Abstract
Chitin is one of the most abundant biopolymers on Earth but under-utilized. The effective conversion of chitin biomass to useful chemicals is a promising strategy to make full use of chitin. Among chitin-derived compounds, some furan derivatives, typically 5-hydroxymethylfurfural and 3-acetamido-5-acetylfuran, have shown great potential as platform compounds in future industries. In this review, different catalytic systems for the synthesis of nitrogen-free 5-hydroxymethylfurfural and nitrogen-containing 3-acetamido-5-acetylfuran from chitin or its derivatives are summarized comparatively. Some efficient technologies for enhancing chitin biomass conversion have been introduced. Last but not least, future challenges are discussed to enable the production of valuable compounds from chitin biomass via greener processes.
1. Introduction
The fast consumption of unrenewable fossil resources for the production of bulk chemicals and fuels has led to serious resource and environmental problems, which has attracted more interest in the utilization of renewable resources [1,2,3,4,5]. Due to renewability, abundance, and structure diversity, biomass is regarded as the best candidate to replace fossil resources. Cellulose, chitin, hemicellulose, and lignin are typical biomass resources [6,7]. Among these, chitin is one of the most abundant biopolymers and can be extracted from cheap crustacean shells, exoskeletons of insects, and the cell walls of fungi [8,9]. Similar to cellulosic biomass-based biorefinery, the novel concept of “Shell Biorefinery” was proposed last decade [10,11]. This concept has driven the development in the extraction of major components (typically chitin) from crustacean shells and the transformation of them into useful products. Owing to ca. 7 wt% of bio-fixed nitrogen in chitin, the utilization of chitin or its derivatives provides an alternative method for the preparation of nitrogen-containing chemicals and materials, which is independent of ammonia synthesis by the Haber process.
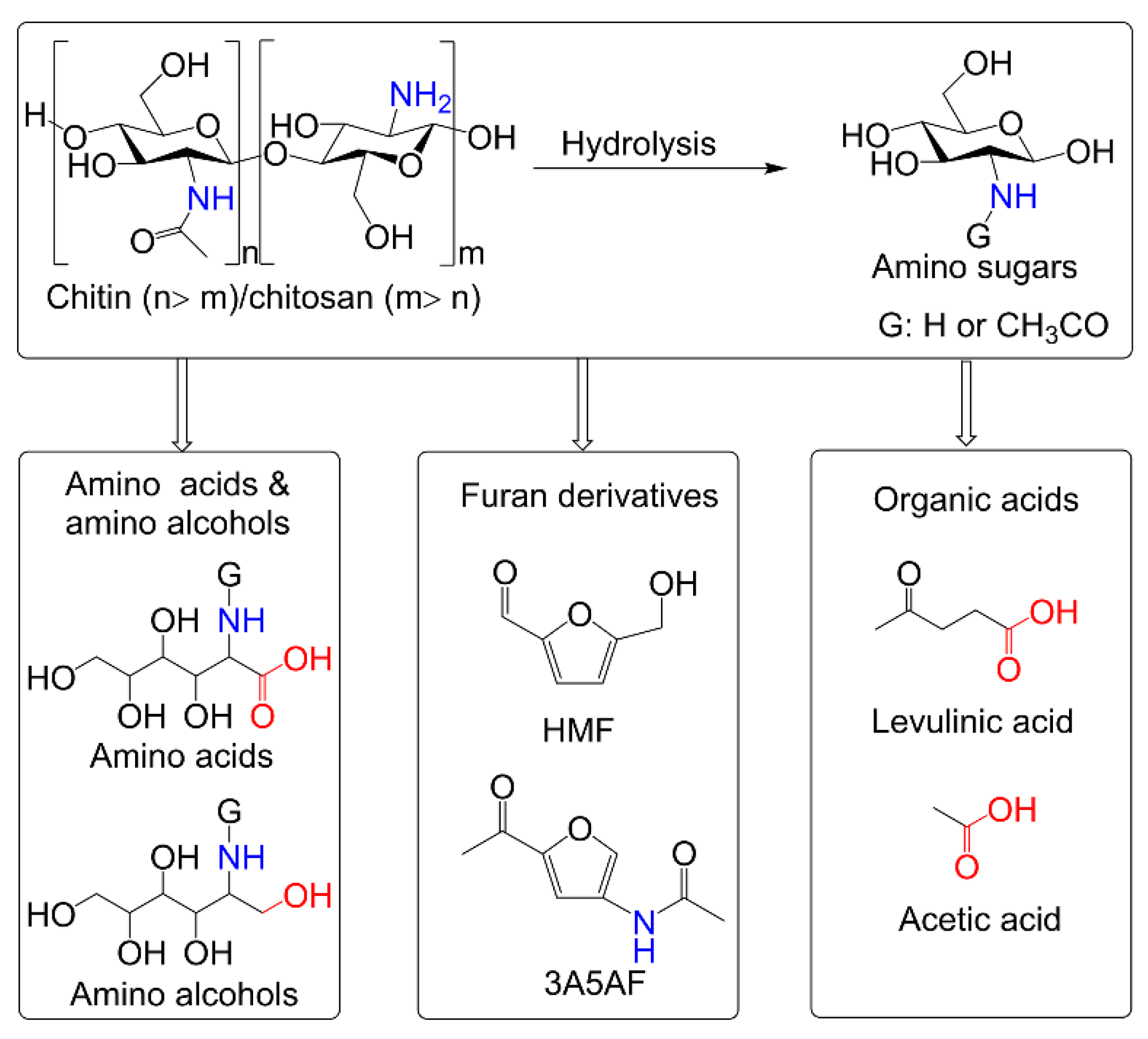
Chitosan is the deacetylation product of chitin. Nitrogen-containing glucosamine (GlcNH2) and N-acetylglucosamine (GlcNAc) can be prepared by the hydrolysis of chitin and chitosan, respectively. Some interesting works have been devoted to the catalytic conversion of chitin biomass (mainly referring to chitin-based polymers and monomers) to value-added chemicals, such as 5-hydroxymethylfurfural (HMF), 3-acetamido-5-acetylfuran (3A5AF), 5-(chloromethyl)furfural, levulinic acid, acetic acid, glucosaminic acid, and 2-acetamido-2-deoxysorbitol [12,13,14,15], as shown in Scheme 1. Among these, both as furan derivatives, HMF and 3A5AF are typical products of chitin biomass conversion. HMF, usually obtained from fructose, glucose, and their polysaccharides, is a well-known versatile platform compound and can be converted into building blocks of polymers, liquid fuel additives, pharmaceuticals, and other fine chemicals [16,17,18]. Compared to HMF that has been extensively investigated, 3A5AF is an emerging platform chemical, which has exhibited the potential to be a substrate for the synthesis of nitrogen-containing fine chemicals [19]. In this review, we focus on the production of furan-derivatives (typically HMF and 3A5AF) from chitin biomass, where different feedstocks and catalytic systems are discussed briefly.
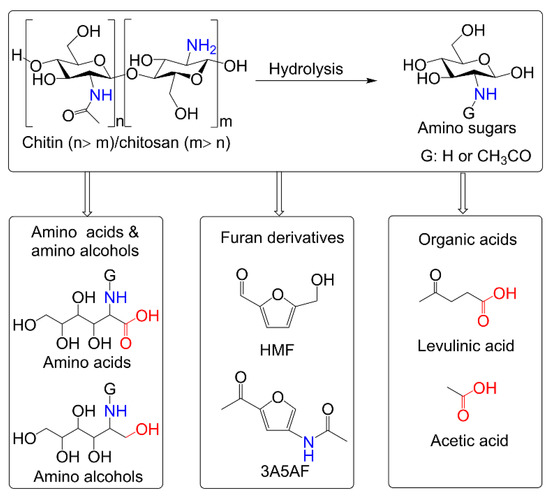
Scheme 1.
Some useful chemicals from chitin biomass.
