1. Introduction
Multiple toxic compounds of natural and synthetic origins can be converted to less dangerous substances via application of (bio)catalysis. Mycotoxins are among the toxins present around us in foods surfaces, water sources, etc. are airborne in industrial, residential, and public premises [
1]. To reduce the health threat, researchers are continuously looking for and suggesting new (bio)catalysts to solve the issues [
2,
3]. In this perpetual race, some fundamentals could be missed which are concerning the (bio)catalysis itself.
Enzymes being specific biocatalysts completely fall under this trend. Most efforts are applied on rational design and engineering [
4] or directed evolution [
5] of the enzymes to improve the specificity or efficiency of enzyme action. To date, much of the works have shown successful conversion of one type of enzyme activity into another [
6] via genetic or chemical modifications of prototype enzymes. In the future, this will allow for the flexible configuring of any enzyme for any practical needs. That means some prototype enzymes already having potential activity can be trimmed as a rock is sculpted into masterpiece. One of the real problems here is the right choice for the initial prototype enzymes.
An alternative is the selection of known and well-studied enzymes, owing to the analysis of their action mechanism, to realize atypical reactions [
7,
8]. This is a nature-like approach to the use of existing biocatalysts since the original naïve enzymes could have completely different functions before evolving in the wild under changed environmental conditions. That is quite possible from a scientific standpoint and appears to be effective from an applicability point of view.
Hexahistidine-tagged organophosphorus hydrolase (His
6-OPH) is an excellent example of an enzyme with promiscuous multiple catalytic activities. It hydrolyzes various organophosphate compounds (pesticides, chemical warfare agents, and products of their degradation) [
9] and bacterial quorum-regulating molecules (various
N-acyl homoserine lactones) [
10]. Recently, the enzyme was shown to hydrolyze some mycotoxins [
11], and the detailed investigation of this mode of its catalytic action became the main purpose of the present study. Special attention was paid to possible modulation of its activity in reactions with mycotoxins. Additionally, some prototype personal protective materials possessing catalytic activity with mycotoxins were obtained and investigated.
