Abstract
The high viscosity of heavy oil is the main challenge hindering its production. Catalytic thermolysis can be an effective solution for the upgrading of heavy oil in reservoir conditions that leads to the viscosity reduction of native oil and increases the yield of light fractions. In this study, the thermolysis of heavy oil produced from Ashalchinskoye field was carried out in the presence of FeP and Al(H2PO4) nanocatalysts at a temperature of 250 °C in N2 gas environment. It was shown that Al(H2PO4)3 and FeP catalysts at a concentration of 0.5% significantly promoted the efficiency of the heavy oil thermolysis and are key controlling factors contributing to the acceleration of chemical reactions. The Al(H2PO4)3 + NiCO3 nanoparticles were active in accelerating the main chemical reactions during upgrading of heavy oil: desulfurization, removal of the side alkyl chains from polyaromatic hydrocarbons, the isomerization of the molecular chain, hydrogenation and ring opening, which led to the viscosity reduction in heavy oil by 42%wt. Moreover, the selectivity of the Al(H2PO4)3 + NiCO3 catalyst relative to the light distillates increased up to 33.56%wt., which is more than two times in contrast to the light distillates of initial crude oil. The content of resins and asphaltenes in the presence of the given catalytic complex was reduced from 34.4%wt. to 14.7%wt. However, FeP + NiCO3 nanoparticles contributed to the stabilization of gasoline fractions obtained after upgraded oil distillation. Based on the results, it is possible to conclude that the thermolysis of heavy oil in the presence of FeP and Al(H2PO4)3 is a promising method for upgrading heavy oil and reducing its viscosity.
1. Introduction
With the ever-increasing demand for energy and declining traditional oil reserves, the petroleum industry is constantly looking for innovative approaches to optimize hydrocarbon production. High-quality oil resources are in critically short supply, so there is a clear trend that oil will be heavier than ever. Despite the importance and large reserves of heavy oil, the development, extraction and refining of heavy hydrocarbons are challenging due to their high density, viscosity and metal content; significant content of resins and asphaltenes; and, hence, heteroatom-containing compounds, low hydrogen-to-carbon (H/C) ratio and low yield of light fractions [1]. Asphaltenes are capable of clogging and plugging up wells, pipelines, downstream equipment and permeable reservoir rocks, as well as influencing the stability of oil−water emulsions and the wettability of rock surfaces, which creates problems in terms of reservoir engineering and field development. Moreover, the higher concentration of metals and heteroelements in heavy crude oil significantly impacts the technological procedures of their processing due to coke formation and catalyst poisoning. All these problems are caused by the chemical and physical nature of asphaltenes that occur in oil-dispersed systems in the form of supramolecular structures representing associated molecule formation with sizes that vary according to the oil composition and external factors such as temperature and pressure. At a high degree of association, asphaltenes coagulate and precipitate. At a low degree of association, they are present, as a rule, in a suspended state. Asphaltenes can also be stable without sedimentation in high-viscosity bitumens at exclusively high concentrations. In any case, their behavior is ambiguous and unpredictable and defines the main oil-dispersed system’s properties, such as density, viscosity and stability in the processes of oil production, transportation and processing. Therefore, it is important to have information about the changes in the phase-dispersed structure of asphaltenes and their properties in various technological processes. Upgrading heavy oil and bitumen, at least to a degree close to that of light oil, is cost-effective. The downstream upgrading of heavy crude oil or oil residues requires a combination of different processes, such as delayed coking, hydroconversion, catalytic cracking, visbreaking, deasphalting, etc., to obtain a synthetic oil suitable for further processing in traditional refineries. This is a cost-effective approach that requires huge investments. In this regard, upstream heavy oil upgrading, which can be carried out in natural reactor reservoir formations, is attractive in terms of efficiency. The in-place upgrading of heavy oil can significantly improve the physical and chemical properties of heavy oil, which leads to the enhancement of heavy oil production, transportation and processing [2]. The complex compositions, as well as the internal interactions in heavy crude oil, are cognized as the domestic origins of the high viscosity of heavy crude oil [3,4]. Reducing its high viscosity is consequently the most demanded route for the exploitation and utilization of heavy crude oil resources. To date, many viscosity-upgrading technologies have been developed [5]. Thermal-enhanced oil recovery methods are considered one of the successful industrial-scale approaches for unlocking heavy hydrocarbon resources and are mainly directed toward reducing the viscosity of heavy oil under the impact of temperature. However, one of the alternative methods for modifying steam-based heavy oil recovery methods was proposed by petrochemicals—catalytic aquathermolysis [6]. The method is carried out by co-injecting suitable catalysts and hydrogen donors within the steam into heavy oil reservoir formations. The application of the catalyst’s nanoparticles during the aquathermolysis of heavy oil is reasonable as catalysts promote destructive hydrogenation reactions such as the cracking of alkyl chains from asphaltene fragments, the isomerization of the molecular chains, hydrogenation and ring opening. The hydrogen-donating capacity of hydrogen donors and steam is intensified in the presence of the appropriate catalysts; thus, catalysts contribute to the polymerization of formed radicals. Moreover, the catalyst’s nanoparticles promote the pyrolysis of heavy oil to break down large heavy oil molecules, such as resins and asphaltenes, into smaller ones. These catalytic nanoparticles can shift the cracking temperature to lower values by accelerating various bond cleavage in resins and asphaltenes molecules with an increase in the content of lighter substances [7]. The cracking of bonds usually occurs via the radical varieties of heteroatoms (S, N and O). These compounds can trigger polymerization reactions in the absence of enough free hydrogen molecules and highly selective catalysts and reproduce giant molecules that make the viscosity of heavy oil high. In addition, a suitable catalyst can provide the decay and removal of O, S and N derivatives from the oil [8]. Even a small breakdown of resins and asphaltenes can significantly reduce the viscosity and increase the mobility of crude oil, and as a result, it is easier to produce them. One of the advantages of nanosized catalysts is their relatively large surface area, which makes them ideal for catalytic aquathermolysis in order to increase the light fraction of crude oil and therefore improve the overall quality of heavy oil [9]. Yang et al. [10] developed a new catalyst based on the Fe nanoparticles of different sizes supported at 10 nm SiO2 as a result of the interaction of iron chloride precursors with silica using the traditional NaBH4 reduction in the liquid phase. The catalyst was very active, with only 1.0 wt% reducing the viscosity of Shengli heavy oil from 184 to less than 45 Pa s. Its reaction mechanism involves the inhibition of oxidation and cleavage of CAS bonds in resins and asphaltenes. Wu et al. [11] synthesized a nickel chelate (i.e., aromatic sulfonyl) with amphiphilic properties. The catalyst was very active at 200 °C because it provided a reduction in viscosity by more than 96%. The results obtained by Aliev et al. also support the efficiency of amphiphilic nickel-based catalysts on the viscosity reduction in heavy oil [12]. The results of mechanistic studies show that the combined hydrophobic–hydrophilic characteristics of the catalyst enhanced reactions involving pyrolysis and heteroatom removal. Similarly, ring opening and closing reactions were observed. Wen et al. used molybdenum oleate as a catalyst in the aquathermolysis of heavy oil from the Liaohe field, and viscosity was reduced by more than 90% [13]. The research has proved the capability of steam at temperature ranges of 240–300 °C in breaking down large asphaltene and resin molecules into small ones [14,15]. Li et al. prepared Fe (III) oleate and applied it in the low-temperature aquathermolysis of Shengli crude oil. The authors imply the activity of Fe metal than Ni and Co, and the viscosity reduction degree of heavy oil is up to 86.1% [16]. However, a study by Wu et al. [17] showed that a 96% viscosity reduction can be achieved with sulfonic acid Ni at a lower temperature of 200 °C even without the addition of a hydrogen donor. Hollow FeP nanoparticles densely encapsulated in carbon nanosheet frameworks (donated as hollow FeP/C nanosheets), in situ converted from Fe-glycolate precursor nanosheets via carbonization and subsequent phosphorization, are designed and synthesized as an advanced electrocatalyst for the hydrogen evolution reaction [18]. Phosphides can exhibit acidic character, and hydrogen ions can form on the acidic centers, which is important for the protonation of cracking products such as free radicals. Phosphides participate in the carbonic-ion reaction mechanism, which results in an increase in the content of isomers in the benzene products of thermolysis.
The determination of an effective water-soluble catalyst and reaction conditions that promote the breakdown of large hydrocarbon molecules to reduce the viscosity of oil is the motivation for this study. The purpose of this work is to study the catalytic performance of FeP and Al(H2PO4)3 nanoparticles in a nitrogen medium on the upgrading of heavy oil by promoting thermolytic reactions. In addition, the impact of Ni(CO3) catalytic additives on hydrogenation reactions was evaluated.