2. Conversion of Chitin Biomass to HMF
2.1. Bronsted Acid as Catalyst
Bronsted acids (i.e., H2SO4, HCl) have been widely used in the acid-catalyzed conversion of biomass. They have some advantages, such as low cost and easy production, on a large scale. Jeong [20] studied the hydrothermal transformation of GlcNH2·HCl by dilute H2SO4. The highest HMF yield was about 3.0 mol% at 175 °C for 5 min employing 2 wt% H2SO4. Levulinic acid, a rehydration product of HMF, was another main product. With the increase in reaction time, the HMF yield decreased and the levulinic acid yield remarkably increased. After 65 min, a 44 mol% yield of levulinic acid was obtained while HMF was not detectable. Due to the instability of HMF especially in an acidic medium, the catalytic conversion of chitin biomass to HMF need be conducted under relatively mild conditions. Interestingly, Jeong et al. [21] subsequently reported a similar catalytic system to produce HMF from chitosan, the polymer of GlcNH2, giving the highest HMF yield of 15 mol% at 174 °C for 36.9 min in a 2.2 wt% aqueous H2SO4 solution. Both long reaction time and high temperature could enhance the further decomposition of HMF to levulinic acid and formic acid.
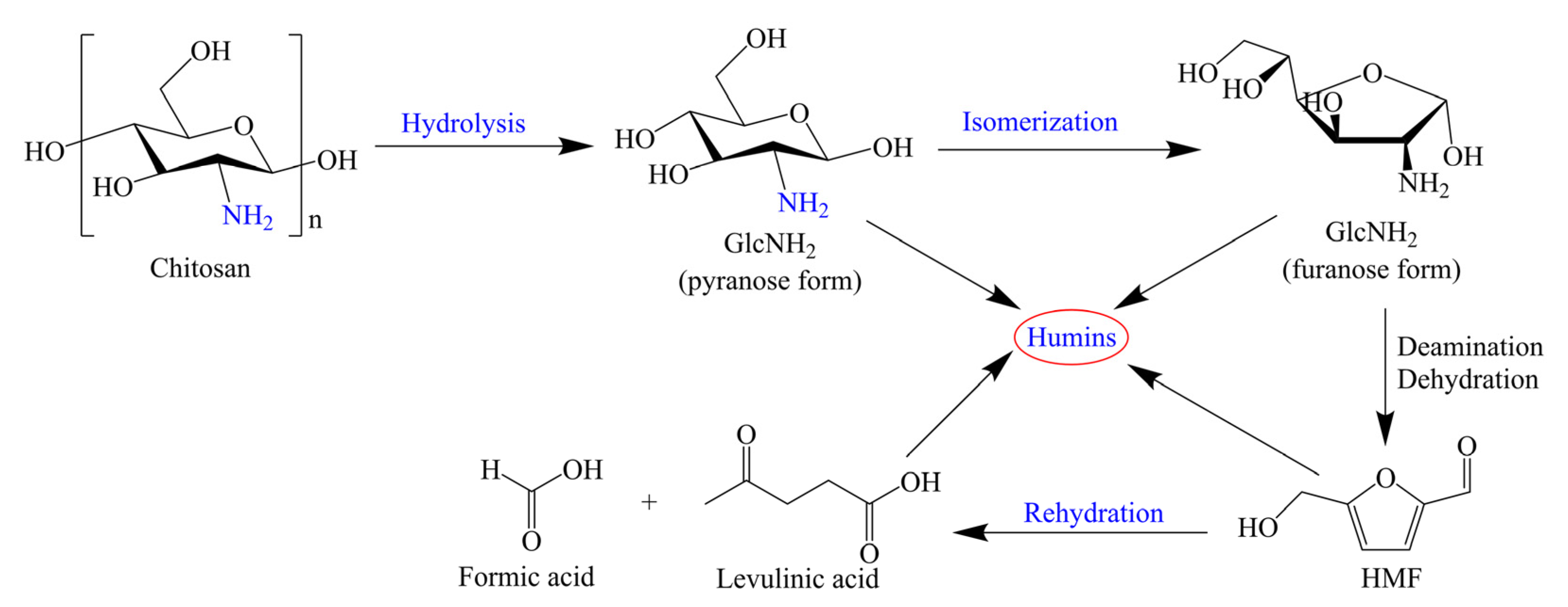
In addition to H2SO4, Jeong’s group has also studied the synthesis of HMF and levulinic acid over other Bronsted acids [22,23,24]. Sulfamic acid bearing both acidic and basic sites has nonvolatile, odorless, nonhygroscopic, and noncorrodible physical properties. Sulfamic acid could effectively catalyze the sequential hydrolysis of chitosan and the dehydration/deamination of GlcNH2, giving an HMF yield of 27 mol% at 200 °C for 2 min, while the yield of levulinic acid was only 0.6 mol%. In comparison with the H2SO4-catalyzed chitosan conversion, the short reaction time employing sulfamic acid is an advantage. The synergetic effect of Bronsted–Lewis acid sites might contribute to good catalytic performance. A possible reaction pathway of sulfamic acid-catalyzed HMF production from chitosan has been proposed, as shown in Scheme 2. First, the hydrolysis of chitosan over Bronsted acid sites produces GlcNH2 bearing an amine group (–NH2). Then, GlcNH2 in pyranose form is isomerized to form furanose one. Under the acidic conditions, the amine group in GlcNH2 is eliminated through a deamination reaction, and the further dehydration and keto-enol tautomerization afford HMF. Under harsh conditions (i.e., strong acidic medium), HMF is remarkably unstable, which reduces the yield of HMF and produces byproducts (i.e., levulinic acid, humins) [22]. Another example using Bronsted acid is methanesulfonic acid [23]. Methanesulfonic acid was effective for the conversion of GlcNH2 and chitosan, and levulinic acid was the main product in most cases due to the strong acidity of the catalyst. Starting from chitosan (2 wt%), the low methanesulfonic acid concentration (0.1 M) and short reaction time (15 min) at 200 °C gave a 19 mol% yield of HMF (Table 1, entry 4), but the HMF yield significantly decreased to 8.2 mol% with a reaction time of 30 min (Table 1, entry 5).
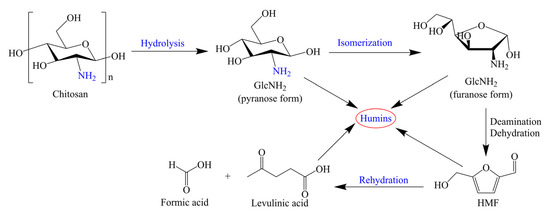
Scheme 2.
Possible reaction pathways of HMF formation from chitosan by sulfamic acid-catalyzed hydrothermal conversion. Adapted with permission from Ref. [22]. Copyright 2018, Wiley.

Table 1.
Catalytic conversion of chitin biomass to HMF over Bronsted acid catalysts.
Some typical results of the Bronsted acid-catalyzed production of HMF from chitin biomass are summarized in Table 1.
2.2. Lewis Acid as Catalyst
A number of Lewis acids have been used for chitin biomass conversion. Omari et al. [25] investigated the catalytic performance of several Lewis acids for the production of HMF and levulinic acid. Some interesting findings have been summarized as follows. Metal salts as Lewis acids consist of anions and cations. The experiment results reveal that both anions and cations have an effect on chitosan conversion and anions play more important roles. Under relatively dilute conditions (100 mg of chitosan, 0.12 mmol of catalyst, and 15 mL of water), the HMF yield was dependent significantly on the anion and followed the trend: CF3SO3− < ClO4− < Cl−. Among these Lewis acids, SnCl4·5H2O gave the best results with an HMF yield up to 13 mol%, while no levulinic acid was detected. Notably, with the increase in SnCl4·5H2O concentration to 0.06 M (100 mg of chitosan, 0.24 mmol of catalyst, and 4 mL of water), a 33 mol% yield of levulinic acid with a small amount of HMF (less than 1 mol% yield) was obtained, indicating the significant effect of acid usage on product selectivity. In addition, the application of microwave irradiation as a heating technology enhanced the production of HMF and other compounds. This catalytic system was effective for the conversion of different chitosan materials, including commercial chitosan with different molecular weights, and extracted chitosan from shrimp or crab, giving an 11–13 mol% yield of HMF. Wang et al. [26] studied the conversion of chitin biomass to HMF over a concentrated ZnCl2 aqueous solution. Owing to the strong interaction of Zn2+ with the hydroxyl/amino/acetamide group, the solubility of chitin biomass in concentrated ZnCl2 aqueous solution was improved. In a 67 wt% ZnCl2 solution, the HMF yields from GlcNH2 and Chitosan-1K reached 22 mol% and 10 mol%, respectively. Though HMF could be produced in this medium, the high ZnCl2 usage (mass ratio of ZnCl2/substrate as 20/1) might result in separation difficulty.
