Designed Synthesis of PDI/BiOCl-BiPO4 Composited Material for Boosted Photocatalytic Contaminant Degradation
Abstract
1. Introduction
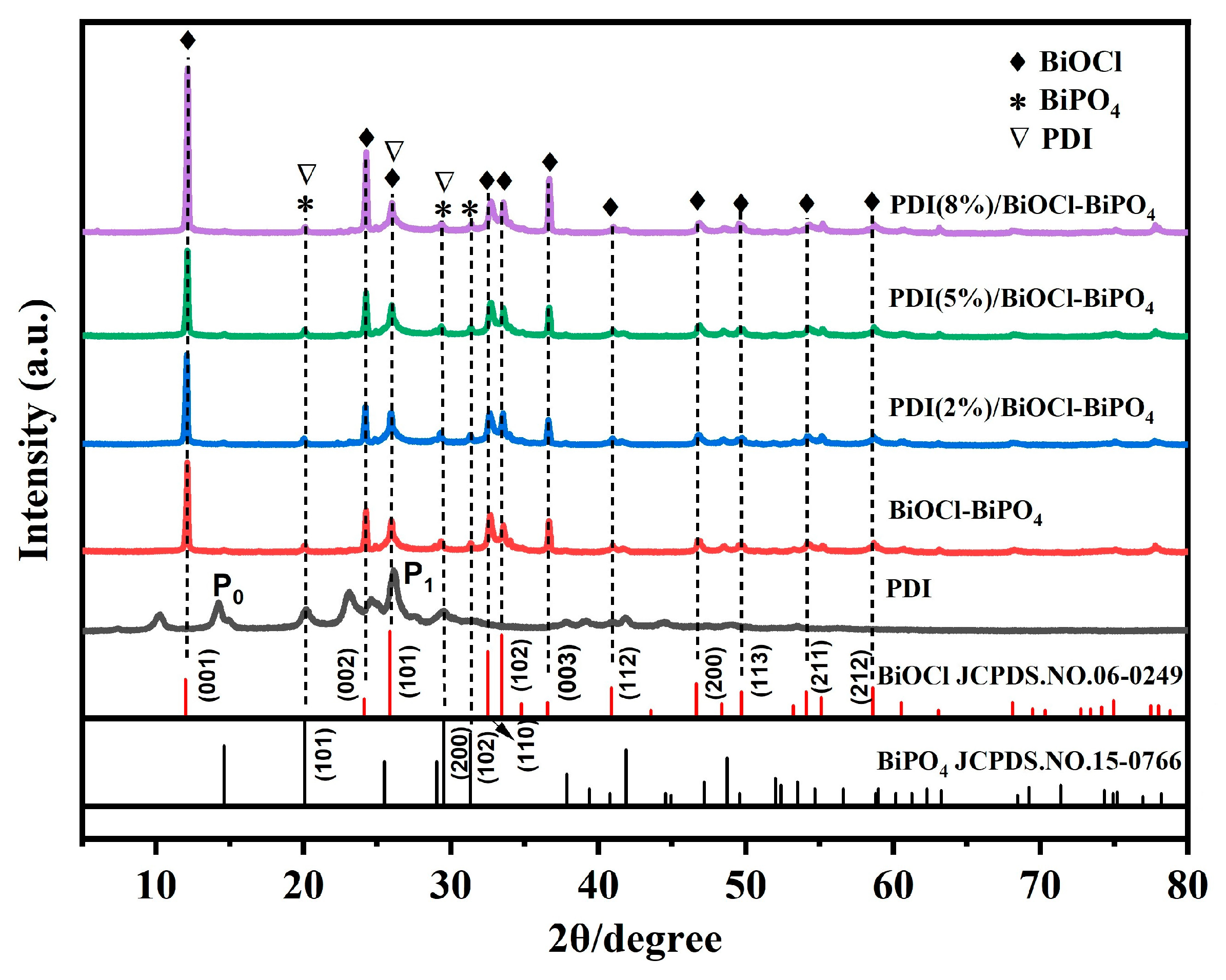
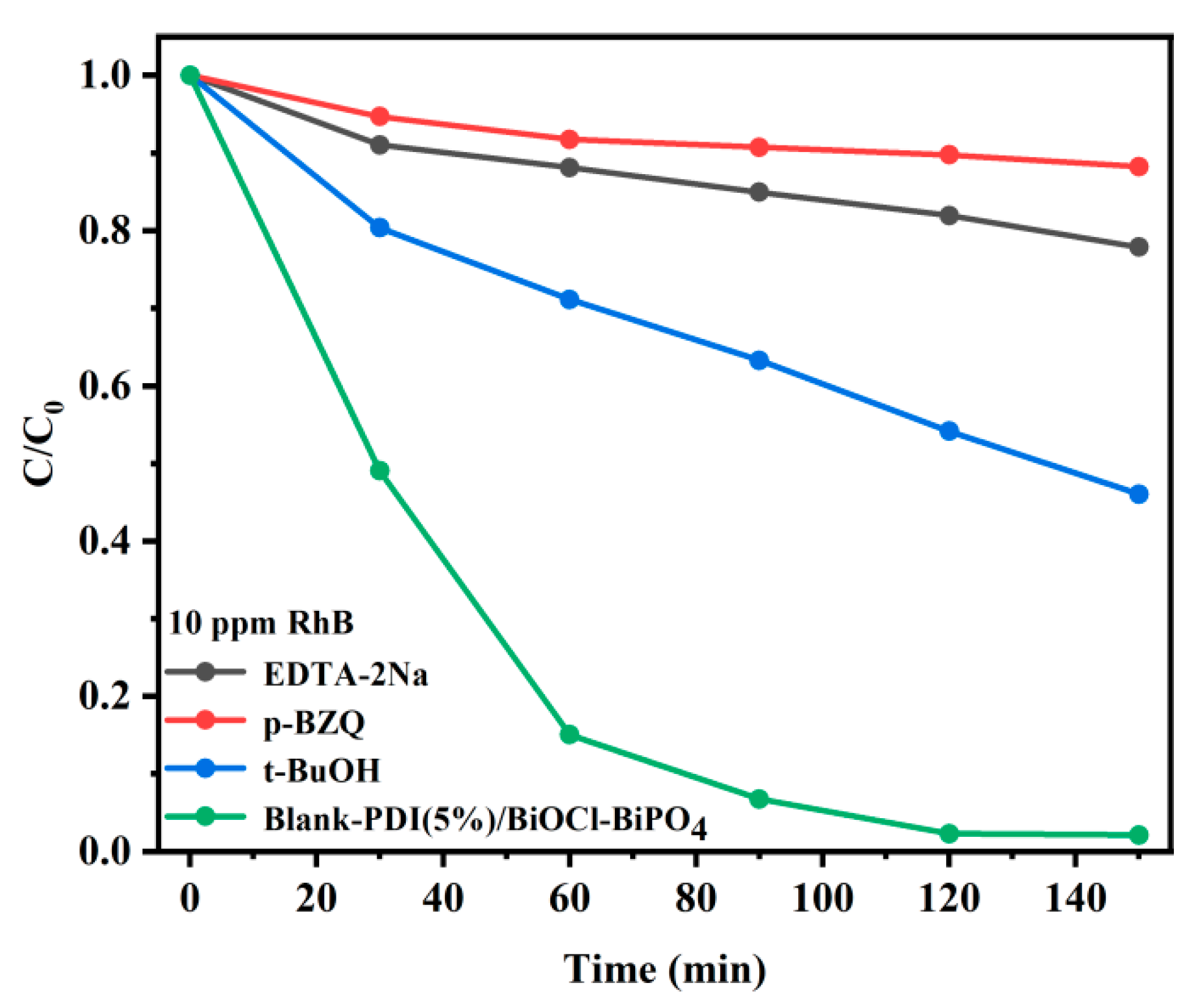
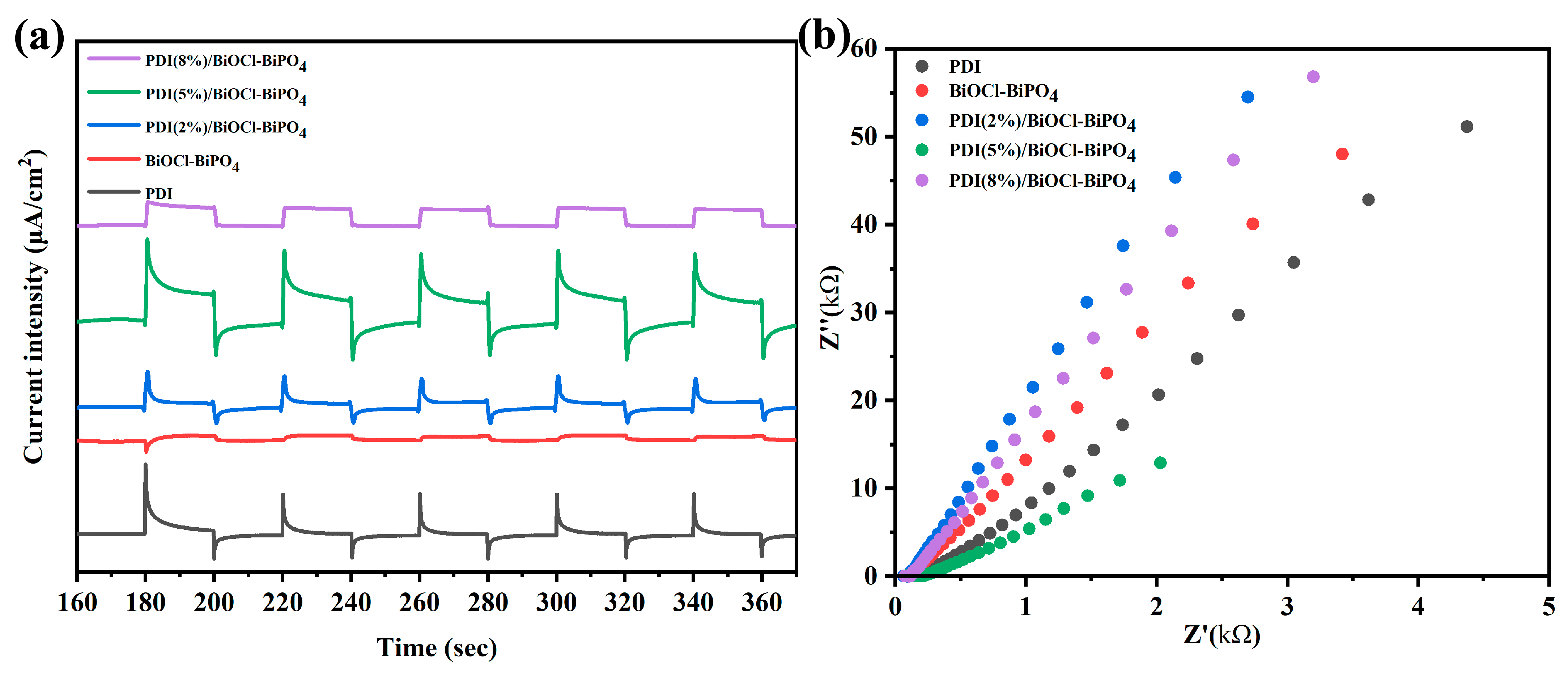
2. Results and Discussion
3. Experimental Section
3.1. Experimental Materials
3.2. Catalyst Preparation
3.3. Characterization of Samples
3.4. Photocatalytic Activity Measurement
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Wu, C.-J.; Valerie Maggay, I.; Chiang, C.-H.; Chen, W.; Chang, Y.; Hu, C.; Venault, A. Removal of tetracycline by a photocatalytic membrane reactor with MIL-53(Fe)/PVDF mixed-matrix membrane. Chem. Eng. J. 2023, 451, 138990. [Google Scholar] [CrossRef]
- Guo, F.; Huang, X.; Chen, Z.; Sun, H.; Shi, W. Investigation of visible-light-driven photocatalytic tetracycline degradation via carbon dots modified porous ZnSnO3 cubes: Mechanism and degradation pathway. Sep. Purif. Technol. 2020, 253, 117518. [Google Scholar] [CrossRef]
- Jiang, H.; Wang, Q.; Chen, P.; Zheng, H.; Shi, J.; Shu, H.; Liu, Y. Photocatalytic degradation of tetracycline by using a regenerable (Bi)BiOBr/rGO composite. J. Clean. Prod. 2022, 339, 130771. [Google Scholar] [CrossRef]
- Olusegun, S.J.; Larrea, G.; Osial, M.; Jackowska, K.; Krysinski, P. Photocatalytic Degradation of Antibiotics by Superparamagnetic Iron Oxide Nanoparticles. Tetracycline Case. Catalysts 2021, 11, 1243. [Google Scholar] [CrossRef]
- He, Z.; Siddique, M.S.; Yang, H.; Xia, Y.; Su, J.; Tang, B.; Wang, L.; Kang, L.; Huang, Z. Novel Z-scheme In2S3/Bi2WO6 core-shell heterojunctions with synergistic enhanced photocatalytic degradation of tetracycline hydrochloride. J. Clean. Prod. 2022, 339, 130634. [Google Scholar] [CrossRef]