2. Results and Discussions
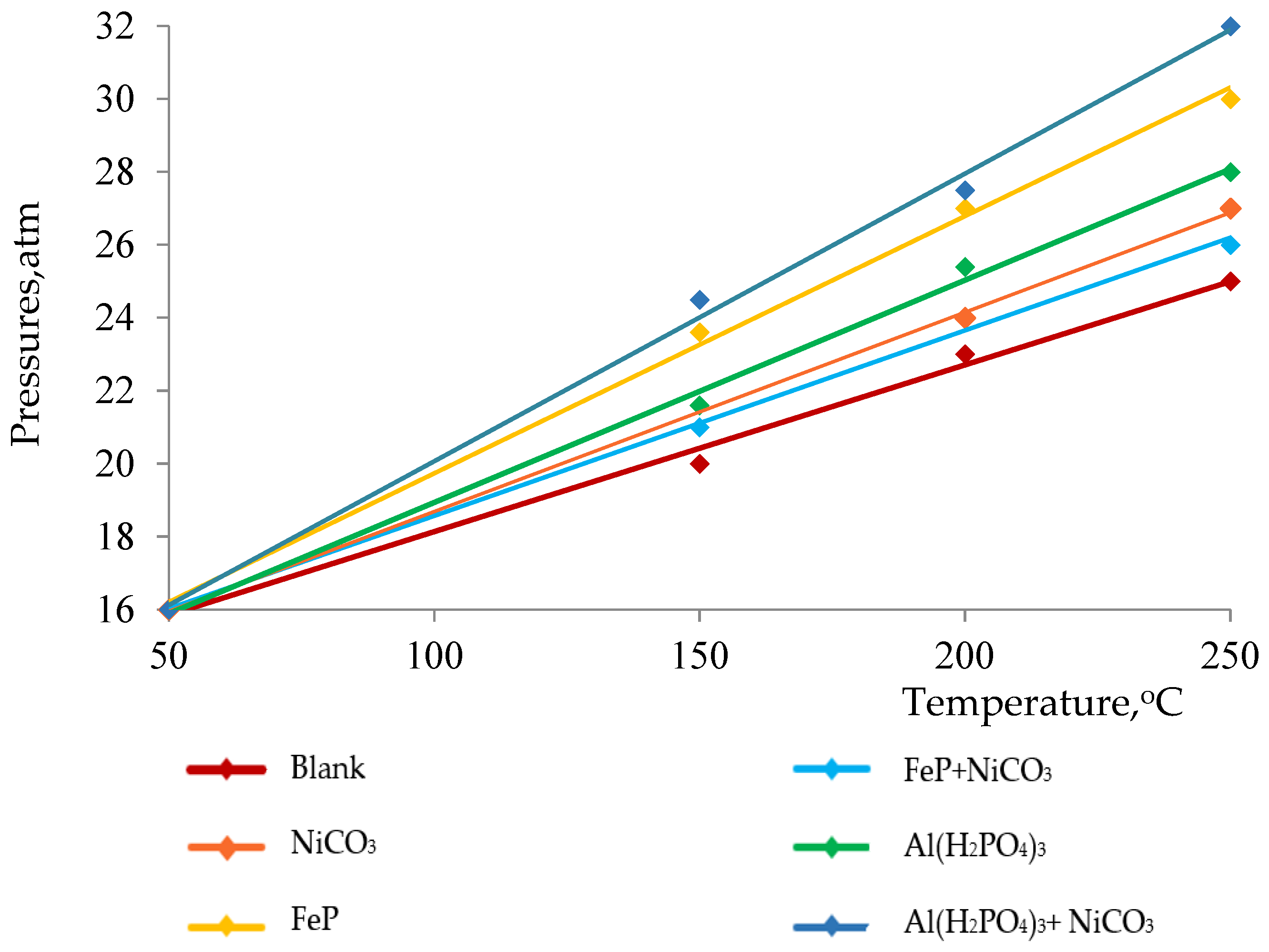
The thermolysis of heavy oil is a process by which heavy components are cracked into lighter ones because of heat energy. Moreover, the thermolysis of heavy oil yields large amounts of non-condensable and hydrocarbon gasses such as hydrogen, CO2, methane, ethane, etc. The number of evolved gasses determines the depth of the thermolytic process, which can be tracked by the increase in pressure in the reaction medium. On the contrary, it is common to observe the presence of high-molecular-weight compounds in thermolysis products, which form via radical chain mechanisms. In the absence of proper catalysts, this mechanism assists the polycondensation of the cracked fragments of hydrocarbons. In its turn, polymerization leads to the formation of resins, asphaltenes, carbenes and carboids. In Figure 1, we demonstrated the influence of various catalysts on the depth of cracking by observing pressure changes during the thermolysis of heavy oil at 250 °C. The results show that all nanoparticles have almost directly proportional influence on the pressure change, while Al(H2PO4)3 + NiCO3 significantly increases the pressure in the reactor and, consequently, the cracking depth. This is certainly reflected in the effective reduction in the viscosity of heavy crude oil (Figure 2).
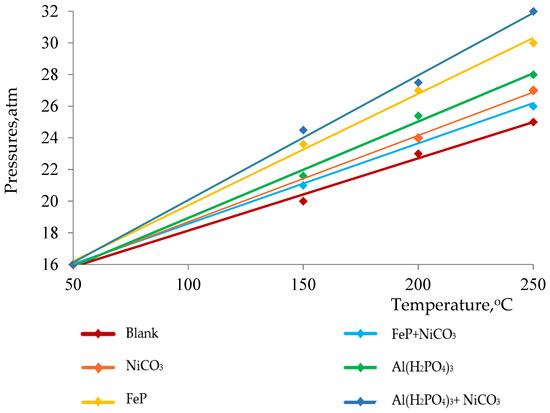
Figure 1.
Pressure changes during thermolysis at 250 °C.
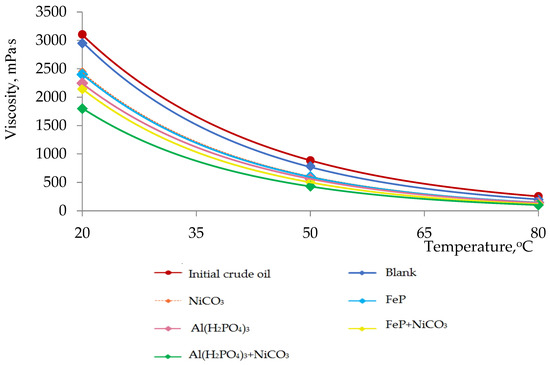
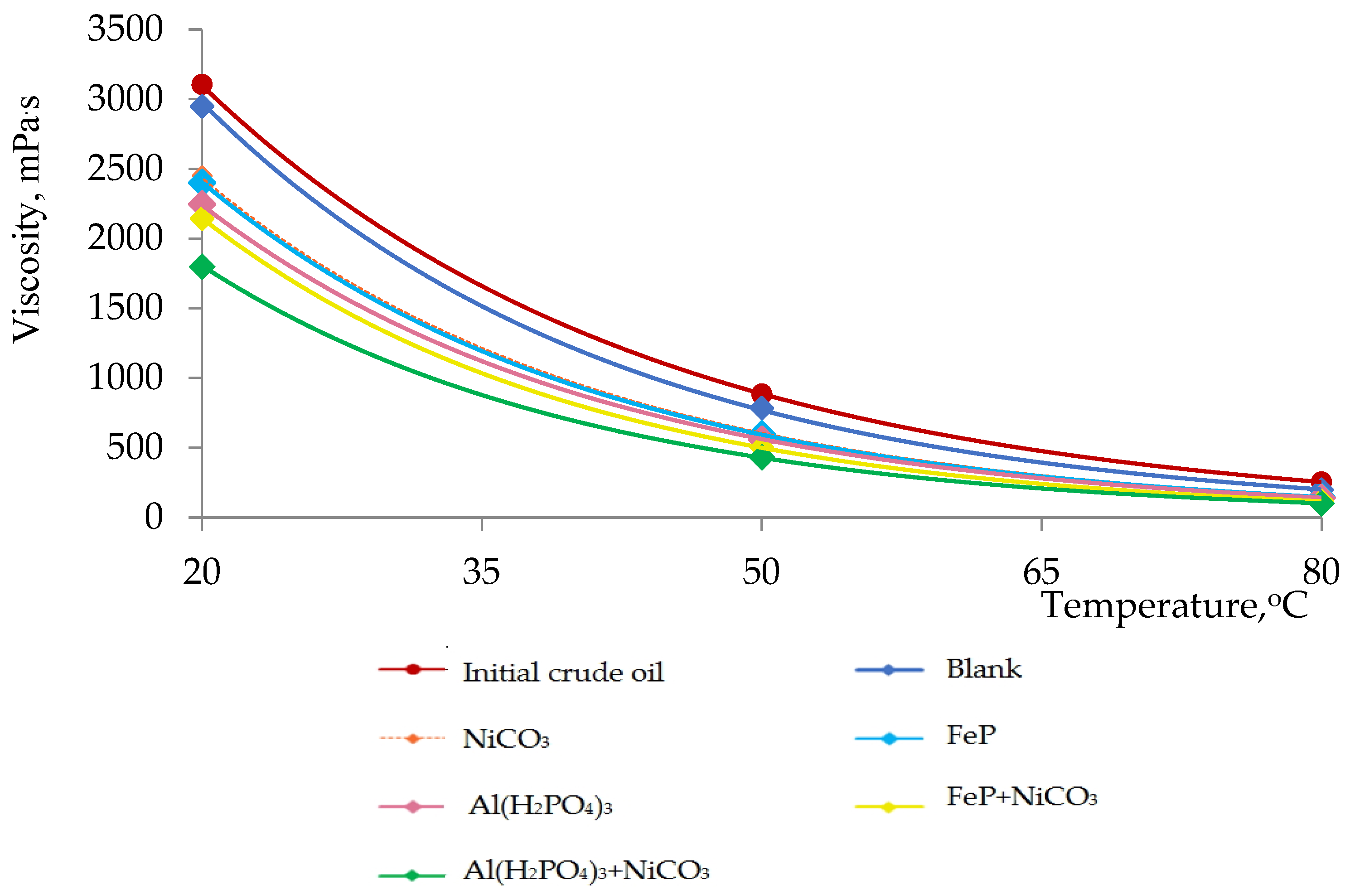
Figure 2.