In the above-mentioned studies, green and cheap water is the sole solvent. However, the poor stability of HMF in water usually leads to the formation of many unwanted side-products. The addition of an appropriate organic solvent is a good approach to improve HMF selectivity. For example, dimethyl sulfoxide (DMSO) is a typical organic solvent that benefits the production of HMF from carbohydrates (i.e., fructose and glucose) because of its unique features [27]. DMSO is not only a good catalyst for the dehydration of fructose to HMF, but also a solvent that can inhibit some undesirable side-reactions effectively. Yu et al. [28] demonstrated the conversion of chitin biomass to HMF in a DMSO-water mixture over metal salts, affording 27 mol% and 38 mol% yields of HMF over FeCl2·4H2O from chitosan-350K and GlcNAc at 180 °C for 5 h, respectively. In addition to DMSO, the combination of water and tetrahydrofuran as a reaction medium also gave an enhanced HMF yield from GlcNAc. This FeCl2·4H2O-DMSO-water catalysis system was effective for the conversion of chitin-400K to HMF with a 19 mol% yield. Due to the poor solubility of biopolymers caused by abundant intra- and intermolecular hydrogen bonds, the yields of HMF from chitin and chitosan were lower than that from GlcNAc. In fact, the bad solubility is still a major challenge for the efficient utilization of polysaccharides, including cellulose, chitin, and chitosan [15,29].
Some typical results of the Lewis acid-catalyzed production of HMF from chitin biomass are summarized in Table 2.

Table 2.
Catalytic conversion of chitin biomass to HMF over Lewis acid catalysts.
2.3. Ionic Liquid as Catalyst
As mentioned earlier, the large number of hydrogen bonds in chitin-based biopolymers lead to difficulties in the dissolution and degradation of chitin and chitosan. Ionic liquids exhibit the ability to cleave hydrogen bonds in polysaccharides and form new hydrogen bonds between anions and the hydroxyl groups of biopolymers, improving the dissolution [30,31].
Several studies from Zang’s group have proven the potential of ionic liquids as catalysts for chitin biomass conversion to HMF. Acidic ionic liquids exhibited a better catalytic performance [32]. Among these ionic liquids, N-methyl imidazolium hydrogen sulfate ([MIM]HSO4) as a catalyst in aqueous medium gave the highest HMF yield of 30 mol% from low-molecular-weight chitosan at 180 °C for 5 h. The protons generated from acidic ionic liquids in water were believed to play a key role in destroying existing hydrogen bonds and forming new ones, which enhanced the hydrolysis of chitosan. Notably, the recovered [MIM]HSO4 showed no obvious activity loss for chitosan-HMF conversion after five catalytic runs.
The ionic liquid type, solvent, and other reaction parameters have remarkable effects on HMF formation from chitin biomass. Zhang et al. [33] reported that benzimidazolium chloride ([Hbim]Cl) could effectively catalyze the synthesis of HMF from chitosan in water, and the addition of DMSO with a mass ratio of DMSO/water of 1/9 increased the HMF yield from 31 mol% to 35 mol%. The further increase in DMSO/water ratio gave a lower HMF yield, indicating the synergistic effect of DMSO and water in this conversion. In brief, water is beneficial for HMF formation and DMSO suppresses the undesirable conversion of HMF. Zang et al. [34] focused on the degradation of the monomer of chitin, GlcNAc, to HMF. N-methylimidazolium hydrogen sulfate ([Hmim][HSO4]) exhibited good catalytic performance in a DMSO-water medium, giving a 65 mol% yield of HMF at 180 °C for 6 h. In addition, this catalytic system could catalyze the conversion of GlcNH2·HCl, chitosan, and chitin to HMF in 55 mol%, 35 mol%, and 26 mol% yields, respectively, and the recycled [Hmim][HSO4] also exhibited good activity. As ionic liquids have some drawbacks, including high cost, complicated manufactory process, and poor biodegradability, the use of ionic liquids as catalysts instead of solvents is a more practical method in biorefinery.
Some typical results of the ionic liquid-catalyzed production of HMF from chitin biomass are summarized in Table 3.

Table 3.
Catalytic conversion of chitin biomass to HMF over ionic liquid catalysts.
2.4. Other Catalyst Types
In addition to traditional Bronsted acids, Lewis acids, and emerging ionic liquids, there are a few examples of HMF production from chitin biomass using other catalysts or combinations of them. Recently, Islam et al. [35] demonstrated that polyoxometalate as a catalyst could catalyze the conversion of pretreated chitin to HMF in DMSO-water medium. It is well-known that the high crystallinity and poor solubility are two main challenges for the effective degradation of chitin. Therefore, compared to chitosan, GlcNH2, and GlcNAc, the use of chitin usually gives a lower HMF yield. Consistent with previous reports [36], the ball milling treatment of the chitin-H2SO4 mixture obviously reduced the crystallinity and improved the solubility. The H2SO4-assisted ball milling produced a large amount water-soluble products with a total yield of nearly 80%. Overall, the combination of pretreatment and microwave heating technology afforded an enhanced HMF yield of 23 mol% over H4SiW12O40 at 180 °C for 3 min.
Another interesting example has been reported by Dandekar’s group [37]. This investigation employed a combination of homogenous and heterogeneous acid catalysts for the conversion of chitosan to HMF. Solid acids can be easily isolated from the reaction mixture, and it is easier for homogeneous acids to gain access to the glycosidic linkage because of less steric hindrance. Under optimized conditions, the combination of H-β zeolite and dilute acetic acid aqueous solution gave the highest HMF yield of 15 mol% to 28 mol% depending on the molecular weight of the parent chitosan. Acetic acid as a “green” organic acid can be produced from biomass-based feedstock [38,39]. Unfortunately, the obvious activity loss of recovered H-β zeolite was observed, and only half the HMF yield compared to fresh H-β zeolite was obtained. Thus, despite being a promising strategy, the development of stable heterogeneous solid acid catalysts for chitin biomass conversion still needs more effort.