- Cheng, M.; Zhang, Y.; Lai, B.; Wang, L.; Yang, S.; Li, K.; Wang, D.; Wu, Y.; Chen, G.-H.; Qian, J. Nitrogen and phosphorus co-doped porous carbons (NPCs) for peroxydisulfate (PDS) activation towards tetracycline degradation: Defects enhanced adsorption and non-radical mechanism dominated by electron transfer. Chem. Eng. J. 2022, 455, 140615. [Google Scholar] [CrossRef]
- Chen, W.; Huang, J.; He, Z.-C.; Ji, X.; Zhang, Y.-F.; Sun, H.-L.; Wang, K.; Su, Z.-W. Accelerated photocatalytic degradation of tetracycline hydrochloride over CuAl2O4/g-C3N4 p-n heterojunctions under visible light irradiation. Sep. Purif. Technol. 2021, 277, 119461. [Google Scholar] [CrossRef]
- Pei, C.-Y.; Chen, Y.-G.; Wang, L.; Chen, W.; Huang, G.-B. Step-scheme WO3/CdIn2S4 hybrid system with high visible light activity for tetracycline hydrochloride photodegradation. Appl. Surf. Sci. 2021, 535, 147682. [Google Scholar] [CrossRef]
- Liu, S.-y.; Zada, A.; Yu, X.; Liu, F.; Jin, G. NiFe2O4/g-C3N4 heterostructure with an enhanced ability for photocatalytic degradation of tetracycline hydrochloride and antibacterial performance. Chemosphere 2022, 307, 135717. [Google Scholar] [CrossRef]
- Zhang, X.-W.; Wang, F.; Wang, C.-C.; Wang, P.; Fu, H.; Zhao, C. Photocatalysis activation of peroxodisulfate over the supported Fe3O4 catalyst derived from MIL-88A(Fe) for efficient tetracycline hydrochloride degradation. Chem. Eng. J. 2021, 426, 131927. [Google Scholar] [CrossRef]
- Abdullah, F.H.; Bakar, N.H.H.A.; Bakar, M.A. Current advancements on the fabrication, modification, and industrial application of zinc oxide as photocatalyst in the removal of organic and inorganic contaminants in aquatic systems. J. Hazard. Mater. 2022, 424, 127416. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Zhuang, H. Recent progresses in photocatalytic hydrogen production: Design and construction of Ni-based cocatalysts. Int. J. Energy Res. 2021, 45, 1480–1495. [Google Scholar] [CrossRef]
- Zhuang, H.; Chen, X.; Xia, J.; Lu, K.; Huang, W.; Liu, X.; Yu, C.; Yang, K. State-of-the-art progress in Ag3PO4-based photocatalysts: Rational design, regulation and perspective. Appl. Mater. Today 2023, 31, 101742. [Google Scholar] [CrossRef]
