation. In Engineering Electromagnetics; Springer: Cham, Switzerland, 2015; pp. 597–663. [Google Scholar] [CrossRef]
- Vaškova, H., II. Raman And Infrared Spectroscopy. Int. J. Math. Model. Methods Appl. Sci. A 2011, 5, 1205–1212. [Google Scholar]
- Garcia-Rico, E.; Alvarez-Puebla, R.A.; Guerrini, L. Direct Surface-Enhanced Raman Scattering (SERS) Spectroscopy of Nucleic Acids: From Fundamental Studies to Real-Life Applications. Chem. Soc. Rev. 2018, 47, 4909–4923. [Google Scholar] [CrossRef]
- Abalde-Cela, S.; Aldeanueva-Potel, P.; Mateo-Mateo, C.; Rodríguez-Lorenzo, L.; Alvarez-Puebla, R.A.; Liz-Marzán, L.M. Surface-Enhanced Raman Scattering Biomedical Applications of Plasmonic Colloidal Particles. J. R. Soc. Interface 2010, 7 (Suppl. S4), S435–S450. [Google Scholar] [CrossRef]
- Stöckle, R.M.; Suh, Y.D.; Deckert, V.; Zenobi, R. Nanoscale Chemical Analysis by Tip-Enhanced Raman Spectroscopy. Chem. Phys. Lett. 2000, 318, 131–136. [Google Scholar] [CrossRef]
- Liu, H.; Zhang, Z.; Liu, S.; Yan, L.; Liu, T.; Zhang, T. Joint Baseline-Correction and Denoising for Raman Spectra. Appl. Spectrosc. 2015, 69, 1013–1022. [Google Scholar] [CrossRef]
- Guo, S.; Bocklitz, T.; Popp, J. Optimization of Raman-Spectrum Baseline Correction in Biological Application. Analyst 2016, 141, 2396–2404. [Google Scholar] [CrossRef] [PubMed]
- De Beer, T.R.M.; Vergote, G.J.; Baeyens, W.R.G.; Remon, J.P.; Vervaet, C.; Verpoort, F. Development and Validation of a Direct, Non-Destructive Quantitative Method for Medroxyprogesterone Acetate in a Pharmaceutical Suspension Using FT-Raman Spectroscopy. Eur. J. Pharm. Sci. 2004, 23, 355–362. [Google Scholar] [CrossRef] [PubMed]
- Jameson, C.J. Fundamental Intramolecular and Intermolecular Information from NMR in the Gas Phase. In New Developments in NMR; Jackowski, K., Jaszuński, M., Eds.; Royal Society of Chemistry: London, UK, 2016; pp. 1–51. [Google Scholar] [CrossRef]
- Tampieri, A.; Szabó, M.; Medina, F.; Gulyás, H. A Brief Introduction to the Basics of NMR Spectroscopy and Selected Examples of Its Applications to Materials Characterization. Phys. Sci. Rev. 2021, 6, 20190086. [Google Scholar] [CrossRef]
- Westphal, H.; Warias, R.; Becker, H.; Spanka, M.; Ragno, D.; Gläser, R.; Schneider, C.; Massi, A.; Belder, D. Unveiling Organocatalysts Action–Investigating Immobilized Catalysts at Steady-State Operation via Lab-on-a-Chip Technology. Chem. Cat. Chem. 2021, 13, 5089–5096. [Google Scholar] [CrossRef]
- Giraudeau, P.; Frydman, L. Single-Scan 2D NMR: An Emerging Tool in Analytical Spectroscopy. Annu. Rev. Anal. Chem. 2014, 7, 129. [Google Scholar] [CrossRef]
- Jacquemmoz, C.; Giraud, F.; Dumez, J.-N. Online Reaction Monitoring by Single-Scan 2D NMR Under Flow Conditions. Analyst 2020, 145, 478–485. [Google Scholar] [CrossRef]
- Kazimierczuk, K.; Orekhov, V. Non-Uniform Sampling: Post-Fourier Era of NMR Data Collection and Processing. Magn. Reson. Chem. 2015, 53, 921–926. [Google Scholar] [CrossRef]
- Kang, C. Applications of In-Cell NMR in Structural Biology and Drug Discovery. Int. J. Mol. Sci. 2019, 20, 139. [Google Scholar] [CrossRef]
- Matlahov, I.; van der Wel, P.C.A. Hidden Motions and Motion-Induced Invisibility: Dynamics-Based Spectral Editing in Solid-State NMR. Methods 2018, 148, 123–135. [Google Scholar] [CrossRef]