3. Conversion of Chitin Biomass to 3A5AF
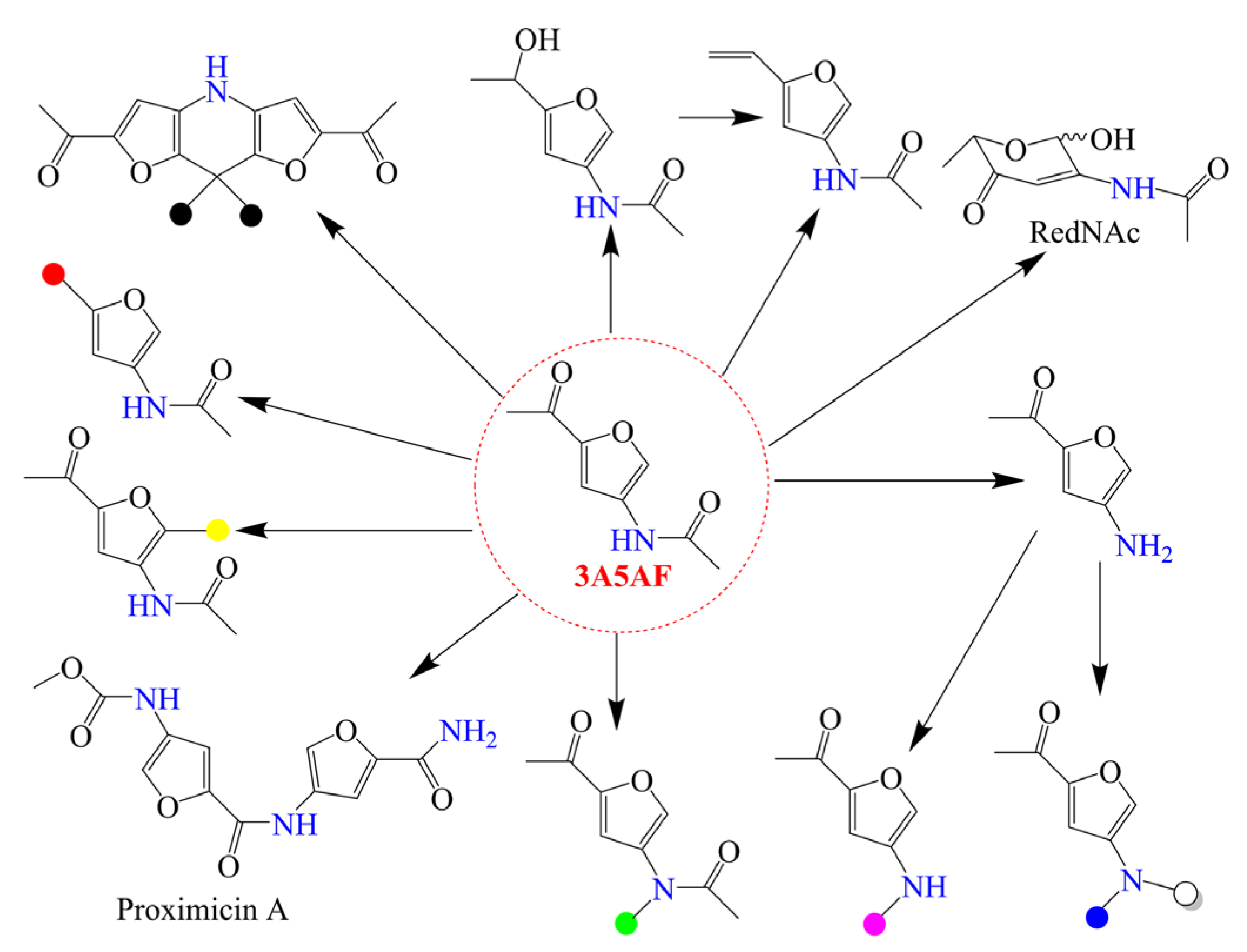
The deamination and dehydration of chitin biomass in a water-containing solvent produce HMF as a product employing different acid catalysts. Although some efforts have been devoted to this research topic, in comparison with cellulose-based feedstock (cellulose and its degradation derivatives), the use of chitin, chitosan, and their monomers to produce HMF is not such a good option. First, both the organic nitrogen-containing groups in chitin biomass and NH3 generated by deamination reaction can interact with acid catalysts, resulting in a loss of catalytic activity. Moreover, organonitrogen compounds are mainly obtained starting from the energy-intensive Haber ammonia synthesis. Therefore, the direct production of organonitrogen compounds by the retention of bio-fixed nitrogen is an attractive strategy for chitin biomass valorization. Under some reaction conditions, the dehydration of GlcNAc with the retention of nitrogen element gives another furan-derivative 3A5AF. As an emerging biochemical with high reactivity, 3A5AF can be converted into a number of value-added compounds, as shown in Scheme 3. For example, Sadiq et al. [40] demonstrated the total synthesis of anticancer alkaloid proximicin A starting from 3A5AF. Compared to previous methods [41,42], safer reagents, reactants, and mediums were used, and biorenewable nitrogen in 3A5AF was transferred to a biologically active alkaloid. In fact, it is difficult to install a nitrogen substituent at the C3 position of the furan heterocycle, and 3A5AF with its special structure is a promising precursor for the synthesis of many useful 3-amidofurans [43,44]. Recently, the catalytic conversion of GlcNAc and chitin toward 3A5AF has attracted increasing attention.
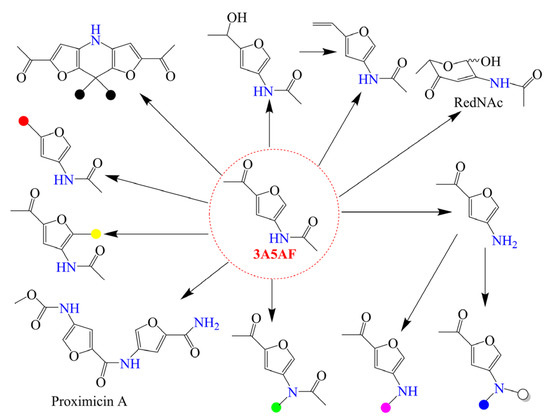
Scheme 3.
Some useful derivatives from 3A5AF, where the colored circles represent different chemical groups. Reproduced with permission from Ref. [19], copyright Wiley.
3.1. GlcNAc as Reactant
The selective removal of three water molecules from GlcNAc produces 3A5AF, which is similar to the dehydration of glucose to HMF [45]. Franich et al. [46] investigated the direct pyrolysis of GlcNAc in vacuo, and a 2% isolated yield of 3A5AF with 5% 3-acetamidofuran was obtained. This is a simple method to produce 3A5AF, but the selectivity is unsatisfactory.
Compared to unselective pyrolysis, the catalytic conversion of GlcNAc in appropriate solvents can remarkably enhance 3A5AF selectivity. Both organic solvents and ionic liquids have been widely used as reaction mediums. Omari et al. [47] studied the conversion of GlcNAc to 3A5AF over NaCl and B(OH)3. It is notable that the solvent has a significant effect on adjusting the product selectivity. Some dipolar aprotic solvents, such as dimethylformamide (DMF), N-methyl-2-pyrrolidone (NMP), acetonitrile (CH3CN), dimethylacetamide (DMA), and DMSO, favored the formation of 3A5AF. Considering environmental, health, and safety issues of these solvents, DMA was chosen for further investigation. After systematic optimization, a 77 mol% yield of 3A5AF was obtained in DMA at 220 °C for 15 min using microwave irradiation, where 100 mol% B(OH)3 and 200 mol% NaCl were used. The increase in NaCl usage to 400 mol% gave a slight increase in 3A5AF yield to 82 mol%. Unlike HMF production from chitin biomass in water-containing medium [12,15], the presence of water had an obvious negative effect on 3A5AF synthesis. Under identical conditions, the addition of a small amount of water into DMA (water/DMA, v/v, 1/99) led to the decrease in 3A5AF yield from 77 mol% to ca. 53 mol%. Padovan et al. [48] reported a simple method for 3A5AF synthesis from GlcNAc in DMF over commercial AlCl3·6H2O by conventional heating. Under identical conditions, the use of 100 mol% AlCl3·6H2O afforded a much higher 3A5AF yield than those over the combination of 100 mol% B(OH)3 and 200 mol% NaCl. However, the optimized yield of 3A5AF (30 mol%) over AlCl3·6H2O was much lower than the previous record [47,48]. Very recently, Wang et al. [49] demonstrated that the combination of NH4Cl and LiCl gave a 43 mol% yield in DMF at 160 °C for 5 min. Significantly, the solvent (DMF), catalyst (NH4Cl), and additive (LiCl) could be recycled by a simple procedure, and a residual catalytic activity of over 90% could be obtained after five catalytic runs. In addition, the produced 3A5AF was also isolated from reaction mixtures with high purity. Therefore, this work provides a promising example of a recyclable catalysis system for chitin biomass utilization.
Recently, several investigations on the preparation of 3A5AF from GlcNAc employing ionic liquid catalysts have been reported [50,51,52,53]. NMP, DMA, and DMF have been proven as favorable reaction mediums for GlcNAc transformation. Different ionic liquids, such as amino acid ionic liquids, quaternary ammonium ionic liquids, ethanolamine ionic liquids, and pyridinium-based ionic liquids, have shown good catalytic performance for 3A5AF formation, and the presence of additives (i.e., B(OH)3 and CaCl2) further enhanced the 3A5AF yield significantly. For example, Wang et al. [50] studied the conversion of GlcNAc to 3A5AF employing amino acid chloride as an ionic liquid catalyst. The use of 200 mol% glycine chloride gave a 43 mol% yield of 3A5AF in DMA at 200 °C for 10 min without other additives. The addition of CaCl2 improved the 3A5AF yield to 53 mol%. The spent glycine chloride could be recycled and exhibited stable catalytic performance, with the 3A5AF yield slightly decreasing from 43 mol% (1st run) to 37 mol% (8th run). This work used renewable and nontoxic amino acids as precursors to prepare recyclable catalysts, which is highly attractive. Subsequently, more ionic liquids have been reported for this reaction, and the 3A5AF yield reached 70 mol% in DMA using pyrazine hydrochloride ionic liquid and additives (B(OH)3 and CaCl2) [51].