Dependence of viscosity on temperature in the process of thermolysis.
The irreversible viscosity reduction in heavy oil is the main goal of both in situ and ex situ upgrading techniques. The mobility of heavy oil strongly depends on the viscosity values. In its turn, mobility determines incremental oil recovery. The viscosity of heavy oil is crucial even after the production step, because one of the major challenges in the pipeline transportation of heavy oil is also its high viscosity, which requires high energy to pump it. Many attempts have been carried out to reduce the viscosity of heavy crudes, including heating, diluting and forming oil-in-water emulsions. The viscosity of heavy oil is very sensitive to heat, but the effect is temporary and it fails irreversibly upon temperature decrease. The application of various diluents such as chemicals, additives, diesel and kerosene distillates are cost-effective. The blending of light crude oils with heavy ones is not practical and sustainable. Therefore, the industry needs more feasible and practical solutions to decrease heavy oil’s viscosity irreversibly.
In Figure 2, the viscosity–temperature characteristics of the initial crude oil sample and thermolysis products in the presence of different catalytic systems are presented. The maximum viscosity reduction degree was observed in the case of Al(H2PO4)3 + NiCO3, the viscosity of which is lower than the initial crude oil sample by 42%. The enhancement in the flow behavior of heavy crude oil is explained by destructive processes mainly in resins and asphaltenes. It is well-known that the content of high-molecular-weight molecules determines the viscosity of heavy oil [19,20]. The results of viscosity measurements are in accordance with the results obtained in Figure 1.
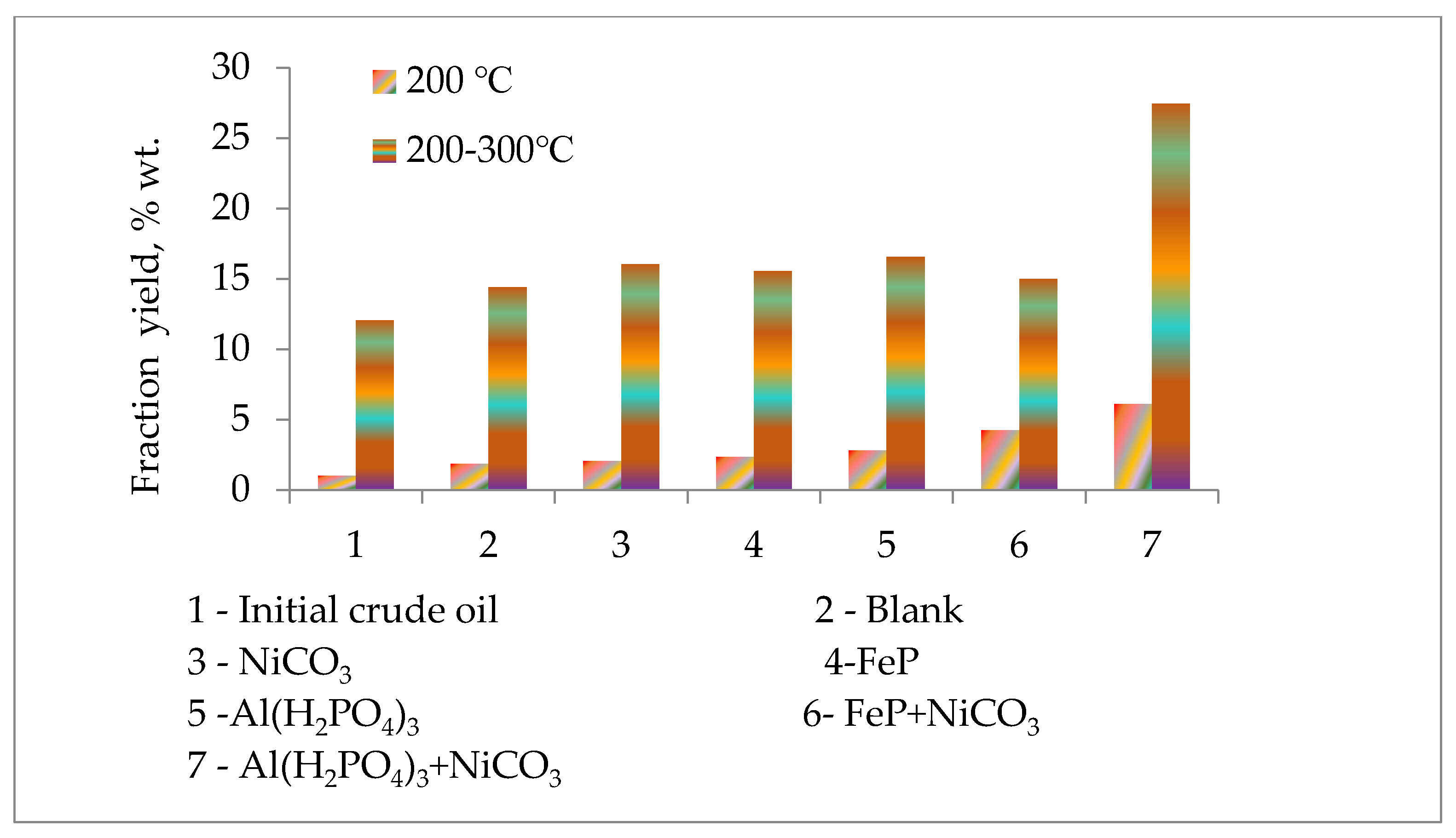
The atmospheric distillation of initial crude oil and conversion products after thermolysis was carried out to evaluate the catalytic upgrading performance of Al(H2PO4)3 and FeP nanoparticles. The yield of low boiling fractions was roughly divided into the following: Fractions obtained below 200 °C refer to gasoline, and fractions obtained at 200–300 °C refer to diesel. The yields of these fractions depending on the used catalyst are presented in Figure 3. The yield of the benzene fraction increases in the presence of FeP and Al(H2PO4)3 catalysts. The former catalyst leads to an increase in the yield of benzene fractions more than two times in contrast to the initial crude oil sample and more than 25% in contrast to the blank sample (thermolysis of crude oil sample without catalyst) under the same conditions. In its turn, Al(H2PO4)3 nanoparticles increased the yield of fraction with a boiling temperature below 200 °C almost three times compared to the initial crude oil sample and more than 50% compared to the blank sample. The increase in the content of low boiling fractions in the presence of catalysts shows the intensification of the thermal cracking of heavy fragments with a boiling point above 300 °C and their transformation into light value-added products. The Al(H2PO4)3 nanoparticles with the NiCO3 additive demonstrated the maximum yield of the benzene fraction, which is six times higher than the benzene fraction of the initial crude oil sample, three times more than the blank sample and more than two times the crude oil sample with Al(H2PO4)3 nanoparticles. The fraction with the boiling point range of 200–300 °C in the case of Al(H2PO4)3 + NiCO3 particles was enhanced by almost two times compared to the blank sample.