- Jaroniec, C.P.; MacPhee, C.E.; Bajaj, V.S.; McMahon, M.T.; Dobson, C.M.; Griffin, R.G. High-Resolution Molecular Structure of a Peptide in an Amyloid Fibril Determined by Magic Angle Spinning NMR Spectroscopy. Proc. Natl. Acad. Sci. USA 2004, 101, 711–716. [Google Scholar] [CrossRef]
- Oosthoek-De Vries, A.J.; Bart, J.; Tiggelaar, R.M.; Janssen, J.W.G.; Van Bentum, P.J.M.; Gardeniers, H.J.G.E.; Kentgens, A.P.M. Continuous Flow 1H and 13C NMR Spectroscopy in Microfluidic Stripline NMR Chips. Anal. Chem. 2017, 89, 2296–2303. [Google Scholar] [CrossRef] [PubMed]
- Smits, J.; Damron, J.T.; Kehayias, P.; McDowell, A.F.; Mosavian, N.; Fescenko, I.; Ristoff, N.; Laraoui, A.; Jarmola, A.; Acosta, V.M. Two-Dimensional Nuclear Magnetic Resonance Spectroscopy with a Microfluidic Diamond Quantum Sensor. Sci. Adv. 2019, 5, eaaw7895. [Google Scholar] [CrossRef] [PubMed]
- Glish, G.L.; Vachet, R.W. The Basics of Mass Spectrometry in the Twenty-First Century. Nat. Rev. Drug Discov. 2003, 2, 140–150. [Google Scholar] [CrossRef] [PubMed]
- Ho, C.S.; Lam, C.W.K.; Chan, M.H.M.; Cheung, R.C.K.; Law, L.K.; Lit, L.C.W.; Ng, K.F.; Suen, M.W.M.; Tai, H.L. Electrospray Ionisation Mass Spectrometry: Principles and Clinical Applications. Clin. Biochem. Rev. 2003, 24, 3. [Google Scholar]
- Pomastowski, P.; Buszewski, B. Complementarity of Matrix- and Nanostructure-Assisted Laser Desorption/Ionization Approaches. Nanomaterials 2019, 9, 260. [Google Scholar] [CrossRef]
- Hosseini, S.; Martinez-Chapa, S.O. Principles and Mechanism of MALDI-ToF-MS Analysis. Briefs Appl. Sci. Technol. 2017, 1–19. [Google Scholar] [CrossRef]
- Li, D.; Yi, J.; Han, G.; Qiao, L. MALDI-TOF Mass Spectrometry in Clinical Analysis and Research. ACS Meas. Sci. Au 2022, 2, 385–404. [Google Scholar] [CrossRef]
- Duncan, M.W.; Roder, H.; Hunsucker, S.W. Quantitative Matrix-Assisted Laser Desorption/Ionization Mass Spectrometry. Brief. Funct. Genomics Proteomics 2008, 7, 355. [Google Scholar] [CrossRef]
- Bae, Y.J.; Kim, M.S. A Thermal Mechanism of Ion Formation in MALDI. Annu. Rev. Anal. Chem. 2015, 8, 41–60. [Google Scholar] [CrossRef]
- Darie-Ion, L.; Whitham, D.; Jayathirtha, M.; Rai, Y.; Neagu, A.N.; Darie, C.C.; Petre, B.A. Applications of MALDI-MS/MS-Based Proteomics in Biomedical Research. Molecules 2022, 27, 6196. [Google Scholar] [CrossRef]
- Gopal, J.; Hasan, N.; Wu, H.F. Fabrication of Titanium Based MALDI Bacterial Chips for Rapid, Sensitive and Direct Analysis of Pathogenic Bacteria. Biosens. Bioelectron. 2013, 39, 57–63. [Google Scholar] [CrossRef] [PubMed]
- Manikandan, M.; Hasan, N.; Wu, H.F. Platinum Nanoparticles for the Photothermal Treatment of Neuro 2A Cancer Cells. Biomaterials 2013, 34, 5833–5842. [Google Scholar] [CrossRef] [PubMed]
- Torres-Sangiao, E.; Leal Rodriguez, C.; García-riestra, C. Application and Perspectives of Maldi–Tof Mass Spectrometry in Clinical Microbiology Laboratories. Microorganisms 2021, 9, 1539. [Google Scholar] [CrossRef] [PubMed]
- Buchberger, A.R.; DeLaney, K.; Johnson, J.; Li, L. Mass Spectrometry Imaging: A Review of Emerging Advancements and Future Insights. Anal. Chem. 2018, 90, 240–265. [Google Scholar] [CrossRef]
- Tuck, M.; Grélard, F.; Blanc, L.; Desbenoit, N. MALDI-MSI Towards Multimodal Imaging: Challenges and Perspectives. Front. Chem. 2022, 10, 4688. [Google Scholar] [CrossRef]
- Konermann, L.; Ahadi, E.; Rodriguez, A.D.; Vahidi, S. Unraveling the Mechanism of Electrospray Ionization. Anal. Chem. 2013, 85, 2–9. [Google Scholar] [CrossRef]