2. Results
2.1. Hydrolysis of Mycotoxins by His6-OPH
Hydrolysis of patulin, deoxynivalenol, aflatoxin B
1, aflatoxin B
2, zearalenone, sterigmatocystin, and ochratoxin A by His
6-OPH was investigated under optimal pH 10.5. However, only four mycotoxins (patulin, deoxynivalenol, zearalenone, and sterigmatocystin) were hydrolyzed (
Figure 1) as it was determined with the AChE assay and ELISA. It should be noted here that there were over-estimations in ochratoxin A concentrations using the AChE assay (i.e., toxicity increased five-fold) during enzymatic hydrolysis, and thus, the data were discarded.
Using the method of analysis of integral enzyme kinetics with competitive product inhibition [
12], catalytic characteristics of His
6-OPH in these reactions were estimated (
Table 1). The best catalytic constant was revealed in the case of patulin, while the best Michaelis constant was obtained in the case of sterigmatocystin.
In toto, the efficiency of the catalytic action towards sterigmatocystin was better, but it was 1.7 million times lower compared to the typical and best substrate (paraoxon) of the enzyme [
13]. At the same time, it was 2.8 times better compared to the same value of His
6-OPH with di(isobutyl) methylphosphonate [
14], which is a product of Vx degradation.
The inhibition constants by product(s) were 3.3 ± 1.5, 15.5 ± 6.7, 42.5 ± 2.9, and 2.5 ± 1.0 μM for patulin, deoxynivalenol, zearalenone, and sterigmatocystin, respectively.
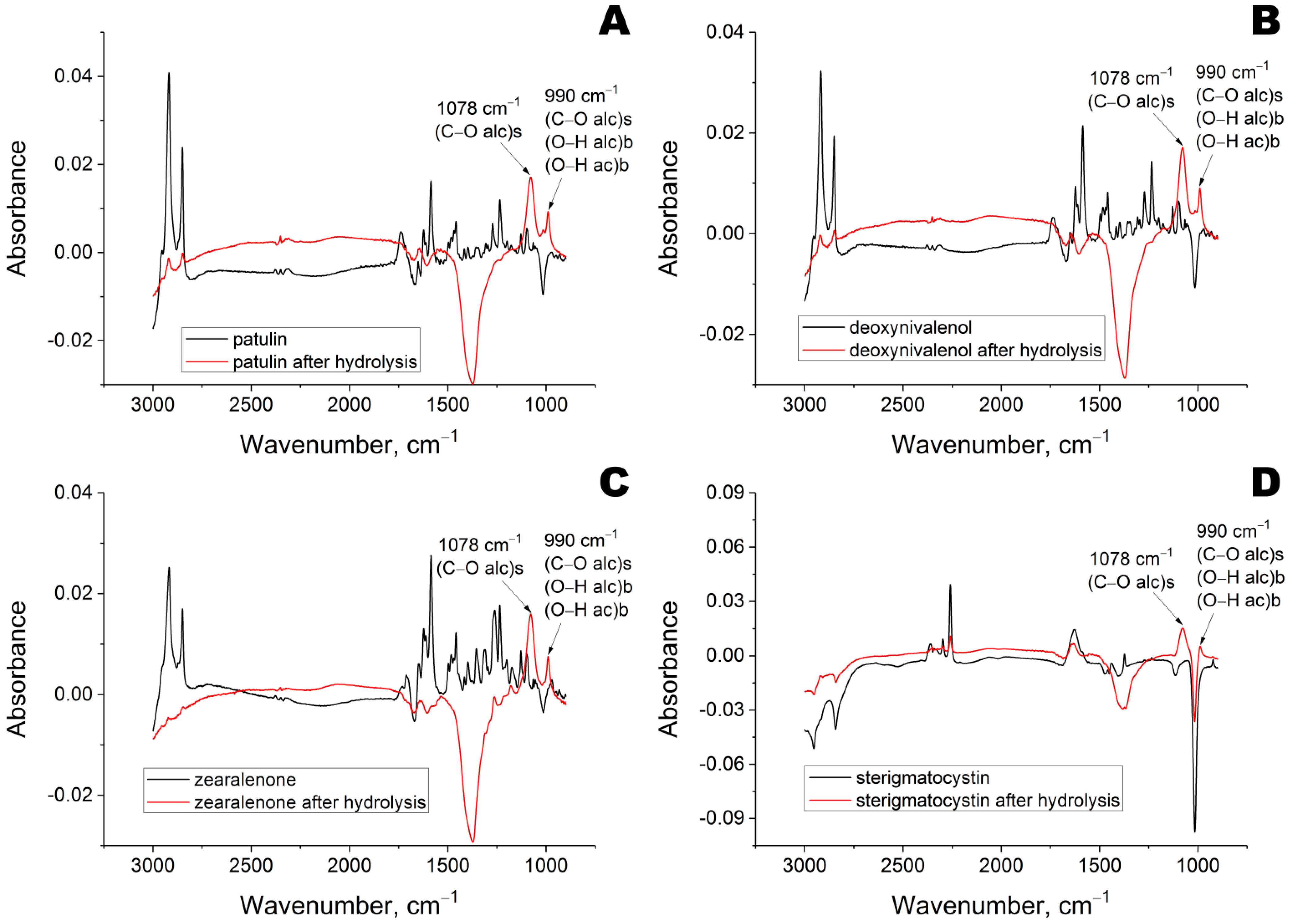
Previously, the GC/MS analysis of reaction medium after enzymatic hydrolysis of mycotoxins was performed [
11], but the reaction product(s) could not be isolated. Therefore, the reaction media after enzyme hydrolysis of mycotoxins were analyzed using Fourier-transform infrared spectroscopy (
Figure 2). The stretching modes of the C–O bond and the bending modes of the O–H bond within alcohols and carboxylic acids intensified significantly.
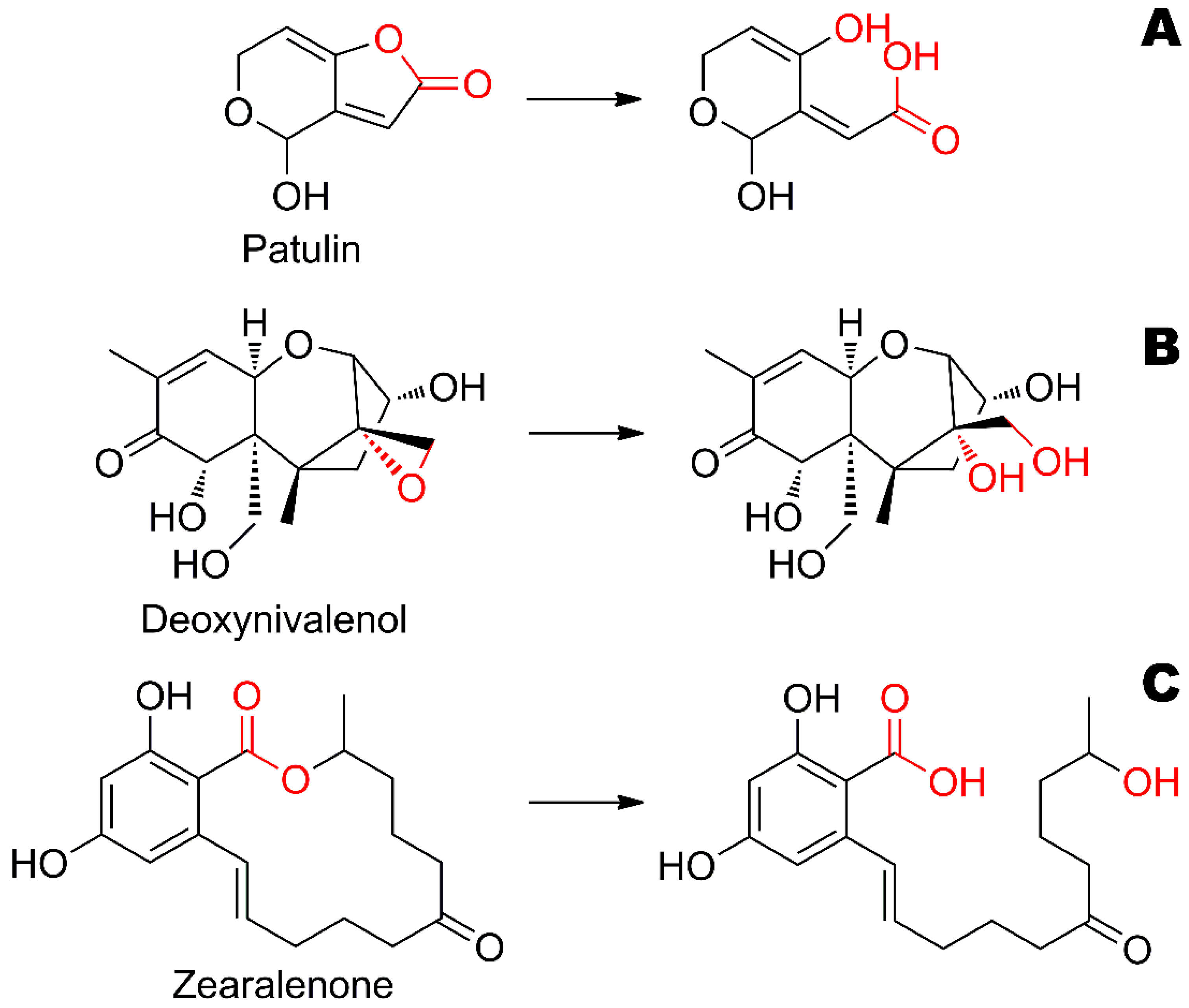
The most probable mechanism of patulin and zearalenone hydrolysis by His
6-OPH was an opening of the lactone ring (
Figure 3) as it occurred with
N-acyl homoserine lactones also hydrolyzed by the enzyme [
10]. Deoxynivalenol contains epoxy-cycle in its structure which is likely to pass de-cyclization in the presence and/or under the action of His
6-OPH.