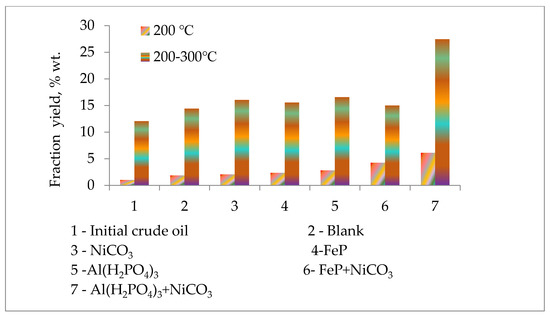
Figure 3.
Light distillate yields before and after thermolysis.
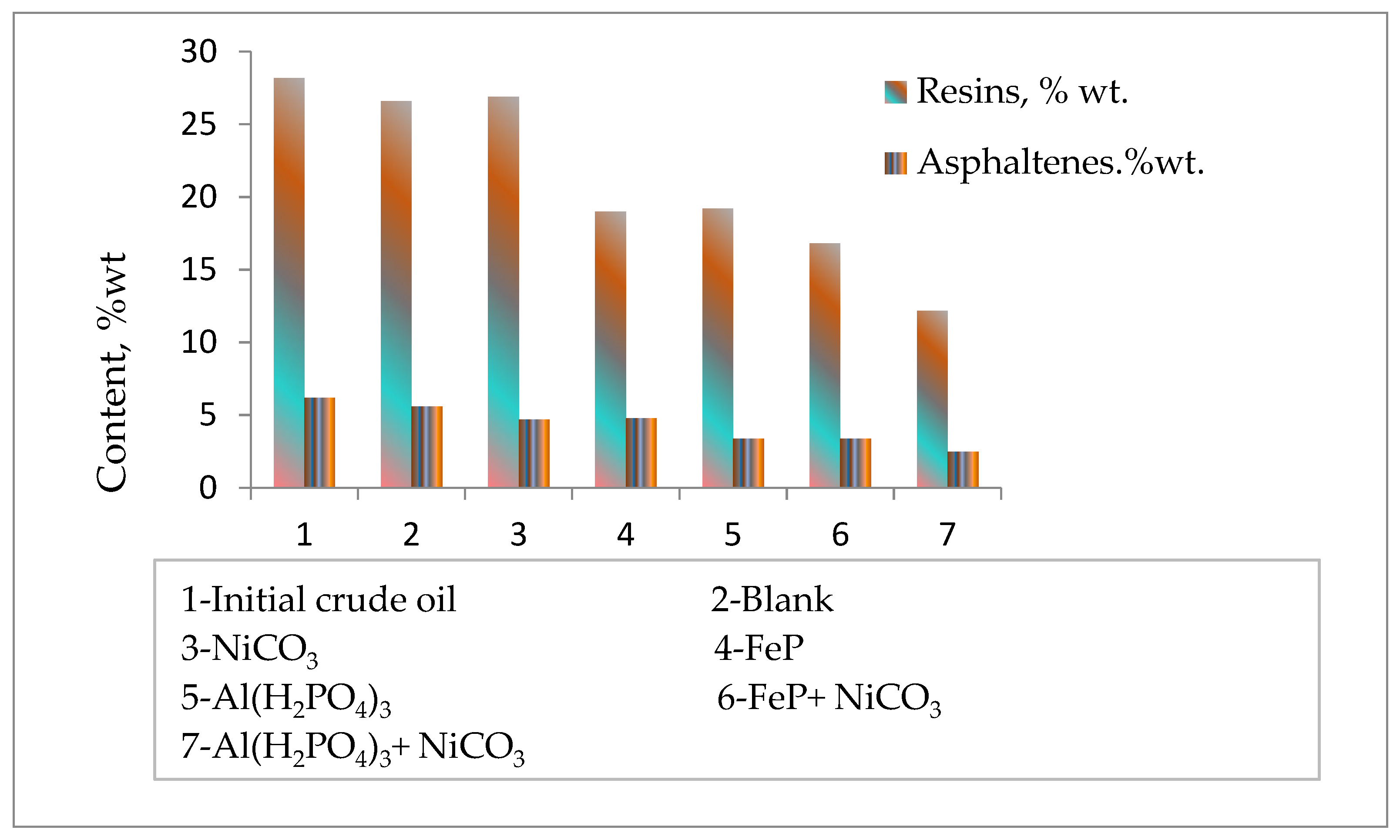
The group composition analysis of initial heavy crude oil and after thermolytic reactions in the absence and presence of nanoparticles revealed changes in the content of the high-molecular-weight components of crude oil resins and asphaltenes (Figure 4). The content of resins and asphaltenes reduces a little after the thermolysis of heavy oil. However, the addition of catalytic complexes to the reaction medium significantly reduces the content of resins and asphaltenes by promoting destructive hydrogenation processes. Here, Al(H2PO4)3 + NiCO3 also demonstrates the most effective catalytic performance by reducing the content of resins and asphaltenes by more than two times in contrast to the blank sample. The obtained experimental results on the group composition of thermolysis products in the absence and presence of catalytic complexes are in agreement with the results of light distillate formations.
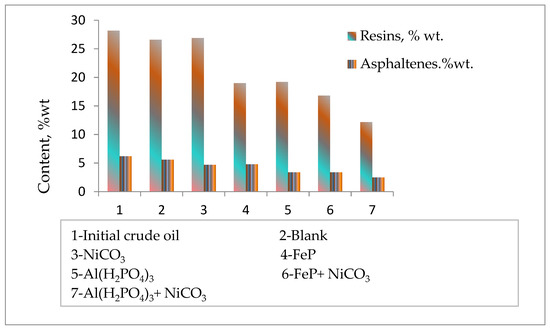
Figure 4.
The content of resins and asphaltenes before and after thermolysis.
The upgrading efficiency of heavy oil can be followed by the oil’s boiling point as well (Table 1). The presence of both Al(H2PO4)3 + NiCO3 and FeP + NiCO3 catalytic complexes reduced the crude oil boiling point from 170 °C to 75 °C due to the formation of light distillates, which compose of normal alkanes, acyclic isoprenoids and cyclic alkanes. Moreover, the data provided in Table 1 show the reduction in the total content of resins and asphaltenes by 2.5 times from 34%wt. to 14.7%wt. At the same time, the total content of sulfur decreased from 4.72%wt. to 4.09%wt., which is explained by the thermal cracking of the least stable sulfur compounds, such as sulfides, into gaseous compounds in the form of hydrogen sulfide and mercaptans.

Table 1.
Composition and properties of heavy oil before and after thermolysis.