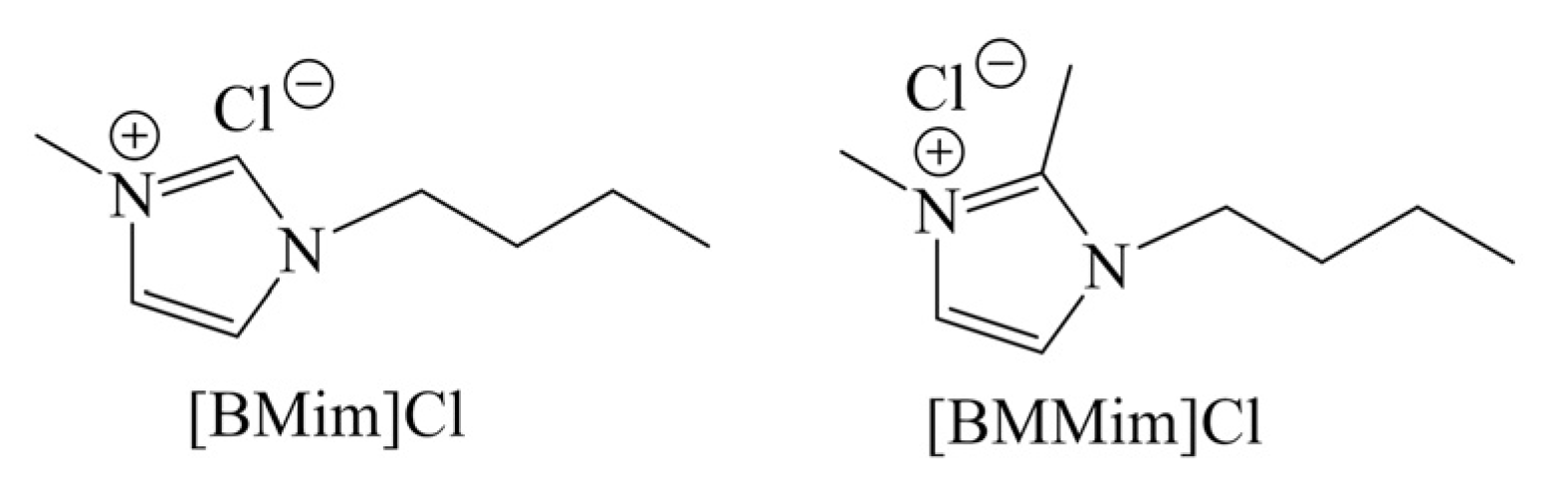
Because of unique characteristics, ionic liquids can act as both solvent and catalyst. Drover et al. [54] demonstrated the direct conversion of GlcNAc to 3A5AF in ionic liquids, giving 34 mol% and 33 mol% yields of 3A5AF in 1-butyl-3-methylimidazolium chloride ([BMim]Cl) and 1,2-dimethyl-3-butylimidazolium chloride [BMMim]Cl, respectively, in the absence of any other catalysts or additives (Table 4, entry 7–8). The structures of [BMim]Cl and [BMMim]Cl are shown as Scheme 4. The anion of ionic liquid has a significant effect on 3A5AF selectivity, where a chloride counterion was found necessary to form 3A5AF compared to bromide and acetate. Due to the advantages of microwave heating, the reaction time was short as 3 min. The addition of 10 mol% B(OH)3 in [BMim]Cl and the use of oil bath heating gave a 79 mol% yield of 3A5AF for 60 min. Considering the cost and high viscosity, the use of ionic liquid as a medium is uneconomic and difficult to operate.

Table 4.
Catalytic conversion of GlcNAc to 3A5AF.
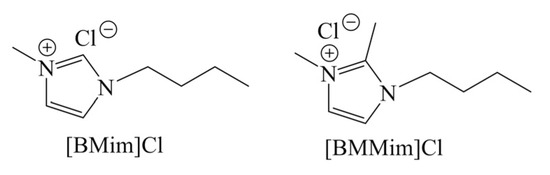
Scheme 4.
Two typical ionic liquids for 3A5AF synthesis.
Some typical results of GlcNAc conversion to 3A5AF are summarized in Table 4.
3.2. Chitin as Reactant
As discussed in Section 3.1, a considerable 3A5AF yield has been obtained from GlcNAc. However, the selective production of GlcNAc from chitin is still challenging, which requires the cleavage of the glucosidic bond and retention of the acetamido group [55]. Therefore, the selective production of 3A5AF from chitin can avoid the purification of intermediate GlcNAc, which is desired but difficult. Limited efforts have been devoted to this topic.
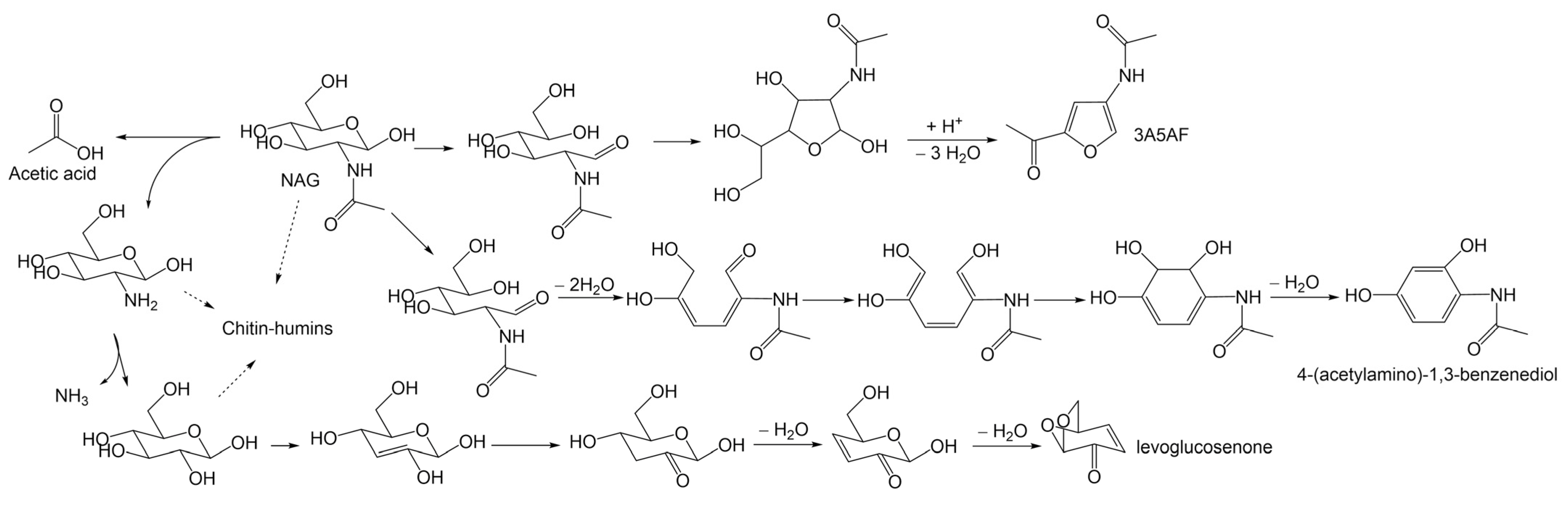
Chen et al. [56] explored the preparation of 3A5AF from chitin in a “one-pot, one-step” process, affording a ca. 8 mol% yield of 3A5AF with 50% conversion in NMP at 215 °C for 2 h. The use of chitin as a substrate required harsh conditions, such as a larger usage of B(OH)3 and NaCl, higher reaction temperature, and longer reaction time, but produced less 3A5AF and a large amount of side-products, such as levoglucosenone, 4-(acetylamino)-1,3-benzenediol, acetic acid, and chitin-humins. A proposed reaction network is shown as Scheme 5. Notably, the degradation of the crystalline region in chitin was found to be the rate-determining step, which was supported by kinetic study results and provided a clue to promote chitin conversion.
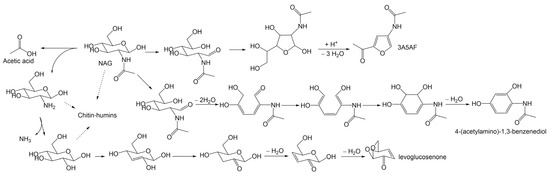
Scheme 5.
Proposed reaction pathways for the conversion of chitin to identified products. Adapted from Ref. [56], copyright Royal Society of Chemistry.