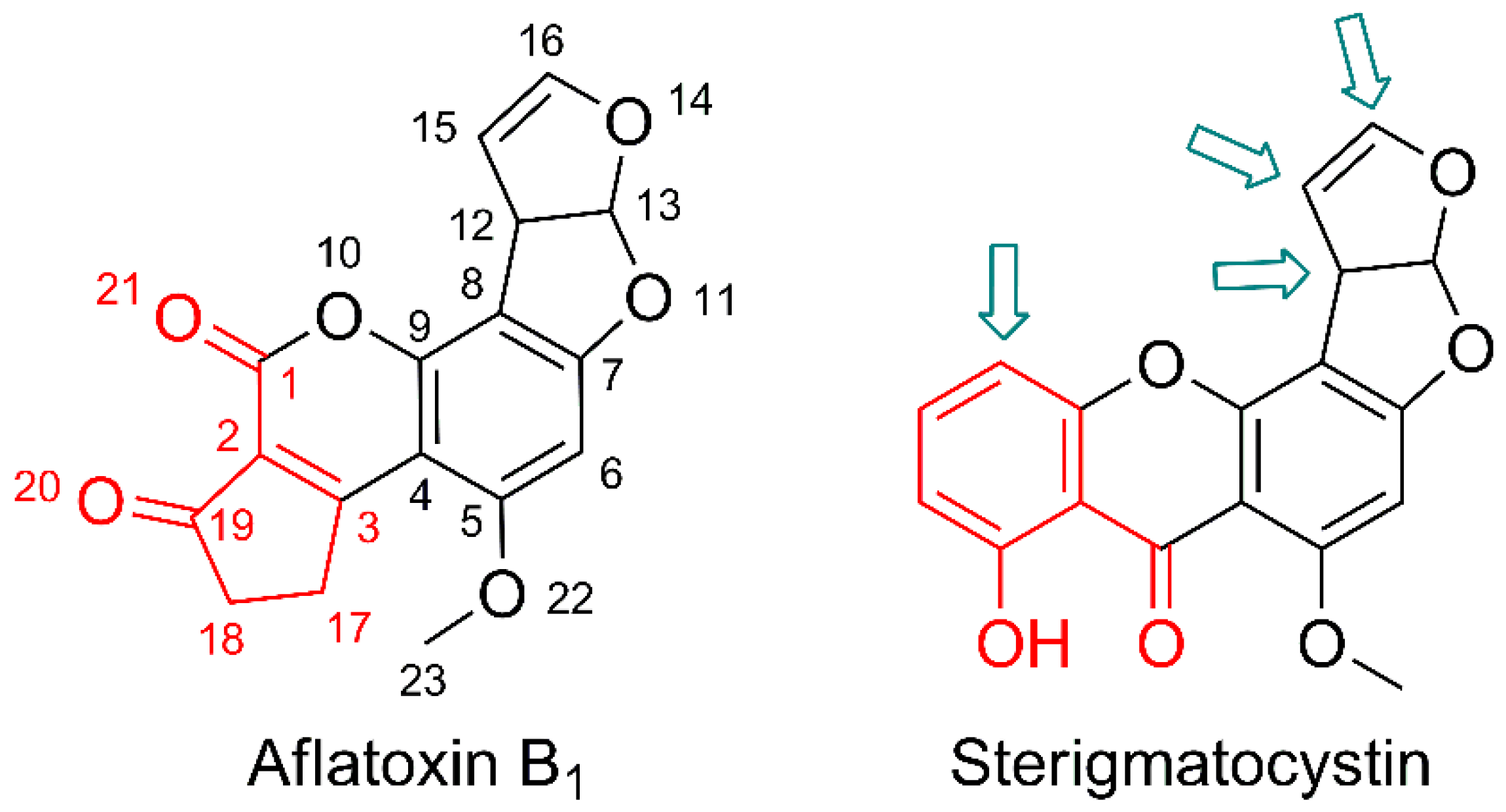
Sterigmatocystin did not contain any of abovementioned structural motifs (
Figure 4). Moreover, His
6-OPH had no detectable activity towards both aflatoxins B
1 and B
2, and thus, the key(s) for the enzyme mechanism of action should be examined within differing structural moieties in future work(s).
2.2. Influence of Sorbents on Efficiency of Enzymatic Mycotoxin Removal
Since the sorbents are widely used for the elimination of different mycotoxins from the water and agricultural (food/feed) products [
16], the joint application of some inorganic sorbents with enzymatic degradation for the decrease of the mycotoxins’ concentrations could be useful. Based on that, several usual inorganic sorbents (zeolite, diatomite, and silica gel) with or without low-temperature plasma pre-treatment were tried in the sorption of zearalenone from water media (
Table 2). Low-temperature plasma treatment is known [
17] to have an effect on sorbents in multiple ways: surface morphology, porosity, pore diameter, etc. Several bentonites were tried also but have not shown any zearalenone adsorption under experimental conditions.
Pretreatment of diatomite with low-temperature plasma leads to a deterioration of its capacity and at the same time, to an improvement in the characteristics of silica gel. Thus, silica gel with medium size particles (0.63–1.25 mm) treated by low-temperature plasma was selected to combine with His6-OPH for further experiments.
To obtain a combination of silica gel and enzyme, its stabilized enzyme–polyelectrolyte nanocomplexes with poly-L-glutamic acid (PLE50) were applied.