- Daub, C.D.; Cann, N.M. How Are Completely Desolvated Ions Produced in Electrospray Ionization: Insights from Molecular Dynamics Simulations. Anal. Chem. 2011, 83, 8372–8376. [Google Scholar] [CrossRef]
- Banerjee, S.; Mazumdar, S. Electrospray Ionization Mass Spectrometry: A Technique to Access the Information beyond the Molecular Weight of the Analyte. Int. J. Anal. Chem. 2012, 2012, 282574. [Google Scholar] [CrossRef]
- Murayama, C.; Kimura, Y.; Setou, M. Imaging Mass Spectrometry: Principle and Application. Biophys. Rev. 2009, 1, 131–139. [Google Scholar] [CrossRef]
- Shimma, S. Mass Spectrometry Imaging. Mass Spectrom. 2022, 11, A0102. [Google Scholar] [CrossRef]
- Kumar, B.R. Application of HPLC and ESI-MS Techniques in the Analysis of Phenolic Acids and Flavonoids from Green Leafy Vegetables (GLVs). J. Pharm. Anal. 2017, 7, 349. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Šimek, H.; Ilioae, D.; Jung, N.; Bräse, S.; Zappe, H.; Dittmeyer, R.; Ladewig, B.P. In Situ Sensors for Flow Reactors—A Review. React. Chem. Eng. 2021, 6, 1497–1507. [Google Scholar] [CrossRef]
- Rizkin, B.A.; Popovic, F.G.; Hartman, R.L. Review Article: Spectroscopic Microreactors for Heterogeneous Catalysis. J. Vac. Sci. Technol. A Vacuum Surfaces Film. 2019, 37, 050801. [Google Scholar] [CrossRef]
- Li, D.E.; Lin, C.H. Microfluidic Chip for Droplet-Based AuNP Synthesis with Dielectric Barrier Discharge Plasma and on-Chip Mercury Ion Detection. RSC Adv. 2018, 8, 16139–16145. [Google Scholar] [CrossRef]
- Bressan, L.P.; Robles-Najar, J.; Adamo, C.B.; Quero, R.F.; Costa, B.M.C.; de Jesus, D.P.; da Silva, J.A.F. 3D-Printed Microfluidic Device for the Synthesis of Silver and Gold Nanoparticles. Microchem. J. 2019, 146, 1083–1089. [Google Scholar] [CrossRef]
- Duncombe, T.A.; Ponti, A.; Seebeck, F.P.; Dittrich, P.S. UV-Vis Spectra-Activated Droplet Sorting for Label-Free Chemical Identification and Collection of Droplets. Anal. Chem. 2021, 93, 13008–13013. [Google Scholar] [CrossRef]
- Bhat, M.P.; Kurkuri, M.; Losic, D.; Kigga, M.; Altalhi, T. New Optofluidic Based Lab-on-a-Chip Device for the Real-Time Fluoride Analysis. Anal. Chim. Acta 2021, 1159, 338439. [Google Scholar] [CrossRef]
- Rodríguez-Ruiz, I.; Lamadie, F.; Charton, S. Uranium(VI) On-Chip Microliter Concentration Measurements in a Highly Extended UV-Visible Absorbance Linearity Range. Anal. Chem. 2018, 90, 2456–2460. [Google Scholar] [CrossRef]
- Adbolusattar, I.; Hadi, H. Paper microfluidic device and spectrophotometry methods for colorimetric detection of phenylephrine hydrochloride in pure and pharmaceutical preparations. Bull. Chem. Soc. Ethiop. 2022, 36, 727–736. [Google Scholar]
- Suarnaba, E.G.T.; Lee, Y.F.; Yamada, H.; Tagawa, T. Ultraviolet-Visible (UV-Vis) Microspectroscopic System Designed for the in Situ Characterization of the Dehydrogenation Reaction over Platinum Supported Catalytic Microchannel Reactor. Appl. Spectrosc. 2016, 70, 1806–1812. [Google Scholar] [CrossRef]
- Ponce, S.; Munoz, M.; Cubillas, A.M.; Euser, T.G.; Zhang, G.R.; Russell, P.S.J.; Wasserscheid, P.; Etzold, B.J.M. Stable Immobilization of Size-Controlled Bimetallic Nanoparticles in Photonic Crystal Fiber Microreactor. Chem.Ing. Tech. 2018, 90, 653–659. [Google Scholar] [CrossRef]