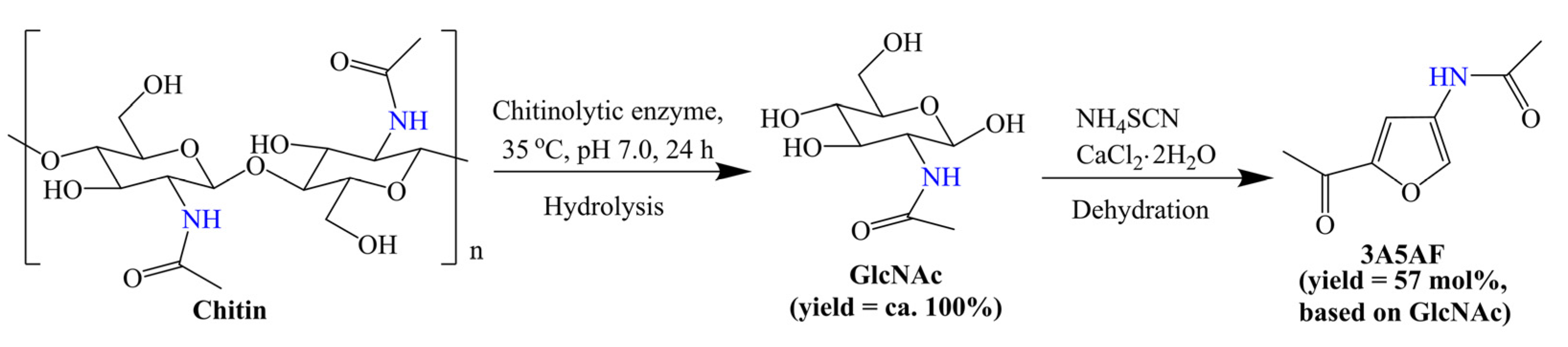
Some methods have been used to destroy the crystalline structure of chitin, which facilitates the further conversion of chitin to small-molecule products. As mentioned earlier, some ionic liquids can promote the dissolution of biopolymers. Chen et al. [57] investigated 3A5AF synthesis from chitin in a range of ionic liquids. Notably, the Cl− in the ionic liquids not only promoted the dissolution of chitin, but also participated in the degradation of it. After optimization, a ca. 7 mol% yield of 3A5AF, which is comparable to that in NMP, was obtained in the presence of B(OH)3 and HCl under milder conditions [56,57]. In addition to the use of special solvents, the pretreatment of chitin is another useful method. The same group [58] found that ball mill treatment without additives was beneficial to the destruction of the hydrogen-bond network and crystalline region, which significantly enhanced the reactivity of chitin and gave a 29 mol% yield of 3A5AF in [BMIm]Cl. Very recently, Chen et al. [59] demonstrated a novel chemo-enzymatic protocol for the transformation of chitin to 3A5AF, as shown in Scheme 6. Unlike previous investigations [56,57,58], chitin was nearly 100% converted into GlcNAc over the recyclable chitinolytic enzyme. The produced GlcNAc was separated and used as a substrate for 3A5AF synthesis in organic solvents. A new finding is that both ammonium thiocyanate (NH4SCN) and thiourea (CH4N2S) were effective for the dehydration of GlcNAc to 3A5AF, and a 57 mol% yield of 3A5AF was obtained in DMA over NH4SCN with CaCl2 2H2O as an additive. Unfortunately, the recovered enzyme and NH4SCN exhibited decreasing catalytic performance. This protocol provides a step-wise conversion of chitin over enzymes and inorganic catalysts, giving an enhanced overall yield of 3A5AF. In addition, the recyclability of catalysts was also considered, which pushes the effective and green production of 3A5AF from chitin.
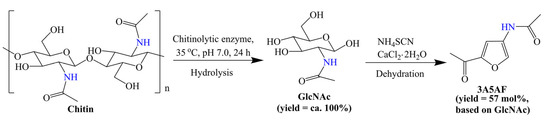
Scheme 6.
Step-wise conversion of chitin to 3A5AF. Adapted with permission from Ref. [59]. Copyright 2021, copyright Elsevier.
4. Conclusions and Perspectives
4.1. Conclusions
Chitin is an abundant nitrogen-containing polysaccharide. The effective transformation of chitin and its derivatives (i.e., chitosan, GlcNAc, and GlcNH2) to valuable chemicals has become an emerging research topic. Among chitin-derived compounds, two typical furan-derivatives, including HMF and 3A5AF, have attracted the most attention.
HMF is a nitrogen-free platform compound that is widely obtained by the dehydration of C6 carbohydrates and their oligosaccharides/polysaccharides. Chitin biomass has also been converted into HMF and its downstream products. Different acid catalysts, such as Bronsted acids, Lewis acids, acidic ionic liquids, heteropolyacids, and the combination of solid acid and homogeneous acids, have been used for HMF production. Moderate HMF yields (up to 66 mol% and 55 mol%) have been obtained from GlcNAc and GlcNH2·HCl, respectively. In contrast, the use of biopolymers (chitin and chitosan) usually gives lower HMF yields (lower than 40 mol%), which can be explained by the difficulties in dissolution and hydrolysis of these biopolymers. The special structure characteristics, mainly including high crystallization and abundant hydrogen bonds, should be the reasons for worse selectivity. Limited examples using heterogeneous catalysts have been reported, but the catalytic performance and stability are still unsatisfactory.
Compared to the synthesis of HMF, the production of 3A5AF from chitin biomass is a more interesting option because of its organonitrogen group. A few efforts have been devoted to 3A5AF production from GlcNAc. The selectivity of 3A5AF is highly dependent on the reaction medium. Some polar aprotic organic solvents (i.e., DMA, DMF, and NMP) and ionic liquids bearing Cl− are favorable choices, while the addition of a small amount of water decreases the 3A5AF yield remarkably. Chlorine-containing metal salts/ionic liquids and B(OH)3 as catalysts/additives are beneficial to the formation of 3A5AF. A ca. 80 mol% yield of 3A5AF has been obtained in DMA or [BMim]Cl. The conversion of chitin is a more promising strategy, which can avoid the isolation of GlcNAc. However, a larger amount of catalysts/additives and harsher conditions are usually required. The use of appropriate solvents (typically chlorine-containing ionic liquids) can enhance the dissolution and degradation, resulting in chitin conversion under milder conditions. In addition, the pretreatment of chitin by mechanical force can destroy the regular crystal region in chitin and increase the reactivity. A step-wise conversion of chitin to 3A5AF is an effective method to enhance the overall yield, and biocatalysis has shown its advantages such as high efficiency, mild conditions, and relative stability.
In comparison to cellulose, the investigations on the catalytic conversion of chitin biomass to value-added furan derivatives are still not sufficient. Particularly, the direct utilization of biopolymers encounters a number of challenges, and many catalytic systems do not meet the principles of green chemistry. Hence, many more efforts need be devoted to this area.
4.2. Perspectives
Based on our knowledge, we provide some research suggestions as follows. (1) The selective depolymerization of chitin/chitosan to monomers is a crucial step to produce valuable chemicals with low molecular weight. Some new technologies, such as biocatalysis, pretreatment (i.e., ball mill grinding), and microwave heating, might be potential methods for the effective dissolution/degradation of chitin and chitosan. (2) Novel, green, and efficient catalytic systems are still lacking. Both catalytic performance and environmental impact should be considered. The recyclability of catalysts/additives/solvents and the isolation of products need be further investigated. (3) There are few insightful studies on the reaction mechanism, which limits the deep understanding of the investigated reactions. (4) Compared to the synthesis of HMF-based products, the production of nitrogen-containing 3A5AF and its derivatives is of more significance, which makes full use of bio-fixed nitrogen in chitin biomass.
Author Contributions
Conceptualization, B.X. and J.D.; methodology, B.X. and Z.D.; formal analysis, Z.D. and J.D.; writing—original draft preparation, B.X., Z.D., R.Y. and D.Y.; writing—review and editing, B.X. and J.D.; visualization, X.G.; project administration, J.D.; funding acquisition, J.D., N.L. and F.L. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the Natural Science Foundation of Chongqing (cstc2020jcyj-msxmX0672, cstc2020jscx-msxmX0076, and cstc2021ycjh-bgzxm0242), National Natural Science Foundation of China (No.21808021), Chongqing Talent Innovation and Entrepreneurship Demonstration Team (CQYC201903189), Science and Technology Project of Chongqing Technology and Business University (KFJJ2019089, 1956037 and 2152027), and Key Disciplines of Chemical Engineering and Technology in Chongqing Colleges and Universities during the 13th Five-Year Plan (950619013).
Conflicts of Interest
The authors declare no conflict of interest.