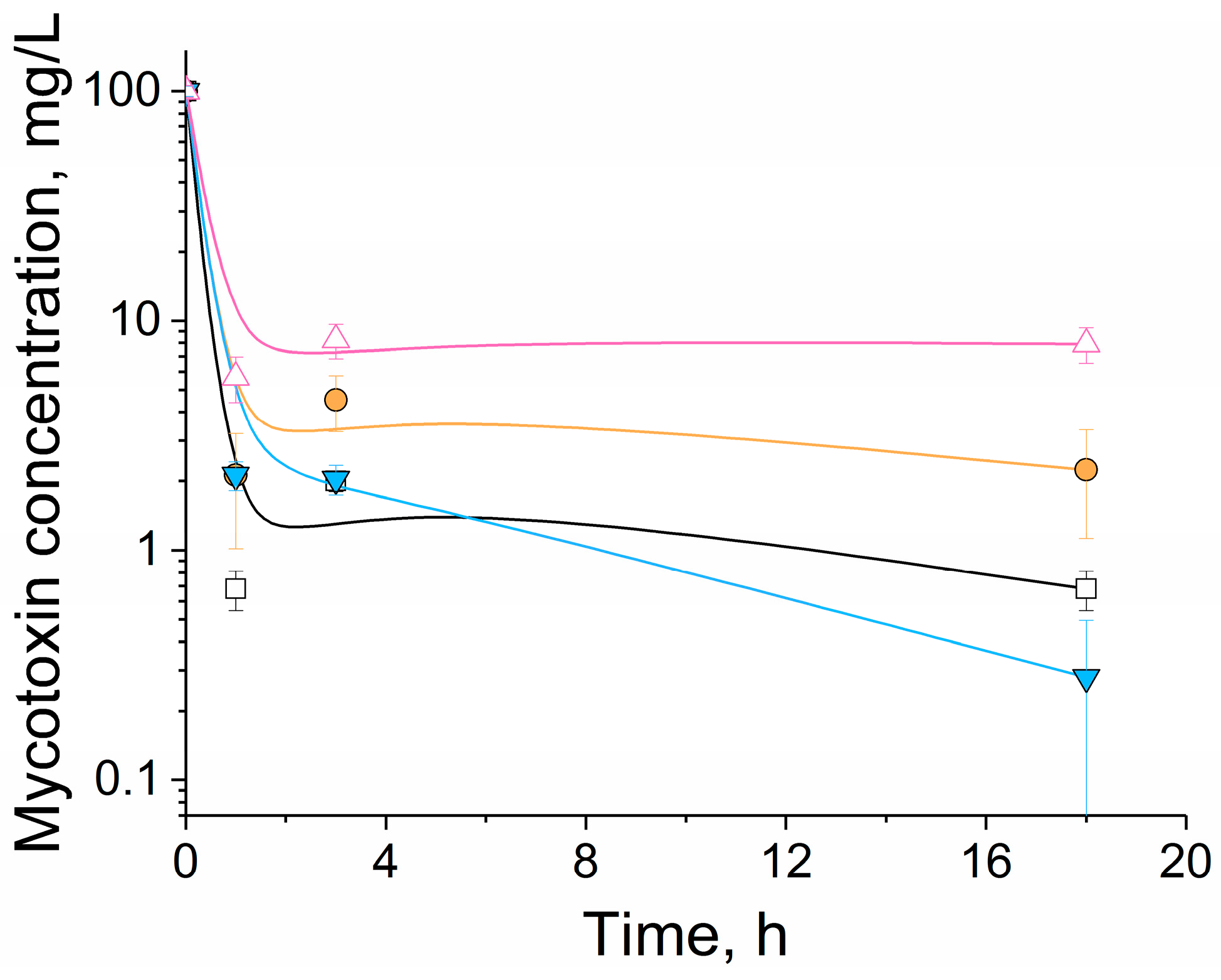
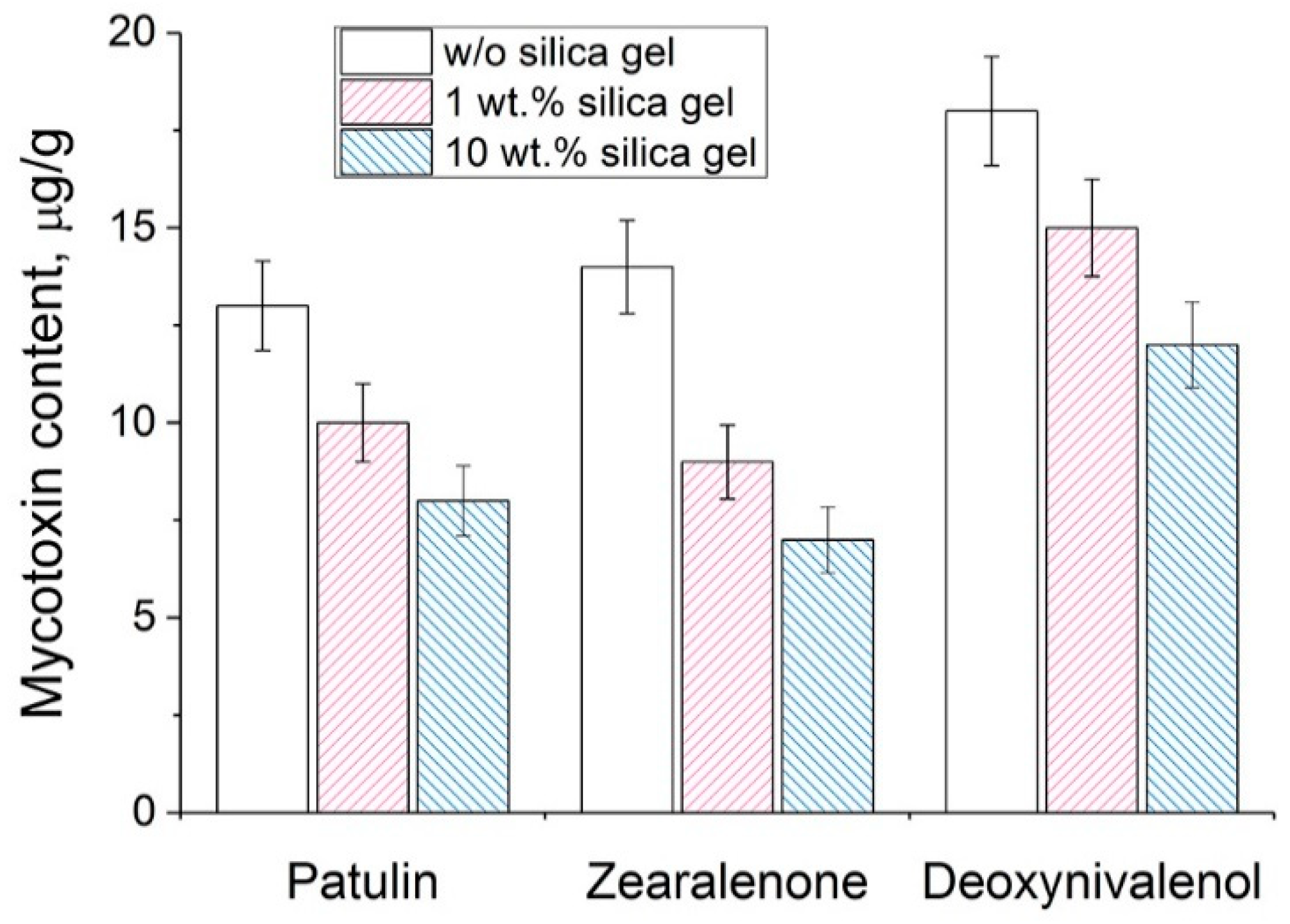
The simultaneous addition of nanocomplex His
6-OPH/PLE
50 and silica gel resulted in an improvement of mycotoxin removal from the grain by 17-36% during 1 h of exposure (
Figure 5). However, longer exposure to the enzyme led to complete removal of toxicity by a single enzyme and incomplete removal by a combination of the enzyme and silica gel (
Table 3). It was likely a result of mycotoxin being retained by a carrier, thereby, preventing their catalytic decomposition. However, additional investigations will be needed to quantify the contribution of each component.
2.3. Hydrolysis of Mycotoxins by Enzyme Nanocomplexes Immobilized onto Fiber Materials
The nanocomplex His
6-OPH/PLE
50 was immobilized onto various fiber materials; the characteristics were described previously [
18]. The catalytic characteristics of the prepared samples were initially determined in reaction with the best substrate (paraoxon) of His
6-OPH (
Table 4).
Despite the apparent variation, there were no statistically significant differences according to ANOVA on ranks (p = 0.071, 0.050, and 0.543 for values
Vmax /E
o,
Km and
Vmax /(E
o ×
Km), respectively). Thereby, His
6-OPH/PLE
50 immobilized on these carriers possessed similar catalytic characteristics with a high degree of certainty. As compared to catalytic characteristics of His
6-OPH/PLE
50 estimated previously [
19], values
Vmax /E
o and
Vmax /(E
o ×
Km) decreased 27–34 and 699–1052 times, respectively, while
Km values increased 22–39 times.
Catalytic characteristics of His
6-OPH/PLE
50 immobilized on fibers were investigated in reactions with mycotoxins (
Table 5). For comparison, another known mycotoxin degrader (thermolysin) [
20] was also taken in a form of similar polyelectrolyte nanocomplex (thermolysin/PLE
50) [
11] and was issued under comparable conditions.