- Aillet, T.; Loubiere, K.; Dechy-Cabaret, O.; Laurent, P. Microreactors as a Tool for Acquiring Kinetic Data on Photochemical Reactions. Chem. Eng. Technol. 2016, 39, 115–122. [Google Scholar] [CrossRef]
- Ye, W.Q.; Wei, Y.Y.; Wang, D.N.; Yang, C.G.; Xu, Z.R. A Digital Microfluidic Platform Based on a Near-Infrared Light-Responsive Shape-Memory Micropillar Array. Lab Chip 2021, 21, 1131–1138. [Google Scholar] [CrossRef] [PubMed]
- Isikawa, M.; Guidelli, E. Microfluidic Synthesis of Theranostic Nanoparticles with Near-Infrared Scintillation: Toward Next-Generation Dosimetry in X-Ray-Induced Photodynamic Therapy. ACS Appl. Mater. Interfaces 2022, 14, 324–336. [Google Scholar] [CrossRef] [PubMed]
- Bello, V.; Bodo, E. A NIR-Spectroscopy-Based Approach for Detection of Fluids in Rectangular Glass Micro-Capillaries. Eng. Proc. 2020, 2, 43. [Google Scholar] [CrossRef]
- Iiyama, T.; Furuya, M.; Arai, T. Near-Infrared Imaging to Quantify the Diffusion Coefficient of Sodium Pentaborate Aqueous Solution in a Microchannel. Chem. Eng. Sci. 2022, 254, 117630. [Google Scholar] [CrossRef]
- Uema, T.; Ohata, T.; Washizuka, Y.; Nakanishi, R.; Kawashima, D.; Kakuta, N. Near-Infrared Imaging in a Microfluidic Channel of Aqueous Acid–Base Reactions. Chem. Eng. J. 2021, 403, 126338. [Google Scholar] [CrossRef]
- Galaverna, R.; Ribessi, R.L.; Rohwedder, J.J.R.; Pastre, J.C. Coupling Continuous Flow Microreactors to MicroNIR Spectroscopy: Ultracompact Device for Facile In-Line Reaction Monitoring. Org. Process Res. Dev. 2018, 22, 780–788. [Google Scholar] [CrossRef]
- Loiland, J.A.; Lobo, R.F. Oxidation of Zeolite Acid Sites in NO/O2 Mixtures and the Catalytic Properties of the New Site in NO Oxidation. J. Catal. 2015, 325, 68–78. [Google Scholar] [CrossRef]
- Fath, V.; Lau, P.; Greve, C.; Kockmann, N.; Röder, T. Efficient Kinetic Data Acquisition and Model Prediction: Continuous Flow Microreactors, Inline Fourier Transform Infrared Spectroscopy, and Self-Modeling Curve Resolution. Org. Process Res. Dev. 2020, 24, 1955–1968. [Google Scholar] [CrossRef]
- Fath, V.; Lau, P.; Greve, C.; Weller, P.; Kockmann, N.; Röder, T. Simultaneous Self-Optimisation of Yield and Purity through Successive Combination of Inline FT-IR Spectroscopy and Online Mass Spectrometry in Flow Reactions. J. Flow Chem. 2021, 11, 285–302. [Google Scholar] [CrossRef]
- Chevalier, S.; Tourvieille, J.N.; Sommier, A.; Pradère, C. Infrared Thermospectroscopic Imaging of Heat and Mass Transfers in Laminar Microfluidic Reactive Flows. Chem. Eng. J. Adv. 2021, 8, 100166. [Google Scholar] [CrossRef]
- Ryu, M.; Kimber, J.A.; Sato, T.; Nakatani, R.; Hayakawa, T.; Romano, M.; Pradere, C.; Hovhannisyan, A.A.; Kazarian, S.G.; Morikawa, J. Infrared Thermo-Spectroscopic Imaging of Styrene Radical Polymerization in Microfluidics. Chem. Eng. J. 2017, 324, 259–265. [Google Scholar] [CrossRef]
- Nelson, G.L.; Lines, A.M.; Bello, J.M.; Bryan, S.A. Online Monitoring of Solutions within Microfluidic Chips: Simultaneous Raman and Uv-Vis Absorption Spectroscopies. ACS Sens. 2019, 4, 2288–2295. [Google Scholar] [CrossRef]
- Mattio, E.; Caleyron, A.; Miguirditchian, M.; Lines, A.M.; Bryan, S.A.; Lackey, H.E.; Rodriguez-Ruiz, I.; Lamadie, F. Microfluidic In-Situ Spectrophotometric Approaches to Tackle Actinides Analysis in Multiple Oxidation States. Appl. Spectrosc. 2022, 76, 580–589. [Google Scholar] [CrossRef]