References
- Kang, Y.; Yang, Q.; Bartocci, P.; Wei, H.; Liu, S.S.; Wu, Z.; Zhou, H.; Yang, H.; Fantozzi, F.; Chen, H. Bioenergy in china: Evaluation of domestic biomass resources and the associated greenhouse gas mitigation potentials. Renew. Sustain. Energy Rev. 2020, 127, 109842. [Google Scholar] [CrossRef]
- Liao, Y.; Koelewijn, S.-F.; Van den Bossche, G.; Van Aelst, J.; Van den Bosch, S.; Renders, T.; Navare, K.; Nicolai, T.; Van Aelst, K.; Maesen, M.; et al. A sustainable wood biorefinery for low-carbon footprint chemicals production. Science 2020, 367, 1385–1390. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Luo, N.; Xie, S.; Zhang, H.; Zhang, Q.; Wang, F.; Wang, Y. Photocatalytic transformations of lignocellulosic biomass into chemicals. Chem. Soc. Rev. 2020, 49, 6198–6223. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Guo, H.; Fang, Z.; Aida, T.M.; Smith, R.L., Jr. Cycloamination strategies for renewable N-heterocycles. Green Chem. 2020, 22, 582–611. [Google Scholar] [CrossRef] [Green Version]
- Kim, Y.; Li, C.-J. Perspectives on green synthesis and catalysis. Green Synth. Catal. 2020, 1, 1–11. [Google Scholar] [CrossRef]
- Zhang, Z.; Song, J.; Han, B. Catalytic transformation of lignocellulose into chemicals and fuel products in ionic liquids. Chem. Rev. 2017, 117, 6834–6880. [Google Scholar] [CrossRef]
- Duan, B.; Huang, Y.; Lu, A.; Zhang, L. Recent advances in chitin based materials constructed via physical methods. Prog. Polym. Sci. 2018, 82, 1–33. [Google Scholar] [CrossRef]
- Bastiaens, L.; Soetemans, L.; D’Hondt, E.; Elst, K. Sources of chitin and chitosan and their isolation. In Chitin and Chitosan; Wiley: Hoboken, NJ, USA, 2019; pp. 1–34. [Google Scholar]
- Santos, V.P.; Marques, N.S.S.; Maia, P.C.S.V.; Barbosa de Lima, M.A.; de Oliveira Franco, L.; de Campos-Takaki, G.M. Seafood waste as attractive source of chitin and chitosan production and their applications. Int. J. Mol. Sci. 2020, 21, 4290. [Google Scholar] [CrossRef]
- Yan, N.; Chen, X. Don’t waste seafood waste. Nature 2015, 524, 155–157. [Google Scholar] [CrossRef]
- Chen, X.; Yang, H.; Yan, N. Shell biorefinery: Dream or reality? Chem. Eur. J. 2016, 22, 13402–13421. [Google Scholar] [CrossRef]
- Zhou, D.; Shen, D.; Lu, W.; Song, T.; Wang, M.; Feng, H.; Shentu, J.; Long, Y. Production of 5-hydroxymethylfurfural from chitin biomass: A review. Molecules 2020, 25, 541. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mascal, M.; Nikitin, E.B. Dramatic advancements in the saccharide to 5-(chloromethyl)furfural conversion reaction. ChemSusChem 2009, 2, 859–861. [Google Scholar] [CrossRef] [PubMed]
- Liu, L. Production of chemicals from marine biomass catalysed by acidic ionic liquids. Green Chem. 2021, 23, 9800–9814. [Google Scholar] [CrossRef]
- Cai, X.; Wang, Z.; Ye, Y.; Wang, D.; Zhang, Z.; Zheng, Z.; Liu, Y.; Li, S. Conversion of chitin biomass into 5-hydroxymethylfurfural: A review. Renew. Sustain. Energy Rev. 2021, 150, 111452. [Google Scholar] [CrossRef]
- Hou, Q.; Qi, X.; Zhen, M.; Qian, H.; Nie, Y.; Bai, C.; Zhang, S.; Bai, X.; Ju, M. Biorefinery roadmap based on catalytic production and upgrading 5-hydroxymethylfurfural. Green Chem. 2021, 23, 119–231. [Google Scholar] [CrossRef]
- Kong, X.; Zhu, Y.; Fang, Z.; Kozinski, J.A.; Butler, I.S.; Xu, L.; Song, H.; Wei, X. Catalytic conversion of 5-hydroxymethylfurfural to some value-added derivatives. Green Chem. 2018, 20, 3657–3682. [Google Scholar] [CrossRef]
- Chen, L.; Xiong, Y.; Qin, H.; Qi, Z. Advances of ionic liquids and deep eutectic solvents in green processes of biomass-derived 5-hydroxymethylfurfura. ChemSusChem 2022, e202102635. [Google Scholar] [CrossRef]
- Dai, J.; Li, F.; Fu, X. Towards shell biorefinery: Advances in chemical-catalytic conversion of chitin biomass to organonitrogen chemicals. ChemSusChem 2020, 13, 6498–6508. [Google Scholar] [CrossRef]
- Jeong, G.-T. Production of levulinic acid from glucosamine by dilute-acid catalyzed hydrothermal process. Ind. Crops Prod. 2014, 62, 77–83. [Google Scholar] [CrossRef]
- Lee, S.-B.; Jeong, G.-T. Catalytic conversion of chitosan to 5-hydroxymethylfurfural under low temperature hydrothermal process. Appl. Biochem. Biotechnol. 2015, 176, 1151–1161. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.S.; Park, M.-R.; Jeon, Y.J.; Kim, S.-K.; Hong, Y.-K.; Jeong, G.-T. Valorization of chitosan as food waste of aquatic organisms into 5-hydroxymethylfurfural by sulfamic acid-catalyzed conversion process. Energy Technol. 2018, 6, 1747–1754. [Google Scholar] [CrossRef]
- Kim, H.S.; Park, M.-R.; Kim, S.-K.; Jeong, G.-T. Valorization of chitosan into levulinic acid by hydrothermal catalytic conversion with methanesulfonic acid. Korean J. Chem. Eng. 2018, 35, 1290–1296. [Google Scholar] [CrossRef]
- Park, M.-R.; Kim, H.S.; Kim, S.-K.; Jeong, G.-T. Thermo-chemical conversion for production of levulinic and formic acids from glucosamine. Fuel Process. Technol. 2018, 172, 115–124. [Google Scholar] [CrossRef]
- Omari, K.W.; Besaw, J.E.; Kerton, F.M. Hydrolysis of chitosan to yield levulinic acid and 5-hydroxymethylfurfural in water under microwave irradiation. Green Chem. 2012, 14, 1480–1487. [Google Scholar] [CrossRef] [Green Version]
- Wang, Y.; Pedersen, C.M.; Deng, T.; Qiao, Y.; Hou, X. Direct conversion of chitin biomass to 5-hydroxymethylfurfural in concentrated ZnCl2 aqueous solution. Bioresour. Technol. 2013, 143, 384–390. [Google Scholar] [CrossRef]
- Zhu, L.; Fu, X.; Hu, Y.; Hu, C. Controlling the reaction networks for efficient conversion of glucose into 5-hydroxymethylfurfural. ChemSusChem 2020, 13, 4812–4832. [Google Scholar] [CrossRef]
- Yu, S.; Zang, H.; Chen, S.; Jiang, Y.; Yan, B.; Cheng, B. Efficient conversion of chitin biomass into 5-hydroxymethylfurfural over metal salts catalysts in dimethyl sulfoxide -water mixture under hydrothermal conditions. Polym. Degrad. Stab. 2016, 134, 105–114. [Google Scholar] [CrossRef]
- Shrotri, A.; Kobayashi, H.; Fukuoka, A. Cellulose depolymerization over heterogeneous catalysts. Acc. Chem. Res. 2018, 51, 761–768. [Google Scholar] [CrossRef] [Green Version]
- Li, Y.; Wang, J.; Liu, X.; Zhang, S. Towards a molecular understanding of cellulose dissolution in ionic liquids: Anion/cation effect, synergistic mechanism and physicochemical aspects. Chem. Sci. 2018, 9, 4027–4043. [Google Scholar] [CrossRef] [Green Version]
- Uto, T.; Idenoue, S.; Yamamoto, K.; Kadokawa, J.-i. Understanding dissolution process of chitin crystal in ionic liquids: Theoretical study. Phys. Chem. Chem. Phys. 2018, 20, 20669–20677. [Google Scholar] [CrossRef]