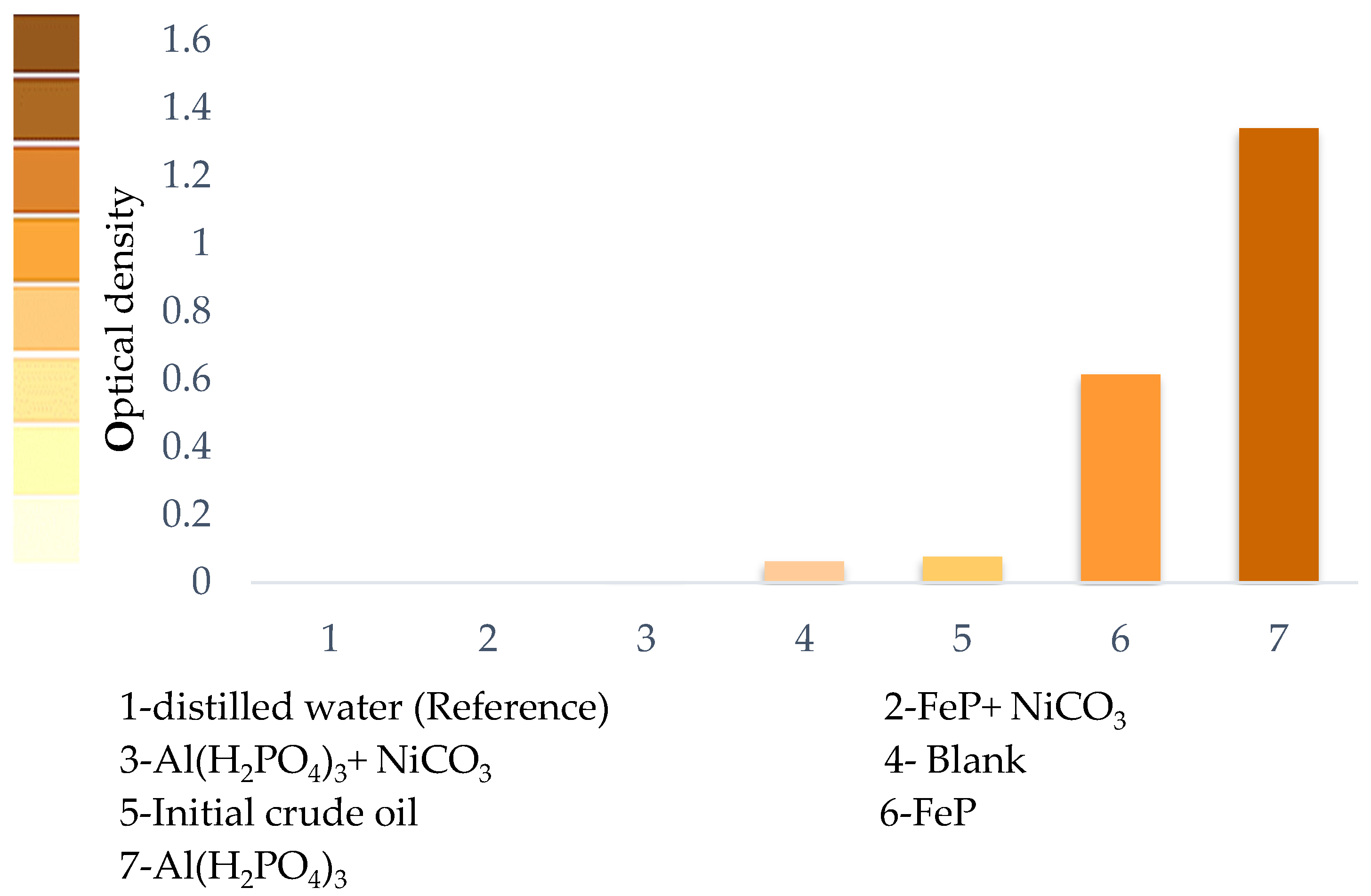
One of the important exploitation parameters of light fractions obtained after the thermocatalytic upgrading of heavy oil is their stability. The photocolorimetric analysis method (Figure 5) has been employed to study the stability of gasoline fractions of heavy oil after thermolytic and thermocatalytic upgrading processes. The results show that some gasoline fractions are not stable during short storage times due to polymerization reactions. Distillation water was used as a reference with zero optical density. The highest stability with the minimum optical density was reached for the thermolysis products of heavy oil in the presence of FeP + NiCO3 and Al(H2PO4)3 + NiCO3 nanoparticles. The optical density of the initial crude oil sample was equal to 0.08. The optical density of gasoline fraction of non-catalytic thermolysis products is almost unchanged—0.06. However, the samples in the presence of FeP (sample #6) and Al(H2PO4)3(sample #7) show the highest values of optical density at −0.61 and 1.34, respectively, which indicates the significant content of unsaturated hydrocarbons in the composition of gasoline fractions. This, in turn, determines the instability of such products.
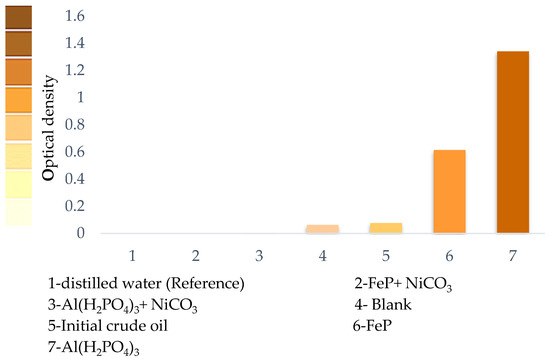
Figure 5.
The photo colorimetric analysis results of gasoline fractions before and after the thermolysis of heavy oil samples.
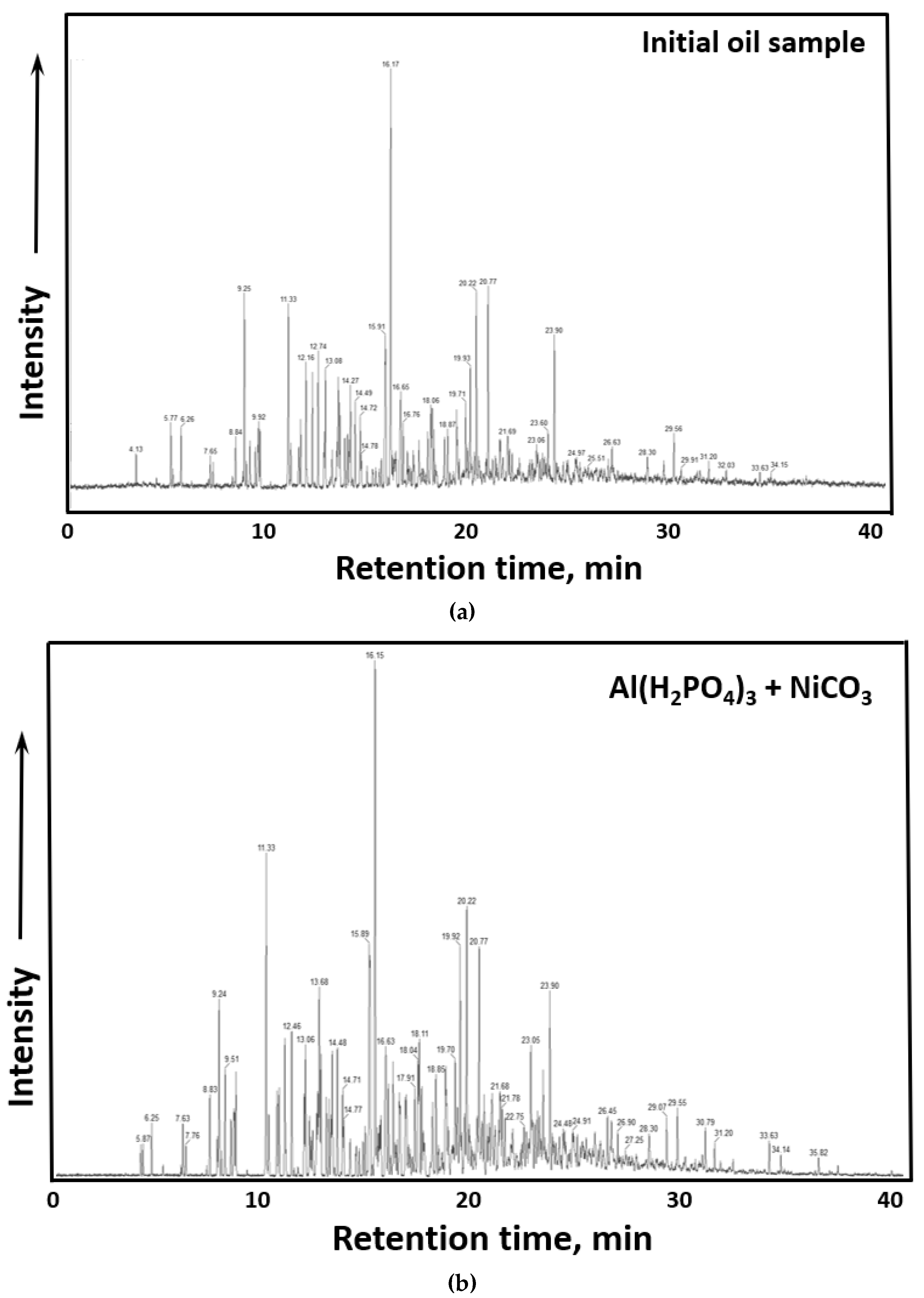
The spectra of GC-MS analysis of gasoline fractions of initial crude oil and after thermolytic upgrading in the presence of Al(H2PO4)3 + NiCO3 are illustrated in Figure 6.
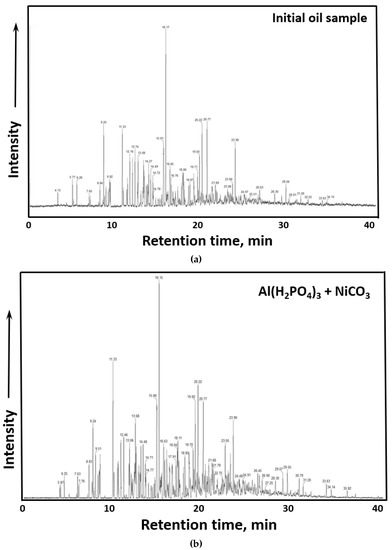
Figure 6.
GC-MS results. (a) Spectra of benzene fraction of initial crude oil before thermolysis, (b) Spectra of benzene fraction of crude oil after thermolysis in the presence of Al(H2PO4)3 + NiCO3.
The distribution of fractional composition is inevitably accompanied by changes in the group composition of gasoline fractions. It was found (Table 2) that in the presence of Al(H2PO4)3 + NiCO3 nanoparticles, the share of unsaturated hydrocarbons has been significantly reduced (more than six times), while the content of isoalkanes increased significantly (by 63%wt.) compared to the initial crude oil.

Table 2.