- Raja, A.; Son, N.; Pandey, S.; Kang, M. Fabrication of solar-driven hierarchical ZnIn2S4/rGO/SnS2 heterojunction photocatalyst for hydrogen generation and environmental pollutant elimination. Sep. Purif. Technol. 2022, 293, 121119. [Google Scholar] [CrossRef]
- Ouyang, C.; Quan, X.; Zhang, C.; Pan, Y.; Li, X.; Hong, Z.; Zhi, M. Direct Z-scheme ZnIn2S4@MoO3 heterojunction for efficient photodegradation of tetracycline hydrochloride under visible light irradiation. Chem. Eng. J. 2021, 424, 130510. [Google Scholar] [CrossRef]
- Sun, L.; Xiang, L.; Zhao, X.; Jia, C.-J.; Yang, J.; Jin, Z.; Cheng, X.; Fan, W. Enhanced visible-light photocatalytic activity of BiOI/BiOCl heterojunctions: Key role of crystal facet combination. ACS Catal. 2015, 5, 3540–3551. [Google Scholar] [CrossRef]
- Zhuang, H.; Huang, Y.; Lin, l.; Xu, W.; Liu, X. Fabrication of BiOCl nanosheets with oxygen vacancies via a novel strategy for enhanced photocatalytic degradation of rhodamine B. Mater. Lett. 2022, 324, 132623. [Google Scholar] [CrossRef]
- Su, X.; Yang, J.; Yu, X.; Zhu, Y.; Zhang, Y. In situ grown hierarchical 50%BiOCl/BiOI hollow flowerlike microspheres on reduced graphene oxide nanosheets for enhanced visible-light photocatalytic degradation of rhodamine B. Appl. Surf. Sci. 2018, 433, 502–512. [Google Scholar] [CrossRef]
- Zhu, X.; Wang, Y.; Guo, Y.; Wan, J.; Yan, Y.; Zhou, Y.; Sun, C. Environmental-friendly synthesis of heterojunction photocatalysts g-C3N4/BiPO4 with enhanced photocatalytic performance. Appl. Surf. Sci. 2021, 544, 148872. [Google Scholar] [CrossRef]
- Wang, Y.; Qiang, Z.; Zhu, W.; Yao, W.; Tang, S.; Yang, Z.; Wang, J.; Duan, J.; Ma, C.; Tan, R. BiPO4 Nanorod/Graphene Composite heterojunctions for photocatalytic degradation of tetracycline hydrochloride. ACS Appl. Nano Mater. 2021, 4, 8680–8689. [Google Scholar] [CrossRef]