- Nelson, G.L.; Asmussen, S.E.; Lines, A.M.; Casella, A.J.; Bottenus, D.R.; Clark, S.B.; Bryan, S.A. Micro-Raman Technology to Interrogate Two-Phase Extraction on a Microfluidic Device. Anal. Chem. 2018, 90, 8345–8353. [Google Scholar] [CrossRef]
- Lines, A.M.; Nelson, G.L.; Casella, A.J.; Bello, J.M.; Clark, S.B.; Bryan, S.A. Multivariate Analysis to Quantify Species in the Presence of Direct Interferents: Micro-Raman Analysis of HNO3 in Microfluidic Devices. Anal. Chem. 2018, 90, 2548–2554. [Google Scholar] [CrossRef]
- Nelson, G.L.; Lines, A.M.; Casella, A.J.; Bello, J.M.; Bryan, S.A. Development and Testing of a Novel Micro-Raman Probe and Application of Calibration Method for the Quantitative Analysis of Microfluidic Nitric Acid Streams. Analyst 2018, 143, 1188–1196. [Google Scholar] [CrossRef]
- Engeldinger, J.; Radnik, J.; Kreyenschulte, C.; Devred, F.; Gaigneaux, E.M.; Fischer, A.; Zanthoff, H.W.; Bentrup, U. Probing the Structural Changes and Redox Behavior of Mixed Molybdate Catalysts under Ammoxidation Conditions: An Operando Raman Spectroscopy Study. ChemCatChem 2016, 8, 976–983. [Google Scholar] [CrossRef]
- Gentleman, A.S.; Miele, E.; Lawson, T.; Kohler, P.; Kim, S.; Yousaf, S.; Garcia, D.A.; Lage, A.; Grey, C.P.; Baumberg, J.J.; et al. In-Situ Raman Spectroscopy of Reaction Products in Optofluidic Hollow-Core Fiber Microreactors. In Proceedings of the 14th Pacific Rim Conference on Lasers and Electro-Optics (CLEO PR 2020), Sydney, Australia, 3–5 August 2020. [Google Scholar] [CrossRef]
- Pinho, B.; Hartman, R.L. Microfluidics with in Situ Raman Spectroscopy for the Characterization of Non-Polar/Aqueous Interfaces. React. Chem. Eng. 2017, 2, 189–200. [Google Scholar] [CrossRef]
- Zhu, J.; Chen, Q.; Kutsanedzie, F.Y.H.; Yang, M.; Ouyang, Q.; Jiang, H. Highly Sensitive and Label-Free Determination of Thiram Residue Using Surface-Enhanced Raman Spectroscopy (SERS) Coupled with Paper-Based Microfluidics. Anal. Methods 2017, 9, 6186–6193. [Google Scholar] [CrossRef]
- Zhang, Z.; Gernert, U.; Gerhardt, R.F.; Höhn, E.M.; Belder, D.; Kneipp, J. Catalysis by Metal Nanoparticles in a Plug-In Optofluidic Platform: Redox Reactions of p-Nitrobenzenethiol and p-Aminothiophenol. ACS Catal. 2018, 8, 2443–2449. [Google Scholar] [CrossRef]
- Willner, M.R.; McMillan, K.S.; Graham, D.; Vikesland, P.J.; Zagnoni, M. Surface-Enhanced Raman Scattering Based Microfluidics for Single-Cell Analysis. Anal. Chem. 2018, 90, 12004–12010. [Google Scholar] [CrossRef] [PubMed]
- Krafft, B.; Panneerselvam, R.; Geissler, D.; Belder, D. A Microfluidic Device Enabling Surface-Enhanced Raman Spectroscopy at Chip-Integrated Multifunctional Nanoporous Membranes. Anal. Bioanal. Chem. 2020, 412, 267–277. [Google Scholar] [CrossRef]
- Nie, Y.; Jin, C.; Zhang, J.X.J. MicrofluidicIn SituPatterning of Silver Nanoparticles for Surface-Enhanced Raman Spectroscopic Sensing of Biomolecules. ACS Sens. 2021, 6, 2584–2592. [Google Scholar] [CrossRef]
- Bi, H.; Fernandes, A.C.; Cardoso, S.; Freitas, P. Interference-Blind Microfluidic Sensor for Ascorbic Acid Determination by UV/Vis Spectroscopy. Sens. Actuators B Chem. 2016, 224, 668–675. [Google Scholar] [CrossRef]
- Cao, E.; Brett, G.; Miedziak, P.J.; Douthwaite, J.M.; Barrass, S.; McMillan, P.F.; Hutchings, G.J.; Gavriilidis, A. A Micropacked-Bed Multi-Reactor System with in Situ Raman Analysis for Catalyst Evaluation. Catal. Today 2017, 283, 195–201. [Google Scholar] [CrossRef]