As before, catalytic characteristics of thermolysin/PLE50 with zearalenone and ochratoxin A were independent on support (ANOVA on ranks, p > 0.62 and >0.82, respectively). In the case of His6-OPH/PLE50, there were statistical differences only for Vmax /Eo values (p = 0.004, which was due to the differing pair of #1 and #2), while other characteristics were undistinguishable (p = 0.139 for both).
As compared to the
Km value (26 μM) of covalently bound thermolysin with ochratoxin A [
20],
Km of sorbed nanocomplex thermolysin/PLE
50 increased 47 times. It is not possible to directly compare
Vmax /E
o values now. However, more than 20 min were required to achieve a stabile biosensor signal in the reaction containing 4 μg/mL enzyme and 100 μM ochratoxin A [
20]. Under these conditions, sorbed thermolysin/PLE
50 could eliminate two thirds of the mycotoxin as a substrate.
Adsorption of His6-OPH/PLE50 onto fibers had only limited or even no effect on Km values, while Vmax /Eo values increased dramatically (more than 43-fold).
Probably the main reason for such behavior was a pH that shifted from the optimal 10.5 to non-optimal 7.5. Another point could be the method of calculation of catalytic characteristics (i.e., integral kinetics was utilized for free His
6-OPH and method of initial rates was used for immobilized His
6-OPH/PLE
50). Nanocomplexing of His
6-OPH with polymers is unlikely to result in such improvement of
Vmax /E
o values and previously have retained or even decreased catalytic characteristics with organophosphate substrates [
19].
3. Discussion
Although mycotoxins have huge variations in chemical structures, there are some distinguishable motifs. All mycotoxins are biosynthesized through multiple but limited biochemical routes. Moreover, prokaryotes and even non-fungal eukaryotes are quite successfully living and evolving under the pressure of mycotoxins. Thus, many pathways for mycotoxin removal have been developed already, and the purpose of humans wishing to possess something useful is to look thoroughly around. First catalysts for mycotoxins were selected this way a long time ago while utilizing easily accessible lipases, proteases, etc.