- Mahendran, N.; Praveen, K. BiPO4/Fe-metal organic framework composite: A promising photocatalyst toward the abatement of tetracycline hydrochloride, Indigo Carmine and reduction of 4-nitrophenol. J. Ind. Eng. Chem. 2021, 100, 220–232. [Google Scholar] [CrossRef]
- Wang, Y.-J.; Zhang, J.-Y.; Hou, S.-S.; Wu, J.-X.; Wang, C.; Li, Y.-M.; Jiang, G.-Y.; Cui, G.-Q. Novel CoAl-LDH Nanosheets/BiPO4 nanorods composites for boosting photocatalytic degradation of phenol. Petrol. Sci. 2022, 19, 3080–3087. [Google Scholar] [CrossRef]
- Yang, W.; Tang, S.; Wei, Z.; Chen, X.; Ma, C.; Duan, J.; Tan, R. Separate-free BiPO4/graphene aerogel with 3D network structure for efficient photocatalytic mineralization by adsorption enrichment and photocatalytic degradation. Chem. Eng. J. 2021, 421, 129720. [Google Scholar] [CrossRef]
- Cai, T.; Zeng, W.; Liu, Y.; Wang, L.; Dong, W.; Chen, H.; Xia, X. A promising inorganic-organic Z-scheme photocatalyst Ag3PO4/PDI supermolecule with enhanced photoactivity and photostability for environmental remediation. Appl. Catal. B Environ. 2020, 263, 118327. [Google Scholar] [CrossRef]
- Ben, H.; Liu, Y.; Liu, X.; Liu, X.; Ling, C.; Liang, C.; Zhang, L. Diffusion-controlled Z-scheme-steered charge separation across PDI/BiOI heterointerface for ultraviolet, visible, and infrared light-driven photocatalysis. Adv. Funct. Mater. 2021, 31, 2102315. [Google Scholar] [CrossRef]
- Yang, J.; Miao, H.; Li, W.; Li, H.; Zhu, Y. Designed synthesis of a p-Ag2S/n-PDI self-assembled supramolecular heterojunction for enhanced full-spectrum photocatalytic activity. J. Mater. Chem. A 2019, 7, 6482–6490. [Google Scholar] [CrossRef]
- Li, B.; Chen, S.; Huang, D.; Cheng, M.; Du, C.; Feng, C.; Yang, Y.; Lei, L.; Chen, Y.; Xue, W.; et al. PDI supermolecule-encapsulated 3D BiVO4 toward unobstructed interfacial charge transfer for enhanced visible-light photocatalytic activity. J. Phys. Chem. C 2021, 125, 18693–18707. [Google Scholar] [CrossRef]