- Navin, C.V.; Krishna, K.S.; Theegala, C.S.; Kumar, C.S.S.R. Space and Time-Resolved Probing of Heterogeneous Catalysis Reactions Using Lab-on-a-Chip. Nanoscale 2016, 8, 5546–5551. [Google Scholar] [CrossRef]
- Lauterbach, F.; Abetz, V.; Lauterbach, F.; Abetz, V. Continuous Kinetic Sampling of Flow Polymerizations via Inline UV–Vis Spectroscopy. Macromol. Rapid Commun. 2020, 41, 2000029. [Google Scholar] [CrossRef]
- Wang, N.; Tan, F.; Zhao, Y.; Tsoi, C.C.; Fan, X.; Yu, W.; Zhang, X. Optofluidic UV-Vis Spectrophotometer for Online Monitoring of Photocatalytic Reactions. Sci. Rep. 2016, 6, 28928. [Google Scholar] [CrossRef]
- Hess, C. New Advances in Using Raman Spectroscopy for the Characterization of Catalysts and Catalytic Reactions. Chem. Soc. Rev. 2021, 50, 3519–3564. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Li, S.; Yao, F.; Bao, F.; Ge, Y.; Zou, M.; Liang, P.; Chen, Q. Progress of Microfluidics Combined with SERS Technology in the Trace Detection of Harmful Substances. Chemosensors 2022, 10, 449. [Google Scholar] [CrossRef]
- Vis, C.M.; Nieuwelink, A.E.; Weckhuysen, B.M.; Bruijnincx, P.C.A. Continuous Flow Pickering Emulsion Catalysis in Droplet Microfluidics Studied with In Situ Raman Microscopy. Chem. Eur. J. 2020, 26, 15099–15102. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Sun, H.; Yin, L.; Yuan, Y.; Xu, M.; Yao, J. On-Line Monitoring on the Micro-Synthesis of α-Phenylethanol by Microfluidic Chip Combined with Surface Enhanced Raman Spectroscopy. Acta Chim. Sin. 2019, 77, 257–262. [Google Scholar] [CrossRef]
- Wu, Y.; Jiang, Y.; Zheng, X.; Jia, S.; Zhu, Z.; Ren, B.; Ma, H. Facile Fabrication of Microfluidic Surface-Enhanced Raman Scattering Devices via Lift-up Lithography. R. Soc. Open Sci. 2018, 5, 172034. [Google Scholar] [CrossRef]
- Panneerselvam, R.; Sadat, H.; Höhn, E.M.; Das, A.; Noothalapati, H.; Belder, D. Microfluidics and Surface-Enhanced Raman Spectroscopy, a Win–Win Combination? Lab Chip 2022, 22, 665–682. [Google Scholar] [CrossRef]
- Dedovets, D.; Zhang, S.; Leng, J.; Pera-Titus, M. Microfluidic Device for Monitoring Catalytic Events on Armored Bubbles. Adv. Mater. Interfaces 2022, 9, 2200759. [Google Scholar] [CrossRef]
- Zhu, J.; Wang, F.; Chen, R.; Cheng, K.; Xu, B.; Guo, Z.; Liang, X.; Ye, M.; Zou, H. Centrifugation Assisted Microreactor Enables Facile Integration of Trypsin Digestion, Hydrophilic Interaction Chromatography Enrichment, and on-Column Deglycosylation for Rapid and Sensitive N-Glycoproteome Analysis. Anal. Chem. 2012, 84, 5146–5153. [Google Scholar] [CrossRef]
- Meller, K.; Pomastowski, P.; Szumski, M.; Buszewski, B. Preparation of an Improved Hydrophilic Monolith to Make Trypsin-Immobilized Microreactors. J. Chromatogr. B 2017, 1043, 128–137. [Google Scholar] [CrossRef]
- Wu, N.; Wang, S.; Yang, Y.; Song, J.; Su, P.; Yang, Y. DNA-Directed Trypsin Immobilization on a Polyamidoamine Dendrimer-Modified Capillary to Form a Renewable Immobilized Enzyme Microreactor. Int. J. Biol. Macromol. 2018, 113, 38–44. [Google Scholar] [CrossRef]
- Szymańska, K.; Pietrowska, M.; Kocurek, J.; Maresz, K.; Koreniuk, A.; Mrowiec-Białoń, J.; Widłak, P.; Magner, E.; Jarzebski, A. Low Back-Pressure Hierarchically Structured Multichannel Microfluidic Bioreactors for Rapid Protein Digestion—Proof of Concept. Chem. Eng. J. 2016, 287, 148–154. [Google Scholar] [CrossRef]