Group composition of gasoline fractions from oil after thermolysis.
3. Experimental Section
3.1. Experiments in Autoclave
The object of this study is a heavy oil sample produced from the Ashalchinskoye field (Republic of Tatarstan, Russia). The Republic of Tatarstan is one of the leaders of heavy oil-producing petroleum provinces in the Russia Federation. The estimated unconventional hydrocarbon resources exceed conventional petroleum stocks. However, processing heavy crude oil in this province is a big challenge mainly connected with the high content of high-molecular-weight resins and asphaltenes in them; the major heteroatom compounds are concentrated in high-molecular-weight compounds. The operating companies are looking for emerging technologies to process the hydrocarbon in a feasible way.
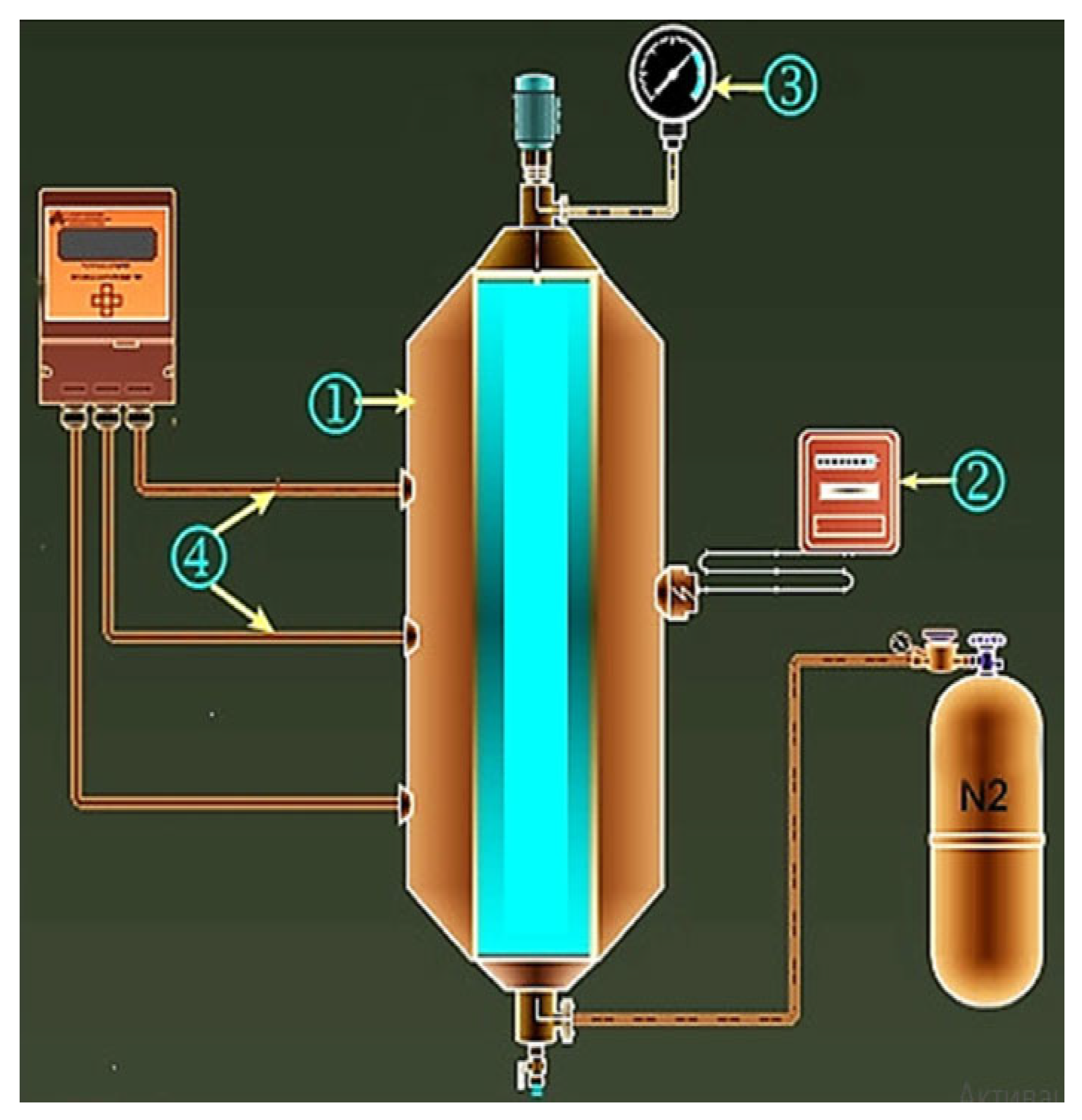
The catalysts are industrially used and were purchased from LLC ChemTec. The particle size distribution of FeP and Al(H2PO4)3 catalysts based on the passport of the reactants ranges between 7–15 nm and 60–80 nm, correspondingly. The effect of the given nanocatalysts with 0.5%wt. concentration on Ashalchinskoye oil during catalytic thermolysis in N2 inert gas medium was evaluated. Moreover, NiCO3 was added as a catalytic additive to the above-mentioned nanoparticles in order to increase the rate of hydrogenation reactions. Experiments were carried out in a laboratory high-pressure reactor made of stainless steel, heating and stirring devices, as well as the means for controlling the course of the process (Figure 7). N2 was injected into the autoclave and pressure was maintained at 1.6 MPa until the reactor temperature was raised to 250 °C and maintained at this temperature for 2 h. At the end of the reaction, the autoclave reactor was cooled to room temperature, and the oil sample was collected for further analysis.
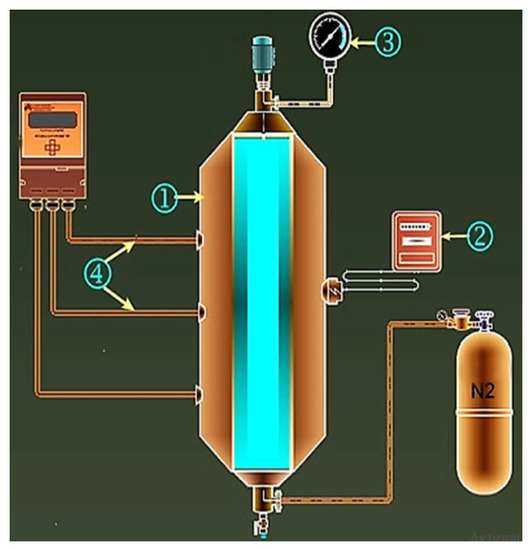
Figure 7.
Schematic illustration of laboratory high-pressure reactor: 1—autoclave; 2—temperature-controlling unit; 3—manometer; 4—thermocouples.
The evaluation of the catalytic performance of the given nanoparticles requires information on the group’s composition, elemental analysis, viscosity values, the yield of light distillates and the composition of the gasoline fraction of heavy oil.
3.2. SARA-Analysis
The composition of oil disperse systems is a universal characteristic that is necessary for the estimation of the stability of a system and the quality of the hydrocarbon feedstocks used in processes of oil refining, petrochemistry and enhanced oil recovery methods. The most practiced method to characterize the group composition of oil-dispersed systems in the world is SARA-analysis. The application of the SARA-analysis method for the characterization of oil disperse systems allows describing the stability of heavy oil and its components, as well as revealing the asphaltene content that determines the viscosity of heavy oil. This method is based on separating crude oil samples into saturated hydrocarbons, aromatic compounds, resins and asphaltenes according to their solubility and polarity. Saturates (aliphatic hydrocarbons) are not polar hydrocarbons and do not contain double bonds. Saturated hydrocarbons are composed of alkanes with a straight chain and branched alkanes, as well as cycloalkanes (naphthenes). Cycloalkanes contain one or more rings, which may have several alkyl side chains. The saturation of oil typically decreases with the increasing molecular weight of fractions. Thus, saturated hydrocarbons tend to predominate in conventional light crude oils. The "wax" fraction refers to a subclass of saturated hydrocarbons consisting mainly of alkanes with straight-chain alkanes from C20 to C30.
Aromatics comprise a group of aromatic hydrocarbons that includes benzene and its derivative structures. Aromatic compounds are an integral part of all oil-dispersed systems, and today most petroleum aromatic compounds can contain alkyl chains and cycloalkane rings today, along with additional aromatic rings. Aromatic hydrocarbons are often classified as mono-, di- and tri-aromatic substances depending on the number aromatic rings in the molecule.
Resins comprise polar molecules containing heteroatoms, such as nitrogen, oxygen or sulfur. In spite of the fact that the resins play a major role in the formation of dispersed oil systems, in contrast to the asphaltenes, these components are less studied. Nevertheless, some of their characteristics are known and defined. Resins have a higher H/C ratio (1.2–1.7) compared to asphaltenes (0.9–1.2). Resins are structurally similar to asphaltenes but have a lower molecular weight (1000 g/mol). Naphthenes acids are generally considered to be a part of resin fractions.