- Li, M.; Zang, H.; Feng, J.; Yan, Q.; Yu, N.; Shi, X.; Cheng, B. Efficient conversion of chitosan into 5-hydroxymethylfurfural via hydrothermal synthesis in ionic liquids aqueous solution. Polym. Degrad. Stab. 2015, 121, 331–339. [Google Scholar] [CrossRef]
- Zhang, M.; Zang, H.; Ma, B.; Zhang, X.; Xie, R.; Cheng, B. Green synthesis of 5-hydroxymethylfurfural from chitosan biomass catalyzed by benzimidazole-based ionic liquids. ChemistrySelect 2017, 2, 10323–10328. [Google Scholar] [CrossRef]
- Zang, H.; Yu, S.; Yu, P.; Ding, H.; Du, Y.; Yang, Y.; Zhang, Y. Hydrothermal conversion of N-acetyl-D-glucosamine to 5-hydroxymethylfurfural using ionic liquid as a recycled catalyst in a water-dimethyl sulfoxide mixture. Carbohydr. Res. 2017, 442, 1–8. [Google Scholar] [CrossRef]
- Islam, M.S.; Nakamura, M.; Rabin, N.N.; Rahman, M.A.; Fukuda, M.; Sekine, Y.; Beltramini, J.N.; Kim, Y.; Hayami, S. Microwave-assisted catalytic conversion of chitin to 5-hydroxymethylfurfural using polyoxometalate as catalyst. RSC Adv. 2021, 12, 406–412. [Google Scholar] [CrossRef]
- Kobayashi, H.; Techikawara, K.; Fukuoka, A. Hydrolytic hydrogenation of chitin to amino sugar alcohol. Green Chem. 2017, 19, 3350–3356. [Google Scholar] [CrossRef] [Green Version]
- Kalane, N.D.; Krishnan, R.A.; Yadav, V.D.; Jain, R.; Dandekar, P. Synergistic effect of hetero- and homo-catalysts on the “green” synthesis of 5-hydroxymethylfurfural from chitosan biomass. Cellulose 2019, 26, 2805–2819. [Google Scholar] [CrossRef]
- Qi, M.; Chen, X.; Zhong, H.; Wu, J.; Jin, F. Base-free, vanadium-catalyzed conversion of chitin into acetic acid under low oxygen pressure. ACS Sustain. Chem. Eng. 2020, 8, 18661–18670. [Google Scholar] [CrossRef]
- Su, H.; Wang, J.; Yan, L. Homogeneously synchronous degradation of chitin into carbon dots and organic acids in aqueous solution. ACS Sustain. Chem. Eng. 2019, 7, 18476–18482. [Google Scholar] [CrossRef]
- Sadiq, A.D.; Chen, X.; Yan, N.; Sperry, J. Towards the shell biorefinery: Sustainable synthesis of the anticancer alkaloid proximicin A from chitin. ChemSusChem 2018, 11, 532–535. [Google Scholar] [CrossRef]
- Wolter, F.E.; Schneider, K.; Davies, B.P.; Socher, E.R.; Nicholson, G.; Seitz, O.; Süssmuth, R.D. Total synthesis of proximicin A−C and synthesis of new furan-based DNA binding agents. Org. Lett. 2009, 11, 2804–2807. [Google Scholar] [CrossRef]
- Brucoli, F.; Natoli, A.; Marimuthu, P.; Borrello, M.T.; Stapleton, P.; Gibbons, S.; Schätzlein, A. Efficient synthesis and biological evaluation of proximicins A, B and C. Bioorg. Med. Chem. 2012, 20, 2019–2024. [Google Scholar] [CrossRef] [PubMed]
- Thuy Trang, P.; Lindsay, A.C.; Kim, S.-W.; Persello, L.; Chen, X.; Yan, N.; Sperry, J. Two-step preparation of diverse 3-amidofurans from chitin. ChemistrySelect 2019, 4, 10097–10099. [Google Scholar]
- Pham, T.T.; Lindsay, A.C.; Chen, X.; Gözaydin, G.; Yan, N.; Sperry, J. Transferring the biorenewable nitrogen present in chitin to several N-functional groups. Sustain. Chem. Pharm. 2019, 13, 100143. [Google Scholar] [CrossRef]
- Shao, Y.; Ding, Y.; Dai, J.; Long, Y.; Hu, Z.-T. Synthesis of 5-hydroxymethylfurfural from dehydration of biomass-derived glucose and fructose using supported metal catalysts. Green Synth. Catal. 2021, 2, 187–197. [Google Scholar] [CrossRef]
- Franich, R.A.; Goodin, S.J.; Wilkins, A.L. Acetamidofurans, acetamidopyrones, and acetamidoacetaldehyde from pyrolysis of chitin and N-acetylglucosamine. J. Anal. Appl. Pyrolysis 1984, 7, 91–100. [Google Scholar] [CrossRef]
- Omari, K.W.; Dodot, L.; Kerton, F.M. A simple one-pot dehydration process to convert N-acetyl-D-glucosamine into a nitrogen-containing compound, 3-acetamido-5-acetylfuran. ChemSusChem 2012, 5, 1767–1772. [Google Scholar] [CrossRef] [Green Version]
- Padovan, D.; Kobayashi, H.; Fukuoka, A. Facile preparation of 3-acetamido-5-acetylfuran from N-acetyl-D-glucosamine by using commercially available aluminum salts. ChemSusChem 2020, 13, 3594–3598. [Google Scholar] [CrossRef]
- Wang, C.; Wu, C.; Zhang, A.; Chen, K.; Cao, F.; Ouyang, P. Conversion of N-acetyl-D-glucosamine into 3-acetamido-5-acetylfuran using cheap ammonium chloride as catalyst. ChemistrySelect 2022, 7, e202104574. [Google Scholar] [CrossRef]
- Wang, J.; Zang, H.; Jiao, S.; Wang, K.; Shang, Z.; Li, H.; Lou, J. Efficient conversion of N-acetyl-D-glucosamine into nitrogen-containing compound 3-acetamido-5-acetylfuran using amino acid ionic liquid as the recyclable catalyst. Sci. Total Environ. 2020, 710, 136293. [Google Scholar] [CrossRef]
- Du, Y.; Zang, H.; Feng, Y.; Wang, K.; Lv, Y.; Liu, Z. Efficient catalytic system for converting N-acetyl-D-glucosamine into valuable chemical 3-acetylamino-5-acetylfuran. J. Mol. Liq. 2022, 347, 117970. [Google Scholar] [CrossRef]
- Zang, H.; Li, H.; Jiao, S.; Lou, J.; Du, Y.; Huang, N. Green conversion of N-acetylglucosamine into valuable platform compound 3-acetamido-5-acetylfuran using ethanolamine ionic liquids as recyclable catalyst. ChemistrySelect 2021, 6, 3848–3857. [Google Scholar] [CrossRef]
- Zang, H.; Lou, J.; Jiao, S.; Li, H.; Du, Y.; Wang, J. Valorization of chitin derived N-acetyl-D-glucosamine into high valuable N-containing 3-acetamido-5-acetylfuran using pyridinium-based ionic liquids. J. Mol. Liq. 2021, 330, 115667. [Google Scholar] [CrossRef]
- Drover, M.W.; Omari, K.W.; Murphy, J.N.; Kerton, F.M. Formation of a renewable amide, 3-acetamido-5-acetylfuran, via direct conversion of N-acetyl-D-glucosamine. RSC Adv. 2012, 2, 4642–4644. [Google Scholar] [CrossRef] [Green Version]
- Gozaydin, G.; Song, S.; Yan, N. Chitin hydrolysis in acidified molten salt hydrates. Green Chem. 2020, 22, 5096–5104. [Google Scholar] [CrossRef]
- Chen, X.; Chew, S.L.; Kerton, F.M.; Yan, N. Direct conversion of chitin into a N-containing furan derivative. Green Chem. 2014, 16, 2204–2212. [Google Scholar] [CrossRef] [Green Version]
- Chen, X.; Liu, Y.; Kerton, F.M.; Yan, N. Conversion of chitin and N-acetyl-D-glucosamine into a N-containing furan derivative in ionic liquids. RSC Adv. 2015, 5, 20073–20080. [Google Scholar] [CrossRef]
- Chen, X.; Gao, Y.; Wang, L.; Chen, H.; Yan, N. Effect of treatment methods on chitin structure and its transformation into nitrogen-containing chemicals. ChemPlusChem 2015, 80, 1565–1572. [Google Scholar] [CrossRef]
- Chen, K.; Wu, C.; Wang, C.; Zhang, A.; Cao, F.; Ouyang, P. Chemo-enzymatic protocol converts chitin into a nitrogen-containing furan derivative, 3-acetamido-5-acetylfuran. Mol. Catal. 2021, 516, 112001. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).