- Zhou, W.; Yang, B.; Liu, G.; Xu, C.; Ji, Q.; Xiang, W.; Sun, D.; Zhong, Q.; He, H.; Yazi, L.; et al. Perylene diimide supermolecule (PDI) as a novel and highly efficient cocatalyst for photocatalytic degradation of tetracycline in water: A case study of PDI decorated graphitic carbon nitride/bismuth tungstate composite. J. Colloid. Interf. Sci. 2022, 615, 849–864. [Google Scholar] [CrossRef]
- Hong, W.; Wang, L.; Liu, K.; Han, X.; Zhou, Y.; Gao, P.; Ding, R.; Liu, E. Asymmetric supercapacitor constructed by self-assembled camellia-like BiOCl and activated carbon microspheres derived from sweet potato starch. J. Alloy. Compd. 2018, 746, 292–300. [Google Scholar] [CrossRef]
- Maisang, W.; Phuruangrat, A.; Randorn, C.; Kungwankunakorn, S.; Thongtem, S.; Wiranwetchayan, O.; Wannapop, S.; Choopun, S.; Kaowphong, S.; Thongtem, T. Enhanced photocatalytic performance of visible-light-driven BiOBr/BiPO4 composites. Mat. Sci. Semicon. Proc. 2018, 75, 319–326. [Google Scholar] [CrossRef]
- Wang, J.; Shi, W.; Liu, D.; Zhang, Z.; Zhu, Y.; Wang, D. Supramolecular organic nanofibers with highly efficient and stable visible light photooxidation performance. Appl. Catal. B Environ. 2017, 202, 289–297. [Google Scholar] [CrossRef]
- Datar, A.; Balakrishnan, K.; Zang, L. One-dimensional self-assembly of a water soluble perylene diimide molecule by pH triggered hydrogelation. Chem. Commun. 2013, 49, 6894–6896. [Google Scholar] [CrossRef] [PubMed]
- Naresh, G.; Malik, J.; Meena, V.; Mandal, T.K. pH-mediated collective and selective solar photocatalysis by a series of layered aurivillius perovskites. ACS Omega 2018, 3, 11104–11116. [Google Scholar] [CrossRef] [PubMed]
- Yin, B.; Fang, Z.; Luo, B.; Zhang, G.; Shi, W. Facile Preparation of Bi24O31Cl10 Nanosheets for Visible-Light-Driven Photocatalytic Degradation of Tetracycline Hydrochloride. Catal. Lett. 2017, 147, 2167–2172. [Google Scholar] [CrossRef]