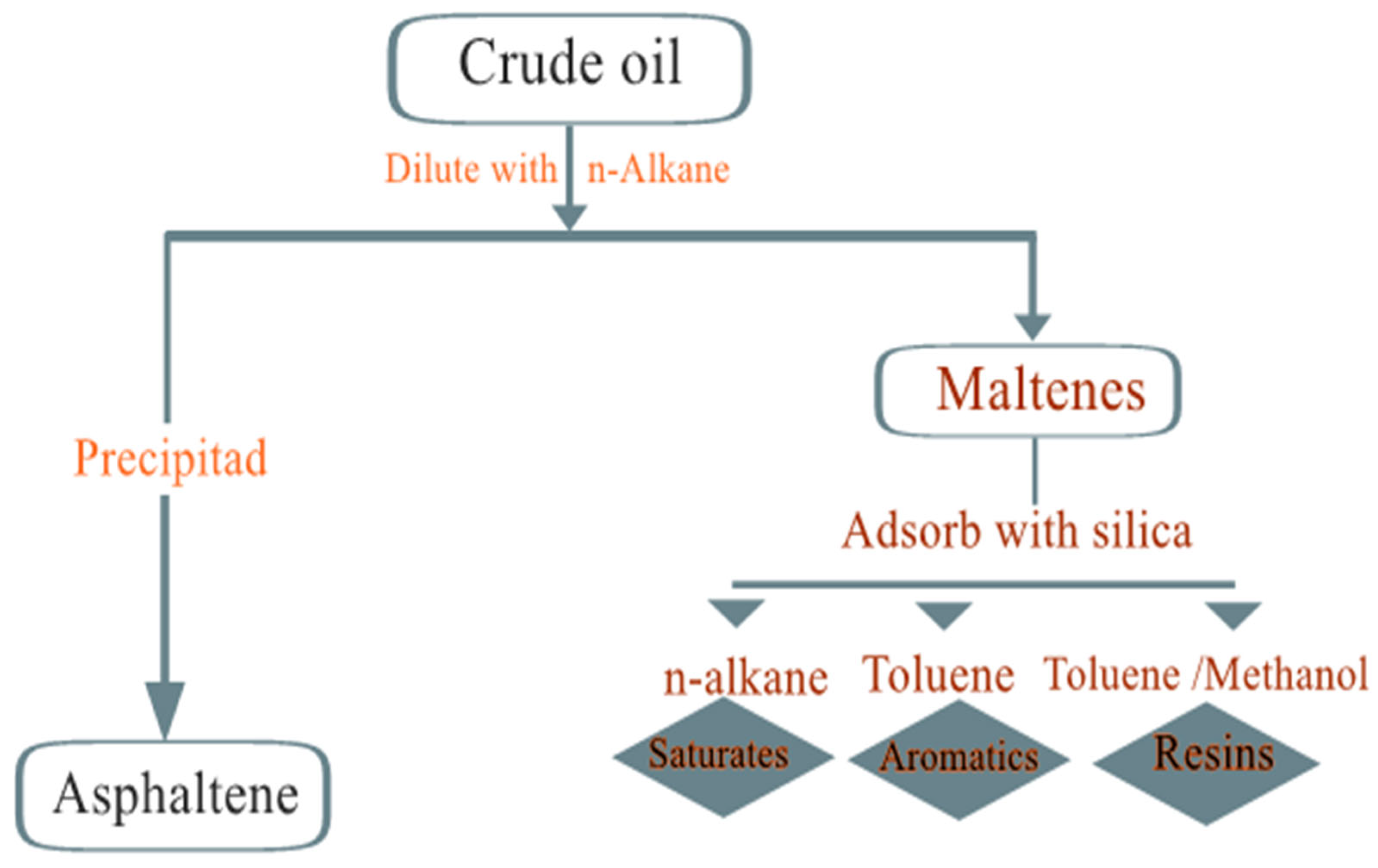
Asphaltenes (asphaltenes): The content of asphaltenes is determined by their precipitation from petroleum by the addition of light alkanes such as pentane, hexane or heptane. This precipitate is soluble in aromatic solvents such as toluene and benzene. The asphaltene fraction of crude oil contains a high percentage of heteroatoms (O, S and N) and organometallic components (Ni, V and Fe). The structures of asphaltenes have been the subject of many studies, but at present, one theory proposes that they are made up of polycyclic aromatic clusters. The molecular weight of asphaltene molecules is difficult to estimate because of their self-association, and their molecular weight ranges from 500 to 2000 g/mol. The SARA analysis (Figure 8) method can be described as follows: Asphaltenes are separated from other hydrocarbon components by adding n-alkanes, such as n-hexane, n-heptane or n-pentane. The solvent-to-solvent ratio should be 1:40 by weight. The nature of the precipitant solvent determines the nature of the asphaltenes isolated from oil asphaltenes. Then, the remaining components, called maltenes, are then separated by passing their mixture through a chromatographic column filled with an adsorbent (aluminum oxides and silica gels are used as active adsorbers). As an adsorbent, the ASK silica gel of fraction 0.25-0.35 was preliminary calcined at a temperature of 1500 °C for 3–4 h.
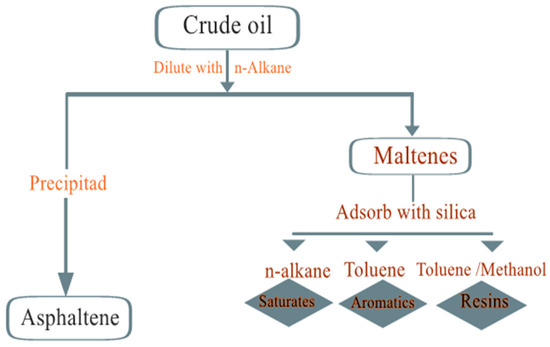
Figure 8.
Schematics of the SARA fractionation.
3.3. Total Sulfur Analysis
The total sulfur content in the oil bulk was determined using an energy-dispersive X-ray fluorescence sulfur analyzer: SPECTRO-SCAN SL (SPECTRON Ltd., St. Petersburg, Russia).
3.4. Viscosity Measurements
The viscosity of the oil samples before and after the reaction was measured using the BROOKFIELD TC500 programmable viscometer at 20, 50 and 80 °C. The constant temperature during the measurements was provided by “Huber” circulation thermostat, and the measurements were carried out after one day of dewatering (centrifuging) in order to properly compare the viscosity reduction degree of upgraded oil samples. It is well-known that at lower temperatures, the influence of supramolecular asphaltene structures on viscosity can be complicated [21]. The volume of crude oil samples was 6.7 mL, and the spindle used was TL5. For the viscosity comparison study, the shear rate (s−1), which was calculated by multiplying 1.32 by the RPM, was the same for all viscosity values of crude oil samples. The RPM, in turn, was read when the spring torque was within 50% and 90%. In such conditions, manufacturers imply that the “Relative Error” and “Repeatability” of the viscosity values should not exceed ±1.0% and 0.2%, respectively.
3.5. Atmospheric Distillation of Crude Oil
The atmospheric distillation of crude oil and conversion products after the autoclave was carried out using the LOIP LP-086 (LOIP Ltd., St. Petersburg, Russia), as per ASTM D86. The distillation unit comprises a flask support mechanism, heater elements and the heater controller. The flask is supported by a Ceran ceramic-glass support board installed on a height-adjustable platform. The low-voltage heating element is built into the insulation of the platform and is powered by a stepless regulator with a calibrated control knob. A glass observation window is fitted in the front of the distillation unit. The condenser unit includes a thermal-insulated cooling bath with a permanently installed condenser tube, a cover, drain taps and an overflow outlet. Coolant fittings are mounted on the rear of the unit.
3.6. Chromatography-Mass Spectroscopy
Chromatograms of the benzene fractions of crude oil before and after catalytic aquathermolysis were recorded using a chromato-mass-spectrometric system, including a Chromatec-Crystal 5000 gas chromatography device with an ISQ mass-selective detector and Xcalibur software for processing the results. The chromatograph was equipped with a capillary column of 30 m long and 0.25 mm in diameter. The gas flow rate of helium was set at 1 mL/min, and the injector temperature was fixed at 310 °C. The thermostat’s temperature program increased from 100 to 300 °C at a rate of 3 °C/min followed by an isotherm until the end of the analysis. The voltage of the ion source was 70 eV, and the temperature was 250 °C. The compounds were identified using the NIST electronic library of mass spectra and according to the literature’s data.