Recent research implemented more intensive workflow to select such hydrolytically active enzymes using computer modeling [
21]. Specifically, it was predicted and confirmed that some mycotoxins could be hydrolyzed by His
6-OPH [
11]. From a standpoint of kinetics, catalytic characteristics of His
6-OPH with all mycotoxins are orders of magnitude lower compared to the best substrate (paraoxon). At the same time, they were found to be even better in comparison with bad substrates (methylphosphonates) [
14]. Interestingly, the best catalytic characteristics of enzyme with mycotoxins were quite close to the same ones of His
6-OPH with
N-acyl homoserine lactones [
10]. The closest structural analogue of
N-acyl homoserine lactones among mycotoxins is patulin. Its
Vmax /E
o and
Km values were 4-10 and 18-108 times worse, respectively, as compared to the same ones with
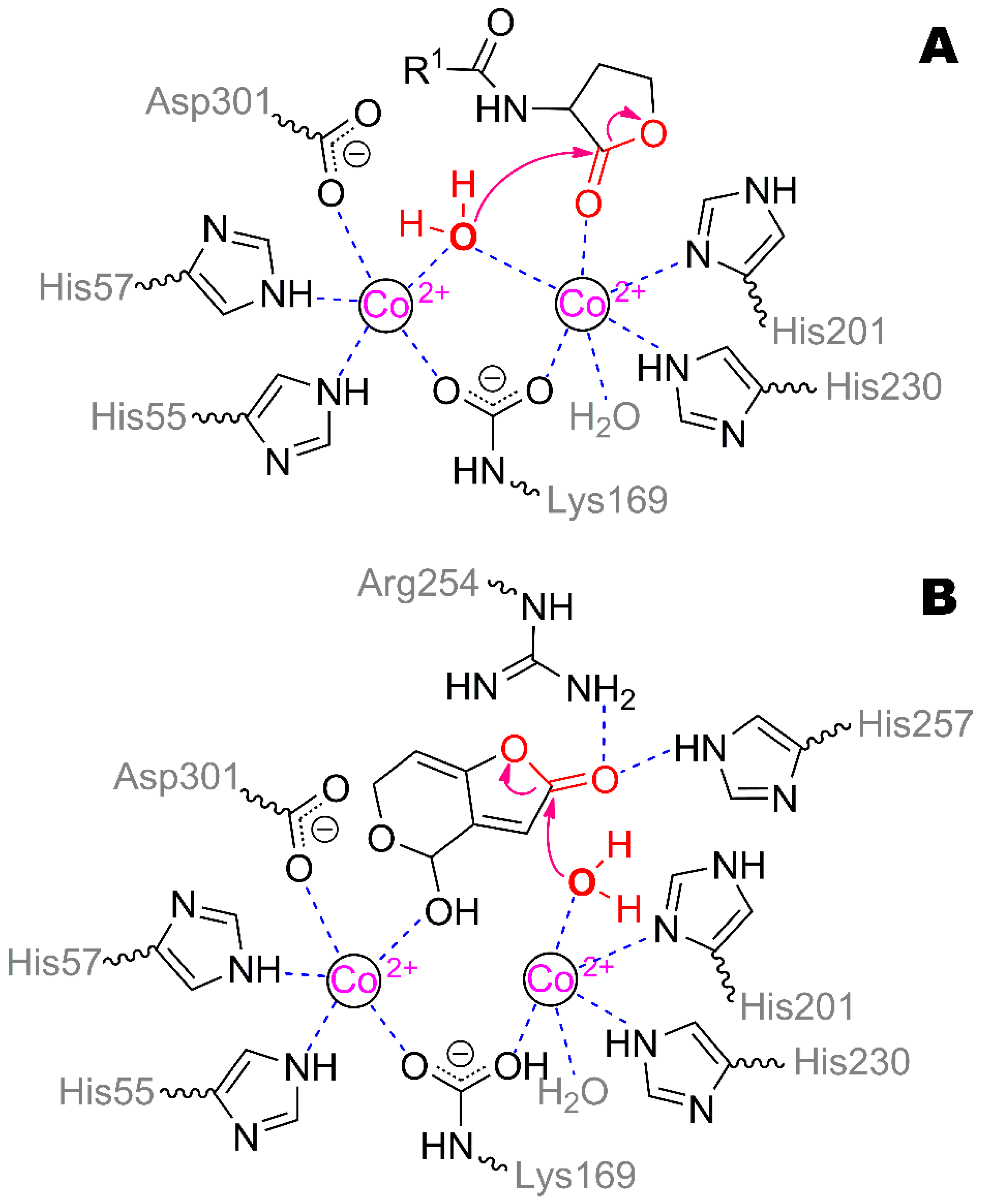
N-acyl homoserine lactones. The structure of patulin is complicated for catalysis in several ways (
Figure 6): (i) the conjugated heterocycle is too rigid and fixes a ‘leaving’ OH-group too close to the forming carboxylic group; (ii) the unsaturated bond within the opening lactone ring reduces flexibility even more. These factors could affect the energy barrier for the reaction path and thus reduce the catalytic constant.
Normally, lactones should bind to the one metal ion via oxygen of the C=O group, while the other metal ion promotes a water molecule (or hydroxide ion) followed by a nucleophilic attack on carbon [
10]. It seems that was not the case with patulin: the heterocycle could not fit in a proper way during binding and should enter deeper into the active site for at least the single metal ion catalysis. This way, patulin is bonded via the OH-group to the second metal ion (3.5 Å) and via carboxylic oxygen to His257 (3.2 Å) and Arg254 (3.3 Å). Then, the first metal ion (5.3 Å) could promote a water molecule for nucleophilic attack on carbon. The multiple obstacles will affect the overall efficiency of the productive binding while worsening
Km.
Zearalenone also contains a lactone ring similar to patulin and
N-acyl homoserine lactones. However, theoretically, [
11] it was too large to accommodate within the active site of His
6-OPH. Moreover, the native zearalenone hydrolases appear to utilize a mainspring mechanism to release the long alcohol residue from the catalytic domain [
15].