- Shi, Y.; Yang, Z.; Wang, B.; An, H.; Chen, Z.; Cui, H. Adsorption and photocatalytic degradation of tetracycline hydrochloride using a palygorskite-supported Cu2O–TiO2 composite. Appl. Clay Sci. 2016, 119, 311–320. [Google Scholar] [CrossRef]
- Li, W.; Ding, H.; Ji, H.; Dai, W.; Guo, J.; Du, G. Photocatalytic degradation of tetracycline hydrochloride via a CdS-TiO2 heterostructure composite under visible light irradiation. Nanomaterials 2018, 8, 415. [Google Scholar] [CrossRef]
- Malik, J.; Kumar, S.; Mandal, T.K. Reactive species specific RhB assisted collective photocatalytic degradation of tetracycline antibiotics with triple-layer Aurivillius perovskites. Catal. Sci. Technol. 2022, 12, 6704–6716. [Google Scholar] [CrossRef]
- Liu, X.; Xu, W.; Wang, N.; Lin, L.; Zhuang, H.; Li, Q. An ultra-high performance for reduction of 4-nitroaniline with optimized noble-metal-free Bi4NbO8Cl nanosheets under visible light irradiation. Adv. Powder Technol. 2022, 33, 103601. [Google Scholar] [CrossRef]
- Hu, Y.; Jia, Z.; Lv, R.; Fan, C.; Zhang, H. One-pot electrochemical preparation of BiOCl/BiPO4 double-layer heterojunction film with efficient photocatalytic performance. Mater. Res. Bull. 2017, 94, 222–230. [Google Scholar] [CrossRef]
- Duo, F.; Wang, Y.; Mao, X.; Zhang, X.; Wang, Y.; Fan, C. A BiPO4/BiOCl heterojunction photocatalyst with enhanced electron-hole separation and excellent photocatalytic performance. Appl. Surf. Sci. 2015, 340, 35–42. [Google Scholar] [CrossRef]
- Mei, J.; Tao, Y.; Gao, C.; Zhu, Q.; Zhang, H.; Yu, J.; Fang, Z.; Xu, H.; Wang, Y.; Li, G. Photo-induced dye-sensitized BiPO4/BiOCl system for stably treating persistent organic pollutants. Appl. Catal. B: Environ. 2021, 285, 119841. [Google Scholar] [CrossRef]
- Ji, X.; Liu, X.; Guo, Y.; Zhang, J. Developing visible light responsive Z-scheme BN-PDI photocatalysts with good degradation performance for antibiotics. Chem. Eng. J. 2021, 425, 131260. [Google Scholar] [CrossRef]