3.7. Optical Density Photocolorimeter
Detecting the optical density or using a photocolorimeter is a method of quantifying the relative stability of petroleum distillate fractions in contrast to the distilled water chosen as a reference point. The optical density values, which are presented in the y-axis (Figure 5) are the ratio of light falling upon the gasoline samples to the intensity of light transmitted through the samples. This technique provides a comparison of the optical properties of solutions. Moreover, the concentration of a solute, shapes and even the sizes of dispersed particles can be evaluated through the given method. In this study, all petroleum distillate fractions were analyzed in a KFK-3 photoelectric photometer, which is designed to measure transmission coefficients, optical density and concentration of solutions. It operates in a wide range of wavelengths within the spectral range: 325–1000 nm. The working length of the cuvettes is 5-10-20-30-40-50-100 mm.
4. Conclusions
In this study, the catalytic effect of Al(H2PO4)3 and FeP nanoparticles on the upgrading of heavy oil produced from Ashalchinskoye by means of thermolysis was investigated. Moreover, the effects of NiCO3 on the activity of the catalytic nanoparticles were discussed. At 250 °C, both catalytic complexes showed high efficiency in the thermolytic upgrading of heavy oil. The catalytic complex Al(H2PO4)3 + NiCO3 provided the best performance, which can be summarized as follows:
- The viscosity of the heavy oil sample was reduced by 42%.
- The content of asphaltenes was reduced by 59.6%.
- The yield of light fractions increased.
- The boiling point was altered from 170 °C to 75 °C.
- The total content of resins and asphaltenes was reduced from 34%wt. to 14.7%wt.
- The total content of sulfur decreased from 4.72%wt. to 4.09%wt.
- The amount of isoalkanes after catalytic thermolysis increased from 22.26 wt.% to 36.31 wt.%.
The obtained results show that the quality of the heavy oil sample is significantly enhanced because of the catalytic thermolysis processes. The high catalytic activity of the given nanoparticles and their relatively low cost make them promising nanocatalysts for the thermolysis processes of heavy oil.
Author Contributions
Conceptualization, R.F.K. and A.V.V.; methodology, V.E.K.; investigation, Y.I.I.A.; resources, A.V.D.; writing—original draft preparation, F.A.A.; writing—review and editing, F.A.A.; supervision, R.F.K. and A.V.V.; funding acquisition, A.V.D. All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported by the Russian Science Foundation (Grant No. 22-73-00021).
Conflicts of Interest
The authors declare no conflict of interest.
References
- Wang, Y.Q. Study on Catalytic Aquathermolysis of Heavy Oil at Relatively Low Temperature; China University of Geosciences: Wuhan, China, 2010. [Google Scholar]
- Alvarado, V.; Manrique, E. Enhanced Oil Recovery: An Update Review. Energies 2010, 3, 1529–1575. [Google Scholar] [CrossRef]
- Tomczyk, N.A.; Winans, R.E.; Shinn, J.H.; Robinson, R.C. On the Nature and Origin of Acidic Species in Petroleum. 1. Detailed Acid Type Distribution in a California Crude Oil. Energy Fuels 2001, 15, 1498–1504. [Google Scholar]
- Li, G.; Chen, Y.; An, Y.; Chen, Y. Catalytic aquathermolysis of super-heavy oil: Cleavage of C-S bonds and separation of light organosulfurs. Fuel Process. Technol. 2016, 153, 94–100. [Google Scholar] [CrossRef]
- Rana, M.; Sámano, V.; Ancheyta, J.; Diaz, J.A.I. A review of recent advances on process technologies for upgrading of heavy oils and residual. Fuel 2007, 86, 1216–1231. [Google Scholar] [CrossRef]
- Vakhin, A.V.; Aliev, F.A.; Mukhamatdinov, I.I.; Sitnov, S.A.; Kudryashov, S.I.; Afanasiev, I.S.; Petrashov, O.V.; Nurgaliev, D.K. Extra-heavy oil aquathermolysis using nickel-based catalyst: Some aspects of in-situ transformation of catalyst precursor. Catalysts 2021, 11, 189. [Google Scholar] [CrossRef]
- Aliev, F.A.; Mukhamatdinov, I.I.; Sitnov, S.A.; Ziganshina, M.R.; Onishchenko, Y.V.; Sharifullin, A.V.; Vakhin, A.V. In-Situ Heavy Oil Aquathermolysis in the Presence of Nanodispersed Catalysts Based on Transition Metals. Processes 2021, 9, 127. [Google Scholar] [CrossRef]
- Fan, H.F.H.F.; Fan, Y.J.; Liu, X.F.; Zhao, L.G. Studies on effect of metal ions on aquathermolysis reaction ofLiaohe heavy oils under steam treatment. J. Fuel Chem. Technol. 2001, 29, 430–433. [Google Scholar]
- Chao, K.; Chen, Y.; Li, J.; Zhang, X.; Dong, B. Upgrading and visbreaking of superheavy oil by catalytic aquathermolysis with aromatic sulfonic copper. Fuel Process. Technol. 2012, 104, 174–180. [Google Scholar] [CrossRef]
- Mukhamatdinov, I.I.; Sh S, I.S.; Ismael, M.; Aliev, F.A.; Davletshin, R.R.; Vakhin, A.V. Influence of Naphthenic Hydrocarbons and Polar Solvents on the Composition and Structure of Heavy-Oil Aquathermolysis Products. Ind. Eng. Chem. Res. 2021, 60, 13191–13203. [Google Scholar] [CrossRef]
- Yang, Z.; Liu, X.; Li, X.; Zhao, M.; Zhang, Z.; Su, C. Preparation of silica supported nanoscale zero valence iron and its feasibility in viscosity reduction of heavy oil. Micro Nano Lett. 2014, 9, 355–358. [Google Scholar] [CrossRef]
- Wu, C.; Su, J.; Zhang, R.; Lei, G.; Cao, Y. The use of amphiphilic nickel chelate for catalytic aquathermolysis of extra-heavy oil under steam injection conditions. Energy Sources Part A Recovery Util. Environ. Eff. 2014, 36, 1437–1444. [Google Scholar] [CrossRef]
- Aliev, F.A.; Akhunov, A.; Mirzaev, O.; Vakhin, A.V. Development of New Amphiphilic Catalytic Steam Additives for Hydrothermal Enhanced Oil Recovery Techniques. Catalysts 2022, 12, 921. [Google Scholar] [CrossRef]
- Wen, S.; Zhao, Y.; Liu, Y.; Hu, S. A Study on Catalytic Aquathermolysis of Heavy Crude Oil during Steam Stimulation. In Proceedings of the International Symposium on Oilfield Chemistry, Houston, TX, USA, 28 February–2 March 2007. [Google Scholar]
- Clark, P.D.; Hyne, J.B. Steam–oil chemical reactions: Mechanisms for the aquathermolysis of heavy oil. AOSTRA J. Res. 1984, 1, 15–20. [Google Scholar]
- Hyne, J.B. A Synopsis of Work on the Chemical Reactions between Water and Heavy Oil Sands during Stimulated Steam Stimulation; AOSTRA Publication Series; AOSTRA: Calgary, Alberta, 1986. [Google Scholar]
- Li, Y.R.; Li, Q.Y.; Wang, X.D.; Yu, L.G.; Yang, J.J. Aquathermolysis of Heavy crude oil with ferric oleate catalyst. Pet. Sci. 2018, 15, 613–624. [Google Scholar] [CrossRef]
- Elayaraja, M.; Parashu, R.K.; Gavin, L.; Stephanie L, B. Control of Phase in Phosphide Nanoparticles Produced by Metal Nanoparticle Transformation: Fe2P and FeP. ACS Nano 2009, 3, 2383–2393. [Google Scholar] [CrossRef]
- Fei, X.M.; Cheng, Y.X.; Fucong, L.; Shu, C.S.; Yang, Y.L.; Jian, L.; Liang, Z. Construction of FeP Hollow Nanoparticles Densely Encapsulated in Carbon Nanosheet Frameworks for Efficient and Durable Electrocatalytic Hydrogen Production. Adv. Sci. 2019, 6, 1801490. [Google Scholar] [CrossRef]
- Kayukova, G.P.; Mikhailova, A.N.; Kosachev, I.P.; Musin, R.Z.; Nasyrova, Z.R.; Aliev, F.A.; Vakhin, A.V. Hydrothermal Impact on Hydrocarbon Generation from Low-Permeable Domanic Sedimentary Rocks with Different Lithofacies. Energy Fuels 2021, 35, 11223–11238. [Google Scholar] [CrossRef]
- Chen, Y.; He, J.; Wang, Y.; Li, P. GC-MS used in study on the mechanism of the viscosity reduction of heavy oil through aquathermolysis catalyzed by aromatic sulfonic H3PMo12O40. Energy 2010, 35, 3454–3460. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).