Deoxynivalenol could not be contained in the active site of His
6-OPH [
11]. However, its enzymatic hydrolysis was more enabling as in the case of human epoxide hydrolase [
22], which requires only two residues of amino acids for hydrolytic action: Asp333 (for nucleophilic attack) and His523 (for regeneration of nucleophile). Two pairs of Asp–His were accessible on the surface of His
6-OPH: Asp121–His123 (distance was ~6 Å) and Asp35–His29 (3.8 Å). The last one is located at the N-terminus of the protein and involves the histidine residue of His
6-tag being closest to the N-terminus of non-modified (initial) enzyme molecule. Retrospective analysis of previous computational data [
11] has shown possible embedding of deoxynivalenol into the cavity on the N-terminus; namely, the distances between the epoxy-carbon atom and the oxygen of the carboxyl group of Asp35 were 7.3-7.4 Å. Interestingly, the Ser359 residue was located even closer (3.7-3.8 Å) to the epoxy-carbon while being proximal to the same His29 (4.9 Å). It can be hypothesized that other His
6-tagged enzymes containing proper amino acid(s) at the terminus could have some non-specific hydrolytic activity with deoxynivalenol in a similar manner.
All investigated mycotoxins demonstrated a product inhibition towards His
6-OPH. Thus, the removal of reaction product(s) is desirable to improve enzyme efficiency and can be realized via the addition of sorbent(s). Moreover, additives of various sorbents can be considered as a generally accepted method of mycotoxins removal from feed. Multiple investigations of sorbents alone or in combination with enzyme(s) are known to-date [
16,
23]. Such combination(s) can be synergistically much productive for the whole process, but it was rather insufficient in the current work. As a result, nanocomplexes His
6-OPH/PLE
50 alone were preferable on a long run.
Finally, some prototype protective materials against mycotoxins were obtained by simple adsorption of enzymes onto textiles. Though catalytic characteristics of immobilized enzymes were lower in orders of magnitude compared to the free ones, the final materials were quite effective in hydrolysis of multiple mycotoxins. Moreover, some improvement of efficiency was determined in the case of immobilized His6-OPH/PLE50 with sterigmatocystin. If it was not experimental error (e.g., too heavy contribution of background hydrolysis), then it could be an interesting direction for future research.
Other research suggested the immobilization of mycotoxin degrading enzymes into pre-formed silica coating [
24]. However, being fragile, such coatings have very specific applications, such as filtering membrane.
4. Materials and Methods
4.1. Materials
Thermolysin from
Geobacillus stearothermophilus (expressed in
E. coli) and paraoxon (diethyl 4-nitrophenyl phosphate) were purchased from Sigma-Aldrich (Darmstadt, Germany). Polyglutamic acid (PLE
50) was obtained from Alamanda Polymers (Huntsville, AL, USA). His
6-OPH was produced in recombinant
E. coli strain SG13009[pREP4] (Qiagen, Hilden, Germany) and purified by a published procedure [
25]. According to methods described earlier [
13], enzyme concentration was determined by Bradford assay with Coomassie Brilliant Blue G-250 (Sigma-Aldrich, Darmstadt, Germany); protein purity was analyzed by sodium dodecyl sulfate polyacrylamide gel electrophoresis in a 12% polyacrylamide gel using a Mini-PROTEAN II cell (Bio-Rad, Hercules, CA, USA) followed by Coomassie Brilliant Blue R-250 (Sigma-Aldrich, Darmstadt, Germany) staining and was ca. 98 ± 1%. Enzyme–polyelectrolyte complexes at molar ratio 1:1 were obtained by the previously published method [
19,
26].
The fibrous materials were purchased locally and described previously [
18]. Briefly, material #1 contained 30% cotton and 70% meta polyaramide fiber fabric and was covered by poly(vinylidene difluoride)-
co-poly(tetrafluoroethylene) membrane. Materials #2 and #3 were nonwoven, consisted of 70% viscose and 30% polyester, and differed in mass per area. The water vapor transmission rates through materials #1–#3 over 24 h, measured according to ISO 2528:2017, were 480, 367, and 300 g/m
2, respectively. The mass per area of materials #1–#3, measured according to ISO 3801:1977, were 343 ± 26, 32.4 ± 4.5, and 54.7 ± 4.1 g/m
2, respectively. All materials were cut to samples of 1 × 1 cm before their further use.
4.2. Enzyme Activity
The organophosphorus hydrolase activity was determined as described earlier [
26] using the Agilent 8453 UV-visible spectroscopy system (Agilent Technology, Waldbronn, Germany) equipped with a thermostatted analytical cell at 405 nm. Typically, 100 µL of 7.5 mM aqueous paraoxon solution was added to 1.9 mL of ca. 1 nM enzyme in 0.1 M carbonate buffer (pH 10.5). Initial reaction rates were used to calculate enzyme activity. One unit of enzyme activity was defined as the quantity of the enzyme necessary to hydrolyze 1 µmol of paraoxon per min at 25 °C.
Stock solutions of 10 mg/mL patulin, deoxynivalenol, zearalenone, and ochratoxin A (Cayman Chemical, Ann Arbor, MI, USA), or 5 mg/mL sterigmatocystin, aflatoxin B1, and aflatoxin B2 (Sigma-Aldrich, Darmstadt, Germany) were prepared in methanol.