- Zhong, C.; Yang, B.; Huang, W.; Huang, H.; Zhang, S.; Yan, X.; Lu, Q.; Chen, Z.; Lin, Z. Self-Assembly Synthes Is of Trypsin-Immobilized Monolithic Microreactor for Fast and Efficient Proteolysis. J. Chromatogr. A 2021, 1635, 461742. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Musyimi, H.K.; Soper, S.A.; Murray, K.K. Development of an Automated Digestion and Droplet Deposition Microfluidic Chip for MALDI-TOF MS. J. Am. Soc. Mass Spectrom. 2008, 19, 964–972. [Google Scholar] [CrossRef] [PubMed]
- Jiang, H.; Zou, H.; Wang, H.; Zhang, Q.; Ni, J.; Zhang, Q.; Guo, Z.; Chen, X. Combination of MALDI-TOF Mass Spectrometry with Immobilized Enzyme Microreactor for Peptide Mapping. Sci. China Ser. B Chem. 2000, 43, 625–633. [Google Scholar] [CrossRef]
- Peterson, D.S.; Rohr, T.; Svec, F.; Fréchet, J.M.J. High-Throughput Peptide Mass Mapping Using a Microdevice Containing Trypsin Immobilized on a Porous Polymer Monolith Coupled to MALDI TOF and ESI TOF Mass Spectrometers. J. Proteome Res. 2002, 1, 563–568. [Google Scholar] [CrossRef] [PubMed]
- Gao, M.; Zhang, P.; Hong, G.; Guan, X.; Yan, G.; Deng, C.; Zhang, X. Novel Monolithic Enzymatic Microreactor Based on Single-Enzyme Nanoparticles for Highly Efficient Proteolysis and Its Application in Multidimensional Liquid Chromatography. J. Chromatogr. A 2009, 1216, 7472–7477. [Google Scholar] [CrossRef]
- Szymańska, K.; Ciemięga, A.; Maresz, K.; Pudło, W.; Malinowski, J.; Mrowiec-Białoń, J.; Jarzębski, A.B. Catalytic Functionalized Structured Monolithic Micro-/Mesoreactors: Engineering, Properties, and Performance in Flow Synthesis: An Overview and Guidelines. Front. Chem. Eng. 2021, 3, 70. [Google Scholar] [CrossRef]
- Villegas, L.; Pero-Gascon, R.; Benavente, F.; Barbosa, J.; Sanz-Nebot, V. On-Line Protein Digestion by Immobilized Enzyme Microreactor Capillary Electrophoresis-Mass Spectrometry. Talanta 2019, 199, 116–123. [Google Scholar] [CrossRef]
- Zhang, Y.; Liu, Y.; Kong, J.; Yang, P.; Tang, Y.; Liu, B. Efficient Proteolysis System: A Nanozeolite-Derived Microreactor. Small 2006, 2, 1170–1173. [Google Scholar] [CrossRef]
- Liu, J.; Lin, S.; Qi, D.; Deng, C.; Yang, P.; Zhang, X. On-Chip Enzymatic Microreactor Using Trypsin-Immobilized Superparamagnetic Nanoparticles for Highly Efficient Proteolysis. J. Chromatogr. A 2007, 1176, 169–177. [Google Scholar] [CrossRef]
- Ku, K.; Frey, C.; Arad, M.; Ghafourifar, G. Development of Novel Enzyme Immobilization Methods Employing Formaldehyde or Triethoxysilylbutyraldehyde to Fabricate Immobilized Enzyme Microreactors for Peptide Mapping. Anal. Methods 2022, 14, 4053–4063. [Google Scholar] [CrossRef] [PubMed]
- Brivio, M.; Fokkens, R.H.; Verboom, W.; Reinhoudt, D.N.; Tas, N.R.; Goedbloed, M.; Van den Berg, A. Integrated Microfluidic System Enabling (Bio)Chemical Reactions with on-Line MALDI-TOF Mass Spectrometry. Anal. Chem. 2002, 74, 3972–3976. [Google Scholar] [CrossRef] [PubMed]
- Gorbunov, A.; Bardin, A.; Ilyushonok, S.; Kovach, J.; Petrenko, A.; Sukhodolov, N.; Krasnov, K.; Krasnov, N.; Zorin, I.; Obornev, A.; et al. Multiwell Photocatalytic Microreactor Device Integrating Drug Biotransformation Modeling and Sample Preparation on a MALDI Target. Microchem. J. 2022, 178, 107362. [Google Scholar] [CrossRef]