- Malik, J.; Kumar, S.; Srivastava, P.; Bag, M.; Mandal, T.K. Cation disorder and octahedral distortion control of internal electric field, band bending and carrier lifetime in Aurivillius perovskite solid solutions for enhanced photocatalytic activity. Mater. Adv. 2021, 2, 4832–4842. [Google Scholar] [CrossRef]







| Catalyst | Pollutant | Pollutant Concentration | Amount of Catalyst | Photocatalytic Efficiency | References |
|---|---|---|---|---|---|
| Bi5CaTi4FeO18 | MB | 1 × 10−5 M, 100 mL | 100 mg | ∼55%, 180 min | [33] |
| Bi24O31Cl10 | TCH | 10 mg L−1, 100 mL | 100 mg | 80.1%, 150 min | [34] |
| Cu2O–TiO2 | TCH | 30 mg L−1, 50 mL | 50 mg | 81.4%, 240 min | [35] |
| CdS-TiO2 | TCH | 50 mg L−1, 50 mL | 50 mg | 87%, 480 min | [36] |
| Bi2.5Sr1.5Nb2Ti0.5Cr0.5O12 | TC | 6.66 mg L−1, 50 mL | 100 mg | 89%, 300 min | [37] |
| PDI(5%)/BiOCl-BiPO4 | RhB | 10 mg L−1, 50 mL | 25 mg | 98%, 150 min | Present work |
| PDI(5%)/BiOCl-BiPO4 | TCH | 10 mg L−1, 50 mL | 25 mg | 81%, 150 min | Present work |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhuang, H.; Wang, F.; Shi, K.; Yang, K. Designed Synthesis of PDI/BiOCl-BiPO4 Composited Material for Boosted Photocatalytic Contaminant Degradation. Catalysts 2023, 13, 688. https://doi.org/10.3390/catal13040688
Zhuang H, Wang F, Shi K, Yang K. Designed Synthesis of PDI/BiOCl-BiPO4 Composited Material for Boosted Photocatalytic Contaminant Degradation. Catalysts. 2023; 13(4):688. https://doi.org/10.3390/catal13040688
Chicago/Turabian StyleZhuang, Huaqiang, Fulin Wang, Kaiyang Shi, and Kai Yang. 2023. "Designed Synthesis of PDI/BiOCl-BiPO4 Composited Material for Boosted Photocatalytic Contaminant Degradation" Catalysts 13, no. 4: 688. https://doi.org/10.3390/catal13040688
APA StyleZhuang, H., Wang, F., Shi, K., & Yang, K. (2023). Designed Synthesis of PDI/BiOCl-BiPO4 Composited Material for Boosted Photocatalytic Contaminant Degradation. Catalysts, 13(4), 688. https://doi.org/10.3390/catal13040688