To determine enzyme activity with mycotoxins, 5 µL of mycotoxin stock solution was accurately added to 195 µL of His
6-OPH (final activity 225 U/mL) in 0.1 M carbonate buffer (pH 10.5) and maintained at 25 °C under gentle mixing. The same procedure without enzyme was used as a control. Over time, aliquots of reaction medium were sampled and immediately analyzed or frozen at –20 °C before further analysis. Mycotoxin concentrations were determined as described previously [
27] using Ellman’s assay of AChE and toxicity to luminescent
Photobacterium phosphoreum cells immobilized in polyvinyl alcohol cryogel. Briefly, 0.96 mL of 0.1 M phosphate buffer (pH 8.0) in a spectrophotometric cell was supplemented with 10 μL of 20 mM 5,5′-dithiobis(2-nitrobenzoic acid) (Sigma-Aldrich) in 0.1 M phosphate buffer (pH 7.0) containing 1.5 g/L Na
2CO
3. Then, 10 μL of 10 μg/mL AChE (Sigma-Aldrich, Darmstadt, Germany) followed by 10 μL of analyzed mycotoxin was added and vigorously mixed. The reaction was initiated by the addition of 10 μL of 50 mM acetylthiocholine iodide (Sigma-Aldrich, Darmstadt, Germany). The rate of formation of 2-nitro-5-thiobenzoic acid was determined using Agilent 8453 UV-visible spectroscopy system at 412 nm.
The luminescence of the immobilized bacteria was measured using a 3560 microluminometer (New Horizons Diagnostics Co, Columbia, MD, USA). The maximum level of luminescence (I0) was determined in an aqueous media based on 2% NaCl solution at 10 ± 1 °C after thermal equilibration for 10 s. The residual intensity of luminescence (I/I0) was analyzed after exposure of the cells in the medium containing analyzed mycotoxin for 0.5 h. For both assays, the residual mycotoxin concentration was calculated with calibration curves.
The values of the Michaelis constant (
Km) and the maximum rate (
Vmax) for the free enzyme in its initial concentration (E
o) were determined by analysis of integral enzyme kinetics with competitive product inhibition [
12]. The data were linearized in coordinates 1/
t*ln(S
0/P) vs. P/
t and approximated using the least square method in Origin Pro 2017 (OriginLab Corporation, Northampton, MA, USA).
After reaction completion, FTIR absorption spectra of samples of reaction medium were obtained using Tensor 27 IR spectrometer (Bruker, Bremen, Germany) equipped with a liquid nitrogen-cooled MCT (mercury-cadmium-telluride) detector and Ministat 125 thermostat (Huber, GA, USA) as described previously [
28].
The measurements were performed in a thermostatted FTIR (frustrated total internal reflection) cell (BioATR-II, Bruker, Bremen, Germany) using a single-reflection crystal ZnSe at 22 °C and constant rate of dry-air flow provided with a Jun-Air compressor (Gast Manufacturing Inc., Benton Harbor, MI, USA). An aliquot (50 µL) of the sample was applied on the FTIR cell’s crystal, the spectrum was recorded thrice in the range of 4000 cm−1 to 950 cm−1 with 1 cm−1 resolution; 70-pass scanning and averaging was performed. The background spectrum was recorded in a similar way for a buffer alone. The spectra were analyzed with Opus software (ver. 7.0, Bruker, Bremen, Germany).
4.3. Experiments with Sorbents
To determine the sorption capacity of the sorbents, 1 mL of 50 µg/mL zearalenone containing 1 vol.% methanol was mixed with 0.1 g of sorbent (bentonites, diatomites, silica gels, or zeolites) and exposed on a thermoshaker for 1 h at 25 °C. The sorbent was centrifuged (10,000× g, 10 min), and the supernatant was separated. The residual concentration of mycotoxin was determined as described above.
For simultaneous hydrolysis and adsorption, a series of 1 g samples of wheat grain was contaminated by 10 μL of 10 mg/mL patulin, deoxynivalenol, or zearalenone. Shortly, 10 (or 100 mg) of sorbent was added, followed by 2 mL of His6-OPH/PLE50 (200 U/g) in 0.1 M carbonate buffer (pH 10.5). The reaction medium was homogenized and exposed at room temperature. After 1 h (and 2 h) the suspension was centrifuged (10,000× g, 10 min), and the supernatant was separated and analyzed.
4.4. Enzyme Activity of Fiber Materials
To deposit enzymes onto fiber materials, the previously published procedure [
18] was used with minor modifications. Briefly, 10 μL of 0.1 mg/mL His
6-OPH/PLE
50 or thermolysin/PLE
50 in a PBS buffer (pH 7.4) were applied per textile sample (1 × 1 cm) and dried for 20–22 h at +8 °C within Petri dishes under sterile conditions.
The organophosphorus hydrolase activity was determined as described earlier [
18]. Briefly, dried samples of fibrous materials were placed in a stirred vessel with 0.1 M carbonate buffer (pH 10.5) and total reaction volume of 2 mL. Then, a variable volume (20–400 μL) of 7.5 mM paraoxon water solution was added and continuously mixed at 25 °C for 5 min. The kinetics of the product (4-nitrophenol) formation was monitored by regular sampling 0.1 mL of a reaction medium into 0.9 mL of the same buffer and measuring OD with an Agilent 8453 UV-visible spectroscopy system at 405 nm. Similar samples without His
6-OPH were applied analogously and used as controls. The initial linear parts of kinetic curves (
Vo = tg α) were used to calculate
Km and
Vmax values for immobilized enzyme while utilizing hyperbolic approximation in Origin Pro 2017.
To determine the activity of immobilized enzymes with mycotoxins, a similar method was applied as above. To wit, dried samples of fibrous materials were placed in a stirred vessel with 0.9 mL of 50 mM phosphate buffer (pH 7.5). After that, 100 μL of variable concentration of mycotoxin in methanol (0.5–10 mg/mL for zearalenone and ochratoxin A, 0.1–5 mg/mL for sterigmatocystin) was added and continuously mixed at 25 °C for 60 min. Amounts of 100 μL aliquots of reaction media were regularly sampled and immediately frozen at –20 °C before further analysis, as described above. The same procedure was applied with samples without enzymes and used as controls. Catalytic characteristics were calculated using the initial rates as above.
All experiments were carried out at least in triplicate. Statistical analysis was performed using SigmaPlot (ver. 12.5, Systat Software Inc., San Jose, CA, USA), and the data are presented as means ± standard deviation (± SD) unless otherwise stated.