- Zhang, S.; Yuan, H.; Zhao, B.; Zhou, Y.; Jiang, H.; Zhang, L.; Liang, Z.; Zhang, Y. Integrated Platform with a Combination of Online Digestion and 18O Labeling for Proteome Quantification via an Immobilized Trypsin Microreactor. Analyst 2015, 140, 5227–5234. [Google Scholar] [CrossRef]
- Foo, H.C.; Smith, N.W.; Stanley, S.M.R. Fabrication of an On-Line Enzyme Micro-Reactor Coupled to Liquid Chromatography–Tandem Mass Spectrometry for the Digestion of Recombinant Human Erythropoietin. Talanta 2015, 135, 18–22. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.; Zhu, G.; Dovichi, N.J. Integrated CZE-ESI-MS/MS System with an Immobilized Trypsin Microreactor for Online Digestion and Analysis of Picogram Amounts of RAW 264.7 Cell Lysate. Anal. Chem. 2013, 85, 4187. [Google Scholar] [CrossRef]
- Li, Y.; Wojcik, R.; Dovichi, N.J. A Replaceable Microreactor for On-Line Protein Digestion in a Two-Dimensional Capillary Electrophoresis System with Tandem Mass Spectrometry Detection. J. Chromatogr. A 2011, 1218, 2007–2011. [Google Scholar] [CrossRef]
- Nissilä, T.; Sainiemi, L.; Franssila, S.; Ketola, R.A. Fully Polymeric Integrated Microreactor/Electrospray Ionization Chip for on-Chip Digestion and Mass Spectrometric Analysis. Sens. Actuators B Chem. 2009, 143, 414–420. [Google Scholar] [CrossRef]
- Haven, J.J.; Vandenbergh, J.; Junkers, T. Watching Polymers Grow: Real Time Monitoring of Polymerizations via an on-Line ESI-MS/Microreactor Coupling. Chem. Commun. 2015, 51, 4611–4614. [Google Scholar] [CrossRef]
- Haven, J.J.; Baeten, E.; Claes, J.; Vandenbergh, J.; Junkers, T. High-Throughput Polymer Screening in Microreactors: Boosting the Passerini Three Component Reaction. Polym. Chem. 2017, 8, 2972–2978. [Google Scholar] [CrossRef]
- Guo, W.; Heeres, H.J.; Yue, J. Continuous Synthesis of 5-Hydroxymethylfurfural from Glucose Using a Combination of AlCl3 and HCl as Catalyst in a Biphasic Slug Flow Capillary Microreactor. Chem. Eng. J. 2020, 381, 122754. [Google Scholar] [CrossRef]


| UV-Vis Spectroscopy | NIR Spectroscopy | Raman Spectroscopy | |
|---|---|---|---|
| Advantages |
|
|
|
| Disadvantages |
|
|
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jurina, T.; Cvetnić, T.S.; Šalić, A.; Benković, M.; Valinger, D.; Kljusurić, J.G.; Zelić, B.; Jurinjak Tušek, A. Application of Spectroscopy Techniques for Monitoring (Bio)Catalytic Processes in Continuously Operated Microreactor Systems. Catalysts 2023, 13, 690. https://doi.org/10.3390/catal13040690
Jurina T, Cvetnić TS, Šalić A, Benković M, Valinger D, Kljusurić JG, Zelić B, Jurinjak Tušek A. Application of Spectroscopy Techniques for Monitoring (Bio)Catalytic Processes in Continuously Operated Microreactor Systems. Catalysts. 2023; 13(4):690. https://doi.org/10.3390/catal13040690
Chicago/Turabian StyleJurina, Tamara, Tea Sokač Cvetnić, Anita Šalić, Maja Benković, Davor Valinger, Jasenka Gajdoš Kljusurić, Bruno Zelić, and Ana Jurinjak Tušek. 2023. "Application of Spectroscopy Techniques for Monitoring (Bio)Catalytic Processes in Continuously Operated Microreactor Systems" Catalysts 13, no. 4: 690. https://doi.org/10.3390/catal13040690
APA StyleJurina, T., Cvetnić, T. S., Šalić, A., Benković, M., Valinger, D., Kljusurić, J. G., Zelić, B., & Jurinjak Tušek, A. (2023). Application of Spectroscopy Techniques for Monitoring (Bio)Catalytic Processes in Continuously Operated Microreactor Systems. Catalysts, 13(4), 690. https://doi.org/10.3390/catal13040690












